Volatile Organic Compounds from Entomopathogenic and Nematophagous Fungi, Repel Banana Black Weevil (Cosmopolites sordidus)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Isolation from Banana Plantations Soils using Insect Baits
2.2. Morphological Identification of EF Isolates
2.3. Molecular Identification of Fungal Strains
2.4. Pathogenicity Assays
2.5. Gas Chromatography—Mass Spectrometry (GC/MS) Analysis
2.6. Olfactometer Bioassays
2.7. Environmental Conditions
2.8. Fungal VOCs Repellency
2.9. Pheromone and Fungal VOCs Attractiveness
2.10. Olfactometer Bioassays Data Treatment for Analysis
3. Results
3.1. Entomopathogenic Fungi are Present in Soil from Banana Plantations
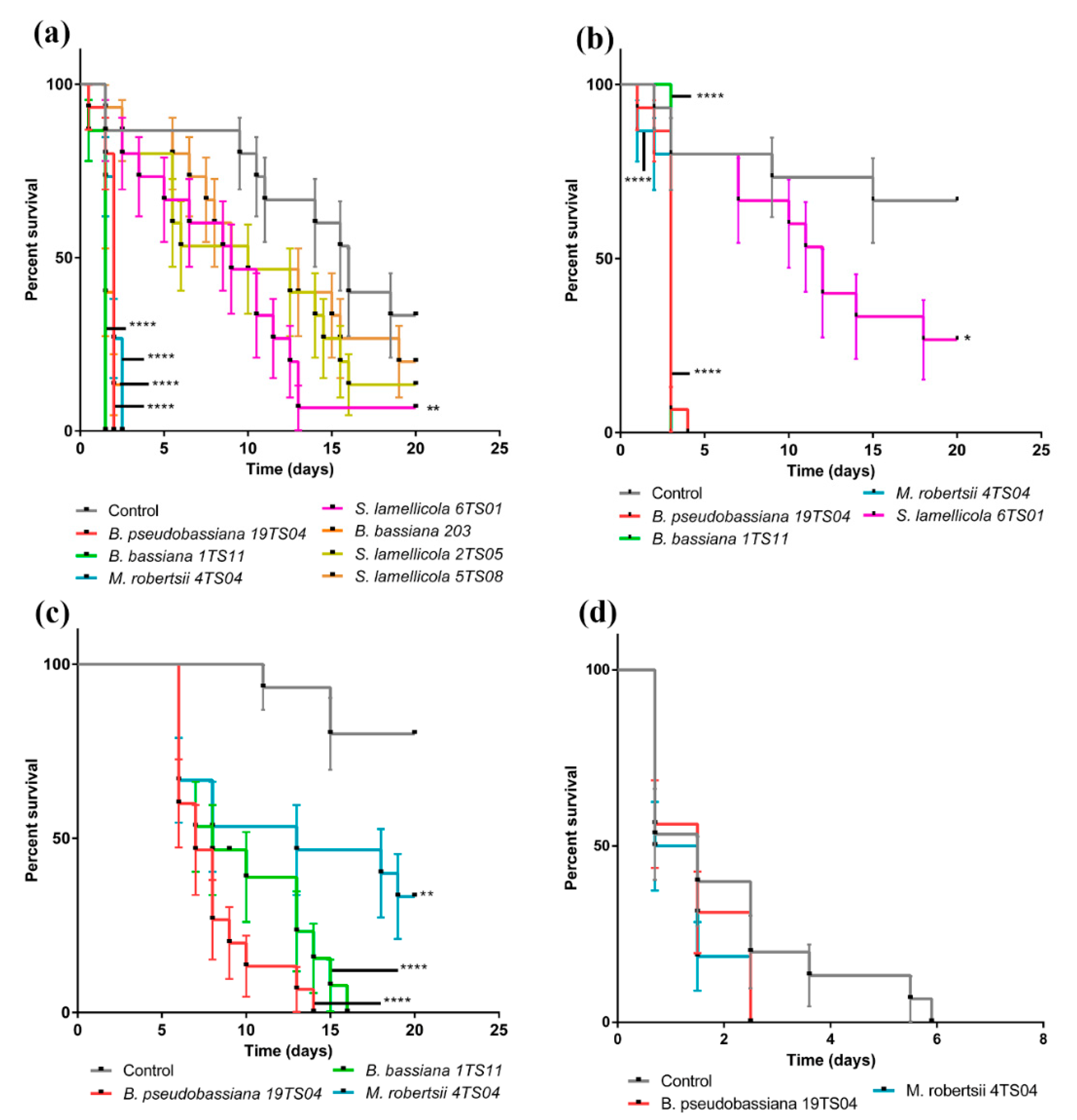
3.2. Beauveria spp. from Banana Soils Are More Virulent than Metarhizium spp. to C. Sordidus
3.3. VOCs Production by Fungal Pathogens of Invertebrates
3.4. Olfactometer Bioassays
3.4.1. Effects of Environmental Conditions on BW Mobility
3.4.2. Fungal VOCs Repel BW
3.4.3. Fungal VOCs Mask Pheromone and Banana Corm BW Attractiveness
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ostmark, H.E. Economic insect pests of bananas. Annu. Rev. Entomol. 1974, 19, 161–176. [Google Scholar] [CrossRef]
- Kennett, C.E.; Mcmurtry, J.A.; Beardsley, J.W. Biological Control in Subtropical and Tropical Crops. Handb. Biol. Control 1999, 1892, 713–742. [Google Scholar] [CrossRef]
- Dubois, T.; Gold, C.S.; Coyne, D.; Paparu, P.; Mukwaba, E.; Athman, S.; Adipala, S.K.E. Merging biotechnology with biological control: Banana Musa tissue culture plants enhanced by endophytic fungi. Uganda J. Agric. Sci. 2004, 9, 445–451. [Google Scholar]
- Waweru, B.; Turoop, L.; Kahangi, E.; Coyne, D.; Dubois, T. Non-pathogenic Fusarium oxysporum endophytes provide field control of nematodes, improving yield of banana (Musa sp.). Biol. Control 2014, 74, 82–88. [Google Scholar] [CrossRef]
- Nankinga, C.M.; Moore, D. Reduction of banana weevil populations using different formulations of the entomopathogenic fungus Beauveria bassiana. Biocontrol Sci. Technol. 2000, 10, 645–657. [Google Scholar] [CrossRef]
- Simmonds, N.W. Bananas, 2nd ed.; Tropical Agricultural Series; Longman: London, UK, 1966; p. 512. [Google Scholar]
- Vilardebo, A. Le coefficient d’infestation, critère d’évaluation du degré d’attaques des bananeraies par Cosmopolites sordidus GERM. le charançon noir du bananier. Fruits 1973, 28, 417–426. [Google Scholar]
- Yuasa, H. Cosmopolites sordidus Germ. found in the Bonin and Loochoo Islands. Oyo Kontyu 1939, 2, 116–118. [Google Scholar]
- Carnero, H.A.; Padilla-Cubas, A.; Montesdeoca, M. Métodos alternativos para el control del picudo de la platanera Cosmopolites sordidus Germar, 1824 (Coleoptera: Curculionidae). Actividades del ICIA en Platanera 2002, 8, 75–82. [Google Scholar]
- Carballo, M. Mortalidad de Cosmopolites sordidus con diferentes formulaciones de Beauveria bassiana. Manejo Integrado de Plagas (CATIE) 1998, 48, 45–48. [Google Scholar]
