Neuroinflammation and Schizophrenia: New Therapeutic Strategies through Psychobiotics, Nanotechnology, and Artificial Intelligence (AI)
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
- (Psychobiotics OR probiotics) AND schizophrenia AND neuroinflammation;
- (Psychobiotics OR probiotics) AND schizophrenia AND (nanotechnology OR artificial intelligence).
2.2. Data Storage and Selection Tools
3. Results
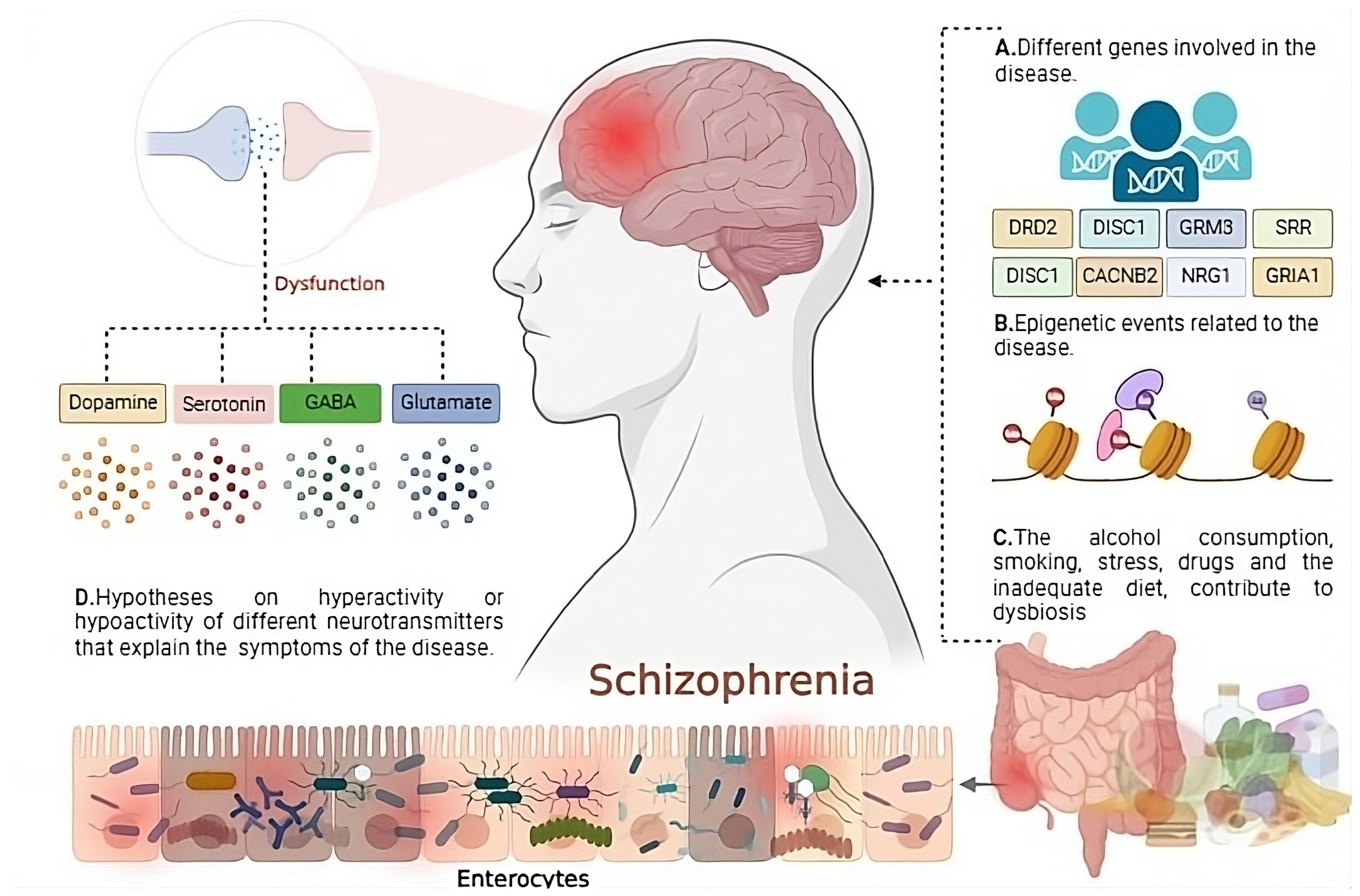
3.1. Schizophrenia: Definition, Symptoms, Risk Factors, Diagnosis, Classification, and Treatment
3.1.1. Definition
3.1.2. Epidemiology
3.2. Neuroinflammation in Schizophrenia
3.3. Gut Microbiota and Schizophrenia
3.4. Impact of Blood–Brain Barrier Dysfunction and Microbiota on Schizophrenia
3.5. The Role of Brain-Derived Neurotrophic Factor in Schizophrenia
3.6. Psychobiotics in the Treatment of Schizophrenia
- ○
- Age: aging is associated with a reduction in the diversity of the intestinal microbiome, altering its configuration and potentially impacting overall health.
- ○
- Diet and Nutrition: eating habits and periods of malnutrition or overnutrition are significant change agents for the gut microbiota.
- ○
- Substance and Medication Use: The use of substances, including laxatives, antibiotics, and antipsychotics, has the potential to alter the composition of the microbiota. These changes can influence the bioavailability and efficacy of various medications.
- ○
- Physical Exercise: Physical activity promotes the diversity of the microbiome, suggesting a positive link between exercise and intestinal health.
- ○
- Geographic Location: the composition of the gut microbiota can vary significantly according to an individual’s geographic location, possibly due to differences in diet and environment.
- ○
- Sampling Time: diurnal variability in sampling can reflect differences in microbial abundance and diversity, related to the food intake cycle.
- Protection of Intestinal Barrier Function: Probiotics help maintain the integrity of the intestinal epithelium by promoting tight junction proteins, such as claudins, Zona occludin-1, and occludin, whose levels are significantly reduced in some disease conditions. Probiotics, like Lactobacillus rhamnosus GG, secrete effector molecules that stimulate the activation of ADAM17 and the release of HB-EGF, resulting in the transactivation of the Epidermal Growth Factor (EGF) receptor, prevention of apoptosis, and preservation of intestinal epithelial function.
- Stimulation of the Immune System: probiotics contribute to the maturation of the immune system, inducing the production of IL-10 in peripheral blood mononuclear cells, which facilitates the production of IgA antibodies at mucosal sites and enhances the immune response.
- Modulation of the CNS: some probiotics can produce neurotransmitters like GABA, influencing neuronal activity in the gastrointestinal tract, possibly affecting the CNS through vagus-nerve-mediated communication.
- Influence on the Host’s Microbiota: probiotics can modify the composition and function of the host’s gut microbiota by producing antimicrobial compounds that suppress or promote the growth of certain microorganisms in the gut, thus contributing to a healthy balance of the microbiota.
- Modulation of Metabolic Responses: probiotics can produce conjugated linoleic acid (CLA) and other compounds that regulate the expression of tight junction proteins and antioxidant enzymes, reducing oxidative stress in colonocytes and modulating inflammation.
