Simple Summary
The genus Homoneura is the most speciose genus of Lauxaniidae. However, no phylogenetic study of Homoneura has been published using morphological data, despite the high diversity and ecological significance. Therefore, we present the first morphological phylogeny of Homoneura. The monophyly of the genus Homoneura and the subgenus Homoneura is not supported. The monophyly of the subgenera Euhomoneura and Neohomoneura is supported, as well as the sister relationship between the subgenera Chaetohomoneura and Neohomoneura. These results provide a comprehensive framework and information toward future research of this genus.
Abstract
The genus Homoneura comprises over 700 described species in eight known subgenera distributed worldwide and has the highest species richness of Lauxaniidae. Five subgenera and more than 200 species have currently been recorded from China. Despite its high diversity, the monophyly of Homoneura and its subgenera, and the phylogenetic relationships among its subgenera remain to be investigated. One maximum-parsimony tree was generated based on 105 morphological characters scored from 24 species, representing all five subgenera of Homoneura recorded from China. The results did not support the monophyly of the genus Homoneura and subgenus Homoneura. The subgenus Chaetohomoneura is a sister to subgenus Neohomoneura. The monophyly of the subgenera Euhomoneura and Neohomoneura is supported. Much of the current classification of the genus Homoneura needs a revision before taxonomy can reflect natural groupings.
1. Introduction
The family Lauxaniidae is species-rich and globally distributed, with three subfamilies, more than 170 genera, and nearly 2100 described species [1]. Lauxaniids have a variety of habitats [2,3], mainly scavenging and fungus feeding [4,5], and have the habit of visiting flowers [6,7]. They play a very important role in the ecological system, degrading organic matter, plant pollination, and maintaining ecological balance [8,9,10]. In addition, lauxaniids are sensitive to environmental change. They have been used as an indicator organism for environmental change assessments of farmland ecosystems in Europe and have also become one of the research hotspots of terrestrial ecosystem assessment indicators [11].
The genus Homoneura is the most speciose genus of Lauxaniidae. Currently, it contains eight subgenera and more than 700 described species worldwide, among which five subgenera and more than 200 species are recorded from China [1]. It is also one of the most diverse genera of the Acalyptratae. Homoneura is widely distributed in all major animal geographical areas except for the Neotropical region.
Stuckenberg hypothesized the relationship among 19 genera of the subfamily Homoneurinae based on morphological characters and divided 19 genera into three groups [12]. Kim used numerical methods to cluster Australian members of the genera Homoneura, Trypeisoma, and allied genera by their similarities in morphology [13]. In the only existing molecular phylogenetic study of the generic level relationships of Lauxaniidae, Shi et al. explored the phylogeny of the subgenus Homoneura based on two mitochondrial and two nuclear genes [14]. The monophyly of the subgenus Homoneura was not recovered. No phylogenetic study of the genus Homoneura has been published using morphological data. Despite the high diversity and ecological significance, the monophyly of the genus Homoneura and its subgenera, and the phylogenetic relationships among the subgenera remain to be investigated.
In this study, we use 105 morphological characters to reconstruct the phylogeny based on all five subgenera of Homoneura that are recorded from China, aiming to (1) test the monophyly of the genus Homoneura; (2) test the monophyly of Chinese subgenera of Homoneura; (3) investigate relationships among five Chinese subgenera of Homoneura.
2. Materials and Methods
2.1. Morphological Study and Terminology
General terminology follows Cumming & Wood and Gaimari & Silva [15,16]. Genitalia preparations were made by removing and macerating the apical portion of the abdomen in cold saturated NaOH for six hours, then rinsing and neutralizing them for dissection and study. After examination in glycerine, they were transferred to fresh glycerine and stored in a microvial pinned below the specimen or moved to an ethanol tube together with the wet specimens. Most characters were illustrated using photographs and line drawings. Photographs were taken using a Canon EOS6D microscope (Canon, Tokyo, Japan) and stacked using HELICO FOCUS v7.0.2.0 (Helicon Soft, Kharkiv, Ukraine). Line drawings were drawn with Adobe Illustrator 2021 v25.2.1 (Adobe, San Jose, CA, USA).
2.2. Specimens Examined and Morphological Characters
In total, 24 lauxaniid species were selected in the analysis, including 17 Homoneura species representing five subgenera. Two species of Lauxaniinae: Minettia (frendelia) longipennis and Pachycerina decemlineata (Figure 1C), and five species of Homoneurinae: Cestrotus liui (Figure 1B), Dioides incurvatus, Noonamyia umbrellata (Figure 1A), Phobeticomyia motuoensis, and Prosopophorella yoshiyasui were selected as outgroup taxa. Table S1 shows the terminals included in the cladistic analysis.
On the basis of our survey, 105 morphological characters obtained from adults from the head (28 characters, Figure 2), thorax (10 characters, Figure 3A–F), legs (9 characters, Figure 3G–L), wing (17 characters, Figure 4), abdomen (7 characters, Figure 5), male genitalia (29 characters, Figure 6, Figure 7 and Figure 8), and female genitalia (5 characters, Figure 9) were numerically coded (Table S2). Eighty-seven characters are binary and 18 are multistate. All characters were treated as unordered and with equal weight. Missing character states were coded with (?), and inapplicable states were scored as (–).
The studied specimens are deposited in the Insect Collection of Henan University of Science and Technology (HAUST).

Figure 1.
Adults of species of Lauxaniidae. (A): Noonamyia umbrellata; (B): Cestrotus liui; (C): Pachycerina decemlineata; (D): Homoneura (Euhomoneura) shatalkini; (E): Homoneura (Homoneura) picta; (F): Homoneura (Homoneura) flavida; (G): Homoneura (Homoneura) dorsacerba; (H): Homoneura (Homoneura) noticomata.
Figure 1.
Adults of species of Lauxaniidae. (A): Noonamyia umbrellata; (B): Cestrotus liui; (C): Pachycerina decemlineata; (D): Homoneura (Euhomoneura) shatalkini; (E): Homoneura (Homoneura) picta; (F): Homoneura (Homoneura) flavida; (G): Homoneura (Homoneura) dorsacerba; (H): Homoneura (Homoneura) noticomata.

2.3. List of Characters Used in the Cladistic Analysis
Head:
- Frons: (0) flat; (1) uplifted; (2) sunken.
- Spot on anterior margin of frons: (0) absent; (1) present.
