Simple Summary
The tobacco cutworm, Spodoptera litura (F.), exemplifies strong reproductive capacities and damages many agricultural crops. The insulin signaling pathway is known as a key determinant of female reproduction in insects. However, the detailed molecular mechanisms in these processes are poorly studied. Here, we injected bovine insulin into the newly emerged moth, resulting in gene expression changes in the insulin pathway, while knockdown of SlInR caused an inverse gene expression change involved in the insulin pathway. Further studies indicated that the content of JH-III, Vg, total proteins and triacylgycerol could be suppressed by SlInR dsRNA injection. Furthermore, stunted ovaries and lower fecundity were observed by RNAi. Our studies indicated that SlInR plays a key role in JH-III synthesis and the ovarian development in S. litura.
Abstract
Insulin signaling can regulate various physiological functions, such as energy metabolism and reproduction and so on, in many insects, including mosquito and locust. However, the molecular mechanism of this physiological process remains elusive. The tobacco cutworm, Spodoptera litura, is one of the most important pests of agricultural crops around the world. In this study, phosphoinositide 3-kinase (SlPI3K), protein kinase B (SlAKT), target of rapamycin (SlTOR), ribosomal protein S6 kinase (SlS6K) and transcription factor cAMP-response element binding protein (SlCREB) genes, except transcription factor forkhead box class O (SlFoxO), can be activated by bovine insulin injection. Then, we studied the influence of the insulin receptor gene (SlInR) on the reproduction of S. litura using RNA interference technology. qRT-PCR analysis revealed that SlInR was most abundant in the head. The SlPI3K, SlAKT, SlTOR, SlS6K and SlCREB genes were decreased, except SlFoxO, after the SlInR gene knockdown. Further studies revealed that the expression of vitellogenin mRNA and protein, Methoprene-tolerant gene (SlMet), could be down-regulated by the injection of dsRNA of SlInR significantly. Furthermore, a depletion in the insulin receptor by RNAi significantly decreased the content of juvenile hormone III (JH-III), total proteins and triacylgycerol. These changes indicated that a lack of SlInR could impair ovarian development and decrease fecundity in S. litura. Our studies contribute to a comprehensive insight into reproduction, regulated by insulin and the juvenile hormone signaling pathway through nutrition, and a provide theoretical basis for the reproduction process in pest insects.
1. Introduction
The reproduction of female insects has been studied extensively because of its importance for insect expansion and its potential as a target for pest control [1]. Vitellogenin (Vg), as the precursor of vitellin, acts as the primary source of nutrients for oocyte maturation and ovarian development [2]. Vg is primarily synthesized in insect body fat, then secreted into the haemolymph and utilized by oocytes through a complicated physiological process [3]. The reproductive success of insects depends on Vg biosynthesis and uptake by developing oocytes [4]. Therefore, Vg can be used as a molecular marker of fecundity [5].
The juvenile hormone (JH), a multifunctional insect hormone, is involved in retaining juvenile characteristics in the larval stages and stimulating gonad development in the adult stage in insects [6]. JH exerts its function through its intracellular receptor Methoprene (Met), a member of the family of the basic helix-loop-helix (bHLH)-Per-Arnt-Sim (PAS) transcription factors [7,8]. JH is synthesized by the corpora allata (CA), which are known as another important factor to regulate a large array of physiological processes, such as embryonic development [9], metamorphosis [10], energy homeostasis [11], reproduction [12] behaviors [13,14], etc. JH binds to intracellular receptor Methoprene (Met), a member of the family of the basic helix-loop-helix (bHLH)-Per-Arnt-Sim (PAS) transcription factors, and then the JH signaling pathway is activated [4]. In the adult stage, nutrition is used to stimulate the production of the major gonadotropin JH [15]. As the biosynthesis of Vg is sensitive to the titer of JH [16], many documents have used Vg as a bio-marker to demonstrate the effect on reproduction by JH in insects [17,18,19].
In insects, the insulin signal responses to energy metabolism are well reported. Insulin-like peptides (ILPs) bind to one or more insulin receptors (InRs) [20]. Then, the phosphoinositide 3-kinase (PI3K)-serine/threonine kinase (Akt) signaling pathway or the GTPase Ras/mitogen is activated, in turn [21,22,23,24,25,26]. In these processes, a series of downstream proteins, including the transcription factor (forkhead box class O, FoxO), the target of rapamycin/ribosomal protein S6 kinase (TOR/S6K), transcription factor cAMP-response element binding protein (CREB), etc., are activated by phosphorylation [27,28,29]. The insulin pathway being involved in fecundity of insects is also documented. When female insects reach an adequate nutritional state, the ILPs are secreted into the hemolymph and bound to the InRs. The insulin pathway is activated [30]. Then, the vitellogenin synthesis begins through the control of some hormonal signaling, such as neuropeptides, juvenile hormones (JH) and ecdysteroids [1]. However, most of these studies focus on the upstream regulatory hierarchy. Therefore, it is of great significance to further elucidate the detailed molecular mechanism, especially to identify which genes and hormones participate in cooperation.
2. Materials and Methods
2.1. Insect Rearing
The S. liturae were reared on an artificial diet at 25 ± 1 °C in a 14 L:10 D photoperiod and 70–80% relative humidity (RH) in an incubator (P400G, Ruihua, China) [31]. The newly emerged adults were collected every 12 h and transferred to a one-end open cylindrical plastic cage (Φ = 12 cm, H = 25 cm) covered with filter paper in inner wall for mating and oviposition, and a cotton ball with 10% honey was added as a diet supplement.
2.2. Bovine Insulin Microinjection
Following this, 25 mg Bovine insulin (Solarbio, Beijing) was solubilized in 50 μL of 1 M hydrochloric acid and diluted to a final concentration of 2 mg/mL with 0.01 M PBS buffer (pH = 8.0). Each new female adult was injected with 7 μL insulin at the fourth and fifth abdominal junctions after being anesthetized on ice for 1 min using a 10 μL micro-syringe syringe (Hamilton) and the injection point was sealed immediately with white Vaseline (Aladdin, China) [32]. Further, 24 h after injection, three individuals were selected at random from each treated group. Further, the abdomens were collected and pooled for RNA isolation. Females injected with PBS buffer were used as control.
