Mitochondrial Dysfunction in Peripheral Blood Mononuclear Cells as Novel Diagnostic Tools for Non-Alcoholic Fatty Liver Disease: Visualizing Relationships with Known and Potential Disease Biomarkers
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Measurement of Laboratory Data
2.3. Isolation of PBMCs
2.4. Extracellular Flux Analysis of PBMCs
2.5. Mitochondrial DNA Quantification
2.6. Measurement of Serum Cytokine IL-6 and TNF-α
2.7. Statistical Analysis
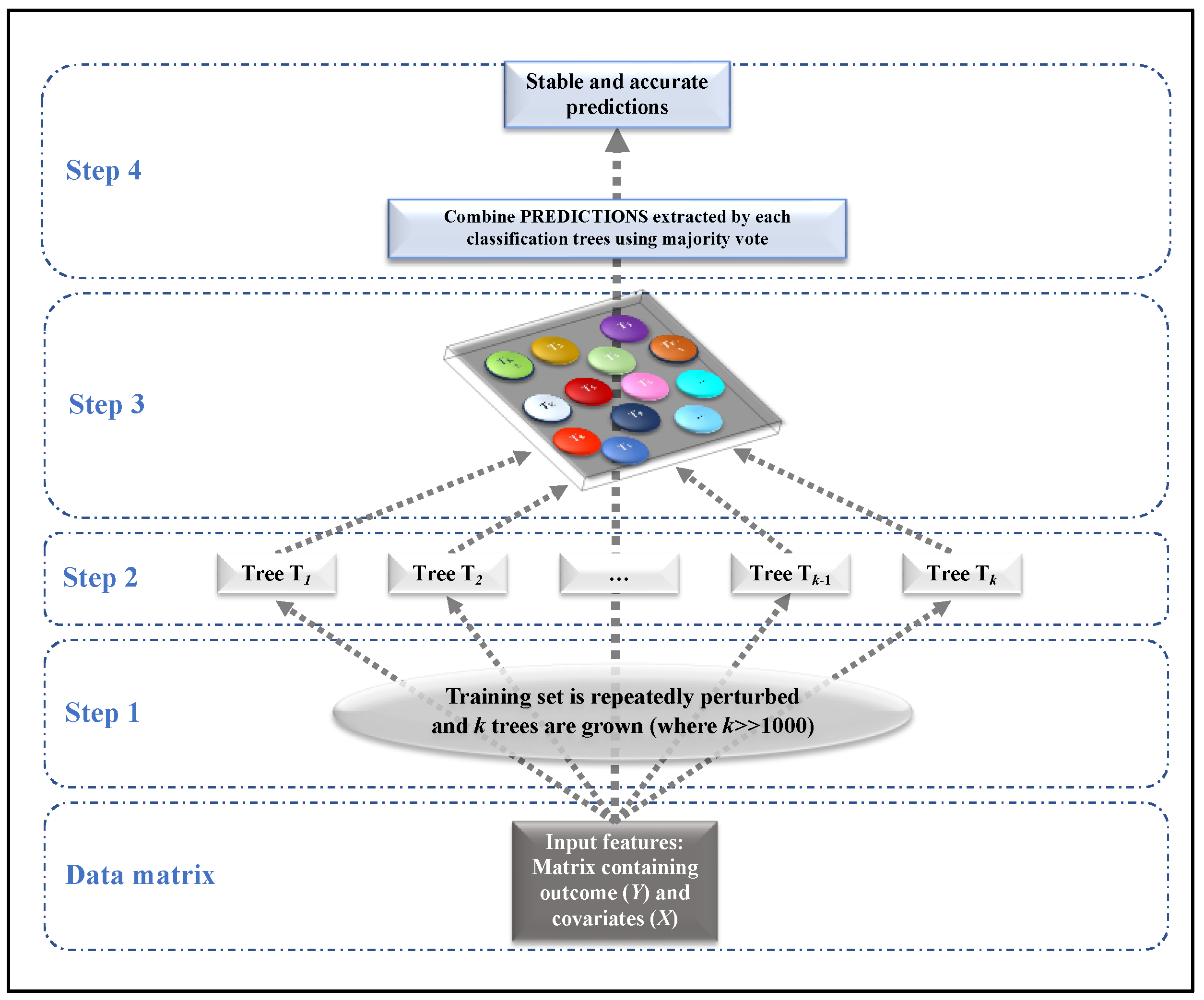
- Step 1: The data matrix is repeatedly perturbed, obtaining k different subsets of observations (usually k = 10,000).
- Step 2: For each perturbed subset obtained at Step 1, a single classification tree is grown.
- Step 3: Each classification tree provides predictions (classes) in correspondence to each subject.
- Step 4: Final prediction, which is stable and accurate, is obtained based on majority voting.
