Multitracer PET/CT with [18F]Fluorodeoxiglucose and [18F]Fluorocholine in the Initial Staging of Multiple Myeloma Patients Applying the IMPeTus Criteria: A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. [18F]FDG PET/CT and [18F]FCH PET/CT Procedures
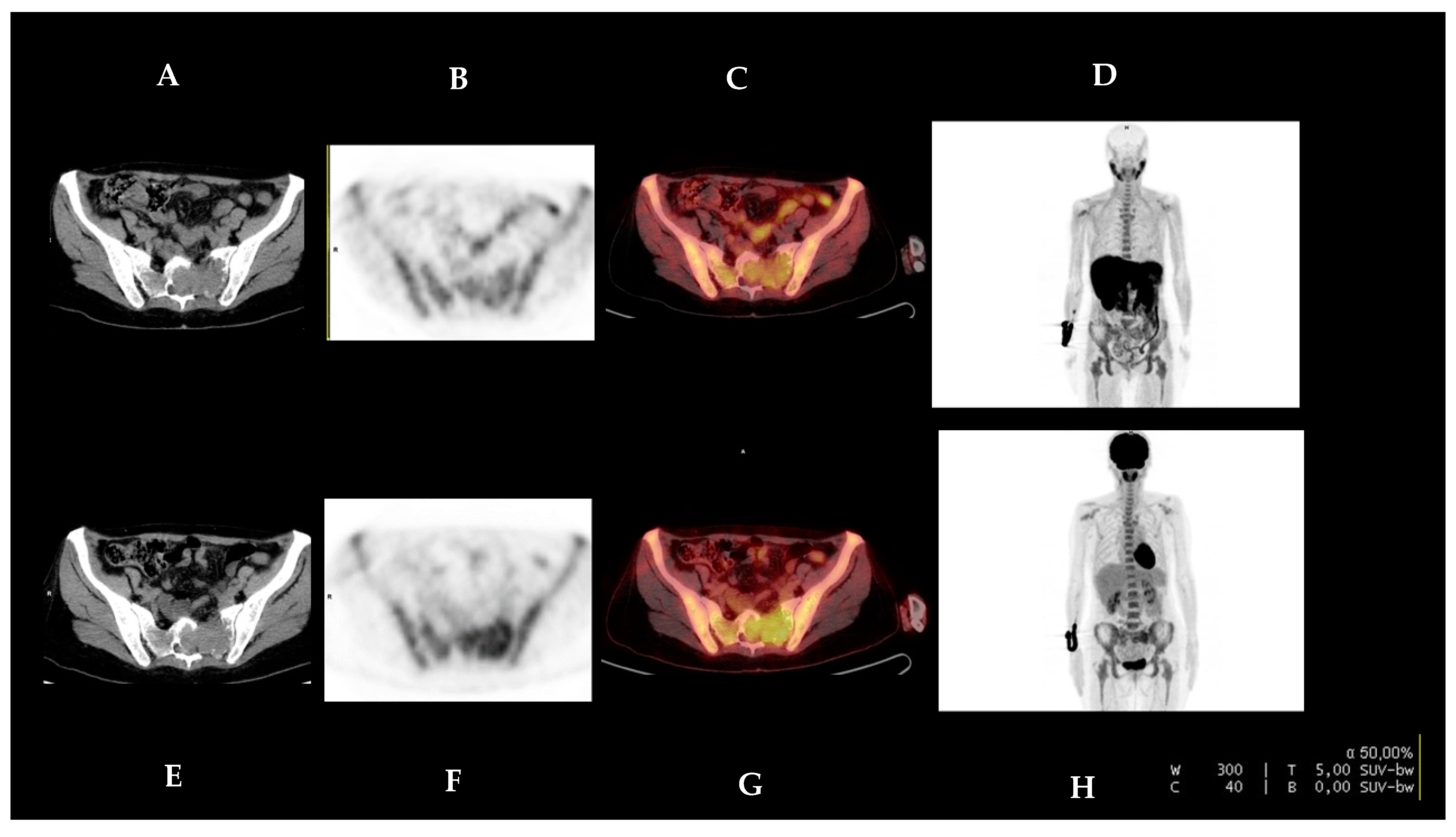
2.3. [18F]FDG PET/CT and [18F]FCH PET/CT Image Analysis
2.4. Reference Standard
2.5. Statistical Analysis
3. Results
3.1. Patient-Based Analysis
3.2. Lesion-Based Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef] [PubMed]
- Hemrom, A.; Tupalli, A.; Alavi, A.; Kumar, R. 18F-FDG versus Non-FDG PET tracers in multiple myeloma. PET Clin. 2022, 17, 415–430. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef]
- Myeloma—CancerStatFacts. SEER. Available online: https://seer.cancer.gov/statfacts/html/mulmy.html (accessed on 5 January 2023).