- Cholesterol Reduction: some probiotics have the ability to produce bile salt hydrolase, which hydrolyzes conjugated bile salts; this releases less-soluble primary bile acids that are excreted rather than reabsorbed, contributing to the reduction in blood cholesterol levels.
3.7. Revolutionizing Gut Health: The Promise of Artificial Intelligence, Nanotechnology, and Synthetic Biology in Psychobiotics
3.8. Advancements and Challenges in the Application of Nanoprobiotics and Nanomedicine
| Material Type | Description | Applications | Advantages | Disadvantages |
|---|---|---|---|---|
| Nanocellulose [16,18] | Available in CNC and CNF forms. Known for its low toxicity, biocompatibility, and adjustable surface properties. It has been shown to improve the properties of probiotic delivery systems when used as an encapsulating material. | Probiotic Encapsulation | Biocompatible and eco-friendly; provides mechanical strength and adjustable surface properties for better encapsulation. | Limited protection against extreme pH and enzymes. |
| Magnesium Oxide Nanoparticles (MgO NPs) [16,18] | Attracted attention due to its high surface area, non-toxicity, mechanical resistance, thermal stability, and low cost. Used for microencapsulation of probiotics, showing an improvement in probiotic viability in acidic environments. | Microencapsulation of Probiotics | Enhances probiotic viability in acidic environments; offers mechanical resistance and thermal stability. | Potential aggregation in biological media; requires careful surface modification. |
| Chitosan Nanoparticles (CSNPs) [16,18,114] | Derived from the alkaline deacetylation of chitin; chitosan is a natural polysaccharide with cationic properties, biocompatibility, non-toxicity, and low cost. Chitosan nanoparticles have shown promise for the encapsulation of probiotic cells, protecting them in the GI tract and improving their mucoadhesive properties. | Encapsulation and Protection in GI Tract | Biocompatible; non-toxic; enhances mucoadhesive properties, protecting probiotics in the GI tract. | Limited solubility in water and some solvents; potential deacetylation challenges. |
| Eudragit S100 Nanoparticles [16,18,114] | A synthetic anionic polymer derived from methacrylic acid and methyl methacrylate ester. Its solubility depends on pH, being insoluble in strongly acidic solutions and slightly soluble in regions of the digestive tract with neutral to weakly alkaline pH. Used to improve viability of probiotic bacteria. | Enhancing Probiotic Viability | Effective at protecting probiotics in acidic GI environments; pH-responsive solubility for targeted release. | Requires careful formulation to achieve desired solubility and release profiles. |
| Starch Nanoparticles [18,80] | One of the most abundant biopolymers in nature; produced by many plants and crops. Starch nanoparticles and nanocrystals have been used for biomedical applications, especially in drug delivery. Although, not the best candidate for probiotic microencapsulation due to potential immediate release in hostile environments. | Potential for Probiotic Encapsulation | Natural and biodegradable; potentially low cost. | Possible immediate release in hostile environments, modifications necessary for stability. |
| Liposomes [124] | Spherical vesicles composed of one or more lipid layers surrounding an aqueous core. Morphologically similar to cell membranes, they can encapsulate hydrophilic drugs in their aqueous core and lipophilic drugs in the lipid bilayer, making them versatile for the delivery of a wide range of therapies. | Wide Range of Therapy Delivery | Can encapsulate both hydrophilic and lipophilic compounds; biocompatible and versatile for various therapies. | Stability issues in the GI tract; potential for leakage or fusion with other lipids. |
| Polymeric Nanoparticles (PNPs) [124] | Colloidal mixtures of biocompatible and biodegradable polymers forming a dense matrix; capable of encapsulating lipophilic drugs within its structure. These nanoparticles offer steric stabilization, protection from enzymatic degradation, and controlled drug release. | Drug Delivery and Stabilization | Protection from enzymatic degradation; controlled release; steric stabilization. | Potential for immune response; complexity in manufacturing. |
| Solid Lipid Nanoparticles (SLNs) [124] | Colloidal dispersions of lipids that solidify at room or body temperature. They offer physical stability, drug protection, and low toxicity. Capable of encapsulating both hydrophilic and lipophilic drugs. | Drug Encapsulation | Physical stability; drug protection; low toxicity. | Limited drug loading capacity; potential for drug expulsion during storage. |
| Micelles [125] | Composed of amphiphilic molecules (having a hydrophilic and a hydrophobic part), micelles form core–shell structures where the hydrophobic core can encapsulate lipophilic drugs, improving their solubility and bioavailability. | Enhancing Solubility and Bioavailability | Improves solubility and bioavailability of lipophilic drugs; simple to prepare. | Critical micelle concentration dependent stability; potential dilution issues in vivo. |
| Nanoemulsions (NE) [124] | Colloidal systems containing oil, water, and surfactants; capable of improving the solubility of water-insoluble drugs and offering controlled drug release. | Solubility Improvement and Controlled Release | Enhances solubility of water-insoluble drugs; controlled release capabilities. | Physical stability over time can be challenging, requiring surfactants for stabilization. |
| Dendrimers [124] | Highly branched polymeric structures providing a platform for drug conjugation, aimed at improving solubility, stability, and efficacy of drug delivery across the blood–brain barrier. | Drug Delivery Efficiency | High drug loading capacity; targeted delivery potential; modifiable surface for functionalization. | Complexity in synthesis; potential toxicity depending on composition and dose. |
