1. Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most aggressive malignancies. Its incidence continues to rise, but it maintains a low 5-years survival rate, below 12% [
1]. The poor prognosis is a result of a lack of early screening biomarkers, late diagnosis, early metastatic dissemination, and tumour resistance to systemic therapies. Surgery remains the only treatment with curative intent but less than 20% of the patients are candidates at the time of the diagnosis.
Differences in response to treatment and in clinical outcomes have been related to PDAC high molecular heterogeneity [
2] and to the presence of a predominant stromal component comprising up to 90% of the total tumour volume [
3]. Based on gene expressions and stromal characteristics, several subtypes of PDAC have been identified which differ in their clinical behaviour and response to treatment [
4].
Inter-patient heterogeneity is a key problem that has led to the failure of many clinical trials, highlighting the need for a more stratified therapeutic approach based on PDAC taxonomy. However, pragmatic approaches to subtyping for clinical implementation and survival prediction are lacking.
The answer may come from the emerging field of radiomics where image-derived features and radiomics features are linked to genomic profiles and beyond. This process, based on sophisticated statistical and machine learning approaches, may provide non-invasively important tumour characteristics, reflecting tumour biology with a major impact on the optimization and personalization of therapeutic strategies, on individual patients’ benefit and on overall disease survival [
5]. The extraction of these quantitative radiomics features can be correlated with different clinical outcomes, highlighting information that is not visible to the human eye which can be obtained from different imaging methods [
6,
7]. Radiomics features can be extracted from conventional imaging modalities and provide information about cancer biology; for example, tumour heterogeneity, which can be manifested at different levels (phenotypic, genomic, …) and reflects tumour aggressiveness, tumour grade, clinical outcomes, response to treatment and survival outcomes [
7,
8,
9,
10,
11].
The usefulness of this new approach has been reported in several cancer types, including PDAC, for predicting response to treatment and survival outcomes [
11]. More recent results of a radiogenomic approach for PDAC have demonstrated the possibility of identifying features correlated with gene mutations (mainly SMAD4, which could be associated with disease-free survival) and stromal content [
12,
13,
14,
15]. Both genetic mutations and stromal components have been considered to be predictive of response to treatment [
2,
3,
15]. Other studies have also demonstrated the role of radiomics in characterising PDAC and predicting resectability, risk of recurrence and overall survival [
6,
11].
However, previous studies in this area have often involved small patient cohorts with limited external validation, leading to findings that may not be generalizable. Notably, multicentric studies in this specific research domain are scarce, which restricts the dependability of the established knowledge [
16,
17,
18,
19]. In response to these gaps, the present study aims to identify predictive biomarkers for overall survival (OS) and progression-free survival (PFS) in patients with PDAC, using the radiomics-based approach of pre-treatment computed tomography (CT) images and clinical data. Our non-invasive approach, which synergizes medical imaging with machine learning, holds promise for improving patient management.
A key contribution and innovation of this study is its multicentric design, involving data from a network of 47 different hospitals, enabling a comprehensive evaluation of the proposed models’ effectiveness in a clinical routine. This large-scale, real-world scenario assessment underscores the practical utility of our methodology across diverse clinical settings. Moreover, the study delves into the significance of both clinical and radiomic features in the predictive models, providing insights into their utility in personalised care strategies based on survival outcomes prediction.
The structure of this article is organised to provide a comprehensive exploration of the study. Following the “Introduction”, which sets the stage by introducing the topic and outlining the rationale of our study, the manuscript is divided into several key sections. The “Materials and Methods” section elaborates on the methodology employed, detailing the patient selection process, the clinical data included in the study and the radiomic procedure—from image selection to the creation and testing of models—as well as the statistical analyses undertaken. The “Results” section presents the findings, focusing on the performance of the models, model explainability, and the survival curves of identified groups with varying risk levels. Finally, the “Discussion” section reflects on these results, comparing them with relevant literature, and addresses the limitations of the current study.
