Simple Summary
Symbiotic relationships are common in nature and have influenced the evolution of life across the cosmos. Bacterial secondary symbionts are linked to the evolution of aphids’ heightened natural enemy defense mechanisms. Due to their significant historical contributions to insect control, entomopathogenic fungi are recognized as potential biocontrol agents. The research highlights the role of symbiotic bacteria in the protection of insects, especially aphids, against the entomopathogenic fungi and discuss potential implications of this symbiosis.
Abstract
The research was focused on the ability of wheat aphids Sitobion avenae, harboring bacterial secondary symbionts (BSS) Hamiltonella defensa or Regiella insecticola, to withstand exposure to fungal isolates of Beauveria bassiana and Metarhizium brunneum. In comparison to aphids lacking bacterial secondary symbionts, BSS considerably increased the lifespan of wheat aphids exposed to B. bassiana strains (Bb1022, EABb04/01-Tip) and M. brunneum strains (ART 2825 and BIPESCO 5) and also reduced the aphids’ mortality. The wheat aphid clones lacking bacterial secondary symbionts were shown to be particularly vulnerable to M. brunneum strain BIPESCO 5. As opposed to wheat aphids carrying bacterial symbionts, fungal pathogens infected the wheat aphids lacking H. defensa and R. insecticola more quickly. When treated with fungal pathogens, bacterial endosymbionts had a favorable effect on the fecundity of their host aphids compared to the aphids lacking these symbionts, but there was no change in fungal sporulation on the deceased aphids. By defending their insect hosts against natural enemies, BSS increase the population of their host society and may have a significant impact on the development of their hosts.
1. Introduction
Symbiosis is a close, long-term, mutual relationship between two organisms of different species that may or may not benefit each other [1]. Symbiotic associations are ubiquitous in nature and have played a vital role in the evolution of life on Earth [2]. Among the diverse examples of symbiotic associations, the most sophisticated ones have been reported to be associated with bacterial endosymbionts, which allow for intimate interactions in the partners involved [3]. Insects are the most diverse order of animals and almost half of all insect species are predicted to harbor bacterial endosymbionts [4]. In most cases, bacterial endosymbionts are vertically transmitted from one generation to the next generation [5]. Bacterial endosymbionts in insects are categorized into two groups: (1) obligate or primary symbionts and (2) facultative or secondary symbionts. Primary symbionts are obligate in nature for host survival. They reside in specialized cells in their hosts, called bacteriocytes, and are mutually beneficial for both partners [6]. Buchnera aphidicola Munson (Enterobacterales: Erwiniaceae) in aphids and Candidatus Portiera aleyrodidarum Thao and Baumann (Oceanospirillales: Halomonadaceae) in whiteflies are typical examples [5,7,8,9]. B. aphidicola and their aphid associations display an outstanding relationship among bacteria and insects, where none of the partners can survive without the other. The bacterium contributes more than 90% of the essential amino acids needed by the pea aphid, Acyrthosiphon pisum Harris (Hemiptera: Aphididae), and directly contributes to nutritional fitness [10].
Bacterial secondary symbionts (BSS) facultatively colonize their hosts, occurring more occasionally and found living freely in the hemolymph or other tissues of their hosts. They have the ability to influence the developmental, morphological, and physiological traits of their hosting individuals, which may benefit their spread and further establishment within host populations [11]. The best-studied BSS–aphid interactions used the pea aphid A. pisum–symbiont system. In this species, BSS contributed to the thermal fitness of aphids against heat shock by improving their survival and fecundity [12,13]. They have also been reported to contribute to the effective utilization of host plants [14,15], and it is hypothesized that geographic variation in host plant use is also influenced by BSS [16]. BSS also contribute to the protection of aphids against natural enemies [17]. Moreover, pea aphids harboring Regiella insecticola Moran (Enterobacterales: Enterobacteriaceae) have been reported to exhibit increased resistance against infection by the entomopathogenic fungus Pandora neoaphidis Remaud and Hennebert (Entomophthorales: Entomophthoraceae) [18]. Similarly, R. insecticola, Rickettsia sp., and Rickettsiella sp. reduced the mortality of their host aphids and also decreased the sporulation of fungus on dead aphids [19]. R. insecticola provided resistance against the aphid-specific fungal pathogen Zoophthora occidentalis (Thaxt.) A. Batko (Entomophthorales: Entomophthoraceae), while no resistance against a generalist fungal entomopathogen such as Beauveria bassiana (Bals.-Criv.) Vuill. (Hypocreales: Cordycipitaceae) was reported [20]. The BSS Hamiltonella defensa Moran (Entomophthorales: Entomophthoraceae) provided protection to pea aphids against the parasitoid Aphidius ervi Haliday (Hymenoptera: Braconidae) by killing its larvae in the aphids [15,21]. H. defensa and Serratia symbiotica Moran (Enterobacterales: Yersiniaceae) in A. pisum also reduced consumption by a predatory ladybird beetle [22]. There is now sufficient evidence that BSS have a prominent influence on the ecology and evolution of their hosts [23] and are also impacting the population dynamics of their hosts [24,25]. By providing protection to their hosts, secondary endosymbionts increase in frequency within host communities [19].
The English grain aphid S. avenae is an economically important insect pest in Western and Central Europe, causing severe damage via quantitative and qualitative losses in wheat crops [26,27,28,29]. Traditionally, insecticides are one of the most common control measures used against aphids, as they are cheap and fast-acting; however, aphids have the ability to rapidly develop insecticide resistance and to reproduce very quickly, making insecticidal treatments less efficient [30,31]. In addition, insecticides are found almost everywhere, and this contamination places the environment and non-target organisms, ranging from beneficial soil microorganisms to insects, fishes, and birds, at increased risk [32].
Thus, there is a need to develop environment friendly and cost-effective measures to reduce pest aphid populations. Biological control using entomopathogenic fungi (EPF) of wheat aphids is a good option to replace chemical control [33]. As compared to the conventional control by pesticides, EPF are environmentally friendly, with no or less risk of leaving pesticide residues in food, and are beneficial for the conservation of the biodiversity of natural enemies in agroecosystems [34]. The mode of action of entomopathogenic fungi involves the germination and penetration of infective spores into the cuticle, followed by developing a germ tube in the hemocoel of the insect host. Thereafter, the fungus reproduces in the insect and causes death due to the production of toxins and/or multiplication to inhabit the entire insect [35].
Approximately 700 fungal species have been described for their entomopathogenic effects, with Beauveriabassiana and Metarhizium brunneum Petch (formerly Metarhizium anisopliae var. anisopliae) (Hypocreales: Clavicipitaceae) being the best-studied species [35,36,37]. B. bassiana is a generalist entomopathogen due to possessing a stereotypical pattern of pathogenicity genes towards many insect species [38].
Several myco-pesticides, using EPF species such as B. bassiana, M. anisopliae, and Akanthomyces lecanii (Zimm.) Spatafora, Kepler & B. Shrestha (Hypocreales: Cordycipitaceae), have been registered for the control of aphids [39,40]. However, biological control of insect pests by these fungal pathogens on a commercial level needs to take into account the diverse ecological interactions between, e.g., the pest species and the entomopathogens on a species and strain level [41].