| DNA-based Nanodevices [120,122,123] | Utilizing the precision of DNA to form nanoscale structures, these devices are tailored for specific interactions within biological systems. They are especially promising for the accurate placement of probiotics or psychobiotics within the gastrointestinal tract. | Targeted Delivery of Probiotics and Psychobiotics | Precise control over delivery location; capable of protecting cargo through harsh conditions; programmable release triggered by environmental factors. | Complexity in design and synthesis; potential for unanticipated interactions with the body’s biochemistry. |
| Quantum Dots (QDs) [124,126] | Nanocrystalline semiconductors offering unique electronic and optical properties, such as high emission and photostability; useful for imaging and diagnosis of CNS disorders, as well as drug delivery. | Imaging, Diagnosis, and Drug Delivery | High photostability and emission for imaging; potential for targeted drug delivery. | Toxicity concerns, especially with heavy-metal-containing QDs; stability in biological environments |
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CNS | Central Nervous System |
| MHC | Major Histocompatibility Complex |
| CD19 and CD20B | Types of Lymphocytes (B Cell Markers) |
| BDNF | Brain-Derived Neurotrophic Factor |
| IFNγ | Interferon Gamma |
| CRP | C-Reactive Protein |
| IBS | Irritable Bowel Syndrome |
| NMDAR | N-Methyl-D-Aspartate Receptor |
| SCFAs | Short-Chain Fatty Acids |
| GRAS | Generally Recognized As Safe |
| AI | Artificial Intelligence |
| DSM-5 | Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition |
| IL-6, IL-8, IL-10, TNF-α | Interleukin 6, Interleukin 8, Interleukin 10, Tumor Necrosis Factor Alpha |
| HPA Axis | Hypothalamic–Pituitary–Adrenal Axis |
| ENS | Enteric Nervous System |
| EGF | Epidermal Growth Factor |
| GABA | Gamma-Aminobutyric Acid |
| MGB Axis | Microbiota–Gut–Brain Axis |
| TLRs | Toll-Like Receptors |
| LGS | Leaky Gut Syndrome |
| LPS | Lipopolysaccharide |
| CCK | Cholecystokinin |
| PYY | Peptide Tyrosine Tyrosine |
| GLP-1 | Glucagon-Like Peptide-1 |
| CRH | Corticotropin-Releasing Hormone |
| NO | Nitric Oxide |
| TPH | Tryptophan Hydroxylase |
| MAO | Monoamine Oxidase |
| PET | Positron Emission Tomography |
| SSRI | Selective Serotonin Reuptake Inhibitors |
| CLA | Conjugated Linoleic Acid |
| TSPO | Translocator Protein |
References
- Patel, K.R.; Cherian, J.; Gohil, K.; Atkinson, D. Schizophrenia: Overview and Treatment Options. Peer Rev. J. Formul. Manag. 2014, 39, 638–645. [Google Scholar]
- Stępnicki, P.; Kondej, M.; Kaczor, A.A. Current Concepts and Treatments of Schizophrenia. Molecules 2018, 23, 2087. [Google Scholar] [CrossRef] [PubMed]
- Saparia, P.; Patel, A.; Shah, H.; Solanki, K.; Patel, A.; Sahayata, M. Schizophrenia: A Systematic Review. Clin. Exp. Psychol. 2022, 9, 8–13. [Google Scholar]
- Correll, C.U.; Schooler, N.R. Negative Symptoms in Schizophrenia: A Review and Clinical Guide for Recognition, Assessment, and Treatment. Neuropsychiatr. Dis. Treat. 2020, 16, 519–534. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, T. The Potential of Probiotics in the Treatment of Schizophrenia. Clin. Neuropsychopharmacol. Ther. 2021, 12, 18–22. [Google Scholar] [CrossRef]
- Vallée, A. Neuroinflammation in Schizophrenia: The Key Role of the WNT/β-Catenin Pathway. Int. J. Mol. Sci. 2022, 23, 2810. [Google Scholar] [CrossRef] [PubMed]
- Munawar, N.; Ahsan, K.; Muhammad, K.; Ahmad, A.; Anwar, M.A.; Shah, I.; Al Ameri, A.K.; Al Mughairbi, F. Hidden Role of Gut Microbiome Dysbiosis in Schizophrenia: Antipsychotics or Psychobiotics as Therapeutics? Int. J. Mol. Sci. 2021, 22, 7671. [Google Scholar] [CrossRef] [PubMed]
- Bleibel, L.; Dziomba, S.; Waleron, K.F.; Kowalczyk, E.; Karbownik, M.S. Deciphering Psychobiotics’ Mechanism of Action: Bacterial Extracellular Vesicles in the Spotlight. Front. Microbiol. 2023, 14, 1211447. [Google Scholar] [CrossRef]
- Del Toro-Barbosa, M.; Hurtado-Romero, A.; Garcia-Amezquita, L.E.; García-Cayuela, T. Psychobiotics: Mechanisms of Action, Evaluation Methods and Effectiveness in Applications with Food Products. Nutrients 2020, 12, 3896. [Google Scholar] [CrossRef]
- Iliopoulou, S.M.; Tsartsalis, S.; Kaiser, S.; Millet, P.; Tournier, B.B. Dopamine and Neuroinflammation in Schizophrenia—Interpreting the Findings from Translocator Protein (18kDa) PET Imaging. Neuropsychiatr. Dis. Treat. 2021, 17, 3345–3357. [Google Scholar] [CrossRef]
- Comer, A.L.; Carrier, M.; Tremblay, M.-È.; Cruz-Martín, A. The Inflamed Brain in Schizophrenia: The Convergence of Genetic and Environmental Risk Factors That Lead to Uncontrolled Neuroinflammation. Front. Cell. Neurosci. 2020, 14, 274. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.E.; Walker, A.K.; Weickert, C.S. Neuroinflammation in Schizophrenia: The Role of Nuclear Factor Kappa B. Transl. Psychiatry 2021, 11, 528. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the Manipulation of Bacteria-Gut-Brain Signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Gupta, D.; Mehrotra, R.; Mago, P. Psychobiotics: The Next-Generation Probiotics for the Brain. Curr. Microbiol. 2021, 78, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Ju, S.; Shin, Y.; Han, S.; Kwon, J.; Choi, T.G.; Kang, I.; Kim, S.S. The Gut–Brain Axis in Schizophrenia: The Implications of the Gut Microbiome and SCFA Production. Nutrients 2023, 15, 4391. [Google Scholar] [CrossRef] [PubMed]
- Dangi, P.; Chaudhary, N.; Chaudhary, V.; Virdi, A.S.; Kajla, P.; Khanna, P.; Jha, S.K.; Jha, N.K.; Alkhanani, M.F.; Singh, V.; et al. Nanotechnology Impacting Probiotics and Prebiotics: A Paradigm Shift in Nutraceuticals Technology. Int. J. Food Microbiol. 2023, 388, 110083. [Google Scholar] [CrossRef] [PubMed]
- Fernanda Barboza Duarte, I.; Lívia Oliveira Nascimento Mergulhão, N.; Da Costa Silva, V.; Carolina Gomes De Bulhões, L.; Diniz Basílio, I., Jr.; Silva, A.C.A. Natural Probiotics and Nanomaterials: A New Functional Food. In Prebiotics and Probiotics—From Food to Health; Franco Robles, E., Ed.; IntechOpen: London, UK, 2022; ISBN 978-1-83969-575-9. [Google Scholar]