2.3. RNA Isolation and Quantitative Real-Time-Polymerase Chain Reaction (qRT-PCR)
Total RNA was isolated from each replicate sample using the Trizol reagent guided by the manufacturer’s suggestion (Invitrogen, Carlsbad, CA, USA). Up to 1 µg of the total was reverse-transcribed into cDNA using M-MLV Reverse Transcriptase (PrimeScript™ II 1st Strand cDNA Synthesis Kit, TaKaRa, Japan). qRT-PCR was performed with a Bio-Rad real-time thermal cycler using the SYBR ExTaq kit (Tli RNaseH Plus) (Takara, Japan) according to the manufacturer’s instructions. Briefly, in a 10 µL total reaction volume containing 1 µL cDNA above, 5 µL TB Green Premix Ex Taq II, 0.4 µL forward and reverse primers (10 µM), respectively, dd water was added to 10 µL. PCR program was as follows: 94 °C for 3 min followed by 40 cycles at 94 °C for 15 s, 58 °C for 30 s and 72 °C for 20 s. The melting curve was analyzed to confirm the amplification of a single fragment. The relative gene expression was analyzed by the 2−ΔΔCt method [33] using (Ribosomal protein L10, RPL10, GenBank Accession No. KC866373) as an internal control. The specific primers for qRT-PCR are shown in Table 1. Quantification of the relative changes in gene transcript level was performed according to the 2−ΔΔCt method. Three biological replicates were performed for all qRT-PCR experiments to obtain the means and standard errors.

Table 1.
The primer information for qRT-PCR and dsRNA production.
2.4. Sample Collection for Different Tissue Expression Analysis
To investigate the expression pattern of different tissues, head, thorax, ovaries and fat body from 5 to 8 individuals were carefully dissected and collected. After the tissues were washed in PBS buffer (0.01 M, pH = 8.0) several times, total RNA was isolated from all these samples. Then cDNA was synthesized and gene transcript levels were performed with qRT-PCR as above.
2.5. RNA Interference
RNA interference was used to investigate the function of SlInR (insulin receptor, GenBank Accession No. XM_022966387.1) with the EGFP gene (enhanced green fluorescent protein, GenBank Accession No. DQ768212) as a parallel control. For dsRNA preparation, SlInR and EGFP genes were first amplified using specific primers conjugated with the T7 RNA polymerase promoter sequence (Table 1). The resultant PCR products were used as templates to synthesize dsRNA (the lengths of dsSlInR and dsEGFP were 458 (3281–3738 bp) and 547 bp (126–672 bp), respectively) according to the manufacturer recommendations of T7 RiboMAX™ Express RNAi System (Promega, Madison, WI, USA). The quality and integrity of dsRNA were analyzed by agarose gel electrophoresis; meanwhile, the quantity was determined by a Nanodrop1000 Ultramicro spectrophotometer (Implen, München, Germany).
One-day-old virgin adult females were used for RNAi experiment. Before injection, the moths were anesthetized on ice for 5 min. As such, 7 µg dsRNA per individual was injected into the abdomen between the 4th and 5th abdominal segment using a 10 μL micro-syringe (Hamilton) and the injection point was sealed with wax immediately. The injected female was then put into a one-end-open cylinder cage with white paper around the inner wall and two normal males were put together for mating. The cage was 30 cm in height and 15 cm in diameter, the open top was enclosed with a piece of nylon mesh to prevent the insects from moving in or out. Further, 10% honey cotton wool balls were added as nutritional supplement. The gene expression changes were calculated using qRT-PCR at 12 h, 24 h, 36 h and 48 h after dsRNA injection as described above.
For fecundity analysis, the paper in the cage was taken out every day and the number of eggs on the paper was counted for a continuous 6 days.
2.6. Determination of Protein and Triglyceride (TAG)
The protein content was determined using a BCA protein concentration assay kit (Beyotime, Shanghai, China). The full abdomen of females was dissected and collected in a 1.5 mL Eppendorf tube. After being weighed, RIPA lysate (250 μL per 20 mg sample) and protease inhibitor (phenylmethylsulfonyl fluoride, PMSF) (final concentration 1 mM) were added. All the mixtures were homogenized on ice for 5 min. Then, 200 μL supernatant was transferred to a 96-well plate and 200 µL BCA developer was added, respectively. After incubating at 37 °C for 30 min, the optical density (OD) values were detected on a HIDEX Sense 425–301 (Turku, Finland) at 562 nm. The standard curve was made according to kit requirements under the same conditions.
TAG assay kit (Solarbio Biotech, Beijing, China) was used for TAG content quantification; the method was described before [34]. In brief, 1 mL n-heptane and isopropanol (1:1) was added to 100 mg dissected ovaries and homogenized for 5 min in an ice bath. After being centrifuged at 8000× g for 10 min at 4 °C, 120 μL supernatant was transferred to a new 1.5 mL Eppendorf tube and 375 μL reagent I along with 75 μL reagent II were added in sequence, shocked 30 s on a vortex and stood 10 min at room temperature, 30 μL supernatant was absorbed and transferred to a clean 1.5 mL Eppendorf tube. Then 100 μL reagent III, 30 μL reagent IV, 100 μL reagent V and 100 μL reagent VI were added in sequence. Further, 200 μL mixture was then transferred to a 96-well plate after being incubated for 15 min at 65 °C in an air oven and the OD value was detected on an HIDEX Sense 425-301 (Turku, Finland) at 420 nm. The DD water was used for control to adjust zero.
2.7. JH- III Determination
JH- III was purchased from Sigma (St. Louis, MI, USA). All of the other reagents were HPLC grade. The JH- III detection was performed by high-performance liquid chromatography (HPLC) method [35]. Briefly, full females without wings and legs were put in a mortar. After adding 4 mL ether: methanol (V:V = 1:1), homogenized in liquid nitrogen and eluting for 5 min under ultrasonic wave in ice bath, the sample was centrifuged at 8000× g for 10 min at 4 °C. The supernatant was transferred to a 15 mL centrifuge tube with 2 mL n-hexane added. After homogenizing in a vortex, the supernatant liquid was extracted and transferred to another new 15 mL centrifuge tube. The extraction was repeated five times. All the supernatant liquids were merged together and the n-hexane was volatilized by vacuum distillation. The residue was re-dissolved using methanol and the final volume was 2 mL. Further, 10 μL sample was separated on a 150 × 2 mm2 C18 reverse-phased column (YMC-Pack Pro C18.5 mm, YMC Co., Ltd., Kyoto, Japan) protected by a guard column (YMC-Pack Pro, sphere ODS- H80, YMC Co., Ltd., Kyoto, Japan) with elution of methanol: ddH2O (V:V = 70:30) at a flow rate of 0.8 mL/min, using an Agilent 1100 HPLC system with autosampler. The standard curve was made according to gradient concentration of the standard JH-III.
2.8. Western Blotting
To analyze the influence of SlInR gene silencing on the expression of Vg protein, a DAB kit was used for Western blot, analyzing its expression. The method was modified according to the protocol previously described [36]. Briefly, about 20 mg female fat body was homogenized in 250 µL RIPA lysate. Proteins were separated on a 12% SDS-PAGE gel; the gel was semi-dry transferred for 40 min at 10 volts to an Immobilon-P PVDF membrane (Millipore, Bedford, MA, USA). The immunoreactivity was tested with the anti-SlVg serum (diluted 1:2000) and an IgG goat anti-rabbit antibody conjugated with HRP was used for a secondary antibody (BOSTER, Wuhan, China, 1:5000 dilution) and finally visualized by 3,3- diaminobenzidine carbon tetrachloride (DAB). Non-specific binding was blocked using a 5% fat-free milk solution.
