Expression Patterns of PAK4 and PHF8 Are Associated with the Survival of Gallbladder Carcinoma Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. GBC Patients
2.2. Immunohistochemical Staining and Scoring
2.3. Statistical Analysis
3. Results
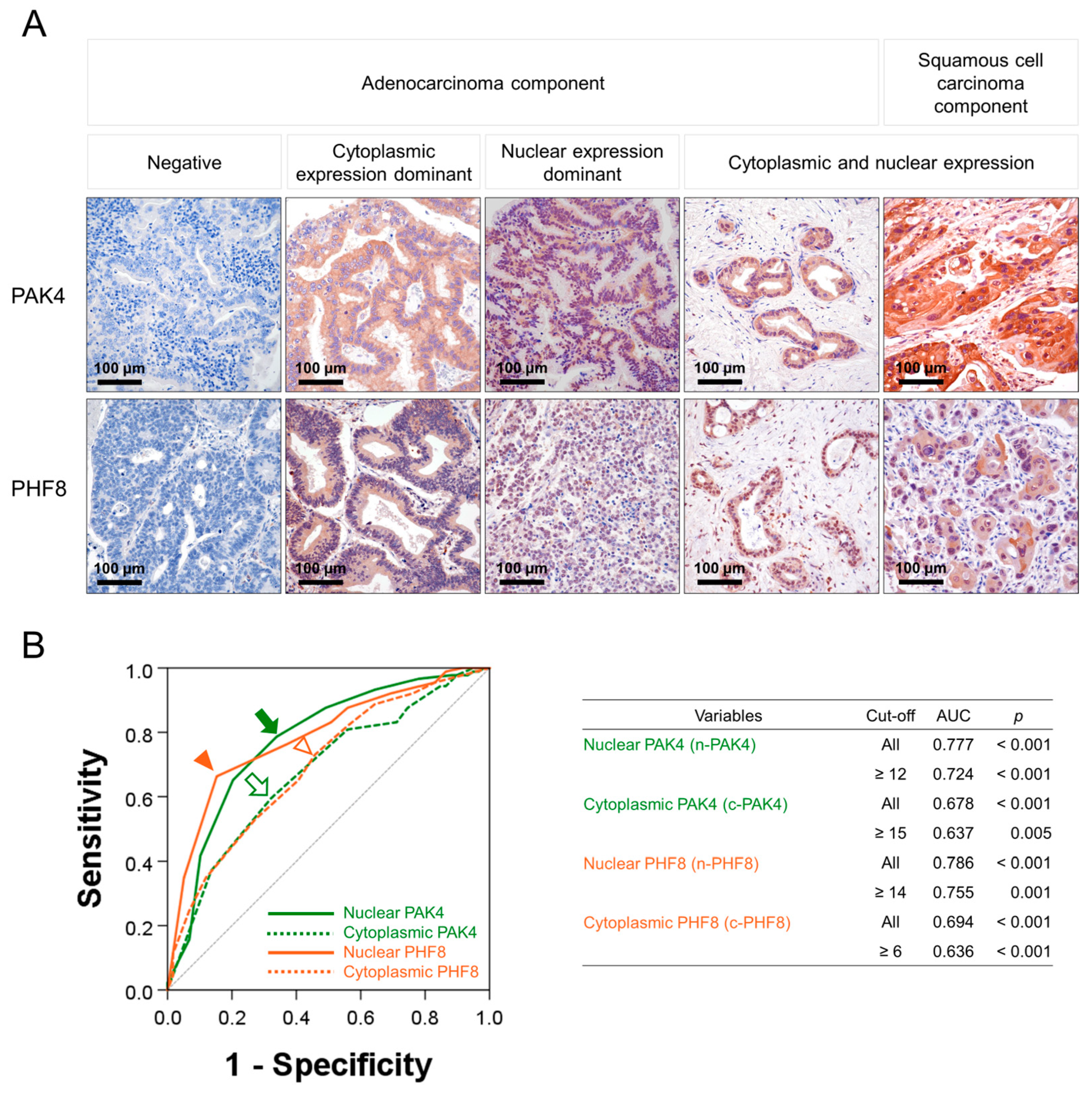
3.1. The Expression of PAK4 and PHF8 in GBCs
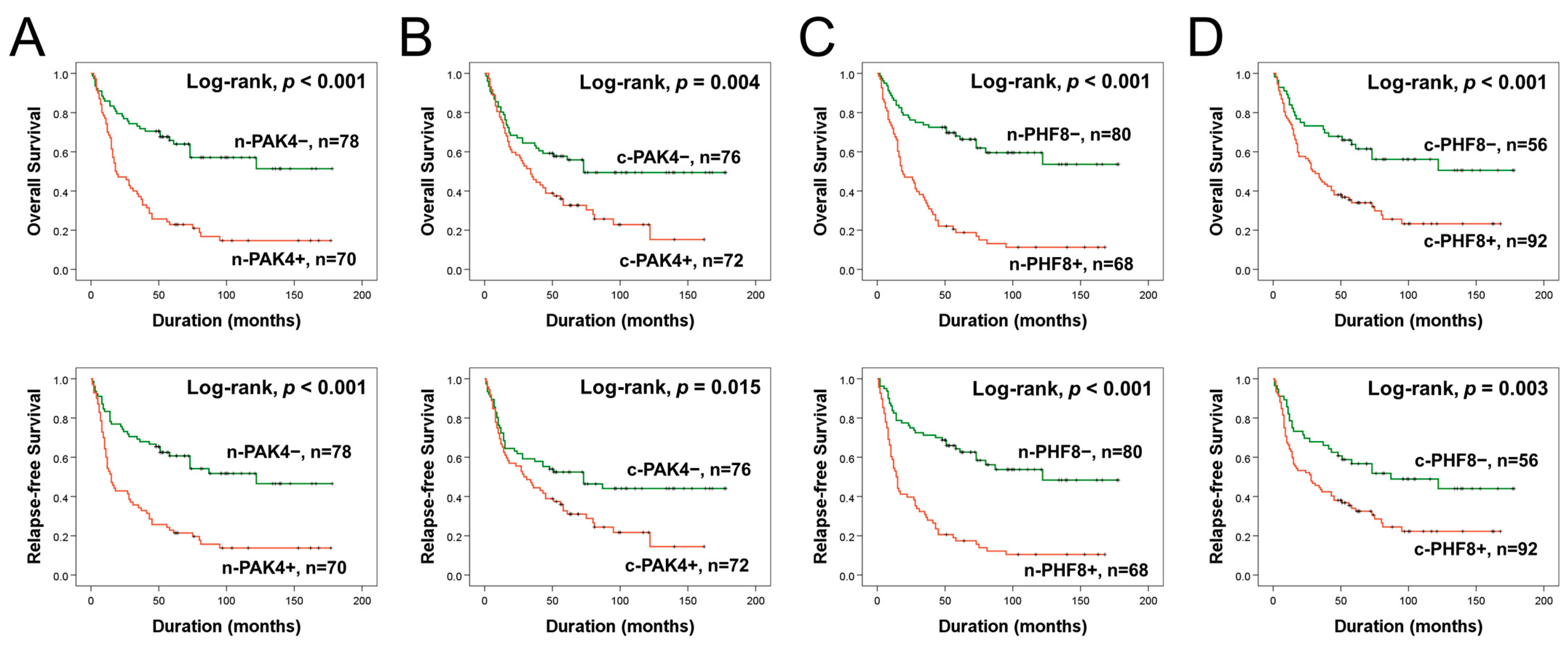
3.2. The Expression Patterns of PAK4 and PHF8 Are Associated with Shorter Survival of GBC Patients
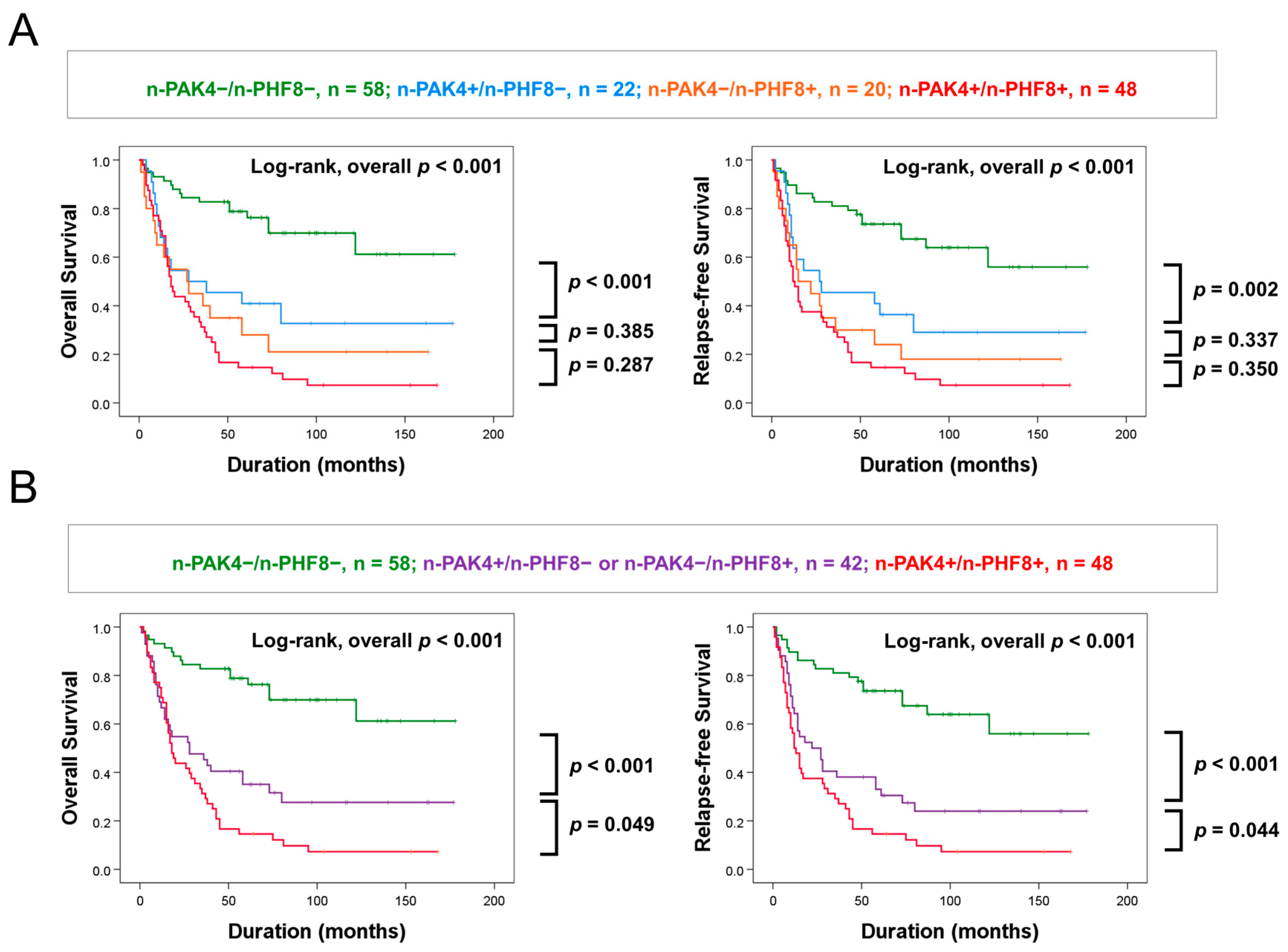
3.3. Coexpression Patterns of Nuclear PAK4 and Nuclear PHF8 Predict Survival of GBC Patients
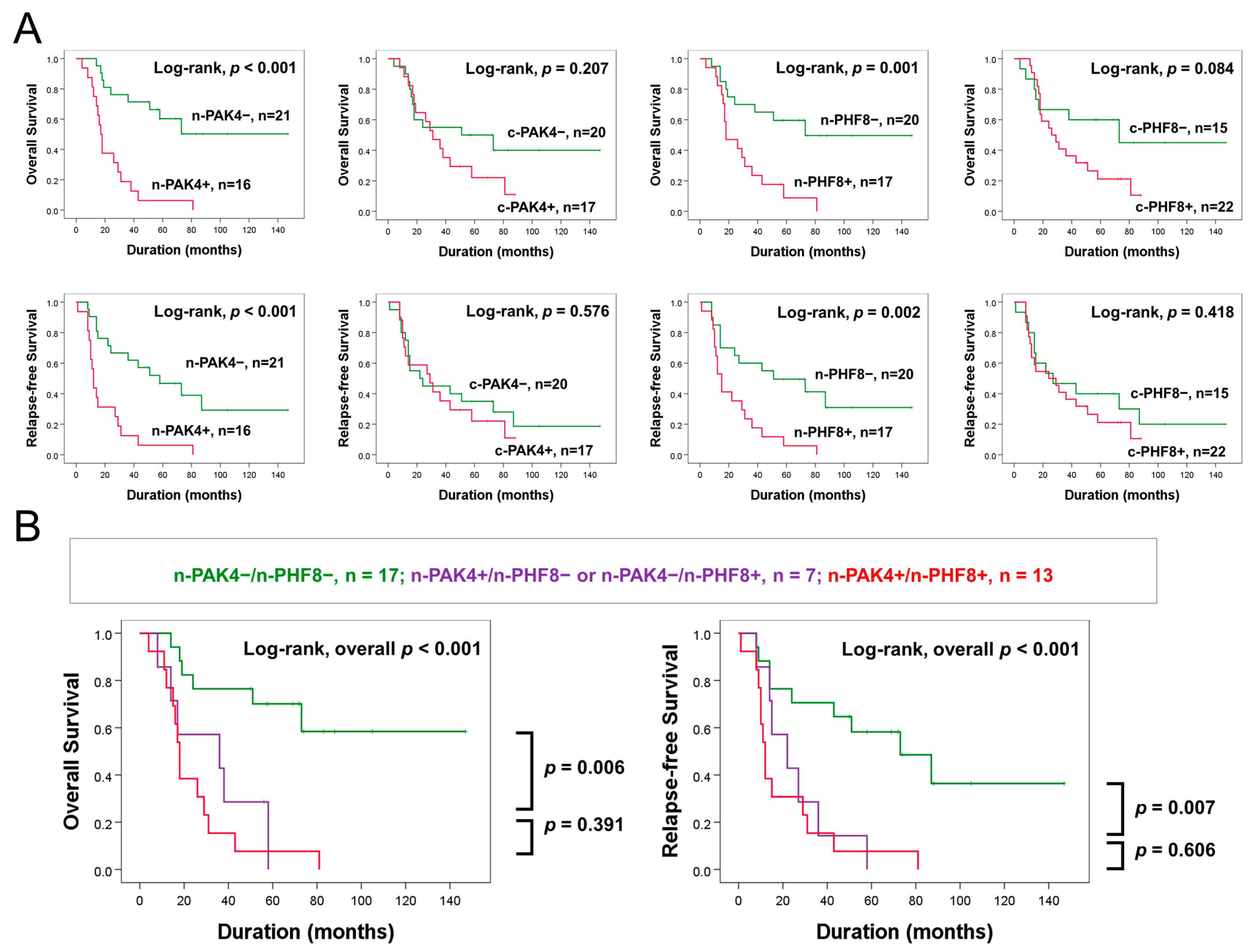
3.4. Expression Patterns of Nuclear PAK4 and Nuclear PHF8 Are Associated with Survival of GBC Patients Who Received Postoperative Therapies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Manser, E.; Leung, T.; Salihuddin, H.; Zhao, Z.S.; Lim, L. A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nature 1994, 367, 40–46. [Google Scholar] [CrossRef]
- Radu, M.; Semenova, G.; Kosoff, R.; Chernoff, J. PAK signalling during the development and progression of cancer. Nat. Rev. Cancer 2014, 14, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Mozibullah, M.; Junaid, M. Biological Role of the PAK4 Signaling Pathway: A Prospective Therapeutic Target for Multivarious Cancers. Arab. J. Chem. 2023, 16, 104438. [Google Scholar] [CrossRef]
- Yu, X.; Huang, C.; Liu, J.; Shi, X.; Li, X. The significance of PAK4 in signaling and clinicopathology: A review. Open Life Sci. 2022, 17, 586–598. [Google Scholar] [CrossRef]
- Costa, T.D.F.; Zhuang, T.; Lorent, J.; Turco, E.; Olofsson, H.; Masia-Balague, M.; Zhao, M.; Rabieifar, P.; Robertson, N.; Kuiper, R.; et al. PAK4 suppresses RELB to prevent senescence-like growth arrest in breast cancer. Nat. Commun. 2019, 10, 3589. [Google Scholar] [CrossRef]
- He, L.F.; Xu, H.W.; Chen, M.; Xian, Z.R.; Wen, X.F.; Chen, M.N.; Du, C.W.; Huang, W.H.; Wu, J.D.; Zhang, G.J. Activated-PAK4 predicts worse prognosis in breast cancer and promotes tumorigenesis through activation of PI3K/AKT signaling. Oncotarget 2017, 8, 17573–17585. [Google Scholar] [CrossRef]