3. Results
3.1. Descriptive Statistics
3.1.1. Clinical, Anthropometric, and Biochemical Characteristics of the Subjects
3.1.2. Mitochondrial DNA Content and Function in PBMCs from the Study Cohort
3.1.3. Correlation
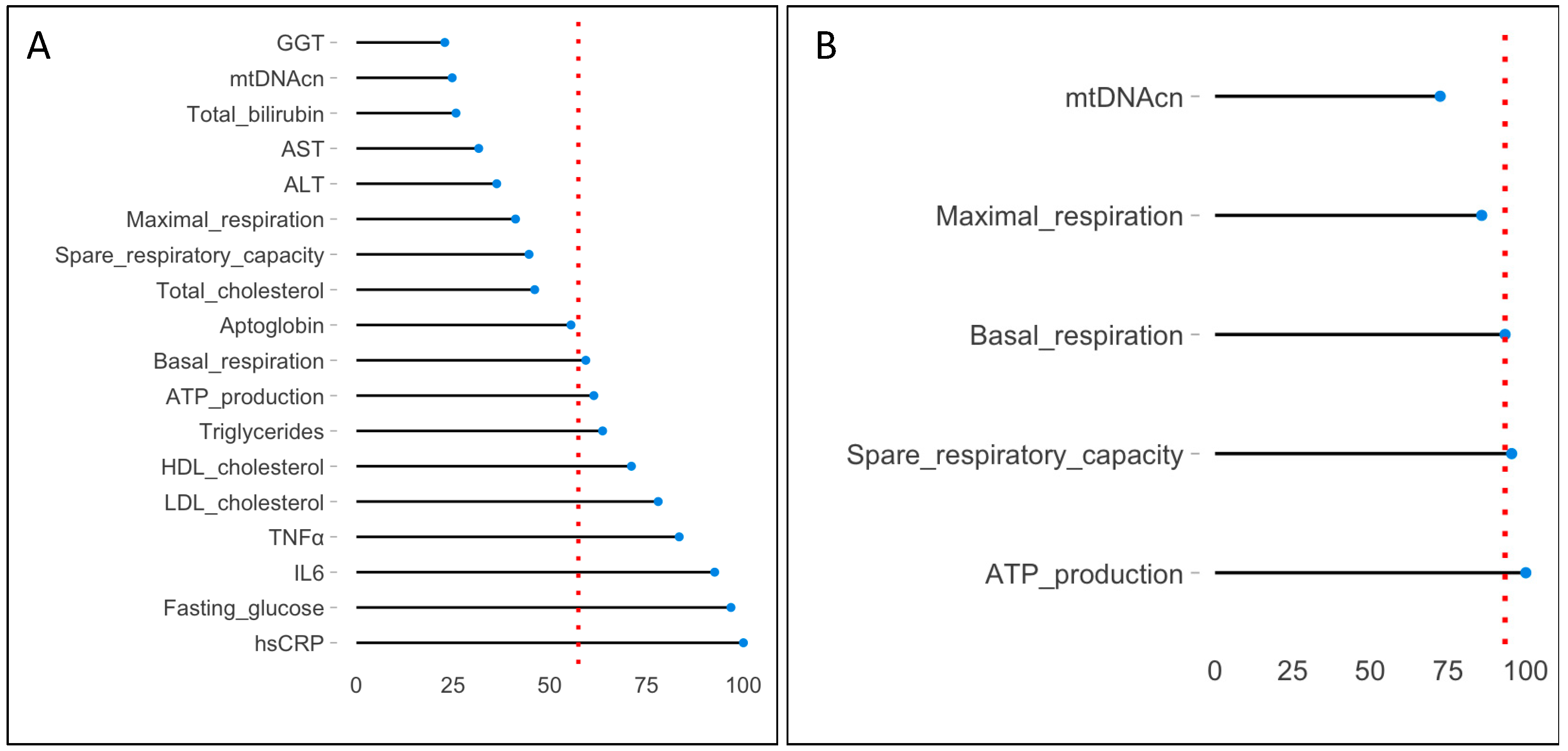
3.1.4. Random Forest and VIMrel
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Powell, E.E.; Wong, V.W.S.; Rinella, M. Non-Alcoholic Fatty Liver Disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL–EASD–EASO Clinical Practice Guidelines for the Management of Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.B.; Mehta, K.J. Liver Biopsy for Assessment of Chronic Liver Diseases: A Synopsis. Clin. Exp. Med. 2023, 23, 273–285. [Google Scholar] [CrossRef]
- Tilg, H.; Effenberger, M. From NAFLD to MAFLD: When Pathophysiology Succeeds. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 387–388. [Google Scholar] [CrossRef]
- Angulo, P.; Hui, J.M.; Marchesini, G.; Bugianesi, E.; George, J.; Farrell, G.C.; Enders, F.; Saksena, S.; Burt, A.D.; Bida, J.P.; et al. The NAFLD Fibrosis Score: A Noninvasive System That Identifies Liver Fibrosis in Patients with NAFLD. Hepatology 2007, 45, 846–854. [Google Scholar] [CrossRef]
- Tomizawa, M.; Kawanabe, Y.; Shinozaki, F.; Sato, S.; Motoyoshi, Y.; Sugiyama, T.; Yamamoto, S.; Sueishi, M. Elevated Levels of Alanine Transaminase and Triglycerides within Normal Limits Are Associated with Fatty Liver. Exp. Ther. Med. 2014, 8, 759–762. [Google Scholar] [CrossRef]
- Duan, Y.; Pan, X.; Luo, J.; Xiao, X.; Li, J.; Bestman, P.L.; Luo, M. Association of Inflammatory Cytokines with Non-Alcoholic Fatty Liver Disease. Front. Immunol. 2022, 13, 880298. [Google Scholar] [CrossRef]
- Kurbatova, I.V.; Topchieva, L.V.; Dudanova, O.P.; Shipovskaya, A.A. The Role of the Soluble Interleukin-6 Receptor in the Progression of Nonalcoholic Fatty Liver Disease. Bull. Exp. Biol. Med. 2022, 174, 585–591. [Google Scholar] [CrossRef]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The Fatty Liver Index: A Simple and Accurate Predictor of Hepatic Steatosis in the General Population. BMC Gastroenterol. 2006, 6, 33. [Google Scholar] [CrossRef]
- Ratziu, V.; Massard, J.; Charlotte, F.; Messous, D.; Imbert-Bismut, F.; Bonyhay, L.; Tahiri, M.; Munteanu, M.; Thabut, D.; Cadranel, J.F.; et al. Diagnostic Value of Biochemical Markers (Fibro Test-FibroSURE) for the Prediction of Liver Fibrosis in Patients with Non-Alcoholic Fatty Liver Disease. BMC Gastroenterol. 2006, 6, 6. [Google Scholar] [CrossRef]
- Stefan, N.; Häring, H.-U.; Cusi, K. Non-Alcoholic Fatty Liver Disease: Causes, Diagnosis, Cardiometabolic Consequences, and Treatment Strategies. Lancet Diabetes Endocrinol. 2019, 7, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Prasun, P.; Ginevic, I.; Oishi, K. Mitochondrial Dysfunction in Nonalcoholic Fatty Liver Disease and Alcohol Related Liver Disease. Transl. Gastroenterol. Hepatol. 2021, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Legaki, A.I.; Moustakas, I.I.; Sikorska, M.; Papadopoulos, G.; Velliou, R.I.; Chatzigeorgiou, A. Hepatocyte Mitochondrial Dynamics and Bioenergetics in Obesity-Related Non-Alcoholic Fatty Liver Disease. Curr. Obes. Rep. 2022, 11, 126–143. [Google Scholar] [CrossRef] [PubMed]