2.9. Ovary Dissection and Microscopy
To determine the impact of gene silencing on the development of ovaries in the S. litura, the ovaries from the 2nd-day females were separated in 1 × PBS (pH = 8.4) followed by fixation in 3.8% formaldehyde in 1 × PBS for 20 min at room temperature. Dissected ovaries were washed three times for 10 min with 0.2% Triton-X 100 (Sigma, USA) in 1× PBS. After washing, ovaries were photographed with a microscope (Nikon, Sendai, Japan).
2.10. Statistical Analysis
The significant differences between the means of multiple samples were analyzed by using one-way analysis of variance (ANOVA), which was followed by the Tukey’s test to perform the multiple comparisons between the means of every two samples. The significant differences between only two samples were determined by using Student’s t-test (* p < 0.05; ** p < 0.01). Statistical analyses were performed using SPSS 20.0 (Chicago, IL, USA) and data are presented as the means ± standard error (SEM).
3. Results
3.1. Tissue-Specific Expression of SlInR
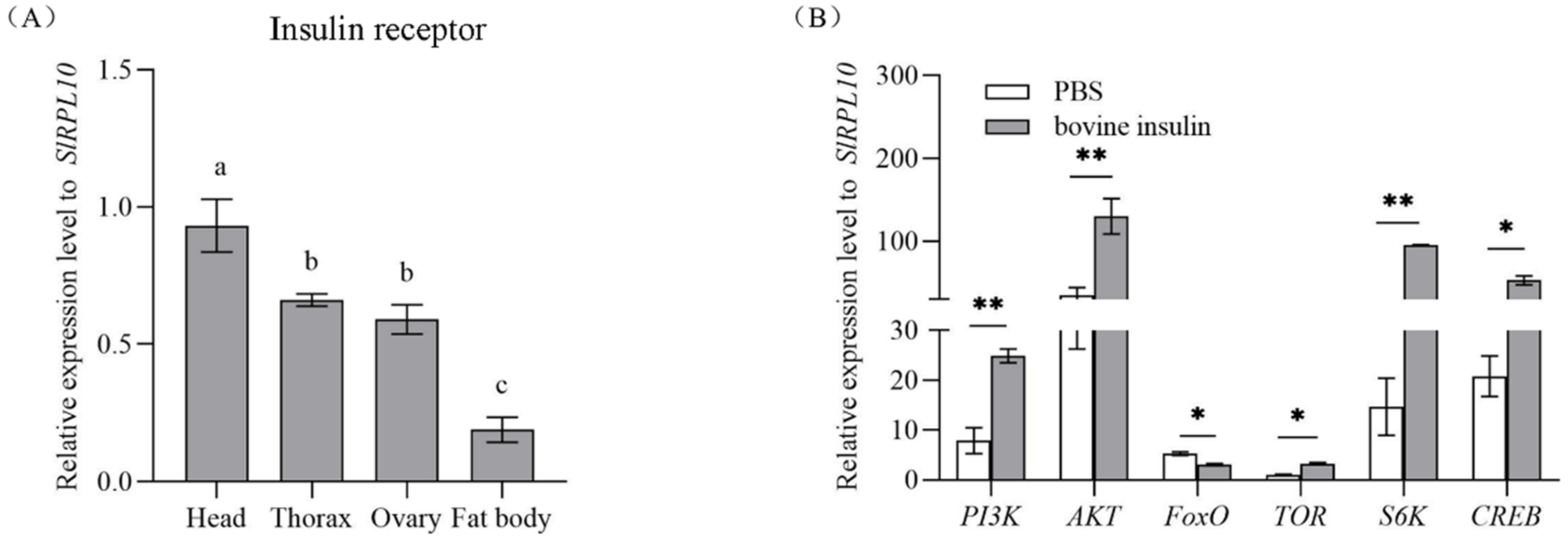
qRT-PCR was used to characterize the expression pattern of SlInR in various tissues in the one-day-old emerging S. litura female adults. The results demonstrated that SlInR mRNA was mainly expressed in head, thorax and ovaries but weakly in fat body (Figure 1A).
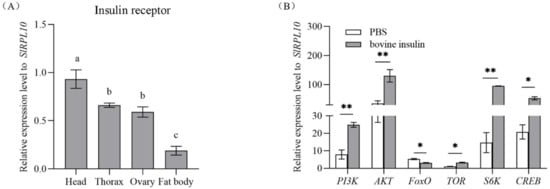
Figure 1.
Expression profile of SlInR in different tissues and effect of bovine insulin injection on gene expression of insulin signaling pathway. (A) qPCR analysis of SlInR expression levels in different tissues from one-day-old adult females. (B) The expression of insulin signaling pathway genes was detected 24 h after bovine insulin injection by qRT-PCR. Results are represented as means ± SD of three independent samples and samples are normalized to SlRPL10 expression levels. Different lowercase letters represent significant differences of SlInR levels among various tissues determined by one-way ANOVA. Statistically significant difference between the PBS and bovine insulin injection groups (* p < 0.05; ** p < 0.01, Student’s t-test).
3.2. Changes in Insulin Pathway Gene Expression after Bovine Insulin Application
Once insects emerged, female adults were injected with 14 μg bovine insulin or PBS buffer. Gene expression changes in the insulin pathway were quantified by qRT-PCR 24 h later. It showed that the expression levels of SlPI3K (GenBank accession No: XM_022968595.1), SlAKT (GenBank accession No: XM_022971351.1), SlTOR (GenBank accession No: XM_022966802.1), SlS6K (GenBank accession No: XM_022972696.1) and SlCREB (GenBank accession No: XM_022973491.1) genes were promoted but the SlFoxO (GenBank accession No: XM_022969964.1) gene was suppressed significantly (Figure 1B).