- Kobayashi, K.; Inokuchi, M.; Takagi, Y.; Otsuki, S.; Fujimori, Y.; Sato, Y.; Yanaka, Y.; Higuchi, K.; Aburatani, T.; Tomii, C.; et al. Prognostic significance of PAK4 expression in gastric cancer. J. Clin. Pathol. 2016, 69, 580–585. [Google Scholar] [CrossRef]
- Siu, M.K.; Chan, H.Y.; Kong, D.S.; Wong, E.S.; Wong, O.G.; Ngan, H.Y.; Tam, K.F.; Zhang, H.; Li, Z.; Chan, Q.K.; et al. p21-activated kinase 4 regulates ovarian cancer cell proliferation, migration, and invasion and contributes to poor prognosis in patients. Proc. Natl. Acad. Sci. USA 2010, 107, 18622–18627. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lu, Y.; Feng, W.; Chen, Q.; Guo, H.; Sun, X.; Bao, Y. A two kinase-gene signature model using CDK2 and PAK4 expression predicts poor outcome in non-small cell lung cancers. Neoplasma 2016, 63, 322–329. [Google Scholar] [CrossRef]
- Li, Y.; Lu, Q.; Xie, C.; Yu, Y.; Zhang, A. Recent advances on development of p21-activated kinase 4 inhibitors as anti-tumor agents. Front. Pharmacol. 2022, 13, 956220. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, T.; Zhu, J.; Li, Z.; Lorent, J.; Zhao, C.; Dahlman-Wright, K.; Stromblad, S. p21-activated kinase group II small compound inhibitor GNE-2861 perturbs estrogen receptor alpha signaling and restores tamoxifen-sensitivity in breast cancer cells. Oncotarget 2015, 6, 43853–43868. [Google Scholar] [CrossRef]
- Moon, S.U.; Kim, J.W.; Sung, J.H.; Kang, M.H.; Kim, S.H.; Chang, H.; Lee, J.O.; Kim, Y.J.; Lee, K.W.; Kim, J.H.; et al. p21-Activated Kinase 4 (PAK4) as a Predictive Marker of Gemcitabine Sensitivity in Pancreatic Cancer Cell Lines. Cancer Res. Treat. 2015, 47, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Wang, H.; Wang, J.; Cheng, M. PB-10, a thiazolo[4,5-d] pyrimidine derivative, targets p21-activated kinase 4 in human colorectal cancer cells. Bioorg. Med. Chem. Lett. 2020, 30, 126807. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Tanasa, B.; Tyurina, O.V.; Zhou, T.Y.; Gassmann, R.; Liu, W.T.; Ohgi, K.A.; Benner, C.; Garcia-Bassets, I.; Aggarwal, A.K.; et al. PHF8 mediates histone H4 lysine 20 demethylation events involved in cell cycle progression. Nature 2010, 466, 508–512. [Google Scholar] [CrossRef]