- Width of ventral margin of face/height of gena: (0) ≥ 3X; (1) < 3X.
- Length of gena/length of eye: (0) ≤ 1; (1) > 1.
- Color of proboscis: (0) black; (1) brown to yellow (Figure 2N).

Figure 2.
Hand characters. (A,N): Homoneura (Homoneura) picta; (B–D,O): Homoneura (Homoneura) flavida; (E): Homoneura (Minettioides) orientis; (F,I): Homoneura (Euhomoneura) yanqingensis; (G): Homoneura (Neohomoneura) tricuspidata; (H,J,K): Prosopophorella yoshiyasui; (L,P): Minettia (Frendelia) longipennis; (M): Pachycerina decemlineata.
Figure 2.
Hand characters. (A,N): Homoneura (Homoneura) picta; (B–D,O): Homoneura (Homoneura) flavida; (E): Homoneura (Minettioides) orientis; (F,I): Homoneura (Euhomoneura) yanqingensis; (G): Homoneura (Neohomoneura) tricuspidata; (H,J,K): Prosopophorella yoshiyasui; (L,P): Minettia (Frendelia) longipennis; (M): Pachycerina decemlineata.

Thorax:
- 29.
- 30.
- 31.
- 32.
- 33.

Figure 3.
Thoracic and leg characters. (A,G,H): Homoneura (Homoneura) flavida; (B): Homoneura (Homoneura) picta; (C): Pachycerina decemlineata; (D,I,J): Homoneura (Euhomoneura) yanqingensis; (E,K): Homoneura (Neohomoneura) tricuspidata; (F): Homoneura (Minettioides) orientis; (L): Minettia (Frendelia) longipennis.
Figure 3.
Thoracic and leg characters. (A,G,H): Homoneura (Homoneura) flavida; (B): Homoneura (Homoneura) picta; (C): Pachycerina decemlineata; (D,I,J): Homoneura (Euhomoneura) yanqingensis; (E,K): Homoneura (Neohomoneura) tricuspidata; (F): Homoneura (Minettioides) orientis; (L): Minettia (Frendelia) longipennis.

- 34.
- 35.
- 36.
- Prescutellar acrostichal seta: (0) present (Figure 3A); (1) absent.
- 37.
- Intra-alar seta: (0) present; (1) absent.
- 38.
Leg:
- 39.
- Length of leg/length of body: (0) ≤ 1; (1) > 1.
- 40.
- 41.
- Ctenidium short seta in fore femur: (0) absent; (1) no more than ten; (2) more than ten (Figure 3G).
- 42.
- Anterior seta in mid femur: (0) more than six; (1) no more than six (Figure 3I).
- 43.
- Posterior ventral seta in mid femur: (0) absent; (1) present.
- 44.
- 45.
- 46.
- Preapical anterior dorsal seta in hind femur; (0) present (Figure 3H); (1) absent.
- 47.
- Anteroventral seta in hind femur: (0) absent; (1) present (Figure 3H).
Wing:
- 48.
- 49.
- Area of transparent area or light-yellow area of wing/area of wing spot area: (0) > 1; (1) ≤ 1.
- 50.
- Area of transparent or light-yellow area above wing R4+5/area of wing spot above R4+5: (0) > 1; (1) ≤ 1.
- 51.
- 52.
- 53.
- 54.
- 55.
- 56.
- 57.
- 58.
- 59.
- 60.
- 2nd (between R1 and R2+3) section/3rd (between R2+3 and R4+5) section: (0) ≥ 3X; (1) < 3X.
- 61.
- 3rd (between R2+3 and R4+5) section/4th (between R4+5 and M1) section: (0) ≥ 1.5X; (1) < 1.5X.
- 62.
- Length of the ultimate section of CuA1/length of the penultimate section of CuA1; (0) < 1/5; (1) ≥ 1/5.
- 63.
- 64.
- Color of knob part of haltere: (0) black; (1) brown or yellow.

Figure 4.
Wing characters. (A): Homoneura (Homoneura) picta; (B): Noonamyia umbrellata; (C): Homoneura (Homoneura) posterotricuspis; (D): Prosopophorella yoshiyasui; (E): Homoneura (Homoneura) flavida; (F): Homoneura (Neohomoneura) zengae; (G): Minettia (Frendelia) longipennis; (H): Homoneura (Euhomoneura) yanqingensis.
Figure 4.
Wing characters. (A): Homoneura (Homoneura) picta; (B): Noonamyia umbrellata; (C): Homoneura (Homoneura) posterotricuspis; (D): Prosopophorella yoshiyasui; (E): Homoneura (Homoneura) flavida; (F): Homoneura (Neohomoneura) zengae; (G): Minettia (Frendelia) longipennis; (H): Homoneura (Euhomoneura) yanqingensis.