2. Material and Methods
2.1. Study Population
This retrospective study has been approved by the ethical review boards of the Hopital Universitaire de Bruxelles (HUB), respectively, of the Hopital Erasme (HE) site and of the Institut Jules Bordet (IJB) site and patient written consent was waived.
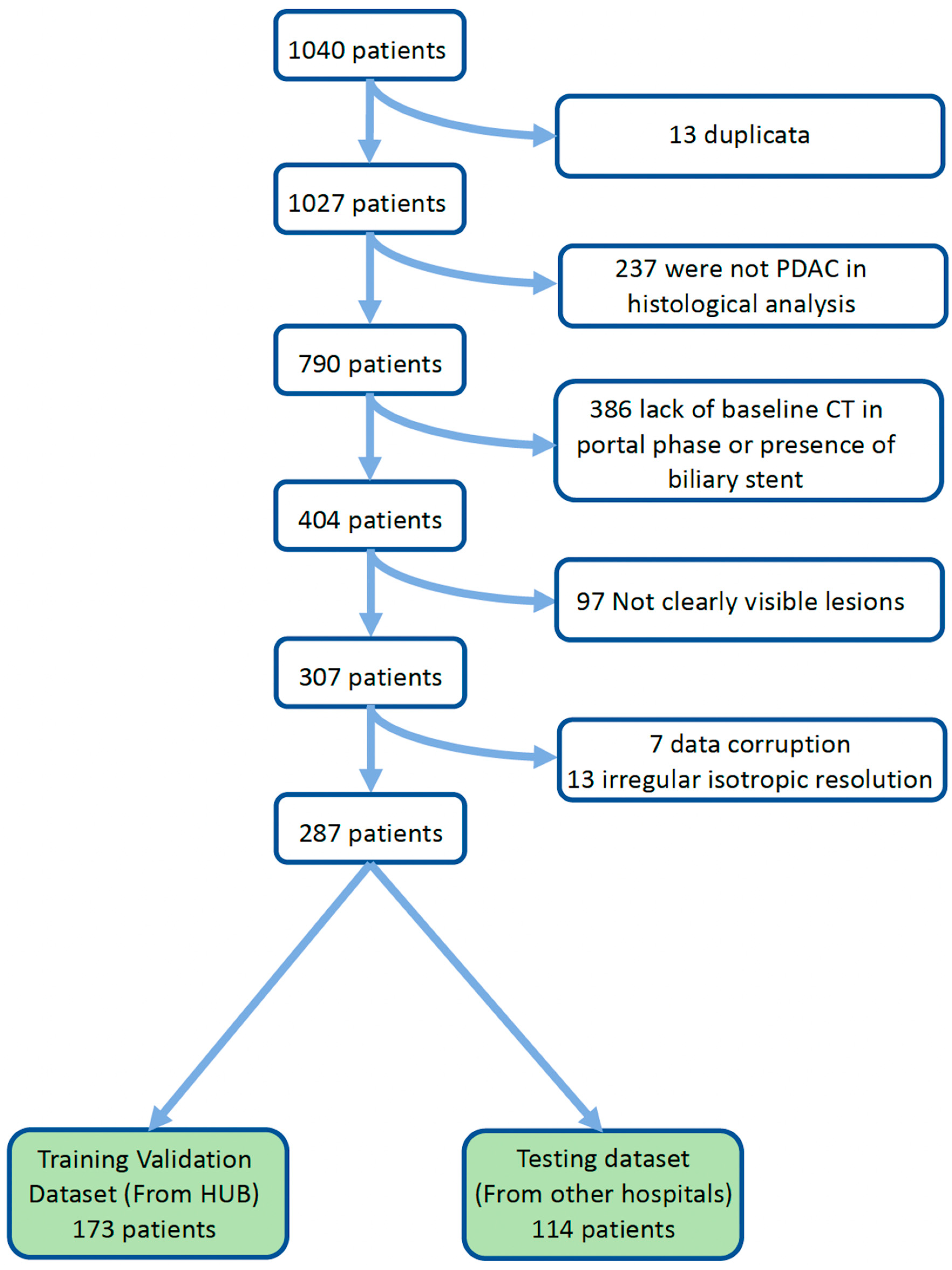
Between January 2010 and December 2020, patients with histologically proven PDAC were identified from HE (dataset 1), from IJB (dataset 2) and from the remaining hospital network connected to HUB (dataset 3—images stored in HUB archive system).
