Pitfalls of Adrenal Washout on CT in the Adult Population: A Pictorial Review
Abstract
1. Introduction
2. Phaeochromocytoma
3. Lipid-Poor Adrenal Adenoma
4. Adrenocortical Carcinoma
5. Hypervascular Metastasis
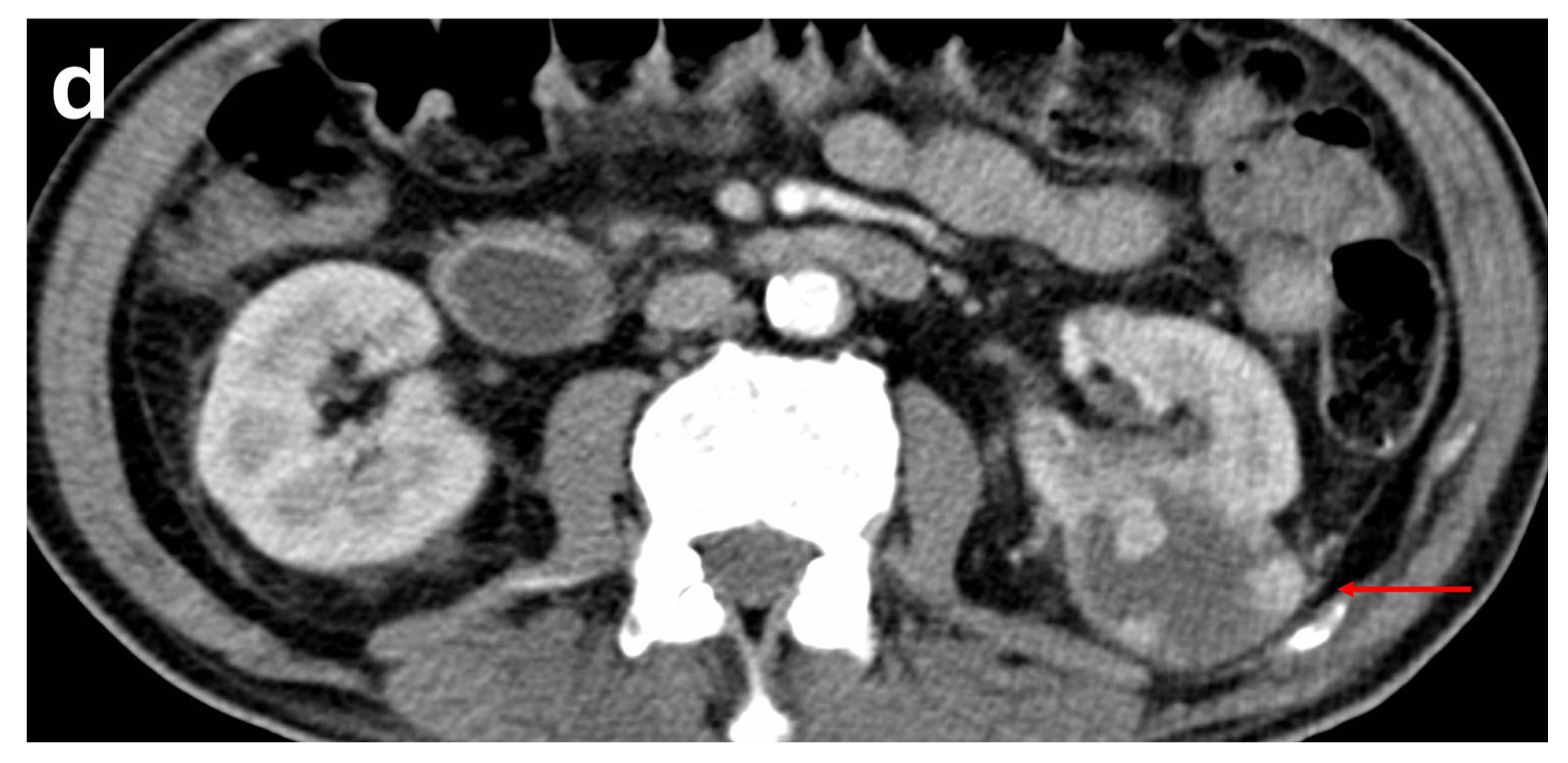
6. Retroperitoneal Haemangioma
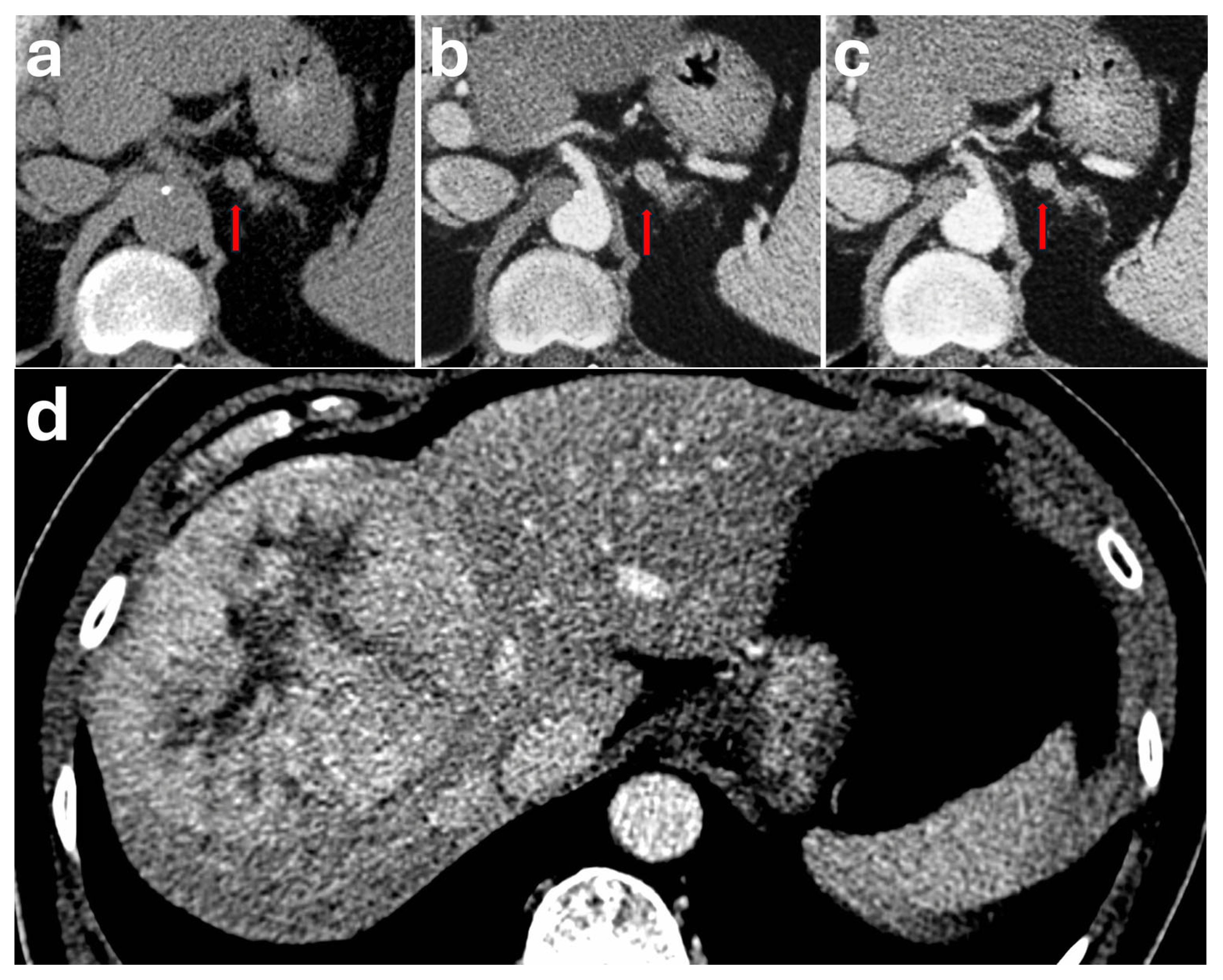
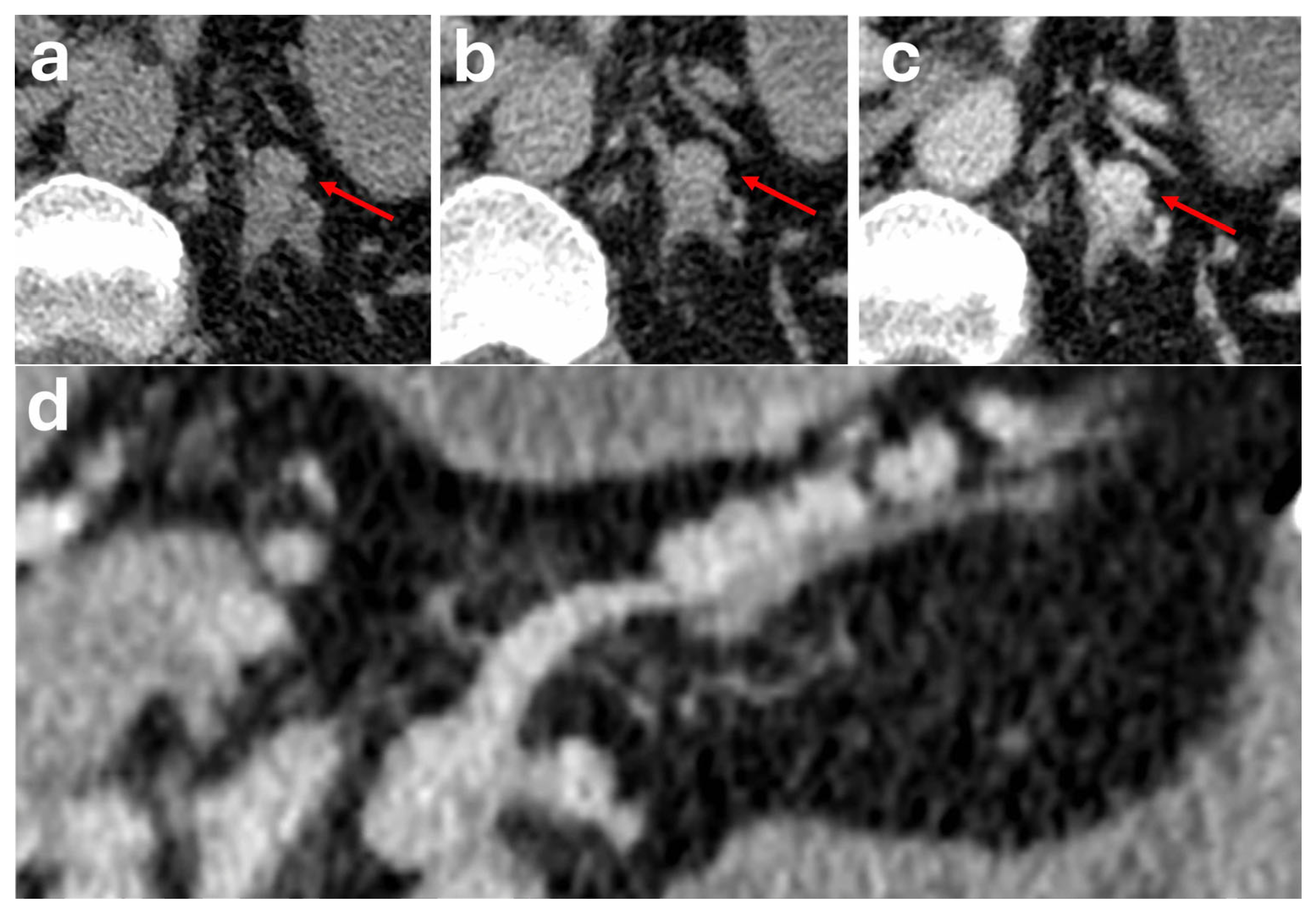
7. Periadrenal Varix
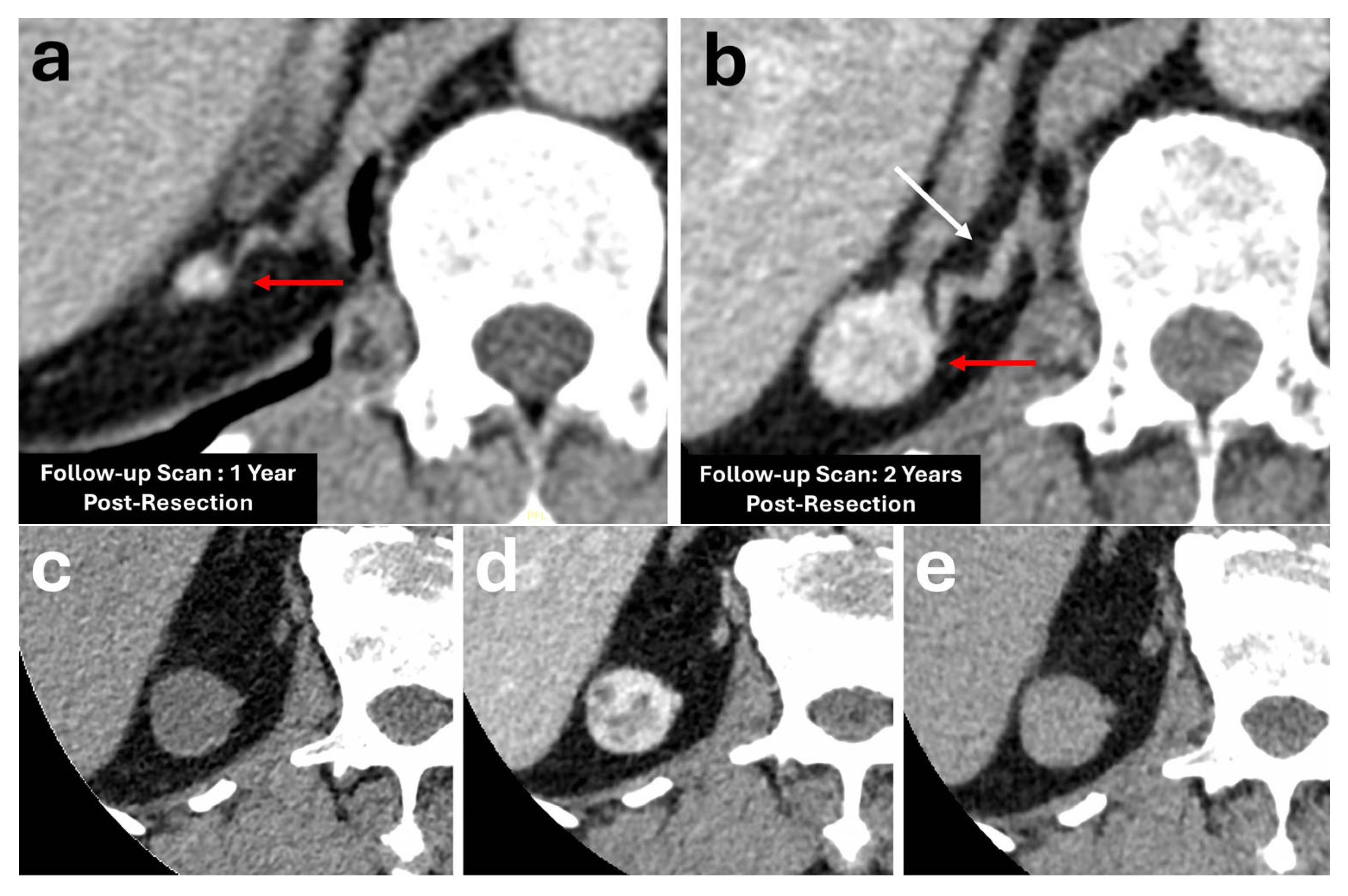
8. Adrenal Haemorrhage
9. Discussion
9.1. Approach to Hypervascular Lesions
9.2. Alternative Imaging Modalities
9.3. Current Recommendations
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bovio, S.; Cataldi, A.; Reimondo, G.; Sperone, P.; Novello, S.; Berruti, A.; Borasio, P.; Fava, C.; Dogliotti, L.; Scagliotti, G.V.; et al. Prevalence of adrenal incidentaloma in a contemporary computerized tomography series. J. Endocrinol. Investig. 2006, 29, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Chaudhry, F.S.; Mayo-Smith, W.W. The incidental adrenal mass on CT: Prevalence of adrenal disease in 1,049 consecutive adrenal masses in patients with no known malignancy. Am. J. Roentgenol. 2008, 190, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Caoili, E.M.; Korobkin, M.; Francis, I.R.; Cohan, R.H.; Platt, J.F.; Dunnick, N.R.; Raghupathi, K.I. Adrenal masses: Characterization with combined unenhanced delayed enhanced CT. Radiology 2002, 222, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Seow, J.H.; Stella, D.L.; Welman, C.J.; Somasundaram, A.J.; Gerstenmaier, J.F. Washed up: The end of an era for adrenal incidentaloma CT. Insights Imaging 2025, 16, 136. [Google Scholar] [CrossRef]
- Fassnacht, M.; Tsagarakis, S.; Terzolo, M.; Tabarin, A.; Sahdev, A.; Newell-Price, J.; Pelsma, I.; Marina, L.; Lorenz, K.; Bancos, I.; et al. European Society of Endocrinology clinical practice guidelines on the management of adrenal incidentalomas, in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 2023, 189, G1–G42. [Google Scholar] [CrossRef]
- van Aswegen, T.; Trinh, B.; Jacques, A.; Lo, G. Adrenal washout CT in patients with no history of cancer: A waste of time? Abdom. Radiol. 2024, 49, 3117–3126. [Google Scholar] [CrossRef] [PubMed]