- Grupo Español de Mieloma. Guía de Mieloma Múltiple; Luzan 5: Madrid, Spain, 2021. [Google Scholar]
- Morales-Lozano, M.I.; Viering, O.; Samnick, S.; Rodríguez-Otero, P.; Buck, A.K.; Marcos-Jubilar, M.; Rasche, L.; Prieto, E.; Kortüm, K.M.; San-Miguel, J.; et al. 18F-FDG and 11C-Methionine PET/CT in newly diagnosed multiple myeloma patients: Comparison of volume-based PET biomarkers. Cancers 2020, 12, 1042. [Google Scholar] [CrossRef] [PubMed]
- Cavo, M.; Terpos, E.; Nanni, C.; Moreau, P.; Lentzsch, S.; Zweegman, S.; Hillengass, J.; Engelhardt, M.; Usmani, S.Z.; Vesole, D.H.; et al. Role of 18F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: A consensus statement by the International Myeloma Working Group. Lancet Oncol. 2017, 18, e206–e217. [Google Scholar] [CrossRef]
- Hillengass, J.; Usmani, S.; Rajkumar, S.V.; Durie, B.G.M.; Mateos, M.V.; Lonial, S.; Joao, C.; Anderson, K.C.; García-Sanz, R.; Riva, E.; et al. International myeloma working group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019, 20, e302–e312. [Google Scholar] [CrossRef]
- Gómez León, N.; Aguado Bueno, B.; Herreros Pérez, M.; León Ramírez, L.F.; Alegre, A.; Colletti, P.M.; Rubello, D.; Carreras, J.L.; Delgado Bolton, R.C. Agreement between 18F-FDG PET/CT and whole-body magnetic resonance compared with skeletal survey for initial staging and response at end-of-treatment evaluation of patients with multiple myeloma. Clin. Nucl. Med. 2021, 46, 310–322. [Google Scholar] [CrossRef]
- Nanni, C.; Versari, A.; Chauvie, S.; Bertone, E.; Bianchi, A.; Rensi, M.; Bellò, M.; Gallamini, A.; Patriarca, F.; Gay, F.; et al. Interpretation criteria for FDG PET/CT in multiple myeloma (IMPeTUs): Final results. IMPeTUs (Italian myeloma criteria for PET USe). Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 712–719. [Google Scholar] [CrossRef]
- Rasche, L.; Angtuaco, E.; McDonald, J.E.; Buros, A.; Stein, C.; Pawlyn, C.; Thanendrarajan, S.; Schinke, C.; Samant, R.; Yaccoby, S.; et al. Low expression of hexokinase-2 is associated with false-negative FDG-positron emission tomography in multiple myeloma. Blood 2017, 130, 30–34. [Google Scholar] [CrossRef]
- Ambrosini, V.; Farsad, M.; Nanni, C.; Schiavina, R.; Rubello, D.; Castellucci, P.; Pasquini, E.; Franchi, R.; Cavo, M.; Fanti, S. Incidental finding of an (11)C-choline PET-positive solitary plasmacytoma lesion. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 1522. [Google Scholar] [CrossRef]
- Mesguich, C.; Hulin, C.; Lascaux, A.; Bordenave, L.; Marit, G.; Hindié, E. Choline PET/CT in multiple myeloma. Cancers 2020, 12, 1394. [Google Scholar] [CrossRef] [PubMed]
- Sachpekidis, C.; Merz, M.; Raab, M.S.; Bertsch, U.; Weru, V.; Kopp-Schneider, A.; Jauch, A.; Goldschmidt, H.; Dimitrakopoulou-Strauss, A. The prognostic significance of [18F]FDG PET/CT in multiple myeloma according to novel interpretation criteria (IMPeTUs). EJNMMI Res. 2021, 11, 100. [Google Scholar] [CrossRef] [PubMed]
- Boellaard, R.; Delgado-Bolton, R.; Oyen, W.J.; Giammarile, F.; Tatsch, K.; Eschner, W.; Verzijlbergen, F.J.; Barrington, S.F.; Pike, L.C.; Weber, W.A.; et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 328–354. [Google Scholar] [CrossRef] [PubMed]