Abdomen:
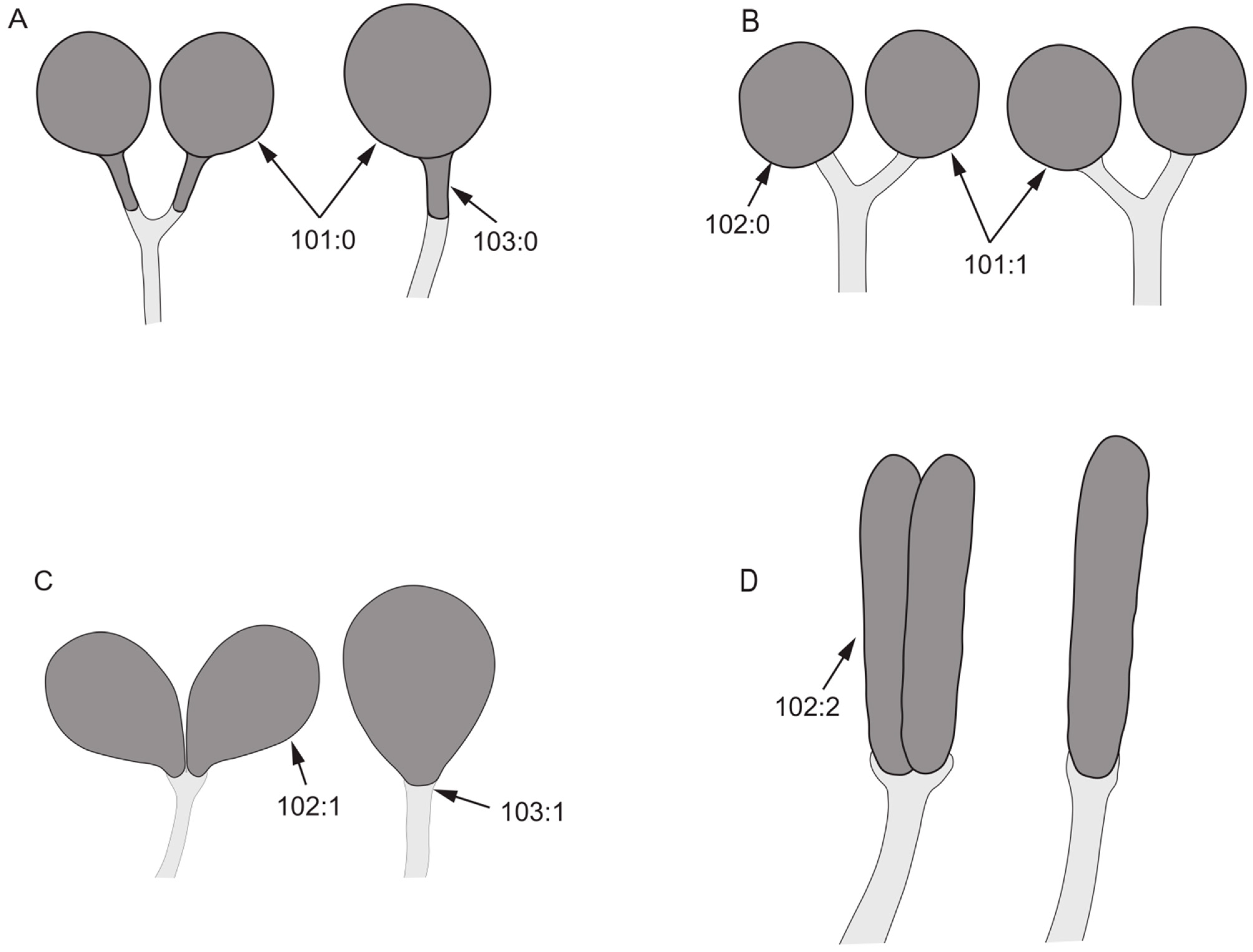
- 65.
- Color of tergites 3–6: (0) black; (1) brown or yellow.
- 66.
- 67.
- 68.
- 69.
- 70.
- 71.

Figure 5.
Abdomen characters. (A,D): Homoneura (Homoneura) picta; (B): Homoneura (Neohomoneura) tricuspidata; (C,F): Homoneura (Euhomoneura) yanqingensis; (E): Homoneura (Minettioides) orientis.
Figure 5.
Abdomen characters. (A,D): Homoneura (Homoneura) picta; (B): Homoneura (Neohomoneura) tricuspidata; (C,F): Homoneura (Euhomoneura) yanqingensis; (E): Homoneura (Minettioides) orientis.

Male genitalia:
- 72.
- 73.
- Length of dorsal margin of syntergosternite/length of posterior margin of syntergosternite: (0) < 1/2; (1) ≥ 1/2.
- 74.
- 75.
- 76.
- Length of dorsal margin of epandrium/length of ventral margin of epandrium: (0) > 1/2; (1) ≤ 1/2.
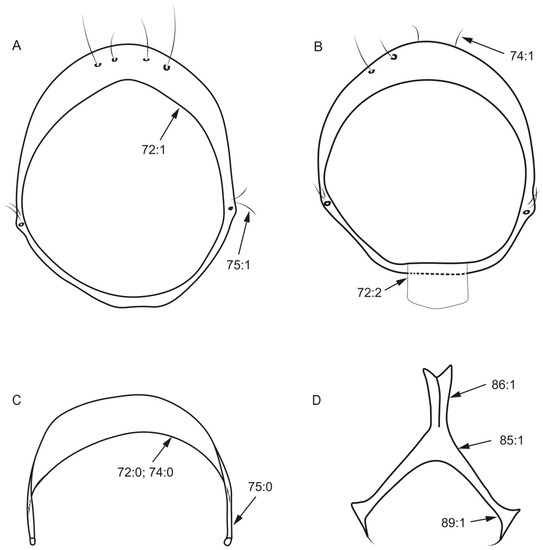
Figure 6.
Syntergosternite and hypandrium characters. (A): Homoneura (Homoneura) dorsacerba; (B): Homoneura (Homoneura) posterotricuspis; (C): Homoneura (Homoneura) procerula; (D): Cestrotus liui.
Figure 6.
Syntergosternite and hypandrium characters. (A): Homoneura (Homoneura) dorsacerba; (B): Homoneura (Homoneura) posterotricuspis; (C): Homoneura (Homoneura) procerula; (D): Cestrotus liui.
- 77.
- 78.
- 79.
- 80.
- 81.
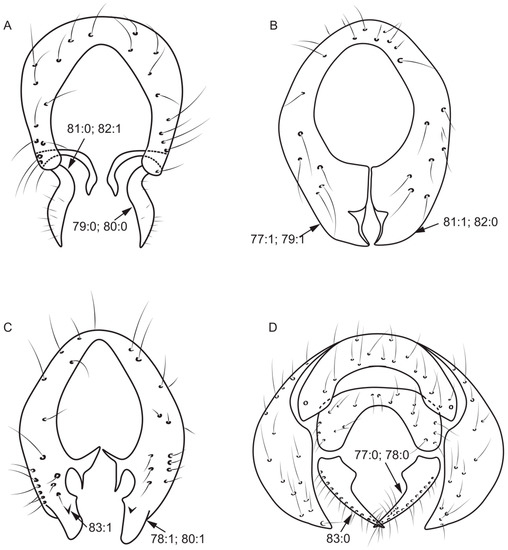
Figure 7.
Epandrium characters. (A): Homoneura (Homoneura) trispina; (B): Homoneura (Homoneura) dorsacerba; (C): Homoneura (Homoneura) posterotricuspis; (D): Pachycerina decemlineata.
Figure 7.
Epandrium characters. (A): Homoneura (Homoneura) trispina; (B): Homoneura (Homoneura) dorsacerba; (C): Homoneura (Homoneura) posterotricuspis; (D): Pachycerina decemlineata.