- Li, S.; Sun, A.; Liang, X.; Ma, L.; Shen, L.; Li, T.; Zheng, L.; Shang, W.; Zhao, W.; Jia, J. Histone demethylase PHF8 promotes progression and metastasis of gastric cancer. Am. J. Cancer Res. 2017, 7, 448–461. [Google Scholar] [CrossRef]
- Lv, Y.; Shi, Y.; Han, Q.; Dai, G. Histone demethylase PHF8 accelerates the progression of colorectal cancer and can be regulated by miR-488 in vitro. Mol. Med. Rep. 2017, 16, 4437–4444. [Google Scholar] [CrossRef]
- Ye, H.; Yang, Q.; Qi, S.; Li, H. PHF8 Plays an Oncogene Function in Hepatocellular Carcinoma Formation. Oncol. Res. 2019, 27, 613–621. [Google Scholar] [CrossRef]
- Zhou, W.; Gong, L.; Wu, Q.; Xing, C.; Wei, B.; Chen, T.; Zhou, Y.; Yin, S.; Jiang, B.; Xie, H.; et al. PHF8 upregulation contributes to autophagic degradation of E-cadherin, epithelial-mesenchymal transition and metastasis in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2018, 37, 215. [Google Scholar] [CrossRef]
- Liu, Q.; Pang, J.; Wang, L.A.; Huang, Z.; Xu, J.; Yang, X.; Xie, Q.; Huang, Y.; Tang, T.; Tong, D.; et al. Histone demethylase PHF8 drives neuroendocrine prostate cancer progression by epigenetically upregulating FOXA2. J. Pathol. 2021, 253, 106–118. [Google Scholar] [CrossRef]
- Liu, Q.; Borcherding, N.C.; Shao, P.; Maina, P.K.; Zhang, W.; Qi, H.H. Contribution of synergism between PHF8 and HER2 signalling to breast cancer development and drug resistance. EBioMedicine 2020, 51, 102612. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Chen, Z.; Jia, G.; Lu, X.; Xie, X.; Jin, W. The histone demethylase PHF8 promotes prostate cancer cell growth by activating the oncomiR miR-125b. OncoTargets Ther. 2015, 8, 1979–1988. [Google Scholar] [CrossRef]
- Tseng, L.L.; Cheng, H.H.; Yeh, T.S.; Huang, S.C.; Syu, Y.Y.; Chuu, C.P.; Yuh, C.H.; Kung, H.J.; Wang, W.C. Targeting the histone demethylase PHF8-mediated PKCalpha-Src-PTEN axis in HER2-negative gastric cancer. Proc. Natl. Acad. Sci. USA 2020, 117, 24859–24866. [Google Scholar] [CrossRef]
- Hu, Y.; Mu, H.; Yang, Y. Histone demethylase PHF8 promotes cell growth and metastasis of non-small-cell lung cancer through activating Wnt/beta-catenin signaling pathway. Histol. Histopathol. 2021, 36, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Sharma, K.L.; Gupta, A.; Yadav, A.; Kumar, A. Gallbladder cancer epidemiology, pathogenesis and molecular genetics: Recent update. World J. Gastroenterol. 2017, 23, 3978–3998. [Google Scholar] [CrossRef]