- Musabyimana, T.; Saxena, R.C.; Kairu, E.W.; Ogol, C.P.K.O.; Khan, Z.R. Effects of Neem Seed Derivatives on Behavioral and Physiological Responses of the Cosmopolites sordidus (Coleoptera: Curculionidae). J. Econ. Entomol. 2001, 94, 449–454. [Google Scholar] [CrossRef]
- Muñoz Ruiz, C. Fluctuación poblacional del picudo negro (Cosmopolites sordidus Germar) del plátano (Musa AAB) en San Carlos, Costa Rica. Tecnol. Marcha 2007, 20, 24–41. [Google Scholar]
- Gold, C.S.; Pena, J.E.; Karamura, E.B. Biology and integrated pest management for the banana weevil Cosmopolites sordidus (Germar) (Coleoptera: Curculionidae). Integr. Pest Manag. Rev. 2001, 6, 79–155. [Google Scholar] [CrossRef]
- Franzmann, B.A. Banana weevil borer in north Queensland. Qld. Agric. J. 1972, 98, 319–321. [Google Scholar]
- De Faria, M.R.; Wraight, S.P. Mycoinsecticides and mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Güerri-Agulló, B.; López-Follana, R.; Asensio, L.; Barranco, P.; Lopez-Llorca, L.V. Use of a Solid Formulation of Beauveria bassiana for Biocontrol of the Red Palm Weevil (Rhynchophorus ferrugineus) (Coleoptera: Dryophthoridae) Under Field Conditions in SE Spain. Fla. Entomol. 2011, 94, 737–747. [Google Scholar] [CrossRef]
- Vega, F.; Kaya, H. Insect Pathology Second Edition; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 978-0-12-384984-7. [Google Scholar]
- Steinhaus, E.A. Disease in a Minor Chord: Being a Semihistorical and Semibiographical Account of a Period in Science When One Could Be Happily Yet Seriously Concerned with the Diseases of Lowly Animals without Backbones, Especially the Insects; The Ohio State University Press: Columbus, OH, USA, 1975; p. 488. [Google Scholar]
- Mesquita, A.L.M.; Lucchini, F.; Alves, E.J.; Caldas, R.C. Influencia dos fatores ambientais no grau de parasitismo de Beauveria bassiana sobre Cosmopolites sordidus e Metamasius hemipterus, em cultivo da bananeira. Pesquisa andamento. 1981, 14, 4. [Google Scholar]
- Larriba, E.; Jaime, M.D.L.A.; Carbonell-Caballero, J.; Conesa, A.; Dopazo, J.; Nislow, C.; Martín-Nieto, J.; Lopez-Llorca, L.V. Sequencing and functional analysis of the genome of a nematode egg-parasitic fungus, Pochonia chlamydosporia. Fungal Genet. Biol. 2014, 65, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Liu, C.; Shen, B.; Bai, M.; Ling, J.; Chen, G.; Mao, Z. Analysis of the complete mitochondrial genome of Pochonia chlamydosporia suggests a close relationship to the invertebrate-pathogenic fungi in Hypocreales. BMC Microbiol. 2015, 15, 5. [Google Scholar] [CrossRef]
- Willcox, J.; Tribe, H.T. Fungal parasitism in cysts of Heterodera. Trans. Br. Mycol. Soc. 1974, 62, 585–594. [Google Scholar] [CrossRef]
- De Leij, F.A.A.M.; Dennehy, J.A.; Kerry, B.R. The effect of temperature and nematode species on interactions between the nematophagous fungus Verticillium chlamydosporium and root-knot nematodes (Meloidogyne spp.). Nematologica 1992, 38, 65–79. [Google Scholar]