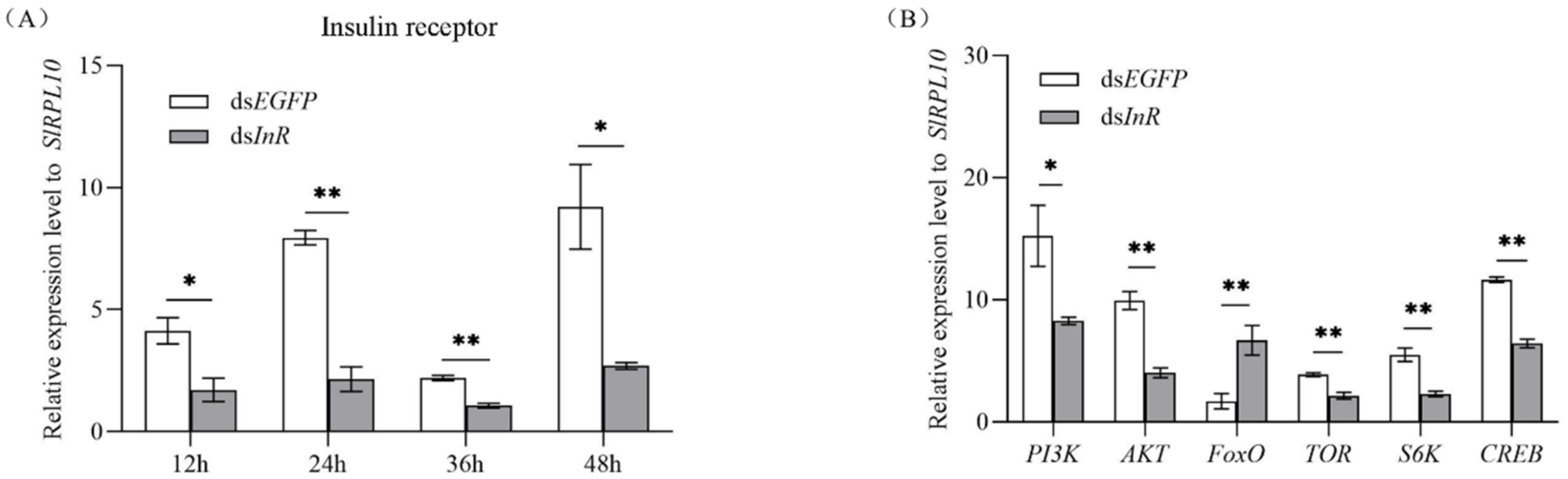
3.3. Effect of SlInR Silence on Insulin Pathway Gene expression
Newly emerged females were injected with dsSlInR or dsEGFP. The efficiency of SlInR gene knockdown was quantified by qRT-PCR at 12 h, 24 h, 36 h and 48 h. For gene expression changes in the insulin pathway, three individuals were sampled as a pool randomly after 24 h injection and gene expression levels were quantified by qRT-PCR also. The results indicated that the SlInR transcripts were decreased 56.4–79.6% after dsSlInR injection compared to control (Figure 2A) and the expression levels of PI3K, AKT, TOR, S6K and CREB genes were significantly decreased, while the FoxO gene was increased 24 h later (Figure 2B).
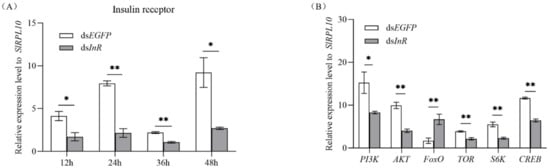
Figure 2.
Efficiency of SlInR gene knockdown and effect of SlInR knockdown on gene expression of insulin signaling pathway. (A) Efficiency of SlInR gene knockdown at 12, 24, 36 and 48 h. (B) The expression of insulin signaling pathway genes was detected 24 h after dsSlInR injection by qRT-PCR. Results are represented as means ± SD of three independent samples and samples are normalized to SlRPL10 expression levels (* p < 0.05; ** p < 0.01, Student’s t-test).
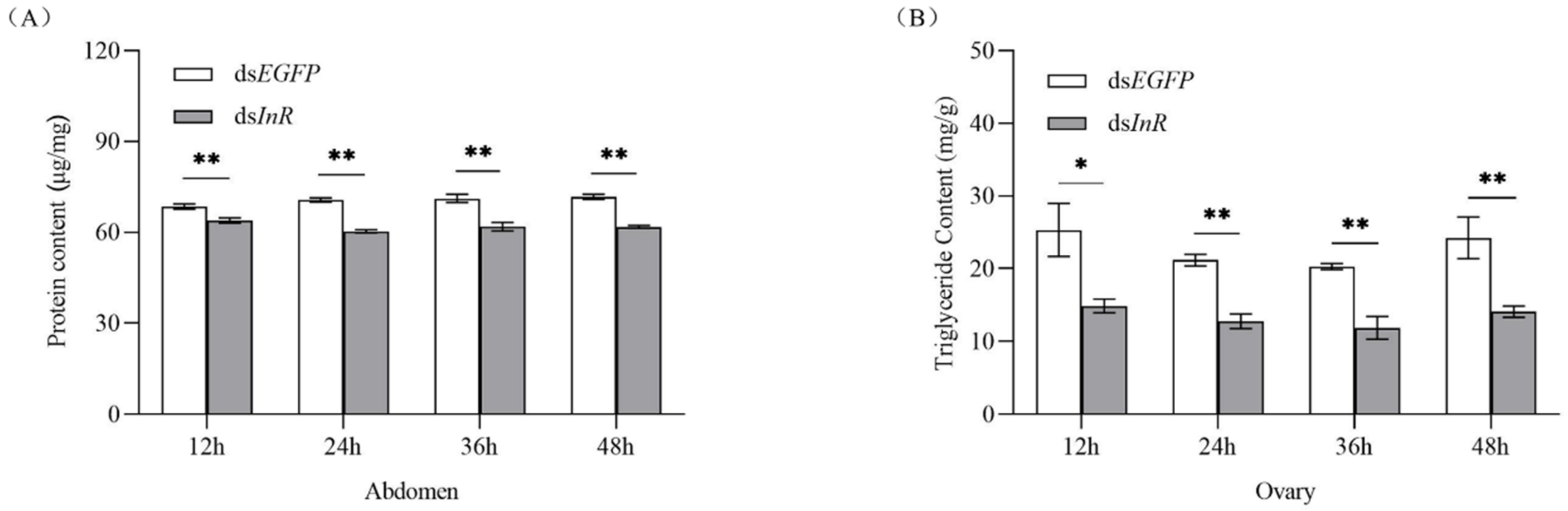
3.4. Effect of SlInR Knockdown on Protein and Triglyceride (TAG) Metabolism
To study whether interference with SlInR affects nutrients related to insect reproduction, protein content in female full abdomen and triglyceride titer in ovaries were simultaneously analyzed. In RNAi SlInR females, protein and TAG levels were reduced significantly at 12 h, 24 h, 36 h and 48 h (Figure 3A,B). Therefore, the nutritional status in S. litura might be regulated by SlInR expression at the mRNA level and then modulate reproduction in response to protein and TAG.
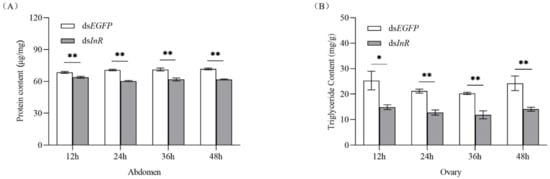
Figure 3.
Effect of SlInR knockdown on the contents of protein and triglyceride. (A) Protein contents in the abdomen after SlInR knockdown (n = 25). (B) Triglyceride contents in ovaries after SlInR knockdown (n = 25). Statistically significant difference between the dsEGFP and dsInR injection groups (* p < 0.05; ** p < 0.01, Student’s t-test).