- Schloetelburg, W.; Ebert, I.; Petritsch, B.; Weng, A.M.; Dischinger, U.; Kircher, S.; Buck, A.K.; Bley, T.A.; Deutschbein, T.; Fassnacht, M. Adrenal wash-out CT: Moderate diagnostic value in distinguishing benign from malignant adrenal masses. Eur. J. Endocrinol. 2022, 186, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Davenport, M.S.; Cohan, R.H.; Caoili, E.M. Can established CT attenuation and washout criteria for adrenal adenoma accurately exclude pheochromocytoma? Am. J. Roentgenol. 2013, 201, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Schieda, N.; Alrashed, A.; Flood, T.A.; Samji, K.; Shabana, W.; McInnes, M.D.F. Comparison of quantitative MRI and CT washout analysis for differentiation of adrenal pheochromocytoma from adrenal adenoma. Am. J. Roentgenol. 2016, 206, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Nasiroglu Imga, N.; Deniz, M.S.; Tural Balsak, B.O.; Aslan, Y.; Tuncel, A.; Berker, D. A comparative analysis of surgically excised hereditary and sporadic pheochromocytomas: Insights from a single-center experience. Kaohsiung J. Med. Sci. 2024, 40, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Čtvrtlïk, F.; Koranda, P.; Schovanek, J.; Škarda, J.; Hartmann, I.; Tudos, Z. Current diagnostic imaging of pheochromocytomas and implications for therapeutic strategy (Review). Exp. Ther. Med. 2018, 15, 3151–3160. [Google Scholar] [CrossRef]
- Falhammar, H.; Kjellman, M.; Calissendorff, J. Initial clinical presentation and spectrum of pheochromocytoma: A study of 94 cases from a single center. Endocr. Connect. 2018, 7, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Lenders, J.W.M.; Duh, Q.-Y.; Eisenhofer, G.; Gimenez-Roqueplo, A.-P.; Grebe, S.K.G.; Murad, M.H.; Naruse, M.; Pacak, K.; Young, W.F., Jr. Pheochromocytoma and Paraganglioma: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2014, 99, 1915–1942. [Google Scholar] [CrossRef] [PubMed]
- Ku, E.J.; Kim, K.J.; Kim, J.H.; Kim, M.K.; Ahn, C.H.; Lee, K.A.; Lee, S.H.; Lee, Y.-B.; Park, K.H.; Choi, Y.M.; et al. Diagnosis for Pheochromocytoma and Paraganglioma: A Joint Position Statement of the Korean Pheochromocytoma and Paraganglioma Task Force. Endocrinol. Metab. 2021, 36, 322–338. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Park, J.Y.; Kim, G.; Jin, S.-M.; Hur, K.Y.; Lee, S.-Y.; Kim, J.H. Urinary free metanephrines for diagnosis of pheochromocytoma and paraganglioma. Endocrinol. Metab. 2021, 36, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Brain, K.L.; Kay, J.; Shine, B. Measurement of urinary metanephrines to screen for pheochromocytoma in an unselected hospital referral population. Clin. Chem. 2006, 52, 2060–2064. [Google Scholar] [CrossRef] [PubMed]