- Bourne, J.M.; Kerry, B.R.; De Leij, F.A.A.M. The importance of the host plant on the interaction between root-knot nematodes Meloidogyne spp. and the nematophagous fungus, Verticillium chlamydosporium Goddard. Biocontrol Sci. Technol. 1996, 6, 539–548. [Google Scholar] [CrossRef]
- Maciá-Vicente, J.G.; Jansson, H.B.; Talbot, N.J.; Lopez-Llorca, L.V. Real-time PCR quantification and live-cell imaging of endophytic colonization of barley (Hordeum vulgare) roots by Fusarium equiseti and Pochonia chlamydosporia. New Phytol. 2009, 182, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Escudero, N.; Lopez-Llorca, L.V. Effects on plant growth and root-knot nematode infection of an endophytic GFP transformant of the nematophagous fungus Pochonia chlamydosporia. Symbiosis 2012, 57, 33–42. [Google Scholar] [CrossRef]
- Domsch, K.H.; Gams, W.; Anderson, T.-H. Compendium of soil fungi. Eur. J. Soil Sci. 1993, 59, 1007. [Google Scholar]
- Zare, R.; Gams, W.; Evans, H.C. A revision of Verticillium section Prostrata. V. The genus Pochonia, with notes on Rotiferophthora. Nova Hedwig. 2001, 73, 51–86. [Google Scholar]
- Manzanilla-López, R.H.; Esteves, I.; Finetti-Sialer, M.M.; Hirsch, P.R.; Ward, E.; Devonshire, J.; Hidalgo-Díaz, L. Pochonia chlamydosporia: Advances and challenges to improve its performance as a biological control agent of sedentary endo-parasitic nematodes. J. Nematol. 2013, 45, 1–7. [Google Scholar]
- Pagans, E.; Font, X.; Sánchez, A. Emission of volatile organic compounds from composting of different solid wastes: Abatement by biofiltration. J. Hazard. Mater. 2006, 131, 179–186. [Google Scholar] [CrossRef]
- Campos, V.P.; Silva Canuto De Pinho, R.; Freire, E.S. Volatiles produced by interacting microorganisms potentially useful for the control of plant pathogens. Ciência e Agrotecnologia 2010, 34, 525–535. [Google Scholar] [CrossRef]
- Splivallo, R.; Ottonello, S.; Mello, A.; Karlovsky, P. Truffle volatiles: From chemical ecology to aroma biosynthesis. New Phytol. 2011, 189, 688–699. [Google Scholar] [CrossRef]
- Kramer, R.; Abraham, W.R. Volatile sesquiterpenes from fungi: What are they good for? Phytochem. Rev. 2012, 11, 15–37. [Google Scholar] [CrossRef]
- Crespo, R.; Pedrini, N.; Juárez, M.P.; Bello, G.M.D. Volatile organic compounds released by the entomopathogenic fungus Beauveria bassiana. Microbiol. Res. 2006, 163, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Müller, A.; Faubert, P.; Hagen, M.; Castell, W.; Polle, A.; Schnitzler, J.P.; Rosenkranz, M. Volatile profiles of fungi–chemotyping of species and ecological functions. Fungal Genet. Biol. 2013, 54, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Korpi, A.; Järnberg, J.; Pasanen, A.L. Microbial volatile organic compounds. Crit. Rev. Toxicol. 2009, 39, 139–193. [Google Scholar] [CrossRef]
- Splivallo, R.; Bossi, S.; Maffei, M.; Bonfante, P. Discrimination of truffle fruiting body versus mycelial aromas by stir bar sorptive extraction. Phytochemistry 2007, 68, 2584–2598. [Google Scholar] [CrossRef] [PubMed]