- Razavi, S.; Janfaza, S.; Tasnim, N.; Gibson, D.L.; Hoorfar, M. Nanomaterial-Based Encapsulation for Controlled Gastrointestinal Delivery of Viable Probiotic Bacteria. Nanoscale Adv. 2021, 3, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- McCoubrey, L.E.; Seegobin, N.; Elbadawi, M.; Hu, Y.; Orlu, M.; Gaisford, S.; Basit, A.W. Active Machine Learning for Formulation of Precision Probiotics. Int. J. Pharm. 2022, 616, 121568. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, H.; Zheng, L.; Li, J.; Hong, Y.; Liang, P.; Kwok, L.-Y.; Zuo, Y.; Zhang, W.; Zhang, H. iProbiotics: A Machine Learning Platform for Rapid Identification of Probiotic Properties from Whole-Genome Primary Sequences. Brief. Bioinform. 2022, 23, bbab477. [Google Scholar] [CrossRef]
- Westfall, S.; Carracci, F.; Estill, M.; Zhao, D.; Wu, Q.; Shen, L.; Simon, J.; Pasinetti, G.M. Optimization of Probiotic Therapeutics Using Machine Learning in an Artificial Human Gastrointestinal Tract. Sci. Rep. 2021, 11, 1067. [Google Scholar] [CrossRef]
- Falagas, M.E.; Pitsouni, E.I.; Malietzis, G.A.; Pappas, G. Comparison of PubMed, Scopus, Web of Science, and Google Scholar: Strengths and Weaknesses. FASEB J. 2008, 22, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Charlesworth, M.; Elrakhawy, M. How to Write a Narrative Review. Anaesthesia 2023, 78, 1162–1166. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.J.; Sawa, A.; Mortensen, P.B. Schizophrenia. Lancet 2016, 388, 86–97. [Google Scholar] [CrossRef]
- McCutcheon, R.A.; Reis Marques, T.; Howes, O.D. Schizophrenia—An Overview. JAMA Psychiatry 2020, 77, 201. [Google Scholar] [CrossRef]
- Orsolini, L.; Pompili, S.; Volpe, U. Schizophrenia: A Narrative Review of Etiopathogenetic, Diagnostic and Treatment Aspects. J. Clin. Med. 2022, 11, 5040. [Google Scholar] [CrossRef]
- Jauhar, S.; Johnstone, M.; McKenna, P.J. Schizophrenia. Lancet 2022, 399, 473–486. [Google Scholar] [CrossRef]
- Wawrzczak-Bargieła, A.; Bilecki, W.; Maćkowiak, M. Epigenetic Targets in Schizophrenia Development and Therapy. Brain Sci. 2023, 13, 426. [Google Scholar] [CrossRef] [PubMed]
- Richetto, J.; Meyer, U. Epigenetic Modifications in Schizophrenia and Related Disorders: Molecular Scars of Environmental Exposures and Source of Phenotypic Variability. Biol. Psychiatry 2021, 89, 215–226. [Google Scholar] [CrossRef]
- Charlson, F.J.; Ferrari, A.J.; Santomauro, D.F.; Diminic, S.; Stockings, E.; Scott, J.G.; McGrath, J.J.; Whiteford, H.A. Global Epidemiology and Burden of Schizophrenia: Findings from the Global Burden of Disease Study 2016. Schizophr. Bull. 2018, 44, 1195–1203. [Google Scholar] [CrossRef]
- Stilo, S.A.; Murray, R.M. The Epidemology of Schizophrenia: Replacing Dogma with Knowledge. Dialogues Clin. Neurosci. 2010, 12, 305–315. [Google Scholar] [CrossRef]
- Solmi, M.; Seitidis, G.; Mavridis, D.; Correll, C.U.; Dragioti, E.; Guimond, S.; Tuominen, L.; Dargél, A.; Carvalho, A.F.; Fornaro, M.; et al. Incidence, Prevalence, and Global Burden of Schizophrenia—Data, with Critical Appraisal, from the Global Burden of Disease (GBD) 2019. Mol. Psychiatry 2023. [Google Scholar] [CrossRef] [PubMed]
- Orrico-Sánchez, A.; López-Lacort, M.; Muñoz-Quiles, C.; Sanfélix-Gimeno, G.; Díez-Domingo, J. Epidemiology of Schizophrenia and Its Management over 8-Years Period Using Real-World Data in Spain. BMC Psychiatry 2020, 20, 149. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.H.; Valencia, G.; Pérez, J.M. Evolution in Schizophrenia Diagnosis in the Department of Caldas, Colombia. 2010–2015. Rev. Fac. Med. 2021, 69, e73159. [Google Scholar] [CrossRef]
- Carteri, R.B.; Oses, J.P.; de Cardoso, T.A.; Moreira, F.P.; Jansen, K.; da Silva, R.A. A Closer Look at the Epidemiology of Schizophrenia and Common Mental Disorders in Brazil. Dement. Neuropsychol. 2020, 14, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Golofast, B.; Vales, K. The Connection between Microbiome and Schizophrenia. Neurosci. Biobehav. Rev. 2020, 108, 712–731. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.; Keating, D.; Young, R.; Wong, M.-L.; Licinio, J.; Wesselingh, S. From Gut Dysbiosis to Altered Brain Function and Mental Illness: Mechanisms and Pathways. Mol. Psychiatry 2016, 21, 738–748. [Google Scholar] [CrossRef]
- Akbarian, S. Epigenetic Mechanisms in Schizophrenia. Dialogues Clin. Neurosci. 2014, 16, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Juckel, G.; Freund, N. Microglia and Microbiome in Schizophrenia: Can Immunomodulation Improve Symptoms? J. Neural Transm. 2023, 130, 1187–1193. [Google Scholar] [CrossRef] [PubMed]
- Socała, K.; Doboszewska, U.; Szopa, A.; Serefko, A.; Włodarczyk, M.; Zielińska, A.; Poleszak, E.; Fichna, J.; Wlaź, P. The Role of Microbiota-Gut-Brain Axis in Neuropsychiatric and Neurological Disorders. Pharmacol. Res. 2021, 172, 105840. [Google Scholar] [CrossRef]
- Szeligowski, T.; Yun, A.L.; Lennox, B.R.; Burnet, P.W.J. The Gut Microbiome and Schizophrenia: The Current State of the Field and Clinical Applications. Front. Psychiatry 2020, 11, 156. [Google Scholar] [CrossRef]
- Müller, N.; Weidinger, E.; Leitner, B.; Schwarz, M.J. The Role of Inflammation in Schizophrenia. Front. Neurosci. 2015, 9, 372. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Stogios, N.; Smith, E.; Lee, J.; Maksyutynsk, K.; Au, E.; Wright, D.C.; De Palma, G.; Graff-Guerrero, A.; Gerretsen, P.; et al. Gut Microbiome in Schizophrenia and Antipsychotic-Induced Metabolic Alterations: A Scoping Review. Ther. Adv. Psychopharmacol. 2022, 12, 204512532210965. [Google Scholar] [CrossRef] [PubMed]
- Góralczyk-Bińkowska, A.; Szmajda-Krygier, D.; Kozłowska, E. The Microbiota–Gut–Brain Axis in Psychiatric Disorders. Int. J. Mol. Sci. 2022, 23, 11245. [Google Scholar] [CrossRef] [PubMed]
