Furthermore, if imaging tests can accurately identify the location of DE lesions, surgeons might have the evidence they need to prepare and enhance their surgical strategy.
Bimanual vaginal examination is positive and supports the diagnosis when encountering a fixed retroverted uterus, pelvic discomfort, tender and shortened uterosacral ligaments, and adnexal masses (palpable nodule, thickened area, or a palpable cystic expansion) [
22]. If deep-infiltrating nodules are palpated on the rectovaginal wall or seen in the posterior vaginal fornix during clinical examination, diagnosis is more straightforward [
22,
23]. Clinical examinations performed during menstruation are the most reliable for detecting deep-infiltrating nodules; however, patient acceptance may be a concern [
23].
3.1.1. Ultrasonography Overview
The accuracy of transvaginal sonography in identifying DE is higher in intestinal and bladder endometriosis, and lower in vaginal, uterosacral, and rectovaginal septum lesions, according to the findings of Bazot M et al. [
27]. The sonographic observations contributed to the morphological criteria classification for diagnosing endometriotic lesions [
27].
Endometriomas are easier to be detected by pelvic examination or TVS than other types of endometriosis; nevertheless, it might be hard to differentiate between endometriomas and ovarian malignancies [
28]. Kupfer et al. defined the appearance of diffuse low-level echoes within cysts as endometriotic involvement of the ovaries [
28].
Homogeneous echoes, often known as ground-glass look, are the characteristic ultrasound aspect of these cysts due to hemorrhagic content, and they do not usually show any vascular echo with a Doppler flow scanner [
29]. The “kissing ovaries” sign, with both the ovaries adherent to the posterior uterine wall, is generally linked with bilateral ovarian endometriosis cysts, and can indicate the existence of pelvic adhesions and DE [
30,
31].
The findings of G. Hudelist et al. define the sonographic diagnostic criteria for different types of endometriotic lesions in the pelvic area [
9].
The anterior pelvic compartment contains the urinary bladder, the urethra, the vesicouterine pouch, and the round ligaments [
20]. If a hypoechogenic nodule with or without cystic characteristics is visible on the posterior wall of the urinary bladder, endometriosis of the bladder is hypothesized [
32].
In a paracervical nodule appearance on TVS, ureteral infiltration should be considered. DE can cause ureteral blockage, which can progress to hydronephrosis and gradual kidney failure; therefore, a genitourinary ultrasound exam followed by specialized renal dysfunction examinations may be required [
33].
The uterus and the adnexa are located in the center pelvic compartment, whereas the posterior compartment hosts the Douglas pouch, the uterine torus, the rectovaginal septum, the uterosacral ligaments, and the rectosigmoid [
33].
Abnormal hypoechogenic linear thickening and/or the hypoechogenic cystic or non-cystic lesion inside the posterior vaginal wall are categorized as vaginal involvement [
9]. The rectovaginal space may have endometriosis implants when hypoechogenic nodules or cysts are visualized [
9].
When the uterus, ovaries, tubes, and rectosigmoid colon form a common block with the disappearance of the peritoneal structures, and peritoneal borders are only partially recognized, the obliteration of the posterior pelvic pouch is deemed complete [
32]. The “kissing ovaries” TVS sign suggests the attachment of the ovaries to the pouch of Douglas through pelvic adhesions [
34]. This observation was linked to higher laparoscopic classification severity scores (#Enzian and rASRM) and longer surgical duration because of more widespread disease in the pouch of Douglas and pelvic area [
30,
34,
35,
36,
37,
38].
According to Bazot’s criteria, the uterosacral ligament involvement is classified as a regular or irregular hypoechogenic nodular structure, or hypoechogenic linear thickening with regular or irregular borders [
27].
One sonographic indicator of rectosigmoid endometriosis is the regular or irregular hypoechogenic mass that alters and modifies the typical look of the muscle layer of the rectosigmoid wall. The rectosigmoid submucosa can be examined along the midsagittal plane as a hypoechogenic subtle difference close to a hyperechogenic stratum [
32].
Applying moderate pressure on the cervical area and the lower abdomen wall to move the uterus when performing TVS helps inspect for pelvic adhesions [
9]. The “sliding sign” examines how the rectum slides against the posterior uterine wall. The sign is positive when there is significant mobility between the uterus and the descendent colon [
35]. A positive sign indicates a lower probability of adhesions [
39]. A negative sliding sign is defined as a lack of mobility of the rectum against the uterus and the posterior vaginal fornix, indicating probable adhesion and endometriosis lesions [
35,
36].