- Song, X.; Hu, Y.; Li, Y.; Shao, R.; Liu, F.; Liu, Y. Overview of current targeted therapy in gallbladder cancer. Signal Transduct. Target. Ther. 2020, 5, 230. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, H.N.; Lee, K.T.; Lee, J.K.; Choi, S.H.; Paik, S.W.; Rhee, J.C.; Lowe, A.W. Gene expression profiles in gallbladder cancer: The close genetic similarity seen for early and advanced gallbladder cancers may explain the poor prognosis. Tumor Biol. 2008, 29, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Li, C.; Kang, B.; Gao, G.; Li, C.; Zhang, Z. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017, 45, W98–W102. [Google Scholar] [CrossRef]
- WHO Classification of Tumours Editorial Board. Digestive System Tumours, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2019. [Google Scholar]
- Amin, M.B.; American Joint Committee on Cancer; American Cancer Society. AJCC Cancer Staging Manual, 8th ed.; American Joint Committee on Cancer; Springer: Chicago, IL, USA, 2017. [Google Scholar]
- Ahn, S.W.; Lee, C.M.; Kang, M.A.; Hussein, U.K.; Park, H.S.; Ahn, A.R.; Yu, H.C.; Yang, J.D.; Yang, Y.H.; Park, K.; et al. IL4Ralpha and IL13Ralpha1 Are Involved in the Development of Human Gallbladder Cancer. J. Pers. Med. 2022, 12, 249. [Google Scholar] [CrossRef]
- Allred, D.; Harvey, J.M.; Berardo, M.; Clark, G.M. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod. Pathol. 1998, 11, 155–168. [Google Scholar] [PubMed]
- Hussein, U.K.; Ahmed, A.G.; Song, Y.; Kim, K.M.; Moon, Y.J.; Ahn, A.R.; Park, H.S.; Ahn, S.J.; Park, S.H.; Kim, J.R.; et al. CK2alpha/CSNK2A1 Induces Resistance to Doxorubicin through SIRT6-Mediated Activation of the DNA Damage Repair Pathway. Cells 2021, 10, 1770. [Google Scholar] [CrossRef]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Abril-Rodriguez, G.; Torrejon, D.Y.; Karin, D.; Campbell, K.M.; Medina, E.; Saco, J.D.; Galvez, M.; Champhekar, A.S.; Perez-Garcilazo, I.; Baselga-Carretero, I.; et al. Remodeling of the tumor microenvironment through PAK4 inhibition sensitizes tumors to immune checkpoint blockade. Cancer Res. Commun. 2022, 2, 1214–1228. [Google Scholar] [CrossRef] [PubMed]