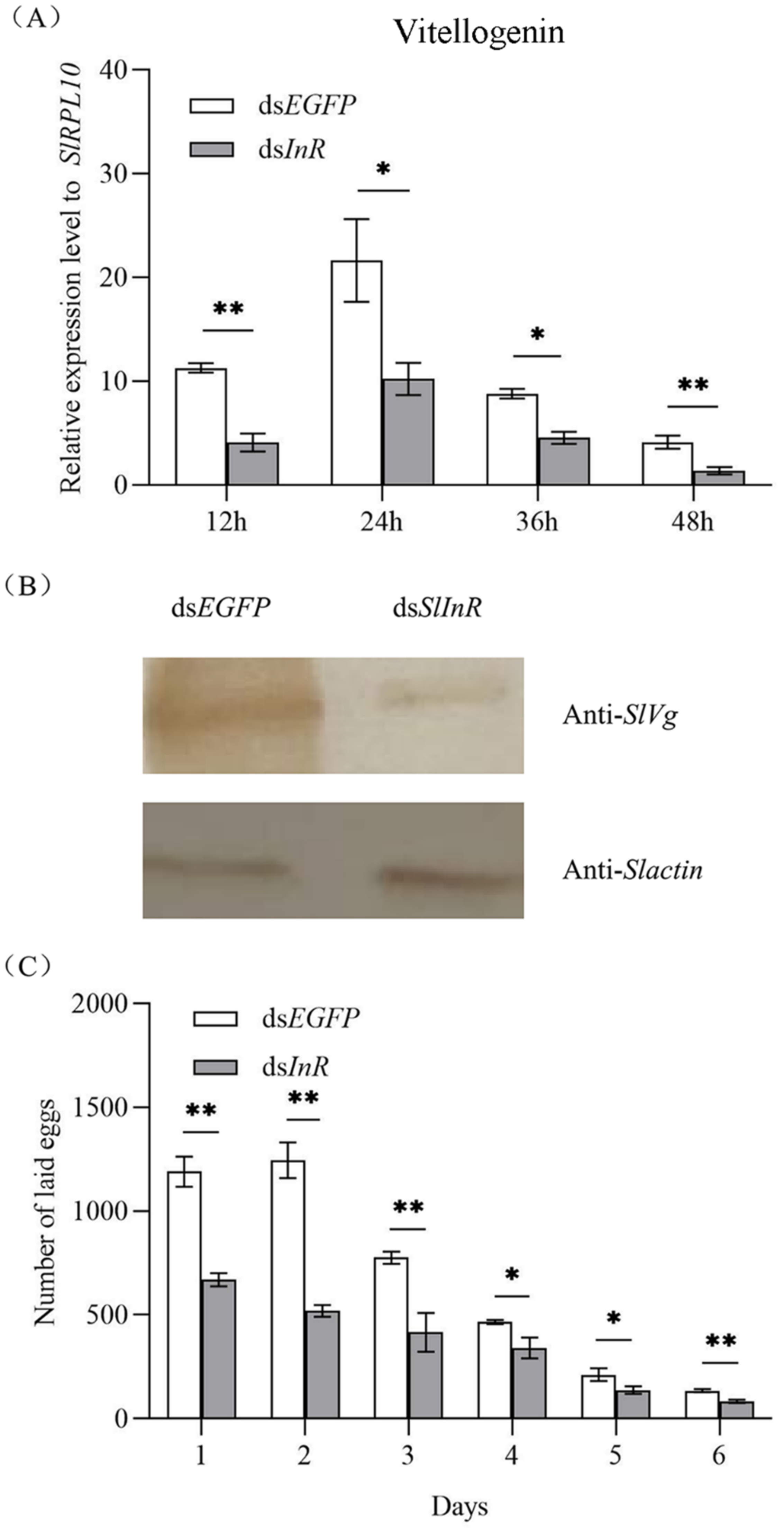
3.5. Effect of SlInR Silence on SlVg Expression and Reproduction
To detect SlVg gene expression in the fat body of S. litura after SlInR silence, the SlVg transcripts were detected at 12 h, 24 h, 36 h and 48 h using the qRT-PCR method. It showed that the SlVg expression level was decreased 51.7–78.3% after dsSlInR RNA injection vs. dsEGFP injection (Figure 4A). For SlVg protein expression change analysis, three individuals were sampled as a pool randomly after 24 h injection and the protein expression was assayed by Western blotting. The results showed a decrease in Vg protein expression too (Figure 4B). The oviposition of the interfering SlInR group was significantly decreased at 6 days compared to the control group (Figure 4C). These results suggest that interference with SlInR may affect the eggs laid by impeding oocyte maturation in S. litura.
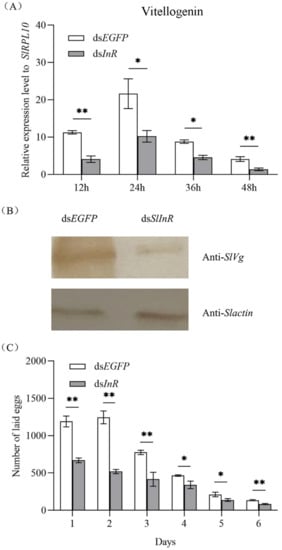
Figure 4.
Effect of SlInR silence on SlVg expression and reproduction. (A) qRT-PCR analysis of Vg transcript abundance in abdomen from females treated with dsSlInR for 12, 24, 36 and 48 h. (B) Western blotting analysis of SlVg contents in fat body. Immunoblot of fat body collected 48 h from females injected with dsSInR and dsEGFP and detection with Vg antibody, an antibody against actin was used as a loading control. (C) Daily fecundity. The newly emerged female adults were treated with dsRNA and the fecundity of single female was recorded for six consecutive days after mating (n = 25). Statistically significant difference between the dsEGFP and dsInR injection groups (* p < 0.05; ** p < 0.01, Student’s t-test).