Although several studies have already addressed the role of BSS in aphid life history parameters, to the best of our knowledge, there are no studies systematically evaluating the aphid genotype–BSS interactions with regard to their role when exposed to different fungal strains. Here, we used two genotypes of S. avenae either harboring or lacking BSS to assess the role of these BSS in conferring resistance against EPF isolates of B. bassiana (strains EABb04/01-Tip and Bb 1022) and M. brunneum (strains ART2825 and BIPESCO 5). We evaluated the role of these BSS in survival, fecundity, and mortality in wheat aphid over time after exposure to EPF.
2. Materials and Methods
2.1. Insect Cultures
Two clones of the English grain aphid, S. avenae, named 5 and 7, were used in this study. These clones were initially collected near Giessen (Giessen, Germany). These clones originally harbored the bacterial secondary symbionts H. defensa and R. insecticola and were reproduced from a single parthenogenetic female aphid [42]. Aphids were maintained under laboratory conditions on winter wheat plants (cv Dekan, KWS GmbH., Einbeck, Germany).
2.2. Exclusion of BSS
Antibiotics have been proven to be a tool for the removal of facultative symbionts from their natural hosts [16,43]. We used an antibiotic micro-injection protocol for the removal of H. defensa from aphid clone 5 [44]. The injection dose contained a mixture of 250 µg of ampicillin, cefotaxime, and gentamycin per ml solution. For the removal of R. insecticola from aphid clone 7, ampicillin was injected with a dose of 1 µg/mg of aphid body weight [45]. Second-instar aphids were paralyzed by exposing them to CO2 for 20 s before injecting the antibiotics. Aphids treated with antibiotics were separately transferred to wheat plants and permitted to reproduce. The nymphs produced from treated aphids were named generation 1 (G1). From these G1 nymphs, 4–5 nymphs were collected randomly from each antibiotic-treated mother aphid and raised on wheat plants until they were able to reproduce sufficient progeny as generation 2 (G2). To confirm the absence of bacterial symbionts, G1 aphids were subjected to diagnostic PCRs. Then, only those G2 nymphs were allowed to proceed to further rearing whose G1 mothers displayed the successful elimination of bacterial secondary symbionts (BSS). Diagnostic PCR was performed up to the eighth generation before trials started, to confirm the exclusion of BSS from aphids. Finally, four aphid lines, named clone +5, clone −5, clone +7, and clone −7, respectively, were prepared. Aphid clones assigned (+) harbored BSS, while clones assigned (–) lacked BSS, based on an identical genetic background.
2.3. PCR Protocol for the Detection of BSS
DNA extraction from aphid clones was carried out by following the CTAB procedure [46]. Diagnostic PCR, by using specific primers, was performed to detect the presence or absence of BSS by amplifying 16S rDNA gene fragments. Forward primer HDFn [5-ATGAAGTCGCGAGACCAAA-3] and reverse primer HDRn [5-GCTTTCCCTCGCAGGTTC-3] were used for H. defensa, and forward primer RIFn [5-GAAGGCGGTAAGAGTAATATGC-3] and reverse primer RIRn [5-CCCCGAAGGTTAAGCTACCTA-3] were used for R. insecticola detection, respectively. The following temperature profile was used for the diagnostic PCR: 94 °C for 3 min followed by 30 cycles of 94 °C for 30 s; 60 °C for 40 s; 72 °C for 90 s, and concluding incubation was performed at 72 °C for 8 min. The PCR was performed in a 25 µL volume with one µL of the DNA template having 0.32 µM of each primer, 2 mM MgCl2, 200 µM dNTP’s, 1× “Bioline” PCR buffer, and 0.25 units of Taq DNA polymerase. PCR products were observed by using ethidium bromide on 1.7% agarose gel. For the verification of BSS, the PCR products were purified from the gel and sent for sequencing to LGC Genomics GmbH, Germany. The final sequences were then correlated with the known sequences of H. defensa and R. insecticola, utilizing the BLAST algorithms at NCBI to confirm the identity of the endosymbionts.
2.4. Fungal Cultures
Fungal isolates (Table 1) were cultured on potato dextrose agar (PDA) (Sigma-Aldrich, Merck KGaA, Darmstadt, Germany) medium in 90 mm Petri dishes. PDA media were prepared by mixing 39 g PDA mixture (potato extract: 4 g, dextrose: 20 g, agar: 15 g) in 1000 mL Bidest water and autoclaving them for 20 min at 121 °C. Petri plates with PDA mixtures were inoculated with fungal spores and incubated for 2 weeks at 20 ± 1 °C with a 16:8 (L:D) hour photoperiod for mass spore production to be used for bioassays.

Table 1.
Isolates of entomopathogenic fungi used in this study with geographic origin, original insect host.
2.5. Bioassay
The roots of young wheat plants were wrapped with moist cotton and placed in 90 mm Petri dishes with filter sheets. In sterile clean bench circumstances, 10 S. avenae nymphs (second or third instar) were transferred to these Petri plates. Using the approach in [51], spore suspensions from entomopathogenic fungi were created after harvesting the spores from PDA plates. In sterilized conical flasks, harvested conidial spores were combined with 100 mL of 0.03% tween TM 20–sterile water solution, and the suspension was agitated using a magnetic stirrer (Bioevopeak Co., Ltd., Jinan, China). By counting the spores in a hemocytometer (Sigma-Aldrich, Merck KGaA, Darmstadt, Germany) while they were being observed under a microscope (Bioevopeak Co., Ltd., Jinan, China the final concentrations of the spore suspensions were determined and corrected for 1 × 108 spores per ml. To apply spores to aphids on Petri plates, the fungal spore suspensions were placed into a hand sprayer (Bürkle GmbH, Bad Bellingen, Germany). Each plate received a complete shower of a spore suspension sprayed from a certain distance (8–10 inches) to cover the whole plate area. All of the plates were para-film-sealed after spore applications, and they were all placed in a climate-controlled room with a 16:8 (L:D) hour photoperiod at 20 ± 1 °C and 65 ± 5% relative humidity. The aphid clones +5, −5, +7, and −7, with 16 replications each, were sprayed using an identical technique for all entomopathogenic fungal strains. The aphids in the control group were sprayed with a 0.03% tween TM 20 solution diluted in 100 mL of sterilized water. For up to five days, dead aphids were counted daily to track the progression of the fungus infection and determine the overall mortality.
2.6. Fecundity Assay
The indirect effects of fungal strains on the fertility of aphids harboring or not harboring bacterial secondary symbionts were evaluated using a fecundity experiment. After recording the final mortality on day five, 16 wingless adult aphids were randomly chosen, and they recovered from the entomopathogenic fungal treatment. These aphids were housed in climate-controlled rooms alongside wheat plants in fresh Petri plates. Daily fecundity data were collected for five days after the females from the first neonate. For a maximum of 16 days, the total number of offspring generated was recorded. Data on fecundity were also gathered for the control treatments in a similar manner.
2.7. Fungal Growth Assay
The effect of bacterial secondary symbionts on fungal development was evaluated in the dead aphids on a semi-selective medium (SM). First, we produced Sabouraud dextrose medium by dissolving glucose and peptone in demineralized water and bringing the mixture’s pH to 6.3 using 1M HCl. The medium included 10 g of peptone, 20 g of glucose, and 18 g of agar per liter of solution. Agar was then added when the solution was transferred to a volumetric flask. The media were autoclaved at 121 °C for 20 min, before being cooled to 50 °C. Before transferring the media to Petri dishes, antibiotics (Thermo Fisher Scientific, Waltham, MA, USA) (cycloheximide: 50 mg, streptomycin: 100 mg, tetracycline: 50 mg per liter) and fungicides (dodine: 100 mg per liter) were applied [52]. On this SM medium, dead aphids from the aforementioned treatments were applied (5 aphids per plate). At 5, 7, and 9 days after inoculation, the diameters of the developing fungi from dead wheat aphids were measured in millimeters using a vernier scale.