- Abril-Rodriguez, G.; Torrejon, D.Y.; Liu, W.; Zaretsky, J.M.; Nowicki, T.S.; Tsoi, J.; Puig-Saus, C.; Baselga-Carretero, I.; Medina, E.; Quist, M.J.; et al. PAK4 inhibition improves PD-1 blockade immunotherapy. Nat. Cancer 2020, 1, 46–58. [Google Scholar] [CrossRef]
- Baskaran, Y.; Ng, Y.W.; Selamat, W.; Ling, F.T.; Manser, E. Group I and II mammalian PAKs have different modes of activation by Cdc42. EMBO Rep. 2012, 13, 653–659. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | No. | n-PAK4 | c-PAK4 | n-PHF8 | c-PHF8 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Positive | p | Positive | p | Positive | p | Positive | p | |||
| Age (years) | <65 y | 69 | 30 (43%) | 0.384 | 30 (43%) | 0.240 | 28 (41%) | 0.221 | 36 (52%) | 0.019 |
| ≥65 y | 79 | 40 (51%) | 42 (53%) | 40 (51%) | 56 (71%) | |||||
| Sex | Male | 73 | 32 (44%) | 0.405 | 37 (51%) | 0.625 | 28 (38%) | 0.068 | 45 (62%) | 0.898 |
| Female | 75 | 38 (51%) | 35 (47%) | 40 (53%) | 47 (63%) | |||||
| CEA * | ≤5.2 ng/mL | 119 | 57 (48%) | 0.853 | 58 (49%) | 0.910 | 54 (45%) | 0.431 | 74 (62%) | 0.678 |
| >5.2 ng/mL | 24 | 11 (46%) | 12 (50%) | 13 (54%) | 16 (67%) | |||||
| CA19-9 ** | ≤37 U/mL | 100 | 44 (44%) | 0.297 | 46 (46%) | 0.282 | 47 (47%) | 0.957 | 64 (64%) | 0.356 |
| >37 U/mL | 43 | 23 (53%) | 24 (56%) | 20 (47%) | 24 (56%) | |||||
| Tumor stage | I and II | 92 | 39 (42%) | 0.125 | 43 (47%) | 0.551 | 37 (40%) | 0.073 | 56 961%) | 0.678 |
| III and IV | 56 | 31 (55%) | 29 (52%) | 31 (55%) | 36 (64%) | |||||
| T category | T1 | 41 | 11 (27%) | 0.004 | 12 (29%) | 0.033 | 14 (34%) | 0.010 | 18 (44%) | 0.018 |
| T2 | 73 | 36 (49%) | 42 (58%) | 30 (41%) | 51 (70%) | |||||
| T3 | 30 | 21 (70%) | 16 (53%) | 21 (70%) | 19 (63%) | |||||
| T4 | 4 | 2 (50%) | 2 (50%) | 3 (75%) | 4 (100%) | |||||
| Lymph node metastasis | Absence | 111 | 52 (47%) | 0.849 | 53 (48%) | 0.704 | 51 (46%) | 1.000 | 70 (63%) | 0.695 |
| Presence | 37 | 18 (49%) | 19 (51%) | 17 (46%) | 22 (59%) | |||||
| Distant metastasis | Absence | 140 | 64 (46%) | 0.107 | 67 (48%) | 0.420 | 63 (45%) | 0.334 | 85 (61%) | 0.129 |
| Presence | 8 | 6 975%) | 5 (63%) | 5 (63%) | 7 (88%) | |||||