- Fassnacht, M.; Assie, G.; Baudin, E.; Eisenhofer, G.; de la Fouchardiere, C.; Haak, H.; de Krijger, R.; Porpiglia, F.; Terzolo, M.; Berruti, A. Adrenocortical carcinomas and malignant phaeochromocytomas: ESMO–EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1476–1490. [Google Scholar] [CrossRef]
- Leung, K.; Stamm, M.; Raja, A.; Low, G. Pheochromocytoma: The Range of Appearances on Ultrasound, CT, MRI, and Functional Imaging. Am. J. Roentgenol. 2013, 200, 370–378. [Google Scholar] [CrossRef]
- Aggarwal, S.; Prete, A.; Chortis, V.; Asia, M.; Sutcliffe, R.P.; Arlt, W.; Ronchi, C.L.; Karavitaki, N.; Ayuk, J.; Elhassan, Y.S. Pheochromocytomas Most Commonly Present As Adrenal Incidentalomas: A Large Tertiary Center Experience. J. Clin. Endocrinol. Metab. 2024, 109, E389–E396. [Google Scholar] [CrossRef] [PubMed]
- Elsayes, K.M.; Elmohr, M.M.; Javadi, S.; Menias, C.O.; Remer, E.M.; Morani, A.C.; Shaaban, A.M. Mimics, pitfalls, and misdiagnoses of adrenal masses on CT and MRI. Abdom. Radiol. 2020, 45, 982–1000. [Google Scholar] [CrossRef]
- Bracci, B.; De Santis, D.; Del Gaudio, A.; Faugno, M.C.; Romano, A.; Tarallo, M.; Zerunian, M.; Guido, G.; Polici, M.; Polidori, T.; et al. Adrenal Lesions: A Review of Imaging. Diagnostics 2022, 12, 2171. [Google Scholar] [CrossRef]
- Albano, D.; Agnello, F.; Midiri, F.; Pecoraro, G.; Bruno, A.; Alongi, P.; Toia, P.; Di Buono, G.; Agrusa, A.; Sconfienza, L.M.; et al. Imaging features of adrenal masses. Insights Imaging 2019, 10, 1. [Google Scholar] [CrossRef]
- Chung, R.; Garratt, J.; Remer, E.M.; Navin, P.; Blake, M.A.; Taffel, M.T.; Hackett, C.E.; Sharbidre, K.G.; Tu, W.; Low, G.; et al. Adrenal Neoplasms: Lessons from Adrenal Multidisciplinary Tumor Boards. RadioGraphics 2023, 43, e220191. [Google Scholar] [CrossRef]
- McDermott, S.; McCarthy, C.J.; Blake, M.A. Images of pheochromocytoma in adrenal glands. Gland Surg. 2015, 4, 350–358. [Google Scholar] [CrossRef]
- Mohammed, M.F.; ElBanna, K.Y.; Ferguson, D.; Harris, A.; Khosa, F. Pheochromocytomas versus adenoma: Role of venous phase CT enhancement. Am. J. Roentgenol. 2018, 210, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Northcutt, B.G.; Trakhtenbroit, M.A.; Gomez, E.N.; Fishman, E.K.; Johnson, P.T. Adrenal Adenoma and Pheochromocytoma: Comparison of Multidetector CT Venous Enhancement Levels and Washout Characteristics. J. Comput. Assist. Tomogr. 