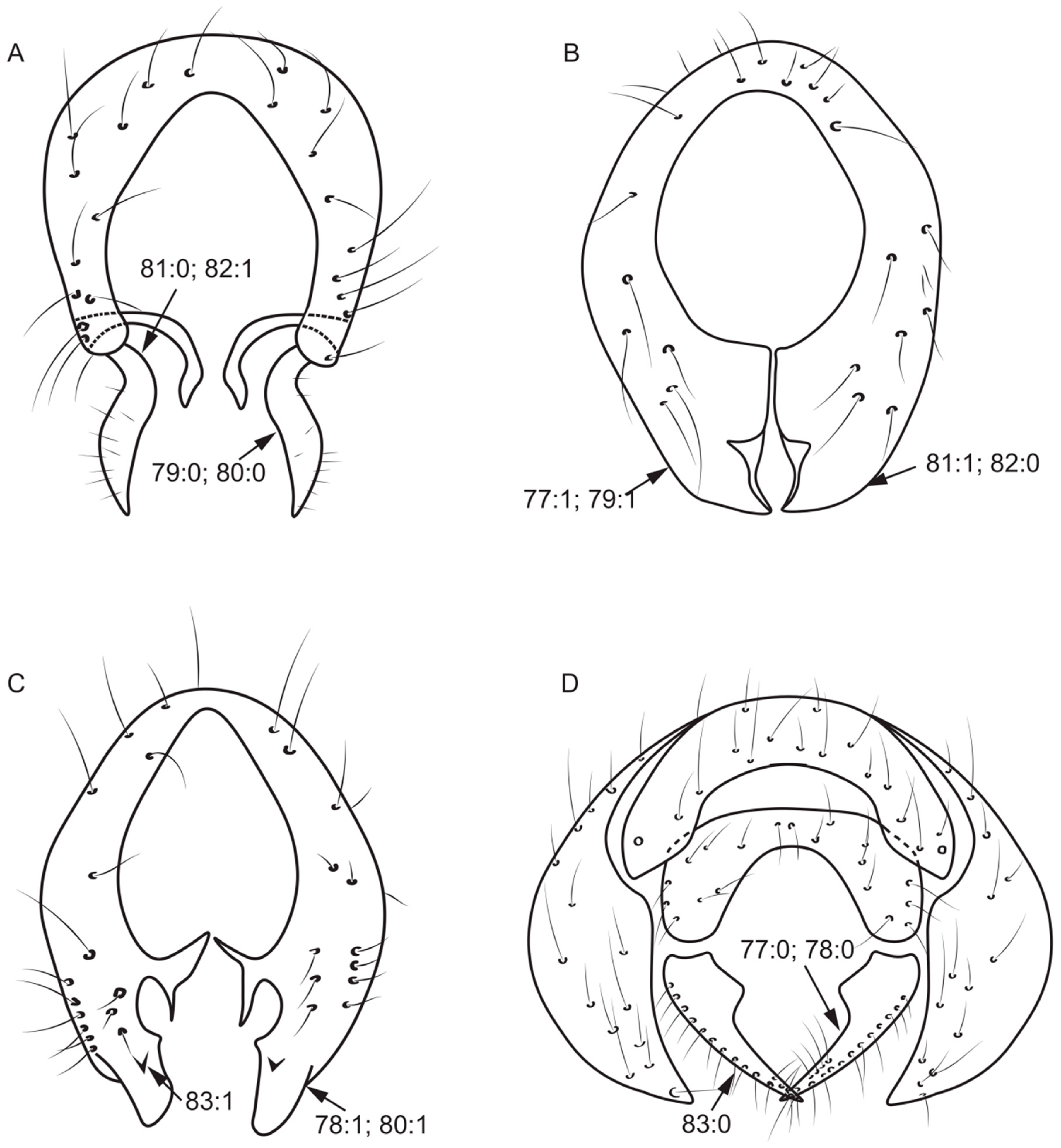
- 82.
- 83.
- 84.
- Surstylus: (0) without seta; (1) with seta.
- 85.
- 86.
- 87.
- Both sides anterior margin of hypandrium: (0) without inner processes; (1) with inner processes (Figure 8A).
- 88.
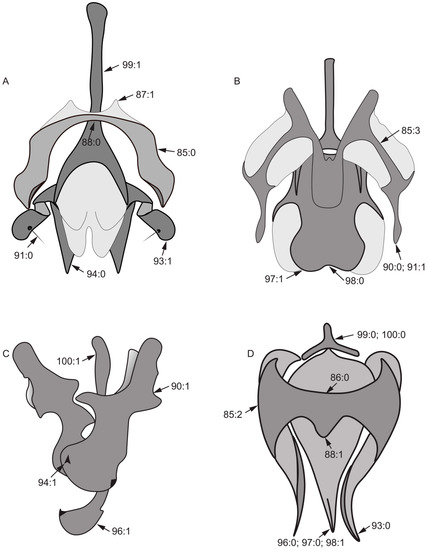
Figure 8.
Aedeagal complex characters. (A): Noonamyia umbrellata; (B): Homoneura (Homoneura) beckeri; (C): Pachycerina decemlineata; (D): Homoneura (Homoneura) procerula.
Figure 8.
Aedeagal complex characters. (A): Noonamyia umbrellata; (B): Homoneura (Homoneura) beckeri; (C): Pachycerina decemlineata; (D): Homoneura (Homoneura) procerula.
- 89.
- Both sides posterior margin of hypandrium: (0) without ventral process; (1) with ventral process (Figure 6D).
- 90.
- 91.
- 92.
- Length of gonite/length of phallus: (0) ≥ 1/2; (1) < 1/2.
- 93.
- 94.
- 95.
- Lateral view of the top of phallus: (0) bent; (1) straight.
- 96.
- 97.
- 98.
- 99.
- 100.