Menakaya et al. discovered that the sliding sign was recognized better in the retrocervix area than in the posterior upper uterine fundus [
39]. They also considered that exceeding the cut-off of 200 performed TVS provides better diagnosing for endometriosis nodules [
39]. The operators with the experience of 2500 scans become proficient in performing the sliding sign technique and detecting the pouch of Douglas obliteration [
39]. Except for the rectovaginal septum (RVS) DE, TVS is a precise and reliable method for non-invasive DE diagnosis [
5].
In 2016, S. Guerreiro et al. formed the International Deep Endometriosis Analysis group (IDEA)-defined parameters to describe the manifestations of endometriosis and DE on TVS, fulfilling the need for established definitions in the sonographic classification and diagnosis of DE [
38]. The research team formed by gynecological surgeons, robotic-assisted surgeons, and radiologists introduced a method for examining the pelvis in women suspected of having endometriosis [
38]. Accordingly, TVS must be performed systematically, with endometriotic lesions measured in a standardized way, with homogeneous nomenclature to describe the DE location and specific expressions (endometriomas, adenomyosis, pelvic adhesions) [
38]. Other studies have attempted to classify endometriosis lesions using ultrasonography characteristics, but none have been externally evaluated and widely adopted [
35].
S. Guerreiro et al. proposes four fundamental screening ultrasound steps with suspected or confirmed endometriosis [
38]. The steps can be followed in any order, as stated in the research item, with the condition that all four prove or rule out various kinds of endometriosis [
38].
The four steps, as stated by S. Guerreiro, are:
Regular uterine and adnexal examinations (with sonographic evidence of adenomyosis or ovarian cysts).
Transvaginal ultrasound “soft markers” evaluation (i.e., site-specific tenderness and adnexal mobility).
POD status is evaluated by utilizing a real-time ultrasound “sliding sign”.
Examination of the anterior and posterior compartments for DE nodules [
38].
The examiner’s experience impacts TVS results and reproducibility. The procedure has its own applications and limitations, but it is becoming more prevalent as a first-line diagnostic method for women suspected of endometriosis [
38]. Bazot M. stated that TVS’s average accuracy in detecting DE is 85.9%, thus encouraging specialists in endometriosis to consider TVS as the first imaging method for diagnosing DE [
27].
The role of ultrasound color Doppler did not prove useful in diagnosing endometriomas or DE nodules. One benefit of color Doppler is to differentiate bowel endometriosis from rectal cancer [
40].
In a 2015 study, Fraser et al. compared the standard TVS with expert-guided transvaginal ultrasound (EGTV) sensitivity for endometriosis assessment [
40]. They found that EGTV is more sensitive than regular pelvic ultrasound when detecting endometriosis, particularly the DE, before surgery. EGTV also contains a detailed classification of the disease’s degree and severity, which can help with surgical strategy and patient assistance [
40].
3.1.2. Magnetic Resonance Imaging Overview
Although MRI is a frequently used tool for diagnosing DE, there is no international agreement on the ideal imaging strategy [
33]. According to local knowledge, indications and imaging techniques may differ between institutions [
33].
The imaging tool with the highest overall accuracy for determining the degree of DE is magnetic resonance imaging, usually used as a second-line diagnosis method after the TVS to obtain an accurate anatomic depiction of the complete pelvic organs [
33].
An MRI performed and interpreted by a specialist in endometriosis can assure the identification of DE based on the juxtaposition between normal pelvic visceral fat and endometriotic nodules or endometriomas [
25]. The MRI aspect of endometriosis lesions is comparable to pelvic adhesions or fibrous tissue [
41]. The radiologist’s experience in interpreting the pelvic MRI of patients suspected of endometriosis is essential in aiding the correct diagnosis of DE [
42].
MRI is the imaging modality with the best average reliability for identifying the degree of DE, and has excellent sensitivity for endometriotic lesions due to its essential soft-tissue sensitivity [
33]. Nonetheless, the examination and imaging interpretation should be adjusted to each patient’s concerns to attain the required accuracy [
43]. Noninvasive tools demand a methodical approach to achieve consistent and comparable results [
43]. When the clinical examination and TVS fail to identify lesions in symptomatic individuals, MRI gives accurate information for DE staging (particularly in parametrial lesions) [
44]. When TVS is not an option, such as in cases of virgo intacta or obesity, MRI is useful. MRI can help determine the size and lateral extension of lesions before surgery, which is essential for surgical planning and approach [
44].
Bruyere proposed that MRI should be conducted by radiologists who are experts in interpreting female pelvic imaging, after he studied the discrepancies in diagnostic accuracy between radiologists with different levels of experience in the MRI evaluation of DE [
45]. Gynecological surgeons should recommend the patients suspected of endometriosis to imaging facilities with adequate expertise [
45].