- Fricker, Z.P.; Pedley, A.; Massaro, J.M.; Vasan, R.S.; Hoffmann, U.; Benjamin, E.J.; Long, M.T. Liver Fat Is Associated with Markers of Inflammation and Oxidative Stress in Analysis of Data from the Framingham Heart Study. Clin. Gastroenterol. Hepatol. 2019, 17, 1157–1164.e4. [Google Scholar] [CrossRef]
- Koliaki, C.; Szendroedi, J.; Kaul, K.; Jelenik, T.; Nowotny, P.; Jankowiak, F.; Herder, C.; Carstensen, M.; Krausch, M.; Knoefel, W.T.; et al. Adaptation of Hepatic Mitochondrial Function in Humans with Non-Alcoholic Fatty Liver Is Lost in Steatohepatitis. Cell Metab. 2015, 21, 739–746. [Google Scholar] [CrossRef]
- Braganza, A.; Annarapu, G.K.; Shiva, S. Blood-Based Bioenergetics: An Emerging Translational and Clinical Tool. Mol. Aspects Med. 2020, 71, 100835. [Google Scholar] [CrossRef]
- Hill, B.G.; Shiva, S.; Ballinger, S.; Zhang, J.; Darley-Usmar, V.M. Bioenergetics and Translational Metabolism: Implications for Genetics, Physiology and Precision Medicine. Biol. Chem. 2019, 401, 3–29. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis Model Assessment: Insulin Resistance and β-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Kramer, P.A.; Chacko, B.K.; Ravi, S.; Johnson, M.S.; Mitchell, T.; Darley-Usmar, V.M. Bioenergetics and the Oxidative Burst: Protocols for the Isolation and Evaluation of Human Leukocytes and Platelets. J. Vis. Exp. 2014, 85, e51301. [Google Scholar] [CrossRef]
- Bonnen, P.E.; Yarham, J.W.; Besse, A.; Wu, P.; Faqeih, E.A.; Al-Asmari, A.M.; Saleh, M.A.M.; Eyaid, W.; Hadeel, A.; He, L.; et al. Mutations in FBXL4 Cause Mitochondrial Encephalopathy and a Disorder of Mitochondrial DNA Maintenance. Am. J. Hum. Genet. 2013, 93, 471–481. [Google Scholar] [CrossRef]
- Picard, M.; Prather, A.A.; Puterman, E.; Cuillerier, A.; Coccia, M.; Aschbacher, K.; Burelle, Y.; Epel, E.S. A Mitochondrial Health Index Sensitive to Mood and Caregiving Stress. Biol. Psychiatry 2018, 84, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, F.; D’Amato, I.; Jensen, P.B.; Ravaglia, S.; Zeviani, M.; Tiranti, V. Microscale Oxygraphy Reveals OXPHOS Impairment in MRC Mutant Cells. Mitochondrion 2012, 12, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, D.; Dusi, S.; Morbin, M.; Uggetti, A.; Moda, F.; D’Amato, I.; Giordano, C.; D’Amati, G.; Cozzi, A.; Levi, S.; et al. Pantothenate Kinase-Associated Neurodegeneration: Altered Mitochondria Membrane Potential and Defective Respiration in Pank2 Knock-out Mouse Model. Hum. Mol. Genet. 2012, 21, 5294–5305. [Google Scholar] [CrossRef] [PubMed]
- Dancelli, L.; Manisera, M.; Vezzoli, M. On Two Classes of Weighted Rank Correlation Measures Deriving from the Spearman’s ρ. In Statistical Models for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2013; pp. 107–114. [Google Scholar]