- Meyer, U.; Schwarz, M.J.; Müller, N. Inflammatory Processes in Schizophrenia: A Promising Neuroimmunological Target for the Treatment of Negative/Cognitive Symptoms and Beyond. Pharmacol. Ther. 2011, 132, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Mongan, D.; Ramesar, M.; Föcking, M.; Cannon, M.; Cotter, D. Role of Inflammation in the Pathogenesis of Schizophrenia: A Review of the Evidence, Proposed Mechanisms and Implications for Treatment. Early Interv. Psychiatry 2020, 14, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Khandaker, G.M.; Meyer, U.; Jones, P.B. (Eds.) Neuroinflammation and Schizophrenia; Current Topics in Behavioral Neurosciences; Springer International Publishing: Cham, Switzerland, 2020; Volume 44, ISBN 978-3-030-39140-9. [Google Scholar]
- Williams, J.A.; Burgess, S.; Suckling, J.; Lalousis, P.A.; Batool, F.; Griffiths, S.L.; Palmer, E.; Karwath, A.; Barsky, A.; Gkoutos, G.V.; et al. Inflammation and Brain Structure in Schizophrenia and Other Neuropsychiatric Disorders: A Mendelian Randomization Study. JAMA Psychiatry 2022, 79, 498. [Google Scholar] [CrossRef] [PubMed]
- Anand, N.; Gorantla, V.R.; Chidambaram, S.B. The Role of Gut Dysbiosis in the Pathophysiology of Neuropsychiatric Disorders. Cells 2023, 12, 54. [Google Scholar] [CrossRef]
- Kelly, J.R.; Minuto, C.; Cryan, J.F.; Clarke, G.; Dinan, T.G. The Role of the Gut Microbiome in the Development of Schizophrenia. Schizophr. Res. 2021, 234, 4–23. [Google Scholar] [CrossRef] [PubMed]
- Lisoway, A.J.; Chen, C.C.; Zai, C.C.; Tiwari, A.K.; Kennedy, J.L. Toward Personalized Medicine in Schizophrenia: Genetics and Epigenetics of Antipsychotic Treatment. Schizophr. Res. 2021, 232, 112–124. [Google Scholar] [CrossRef]
- Liu, J.C.W.; Gorbovskaya, I.; Hahn, M.K.; Müller, D.J. The Gut Microbiome in Schizophrenia and the Potential Benefits of Prebiotic and Probiotic Treatment. Nutrients 2021, 13, 1152. [Google Scholar] [CrossRef]
- Grover, S.; Patil, A.; Kaur, A.; Garg, G. Probiotics: A Potential Immunotherapeutic Approach for the Treatment of Schizophrenia. J. Pharm. Bioallied Sci. 2019, 11, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S. The Environment and Susceptibility to Schizophrenia. Prog. Neurobiol. 2011, 93, 23–58. [Google Scholar] [CrossRef]
- Attademo, L.; Bernardini, F.; Garinella, R.; Compton, M.T. Environmental Pollution and Risk of Psychotic Disorders: A Review of the Science to Date. Schizophr. Res. 2017, 181, 55–59. [Google Scholar] [CrossRef]
- Minichino, A.; Brondino, N.; Solmi, M.; Del Giovane, C.; Fusar-Poli, P.; Burnet, P.; Cipriani, A.; Lennox, B.R. The Gut-Microbiome as a Target for the Treatment of Schizophrenia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials of Add-on Strategies. Schizophr. Res. 2021, 234, 58–70. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, J.; Wu, L.; Luo, J.; Liang, X.; Xiao, B.; Zhu, Y. The Impacts of Delivery Mode on Infant’s Oral Microflora. Sci. Rep. 2018, 8, 11938. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.M.; Mazzoni, C.; Hogstrom, L.; Bryant, A.; Bergerat, A.; Cher, A.; Pochan, S.; Herman, P.; Carrigan, M.; Sharp, K.; et al. Delivery Mode Affects Stability of Early Infant Gut Microbiota. Cell Rep. Med. 2020, 1, 100156. [Google Scholar] [CrossRef]
- Almand, A.T.; Anderson, A.P.; Hitt, B.D.; Sitko, J.C.; Joy, R.M.; Easter, B.D.; Almand, E.A. The Influence of Perceived Stress on the Human Microbiome. BMC Res. Notes 2022, 15, 193. [Google Scholar] [CrossRef]
- Lotti, S.; Dinu, M.; Colombini, B.; Amedei, A.; Sofi, F. Circadian Rhythms, Gut Microbiota, and Diet: Possible Implications for Health. Nutr. Metab. Cardiovasc. Dis. 2023, 33, 1490–1500. [Google Scholar] [CrossRef]
- Seel, W.; Reiners, S.; Kipp, K.; Simon, M.-C.; Dawczynski, C. Role of Dietary Fiber and Energy Intake on Gut Microbiome in Vegans, Vegetarians, and Flexitarians in Comparison to Omnivores—Insights from the Nutritional Evaluation (NuEva) Study. Nutrients 2023, 15, 1914. [Google Scholar] [CrossRef] [PubMed]
- Tsamakis, K.; Galinaki, S.; Alevyzakis, E.; Hortis, I.; Tsiptsios, D.; Kollintza, E.; Kympouropoulos, S.; Triantafyllou, K.; Smyrnis, N.; Rizos, E. Gut Microbiome: A Brief Review on Its Role in Schizophrenia and First Episode of Psychosis. Microorganisms 2022, 10, 1121. [Google Scholar] [CrossRef]
- Generoso, J.S.; Giridharan, V.V.; Lee, J.; Macedo, D.; Barichello, T. The Role of the Microbiota-Gut-Brain Axis in Neuropsychiatric Disorders. Braz. J. Psychiatry 2021, 43, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Borre, Y.E.; Cryan, J.F. Genomics of Schizophrenia: Time to Consider the Gut Microbiome? Mol. Psychiatry 2014, 19, 1252–1257. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, F.; Severance, E.; Yolken, R. The Microbiome, Immunity, and Schizophrenia and Bipolar Disorder. Brain Behav. Immun. 2017, 62, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Skowron, K.; Budzyńska, A.; Wiktorczyk-Kapischke, N.; Chomacka, K.; Grudlewska-Buda, K.; Wilk, M.; Wałecka-Zacharska, E.; Andrzejewska, M.; Gospodarek-Komkowska, E. The Role of Psychobiotics in Supporting the Treatment of Disturbances in the Functioning of the Nervous System—A Systematic Review. Int. J. Mol. Sci. 2022, 23, 7820. [Google Scholar] [CrossRef] [PubMed]
- Najjar, S.; Pahlajani, S.; De Sanctis, V.; Stern, J.N.H.; Najjar, A.; Chong, D. Neurovascular Unit Dysfunction and Blood–Brain Barrier Hyperpermeability Contribute to Schizophrenia Neurobiology: A Theoretical Integration of Clinical and Experimental Evidence. Front. Psychiatry 2017, 8, 83. [Google Scholar] [CrossRef]
- Maurus, I.; Wagner, S.; Campana, M.; Roell, L.; Strauss, J.; Fernando, P.; Muenz, S.; Eichhorn, P.; Schmitt, A.; Karch, S.; et al. The Relationship between Blood–Brain Barrier Dysfunction and Neurocognitive Impairments in First-Episode Psychosis: Findings from a Retrospective Chart Analysis. BJPsych Open 2023, 9, e60. [Google Scholar] [CrossRef]
- Lima Giacobbo, B.; Doorduin, J.; Klein, H.C.; Dierckx, R.A.J.O.; Bromberg, E.; De Vries, E.F.J. Brain-Derived Neurotrophic Factor in Brain Disorders: Focus on Neuroinflammation. Mol. Neurobiol. 2019, 56, 3295–3312. [Google Scholar] [CrossRef]