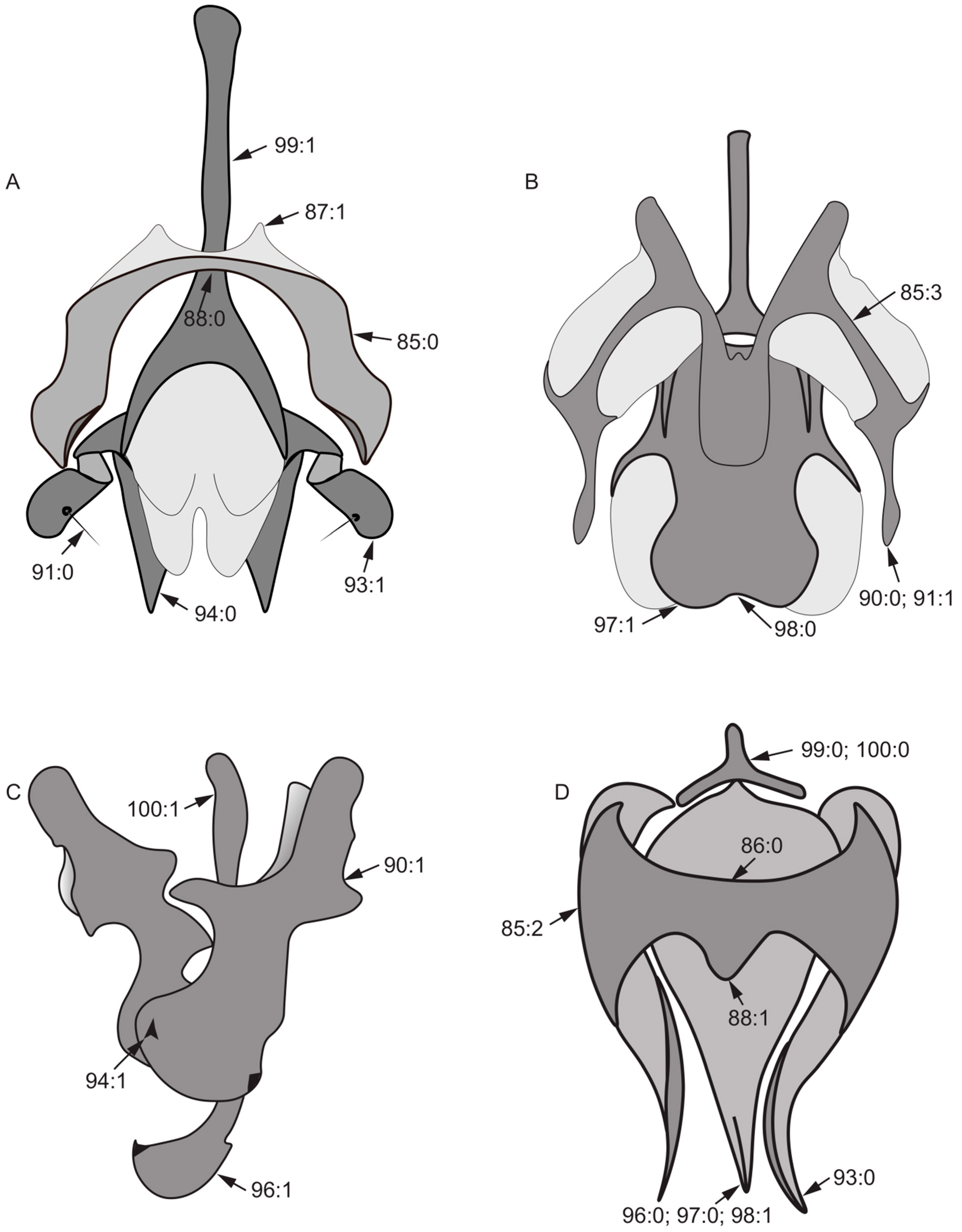
Female genitalia:
- 101.
- 102.
- 103.
- 104.
- Both sides of posterior margin of sternite 8: (0) with process; (1) without process.
- 105.
- Posterior margin of tergite 9: (0) uplifted; (1) sunken.
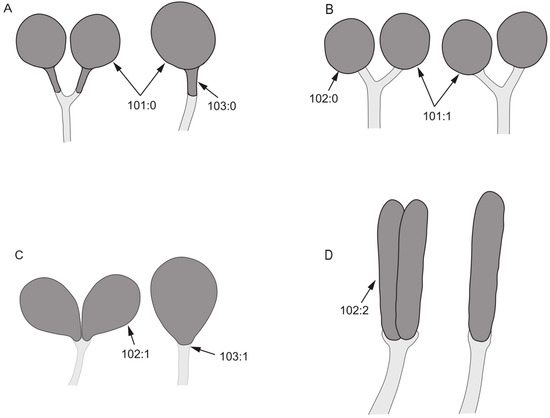
Figure 9.
Spermathecae characters. (A): Minettia (Frendelia) longipennis; (B): Noonamyia umbrellata; (C): Cestrotus liui; (D): Homoneura (Minettioides) orientis.
Figure 9.
Spermathecae characters. (A): Minettia (Frendelia) longipennis; (B): Noonamyia umbrellata; (C): Cestrotus liui; (D): Homoneura (Minettioides) orientis.
2.4. Cladistic Analysis
The phylogenetic reconstruction was conducted using maximum-parsimony analysis using NONA v2.0 (Goloboff, P.A., Tucuman, Argentina) with a heuristic search by 1000 replications, and TNT with 1000 random-addition traditional searches [17,18]. All characters were initially equally weighted. Branch support values were verified through bootstrap analyses on NONA 2.0 with 100 replications. The Bremer support value or decay index for the resultant strict consensus tree was calculated using TNT [19]. The unambiguous characters were mapped on the tree using WinClada version v1.00.08 (Nixon, K.C., Ithaca, NY, USA) [20].