- Morath, S.U.; Hung, R.; Bennett, J.W. Fungal volatile organic compounds: A review with emphasis on their biotechnological potential. Fungal Biol. Rev. 2012, 26, 73–83. [Google Scholar] [CrossRef]
- Budenberg, W.J.; Ndiege, I.O.; Karago, F.W. Evidence for volatile male-produced pheromone in banana weevil Cosmopolites sordidus. J. Chem. Ecol. 1993, 19, 1905–1916. [Google Scholar] [CrossRef]
- Mori, K.; Nakayama, T.; Takikawa, H. Synthesis and absolute configuration of sordidin, the male-produced aggregation pheromone of the banana weevil, Cosmopolites sordidus. Tetrahedron Lett. 1996, 37, 3741–3744. [Google Scholar] [CrossRef]
- Abagale, S.A.; Woodcock, C.; Chamberlain, K.; Osafo-Acquaah, S.; van Emden, H.; Birkett, M.A.; Braimah, H. Attractiveness of host banana leaf materials to the banana weevil, Cosmopolites sordidus in Ghana for development of field management strategies. Pest Manag. Sci. 2019, 75, 549–555. [Google Scholar] [CrossRef]
- Yanagawa, A.; Yokohari, F.; Shimizu, S. The Role of Antennae in Removing Entomopathogenic Fungi from Cuticle of the Termite, Coptotermes formosanus. J. Insect Sci. 2009, 9, 1–9. [Google Scholar] [CrossRef]
- Pasanen, P.; Korpi, A.; Kalliokoski, P.; Pasanen, A.L. Growth and volatile metabolite production of Aspergillus versicolor inhouse dust. Environ. Int. 1997, 23, 425–432. [Google Scholar] [CrossRef]
- Nilsson, A.; Kihlstrom, E.; Lagesson, V.; Wessen, B.; Szponar, B.; Larsson, L.; Tagesson, C. Microorganisms and volatile organic compounds in airborne dust from damp residences. Indoor Air 2004, 14, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, N.; Laumbach, R.; Kelly-McNeil, K.; Lioy, P.; Fan, Z.H.; Zhang, J.; Ottenweller, J.; Ohman-Strickland, P.; Kipen, H. Health effects of a mixture of indoor air volatile organics, their ozone oxidation products, and stress. Environ. Health Perspect. 2005, 113, 1542–1548. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Tian, M.Y.; He, Y.R.; Lei, Y.Y. Differential fluctuation in virulence and VOC profiles among different cultures of entomopathogenic fungi. J. Invertebr. Pathol. 2010, 104, 166–171. [Google Scholar] [CrossRef]
- Strobel, G.; Manker, D.; Mercier, J. Novel Endophytic Fungi and Methods of Use. U.S. Patent Pub. No. 0285543 A1, 11 November 2010. [Google Scholar]
- Daisy, B.H.; Strobel, G.A.; Castillo, U.; Ezra, D.; Sears, J.; Weaver, D.K.; Runyon, J.B. Napthalene, an insect repellent, is produced by Muscodor vitigenus, a novel endophytic fungus. Microbiology 2002, 148, 3737–3741. [Google Scholar] [CrossRef] [PubMed]
- Meyling, N.V.; Pell, J.K. Detection and avoidance of an entomopathogenic fungus by a generalist insect predator. Ecol. Entomol. 2006, 31, 162–171. [Google Scholar] [CrossRef]
- Thompson, S.R.; Brandenburg, R.L.; Roberson, G.T. Entomopathogenic Fungi Detection and Avoidance by Mole Crickets (Orthoptera: Gryllotalpidae). Environ. Entomol. 2007, 36, 165–172. [Google Scholar] [CrossRef]