- Porter, G.A.; O’Connor, J.C. Brain-Derived Neurotrophic Factor and Inflammation in Depression: Pathogenic Partners in Crime? World J. Psychiatry 2022, 12, 77–97. [Google Scholar] [CrossRef]
- Gören, J.L. Brain-Derived Neurotrophic Factor and Schizophrenia. Ment. Health Clin. 2016, 6, 285–288. [Google Scholar] [CrossRef]
- Sandrini, L.; Di Minno, A.; Amadio, P.; Ieraci, A.; Tremoli, E.; Barbieri, S. Association between Obesity and Circulating Brain-Derived Neurotrophic Factor (BDNF) Levels: Systematic Review of Literature and Meta-Analysis. Int. J. Mol. Sci. 2018, 19, 2281. [Google Scholar] [CrossRef]
- Katuri, R.B.; Gaur, G.S.; Sahoo, J.P.; Bobby, Z.; Shanmugavel, K. Association of Circulating Brain-Derived Neurotrophic Factor with Cognition among Adult Obese Population. J. Obes. Metab. Syndr. 2021, 30, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Dhanavade, M.J.; Sonawane, K.D. Amyloid Beta Peptide-Degrading Microbial Enzymes and Its Implication in Drug Design. 3 Biotech 2020, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Sedov, I.; Khaibrakhmanova, D. Molecular Mechanisms of Inhibition of Protein Amyloid Fibril Formation: Evidence and Perspectives Based on Kinetic Models. Int. J. Mol. Sci. 2022, 23, 13428. [Google Scholar] [CrossRef] [PubMed]
- Kishor, K.; Azmeen, F.; Jha, P. Role of Nano Probiotic as Potential Alternative Therapy Source for Oral Health. J. Surv. Fish. Sci. 2023, 10, 3277–3279. [Google Scholar]
- Seeman, M.V. The Gut Microbiome and Treatment-Resistance in Schizophrenia. Psychiatr. Q. 2020, 91, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Daliri, E.B.-M.; Ofosu, F.K.; Xiuqin, C.; Chelliah, R.; Oh, D.-H. Probiotic Effector Compounds: Current Knowledge and Future Perspectives. Front. Microbiol. 2021, 12, 655705. [Google Scholar] [CrossRef] [PubMed]
- Magalhães-Guedes, K.T. Psychobiotic Therapy: Method to Reinforce the Immune System. Clin. Psychopharmacol. Neurosci. 2022, 20, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Jagtiani, E.; Adsare, S. Microencapulsation: Probiotics, Prebiotics, and Nutraceuticals. J. Nanotechnol. Nanomater. 2022, 3, 34–60. [Google Scholar] [CrossRef]
- Ross, K. Psychobiotics: Are They the Future Intervention for Managing Depression and Anxiety? A Literature Review. Explore 2023, 19, 669–680. [Google Scholar] [CrossRef]
- Tomasik, J.; Yolken, R.H.; Bahn, S.; Dickerson, F.B. Immunomodulatory Effects of Probiotic Supplementation in Schizophrenia Patients: A Randomized, Placebo-Controlled Trial. BiomarkInsights 2015, 10, BMI.S22007. [Google Scholar] [CrossRef]
- Perez-Burgos, A.; Wang, B.; Mao, Y.-K.; Mistry, B.; Neufeld, K.-A.M.; Bienenstock, J.; Kunze, W. Psychoactive Bacteria Lactobacillus Rhamnosus (JB-1) Elicits Rapid Frequency Facilitation in Vagal Afferents. Am. J. Physiol.-Gastrointest. Liver Physiol. 2013, 304, G211–G220. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.M.; Jenks, S.M. Ingestion of Mycobacterium Vaccae Decreases Anxiety-Related Behavior and Improves Learning in Mice. Behav. Process. 2013, 96, 27–35. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Wang, M.; Zheng, L.; Cen, Q.; Wang, F.; Zhu, L.; Pang, R.; Zhang, A. Bifidobacterium: A Probiotic for the Prevention and Treatment of Depression. Front. Microbiol. 2023, 14, 1174800. [Google Scholar] [CrossRef]
- Nocera, A.; Nasrallah, H. The Association of the Gut Microbiota with Clinical Features in Schizophrenia. Behav. Sci. 2022, 12, 89. [Google Scholar] [CrossRef]
- Thirion, F.; Speyer, H.; Hansen, T.H.; Nielsen, T.; Fan, Y.; Le Chatelier, E.; Fromentin, S.; Berland, M.; Plaza Oñate, F.; Pons, N.; et al. Alteration of Gut Microbiome in Patients with Schizophrenia Indicates Links Between Bacterial Tyrosine Biosynthesis and Cognitive Dysfunction. Biol. Psychiatry Glob. Open Sci. 2023, 3, 283–291. [Google Scholar] [CrossRef]
- Xu, R.; Wu, B.; Liang, J.; He, F.; Gu, W.; Li, K.; Luo, Y.; Chen, J.; Gao, Y.; Wu, Z.; et al. Altered Gut Microbiota and Mucosal Immunity in Patients with Schizophrenia. Brain Behav. Immun. 2020, 85, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Song, J.; Ke, P.; Kong, L.; Lei, B.; Zhou, J.; Huang, Y.; Li, H.; Li, G.; Chen, J.; et al. The Gut Microbiome Is Associated with Brain Structure and Function in Schizophrenia. Sci. Rep. 2021, 11, 9743. [Google Scholar] [CrossRef]
- Yang, C.; Lin, X.; Wang, X.; Liu, H.; Huang, J.; Wang, S. The Schizophrenia and Gut Microbiota: A Bibliometric and Visual Analysis. Front. Psychiatry 2022, 13, 1022472. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Xu, H.; Chai, C.; Qin, Z.; Zhou, W. Integrated Bioinformatics Analysis of Potential Biomarkers for Pancreatic Cancer. Clin. Lab. Anal. 2022, 36, e24381. [Google Scholar] [CrossRef]
- Nita, I.-B.; Ilie, O.-D.; Ciobica, A.; Hritcu, L.-D.; Dobrin, I.; Doroftei, B.; Dobrin, R. Reviewing the Potential Therapeutic Approaches Targeting the Modulation of Gastrointestinal Microflora in Schizophrenia. Int. J. Mol. Sci. 2022, 23, 16129. [Google Scholar] [CrossRef]
- Minervini, G.; Franco, R.; Marrapodi, M.M.; Fiorillo, L.; Badnjević, A.; Cervino, G.; Cicciù, M. Probiotics in the Treatment of Radiotherapy-Induced Oral Mucositis: Systematic Review with Meta-Analysis. Pharmaceuticals 2023, 16, 654. [Google Scholar] [CrossRef] [PubMed]