| Lymphovascular invasion | Absence | 127 | 60 (47%) | 0.975 | 60 (47%) | 0.401 | 62 (49%) | 0.085 | 79 (62%) | 0.979 |
| Presence | 21 | 10 (48%) | 12 (57%) | 6 (29%) | 13 (62%) | |||||
| Histologic type | Adenocarcinoma | 143 | 67 947%) | 0.566 | 68 (48%) | 0.327 | 65 (45%) | 0.544 | 87 (61%) | 0.207 |
| Adenosquamous carcinoma | 4 | 2 (50%) | 3 (75%) | 2 (50%) | 4 (100%) | |||||
| Squamous cell carcinoma | 1 | 1 (100%) | 1 (100%) | 1 9100%) | 1 (100%) | |||||
| Histologic grade | G1 | 63 | 21 (33%) | 0.003 | 23 (37%) | 0.011 | 20 (32%) | 0.003 | 35 (56%) | 0.154 |
| G2 and G3 | 85 | 49 (58%) | 49 (58%) | 48 (56%) | 57 (67%) | |||||
| c-PHF8 | Negative | 56 | 20 (36%) | 0.028 | 18 (32%) | 0.002 | 14 (25%) | <0.001 | ||
| Positive | 92 | 50 (54%) | 54 (59%) | 54 (59%) | ||||||
| n-PHF8 | Negative | 80 | 22 (28%) | <0.001 | 32 (40%) | 0.022 | ||||
| Positive | 68 | 48 (71%) | 40 (59%) | |||||||
| c-PAK4 | Negative | 76 | 25 (33%) | <0.001 | ||||||
| Positive | 72 | 45 (63%) |
| Characteristics | No. | OS | RFS | ||
|---|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | ||
| Age, y ≥ 65 (vs. <65) | 79/148 | 2.248 (1.444–3.500) | <0.001 | 2.017 (1.317–3.088) | 0.001 |
| Sex, female (vs. male) | 75/148 | 0.704 (0.463–1.070) | 0.100 | 0.708 (0.471–1.063) | 0.096 |
| CEA, >5.2 ng/mL (vs. ≤5.2 ng/mL) * | 24/142 | 1.382 (0.812–2.351) | 0.233 | 1.286 (0.758–2.180) | 0.351 |
| CA19-9, >37 U/mL (vs. ≤37 U/mL) ** | 43/143 | 1.818 (1.177–2.811) | 0.007 | 1.892 (1.239–2.889) | 0.003 |
| Tumor stage, III and IV (vs. I and II) | 56/148 | 3.555 (2.321–5.445) | <0.001 | 3.273 (2.160–4.958) | <0.001 |
| T category, T1 | 41/148 | 1 | <0.001 | 1 | <0.001 |
| T2 | 73/148 | 2.279 (1.241–4.183) | 0.008 | 2.416 (1.345–4.339) | 0.003 |
| T3 | 30/148 | 10.123 (5.231–19.588) | <0.001 | 8.504 (4.458–16.223) | <0.001 |
| T4 | 4/148 | 9.876 (3.157–30.899) | <0.001 | 9.694 (3.118–30.138) | <0.001 |
| Lymph node metastasis, presence (vs. absence) | 37/148 | 1.982 (1.266–3.104) | 0.003 | 1.997 (1.288–3.098) | 0.002 |
| Distant metastasis, presence (vs. absence) | 8/148 | 6.190 (2.849–13.451) | <0.001 | 4.781 (2.224–10.278) | <0.001 |