Inclusion criteria were: histologically proven PDAC (based on endoscopic-driven biopsy), patients older than 18 years, pre-treatment contrast-enhanced CT at the portal phase with a slice thickness greater than 1 mm and with clearly visible PDAC lesion. Exclusion criteria were: patients with no pretreatment CT, or with a pretreatment CT without portal phase or with biliary stent at the moment of CT scanner and patients with final diagnosis other than PDAC.
For each included patient the following clinical, histological and radiological data, representing the clinical features, were collected: age, gender, body mass index (BMI), alcohol consumption, tobacco consumption, Ca19.9, treatment type, treatment strategy, histological grade, resection margin, pancreatic tumour location, presence/absence of metastatic disease, disease subgrouping based on NCCN guidelines (version 2.2022 [
20,
21]).
OS was calculated in months from the date of diagnosis to date of death and PFS was calculated in months from the date of treatment initiation to the date of first progression. For PFS and OS, patients were considered to be censored if they were lost during follow-up. Additional details can be found in the
Supplementary material “Clinical, radiological and histological data”. If clinical features were missing, data were filled using the MissForest package version 0.2.0 (
https://pypi.org/project/MissForest/ (accessed on 13 May 2023)).
2.2. Patients Stratification
The exams in dataset 1 and dataset 2 were used as the training and validation dataset, specifically for feature selection and model evaluation through ten-fold cross-validation. Exams from dataset 3, comprising data from 47 different hospitals with varying CT acquisition protocols, were used as the testing dataset, which remained separate throughout the feature selection and model training phases. This stratification was used for both outcomes, OS and PFS.
2.3. Data Analysis
All patients underwent abdominal pretreatment contrast-enhanced CT. All the DICOM data were stored in the radiological department server of IJB, in accordance with the approbation of the ethical review boards. The portal phase was taken into account for this study.
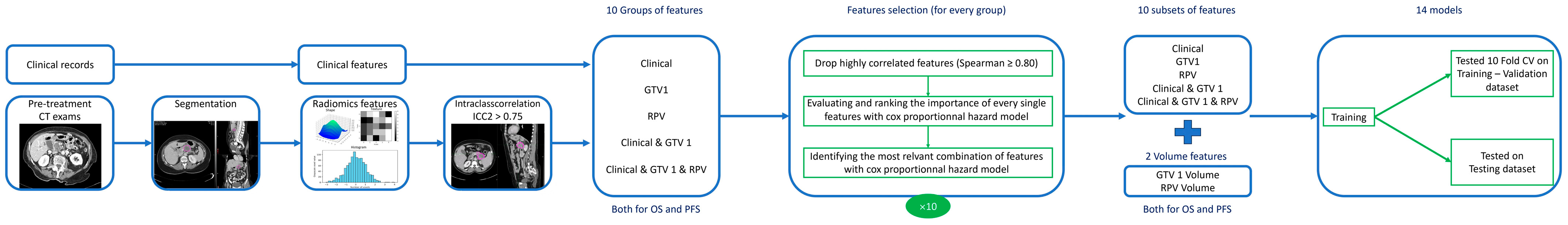
Subsequently, radiomics analysis was applied to the acquired images. This process involved: segmentation of the regions of interest, extracting quantitative features from the CT images (which capture the texture, shape, and intensity of the tumour region), feature selection to identify the most relevant features for our predictive models. These radiomic features were then used in conjunction with the clinical data to develop the predictive models for PFS and OS in PDAC patients. Further details are provided in the following paragraphs.