2.8. Statistical Analysis
In a one-way ANOVA, the total aphid mortality was analyzed using the least significant difference (LSD) test. Using repeated-measure analysis of variance (ANOVA) and a paired t-test, analyses of the fecundity of aphids and fungal radial growth after fungal treatments were performed. Statistix (version 8.1) software was used to conduct statistical analyses, with a 5% threshold of significance [53]. The Kaplan–Meier survival tool in R software (version 3.1) was used to calculate the daily aphid survivability rate.
3. Results
3.1. Total Mortality in Wheat Aphid
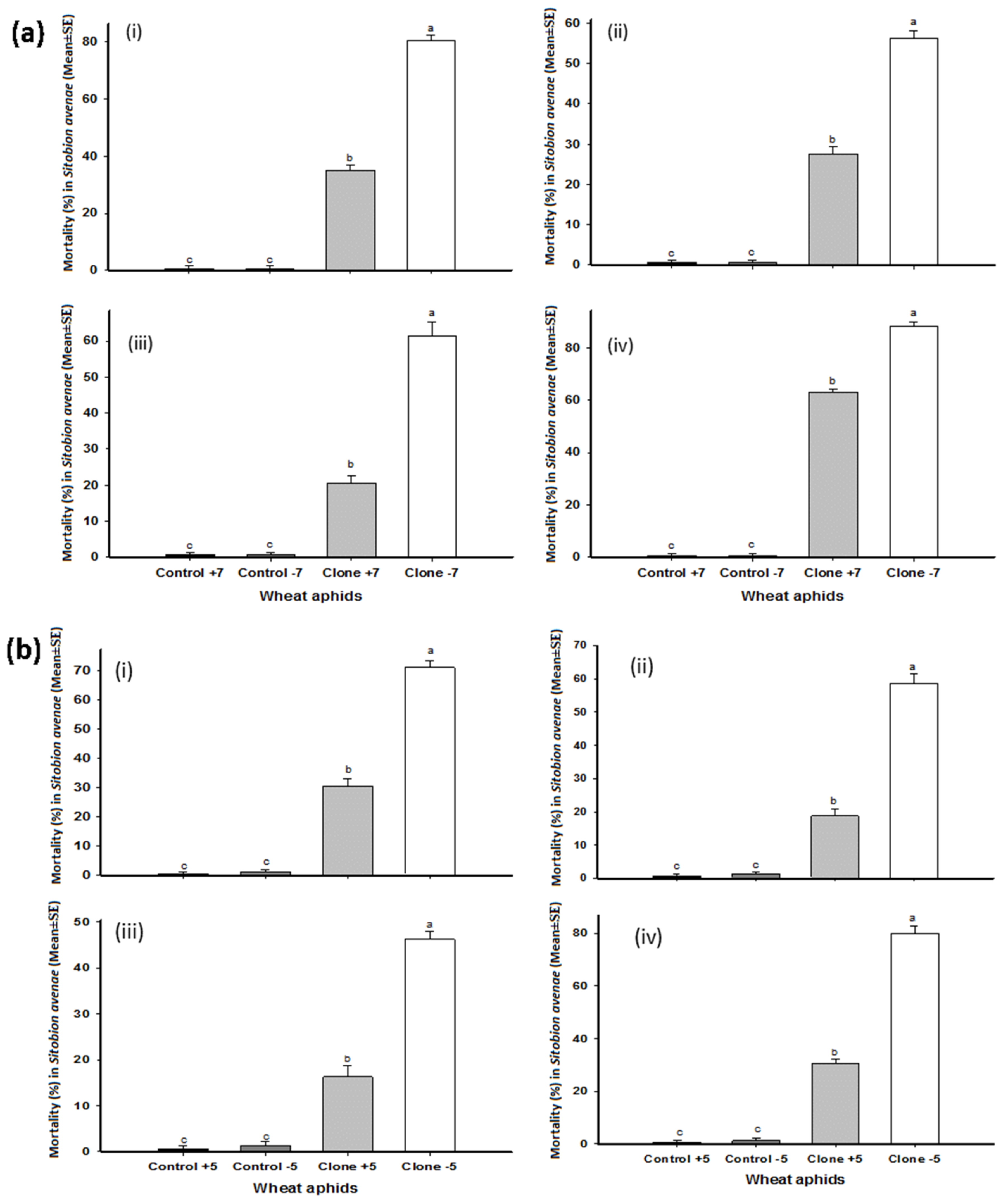
The strains of entomopathogenic fungi, B. bassiana and M. brunneum, varied with regard to pathogenicity and virulence against aphid clones with and without BSS, causing significantly higher mortality in those clones without bacterial symbionts. B. bassiana strains Bb1022 and EABb04/01-Tip and M. brunneum strains ART 2825 and BIPESCO 5 caused 70, 59, 46, and 80% mortality, respectively, in clone –5 after five days, while 30, 19, 16, and 31% mortality, respectively, was recorded in clone +5 (Figure 1a). The bacterial secondary symbiont H. defensa partially protected aphids harboring them, significantly reducing mortality (p < 0.005) as compared aphids lacking H. defensa. Similar results in mortality were recorded for clone −7 when exposed to B. bassiana strains Bb1022 and EABb04/01-Tip and M. brunneum strains ART 2825 and BIPESCO 5, causing 80, 56, 61, and 88% mortality, respectively, after 5 days, while 35, 28, 21, and 63%, respectively, was recorded for clone +7 (Figure 1b). M. brunneum strain BIPESCO 5 was found the most lethal to aphids without BSS as compared to the other fungal strains tested and caused more than 80% mortality to clone −5 and −7. The mortality of wheat aphids in control treatments was less than one percent.
Figure 1.
Total mortality in wheat aphid (Sitobion avenae) clones +7, −7 (a) and +5, −5 (b) after five days of application of Beauveria bassiana strains Bb1022 (i) and EABb04/01-Tip (ii) and Metarhizium brunneum strains ART 2825 (iii) and BIPESCO 5 (iv). The letters (a, b, c) are indicating the significance difference among the treatments.
3.2. Fungal Radial Growth
Both tested entomopathogenic fungi, B. bassiana and M. brunneum, successfully sporulated on dead aphids placed on semi-selective media. The radial growth of B. bassiana strain Bb1022 on dead aphids either harboring or not harboring H. defensa or R. insecticola was not significantly different (p > 0.005) after 5, 7, and 9 days of inoculation and showed an almost similar growth pattern. Similar results were obtained in the case of B. bassiana strain EABb04/01-Tip for both of the aphid clones. Once EPF were able to overcome the resistance conferred by the bacterial secondary symbionts, due to the death of the specimens colonized, no difference in fungal growth on dead aphids with or without bacterial secondary symbionts was observed. Meanwhile, the isolate of M. brunneum ART 2825 showed significantly less radial growth on aphids harboring H. defensa as compared to those lacking symbiotic bacteria up to seven days after plating them on agar (p < 0.005); however, at day nine, this difference was no longer significant (p > 0.005). The same strain, ART 2825, behaved differently on wheat aphid clones 7 and −7. Differences in radial growth from aphids with and without R. insecticola were non-significant from the beginning up to 7 days, but growth significantly accelerated on clone −7 (p < 0.005) from aphids containing R. insecticola. M. brunneum BIPESCO 5 grew equally well (p > 0.005) on dead aphids with or without H. defensa for up to 9 days (Table 2). In the case of aphids harboring R. insecticola, less growth was observed in the case of BIPESCO 5 for the first 5 days after plating dead aphids onto the media, as compared to aphids without R. insecticola. Thereafter, fungal growth increased in aphids infected with R. insecticola and growth differences were non-significant (p > 0.005) when comparing aphids with and without R. insecticola after 7 and 9 days (Table 2)

Table 2.