- Ormond, E.L.; Thomas, A.P.M.; Pell, J.K.; Freeman, S.N.; Roy, H.E. Avoidance of a generalist entomopathogenic fungus by the ladybird, Coccinella septempunctata. FEMS Microbiol. Ecol. 2011, 77, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Inamdar, A.A.; Masurekar, P.; Bennett, J.W. Neurotoxicity of fungal volatile organic compounds in Drosphilla melanogaster. Toxicol. Sci. 2010, 117, 418–426. [Google Scholar] [CrossRef]
- Asensio, L.; Carbonell, T.; Lopez-Jimenez, J.A.; López-Llorca, L.V. Entomopathogenic fungi in soils from Alicante province (Spain). Span. J. Agric. Res. 2003, 1, 3745. [Google Scholar] [CrossRef]
- Lopez-Llorca, L.V.; Duncan, J.M. New media for the estimation of fungal infection in eggs of the cereal cyst nematode, Heterodera avenae Woll. Nematologica 1986, 32, 486–490. [Google Scholar] [CrossRef]
- Gams, W.; Hoekstra, E.S.; Aptroot, A. CBS Course of Mycology, 4th ed.; Centraalbureau voor Schimmelcultures: Baarn, The Netherlands, 1998. [Google Scholar]
- Gams, W.; Van der Aa, H.A.; Van der Plaats-Niterink, A.J.; Samson, R.A.; Stalpers, J.A. CBS Course of Mycology, 3rd ed.; Centraalbureau voor schimmelcultures: Utrecht, The Netherlands, 1987. [Google Scholar]
- Lacey, L.A. Manual of Techniques in Insect Pathology; Elsevier: Amsterdam, The Netherlands, 1997; Volume V-1, pp. 153–185. [Google Scholar]
- O’Donnell, K.; Cigelnik, E.; Nirenberg, H.I. Molecular systematics and phylogeography of the Gibberella fujikuroi species complex. Mycologia 1998, 90, 465–493. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols; Elsevier: Amsterdam, The Netherlands, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Güerri-Agulló, B.; Gómez-Vidal, S.; Asensio, L.; Barranco, P.; Lopez-Llorca, L.V. Infection of the Red Palm Weevil (Rhynchophorus ferrugineus) by the entomopathogenic fungus Beauveria bassiana: A SEM study. Microsc. Res. Tech. 2010, 73, 714–725. [Google Scholar] [CrossRef] [PubMed]
- Ricaño, J.; Güerri-Agulló, B.; Serna-Sarriás, M.J.; Rubio-Llorca, G.; Asensio, L.; Barranco, P.; López-Llorca, L.V. Evaluation of the pathogenicity of multiple isolates of Beauveria bassiana (Hypocreales: Clavicipitaceae) on Rhynochophorus ferrugineus (Coleoptera:Dryophthoridae) for the assessment of a solid formulation under simulated field conditions. Fla. Entomol. 2013, 96, 1311–1324. [Google Scholar] [CrossRef]
- Olivares-Bernabeu, C.M.; López-Llorca, L.V. Fungal egg-parasites of plant-parasitic nematodes from Spanish soils. Rev. Iberoam. Micol. 2002, 19, 104–110. [Google Scholar]
- Purcaro, G.; Moret, S.; Conte, L.S. Microestrazione in fase solida (SPME). Campione L’analisi Chim. 2014, 177–214. [Google Scholar] [CrossRef]
- Bazzocchi, G.G.; Maini, S. Ruolo dei semiochimici volatili nella ricerca dell’ospite da parte del parassitoide Diglyphus isaea (Hymenoptera Eulophidae). Prove olfattometriche. Boll. dell’Istituto Entomol. G. Grandi Univ. Bologna 2000, 54, 143–154. [Google Scholar]
- Lopez-Cepero, J.; COPLACA, Tenerife, Canary Islands, Spain. Personal communication, 2019.