2.3.1. Segmentation and Features Extraction
Contrast-enhanced CT images were anonymized, and portal imaging sequences were sent to an archive connected with MIM 7.1.5™ (MIM Software Inc., Cleveland, OH, USA).
The CT images had different resolutions; therefore, all the exams were resampled to the same resolution, using the SimpleITK sitkBSpline interpolator for exams and the SimpleITK sitkNearestNeighbor interpolator for segmentations.
A radiologist with 12 years’ experience in abdominal imaging (RC, Reader 1) delineated and segmented semi-manually each tumour using MIM 7.1.5™ to obtain gross tumour volume (GTV1) (
Figure 1). To assess the intra-rater reliability of features, Reader 1 performed the tumour segmentations a second time (GTV2) in 45 randomly chosen patients after a few months. To assess the inter-rater reliability of features for GTV, a first-year radiologist resident (AM, Reader 2) delineated and segmented the pancreatic tumours from the same 45 randomly chosen patients (GTV3). Intra-rater and inter-observer reliability evaluation of features was obtained by using the IntraClass Correlation two-way mixed effect single measurement (ICC2). Only features with ICC2 greater than 0.75 were selected [
22,
23].
An automated segmentation tool developed at Maastricht University was used to delineate and automatically segment the non-tumoral pancreas (RPV) [
24]; subsequently, the RPV segmentations were manually verified and modified, using ITK-SNAP [
25] (Version 3.8.0,
http://www.itksnap.org/ (accessed on 13 May 2023)) (an example is provided in
Supplementary Materials Figure S1) by the two readers in consensus.
Radiomics features were extracted from GTV1, GTV2, GTV3 and RPV segmentations with PyRadiomics [
26]. For each segmentation, a total of 107 distinct features were extracted, encompassing seven primary groups: shape (14 features), first order (18 features), GLCM (24 features), GLDM (14 features), GLRLM (16 features), GLSZM (16 features) and NGTDM (5 features). Definitions of extracted features are described at
https://pyradiomics.readthedocs.io/en/latest/features.html (accessed on 13 May 2023). To assure better reproducibility, the intensity discretisation bin width was set to 25 [
27].
2.3.2. Feature Selection
Clinical features and features obtained from segmentations (GTV1, RPV) were used individually and in combination and the following feature subsets were obtained: clinical, GTV1, RPV, clinical>V1, clinical>V1&RPV, for both OS and PFS, resulting in 10 feature groups.
Initially, the features with the highest reliability based on the aforementioned ICC2 cutoff were selected (this step was applied only to GTV1 features).
Subsequently, for each group, highly correlated and redundant features were eliminated by applying a Spearman correlation coefficient threshold of 0.80.
Then, a univariate analysis was conducted to identify the most significant features that exhibit the highest C-index within Cox’s Proportional Hazards [
28,
29].
Lastly, for every group and using the previous assessment of the single features’ importance, the features were progressively combined to determine the most optimal combination, employing a Cox Proportional Hazards model.
In addition, as distinct sets, we used the volume measure for GTV1 and RPV, both for OS and PFS, resulting in a total of 14 sets of features (
Figure 2).
It is worth mentioning that these steps were computed only on the training–validation dataset (dataset 1 and dataset 2).
2.4. Model Building and Evaluation
Using the GradientBoostingSurvivalAnalysis classifier (scikit-survival version 0.19.0.post1) [
28], ten-fold cross-validation was performed on the training–validation dataset to tune the hyperparameter and for evaluation. Fourteen different models were trained, in particular they were based on each set of features, both for OS and PFS and tested on the testing dataset.
2.5. Model Interpretability
To explain the model’s prediction, we used SHapley Additive exPlanations (SHAP), a post hoc interpretability technique that assigns a SHAP value to each feature [
30,
31]. The SHAP value of each feature quantifies its impact on the model’s output. Positive SHAP values show that the presence of a feature pushes the prediction higher than the baseline and negative values suggest the opposite. The magnitude of these values demonstrates the strength of the feature’s influence on the model’s output. To provide a more comprehensive understanding, we used SHAP global summary plots. A SHAP summary plot ranks the features based on their average absolute SHAP values across all predictions and it helps to visualise their overall importance. Hence, SHAP helps to identify the most influential features and their trends in our best models for predicting OS and PFS.