| Lymphovascular invasion, presence (vs. absence) | 21/148 | 2.333 (1.364–3.993) | 0.002 | 2.307 (1.354–3.930) | 0.002 |
| Histologic type, adenocarcinoma | 143/148 | 1 | 0.002 | 1 | 0.002 |
| adenosquamous carcinoma | 4/148 | 3.772 (1.366–10.419) | 0.010 | 4.013 (1.451–11.099) | 0.007 |
| squamous cell carcinoma | 1/148 | 1.804 (1.779–107.101) | 0.012 | 12.753 (1.654–98.308) | 0.015 |
| Histologic grade, G2 and G3 (vs. G1) | 85/148 | 3.066 (1.923–4.890) | <0.001 | 3.043 (1.938–4.778) | <0.001 |
| n-PAK4, positive (vs. negative) | 70/148 | 3.063 (1.972–4.759) | <0.001 | 2.798 (1.835–4.268) | <0.001 |
| c-PAK4, positive (vs. negative) | 72/148 | 1.828 (1.194–2.797) | 0.005 | 1.651 (1.095–2.489) | 0.017 |
| n-PHF8, positive (vs. negative) | 68/148 | 3.713 (2.378–5.799) | <0.001 | 3.471 (2.265–5.319) | <0.001 |
| c-PHF8, positive (vs. negative) | 92/148 | 2.240 (1.397–3.252) | <0.001 | 1.941 (1.243–3.030) | 0.004 |
| Characteristics | OS | RFS | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| Age, y ≥65 (vs. <65) | 2.730 (1.710–4.358) | <0.001 | 2.275 (1.455–3.557) | <0.001 |
| Tumor stage, III and IV (vs. I and II) | 2.543 (1.337–4.836) | 0.004 | 2.008 (1.050–3.839) | 0.035 |
| T category, T1 | 1 | 0.031 | 1 | 0.030 |
| T2 | 1.744 (0.911–3.339) | 0.093 | 1.831 (0.979–3.426) | 0.058 |
| T3 | 3.516 (1.439–8.592) | 0.006 | 3.278 (1.377–7.802) | 0.007 |
| T4 | 5.077 (1.401–18.405) | 0.013 | 5.430 (1.522–19.839) | 0.009 |
| n-PAK4, positive (vs. negative) | 2.003 (1.161–3.458) | 0.013 | 1.794 (1.082–2.975) | 0.024 |
| n-PHF8, positive (vs. negative) | 2.130 (1.229–3.695) | 0.007 | 2.411 (1.408–4.127) | 0.001 |
| Coexpression Pattern of n-PAK4 and n-PHF8 | No. | 5-Year OS (%) | 10-Year OS (%) | 5-Year RFS (%) | 10-Year RFS (%) |
|---|---|---|---|---|---|
| Coexpression Model 1 | |||||
| n-PAK4/n-PHF8, −/− | 58 | 79 | 70 | 74 | 64 |
| n-PAK4/n-PHF8, +/− | 22 | 41 | 33 | 41 | 29 |
| n-PAK4/n-PHF8, −/+ | 20 | 28 | 21 | 24 | 18 |
| n-PAK4/n-PHF8, +/+ | 48 | 15 | 7 | 15 | 7 |
| Coexpression Model 2 | |||||
| n-PAK4/n-PHF8, −/− | 58 | 79 | 70 | 74 | 64 |
| n-PAK4/n-PHF8, −/+ or +/− | 42 | 35 | 28 | 33 | 24 |
| n-PAK4/n-PHF8, +/+ | 48 | 15 | 7 | 15 | 7 |
| Characteristics | OS | RFS | ||
|---|---|---|---|---|
| HR (95% CI) | p | HR (95% CI) | p | |