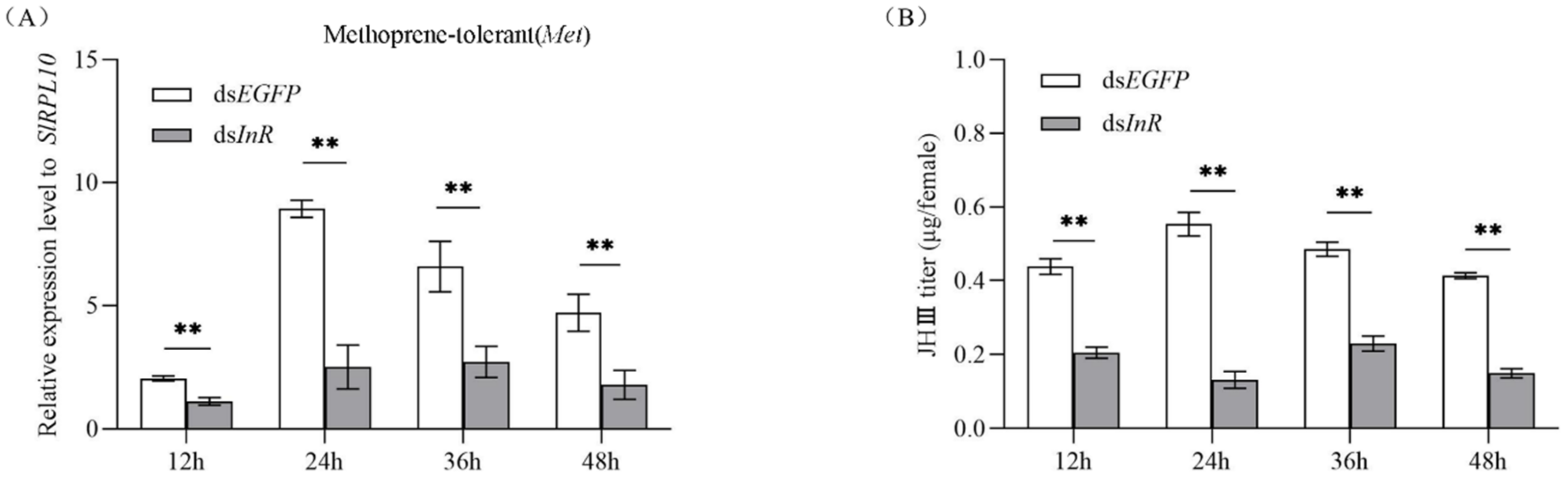
3.6. Effect of SlInR Interference on SlMet Expression and JH-III titer
To study whether interference with SlInR affects the juvenile hormone (JH) pathway, methoprene-tolerant (Met) (GenBank accession No: 2595041) transcripts were detected at 12 h, 24 h, 36 h and 48 h after SlInR RNAi. qRT-PCR analysis showed that silencing of SlInR greatly decreased the SlMet expression in the female full abdomen of dsInR-treated adults compared to the controls (Figure 5A). HPLC detection indicated that JH-III titer in the body was reduced dramatically in the dsSlInR RNA injection group (Figure 5B). These data indicated that the insulin pathway can affect insect JH cascade.
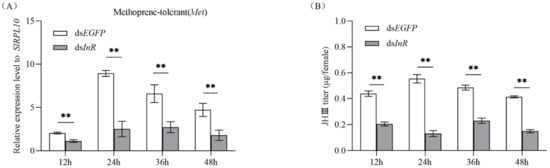
Figure 5.
Effect of SlInR interference on SlMet expression and JH-III titer. (A) qRT-PCR analysis of SlMet expression levels after SlInR knockdown. (B) JH-III titer was evaluated after SlInR gene knockdown by HPLC. Results are represented as means ± SD of three independent replicates. Statistically significant difference between the dsEGFP and dsInR injection groups (** p < 0.01, Student’s t-test).
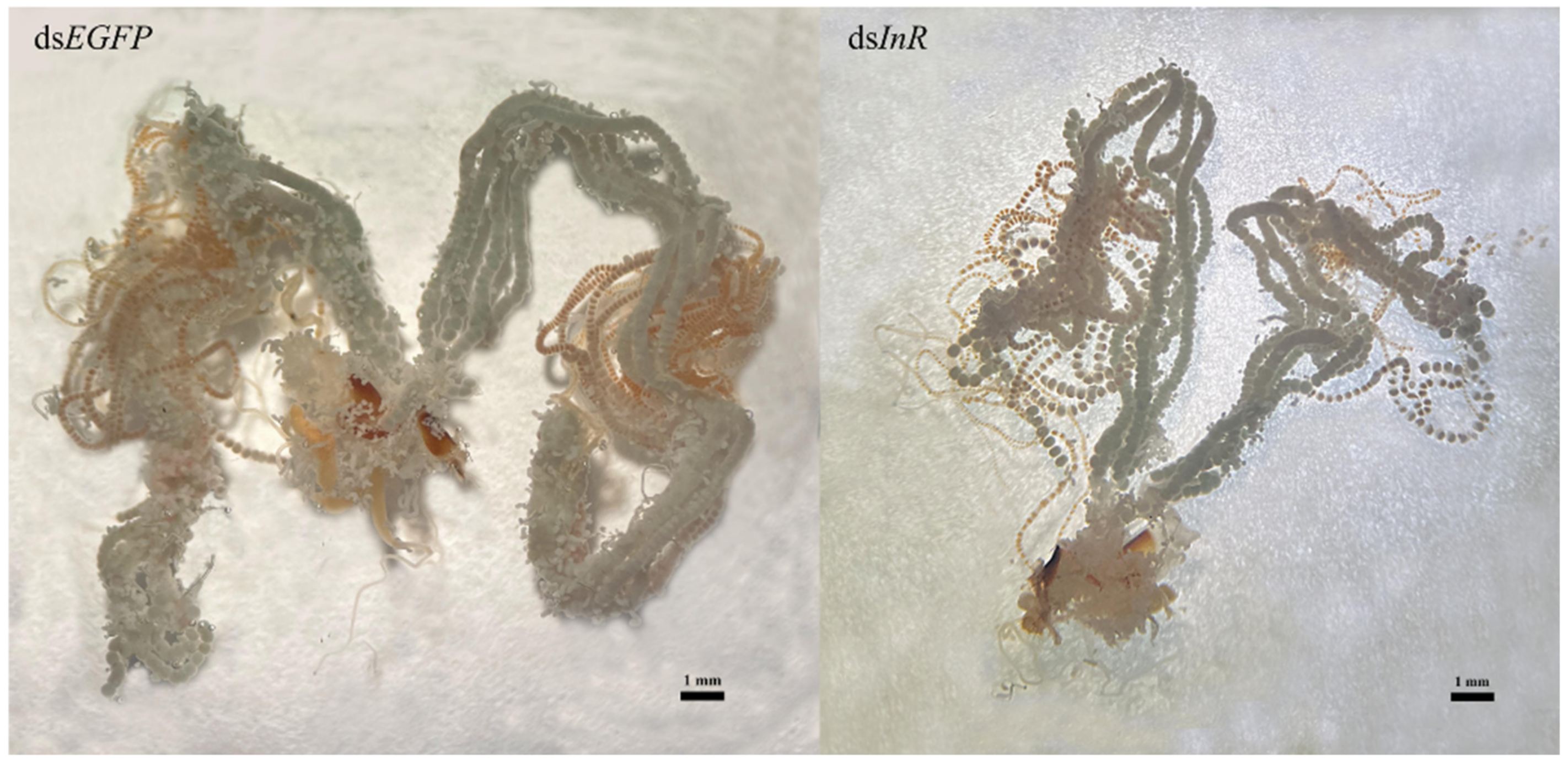
3.7. Effect of SlInR Knockdown on Ovarian Development
Newly emerged insects were injected with dsSlInR or dsEGFP RNA. Further, 10% honey was added as a nutritional supplement and 48 h later, the ovaries were dissected and photographed. The results suggested that RNAi knockdown of dsSlInR resulted in underdeveloped ovaries with abnormal ovarioles in the ovaries (Figure 6).