2016, 40, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Perrier, N.D.; Grubbs, E.G.; Sircar, K.; Ye, Z.X.; Lee, J.E.; Ng, C.S. CT features and quantification of the characteristics of adrenocortical carcinomas on unenhanced and contrast-enhanced studies. Clin. Radiol. 2012, 67, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Timmers, H.J.L.M.; Taïeb, D.; Pacak, K.; Lenders, J.W.M. Imaging of Pheochromocytomas and Paragangliomas. Endocr. Rev. 2024, 45, 414–434. [Google Scholar] [CrossRef]
- Patel, D. Surgical approach to patients with pheochromocytoma. Gland Surg. 2020, 9, 32–42. [Google Scholar] [CrossRef]
- Bechmann, N.; Chiapponi, C.; Groeben, H.-T.; Grasshoff, C.; Zimmermann, P.; Walz, M.; Mogl, M.; Fendrich, V.; Holzer, K.; Rayes, N.; et al. Preoperative Management of Catecholamine-Producing Pheochromocytomas and Paragangliomas—Results from a DELPHI Process. J. Endocr. Soc. 2025, 9, bvaf024. [Google Scholar] [CrossRef]
- Press, D.; Akyuz, M.; Dural, C.; Aliyev, S.; Monteiro, R.; Mino, J.; Mitchell, J.; Hamrahian, A.; Siperstein, A.; Berber, E. Predictors of recurrence in pheochromocytoma. Surgery 2014, 156, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, A.; Taieb, A.; Hattab, F.; Ghachem, A.; Slama, N.B.H.; Halloul, I.; Saafi, W.; Marzouk, H.; Elfekih, H.; Saad, G.; et al. Characterization of Unilateral Adrenal Incidentalomas: Hormonal Analysis, Computed Tomography, and Magnetic Resonance Imaging Correlation. Cureus 2025, 17, e100542. [Google Scholar] [CrossRef] [PubMed]
- Park, J.J.; Park, B.K.; Kim, C.K. Adrenal imaging for adenoma characterization: Imaging features, diagnostic accuracies and differential diagnoses. Br. J. Radiol. 2016, 89, 20151018. [Google Scholar] [CrossRef] [PubMed]
- Boland, G.W.; Lee, M.J.; Gazelle, G.S.; Halpern, E.F.; McNicholas, M.M.; Mueller, P.R. Characterization of adrenal masses using unenhanced CT: An analysis of the CT literature. Am. J. Roentgenol. 1998, 171, 201–204. [Google Scholar] [CrossRef]
- Ho, L.M.; Paulson, E.K.; Brady, M.J.; Wong, T.Z.; Schindera, S.T. Lipid-poor adenomas on unenhanced CT: Does histogram analysis increase sensitivity compared with a mean attenuation threshold? Am. J. Roentgenol. 2008, 191, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Dinnes, J.; Bancos, I.; di Ruffano, L.F.; Chortis, V.; Davenport, C.; Bayliss, S.; Sahdev, A.; Guest, P.; Fassnacht, M.; Deeks, J.J.; et al. MANAGEMENT OF ENDOCRINE DISEASE: Imaging for the diagnosis of malignancy in incidentally discovered adrenal masses: A systematic review and meta-analysis. Eur. J. Endocrinol. 2016, 175, R51–R64. [Google Scholar] [CrossRef]