2.6. Statistical Analysis
Chi-squared and Mann–Whitney were used to compare the clinical features between the training–validation and testing datasets. To analyse the various models, including those based solely on volume, we employed the C-index. This index generalises the area under the receiver operator characteristic curve (AUC) by assessing how well the model can separate the survival curves [
32].
A two-sided permutation test was used to assess the statistical significance of the C-index for survival prediction across the different models in the testing dataset.
For survival prediction, the top-performing models that predicted OS and PFS were analysed and used to obtain the risk score through the GradientBoostingSurvivalAnalysis model. For the classification of high and low-risk groups, we used the median risk score from the training–validation dataset as a threshold [
33]. Specifically, patients in the test set with risk scores above this median were classified as high risk, while those with scores below the median were categorised as low risk.
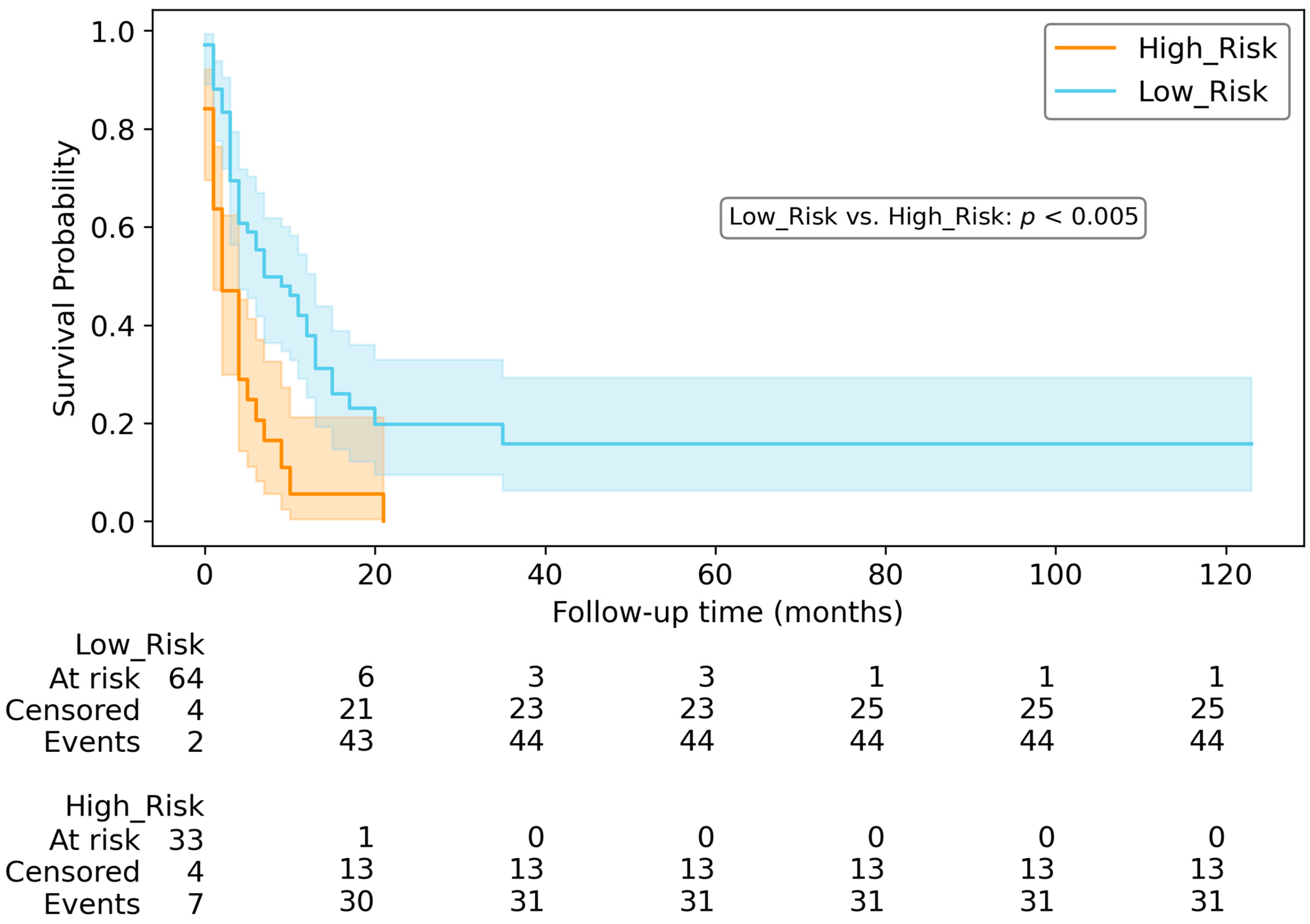
Subsequently, survival curves were generated using the Kaplan–Meier method to showcase the survival probabilities over time for these two groups. To assess the statistical difference between the low-risk group curve and high-risk group curve, in the testing dataset, the two-log-rank test was used.
The Spearman correlation coefficient was used to calculate the inter-correlation among the selected features and with volume.
All the above mentioned parts of the pipeline were computed using Python 3.8; the features were standardised using sklearn.preprocessing.StandardScaler (scikit-learn version 1.1.2) [
34].
4. Discussion
In this study, a comparative evaluation was performed on various models that integrated either or both clinical and radiomics features extracted from pretreatment CT scans to predict OS and PFS. Our findings indicate that the combination of clinical and GTV1 radiomics features yielded the best predictive model for OS, achieving a C-index of 0.72 in the testing dataset. Conversely, for PFS prediction, the best model was obtained using only clinical features, with a C-index of 0.70 for the testing dataset. The efficacy of the alternative models was relatively inferior in comparison.
Examining the statistical comparison among the different models, it is evident that models relying solely on clinical features showcase excellent performance in predicting both OS and PFS. While the Clinical>V1 model slightly surpasses the Clinical model in terms of C-index for OS, the difference between the two models lacks statistical significance (
p-value = 0.212;