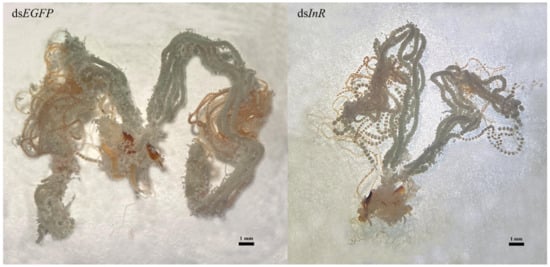
Figure 6.
Effect of SlInR interference on ovarian development. dsSlInR and dsEGFP were injected after eclosion and the ovaries of female adults were dissected 48 h later. The scale bar represents 1 mm.
4. Discussion
Insulin binding to the insulin receptor (InR) is usually associated with the regulation of metabolic responses [37]. Previous studies have shown that the InR predominantly stimulated the PI3K/AKT pathway and the Ras/MAPK pathway [38,39]. Our results indicated that bovine insulin could activate the insulin pathway genes (SlPI3K, SlAKT, SlTOR, SlS6K, SlCREB) (Figure 1B). Bovine insulin is widely used to investigate the physiological functions of ILPs and the related signaling pathway in many insects because the structure and function of ILPs are conservative in different species [21,40,41]. Insect ILPs participate in more physiological processes and act in a different manner compared to mammalian insulins [42]. Each of the ILPs in A. aegypti shared some essential residues in the bovine insulin A and B chain [23]. However, the ILPs in S. litura have not been reported in the NCBI genome database yet. Therefore, we do not know which ILPs exactly perform this process.
Insulin is known to be a key determinant of female reproduction in insects for regulating ovarian development and oogenesis [23,43,44,45]. In this study, the SlInR was effectively knocked down by dsRNA injection (Figure 2A). Contrary to the bovine insulin injection, the expression of critical genes (SlPI3K, SlAKT, SlTOR, SlS6K, SlCREB) in the insulin signal pathway were decreased after SlInR was knocked down (Figure 2B). The expression level of the Vg gene and protein was reduced and, synchronously, fecundity declined after RNAi-mediated knockdown of SlInR. Previous studies revealed that knockdown of InR(s) could affect ovarian development in Culex quinquefasciatus [46], Chrysopa pallens [25], Bactrocera dorsalis [44] and so on. Reports also indicated that AKT, TOR, PI3K, S6K [47,48], FoxO [49] and CREB [25] genes are involved in reproduction regulation, respectively, in many insects. Similarly, our results also indicated the important role of the insulin signal pathway in the ovarian development and fecundity of S. litura.
Insulin-like peptides (ILPs) bind to one or more InR(s). Then, the phosphoinositide 3-kinase (PI3K)-serine/threonine kinase (Akt) signaling pathway is activated, in turn [21,22]. Further, the downstream genes (TOR, FoxO, CREB, S6K) will be activated or have a suppressed response to this process [28,50,51,52]. In this study, our results indicated that the expression levels of genes (SlPI3K, SlAkT, SlTOR, SlS6K and SlCREB) were increased but the SlFoxO gene expression was reduced when the insulin pathway was activated by bovine insulin, while the transcriptions were exactly inverse when the SlInR gene was knocked down (Figure 1B and Figure 2B). Similar results were observed in many other insects, such as Nilaparvata lugens [51] and Blattella germanica [53]. However, a recent report opposed this, as the expression of FoxO was reduced when the InR gene was knocked down in B. dorsalis [44]. Evidence indicated that the FoxO gene is a negative regulatory factor in the insulin pathways [40,51,53]. However, it could be activated in different cascades in the insulin pathway [54]. We found the SlInR gene was expressed in head, thorax, fat body and female ovaries (Figure 1A). It seems that SlInR’s function in different tissues might impact reproduction in a wide variety of ways.
The reproductive success of insects depends on vitellogenin (Vg) biosynthesis and uptake of Vg into developing oocytes mediated by the vitellogenin receptor [4]. Vg synthesis can be induced by JH in many insects [17,19,55]. In this study, the silence of the SlInR gene led to a decrease in the SlMet gene expression level and decline in JH-III titer (Figure 5A,B). Furthermore, ovarian development was blocked with a reduction in Vg gene transcription and protein translation (Figure 4A,B and Figure 6). These results were presented in Tribolium castaneum [40], Anopheles albimanus [19] and Cimex lectularius [55]. Therefore, how the insulin pathway regulates ovarian development through JH-mediated control of the Vg expression in S litura deserves further investigation.
JH has been shown to be involved in the nutrient-mediated regulation of reproduction in many insect species [41,56,57]. The regulation of JH titer by nutrient signals working through the insulin pathway was observed in Drosophila melanogaster, Aedes aegypti [57], Blattella germanica [58] and Nilaparvata lugens [59]. Most insect eggs contain large supplies of nutrients to support embryogenesis and oogenesis is commonly a nutrient-limited process [60]. The proteins accumulate in insects developing oocytes [61] and amounts of lipophorins are found in ovaries at different stages of the reproduction cycle in B. germanica [62] and Panstrongylus megistus [63]. Lipid is also another essential component for the journey to insect reproduction [64,65]. The first nutrition of herbivorous insects is provided by their mothers through the resources they deposit into eggs [66]. Herein, protein concentration in full abdomen and triacylglycerol content in ovaries were significantly decreased after dsSlInR RNA injection (Figure 3A,B). Therefore, nutrition is a main factor for limiting reproduction. However, how this reproduction is regulated by insulin pathway crosstalk with the JH signal pathway and nutrition needs to be further elucidated.