- Owei, L.; Wachtel, H. The Landmark Series: Evaluation and Management of Adrenal Incidentalomas. Ann. Surg. Oncol. 2025, 32, 4712–4719. [Google Scholar] [CrossRef]
- Bharwani, N.; Rockall, A.G.; Sahdev, A.; Gueorguiev, M.; Drake, W.; Grossman, A.B.; Reznek, R.H. Adrenocortical Carcinoma: The Range of Appearances on CT and MRI. Am. J. Roentgenol. 2011, 196, W706–W714. [Google Scholar] [CrossRef]
- Fassnacht, M.; Puglisi, S.; Kimpel, O.; Terzolo, M. Adrenocortical carcinoma: A practical guide for clinicians. Lancet Diabetes Endocrinol. 2025, 13, 438–452. [Google Scholar] [CrossRef] [PubMed]
- Thampi, A.; Shah, E.; Elshimy, G.; Correa, R. Adrenocortical carcinoma: A literature review. Transl. Cancer Res. 2020, 9, 1253–1264. [Google Scholar] [CrossRef]
- El Mjabber, R.; Alami, R.; Filali, N.; Bourial, A.; Dahbi, Z.; Kouhen, F.; Ismaili, N.; El Majjaoui, S.; Naim, A. Radiotherapy in Li-Fraumeni Syndrome: From Biological Concern to Personalized Clinical Decision-Making. Cureus 2026, 18, e100978. [Google Scholar] [CrossRef] [PubMed]
- Cingam, S.R.; Mukkamalla, S.K.R.; Karanchi, H. StatPearls [Internet]. 2023. Adrenal Metastasis. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30611952 (accessed on 5 January 2026). [PubMed]
- Lam, K.Y.; Lo, C.Y. Metastatic tumours of the adrenal glands: A 30-year experience in a teaching hospital. Clin. Endocrinol. 2002, 56, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Sasaguri, K.; Takahashi, N.; Takeuchi, M.; Carter, R.E.; Leibovich, B.C.; Kawashima, A. Differentiation of benign from metastatic adrenal masses in patients with renal cell carcinoma on contrast-enhanced CT. Am. J. Roentgenol. 2016, 207, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Yasaka, K.; Gonoi, W.; Akai, H.; Katsura, M.; Akahane, M.; Kiryu, S.; Ohtomo, K. Differentiation of adrenal tumors in patients with hepatocellular carcinoma: Adrenal adenoma versus metastasis. Eur. J. Radiol. 2013, 82, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.A.; Kim, C.K.; Park, B.K.; Kim, B. Evaluation of adrenal Metastases from renal cell carcinoma and hepatocellular carcinoma: Use of Delayed Contrast-enhanced CT. Radiology 2013, 266, 514–520. [Google Scholar] [CrossRef]