- McGuinness, A.J.; Davis, J.A.; Dawson, S.L.; Loughman, A.; Collier, F.; O’Hely, M.; Simpson, C.A.; Green, J.; Marx, W.; Hair, C.; et al. A Systematic Review of Gut Microbiota Composition in Observational Studies of Major Depressive Disorder, Bipolar Disorder and Schizophrenia. Mol. Psychiatry 2022, 27, 1920–1935. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Kosciolek, T.; Tang, J.; Zhou, Y.; Li, Z.; Ma, X.; Zhu, Q.; Yuan, N.; Yuan, L.; Li, C.; et al. Gut Microbiome and Magnetic Resonance Spectroscopy Study of Subjects at Ultra-High Risk for Psychosis May Support the Membrane Hypothesis. Eur. Psychiatry 2018, 53, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Castro-Nallar, E.; Bendall, M.L.; Pérez-Losada, M.; Sabuncyan, S.; Severance, E.G.; Dickerson, F.B.; Schroeder, J.R.; Yolken, R.H.; Crandall, K.A. Composition, Taxonomy and Functional Diversity of the Oropharynx Microbiome in Individuals with Schizophrenia and Controls. PeerJ 2015, 3, e1140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Pan, L.-Y.; Zhang, Z.; Zhou, Y.-Y.; Jiang, H.-Y.; Ruan, B. Analysis of Gut Mycobiota in First-Episode, Drug-Naïve Chinese Patients with Schizophrenia: A Pilot Study. Behav. Brain Res. 2020, 379, 112374. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, P.; Wang, Y.; Liu, Y.; Li, X.; Kumar, B.U.; Hei, G.; Lv, L.; Huang, X.-F.; Fan, X.; et al. Changes in Metabolism and Microbiota after 24-Week Risperidone Treatment in Drug Naïve, Normal Weight Patients with First Episode Schizophrenia. Schizophr. Res. 2018, 201, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Pełka-Wysiecka, J.; Kaczmarczyk, M.; Bąba-Kubiś, A.; Liśkiewicz, P.; Wroński, M.; Skonieczna-Żydecka, K.; Marlicz, W.; Misiak, B.; Starzyńska, T.; Kucharska-Mazur, J.; et al. Analysis of Gut Microbiota and Their Metabolic Potential in Patients with Schizophrenia Treated with Olanzapine: Results from a Six-Week Observational Prospective Cohort Study. J. Clin. Med. 2019, 8, 1605. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Xu, J.; Li, Z.; Huang, Y.; Yuan, Y.; Wang, J.; Zhang, M.; Hu, S.; Liang, Y. Analysis of Gut Microbiota Diversity and Auxiliary Diagnosis as a Biomarker in Patients with Schizophrenia: A Cross-Sectional Study. Schizophr. Res. 2018, 197, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Kosciolek, T.; Maldonado, Y.; Daly, R.E.; Martin, A.S.; McDonald, D.; Knight, R.; Jeste, D.V. Differences in Gut Microbiome Composition between Persons with Chronic Schizophrenia and Healthy Comparison Subjects. Schizophr. Res. 2019, 204, 23–29. [Google Scholar] [CrossRef]
- Ma, X.; Asif, H.; Dai, L.; He, Y.; Zheng, W.; Wang, D.; Ren, H.; Tang, J.; Li, C.; Jin, K.; et al. Alteration of the Gut Microbiome in First-Episode Drug-Naïve and Chronic Medicated Schizophrenia Correlate with Regional Brain Volumes. J. Psychiatr. Res. 2020, 123, 136–144. [Google Scholar] [CrossRef]
- Vindegaard, N.; Speyer, H.; Nordentoft, M.; Rasmussen, S.; Benros, M.E. Gut Microbial Changes of Patients with Psychotic and Affective Disorders: A Systematic Review. Schizophr. Res. 2021, 234, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zagórska, A.; Marcinkowska, M.; Jamrozik, M.; Wiśniowska, B.; Paśko, P. From Probiotics to Psychobiotics—The Gut-Brain Axis in Psychiatric Disorders. Benef. Microbes 2020, 11, 717–732. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sinha, R.; Shukla, P. Artificial Intelligence and Synthetic Biology Approaches for Human Gut Microbiome. Crit. Rev. Food Sci. Nutr. 2022, 62, 2103–2121. [Google Scholar] [CrossRef] [PubMed]
- Laterza, L.; Putignani, L.; Settanni, C.R.; Petito, V.; Varca, S.; De Maio, F.; Macari, G.; Guarrasi, V.; Gremese, E.; Tolusso, B.; et al. Ecology and Machine Learning-Based Classification Models of Gut Microbiota and Inflammatory Markers May Evaluate the Effects of Probiotic Supplementation in Patients Recently Recovered from COVID-19. Int. J. Mol. Sci. 2023, 24, 6623. [Google Scholar] [CrossRef] [PubMed]
- Benner, S.A.; Sismour, A.M. Synthetic Biology. Nat. Rev. Genet. 2005, 6, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Restrepo, J.C.; Dueñas, D.; Corredor, Z.; Liscano, Y. Advances in Genomic Data and Biomarkers: Revolutionizing NSCLC Diagnosis and Treatment. Cancers 2023, 15, 3474. [Google Scholar] [CrossRef] [PubMed]
- Trejos, M.; Aristizabal, Y.; Aragón-Muriel, A.; Oñate-Garzón, J.; Liscano, Y. Characterization and Classification In Silico of Peptides with Dual Activity (Antimicrobial and Wound Healing). Int. J. Mol. Sci. 2023, 24, 13091. [Google Scholar] [CrossRef] [PubMed]
- Doudna, J.A.; Charpentier, E. The New Frontier of Genome Engineering with CRISPR-Cas9. Science 2014, 346, 1258096. [Google Scholar] [CrossRef] [PubMed]
- Forth, E.; Buehner, B.; Storer, A.; Sgarbossa, C.; Milev, R.; Chinna Meyyappan, A. Systematic Review of Probiotics as an Adjuvant Treatment for Psychiatric Disorders. Front. Behav. Neurosci. 2023, 17, 1111349. [Google Scholar] [CrossRef]
- Mittal, K.R.; Pharasi, N.; Sarna, B.; Singh, M.; Rachana; Haider, S.; Singh, S.K.; Dua, K.; Jha, S.K.; Dey, A.; et al. Nanotechnology-Based Drug Delivery for the Treatment of CNS Disorders. Transl. Neurosci. 2022, 13, 527–546. [Google Scholar] [CrossRef]
- Naqvi, S.; Panghal, A.; Flora, S.J.S. Nanotechnology: A Promising Approach for Delivery of Neuroprotective Drugs. Front. Neurosci. 2020, 14, 494. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.P.; Gunjan; Himanshu; Mukherjee, R.; Chang, C.-M. Nanocarrier-Mediated Probiotic Delivery: A Systematic Meta-Analysis Assessing the Biological Effects. Sci. Rep. 2024, 14, 631. [Google Scholar] [CrossRef] [PubMed]
- Ciont, C.; Mesaroș, A.; Pop, O.L.; Vodnar, D.C. Iron Oxide Nanoparticles Carried by Probiotics for Iron Absorption: A Systematic Review. J. Nanobiotechnol 2023, 21, 124. [Google Scholar] [CrossRef] [PubMed]
- Jalil, A.T.; Thabit, S.N.; Hanan, Z.K.; Alasheqi, M.Q.; Al-Azzawi, A.K.J.; Zabibah, R.S.; Fadhil, A.A. Modulating Gut Microbiota Using Nanotechnology to Increase Anticancer Efficacy of the Treatments. Macromol. Res. 2023, 31, 739–752. [Google Scholar] [CrossRef]
- Posadas, I.; Monteagudo, S.; Ceña, V. Nanoparticles for Brain-Specific Drug and Genetic Material Delivery, Imaging and Diagnosis. Nanomedicine 2016, 11, 833–849. [Google Scholar] [CrossRef] [PubMed]
- Asha Spandana, K.M.; Bhaskaran, M.; Karri, V.V.S.N.R.; Natarajan, J. A Comprehensive Review of Nano Drug Delivery System in the Treatment of CNS Disorders. J. Drug Deliv. Sci. Technol. 2020, 57, 101628. [Google Scholar] [CrossRef]