- Abate, G.; Vezzoli, M.; Sandri, M.; Rungratanawanich, W.; Memo, M.; Uberti, D. Mitochondria and Cellular Redox State on the Route from Ageing to Alzheimer’s Disease. Mech. Ageing Dev. 2020, 192, 111385. [Google Scholar] [CrossRef] [PubMed]
- Codenotti, S.; Vezzoli, M.; Poliani, P.L.; Cominelli, M.; Bono, F.; Kabbout, H.; Faggi, F.; Chiarelli, N.; Colombi, M.; Zanella, I.; et al. Caveolin-1, Caveolin-2 and Cavin-1 Are Strong Predictors of Adipogenic Differentiation in Human Tumors and Cell Lines of Liposarcoma. Eur. J. Cell Biol. 2016, 95, 252–264. [Google Scholar] [CrossRef]
- Vezzoli, M.; Inciardi, R.M.; Oriecuia, C.; Paris, S.; Murillo, N.H.; Agostoni, P.; Ameri, P.; Bellasi, A.; Camporotondo, R.; Canale, C.; et al. Machine Learning for Prediction of In-Hospital Mortality in Coronavirus Disease 2019 Patients: Results from an Italian Multicenter Study. J. Cardiovasc. Med. 2022, 23, 439–446. [Google Scholar] [CrossRef]
- Azzolina, D.; Baldi, I.; Barbati, G.; Berchialla, P.; Bottigliengo, D.; Bucci, A.; Calza, S.; Dolce, P.; Edefonti, V.; Faragalli, A.; et al. Machine Learning in Clinical and Epidemiological Research: Isn’t It Time for Biostatisticians to Work on It? Epidemiol. Biostat. Public Health 2022, 16, 4. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Garrafa, E.; Vezzoli, M.; Ravanelli, M.; Farina, D.; Borghesi, A.; Calza, S.; Maroldi, R. Early Prediction of In-Hospital Death of COVID-19 Patients: A Machine-Learning Model Based on Age, Blood Analyses, and Chest X-Ray Score. eLife 2021, 10, e70640. [Google Scholar] [CrossRef]
- Vezzoli, M.; Ravaggi, A.; Zanotti, L.; Miscioscia, R.A.; Bignotti, E.; Ragnoli, M.; Gambino, A.; Ruggeri, G.; Calza, S.; Sartori, E.; et al. RERT: A Novel Regression Tree Approach to Predict Extrauterine Disease in Endometrial Carcinoma Patients. Sci. Rep. 2017, 7, 10528. [Google Scholar] [CrossRef]
- Minelli, A.; Silva, R.C.; Barlati, S.; Vezzoli, M.; Carletto, S.; Isabello, C.; Bortolomasi, M.; Nibbio, G.; Lisoni, J.; Menesello, V.; et al. The Elephant in the Room: A Cross-Sectional Study on the Stressful Psychological Effects of the COVID-19 Pandemic in Mental Healthcare Workers. Brain Sci. 2022, 12, 408. [Google Scholar] [CrossRef] [PubMed]
- Carpita, M.; Vezzoli, M. Statistical Evidence of the Subjective Work Quality: The Fairness Drivers of the Job Satisfaction. Electron. J. Appl. Stat. Anal. 2012, 5, 89–107. [Google Scholar] [CrossRef]
- Vezzoli, M. Exploring the Facets of Overall Job Satisfaction through a Novel Ensemble Learning. Electron. J. Appl. Stat. Anal. 2011, 4, 23–38. [Google Scholar] [CrossRef]
- Divakaruni, A.S.; Paradyse, A.; Ferrick, D.A.; Murphy, A.N.; Jastroch, M. Analysis and Interpretation of Microplate-Based Oxygen Consumption and PH Data. Methods Enzymol. 2014, 547, 309–354. [Google Scholar] [PubMed]