- Suntornlohanakul, O.; Mandal, S.; Saha, P.; Saygili, E.S.; Asia, M.; Arlt, W.; Elhassan, Y.S.; Prete, A.; Ronchi, C.L. Presentation and management of patients with adrenal masses: A large tertiary centre experience. Eur. J. Endocrinol. 2024, 191, 481–490. [Google Scholar] [CrossRef]
- Mao, J.J.; Dages, K.N.; Suresh, M.; Bancos, I. Presentation, disease progression and outcomes of adrenal gland metastases. Clin. Endocrinol. 2020, 93, 546–554. [Google Scholar] [CrossRef]
- Panwar, V.; Cai, Q.; Jia, L. Metastatic diseases to the adrenal gland: A comprehensive study from an academic institution with emphasis on clinical occult cases. Pathol. Res. Pract. 2024, 261, 155487. [Google Scholar] [CrossRef]
- Xue, X.; Song, M.; Xiao, W.; Chen, F.; Huang, Q. Imaging findings of retroperitoneal anastomosing hemangioma: A case report and literature review. BMC Urol. 2022, 22, 77. [Google Scholar] [CrossRef]
- Debaibi, M.; Sghair, A.; Sahnoun, M.; Zouari, R.; Essid, R.; Kchaou, M.; Dhaoui, A.; Chouchen, A. Primary retroperitoneal cavernous hemangioma: An exceptional disease in adulthood. Clin. Case Rep. 2022, 10, e05850. [Google Scholar] [CrossRef]
- Ghalib, O.H.; Bapir, R.; Hassan, H.A.; Hussain, L.L.; Ismaeil, D.A.; Tahir, S.H.; Najar, K.A.; Hama, J.I.; Abdullah, H.O.; Kakamad, F.H. Retroperitoneal cavernous hemangioma: A case report with literature review. Radiol. Case Rep. 2026, 21, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wu, J. Multimodal imaging features of retroperitoneal anastomosing hemangioma: A case report and literature review. Front. Oncol. 2023, 13, 1269631. [Google Scholar] [CrossRef] [PubMed]
- Brady, T.; Gross, B.; Glazer, G.; Williams, D. Adrenal pseudomasses due to varices: Angiographic-CT-MRI-pathologic correlations. Am. J. Roentgenol. 1985, 145, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Mitty, H.A.; Cohen, B.A.; Sprayregen, S.; Schwartz, K. Adrenal pseudotumors on CT due to dilated portosystemic veins. AJR Am. J. Roentgenol. 1983, 141, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.Y.; Fung, K.K.F.; Ng, J.C.; Lee, B.K.H.; Cheng, K.K.; Cho, D.H.Y. Adrenal Venous Sampling for Recognising the Many Variants of Adrenal Veins: A Pictorial Essay. Hong Kong J. Radiol. 2024, 27, 248–258. [Google Scholar] [CrossRef]
- Sacerdote, M.G.; Johnson, P.T.; Fishman, E.K. CT of the adrenal gland: The many faces of adrenal hemorrhage. Emerg. Radiol. 2012, 19, 53–60. [Google Scholar] [CrossRef]