- Chondrogiannis, S.; Marzola, M.C.; Grassetto, G.; Maffione, A.M.; Rampin, L.; Veronese, E.; Massaro, A.; Rubello, D. New acquisition protocol of 18F-Choline PET/CT in prostate cancer patients: Review of the literature about methodology and proposal of standardization. Biomed. Res. Int. 2014, 2014, 215650. [Google Scholar] [CrossRef] [PubMed]
- Schillaci, O.; Calabria, F.; Tavolozza, M.; Cicciò, C.; Carlani, M.; Caracciolo, C.R.; Danieli, R.; Orlacchio, A.; Simonetti, G. 18F-Choline PET/CT physiological distribution and pitfalls in image interpretation: Experience in 80 patients with prostate cancer. Nucl. Med. Commun. 2010, 31, 39–45. [Google Scholar] [CrossRef]
- de Groot, E.H.; Post, N.; Boellaard, R.; Wagenaar, N.R.; Willemsen, A.T.; van Dalen, J.A. Optimized dose regimen for whole-body FDG-PET imaging. EJNMMI Res. 2013, 3, 63. [Google Scholar] [CrossRef]
- Meignan, M.; Gallamini, A.; Haioun, C. Report on the first international workshop on interim-PET-scan in Lymphoma. Leuk. Lymphoma 2009, 50, 1257–1260. [Google Scholar] [CrossRef]
- Lütje, S.; de Rooy, J.W.; Croockewit, S.; Koedam, E.; Oyen, W.J.; Raymakers, R.A. Role of radiography, MRI and FDG-PET/CT in diagnosing, staging and therapeutical evaluation of patients with multiple myeloma. Ann. Hematol. 2009, 88, 1161–1168. [Google Scholar] [CrossRef]
- Lu, Y.Y.; Chen, J.H.; Lin, W.Y.; Liang, J.A.; Wang, H.Y.; Tsai, S.C.; Kao, C.H. FDG PET or PET/CT for detecting intramedullary and extramedullary lesions in multiple Myeloma: A systematic review and meta-analysis. Clin. Nucl. Med. 2012, 37, 833–837. [Google Scholar] [CrossRef]
- Lapa, C.; Kircher, M.; Da Via, M.; Schreder, M.; Rasche, L.; Kortüm, K.M.; Einsele, H.; Buck, A.K.; Hänscheid, H.; Samnick, S. Comparison of 11C-Choline and 11C-Methionine PET/CT in multiple myeloma. Clin. Nucl. Med. 2019, 44, 620–624. [Google Scholar] [CrossRef]
- Cassou-Mounat, T.; Balogova, S.; Nataf, V.; Calzada, M.; Huchet, V.; Kerrou, K.; Devaux, J.Y.; Mohty, M.; Talbot, J.N.; Garderet, L. 18F-fluorocholine versus 18F-fluorodeoxyglucose for PET/CT imaging in patients with suspected relapsing or progressive multiple myeloma: A pilot study. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1995–2004. [Google Scholar] [CrossRef]
- Meckova, Z.; Lambert, L.; Spicka, I.; Kubinyi, J.; Burgetova, A. Is fluorine-18-fluorocholine PET/CT suitable for the detection of skeletal involvement of multiple myeloma? Hell. J. Nucl. Med. 2018, 21, 167–168. [Google Scholar] [CrossRef]
- Kircher, S.; Stolzenburg, A.; Kortüm, K.M.; Kircher, M.; Da Via, M.; Samnick, S.; Buck, A.K.; Einsele, H.; Rosenwald, A.; Lapa, C. Hexokinase-2 expression in 11C-Methionine-positive, 18F-FDG-negative multiple myeloma. J. Nucl. Med. 2019, 60, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Nanni, C.; Zamagni, E.; Cavo, M.; Rubello, D.; Tacchetti, P.; Pettinato, C.; Farsad, M.; Castellucci, P.; Ambrosini, V.; Montini, G.C.; et al. 11C-choline vs. 18F-FDG PET/CT in assessing bone involvement in patients with multiple myeloma. World J. Surg. Oncol. 2007, 5, 68. [Google Scholar] [CrossRef] [PubMed]
- Talbot, J.N.; Fartoux, L.; Balogova, S.; Nataf, V.; Kerrou, K.; Gutman, F.; Huchet, V.; Ancel, D.; Grange, J.D.; Rosmorduc, O. Detection of hepatocellular carcinoma with PET/CT: A prospective comparison of 18F-fluorocholine and 18F-FDG in patients with cirrhosis or chronic liver disease. J. Nucl. Med. 2010, 51, 1699–1706. [Google Scholar] [CrossRef] [PubMed]