Supplementary Materials Figure S5). The analysis underscores the robustness of clinical-based models in prognostication for OS and PFS.
From another perspective, in the absence of clinical data, the radiomic model based solely on GTV1 can be a viable alternative for predicting OS, as no statistically significant difference was observed between the Clinical and GTV1 models (
p-value = 0.156;
Supplementary Materials Figure S6). Regarding PFS, however, the Clinical model demonstrates a statistically superior C-index compared to the other models based solely on radiomic features.
Examining the Kaplan–Meier curves, the risk score derived from the top-performing models can be utilised to categorise patients into high-risk and low-risk categories for both OS (
Figure 6) and PFS (
Figure 7).
Our findings are consistent with the recent literature. For predicting survival time after the surgical resection, Park et al. [
35] reported a C-index of 0.74 for their clinical and radiomics model; differently from our results, they demonstrated a slight increase in the C-index when adding RPV features to the clinical + GTV model. Cheng et al. [
16] observed that CT texture analysis was associated with PFS and OS; by combining texture features and tumour size, they achieved an AUC of 0.756 for predicting OS. In contrast to our findings, CA19.9 levels were not found to be correlated with either OS or PFS. This discrepancy in the importance of CA19.9 might be attributed to the relatively small dataset, which consisted exclusively of 41 patients with unresectable PDAC. Separate from our findings, both previous studies [
16,
35] did not investigate the inter- or intra-observer reliability of segmentations. In a study by Yang et al. [
36], a radiomics signature was proposed to predict early death (within one year) in patients with advanced PDAC exhibiting stable disease. The radiomics signature achieved an AUC of 0.84 in the internal testing set and 0.87 in the external testing set. However, their inclusion criteria focused on pancreatic tumours with a size greater than or equal to 20 mm and patients with stable disease after chemotherapy. Healy et al. [
37] identified a pre-operative clinical–radiomic model for predicting OS and disease-free survival (DFS) in resectable PDAC patients. In the testing dataset, the model based solely on radiomic features achieved superior results for both OS (C-index of 0.564) and DFS (C-index of 0.573) compared to clinical and AJCC TNM models. In contrast to our findings, the radiomics model outperformed the clinical models. This disparity may be attributed to the exclusion of several clinical features in their models, such as R_margins, alcohol and tobacco consumption. Furthermore, their dataset exclusively included patients who met the criteria for a resectable PDAC.