Radial growth diameters (cm ± SE) of Beauveria bassiana and Metarhizium brunneum from dead wheat aphid (Sitobion avenae) clones with and without bacterial secondary symbionts.
3.3. Fecundity Assay
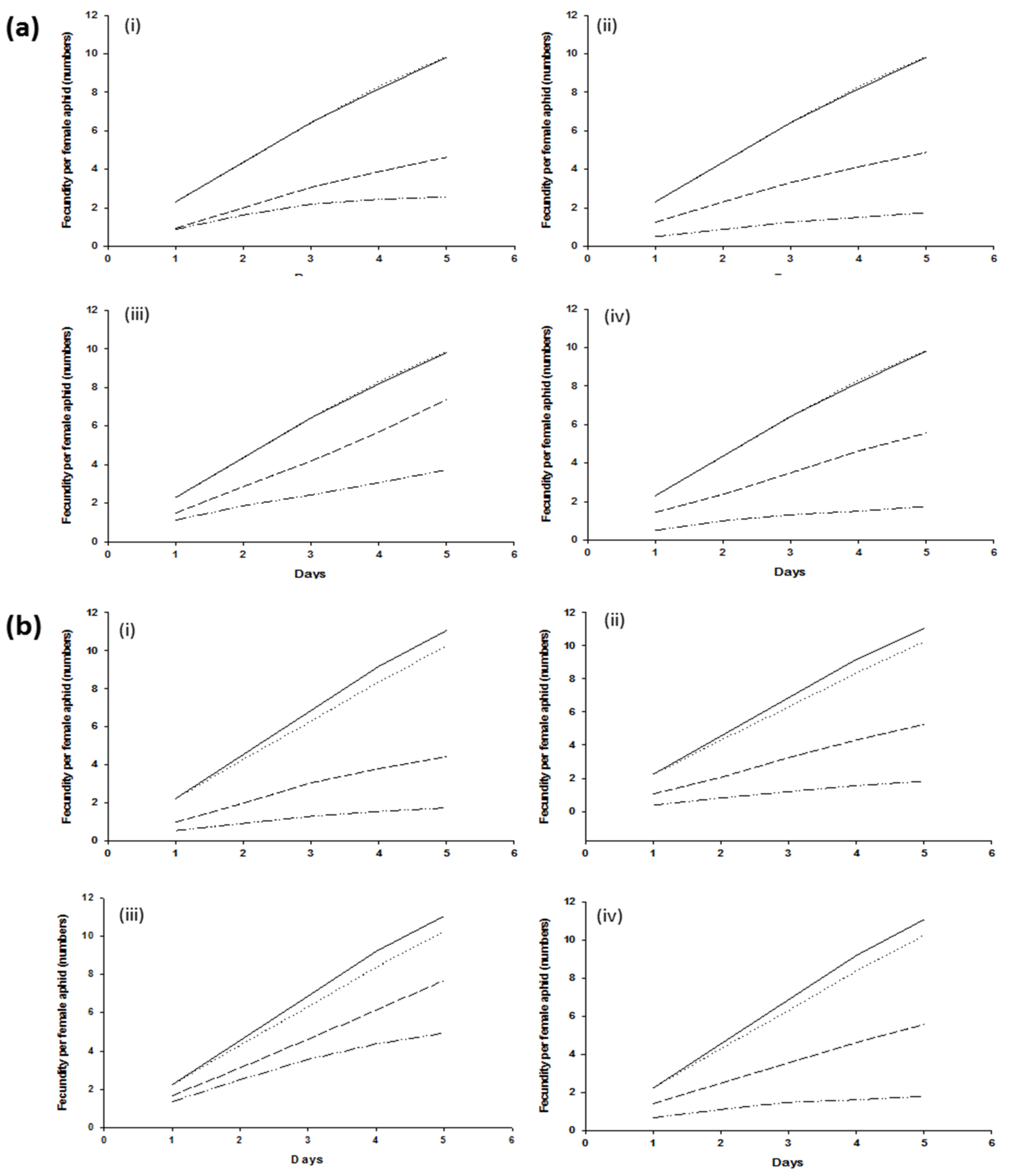
Fecundity in wheat aphids harboring H. defensa or not harboring bacterial symbionts was not significantly different (p > 0.005) in control treatments and resulted in 10 nymphs per female aphid in five days. Similar results were recorded in aphids harboring or not harboring R. insecticola. Female aphids, infected with H. defensa and R. insecticola exposed to the B. bassiana strain Bb1022, exhibited a significantly higher (p < 0.005) reduction in cumulative fecundity. Within five days, female aphids harboring H. defensa and R. insecticola produced an average of 4.62 ± 0.22 and 4.44 ± 0.46 nymphs, respectively, as compared to aphids lacking them (2.56 ± 0.22 and 1.75 ± 0.18 nymphs per female). Similar fecundity effects were observed for B. bassiana strain EABb04/01-Tip, either with or without H. defensa and R. insecticola, producing 4.88 ± 0.42 and 5.25 ± 0.64, or 2.13 ± 0.44 and 1.81 ± 0.18 offspring, respectively. The bacterial symbionts H. defensa and R. insecticola exhibited higher resistance to M. brunneum ART 2825 and resulted in higher fecundity as compared to all other fungal strains; however, differences were still significantly different (p > 0.005) when comparing aphid clones +5, −5, +7, and −7, producing 7.37 ± 0.66, 3.69 ± 0.26, 7.69 ± 0.94, and 4.94 ± 0.55, respectively (Figure 2). The effect of BIPESCO 5 on the fecundity of aphids harboring H. defensa was highly significant (p < 0.005), producing 5.56 ± 0.62 nymphs per female, as compared to wheat aphids without H. defensa, producing only 1.75 ± 0.44 nymphs. Similar effects of M. brunneum BIPESCO 5 were observed in aphid clones with or without R. insecticola (p > 0.005).
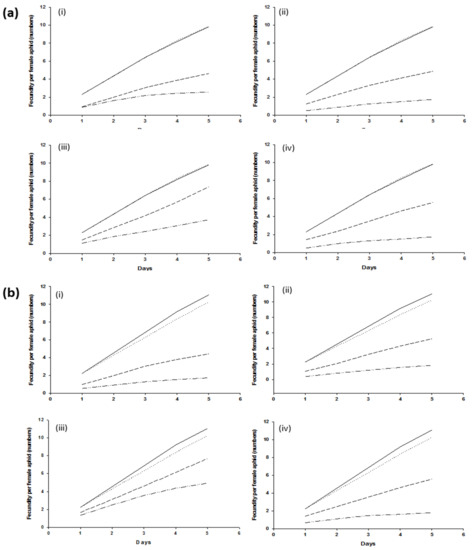
Figure 2.