- Jordan, E.; Poder, L.; Courtier, J.; Sai, V.; Jung, A.; Coakley, F.V. Imaging of nontraumatic adrenal hemorrhage. Am. J. Roentgenol. 2012, 199, W91–W98. [Google Scholar] [CrossRef] [PubMed]
- Elhassan, Y.S.; Ronchi, C.L.; Wijewickrama, P.; Baldeweg, S.E. Approach to the Patient With Adrenal Hemorrhage. J. Clin. Endocrinol. Metab. 2023, 108, 995–1006. [Google Scholar] [CrossRef] [PubMed]
- Świeczkowski-Feiz, S.; Toutounchi, S.; Krajewska, E.; Celejewski, K.; Gelo, R.; Kaszczewski, P.; Jakuczun, W.; Ambroziak, U.; Gałązka, Z. Adrenal hemorrhage: Diagnostics, management, and treatment. Review and clinical update. Wideochirurgia Inne Tech. Maloinwazyjne 2025, 20, 255–260. [Google Scholar] [CrossRef]
- Russell, G.; Navale, P.; Ludwig, D.R.; Strnad, B.S.; Itani, M. Peripheral nodular enhancement in adrenal and renal hematomas: A report of 3 cases. Radiol. Case Rep. 2023, 18, 3371–3375. [Google Scholar] [CrossRef]
- Dong, A.; Cui, Y.; Wang, Y.; Zuo, C.; Bai, Y. 18F-FDG PET/CT of Adrenal Lesions. Am. J. Roentgenol. 2014, 203, 245–252. [Google Scholar] [CrossRef] [PubMed]





| Clinical Presentation/Biochemical Markers (If Any) | Key CT Imaging Features | Alternative Imaging/Confirmatory Studies | |
|---|---|---|---|
| Phaeochromocytoma | Signs and symptoms of excess catecholamines. Triad of headache, palpitations, sweating (highly specific) Germline mutations in 40% (e.g., MEN2A, MEN2B, VHL 2, succinate dehydrogenase B, C and D mutations) Elevated plasma catecholamine and 24 h urinary catecholamine metabolites (highly specific) | Avidly enhancing mass (often >130 HU); homogeneous enhancement if small; heterogeneous appearance if large with necrosis/cystic/haemorrhagic change. If malignant, there may be local invasion, tumour thrombus or metastasis. | MRI if CT is inconclusive or patient deemed not suitable PET/CT or SPECT for evaluation of metastasis or multiplicity. |
| Lipid-Poor Adenoma | Majority are asymptomatic and non-functioning. Some can secrete steroid hormones | Well-circumscribed, rounded, homogenous mass measuring 1–4 cm, lack of aggressive features. Demonstrates stability on follow-up, rapid washout on AWCT (though non-specific). If large, it can show heterogeneous appearance with necrosis/cystic change, haemorrhage or calcification. | MRI to look for inherent fat |