- Usmani, S.Z.; Heuck, C.; Mitchell, A.; Szymonifka, J.; Nair, B.; Hoering, A.; Alsayed, Y.; Waheed, S.; Haider, S.; Restrepo, A.; et al. Extramedullary disease portends poor prognosis in multiple myeloma and is over-represented in high-risk disease even in the era of novel agents. Haematologica 2012, 97, 1761–1767. [Google Scholar] [CrossRef]
- Mesguich, C.; Hulin, C.; Latrabe, V.; Asselineau, J.; Bordenave, L.; Perez, P.; Hindie, E.; Marit, G. Prospective comparison of 18F-Choline Positron Emission Tomography/Computed Tomography (PET/CT) and 18F-Fluorodeoxyglucose (FDG) PET/CT in the Initial Workup of multiple myeloma: Study protocol of a prospective imaging trial. JMIR Res. Protoc. 2020, 9, e17850. [Google Scholar] [CrossRef]
- Calabria, F.; Chiaravalloti, A.; Cicciò, C.; Gangemi, V.; Gullà, D.; Rocca, F.; Gallo, G.; Cascini, G.L.; Schillaci, O. PET/CT with 18F-choline: Physiological whole bio-distribution in male and female subjects and diagnostic pitfalls on 1000 prostate cancer patients: 18F-choline PET/CT bio-distribution and pitfalls. A southern Italian experience. Nucl. Med. Biol. 2017, 51, 40–54. [Google Scholar] [CrossRef]
- Matteucci, F.; Paganelli, G.; Martinelli, G.; Cerchione, C. PET/CT in multiple myeloma: Beyond FDG. Front. Oncol. 2021, 10, 622501. [Google Scholar] [CrossRef]
- Delgado Bolton, R.C.; Calapaquí Terán, A.K.; Fanti, S.; Giammarile, F. New biomarkers with prognostic impact based on multitracer PET/CT imaging in neuroendocrine neoplasms: The light leading out of the darkness in challenging tumors. Clin. Nucl. Med. 2022, 47, 219–220. [Google Scholar] [CrossRef]
- Sachpekidis, C.; Goldschmidt, H.; Dimitrakopoulou-Strauss, A. Positron Emission Tomography (PET) radiopharmaceuticals in multiple myeloma. Molecules 2019, 25, 134. [Google Scholar] [CrossRef]
- Nakamoto, Y.; Kurihara, K.; Nishizawa, M.; Yamashita, K.; Nakatani, K.; Kondo, T.; Takaori-Kondo, A.; Togashi, K. Clinical value of (1)(1)C-Methionine PET/CT in patients with plasma cell malignancy: Comparison with (1)(8)F-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 708–715. [Google Scholar] [CrossRef]
- Ho, C.L.; Chen, S.; Leung, Y.L.; Cheng, T.; Wong, K.N.; Cheung, S.K.; Liang, R.; Chim, C.S. 11C-Acetate PET/CT for metabolic characterization of multiple myeloma: A comparative study with 18F-FDG PET/CT. J. Nucl. Med. 2014, 55, 749–752. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, T.S.; Lee, J.W.; Kwon, H.W.; Kim, Y.I.; Kang, S.H.; Kim, S.K. Incidental finding of an 11C-Acetate PET-positive multiple myeloma. Ann. Nucl. Med. 2010, 24, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Brausi, M.; Hoskin, P.; Andritsch, E.; Banks, I.; Beishon, M.; Boyle, H.; Colecchia, M.; Delgado-Bolton, R.; Höckel, M.; Leonard, K.; et al. ECCO Essential Requirements for Quality Cancer Care: Prostate cancer. Crit. Rev. Oncol. Hematol. 2020, 148, 102861. [Google Scholar] [CrossRef] [PubMed]