- Ajaz, S.; McPhail, M.J.; Gnudi, L.; Trovato, F.M.; Mujib, S.; Napoli, S.; Carey, I.; Agarwal, K. Mitochondrial Dysfunction as a Mechanistic Biomarker in Patients with Non-Alcoholic Fatty Liver Disease (NAFLD). Mitochondrion 2021, 57, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Ritz, T.; Krenkel, O.; Tacke, F. Dynamic Plasticity of Macrophage Functions in Diseased Liver. Cell. Immunol. 2018, 330, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, Y.; Sottas, C.; Lazaris, A.; Petrillo, S.K.; Metrakos, P.; Li, L.; Ishida, Y.; Saito, T.; Garza, S.; et al. Loss of Mitochondrial ATPase ATAD3A Contributes to Nonalcoholic Fatty Liver Disease through Accumulation of Lipids and Damaged Mitochondria. J. Biol. Chem. 2022, 298, 102008. [Google Scholar] [CrossRef]
- Li, Q.; Rempel, J.D.; Yang, J.; Minuk, G.Y. The Effects of Pathogen-Associated Molecular Patterns on Peripheral Blood Monocytes in Patients with Non-Alcoholic Fatty Liver Disease. J. Clin. Exp. Hepatol. 2022, 12, 808–817. [Google Scholar] [CrossRef]
- Kim, J.; Wei, Y.; Sowers, J.R. Role of Mitochondrial Dysfunction in Insulin Resistance. Circ. Res. 2008, 102, 401–414. [Google Scholar] [CrossRef]
- Barazzoni, R.; Gortan Cappellari, G.; Ragni, M.; Nisoli, E. Insulin Resistance in Obesity: An Overview of Fundamental Alterations. Eat. Weight. Disord. Stud. Anorex. Bulim. Obes. 2018, 23, 149–157. [Google Scholar] [CrossRef]
- DeConne, T.M.; Muñoz, E.R.; Sanjana, F.; Hobson, J.C.; Martens, C.R. Cardiometabolic Risk Factors Are Associated with Immune Cell Mitochondrial Respiration in Humans. Am. J. Physiol.-Heart Circ. Physiol. 2020, 319, H481–H487. [Google Scholar] [CrossRef] [PubMed]
- Mocciaro, G.; Allison, M.; Jenkins, B.; Azzu, V.; Huang-Doran, I.; Herrera-Marcos, L.V.; Hall, Z.; Murgia, A.; Susan, D.; Frontini, M.; et al. Non-Alcoholic Fatty Liver Disease Is Characterised by a Reduced Polyunsaturated Fatty Acid Transport via Free Fatty Acids and High-Density Lipoproteins (HDL). Mol. Metab. 2023, 73, 101728. [Google Scholar] [CrossRef]
- Newman, T.M.; Shively, C.A.; Register, T.C.; Appt, S.E.; Yadav, H.; Colwell, R.R.; Fanelli, B.; Dadlani, M.; Graubics, K.; Nguyen, U.T.; et al. Diet, Obesity, and the Gut Microbiome as Determinants Modulating Metabolic Outcomes in a Non-Human Primate Model. Microbiome 2021, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Bonder, M.J.; Cenit, M.C.; Tigchelaar, E.F.; Maatman, A.; Dekens, J.A.M.; Brandsma, E.; Marczynska, J.; Imhann, F.; Weersma, R.K.; et al. The Gut Microbiome Contributes to a Substantial Proportion of the Variation in Blood Lipids. Circ. Res. 2015, 117, 817–824. [Google Scholar] [CrossRef]
- Franco-Obregón, A.; Gilbert, J.A. The Microbiome-Mitochondrion Connection: Common Ancestries, Common Mechanisms, Common Goals. mSystems 2017, 2, e00018-17. [Google Scholar] [CrossRef]
- Archetti, C.; Montanelli, A.; Finazzi, D.; Caimi, L.; Garrafa, E. Clinical Laboratory Automation: A Case Study. J. Public Health Res. 2017, 6, 881. [Google Scholar] [CrossRef] [PubMed]
- Garrafa, E.; Brugnoni, D.; Barbaro, M.; Andreoli, L.; Focà, E.; Salvetti, M.; Castelli, F.; Franceschini, F.; Piva, S.; Muiesan, M.L.; et al. Laboratory Considerations amidst the Coronavirus Disease 2019 Outbreak: The Spedali Civili in Brescia Experience. Bioanalysis 2020, 12, 1223–1230. [Google Scholar] [CrossRef]