Number of offspring produced within 5 days following applications of entomopathogenic fungi Beauveria bassiana strains Bb1022 (i) and EABb04/01-Tip (ii) and Metarhizium brunneum strains ART 2825 (iii) and BIPESCO 5 (iv) to wheat aphid (Sitobion avenae) clones +5 and −5 (a) and clones +7 and −7 (b). ( ) shows control with BSS, (
) shows control with BSS, ( ) shows control without BSS, (
) shows control without BSS, ( ) shows treatment with BSS, and (
) shows treatment with BSS, and ( ) shows treatment without BSS.
) shows treatment without BSS.
 ) shows control with BSS, (
) shows control with BSS, ( ) shows control without BSS, (
) shows control without BSS, ( ) shows treatment with BSS, and (
) shows treatment with BSS, and ( ) shows treatment without BSS.
) shows treatment without BSS.
3.4. Infection Period
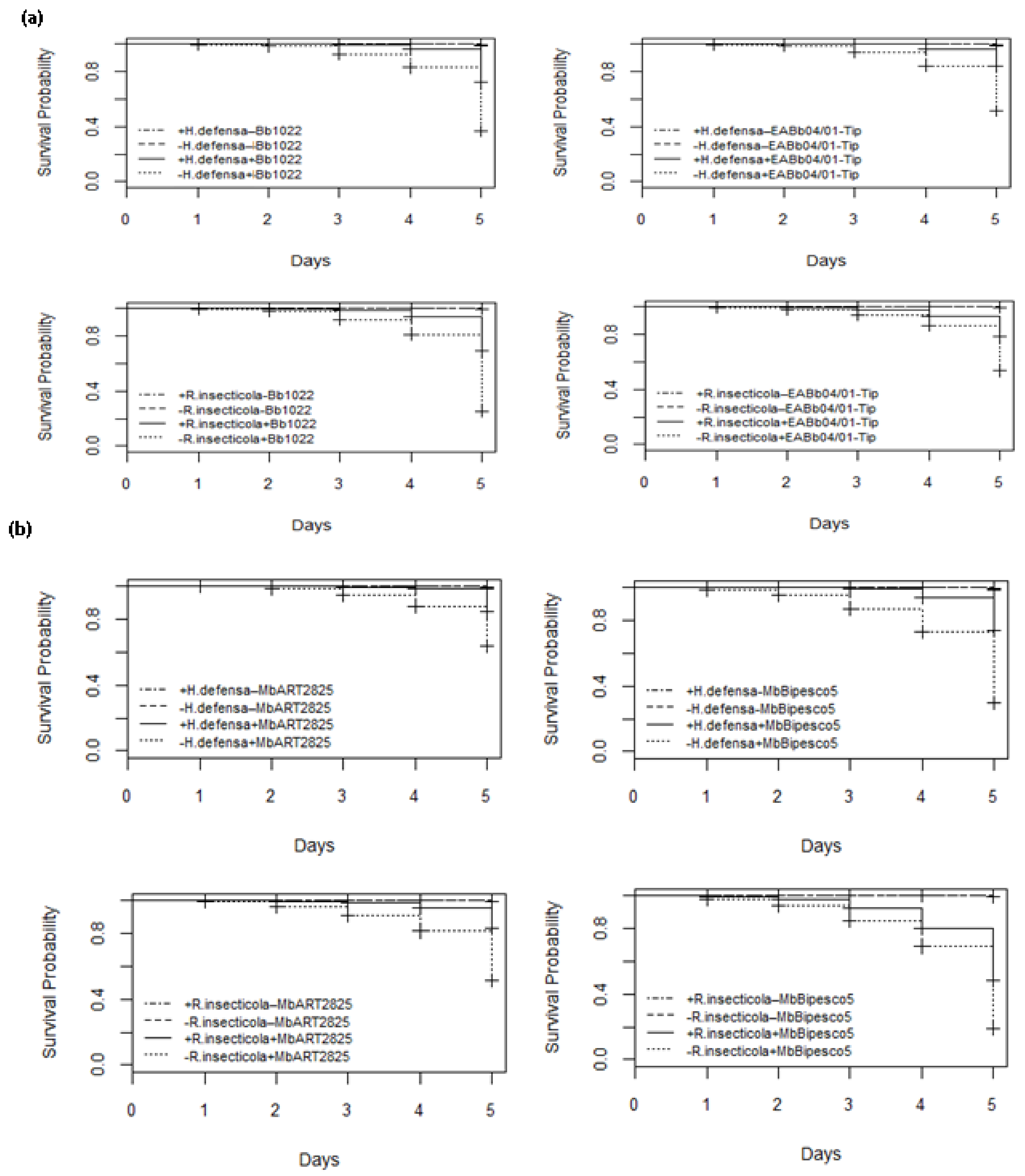
Daily evaluations of aphid survival up to 5 days after fungal spore treatments and of the controls were conducted. The survival of aphids in the control treatments was unaffected by H. defensa and R. insecticola, with 100% of them still alive after 5 days. B. bassiana strain Bb1022 infection began in aphid clones +5 and +7 after the third and second days, respectively, but in aphid clones −5 and −7, infection began within 24 h, leading to early death. The fungal infection was highest on the 5th day after application and resulted in 72% and 70% survival of aphids in clones +5 and +7, while, in clones −5 and −7, survival was significantly reduced to 36% and 25%, respectively. This clearly indicates that R. insecticola confer significant resistance to their hosts, and the difference illustrates that the protection against B. bassiana strain Bb1022 provided by H. defensa and in survival is significant (LR χ2 = 334, 399, df = 3 and p < 0.005). H. defensa and R. insecticola delayed B. bassiana strain EABb04/01-Tip infections in individuals of aphid clones +5 and +7 (51 and 53%, respectively) compared to individuals without bacterial symbionts, which was highly significant (LR χ2 = 270, 224, df = 3 and p < 0.005). M. brunneum ART 2825 significantly reduced the survival of aphids in clonal lines −5 and −7 as compared to aphids harboring H. defensa and R. insecticola (LR χ2 = 189, 290, df = 3; p < 0.005). The infection started earlier in wheat aphids without bacterial symbionts, reducing the survival on the 5th day to 63 and 51%, as compared to the aphid +5 and +7 specimens with a survival rate of 85 and 83%, respectively (Figure 3). M. brunneum BIPESCO 5 proved to be the most virulent strain towards both aphid lines without H. defensa and R. insecticola, reducing their survival to 30 and 19% on day 5 as compared to aphids harboring bacterial symbionts, with a significantly higher survival rate (LR χ2 = 458, 484, df = 3 and p < 0.005) of 74 and 48%, respectively.
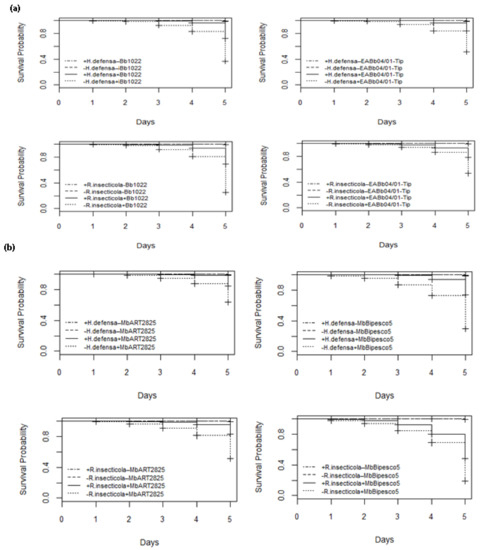
Figure 3.