3. Results
3.1. Phylogenetic Analysis
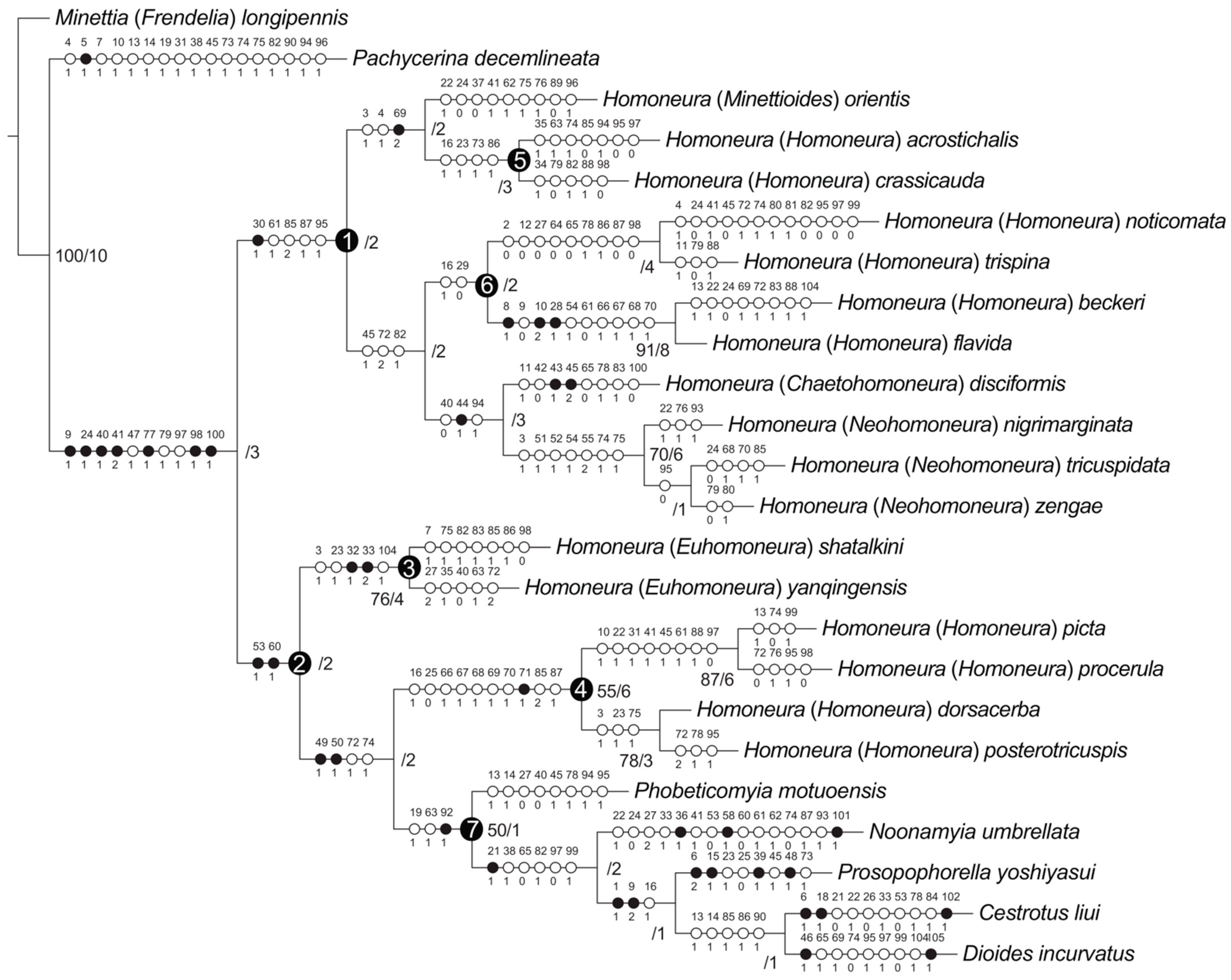
Maximum-parsimony analyses yielded one maximum-parsimonious tree [branch length = 337, consistency index (CI) = 0.36, retention index (RI) = 0.56]. The maximum-parsimony tree is shown in Figure 10, Bootstrap values (BS) and Bremer support (B) values are presented next to the nodes.
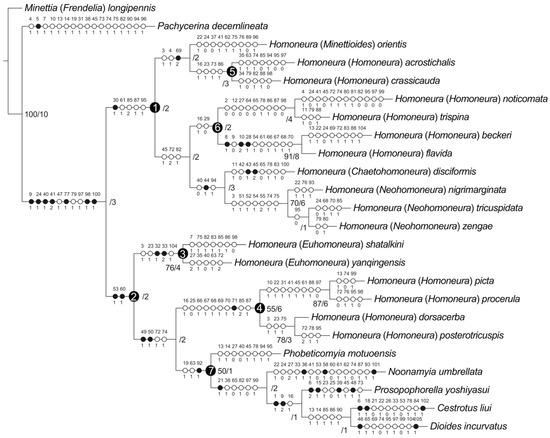
Figure 10.
Phylogenetic relationships of Homoneura. Synapomorphies are marked by filled circles and homoplasious characters by open circles. The character numbers and states are placed above and below the circles, respectively. Bremer support: left, bootstrap values > 50%; right, Bremer support values.
3.2. Subfamily Homoneurinae
The monophyly of the subfamily Homoneurinae was supported (BS = −/B = 3) by seven synapomorphies: between the middle of frons and fronto-orbital setae with two spots (9:1), length of the first flagellomere/width of the first flagellomere < 2X (24:1), posterior ventral seta in fore femur no less than five (40:1), ctenidium short seta in fore femur with more than ten (41:2), surstylus not separated from epandrium (77:1), phallus without distinct apical concave (98:1), and width of the base of aedeagal apodeme/width of the middle of aedeagal apodeme ≤ 2X (100:1).
Two major lineages were recovered within Homoneurinae. The clade 1 was supported by one synapomorphy: mesonotum without stripe (30:1), and four homoplasious characters: wing 3rd (between R2+3 and R4+5) section/4th (between R4+5 and M1) section < 1.5X (61:1), hypandrium H-shaped (85:2), both sides anterior margin of hypandrium with inner processes (87:1), and lateral view of the top of phallus straight (95:1). The clade 2 was supported by two synapomorphies: crossvein r-m with spot (53:1), and wing 2nd (between R1 and R2+3) section/3rd (between R2+3 and R4+5) section < 3X (60:1).
3.3. The Genus Homoneura and Allies
The genus Homoneura was not recovered as monophyletic, and three graded clades (clades 1, 3, and 4) were detected instead. Clade 1 included subgenera Minettioides, Neohomoneura, and Chaetohomoneura, and part of the subgenus Homoneura (Homoneura (H.) acrostichali, Homoneura (H.) crassicauda, Homoneura (H.) noticomata, Homoneura (H.) trispina, Homoneura (H.) beckeri, and Homoneura (H.) flavida). Clade 3 represented subgenus Euhomoneura and was supported by two synapomorphies: pre-sutural dorsocentral seta present (32:1), and two post-sutural dorsocentral setae (33:2). Clade 4 included another part of the subgenus Homoneura (Homoneura (H.) picta, Homoneura (H.) procerula, Homoneura (H.) dorsacerba, and Homoneura (H.) posterotricuspis), which was supported by one synapomorphy: side of tergite 6 without spot (71:1).
The other five included the Homoneurinae genera, Cestrotus, Dioides, Noonamyia, Phobeticomyia, and Prosopophorella, forming a monophyletic group (clade 7) and sister to clade 4 (nested within the genus Homoneura). The monophyly of clade 7 was supported by one synapomorphy: length of gonite/length of phallus < 1 (92:1), and two homoplasious characters: length of gena/length of eye > 1 (19:1), and crossvein r-m located behind the middle of the discal cell (63:1).