- Kratochwil, C.; Fendler, W.P.; Eiber, M.; Baum, R.; Bozkurt, M.F.; Czernin, J.; Delgado Bolton, R.C.; Ezziddin, S.; Forrer, F.; Hicks, R.J.; et al. EANM procedure guidelines for radionuclide therapy with 177Lu-labelled PSMA-ligands (177Lu-PSMA-RLT). Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2536–2544. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.; Sobol, N.; O’Donoghue, J.; Kirov, A.S.; Riedl, C.C.; Min, R.; Smith, E.; Carter, L.M.; Lyashchenko, S.K.; Lewis, J.S.; et al. CD38-targeted immuno-pet of multiple myeloma: From xenograft models to first-in-human imaging. Radiology 2020, 295, 606–615. [Google Scholar] [CrossRef]
- Herrmann, K.; Schottelius, M.; Lapa, C.; Osl, T.; Poschenrieder, A.; Hänscheid, H.; Lückerath, K.; Schreder, M.; Bluemel, C.; Knott, M.; et al. First-in-Human experience of CXCR4-Directed endoradiotherapy with 177Lu- and 90Y-labeled pentixather in advanced-stage multiple myeloma with extensive intra- and extramedullary disease. J. Nucl. Med. 2016, 57, 248–251. [Google Scholar] [CrossRef]
- Delgado Bolton, R.C.; Calapaquí Terán, A.K.; Fanti, S.; Giammarile, F. The concept of strength through synergy applied to the search of powerful prognostic biomarkers in gastroesophageal cancer: An example based on combining clinicopathological parameters, imaging-derived sarcopenia measurements, and radiomic features. Clin. Nucl. Med. 2023, 48, 156–157. [Google Scholar] [CrossRef]
- Delgado Bolton, R.C.; Calapaquí Terán, A.K.; Herrmann, K.; Fanti, S.; Giammarile, F. Are we approaching a change in paradigm in PET/CT imaging of solid gastrointestinal (or digestive) tract tumors with the clinical application of FAPI imaging? Clin. Nucl. Med. 2023, 48, 318–319. [Google Scholar] [CrossRef]


| Patient Characteristics | |||||||
|---|---|---|---|---|---|---|---|
| Patient Number | Age | Sex | Ig | Light Chain | Bence-Jones | % of Plasma Cells | CRAB |
| 1 | 43 | M | IgG | Lambda | No | 13 | - |
| 2 | 68 | F | IgG | Kappa | No | 72.4 | Pathological fracture |
| 3 | 67 | F | IgG | Kappa | Yes | 17 | Anaemia |
| 4 | 73 | M | IgA | Lambda | No | 30 | - |
| 5 | 50 | F | IgA | Lambda | No | 44.6 | - |
| 6 | 59 | M | IgA | Lambda | Yes | 15 | - |
| 7 | 59 | F | IgA | Kappa | No | 55 | Pathological fracture |
| 8 | 68 | F | IgG | Kappa | No | 24.2 | - |
| 9 | 59 | F | IgM | Kappa | Yes | 25.6 | Pathological fracture |
| 10 | 50 | M | IgG | Kappa | Yes | 97 | Anaemia. Renal failure. Lytic lesions. |
| Characteristics of the MM Lesions and Lesion-by-Lesion Comparison between [18F]FDG PET/CT and [18F]FCH PET/CT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient Number | Bone Marrow Affected | Focal Lesions (Number) | Extra-Medullary/Para-Medullary Disease | Lytic Lesions (Number) | SUVmax in Target Lesion (Location) | Deauville Score | ||||||
| [18F]FDG | [18F]FCH | [18F]FDG | [18F]FCH | [18F]FDG | [18F]FCH | [18F]FDG | [18F]FCH | [18F]FDG | [18F]FCH | [18F]FDG | [18F]FCH | |
| 1 | No | Yes | No | Yes (1) | No | No | No | No | - | 4.3 (left iliac) | 2 | 3 |
| 2 | No | Yes | Yes (2) * | Yes (2) | Vagina and liver foci | No | No | No | - | 4.6 (D7) | 4 | 3 |
| 3 | Yes | Yes | Yes (1) † | Yes (1) † | - | - | Yes (1) | Yes (1) | 4.5 | 4.1 | 4 | 3 |
| 4 | No | Yes | No | No | - | - | Yes (1) | Yes (1) | 3.3 (lytic lesion) | 2.1 (lytic lesion) | 2 | 2 |
| 5 | No | Yes (patchy) | No | No | No | No | No | No | - | - | 3 | 3 |