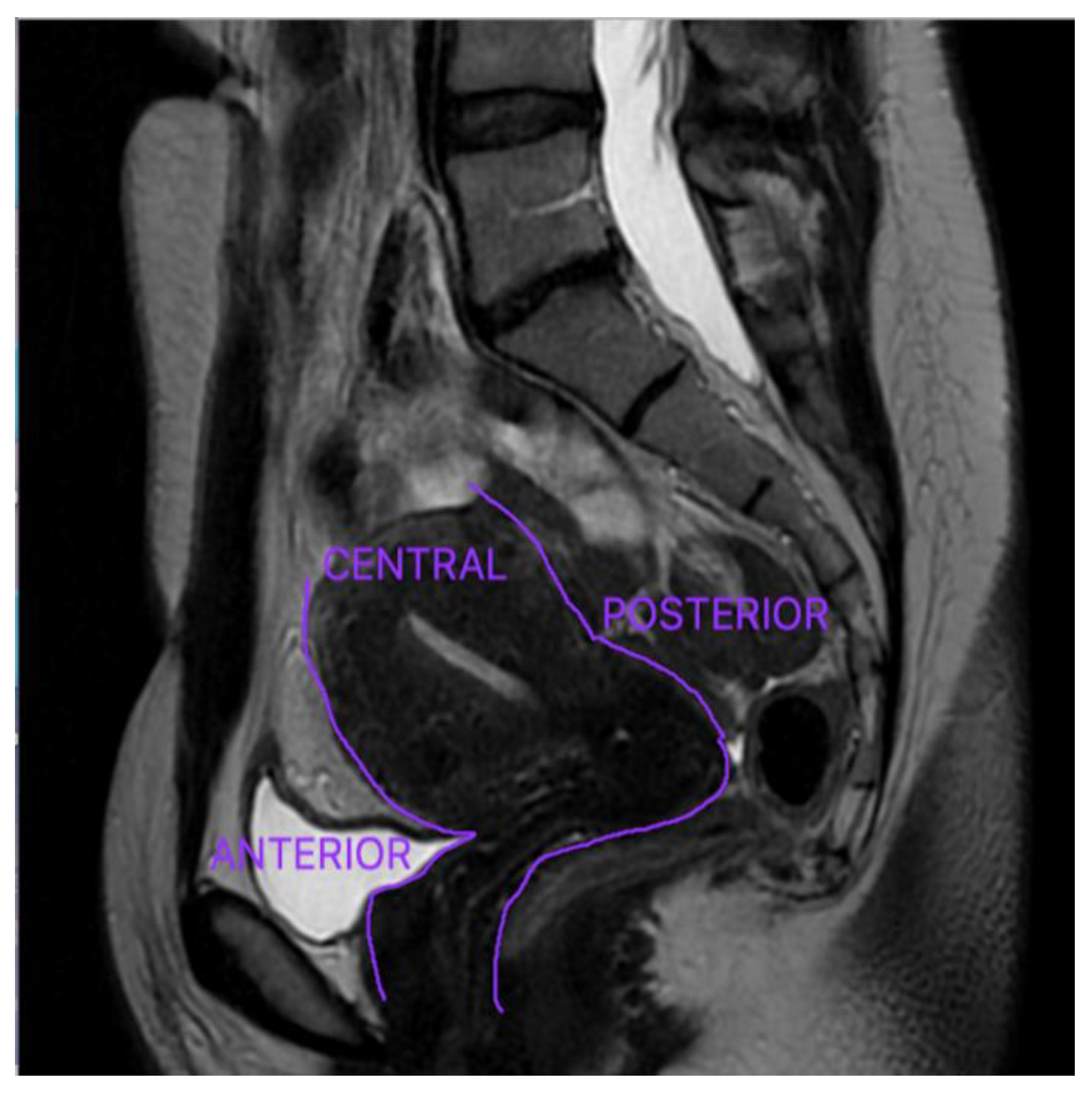
Anterior DE refers to a disease that affects the organs located in the anterior compartment: urinary bladder, the urethra, the vesicouterine pouch, and the round ligaments, and it is significantly less prevalent [
20]. The uterus and ovaries are located in the central compartment, whereas the Douglas pouch, uterine torus, USL, rectum, and sigmoid colon are situated in the posterior compartment—
Figure 2 [
33]. DE is most typically observed in the pelvic posterior compartment, with the USL having the highest prevalence [
17,
46].
Lorusso et al. described a standard MRI methodology for detecting endometriosis lesions that they utilize in their center [
33]. There is no agreement reached on the ideal imaging protocol worldwide. The research team illustrates the essential specifications they applied to acquire the most accurate results in detecting DE lesions in their endometriosis MRI protocol [
33].
The best-quality imaging for DE nodules is acquired when using a 1.5 Tesla or 3 Tesla scanner and high-resolution phased array coils (with 8–16 channels). The TSE (turbo spin-echo)-T2w sequences must be examined in the axial, sagittal, and coronal planes with high resolution (3 mm). Regarding the endometriomas, the authors recommend using the TSE T1w (with and without fat saturation) sequences [
33,
44].