- Lopez-Llorca, L.V.; Jalinas, J.; Marhuenda Egea, F.C. Compuestos Orgánicos Volátiles del Hongo Entomopatógeno Beauveria bassiana Como Repelentes de Insectos. Spanish Patent of Invention P201631534, 9 February 2017. [Google Scholar]
- Chokechaijaroenporn, O.; Bunyapraphatsara, N.; Kongchuensin, S. Mosquito Repellent Activities of Ocimum Volatile Oils. Phytomedicine 1994, 1, 135–139. [Google Scholar] [CrossRef]
- Obeng-Ofori, D.; Reichmuth, C.H.; Bekele, A.J.; Hassanali, A. Toxicity and protectant potential of camphor, a major component of essential oil of Ocimum kilimandscharicum, against four stored product beetles. Int. J. Pest Manag. 1998, 44, 203–209. [Google Scholar] [CrossRef]
- Rozman, V.; Kalinovic, I.; Korunic, Z. Toxicity of naturally occurring compounds of Lamiaceae and Lauraceae to three stored-product insects. J. Stored Prod. Res. 2007, 43, 349–355. [Google Scholar] [CrossRef]
- Lescot, T. La diversité génétique des bananiers. FruiTrop 2011, 89, 58–62. [Google Scholar]
- Ferri, D.V.; Munhoz, C.F.; Neves, P.M.O.; Ferracin, L.M.; Sartori, D.; Vieira, M.L.C.; Fungaro, M.H.P. Genetic Variability of Beauveria bassiana and a DNA Marker for Environmental Monitoring of a Highly Virulent Isolate Against Cosmopolites sordidus. Indian J. Microbiol. 2012, 52, 569–574. [Google Scholar] [CrossRef]
- Kiggundu, A.; Gold, C.S.; Labuschagne, M.T.; Vuylsteke, D.; Louw, S. Levels of host plant resistance to banana weevil, Cosmopolites sordidus (Germar) (Coleoptera:Curculionidae), in Ugandan Musa germplasm. Euphytica 2003, 133, 267–277. [Google Scholar] [CrossRef]
- Kiggundu, A.; Gold, C.S.; Labuschagne, M.T.; Vuylsteke, D.; Louw, S. Components of resistance to banana weevil (Cosmopolites sordidus) in Musa germplasm in Uganda. Entomol. Exp. Appl. 2007, 27–35. [Google Scholar] [CrossRef]
- Night, G.; Gold, C.S.; Power, A.G. Feeding behaviour and efficiency of banana weevil (Cosmopolites sordidus) larvae on banana cultivars of varying resistance levels. J. Appl. Entomol. 2011, 135, 430–437. [Google Scholar] [CrossRef]
- Kaaya, G.P.; Seshu-Reddy, K.V.; Kokwaro, E.D.; Munyinyi, D.M. Pathogenicity of Beauveria bassiana, Metarhizium anisopliae and Serratia marcescens to the banana weevil Cosmopolites sordidus. Biocontrol Sci. Technol. 1993, 3, 177–187. [Google Scholar] [CrossRef]
- Lopes, R.B.; Mesquita, A.L.M.; Tigano, M.S.; Souza, D.A.; Hajek, A.E. Diversity of indigenous Beauveria and Metarhizium spp. in a commercial banana field and their virulence toward Cosmopolites sordidus (Coleoptera:Curculionidae). Fungal Ecol. 2013, 6, 356–364. [Google Scholar] [CrossRef]
- Meyling, N.V.; Eilenberg, J. Occurrence and distribution of soil borne entomopathogenic fungi within a single organic agroecosystem. Agric. Ecosyst. Environ. 2006, 113, 336–341. [Google Scholar] [CrossRef]
- Niu, X.M. Secondary metabolites from Pochonia chlamydosporia and other species of Pochonia. In Perspectives in Sustainable Nematode Management Through Pochonia chlamydosporia Applications for Root and Rhizosphere Health; Springer: Cham, Switzerland, 2017; pp. 131–168. [Google Scholar]