3.4. Monophyly and Relationships among Chinese Subgenera of Homoneura
The monophyly of the subgenera Neohomoneura and Euhomoneura was supported, while the subgenus Homoneura was recovered as polyphyletic (clades 4, 5, and 6). Due to the limited taxon sampling, the monophyly of the subgenera Minettioides and Chaetohomoneura could not be tested.
Within clade 1, Minettioides + clade 5 was sister to clade 6 + (Chaetohomoneura + Neohomoneura). The monophyly of Minettioides + clade 5 was supported by one synapomorphy: side of tergite 5 with circular spot (69:2), and two homoplasious characters: ocellar triangle brown to yellow (3:1), and length of ocellar seta/length of anterior fronto-orbital seta < 1 (4:1). The sister relationship of Chaetohomoneura and Neohomoneura was supported by one synapomorphy: mid tibia with posterior seta (44:1), and two homoplasious characters: posterior ventral seta in fore femur no less than five (40:0), and phallus with thorn or sharp process (94:1). The monophyly of Neohomoneura was supported by seven homoplasies: ocellar triangle brown to yellow (3:1), spot on R2+3 no longer than half the length of R2+3 (51:1), tip of R4+5 with one spot and it is not longer than half of the top of R4+5 (52:1), crossvein dm-cu with spot (54:1), tip of M1 with one spot and it is longer than half of the top of M1 (55:2), dorsal margin of syntergosternite with short hair (74:1), and syntergosternite around the spiracle with short hair (75:1).
Within clade 2, Euhomoneura is sister to clade 4 + clade 7. The monophyly of Euhomoneura was supported by pre-sutural dorsocentral seta present (32:1), and two post-sutural dorsocentral setae (33:2). The sister relationship of clade 4 and clade 7 was supported by two synapomorphies: area of transparent area or light-yellow area of wing/area of wing spot area ≤ 1 (49:1), and area of transparent or light-yellow area above wing R4+5/area of wing spot above R4+5 ≤ 1 (50:1). The monophyly of clade 4 was supported by side of tergite 6 without spot (71:1).
4. Discussion
Five non-Homoneura Homoneurinae genera were included in the present study intended to be an outgroup, but deeply nested into Homoneura based on four synapomorphies on the wing: spot on crossvein r-m absent (53:1), wing 2nd (between R1 and R2+3) section/3rd (between R2+3 and R4+5) section < 3X (60:1), area of transparent area or light-yellow area of wing/area of wing spot area ≤ 1 (49:1), and area of transparent or light-yellow area above wing R4+5/area of wing spot above R4+5 ≤ 1 (50:1). The monophyly of genus Homoneura, therefore, was not supported by our analysis.
Sasakawa proposed the combination of pre-sutural dorsocentral seta absent and post-sutural dorsocentral seta three (dorsocentral setae 0 + 3) as the synapomorphy of subgenus Homoneura [21]. We found that pre-sutural dorsocentral seta present (32:1) was a synapomorphy of Euhomoneura, and pre-sutural dorsocentral seta absent was pleiomorphic and found in the remainder of the included subgenera. Multiple statuses were proposed in the present study regarding the number and location of post-sutural dorsocentral setae. Post-sutural dorsocentral setae three, firstst post-sutural dorsocentral seta behind the transversal suture (33:0) is plesiomorphic and found in non-Euhomoneura Homoneura subgenera, whereas post-sutural dorsocentral setae three, first post-sutural dorsocentral seta in the transversal suture (33:1) occurs in Noonamyia umbrellata and Cestrotus liui; post-sutural dorsocentral setae two (33:2) is a synapomorphy of Euhomoneura. Therefore, dorsocentral setae 0 + 3 cannot be an effective synapomorphy to support the monophyly of subgenus Homoneura.
The non-monophyly of the genus Homoneura and the subgenus Homoneura was also detected based on molecular data. Shi et al. obtained two mitochondrial genes (COI, 16S-rRNA) and two nuclear genes (Elongation factor 1-α, 28S-rRNA), and reconstructed gene trees using four different methods (NJ, ME, MP, and ML). Among 16 resulted gene trees, the monophyly of genus Homoneura and subgenus Homoneura was never recovered (they always had Minettia, or Sapromyza, or both nested) [14].
The sister relationship between the subgenera Chaetohomoneura and Neohomoneura was also suggested by Sasakawa [21]. Sasakawa proposed two potential ‘plesiomorphic’ characters to support Chaetohomoneura + Neohomoneura, among which mid tibia with posterior seta (44:1) was found to be the synapomorphy by our current analysis. Another character, acrostichal seta no less than seven rows (34:0) was found to be homoplastic in multiple species of the subgenera Minettioides and Homoneura. To alleviate the difficulty of distinguishing these two subgenera, Shi et al. summarized three characters: Chaetohomoneura with two supra-alar setae, four strong apical ventral setae on mid tibia, and posterior ventral setae often present in mid femur; Neohomoneura with one supra-alar seta, three strong apical ventral setae on mid tibia, and posterior ventral setae often absent from mid femur [22]. We found that additional characters could help distinguish these two subgenera: Chaetohomoneura with posterior ventral seta in mid femur present, and Neohomoneura with wing with four spots, separately on tip of R2+3, R4+5, M1, and crossvein dm-cu.
Sasakawa suggested intra-alar seta absent as a synapomorphy to support the monophyly of subgenus Euhomoneura + subgenus Homoneura [21]. Based on the present analysis, we found that the character is plesiomorphic on our tree and present in all species of non-Minettioides Chinese subgenera of the genus Homoneura. The synapomorphies support the monophyly of subgenera Euhomoneura, which is consistent with Sasakawa (one pre-sutural dorsocentral setae (32:1) and two post-sutural dorsocentral setae (33:2)).
5. Conclusions
This study presents the first morphological phylogeny of Homoneura, based on 105 characters of adults and 24 species representing all five subgenera of Homoneura recorded from China, underpinning our understanding of the phylogenetic relationships in the group.
Our results show that the monophyly of the genus Homoneura and subgenus Homoneura is not supported. Additionally, the results show that the monophyly of the subgenera Euhomoneura and Neohomoneura is supported, as well as the sister relationship between the subgenera Chaetohomoneura and Neohomoneura. Due to our regional taxon sampling, our result are premature to propose a new classification for genus or subgenus Homoneura, but we discovered the urgent need to revise this diverse group. Future studies with global taxon sampling, morphological evidence from multiple life stages, and molecular data are needed to reconstruct the phylogeny of Homoneurinae and revise the classification.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects13080665/s1, Table S1: The species studied, Table S2: Morphological dataset used for the analysis of the phylogeny.