The detection of DE is predicated on the juxtaposition between the high signal intensity of visceral fat and the low signal level of endometriotic nodules; therefore, fat-saturated T2w images are excluded from the approach [
33,
47].
The protocol does not require the use of routine rectal distension [
33]. However, there are benefits to rectal distention in patients with an endometriotic nodule infiltrating the rectum on standard TSE T2w imaging, which indicates the need for bowel excision [
48]. The patients can undergo an MRI scan independently of the menstrual cycle phase with the condition of a full bladder [
47].
Bazot et al. proposed a new series of guidelines for using MRI in diagnosing DE [
48]. MRI evaluation of DE uses vaginal and rectal opacification with sonographic gel [
8,
48].
The instillation of intravaginal and intrarectal gel relaxes the cavities, allowing for improved visualization of the walls and potential endometriosis nodules, and determining the depth of the infiltration zone [
48,
49]. The gel’s contrast allows for a clearer demarcation of the peritoneal recesses (recto-vaginal and bladder-vaginal recession) [
48,
50]. Peristaltic artifacts are also diminished when the gel is present [
48].
T2 hypointense regions attached to subsequent nodular thickening that induce anatomical distortion are often evident as adhesions [
51]. Superficial endometriosis lesions are more challenging to be detected on MRI, and are best discovered intraoperatively. If the lesions are “active” with bleeding, they are seen on the peritoneal surfaces as tiny T1 hyperintensities [
51].
Endometriosis ovarian cysts appear as thick-walled blood-filled tumors with uniformly high signal strength in the T1w sequence [
52]. These lesions are either hyperintense or hypointense in the T2w series, or can have a typical layered look (shading sign) due to cyclic bleeding and hemosiderin deposited in time [
44,
52]. Endometriomas are frequently known as “chocolate cysts” [
53]. Dark patches may appear within cysts in some situations in T2w sequences [
52]. Atypical thickenings or vegetations should be assessed to rule out malignant progression [
52].
MRI’s sensitivity and specificity for the diagnosis of endometriomas are 95% and 91%, respectively [
52].
Endometrial glands and stroma are densely packed with fibro-muscular and inflammation responses in DE nodules and plaque-like lesions, which have an uneven, spiculated appearance and an MRI signal intensity comparable to that of pelvic muscles [
54].
One of the most common locations for DE is the USL [
54]. Bilateral USL lesions correlate with the existence of other posterior DE nodules, especially rectal endometriosis [
55]. USL endometriosis lesions appear on the MRI sections as hypointense thickening of the ligament with regular or irregular borders [
54,
55]. The sensitivity and specificity of MRI in diagnosing USL endometriosis are 85% and 80%, respectively [
55].
The rectum and sigmoid colon are the most common locations of intestinal DE, and the cecum and ileum are involved in approximately 5% of cases [
33,
50,
56]. The presence of a solid or plaque-like intestinal wall thickening and disappearance of the visceral fat barrier between the rectosigmoid and the uterine wall or adnexa is used to diagnose bowel DE [
33]. The supplementary signs, such as the “mushroom cap” sign, aid in the correct identification of the disease [
56]. The gills of the mushroom are recreated by retractile T2 hypointense growth of the muscular stratum, whereas the mushroom cap is represented by a fine layer of T2 hyperintense submucosa and mucosa [
33,
56]. The sensitivity and specificity of MRI in diagnosing bowel endometriosis are 83% and 88%, respectively [
57].
The” kissing ovaries” sign can be distinguished on MRI as well, in the case of ovaries collapsing in the Douglas pouch and causing pelvic adhesions. When endometriosis lesions spread from the retro-cervical space to the anterior rectum, obliteration of the Douglas pouch can be considered [
57]. Small bowel movements between the uterus and the rectal wall exclude obliteration of the pouch Douglas [
56,
57]. The sensitivity and specificity of MRI for the assessment of endometriosis lesions of the pouch of Douglas are 89% are 94%, respectively [
55].
Bladder and urinary endometriosis are uncommon and specifically affect the vesical dome, paravesical lesions, and ureteral nodules [
58]. The lesions appear as single or diffuse wall thickening and signal intensity abnormalities [
53]. MRI and TVS are more reliable for detecting endometriosis nodules located in the ureters as opposed to the bladder [
58].
The most sensitive images for detecting ureters lesions are axial and sagittal TSE T2w images. Contrast MR urography can follow MRI to investigate the presence of ureterohydronephrosis [
57]. The sensitivity of MRI for diagnosing bladder endometriosis is 88%, the specificity is 99%, and the overall diagnostic accuracy reaches 98% [
55].