- Signorini, S.G.; Brugnoni, D.; Levaggi, R.; Garrafa, E. Less Is More: An Ecological and Economic Point of View on Appropriate Use of Lab Testing for COVID-19 Patients. Bioanalysis 2021, 13, 1781–1783. [Google Scholar] [CrossRef]
- Grattagliano, I.; Montezinho, L.P.; Oliveira, P.J.; Frühbeck, G.; Gómez-Ambrosi, J.; Montecucco, F.; Carbone, F.; Wieckowski, M.R.; Wang, D.Q.-H.; Portincasa, P. Targeting Mitochondria to Oppose the Progression of Nonalcoholic Fatty Liver Disease. Biochem. Pharmacol. 2019, 160, 34–45. [Google Scholar] [CrossRef]
- Ruocco, C.; Segala, A.; Valerio, A.; Nisoli, E. Essential Amino Acid Formulations to Prevent Mitochondrial Dysfunction and Oxidative Stress. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 88–95. [Google Scholar] [CrossRef]

| Variables | NAFLD | Non-NAFLD | Total | p Value |
|---|---|---|---|---|
| (N = 19) | (N = 18) | (N = 37) | ||
| Sex | 0.330 (a) | |||
| Female | 8 (42.1%) | 11 (61.1%) | 19 (51.4%) | |
| Male | 11 (57.9%) | 7 (38.9%) | 18 (48.6%) | |
| Age (years) | 0.068 (b) | |||
| Mean (SD) | 51.84 (4.15) | 48.44 (5.74) | 50.19 (5.21) | |
| Median (Q1, Q3) | 52.00 (50.00, 54.50) | 48.00 (43.25, 52.75) | 51.00 (47.00, 54.00) | |
| Range | 42.00–59.00 | 40.00–59.00 | 40.00–59.00 | |
| BMI (Kg/m2) | <0.001 (b) | |||
| Mean (SD) | 32.62 (6.93) | 23.49 (3.64) | 28.18 (7.18) | |
| Median (Q1, Q3) | 31.30 (27.85, 34.55) | 23.40 (20.50, 25.77) | 27.00 (23.50, 31.50) | |
| Range | 25.20–55.00 | 17.60–31.70 | 17.60–55.00 | |
| Waist Circumference (cm) | <0.001 (b) | |||
| N-Miss | 0 | 2 | 2 | |
| Mean (SD) | 107.34 (17.71) | 86.97 (9.42) | 98.03 (17.64) | |
| Median (Q1, Q3) | 105.00 (96.50, 112.00) | 87.50 (81.00, 94.62) | 96.00 (88.00, 106.25) | |
| Range | 86.00–169.00 | 70.00–99.00 | 70.00–169.00 | |
| Fasting glucose (mg/dL) | 0.014 (b) | |||
| Mean (SD) | 99.37 (22.30) | 86.39 (11.26) | 93.05 (18.76) | |
| Median (Q1, Q3) | 91.00 (86.00, 105.00) | 81.50 (77.75, 95.25) | 88.00 (81.00, 98.00) | |
| Range | 81.00–169.00 | 74.00–115.00 | 74.00–169.00 | |
| Fasting insulin (µUI/mL) | <0.001 (b) | |||
| Mean (SD) | 14.21 (9.76) | 4.44 (3.38) | 9.46 (8.81) | |
| Median (Q1, Q3) | 12.00 (8.50, 18.00) | 4.00 (2.00, 5.00) | 8.00 (4.00, 13.00) | |
| Range | 2.00–42.00 | 1.00–14.00 | 1.00–42.00 | |
| HOMA index | <0.001 (b) | |||
| Mean (SD) | 3.60 (2.78) | 1.02 (0.95) | 2.34 (2.45) | |
| Median (Q1, Q3) | 2.86 (1.72, 4.69) | 0.81 (0.40, 1.16) | 1.60 (0.79, 3.11) | |
| Range | 0.42–11.27 | 0.18–3.98 | 0.18–11.27 | |
| Total cholesterol (mg/dL) | 0.027 (b) | |||
| Mean (SD) | 209.37 (28.51) | 185.94 (31.09) | 197.97 (31.68) | |
| Median (Q1, Q3) | 212.00 (189.50, 230.00) | 185.00 (167.75, 200.25) | 195.00 (176.00, 218.00) | |
| Range | 165.00–262.00 | 117.00–259.00 | 117.00–262.00 | |
| HDL cholesterol (mg/dL) | 0.004 (b) | |||
| Mean (SD) | 51.36 (13.93) | 67.49 (16.96) | 59.21 (17.31) | |
| Median (Q1, Q3) | 47.60 (39.65, 62.80) | 69.50 (53.32, 75.33) | 55.60 (46.10, 71.80) | |
| Range | 33.20–77.60 | 43.30–112.80 | 33.20–112.80 | |
| LDL cholesterol (mg/dL) | 0.008 (b) | |||
| Mean (SD) | 131.53 (29.39) | 102.72 (31.99) | 117.51 (33.59) | |