Author Contributions
Methodology, C.K., W.L. and X.L.; software, C.K. and X.L.; validation, C.K. and X.L.; formal analysis, C.K., W.L. and X.L.; investigation, K.F., S.Z., C.K. and W.L.; resources, W.L.; data curation, K.F. and S.Z.; writing—original draft preparation, C.K.; writing—review and editing, C.K., W.L. and X.L.; project administration, W.L.; funding acquisition, W.L. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded by the National Natural Science Foundation of China (32070477), and the National Science & Technology Fundamental Resources Investigation Program of China (2018FY100400, 2019FY100400).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is contained within the article and supplementary material.
Acknowledgments
We express our sincere thanks to Xulong Chen for identification of the specimens of Homoneura.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Li, W.L.; Qi, L.; Yang, D. Four new species of the subfamily Homoneurinae (Diptera, Lauxaniidae) from southwestern China. ZooKeys 2020, 953, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Dvořáková, K. The results of a preliminary survey of the families Lauxaniidae and Heleomyzidae (Diptera, Acalyptrata) in the Upper and Lower Palatinate forest. Erica 2008, 15, 108–112. [Google Scholar]
- Shewell, G.E. Lauxaniidae. In Manual of Nearctic Diptera McAlpine; McAlpine, J.F., Peterson, B.V., Shewell, G.E., Teskey, H.J., Vockeroth, J.R., Wood, D.M., Eds.; Agriculture Canada: Ottawa, ON, Canada, 1987; Volume 2, pp. 951–964. [Google Scholar]
- Broadhead, E.C. The species of Poecilominettia, Homoeominettia and Floriminettia (Diptera: Lauxaniidae) in Panama. Bull. Br. Mus. (Nat. Hist.) Entomol. 1989, 58, 185–226. [Google Scholar]
- Hering, E. Biology of the Leaf Miners; Springer Netherlands: Berlin, Germany, 1951; pp. 1–420. [Google Scholar]
- Papp, L.; Shatalkin, A.I. Family Lauxaniidae. In Manual of Palaearctic Diptera; Papp, L., Darvas, B., Eds.; Science Herald: Budapest, Hungary, 1998; Volume 3, pp. 383–400. [Google Scholar]
- Silva, V.C.; Mello, R.L. Occurrence of Physoclypeus farinosus Hendel (Diptera: Lauxaniidae) in flowerheads of Asteraceae (Asterales). Neotrop. Entomol. 2008, 37, 92–96. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Miller, R.M.; Foote, B.A. Biology and immature stages of eight species of Lauxaniidae (Diptera) II. Descriptions of immature stages and discussion of larval feeding habits and morphology. P. Entomol. Soc. Wash. 1976, 78, 16–37. [Google Scholar]
- Moretti, T.C.; Ribeiro, O.B.; Thyssen, P.J.; Solis, D.R. Insects on decomposing carcasses of small rodents in a secondary forest in Southeastern Brazil. Eur. J. Entomol. 2008, 105, 691–696. [Google Scholar] [CrossRef]
- Shewell, G.E. Family Lauxaniidae. In A Catalogue of the Diptera of America North of Mexico; Stone, A., Sabrosky, C.W., Wirth, W.W., Foote, R.H., Coulson, J.R., Eds.; United States Department of Agriculture: Washington, DC, USA, 1965; pp. 696–704. [Google Scholar]
- Reddersen, J. Feeding biology of fungivorous insects from Danish cereal fields. Pedobiologia 1995, 39, 370–384. [Google Scholar]
- Stuckenberg, B.R. A review of the Old World genera of Lauxaniidae (Diptera). Ann. Natal Mus. 1971, 20, 499–610. [Google Scholar]
- Kim, S.P. Australian Lauxaniid Flies. Revision of the Australian Species of Homonera van der Wulp, Trypetisoma Malloch, and Allied Genera (Diptera, Lauxaniidae). In Monographs on Invertebrate Taxonomy; Morton, D.W., Nielsen, E.S., Reekie, P.W., Eds.; Commonwealth Scientific and Industrial Research Organisation: Canberra, Australia, 1994; Volume 1, pp. 1–445. [Google Scholar]
- Shi, L.; Zhang, M.J.; Shen, R.R.; Li, S.D.; Bai, J.; Wang, Y.J.; He, G.W.; Cui, Y. Preliminary study on phylogeny of species groups in subgenus Homoneura. J. Environ. Entomol. 2017, 39, 351–356. [Google Scholar]
- Cumming, J.M.; Wood, D.M. Adult morphology and terminology. In Manual of Afrotropical Diptera; Kirk-Spriggs, A.H., Sinclair, B.J., Eds.; South African National Biodiversity Institute: Pretoria, South Africa, 2017; Volume 1, pp. 89–133. [Google Scholar]
- Gaimari, S.D.; Silva, V.C. Lauxaniidae (Lauxaniid flies). In Manual of Afrotropical Diptera; Kirk-Spriggs, A.H., Sinclair, B.J., Eds.; South African National Biodiversity Institute: Pretoria, South Africa, 2021; Volume 3, pp. 1757–1781. [Google Scholar]
- Goloboff, P.A. NONA ver. 2; Goloboff, P.A.: Tucuman, Argentina, 1999. [Google Scholar]
- Goloboff, P.A.; Farris, J.S.; Nixon, K.C. TNT, a free program for phylogenetic analysis. Cladistics 2008, 24, 774–786. [Google Scholar] [CrossRef]
- Bremer, K. Branch support and tree stability. Cladistics 1994, 10, 295–304. [Google Scholar] [CrossRef]
- Nixon, K.C. WinClada; ver. 1.00.08; Nixon, K.C.: Ithaca, NY, USA, 2002. [Google Scholar]
- Sasakawa, M. Lauxaniidae (Diptera) of Malaysia (part 2): A revision of Homoneura van der Wulp. Insect. Mats. 1992, 46, 133–210. [Google Scholar]
- Shi, L.; Wang, J.C.; Yang, D. Nine new species from China of the subgenera Chaetohomoneura and Neohomoneura in the genus Homoneura (Diptera, Lauxaniidae). Zootaxa 2011, 2975, 1–28. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).