- Fischer, G.; Schwalbe, R.; Möller, M.; Ostrowski, R.; Dott, W. Species-specific production of microbial volatile organic compounds (MVOC) by airborne fungi from a compost facility. Chemosphere 1999, 39, 795–810. [Google Scholar] [CrossRef]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Wnuk, S.; Kinastowski, S.; Kaminski, E. Synthesis and analysis of l-octen-3-ol, the main flavour component of mushrooms. Food/Nahrung 1983, 27, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Venkateshwarlu, G.; Chandravadana, M.V.; Tewari, R.P. Volatile flavour components of some edible mushrooms (Basidiomycetes). Flavour Fragr. J. 1999, 194, 191–194. [Google Scholar] [CrossRef]
- Lozano-Soria, A.; Picciotti, U.; Lopez-Moya, F.; Lopez-Cepero, J.; Porcelli, F.; Lopez-Llorca, L.V. Volatile organic compounds from entomopathogenic and nematophagous fungi, repel banana black weevil (Cosmopolites sordidus). BioRxiv 2020. [Google Scholar] [CrossRef]



| Fungi spp. | Code | CECT Number | NCBI Number | Soil Code | Fungal Genera | Blastn | |
|---|---|---|---|---|---|---|---|
| ITS | Tef1a | ||||||
| Simplicillium lamellicola | A.l.6TS01 | CECT 21125 | MK156720.1 | TS01 | Simplicillium sp. | Simplicillium lamellicola | - |
| Metarizhium robertsii | M.r.4TS04 | CECT 21126 | MK156715.1 | TS04 | Metarhizium sp. | Metarhizium robertsii | M. robertsii |
| Beauveria pseudobassiana | B.p.19TS04 | CECT 21122 | MK156716.1 | TS04 | Beauveria sp. | Beauveria pseudobassiana | B. pseudobasiana |
| Simplicillium lamellicola | A.l.2TS05 | CECT 21123 | MK156718.1 | TS05 | Simplicillium sp. | Simplicillium lamellicola | - |
| Simplicillium lamellicola | A.l.5TS08 | CECT 21124 | MK156719.1 | TS08 | Simplicillium sp. | Simplicillium lamellicola | - |
| Beauveria bassiana | B.b.1TS11 | CECT 21121 | MK156717.1 | TS11 | Beauveria sp. | Beauveria bassiana | B. bassiana |
| Metarhizium brunneum | M.b.8TS21 | - | MT703853 | TS21 | Metarhizium sp. | Metarhizium brunneum | M. brunneum |
| Beauveria bassiana 203 | 10d | 20d | 30d | 40d | 50d | 60d |
|---|---|---|---|---|---|---|
| M-VOCs | ||||||
| 3-cyclohepten-1-one | X | X | X | X | X | X |
| 4-fluoro-1,2-xylene | X | |||||
| m-VOCs | ||||||
| p-Trimethylsilyloxyphenyl-bis(trimethylsilyloxy)ethane | X | |||||
| Borneol | X | X | X | X | X | X |
| 3-octanone | X | |||||
| (2R*,6R*,8AS*)-6-hydroxyedulan | X | |||||
| β-pinene | X | |||||
| Beauveria bassiana 1TS11 | 10d | 20d | 30d | 40d | 50d | 60d |
|---|---|---|---|---|---|---|
| M-VOCs | ||||||
| 1,3-octadiene | X | |||||
| 3-cyclohepten-1-one | X (m-VOCs) | X | X | X | X | |
| m-VOCs | ||||||
| Methyl-1,4-dioxide-pyrazine | X | |||||
| 2-methyl-2-bornene | X | |||||
| 2-(2-ethoxyethoxy)-ethanol | X | |||||
| 2,2,4,4,6,8,8-heptamethyl-nonane | X | |||||
| 1-methyl-1H-1,2,4-triazole | X | |||||
| Borneol | X | X | X | X | ||
| Benzothiazole | X | |||||
| 5-ethyl-3-hydroxy-4-methyl-2(5H)-furanone | X | |||||
| (+-)-gymnomitrene | X | |||||
| Metarizhium robertsii 4TS04 | 10d | 20d | 30d | 40d | 50d | 60d |
|---|---|---|---|---|---|---|
| M-VOCs | ||||||
| 3-cyclohepten-1-one | X | X | X | |||
| 1-octen-3-ol | X(m-VOCs) | X | X | X(m-VOCs) | ||
| 4-fluoro-1,2-xylene | X | X | ||||
| 1,3-dimethoxy-benzene | X | |||||
| m-VOCs | ||||||
| 1,3-octadiene | X | |||||
| 3,5-dimethyl-2-propylthiophene | X | |||||
| 1-β-pinene | X | |||||
| 1-methyl-4-(1-methylethyl)-benzene | X | |||||
| 2-amino-3,5-dibromo-6-methylpyridine | X | |||||