Survival curves showing infection period over 5 days post-application of entomopathogenic fungi Beauveria bassiana strains Bb1022 and EABb04/01-Tip (a) and Metarhizium brunneum strains ART 2825 and BIPESCO 5 (b) against wheat aphid (Sitobion avenae) clones +5, −5, +7, and −7.
4. Discussion
It is evidenced through our results that bacterial secondary symbionts H. defensa and R. insecticola can influence their host wheat aphids in multiple ways, such as host survival over the infection period against entomopathogenic fungi B. bassiana and M. brunneum strains. Our results indicate that the presence of bacterial secondary symbionts delays the infection penetration of entomopathogenic fungi into the wheat aphids harboring H. defensa and R. insecticola as compared to those lacking them, and increases the probability of host survival. Some previous studies have reported that R. insecticola increased the survival of its host pea aphid against fungal entomopathogen P. neoaphidis, although this effect differed in magnitude among pea aphid clones [18]. Carrying R. insecticola and Rickettsia spp. was beneficial to pea aphids and showed strong protection effects against entomopathogenic fungi P. neoaphidis by enhancing their survival [19]. Our results also highlight that M. brunneum strain BIPESCO 5 is the most virulent against wheat aphids, causing up to 90% mortality.
At present, M. brunneum strain BIPESO 5 and strain CB15-III are the only certified Metarhizium strains for pest control on commercial levels in many European countries [37].
The comparison of the entomopathogenic fungi B. bassiana and M. brunneum’s fungal sporulation growth on dead aphids with and without H. defensa and R. insecticola proved that bacterial secondary symbionts cannot influence the fungal growth on their hosts after death, because no appreciable difference in growth rates was found on aphids with or without these bacterial symbionts. This indicates that when the fungal pathogen effectively overcame the resistance provided by the bacterial secondary symbionts at the beginning of the infection, the wheat aphids sporulated successfully, without any difference regarding the presence or absence of bacterial symbionts.
Some previous studies also reported similar results regarding the entomopathogenic fungi P. neoaphidis on dead pea and wheat aphids with or without H. defense. In fact, the sporulation of P. neoaphidis was not affected by the presence of bacterial symbionts after the death of their hosts [19,54]. It is possible that secondary symbionts are only effective against entomopathogenic fungal spread while in combination with the natural immune system of their host aphid, but they lacked effectiveness after the death of their host.
The tested fungal pathogen strains of B. bassiana and M. brunneum had negative effects on the fecundity of wheat aphids without H. defensa and R. insecticola. In our results, this was an indirect effect of the entomopathogenic fungi, which caused higher mortality in wheat aphids without bacterial secondary symbionts after 5 days, resulting in reduced fecundity indirectly. This adverse effect caused by the entomopathogenic fungi was successfully reduced in wheat aphids carrying bacterial secondary symbionts by enhancing their longevity under stress. Such effects of entomopathogenic fungi on the aphid’s fecundity are important when considering them as biocontrol agents against aphids, because a decrease in fecundity after infection must contribute to a reduction in populations of wheat aphids. Negative effects on fecundity caused by M. anisopliae and Aspergillus ochraceus Wilhelm (Eurotiales: Trichocomaceae) fungi have already been reported in Ceratitis capitata (Diptera: Tephritidae) [55], while the application of P. neoaphidis reduced the fecundity of pea aphids carrying Rickettsia and Spiroplasma bacterial symbionts [50]. It is inferred that different bacterial symbionts express themselves differently depending on their host and natural enemy’s genetic backgrounds.
Previous studies demonstrated that the host defense of bacterial secondary symbionts was limited to P. neoaphidis mainly in aphids, but there should be more explanatory and diversified effects on their host phenotypes against the natural enemies for their persistence in aphid populations. The effectiveness of bacterial symbionts is dependent upon the genotypes of the host and fungal strains; for example, the presence of H. defensa had no effect on the fungal sporulation or on the survival of pea aphids against P. neoaphidis [19], but, in contrast, it exhibited protection towards its host in our trials. In addition, R. insecticola potentially defends its host against fungal pathogens [15,21,54]. All these variations, including those noticed here, lead to the conclusion that the diverse functions performed by bacterial symbionts for their host aphids under various biotic and abiotic stresses have helped them to maintain their populations.
The possibility that bacterial secondary symbionts have the ability to act as a defensive wall for their hosts against natural enemies such as entomopathogenic fungi, parasitoids, and predators needs further consideration [23]. The mechanism behind H. defensa and R. insecticola’s resistance to fungal pathogens is still unknown, regardless of the symbiont’s genome availability [56]. It would be interesting to identify whether these two bacterial symbionts have developed these characteristics independently or whether there is some genetic aspect involved in their dissemination. There is also a probability that bacterial secondary symbionts boost the aphids’ defense against pathogens indirectly by galvanizing the immune system of the host and competing with pathogens for nutrition or proliferating essential nutrients accessible to the immune system [23]. In addition, the resistance conferred by bacterial symbiont genotypes against natural enemies may rely on abiotic components such as temperature [57], heat stress [58], and drought stress [59], and also host attributes such as age [60].
Our results broaden the significance of H. defensa and R. insecticola in wheat aphids, where they significantly reduced the mortality of wheat aphids caused by strains of entomopathogenic fungi B. bassiana (EABb04/01-Tip and Bb 1022) and M. brunneum (ART 2825 and BIPESCO 5). In addition to host aphid protection against parasitoids [21], H. defensa and R. insecticola also play a significant role in the host defense system by conferring resistance upon their hosts against fungal pathogens. This finding is another clue by which to understand the wide spread of these bacterial secondary symbionts among the insect communities. As these entomopathogenic fungi are being used as biocontrol tools against insect pests such as aphids [61], our work suggests that bacterial secondary symbiont-based protection against fungal pathogens may be a crucial consideration during the selection and development of biocontrol agents.
5. Conclusions
In general, researchers still need to develop a complete understanding of the role of bacterial secondary symbionts in insect host populations. Our results may assist in a better understanding of the interactions between BSS (H. defensa and R. insecticola) of S. avenae and EPF (B. bassiana and M. brunneum). It is concluded that these BSS provide their host aphids with increased resistance against the infection and toxicity caused by the application of EPF by lowering their mortality. Thus, it would be important to consider the presence of such bacterial secondary symbionts in the biological control of not only wheat aphids but also other insect pests of crops.
Author Contributions
Conceptualization, S.A. and A.S.; methodology, Q.S. and S.A.; software, M.A.F., M.A.A. and M.F.S.; validation, K.T., M.F.S. and S.A.; formal analysis, S.A. and M.I.U.; investigation, S.A. and A.S.; resources, M.F.S., A.J. and M.I.U.; data curation, S.A., M.A.A., A.I. and A.I., M.F.S.; writing—original draft preparation, S.A., A.I., M.A.A. and M.I.U.; writing—review and editing, M.F.S., A.J., M.I.U. and B.M.; visualization, M.F.S.; supervision, A.S.; project administration, M.F.S.; funding acquisition, A.J., M.F.S. and B.M. All listed authors met all criteria of the International Committee of Medical Journal Editors (ICMJE) and, more specifically, they were involved as described above. All authors have read and agreed to the published version of the manuscript.