Regarding the explainability of radiomics features, firstorder_90Percentile (from GTV1) was the unique radiomics features, selected in the OS Clinical>V1 model; a lower value showed a higher risk score, meaning that tumours with lower attenuation were associated with a poorer prognosis. This observation aligns with several studies [
37,
38], indicating that tumours exhibiting reduced enhancement on CT imaging, indicative of decreased vascularity, are associated with poorer OS. Lower density could be related to regions of tissue death due to venous invasion or hypoxia necrosis, both patterns of a more aggressive tumour behaviour [
39,
40,
41,
42].
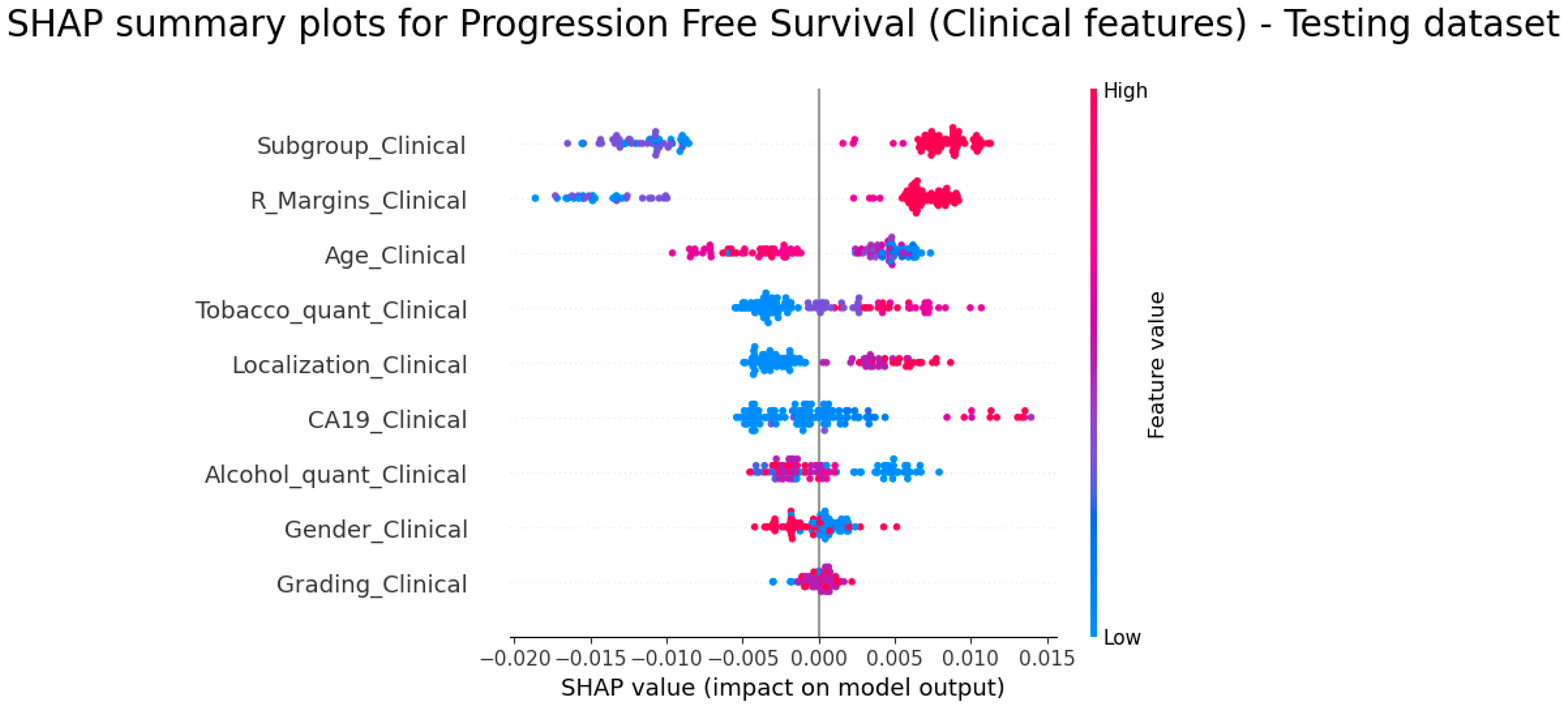
In relation to the explainability of other features in the OS Clinical>V1 model, the SHAP analysis proved that high values of CA19, Subgroup, Age, R_Margins and Grading are associated with a higher risk score; it is to be noted that, according to our feature mapping, the highest value for R_Margins represented patients that did not undergo surgery.
With regard to PFS prediction, the SHAP analysis showed a similar trend for Subgroup, CA19, R_Margins and Localization. Investigating the last mentioned feature, the highest value for Localisation meant tumours located at the tail; there are several studies showing different impacts based on the localization of the tumours, which also have contradictory results. Some articles [
43,
44,
45] did not discover a difference in mortality among tumours localised in the head and body/tail; however, other studies [
46,
47,
48] demonstrated that tail or body/tail localisation had a worse prognosis. Moreover, for PFS we observed a trend indicating higher alcohol consumption may lead to a lower risk; as indicated by published research, several studies have presented conflicting outcomes regarding the prognostic significance of alcohol in patients diagnosed with PDAC [
49,
50,
51,
52,
53].
To summarise, the SHAP analysis reveals and emphasises that, in predicting survival outcomes, clinical features exert a stronger influence compared to radiomics features.
There are several limitations in this study that need to be acknowledged.