| Median (Q1, Q3) | 127.00 (118.00, 154.50) | 95.50 (86.25, 118.75) | 119.00 (94.00, 142.00) | |
| Range | 72.00–194.00 | 37.00–166.00 | 37.00–194.00 | |
| Triglycerides (mg/dL) | 0.006 (b) | |||
| Mean (SD) | 132.37 (73.47) | 78.78 (45.27) | 106.30 (66.37) | |
| Median (Q1, Q3) | 126.00 (71.00, 155.00) | 64.00 (49.75, 91.75) | 77.00 (60.00, 131.00) | |
| Range | 57.00–301.00 | 32.00–219.00 | 32.00–301.00 | |
| ALT (U/L) | 0.163 (b) | |||
| N-Miss | 0 | 1 | 1 | |
| Mean (SD) | 33.89 (14.49) | 31.12 (23.40) | 32.58 (18.98) | |
| Median (Q1, Q3) | 32.00 (22.50, 40.50) | 23.00 (18.00, 32.00) | 30.50 (19.00, 35.50) | |
| Range | 13.00–64.00 | 15.00–108.00 | 13.00–108.00 | |
| AST (U/L) | 0.867 (b) | |||
| Mean (SD) | 16.79 (6.24) | 18.28 (10.81) | 17.51 (8.67) | |
| Median (Q1, Q3) | 16.00 (11.50, 22.00) | 14.50 (10.25, 22.75) | 15.00 (11.00, 22.00) | |
| Range | 8.00–27.00 | 6.00–43.00 | 6.00–43.00 | |
| Total bilirubin (mg/dL) | 0.648 (b) | |||
| Mean (SD) | 1.36 (3.43) | 0.66 (0.29) | 1.02 (2.46) | |
| Median (Q1, Q3) | 0.55 (0.48, 0.66) | 0.56 (0.53, 0.69) | 0.56 (0.50, 0.66) | |
| Range | 0.29–15.50 | 0.35–1.42 | 0.29–15.50 | |
| GGT (U/L) | 0.564 (b) | |||
| Mean (SD) | 33.58 (40.86) | 27.85 (34.71) | 30.79 (37.58) | |
| Median (Q1, Q3) | 22.70 (11.60, 35.50) | 16.40 (12.67, 21.55) | 17.10 (12.40, 32.70) | |
| Range | 0.50–178.60 | 5.30–152.40 | 0.50–178.60 | |
| Haptoglobin (mg/dL) | 0.098 (b) | |||
| Mean (SD) | 133.71 (57.92) | 106.71 (48.92) | 120.58 (54.73) | |
| Median (Q1, Q3) | 148.00 (87.30, 170.00) | 119.00 (68.10, 134.75) | 121.00 (79.40, 165.00) | |
| Range | 21.00–224.00 | 21.00–202.00 | 21.00–224.00 | |
| hsCRP (mg/L) | 0.007 (b) | |||
| Mean (SD) | 3.26 (3.36) | 1.32 (1.49) | 2.31 (2.77) | |
| Median (Q1, Q3) | 1.43 (1.02, 4.25) | 0.61 (0.34, 1.70) | 1.13 (0.56, 2.70) | |
| Range | 0.33–10.00 | 0.08–4.96 | 0.08–10.00 | |
| TNF-α (pg/mL) | 0.025 (b) | |||
| N-Miss | 0 | 1 | 1 | |
| Mean (SD) | 3.26 (1.64) | 2.34 (1.32) | 2.83 (1.55) | |
| Median (Q1, Q3) | 3.02 (2.59, 3.38) | 2.38 (1.47, 2.60) | 2.60 (2.15, 3.37) | |
| Range | 1.62–9.37 | 0.00–5.68 | 0.00–9.37 | |
| IL-6 (pg/mL) | 0.006 (b) | |||
| Mean (SD) | 2.40 (2.11) | 1.07 (0.73) | 1.75 (1.72) | |
| Median (Q1, Q3) | 1.55 (1.11, 2.70) | 0.95 (0.61, 1.30) | 1.19 (0.80, 1.58) | |
| Range | 0.53–8.40 | 0.24–3.65 | 0.24–8.40 | |
| Fatty Liver Index | <0.001 (b) | |||
| N-Miss | 0 | 2 | 2 | |
| Mean (SD) | 67.95 (29.34) | 20.13 (18.19) | 46.09 (34.44) | |
| Median (Q1, Q3) | 80.59 (55.52, 89.81) | 14.53 (3.78, 33.95) | 39.57 (14.53, 80.68) | |
| Range | 3.35–98.61 | 1.32–60.69 | 1.32–98.61 |
| Variables | NAFLD | Non-NAFLD | Total | p Value |
|---|---|---|---|---|
| (N = 19) | (N = 18) | (N = 37) | ||
| mtDNAcn | 0.318 (a) | |||
| N-Miss | 0 | 1 | 1 | |
| Mean (SD) | 3.32 (0.43) | 3.46 (0.37) | 3.39 (0.40) | |
| Median (Q1, Q3) | 3.32 (2.91, 3.76) | 3.54 (3.15, 3.80) | 3.39 (3.02, 3.80) | |
| Range | 2.78–3.97 | 2.89–3.94 | 2.78–3.97 | |
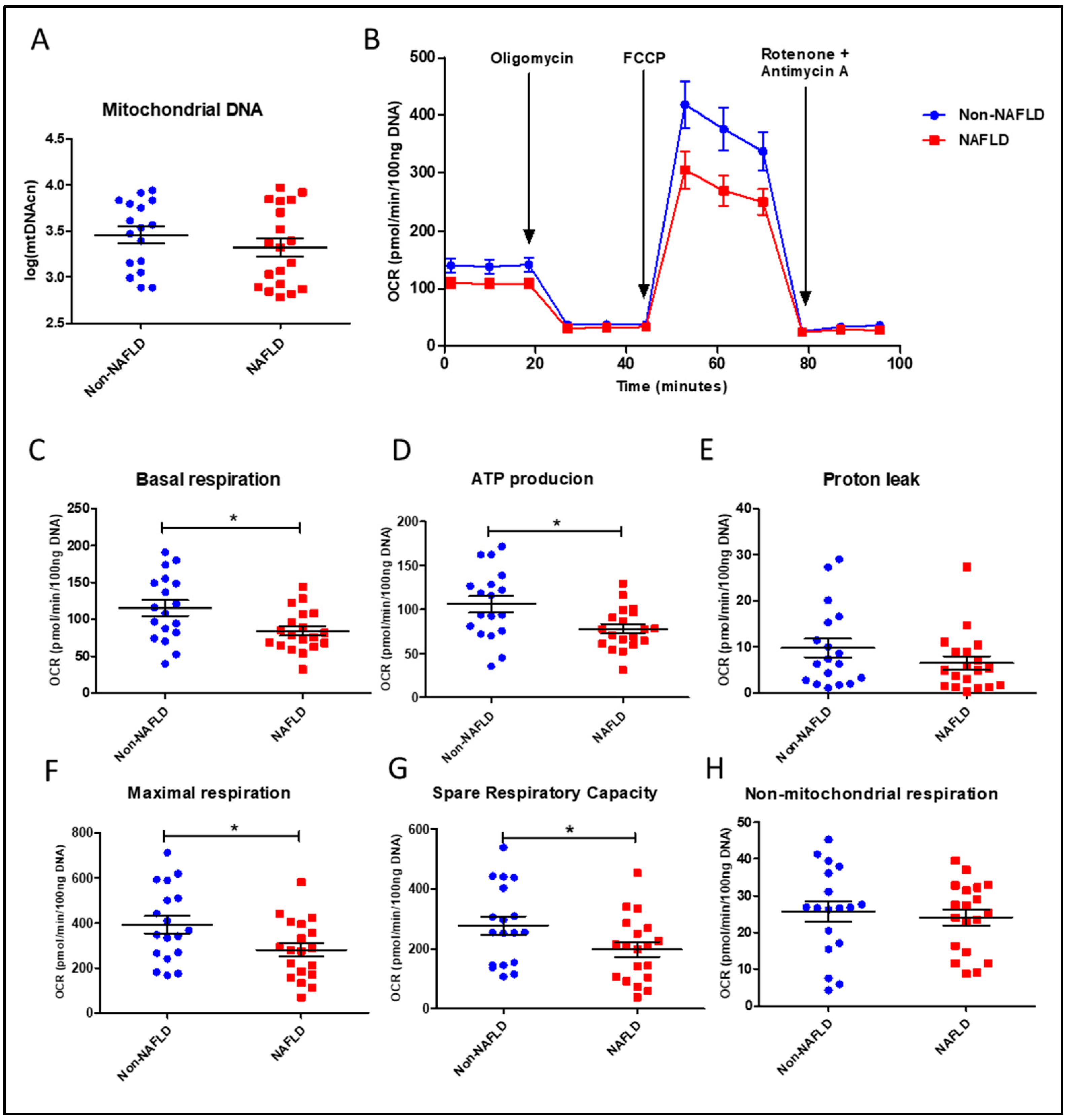
| Basal respiration | 0.023 (a) | |||