Funding
This study was partially supported by Università degli Studi di Palermo (35-161, PRJ-0510).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We wish to express our appreciation to Zaheer Abbas for his help and support. We would like to thank the anonymous reviewers for their valuable comments and suggestions that improved the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Louie, A. Relational biology of symbiosis. Axiomathes 2010, 20, 495–509. [Google Scholar] [CrossRef]
- Zientz, E.; Silva, F.J.; Gross, R. Genome interdependence in insect-bacterium symbioses. Genome Biol. 2001, 2, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, Y. Endosymbiotic bacteria in insects: Their diversity and culturability. Microbes Environ. 2009, 24, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Bourtzis, K.; Miller, T.A. Insect Symbiosis, 1st ed.; Bourtzis, K., Miller, T.A., Eds.; CRC Press: Boca Raton, FL, USA, 2006; Volume 2, pp. 1–304. [Google Scholar]
- Moran, N.A. Bacterial menageries inside insects. Proc. Natl. Acad. Sci. USA 2001, 98, 1338–1340. [Google Scholar] [CrossRef]
- Bright, M.; Bulgheresi, S. A complex journey: Transmission of microbial symbionts. Nat. Rev. Microbiol. 2010, 8, 218–230. [Google Scholar] [CrossRef]
- Douglas, A. Phloem-sap feeding by animals: Problems and solutions. J. Exp. Bot. 2006, 57, 747–754. [Google Scholar] [CrossRef]
- Prado, S.S.; Almeida, R.P. Role of symbiotic gut bacteria in the development of Acrosternum hilare and Murgantia histrionica. Entomol. Exp. Appl. 2009, 32, 21–29. [Google Scholar] [CrossRef]
- Feldhaar, H. Bacterial symbionts as mediators of ecologically important traits of insect hosts. Ecol. Entomol. 2011, 36, 533–543. [Google Scholar] [CrossRef]
- Oliver, K.M.; Degan, P.H.; Burke, G.R.; Moran, N.A. Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu. Rev. Entomol. 2010, 55, 247–266. [Google Scholar] [CrossRef]
- Dion, E.; Polin, S.E.; Simon, J.C.; Outreman, Y. Symbiont infection affects aphid defensive behaviours. Biol. Lett. 2011, 7, 743–746. [Google Scholar] [CrossRef]
- Montllor, C.B.; Maxmen, A.; Purcell, A.H. Facultative bacterial endosymbionts benefit pea aphids Acyrthosiphon pisum under heat stress. Ecol. Entomol. 2002, 27, 189–195. [Google Scholar] [CrossRef]
- Russell, J.A.; Moran, N.A. Costs and benefits of symbiont infection in aphids: Variation among symbionts and across temperatures. Proc. R. Soc. B 2006, 273, 603–610. [Google Scholar] [CrossRef]
- Tsuchida, T.; Coga, R.; Shibao, H.; Matsumoto, T.; Fukatsu, T. Diversity and geographic distribution of secondary endosymbiotic bacteria in natural populations of the pea aphid, Acyrthosiphon pisum. Mol. Ecol. 2002, 11, 2123–2135. [Google Scholar] [CrossRef]
- Ferrari, J.; Darby, A.C.; Daniell, T.J.; Godfray, C.H.; Douglas, A.E. Linking the bacterial community in pea aphids with host-plant use and natural enemy resistance. Ecol. Entomol. 2004, 29, 60–65. [Google Scholar] [CrossRef]
- McLean, A.; van Ash, M.; Ferrari, J.; Godfray, C.H. Effects of bacterial secondary symbionts on host plant use in pea aphids. Proc. R. Soc. B 2011, 278, 760–766. [Google Scholar] [CrossRef]
- Moran, N.A.; Russel, J.A.; Koga, R.; Fukatsu, T. Evolutionary relationships of three new species of Enterobacteriaceae living as symbionts of aphids and other insects. Appl. Environ. Microbiol. 2005, 71, 3302–3310. [Google Scholar] [CrossRef]
- Scarborough, C.L.; Ferrari, J.; Godfray, C.H. Aphid protected from pathogen by endosymbiont. Science 2005, 310, 1781. [Google Scholar] [CrossRef]
- Łukasik, P.; Guo, H.; van Asch, M.; Ferrari, J.; Godfray, C.H. Protection against a fungal pathogen conferred by the aphid facultative endosymbionts Rickettsia and Spiroplasma is expressed in multiple host genotypes and species and is not influenced by co-infection with another symbiont. J. Evol. Biol. 2013, 26, 2654–2661. [Google Scholar] [CrossRef]
- Parker, B.J.; Sprag, C.; Altincicek, B.; Gerardo, N.M. Symbiont-mediated protection against fungal pathogens in pea aphids: A role for pathogen specificity? Appl. Environ. Microbiol. 2013, 79, 2455–2458. [Google Scholar] [CrossRef]
- Oliver, K.M.; Russell, J.A.; Moran, N.A.; Hunter, S.H. Facultative bacterial symbionts in aphids confer resistance to parasitic wasps. Proc. Natl. Acad. Sci. USA 2003, 100, 1803–1807. [Google Scholar] [CrossRef]
- Costopoulos, K.; Kovacs, J.L.; Kamins, A.; Gerardo, N.M. Aphid facultative symbionts reduce survival of the predatory lady beetle Hippodamia convergens. BMC Ecol. 2014, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Haine, E.R. Symbiont-mediated protection. Proc. Biol. Sci. R. Soc. B 2008, 275, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Oliver, K.M.; Campos, J.; Moran, N.A.; Hunter, M.S. Population dynamics of defensive symbionts in aphids. Proc. Biol. Sci. R. Soc. B 2008, 275, 293–299. [Google Scholar] [CrossRef]
- Ferrari, J.; Vavre, F. Bacterial symbionts in insects or the story of communities affecting communities. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 1389–1400. [Google Scholar] [CrossRef] [PubMed]
- Larsson, H. A crop loss model and economic thresholds for the grain aphid, Sitobion avenae (F.), in winter wheat in southern Sweden. Crop Prot. 2005, 24, 397–405. [Google Scholar] [CrossRef]
- Dedryver, C.-A.; Le Gallic, J.-F.; Haack, L.; Halkett, F.; Outreman, Y.; Simon, J.C. Seasonal and annual genotypic variation and the effect of climate on population genetic structure of the cereal aphid Sitobion avenae in northern France. Bull. Entomol. Res. 2008, 98, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Alkhedir, H.; Karlovsky, P.; Vidal, S. Effect of light intensity on colour morph formation and performance of the grain aphid Sitobion avenae F. (Homoptera: Aphididae). J. Insect Physiol. 2010, 56, 1999–2005. [Google Scholar] [CrossRef]
- Liu, X.; Yang, X.F.; Wang, C.Y.; Wang, J.Y.; Zang, H.; Ji, W.Q. Molecular mapping of resistance gene to English grain aphid (Sitobion avenae F.) in Triticum durum wheat line C273. Theor. Appl. Genet. 2012, 124, 287–293. [Google Scholar] [CrossRef]
- Ambethgar, V. Potential of entomopathogenic fungi in insecticide resistance management (IRM): A review. J. Biopest. 2009, 2, 177–193. [Google Scholar]
- Dewar, A.M.; Dewar, A.J.; Haylock, L.A.; Foster, S.P.; Williamson, M.S. Alternative insecticides to control cereal aphids, Sitobion avenae, that are resistant to pyrethroids. In Proceedings of the Crop Protection in Northern Britain, Dundee, UK, 25–26 February 2014; pp. 131–136. [Google Scholar]