The first limitation is the inclusion of data from 49 different centres, resulting in CT exams being performed on various CT scanners with different acquisition protocols. Unfortunately, the utilisation of ComBat for image harmonisation was not feasible due to the unavailability of at least 20 scans per centre, as suggested in [
54,
55]. To mitigate this bias, we utilised portal phases and resampled the voxel size to a consistent value for all exams.
The second limitation is the statistical differences observed between the training–validation dataset and the testing dataset within patient subgroups (p-value of 0.01) and R_margins (p-value of 0.02). It is worth considering that the baseline CT scans performed at the HUB IJB and HUB HE (the training–validation datasets) included cases spanning the entire spectrum of severity, and subsequently received surgical or non-surgical treatment options. On the other side, a most important part of exams in the testing dataset (performed in the hospital network connected to HUB) were redirected to the HUB IJB and HUB HE for surgical intervention (without repeating CT exam). This could explain the difference between the two datasets.
The third limitation is the explainability of radiomics features for temporal data. Radiomics features primarily capture static tumour characteristics, potentially limiting their ability to reflect dynamic changes in disease progression over time. Addressing this concern, delta–radiomics features can be utilised to elucidate the underlying biological mechanisms. Future investigations should aim to incorporate histological and genetic information, enhancing a more accurate representation of temporal changes and boosting the predictive capacity of radiomics models.
In conclusion, the clinical and radiomics models presented in this study, derived from a multicentric study and designed to predict OS and PFS in patients with PDAC, offer a non-invasive approach with the potential to improve patient management and care. Additionally, we utilised SHAP analysis to gain insights into the interpretability of the model. In order to validate and further enhance the clinical applicability of our findings, it will be mandatory to conduct prospective studies in a real clinical setting and to integrate histological and genetic data.