| Adrenocortical Carcinoma (ACC) | Features of excess steroid hormone in up to 60% | Large (>5 cm); heterogeneous mass due to haemorrhage or necrosis; may show central calcification (30%). Can show intratumoural macroscopic fat Features of malignancy: locoregional invasion of adjacent structures, venous invasion, tumour thrombus or metastatic spread | MRI is an alternative equally as effective as CT. FDG-PET/CT can be used, especially if there is suspicion of bone metastasis |
| Hypervascular Metastasis | History of primary cancer (e.g., RCC, HCC) | High attenuation of >140 HU, often bilateral (43%). Can show rapid growth and clinical history of extra-adrenal malignancy. | Histopathological correlation or correlation with known primary malignancy |
| Retroperitoneal Haemangioma | Non-specific presentation | Non-specific features, although classically show centripetal filling and progressive enhancement reaching >150 HU on venous phase | At times, histopathological correlation is required due to non-specific features |
| Periadrenal Varix | Non-specific presentation. Patients may have history of chronic liver disease leading to portal hypertension | Ovoid lesion mimicking a hypervascular adrenal mass if close to the adrenal gland. Often requires MPR to show its periadrenal location. Typically showing a serpiginous appearance and enhancing in tandem with vessels (often showing continuation with renal/phrenic veins | Nil specific |
| Adrenal Haemorrhage | Non-specific presentation. Common aetiologies: trauma, stress, coagulopathy, underlying adrenal lesion | Highly variable features. Typically demonstrates high unenhanced HU (50–90 HU), temporal evolution is a hallmark feature (reduction in size and HU on follow-up) | Serial follow-up CT imaging showing resolution or size reduction |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tay, B.H.B.; Sim, J.Z.T. Pitfalls of Adrenal Washout on CT in the Adult Population: A Pictorial Review. Diagnostics 2026, 16, 920. https://doi.org/10.3390/diagnostics16060920
Tay BHB, Sim JZT. Pitfalls of Adrenal Washout on CT in the Adult Population: A Pictorial Review. Diagnostics. 2026; 16(6):920. https://doi.org/10.3390/diagnostics16060920
Chicago/Turabian StyleTay, Benjamin Hao Bai, and Jordan Zheng Ting Sim. 2026. "Pitfalls of Adrenal Washout on CT in the Adult Population: A Pictorial Review" Diagnostics 16, no. 6: 920. https://doi.org/10.3390/diagnostics16060920
APA StyleTay, B. H. B., & Sim, J. Z. T. (2026). Pitfalls of Adrenal Washout on CT in the Adult Population: A Pictorial Review. Diagnostics, 16(6), 920. https://doi.org/10.3390/diagnostics16060920