- Pimienta, D.A.; Cruz Mosquera, F.E.; Palacios Velasco, I.; Giraldo Rodas, M.; Oñate-Garzón, J.; Liscano, Y. Specific Focus on Antifungal Peptides against Azole Resistant Aspergillus Fumigatus: Current Status, Challenges, and Future Perspectives. JOF J. Fungi 2022, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Chao, J.; Liu, H.; Su, S.; Wang, L.; Huang, W.; Fan, C. Structural DNA Nanotechnology for Intelligent Drug Delivery. Small 2014, 10, 4626–4635. [Google Scholar] [CrossRef] [PubMed]
- Endo, M.; Sugiyama, H. DNA Origami Nanomachines. Molecules 2018, 23, 1766. [Google Scholar] [CrossRef]
- Rajendran, A.; Endo, M.; Sugiyama, H. Single-Molecule Analysis Using DNA Origami. Angew. Chem. Int. Ed. 2012, 51, 874–890. [Google Scholar] [CrossRef]
- Shishparenok, A.N.; Furman, V.V.; Zhdanov, D.D. DNA-Based Nanomaterials as Drug Delivery Platforms for Increasing the Effect of Drugs in Tumors. Cancers 2023, 15, 2151. [Google Scholar] [CrossRef] [PubMed]
- Sangnim, T.; Puri, V.; Dheer, D.; Venkatesh, D.N.; Huanbutta, K.; Sharma, A. Nanomaterials in the Wound Healing Process: New Insights and Advancements. Pharmaceutics 2024, 16, 300. [Google Scholar] [CrossRef] [PubMed]
- Janik, M.; Hanula, M.; Khachatryan, K.; Khachatryan, G. Nano-/Microcapsules, Liposomes, and Micelles in Polysaccharide Carriers: Applications in Food Technology. Appl. Sci. 2023, 13, 11610. [Google Scholar] [CrossRef]
- Wu, Y.; Yang, G.; Van Der Mei, H.C.; Shi, L.; Busscher, H.J.; Ren, Y. Synergy between “Probiotic” Carbon Quantum Dots and Ciprofloxacin in Eradicating Infectious Biofilms and Their Biosafety in Mice. Pharmaceutics 2021, 13, 1809. [Google Scholar] [CrossRef] [PubMed]

| Factor | Impact on Gut Microbiota |
|---|---|
| Mode of Delivery [50,57,58] | Influences the initial colonization of a newborn’s GI tract. Vaginal delivery (VD) leads to a microbiota similar to maternal vaginal microbiota with a dominance of Lactobacillus spp. Cesarean section (CS) results in decreased diversity and an imbalance, with infants showing higher abundance of hospital pathogens and lower abundance of Bifidobacteria spp., Bacteroides spp., Staphylococcus spp., Corynebacterium spp., and Propionibacterium spp. |
| Probiotics [50] | Regulates the immune system, supports gut barrier integrity, and has been shown to alleviate symptoms of depression, anxiety, and stress. Specific strains like Lactobacillus helveticus R0052 and B. longum R0175 have shown benefits in mental health. |
| Stress [50,59] | Stress can decrease the number of beneficial species like Lactobacillus spp. and Bifidobacterium spp. while increasing pathogenic and non-pathogenic strains of E. coli and species from the genus Clostridium spp. |
| Circadian Clock System [50,60] | The circadian rhythm affects the diurnal fluctuations of GM. Stress and changes in the circadian clock system can cause dysregulation of the intestinal microbiota, leading to decreases in Lactobacillus and increases in pathogenic bacteria. |
| Occupational and Environmental Exposure [50] | Occupational and environmental pollutants, including heavy metals, pesticides, and PAHs can modify GM composition. Shift work and exposure to specific work environments can alter the microbiota, indicating potential health risks. |
| Diet [50,61] | Diet influences GM diversity and abundance, affecting metabolism and immune responses. Dietary fibers are fermented by GM to produce SCFAs, beneficial for colon health. Variations in diet, such as Mediterranean, ketogenic, vegetarian, or vegan diets, have significant impacts on GM composition. |
| Psychobiotic | Effect on Schizophrenia | Potential Mechanism |
|---|---|---|
| Lactobacillus rhamnosus (JB-1) [83] | Reduced stress-induced corticosterone levels; decreased depressive behavior | Downregulation of HPA axis activity; alteration in GABA receptor expression |
| Mycobacterium vaccae [7,84] | Reduced anxiety in maze-learning tasks | Not specified |
| Bifidobacteria infantis [7,85] | Increased tryptophan and serotonin levels; decreased pro-inflammatory cytokines | Immunomodulatory effects; modulation of tryptophan metabolism |
| Lactobacillus helveticus NS8 [7] | Lowered post-restraint anxiety; enhanced memory; reduced corticosterone and ACTH levels | Anti-inflammatory effects; increase in hippocampal BDNF mRNA |
| Lactobacillus rhamnosus JB-1 (additional study) [7] | Elevated concentrations of glutamate, GABA, and tNAA, indicating changes in neural metabolism | Modulation of neurotransmitter concentrations, particularly in excitatory and inhibitory balance |
| General Probiotics [7] | Observations of reduced gastrointestinal inflammation, immune activation, and modulation of physiological variables including inflammatory markers | Anti-inflammatory properties; vagus nerve stimulation; cytokine modulation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mosquera, F.E.C.; Guevara-Montoya, M.C.; Serna-Ramirez, V.; Liscano, Y. Neuroinflammation and Schizophrenia: New Therapeutic Strategies through Psychobiotics, Nanotechnology, and Artificial Intelligence (AI). J. Pers. Med. 2024, 14, 391. https://doi.org/10.3390/jpm14040391
Mosquera FEC, Guevara-Montoya MC, Serna-Ramirez V, Liscano Y. Neuroinflammation and Schizophrenia: New Therapeutic Strategies through Psychobiotics, Nanotechnology, and Artificial Intelligence (AI). Journal of Personalized Medicine. 2024; 14(4):391. https://doi.org/10.3390/jpm14040391
Chicago/Turabian StyleMosquera, Freiser Eceomo Cruz, Maria Camila Guevara-Montoya, Valentina Serna-Ramirez, and Yamil Liscano. 2024. "Neuroinflammation and Schizophrenia: New Therapeutic Strategies through Psychobiotics, Nanotechnology, and Artificial Intelligence (AI)" Journal of Personalized Medicine 14, no. 4: 391. https://doi.org/10.3390/jpm14040391
APA StyleMosquera, F. E. C., Guevara-Montoya, M. C., Serna-Ramirez, V., & Liscano, Y. (2024). Neuroinflammation and Schizophrenia: New Therapeutic Strategies through Psychobiotics, Nanotechnology, and Artificial Intelligence (AI). Journal of Personalized Medicine, 14(4), 391. https://doi.org/10.3390/jpm14040391