| 4-octen-3-ol | X | |||||
| 2,4-bis(1,1-dimethylethyl)-phenol | X | |||||
| 5-oxoisoboldine | X | |||||
| Pochonia clamydosphoria 123 | 10d | 20d | 30d | 40d | 50d | 60d |
|---|---|---|---|---|---|---|
| M-VOCs | ||||||
| Dimethyldisulfide | X | X | ||||
| 3-methyl-2-pentanone | X | |||||
| 3-cyclohepten-1-one | X | X | ||||
| Propyl-cyclohexane | X | |||||
| Dimethyl-trisulfide | X | |||||
| 1-octen-3-ol | X | X | X | X | ||
| 3-octanone | X | X | X | |||
| 2-dodecanone | X | X | ||||
| 1-methylallyl(cyclooctatetraene)titanium | X | |||||
| 1-phenyl-2-propanone | X | X | ||||
| 1,3-dimethoxy-benzene | X | X | X | X | X | X |
| β-elemene | X | |||||
| m-VOCs | ||||||
| 2-butanone | X | X | ||||
| 3-methylbutanal | X | X | X | |||
| 3-hydroxy-2-butanone | X | |||||
| 2-methylbutanal | X | |||||
| 2,4-octadiene | X | |||||
| 4-methyl-3-hexanone | X | |||||
| Nonane | X | |||||
| (1-methylethenyl) cyclopropene | X | |||||
| 5-methyl-2-hexanone | X | |||||
| 2-butoxy-ethanol | X | |||||
| Propionoin | X | X | ||||
| 2,4-dimethyl-3-hexene | X | |||||
| 3,4-dimethyl-2-hexene | X | |||||
| Trans-3,4-dimethyl-2-hexene | X | |||||
| β-pinene | X | |||||
| Anisole | X | |||||
| 2-isopropyl-5-oxohexanal | X | |||||
| 6-methyl-2-heptanone | X | |||||
| Trimethyl-pyrazine | X | |||||
| 2-methyl-2-bornene | X | |||||
| 2,4-dimethyl-methyl ester-hexanoic acid | X | |||||
| 1-methyl-4-(1-methylethyl)-benzene | X | |||||
| Isocyano-benzene | X | |||||
| 2-(2-ethoxyethoxy)-ethanol | X | X | ||||
| 2,4-dimethylfuran | X | |||||
| 2-dodecanone | X | X | ||||
| 2-methyl-2-(2-methyl-2-butenyl)-furan | X | |||||
| Benzenemethanol | X | |||||
| 3-hydroxymandelic acid ethyl ester di-TMS | X | X | ||||
| 4-hydroxymandelic acid ethyl ester di-TMS | X | |||||
| 4-trimethylsilyl-9,9-dimethyl-9-silafluorene | X | |||||
| 6,7-dimethoxy-2,2-dimethyl-2H-1-benzopyran | X | |||||
| Bicyclo[2 .2.1]heptane-2-carboxylic acid | X | |||||
| 2-amino-4-methylpyrimidine | X | |||||
| 2,4-bis(1,1-dimethylethyl)-phenol | X | |||||
| 2-undecanone | X | |||||
| 2-octanone | X | |||||
| 1-ethyl-2-methyl-cyclohexane | X | |||||
| 16-oxosalutaridine | X | X | ||||
| 5-acetyl-2-hydrazino-4-methylpyridine | X | |||||
| Cyclotetradecane | X | |||||
| trans-β-farnesene | X | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozano-Soria, A.; Picciotti, U.; Lopez-Moya, F.; Lopez-Cepero, J.; Porcelli, F.; Lopez-Llorca, L.V. Volatile Organic Compounds from Entomopathogenic and Nematophagous Fungi, Repel Banana Black Weevil (Cosmopolites sordidus). Insects 2020, 11, 509. https://doi.org/10.3390/insects11080509
Lozano-Soria A, Picciotti U, Lopez-Moya F, Lopez-Cepero J, Porcelli F, Lopez-Llorca LV. Volatile Organic Compounds from Entomopathogenic and Nematophagous Fungi, Repel Banana Black Weevil (Cosmopolites sordidus). Insects. 2020; 11(8):509. https://doi.org/10.3390/insects11080509
Chicago/Turabian StyleLozano-Soria, Ana, Ugo Picciotti, Federico Lopez-Moya, Javier Lopez-Cepero, Francesco Porcelli, and Luis Vicente Lopez-Llorca. 2020. "Volatile Organic Compounds from Entomopathogenic and Nematophagous Fungi, Repel Banana Black Weevil (Cosmopolites sordidus)" Insects 11, no. 8: 509. https://doi.org/10.3390/insects11080509