| Mean (SD) | −0.37 (0.74) | 0.45 (1.17) | 0.03 (1.05) | |
| Median (Q1, Q3) | −0.52 (−0.84, 0.09) | 0.37 (−0.39, 1.34) | −0.24 (−0.73, 0.63) | |
| Range | −1.74–1.22 | −1.54–2.45 | −1.74–2.45 | |
| ATP production | 0.021 (a) | |||
| Mean (SD) | −0.37 (0.71) | 0.46 (1.17) | 0.04 (1.04) | |
| Median (Q1, Q3) | −0.39 (−0.83, 0.10) | 0.43 (−0.40, 1.12) | −0.11 (−0.63, 0.77) | |
| Range | −1.77–1.17 | −1.64–2.41 | −1.77–2.41 | |
| Proton leak | 0.191 (a) | |||
| Mean (SD) | −0.19 (0.80) | 0.22 (1.08) | 0.01 (0.96) | |
| Median (Q1, Q3) | −0.39 (−0.81, 0.11) | −0.15 (−0.64, 0.79) | −0.27 (−0.75, 0.30) | |
| Range | −0.97–2.41 | −0.87–2.62 | −0.97–2.62 | |
| Maximal respiration | 0.045 (a) | |||
| Mean (SD) | −0.30 (0.81) | 0.38 (1.01) | 0.03 (0.96) | |
| Median (Q1, Q3) | −0.31 (−0.93, 0.28) | 0.16 (−0.39, 1.08) | 0.02 (−0.73, 0.57) | |
| Range | −1.60–1.52 | −1.00–2.33 | −1.6–2.33 | |
| Spare respiratory capacity | 0.045 (a) | |||
| Mean (SD) | −0.26 (0.81) | 0.33 (0.98) | 0.02 (0.93) | |
| Median (Q1, Q3) | −0.18 (−0.94, 0.20) | 0.16 (−0.63, 1.09) | −0.06 (−0.69, 0.55) | |
| Range | −1.46–1.63 | −0.93–2.27 | −1.46–2.27 | |
| Non-mitochondrial respiration | 0.715 (a) | |||
| Mean (SD) | −0.06 (0.82) | 0.08 (1.04) | 0.01 (0.92) | |
| Median (Q1, Q3) | 0.07 (−0.79, 0.60) | 0.17 (−0.58, 0.86) | 0.17 (−0.72, 0.64) | |
| Range | −1.37–1.26 | −1.75–1.76 | −1.75–1.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrafa, E.; Segala, A.; Vezzoli, M.; Bottani, E.; Zanini, B.; Vetturi, A.; Bracale, R.; Ricci, C.; Valerio, A. Mitochondrial Dysfunction in Peripheral Blood Mononuclear Cells as Novel Diagnostic Tools for Non-Alcoholic Fatty Liver Disease: Visualizing Relationships with Known and Potential Disease Biomarkers. Diagnostics 2023, 13, 2363. https://doi.org/10.3390/diagnostics13142363
Garrafa E, Segala A, Vezzoli M, Bottani E, Zanini B, Vetturi A, Bracale R, Ricci C, Valerio A. Mitochondrial Dysfunction in Peripheral Blood Mononuclear Cells as Novel Diagnostic Tools for Non-Alcoholic Fatty Liver Disease: Visualizing Relationships with Known and Potential Disease Biomarkers. Diagnostics. 2023; 13(14):2363. https://doi.org/10.3390/diagnostics13142363
Chicago/Turabian StyleGarrafa, Emirena, Agnese Segala, Marika Vezzoli, Emanuela Bottani, Barbara Zanini, Alice Vetturi, Renata Bracale, Chiara Ricci, and Alessandra Valerio. 2023. "Mitochondrial Dysfunction in Peripheral Blood Mononuclear Cells as Novel Diagnostic Tools for Non-Alcoholic Fatty Liver Disease: Visualizing Relationships with Known and Potential Disease Biomarkers" Diagnostics 13, no. 14: 2363. https://doi.org/10.3390/diagnostics13142363
APA StyleGarrafa, E., Segala, A., Vezzoli, M., Bottani, E., Zanini, B., Vetturi, A., Bracale, R., Ricci, C., & Valerio, A. (2023). Mitochondrial Dysfunction in Peripheral Blood Mononuclear Cells as Novel Diagnostic Tools for Non-Alcoholic Fatty Liver Disease: Visualizing Relationships with Known and Potential Disease Biomarkers. Diagnostics, 13(14), 2363. https://doi.org/10.3390/diagnostics13142363