- Mulé, R.; Sabella, G.; Robba, L.; Manachini, B. Systematic Review of the Effects of Chemical Insecticides on Four Common Butterfly Families. Front. Environ. Sci. 2017, 5, 32. [Google Scholar] [CrossRef]
- Hong, S.I.; Kim, K. Selection of entomopathogenic fungi for aphid control. J. Biosci. Bioeng. 2007, 104, 498–505. [Google Scholar]
- Lacey, L.A.; Frutos, R.; Kaya, H.K.; Vail, P. Insect pathogens as biological control agents: Do they have a future? Biol. Control 2001, 21, 230–248. [Google Scholar] [CrossRef]
- Goettel, M.; Eilenberg, J.; Glare, T. Entomopathogenic Fungi and their Role in Regulation of Insect Populations. In Insect Control: Biological and Synthetic Agents, 1st ed.; Lawrence, I., Gill, S., Eds.; Academic Press: London, UK, 2005; Volume 1, pp. 387–438. [Google Scholar]
- Meyling, N.V.; Eilenberg, J. Ecology of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae in temperate agroecosystems: Potential for conservation biological control. Biol. Control 2007, 43, 145–155. [Google Scholar] [CrossRef]
- EC (European Commission). Commission Regulation (EU) 2021/2010 of 17 November 2021 amending Regulation (EC) No 1185/2009 of the European Parliament and of the Council concerning statistics on pesticides, as regards the list of active substances. Off. J. Eur. Union 2021, L 410, 1–47. [Google Scholar]
- Pathan, A.A.K.; Devi, K.U.; Vogel, H.; Reineke, A. Analysis of differential gene expression in the generalist entomopathogenic fungus Beauveria bassiana (Bals.) Vuillemin grown on different insect cuticular extracts and synthetic medium through cDNA-AFLPs. Fungal Genet. Biol. 2007, 44, 1231–1241. [Google Scholar] [CrossRef]
- De Faria, M.R.; Wraight, S.P. Mycoinsecticides and mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biol. Control 2007, 43, 237–256. [Google Scholar] [CrossRef]
- Kim, J.J.; Jeong, G.; Han, J.H.; Lee, S. Biological control of aphid using fungal culture and culture filtrates of Beauveria bassiana. Mycobiology 2013, 41, 221–224. [Google Scholar] [CrossRef]
- Jackson, M.A.; Dunlap, C.A.; Jaronski, S.T. Ecological considerations in producing and formulating fungal entomopathogens for use in insect biocontrol. Bio. Control 2010, 55, 129–145. [Google Scholar] [CrossRef]
- Alkhedir, H.; Karlovsky, P.; Vidal, S. Relationship between water soluble carbohydrate content, aphid endosymbionts and clonal performance of Sitobion avenae on cocksfoot cultivars. PLoS ONE 2013, 8, e54327. [Google Scholar] [CrossRef]
- Leonardo, T.E. Removal of a specialization-associated symbiont does not affect aphid fitness. Ecol. Lett. 2004, 7, 461–468. [Google Scholar] [CrossRef]
- Simon, J.C.; Boutin, S.; Tsuchida, T.; Koga, R.; Le Gallic, J.F.; Frantz, A.; Outreman, Y.; Fukatsu, T. Facultative symbiont infections affect aphid reproduction. PLoS ONE 2011, 6, e21831. [Google Scholar] [CrossRef] [PubMed]
- Koga, R.; Tsuchida, T.; Sakurai, M.; Fukatsu, T. Selective elimination of aphid endosymbionts: Effects of antibiotic dose and host genotype, and fitness consequences. FEMS Microbiol. Ecol. 2007, 60, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Rangasamy, M.; Tan, S.Y.; Wang, H.; Siegfried, B.D. Evaluation of five methods for total DNA extraction from western corn rootworm beetles. PLoS ONE 2010, 5, e11963. [Google Scholar] [CrossRef] [PubMed]
- Vidal, S.; Jaber, L.R. Entomopathogenic fungi as endophytes: Plant-endophyte-herbivore interactions and prospects for use in biological control. Curr. Sci. 2015, 109, 46–54. [Google Scholar]
- Quesada-Moraga, E.; Muñoz-Ledesma, F.J.; Santiago-Álvarez, C. Systemic protection of Papaver somniferum L. against Iraella luteipes (Hymenoptera: Cynipidae) by an endophytic strain of Beauveria bassiana (Ascomycota: Hypocreales). Environ. Entomol. 2009, 38, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Eckard, S.; Ansari, M.A.; Bacher, S.; Butt, T.M.; Enkerli, J.; Grabenweger, G. Virulence of in vivo and in vitro produced conidia of Metarhizium brunneum strains for control of wireworms. Crop Prot. 2014, 64, 137–142. [Google Scholar] [CrossRef]
- Kölliker, U.; Biasio, L.; Jossi, W. Potential control of Swiss wireworms with entomopathogenic fungi. Potential Control. Swiss Wireworms Entomopathog. Fungi 2011, 66, 517–520. [Google Scholar]
- Johnson, D.M.; White, R.L.; Pereira, R.M.; Geden, C.J. Beauveria bassiana culturing and harvesting for bioassays with house flies. J. Insect Sci. 2020, 20, 14. [Google Scholar] [CrossRef]
- Strasser, H.; Forer, A.; Schinner, F. Development of media for the selective isolation and maintenance of virulence of Beauveria brongniartii. In Proceedings of the 3rd International Workshop on Microbial Control of Soil Dwelling Pests, Lincoln, New Zealand, 21–23 February 1996. [Google Scholar]
- Statistix 8.1, (Version 8.1); Analytical Software: Tallahassee, FL, USA, 2003.
- Łukasik, P.; van Asch, M.; Guo, H.; Ferrari, J.; Godfray, C.H. Unrelated facultative endosymbionts protect aphids against a fungal pathogen. Ecol. Lett. 2013, 16, 214–218. [Google Scholar] [CrossRef]
- Castillo, M.A.; Moya, P.; Hernández, E.; Primo-Yufera, E. Susceptibility of Ceratitis capitata Wiedemann (Diptera, Tephritidae) to entomopathogenic fungi and their extracts. Biol. Control 2000, 19, 274–282. [Google Scholar] [CrossRef]
- Hansen, A.K.; Vorburger, C.; Moran, N.A. Genomic basis of endosymbiont-conferred protection against an insect parasitoid. Genome Res. 2012, 22, 106–114. [Google Scholar] [CrossRef]
- Guay, J.F.; Boudreault, S.; Michaud, D.; Cloutier, C. Impact of environmental stress on aphid clonal resistance to parasitoids: Role of Hamiltonella defensa bacterial symbiosis in association with a new facultative symbiont of the pea aphid. J. Insect Physiol. 2009, 55, 919–926. [Google Scholar] [CrossRef]
- Cayetano, L.; Vorburger, C. Genotype-by-genotype specificity remains robust to average temperature variation in an aphid/endosymbiont/parasitoid system. J. Evol. Biol. 2013, 26, 1603–1610. [Google Scholar] [CrossRef]
- Romo, C.M.; Tylianakis, J.M. Elevated temperature and drought interact to reduce parasitoid effectiveness in suppressing hosts. PLoS ONE 2013, 8, e58136. [Google Scholar] [CrossRef]
- Schmid, M.; Sieber, R.; Zimmermann, Y.S.; Vorburger, C. Development, specificity and sublethal effects of symbiont-conferred resistance to parasitoids in aphids. Funct. Ecol. 2012, 26, 207–215. [Google Scholar] [CrossRef]
- Hajek, A.E.; Delalibera, I. Fungal pathogens as classical biological control agents against arthropods. Bio. Control 2010, 55, 147–158. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).