| 6 | Yes | Yes | No | Yes (3) | Parotid adenopathy | Yes (1) | Yes (1) | Yes (1) | - | 7 (D5) | 3 | 3 |
| 7 | Yes | Yes | Yes (1) # | Yes (7) | Yes (1 PMD) | Yes (1 PMD) | Yes (2) § | Yes (5) § | 6.9 (left sacrum) | 8.9 (left sacrum) | 4 | 3 |
| 8 | No | Yes | No | No | - | Preauricular adenopathy | No | No | - | 2.7 (adenopathy) | 3 | 3 |
| 9 | Yes | Yes | Yes (4) | Yes (7) | Yes (1, rib PMD) | Yes (1, rib PMD) | No | No | 4.6 (D9) | 6.3 (D9) | 4 | 3 |
| 10 | Yes | yes | Yes (>10) | Yes (>10) | No | No | Yes (>10) | Yes (>10) | 5 | 3 | ||
| Description of SUVmax of the MM Lesions and Lesion-by-Lesion Comparison between [18F]FDG PET/CT and [18F]FCH PET/CT | |||||||
|---|---|---|---|---|---|---|---|
| Patient Number | Axial or Extra-Axial Lesions | [18F]FCH | [18F]FDG | ||||
| Location | SUVmax | Deauville Score | Location | SUVmax | Deauville Score | ||
| 1 | Axial | Iliac bone | 4.3 | 3 | No uptake | ||
| 2 | Axial | Spinous L5 | 2.9 | 3 | Spinous L5 | 2.6 | 4 |
| 10th right rib | 5.3 | 3 | 10th right rib | 3.0 | 4 | ||
| D6 | 3.6 | 3 | D6 | 1.6 | 1 | ||
| D7 | 4.6 | 3 | D7 | 1.7 | 1 | ||
| 3 | Axial | Left sacrum | 4.1 | 3 | Left sacrum | 4.5 | 4 |
| 4 | Extra-axial | Internal femoral condyle | 2.1 | 2 | Internal femoral condyle | 3.3 | 2 |
| 5 | Axial | No uptake | No uptake | ||||
| 6 | Axial | D5 | 7.0 | 3 | No uptake | ||
| Left sacrum | 5.6 | No uptake | |||||
| Iliac bone | 6.1 | 3 | No uptake | ||||
| 7 | Axial | Sacrum para-medullary lesion | 8.9 | 3 | Sacrum para-medullary lesion | 6.9 | 4 |
| D6 | 8.0 | 3 | No uptake | ||||
| Calvarium | 1.6 | 3 | No uptake | ||||
| 8 | Axial | No uptake | No uptake | ||||
| 9 | Axial | D9 | 6.3 | 3 | D9 | 4.6 | 4 |
| 3rd left rib | 4.0 | 3 | 3rd left rib | 3.5 | 3 | ||
| 10 | Axial and extra-axial | Sternum | 5.9 | 3 | Sternum | 8.3 | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrastachu Zumarán, P.; García Megías, I.; Mangas Losada, M.; Mendoza Melero, A.; Villanueva Torres, A.; Boulvard Chollet, X.; Romero Robles, L.; Hernández Pérez, P.M.; Ramírez Lasanta, R.; Delgado Bolton, R.C. Multitracer PET/CT with [18F]Fluorodeoxiglucose and [18F]Fluorocholine in the Initial Staging of Multiple Myeloma Patients Applying the IMPeTus Criteria: A Pilot Study. Diagnostics 2023, 13, 1570. https://doi.org/10.3390/diagnostics13091570
Garrastachu Zumarán P, García Megías I, Mangas Losada M, Mendoza Melero A, Villanueva Torres A, Boulvard Chollet X, Romero Robles L, Hernández Pérez PM, Ramírez Lasanta R, Delgado Bolton RC. Multitracer PET/CT with [18F]Fluorodeoxiglucose and [18F]Fluorocholine in the Initial Staging of Multiple Myeloma Patients Applying the IMPeTus Criteria: A Pilot Study. Diagnostics. 2023; 13(9):1570. https://doi.org/10.3390/diagnostics13091570
Chicago/Turabian StyleGarrastachu Zumarán, Puy, Irene García Megías, María Mangas Losada, Alejandro Mendoza Melero, Amós Villanueva Torres, Xavier Boulvard Chollet, Leonardo Romero Robles, Prisma Montserrat Hernández Pérez, Rafael Ramírez Lasanta, and Roberto C. Delgado Bolton. 2023. "Multitracer PET/CT with [18F]Fluorodeoxiglucose and [18F]Fluorocholine in the Initial Staging of Multiple Myeloma Patients Applying the IMPeTus Criteria: A Pilot Study" Diagnostics 13, no. 9: 1570. https://doi.org/10.3390/diagnostics13091570