| Univariate analysis | ||||
| n-PAK4/n-PHF8, −/− | 1 | <0.001 | 1 | <0.001 |
| −/+ or +/− | 3.727 (2.021–6.873) | <0.001 | 3.277 (1.847–5.814) | <0.001 |
| +/+ | 6.019 (3.369–10.751) | <0.001 | 5.309 (3.077–9.159) | <0.001 |
| Multivariate analysis * | ||||
| Age, y ≥65 (vs. <65) | 3.139 (1.944–5.068) | <0.001 | 2.663 (1.699–4.173) | <0.001 |
| CA19-9, >37 U/mL (vs. ≤37 U/mL) | 1.569 (1.005–2.451) | 0.048 | ||
| Tumor stage, III and IV (vs. I and II) | 2.812 (1.466–5.397) | 0.002 | 3.373 (2.118–5.373) | <0.001 |
| T category, T1 | 1 | 0.030 | ||
| T2 | 1.569 (0.820–3.000) | 0.173 | ||
| T3 | 3.283 (1.348–7.996) | 0.009 | ||
| T4 | 5.368 (1.478–19.487) | 0.011 | ||
| Histologic grade, G2 and G3 (vs. G1) | 2.164 (1.338–3.500) | 0.002 | ||
| n-PAK4/n-PHF8, −/− | 1 | <0.001 | 1 | <0.001 |
| −/+ or +/− | 4.975 (2.611–9.479) | <0.001 | 4.225 (2.323–7.687) | <0.001 |
| +/+ | 5.173 (2.769–9.665) | <0.001 | 5.127 (2.872–9.152) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, A.R.; Karamikheirabad, M.; Park, M.S.; Zhang, J.; Kim, H.S.; Jeong, J.S.; Kim, K.M.; Park, H.S.; Jang, K.Y. Expression Patterns of PAK4 and PHF8 Are Associated with the Survival of Gallbladder Carcinoma Patients. Diagnostics 2023, 13, 1149. https://doi.org/10.3390/diagnostics13061149
Ahn AR, Karamikheirabad M, Park MS, Zhang J, Kim HS, Jeong JS, Kim KM, Park HS, Jang KY. Expression Patterns of PAK4 and PHF8 Are Associated with the Survival of Gallbladder Carcinoma Patients. Diagnostics. 2023; 13(6):1149. https://doi.org/10.3390/diagnostics13061149
Chicago/Turabian StyleAhn, Ae Ri, Maryam Karamikheirabad, Min Su Park, Junyue Zhang, Hyun Sun Kim, Ji Su Jeong, Kyoung Min Kim, Ho Sung Park, and Kyu Yun Jang. 2023. "Expression Patterns of PAK4 and PHF8 Are Associated with the Survival of Gallbladder Carcinoma Patients" Diagnostics 13, no. 6: 1149. https://doi.org/10.3390/diagnostics13061149
APA StyleAhn, A. R., Karamikheirabad, M., Park, M. S., Zhang, J., Kim, H. S., Jeong, J. S., Kim, K. M., Park, H. S., & Jang, K. Y. (2023). Expression Patterns of PAK4 and PHF8 Are Associated with the Survival of Gallbladder Carcinoma Patients. Diagnostics, 13(6), 1149. https://doi.org/10.3390/diagnostics13061149