Author Contributions
Conceptualization, X.D. and X.C.; methodology, X.P., Y.P. and C.Z.; software, Y.H. and C.L.; validation, X.P., Y.P., and L.W.; formal analysis, X.P., C.L.; investigation, C.L.; resources, X.P., Y.P., L.W.; data curation, L.C.; writing-original draft preparation, X.P. and L.C.; writing-review and editing, X.D. and X.C.; visualization, L.W.; supervision, X.D. and X.C.; project administration, X.D. and X.C.; funding acquisition, X.D. and X.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (31772168) and Fujian Provincial Key Laboratory of Ecology-Toxicological Effects & Control for Emerging Contaminants (PY21001).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data is provided in the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Roy, S.; Saha, T.T.; Zou, Z.; Raikhel, A.S. Regulatory Pathways Controlling Female Insect Reproduction. Annu. Rev. Entomol. 2018, 63, 489–511. [Google Scholar] [CrossRef] [PubMed]
- Shang, F.; Niu, J.Z.; Ding, B.Y.; Zhang, Q.; Ye, C.; Zhang, W.; Smagghe, G.; Wang, J.J. Vitellogenin and its receptor play essential roles in the development and reproduction of the brown citrus aphid, Aphis (Toxoptera) citricidus. Insect Mol. Biol. 2018, 27, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Hagedorn, H.H.; Kunkel, J.G. Vitellogenin and Vitellin in Insects. Annu. Rev. Entomol. 1979, 24, 475–505. [Google Scholar] [CrossRef]
- Miao, L.; Zhang, N.; Jiang, H.; Dong, F.; Yang, X.; Xu, X.; Qian, K.; Meng, X.; Wang, J. Involvement of Two Paralogous Methoprene-Tolerant Genes in the Regulation of Vitellogenin and Vitellogenin Receptor Expression in the Rice Stem Borer, Chilo suppressalis. Front. Genet. 2020, 11, 609. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, D.; Zhang, M.; Chen, W.; Zhang, G. Food source affects the expression of vitellogenin and fecundity of a biological control agent, Neoseiulus cucumeris. Exp. Appl. Acarol. 2014, 63, 333–347. [Google Scholar] [CrossRef]
- Gilbert, L.I.; Granger, N.A.; Roe, R.M. The juvenile hormones: Historical facts and speculations on future research directions. Insect Biochem. Mol. Biol. 2000, 30, 617–644. [Google Scholar] [CrossRef]
- Jindra, M.; Bellés, X.; Shinoda, T. Molecular basis of juvenile hormone signaling. Curr. Opin. Insect Sci. 2015, 11, 39–46. [Google Scholar] [CrossRef]
- Li, M.; Mead, E.A.; Zhu, J. Heterodimer of two bHLH-PAS proteins mediates juvenile hormone-induced gene expression. Proc. Natl. Acad. Sci. USA 2011, 108, 638–643. [Google Scholar] [CrossRef]
- Naruse, S.; Washidu, Y.; Miura, K.; Shinoda, T.; Minakuchi, C. Methoprene-tolerant is essential for embryonic development of the red flour beetle Tribolium castaneum. J. Insect Physiol. 2020, 121, 104017. [Google Scholar] [CrossRef]
- Hiruma, K.; Kaneko, Y. Hormonal regulation of insect metamorphosis with special reference to juvenile hormone biosynthesis. Curr. Top. Dev. Biol. 2013, 103, 73–100. [Google Scholar]
- Xu, J.; Sheng, Z.; Palli, S.R. Juvenile hormone and insulin regulate trehalose homeostasis in the red flour beetle, Tribolium castaneum. PLoS Genet. 2013, 9, e1003535. [Google Scholar] [CrossRef] [PubMed]
- Riddiford, L.M. How does juvenile hormone control insect metamorphosis and reproduction? Gen. Comp. Endocrinol. 2012, 179, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Leinwand, S.G.; Scott, K. Juvenile hormone drives the maturation of spontaneous mushroom body neural activity and learned behavior. Neuron 2021, 109, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Amsalem, E.; Malka, O.; Grozinger, C.; Hefetz, A. Exploring the role of juvenile hormone and vitellogenin in reproduction and social behavior in bumble bees. BMC Evol. Biol. 2014, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Fronstin, R.B.; Hatle, J.D. A cumulative feeding threshold required for vitellogenesis can be obviated with juvenile hormone treatment in lubber grasshoppers. J. Exp. Biol. 2008, 211 Pt 1, 79–85. [Google Scholar] [CrossRef][Green Version]
- Hatle, J.D.; Juliano, S.A.; Borst, D.W. Hemolymph ecdysteroids do not affect vitellogenesis in the lubber grasshopper. Arch. Insect Biochem. Physiol. 2003, 52, 45–57. [Google Scholar] [CrossRef]
- Parthasarathy, R.; Sun, Z.; Bai, H.; Palli, S.R. Juvenile hormone regulation of vitellogenin synthesis in the red flour beetle, Tribolium castaneum. Insect Biochem. Mol. Biol. 2010, 40, 405–414. [Google Scholar] [CrossRef]
- Gujar, H.; Palli, S.R. Juvenile hormone regulation of female reproduction in the common bed bug, Cimex lectularius. Sci. Rep. 2016, 6, 35546. [Google Scholar] [CrossRef]
- Hernández-Martínez, S.; Cardoso-Jaime, V.; Nouzova, M.; Michalkova, V.; Ramirez, C.E.; Fernandez-Lima, F.; Noriega, F.G. Juvenile hormone controls ovarian development in female Anopheles albimanus mosquitoes. Sci. Rep. 2019, 9, 2127. [Google Scholar] [CrossRef]
- Wu, Q.; Brown, M.R. Signaling and function of insulin-like peptides in insects. Annu. Rev. Entomol. 2006, 51, 1–24. [Google Scholar] [CrossRef]
- Zhang, T.T.; Zhang, G.C.; Zeng, F.F.; Liu, C.Y.; Mao, J.J. Insulin-like peptides regulate vitellogenesis and oviposition in the green lacewing, Chrysopa septempunctata. Bull. Entomol. Res. 2017, 107, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Colombani, J.; Andersen, D.S.; Boulan, L.; Boone, E.; Romero, N.; Virolle, V.; Texada, M.; Léopold, P. Drosophila Lgr3 Couples Organ Growth with Maturation and Ensures Developmental Stability. Curr. Biol. 2015, 25, 2723–2729. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.R.; Clark, K.D.; Gulia, M.; Zhao, Z.; Garczynski, S.F.; Crim, J.W.; Suderman, R.J.; Strand, M.R. An insulin-like peptide regulates egg maturation and metabolism in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2008, 105, 5716–5721. [Google Scholar] [CrossRef]
- Badisco, L.; Marchal, E.; Van Wielendaele, P.; Verlinden, H.; Vleugels, R.; Vanden Broeck, J. RNA interference of insulin-related peptide and neuroparsins affects vitellogenesis in the desert locust Schistocerca gregaria. Peptides 2011, 32, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Zhang, T.; Feng, Y.; Liu, X.; Zhang, L.; Chen, H.; Zeng, F.; Wang, M.; Liu, C.; Li, Y.; et al. Two insulin receptors coordinate oogenesis and oviposition via two pathways in the green lacewing, Chrysopa pallens. J. Insect Physiol. 2020, 123, 104049. [Google Scholar] [CrossRef] [PubMed]
- Werz, C.; Köhler, K.; Hafen, E.; Stocker, H. The Drosophila SH2B family adaptor Lnk acts in parallel to chico in the insulin signaling pathway. PLoS Genet 2009, 5, e1000596. [Google Scholar] [CrossRef]
- Kim, S.E.; Cho, J.Y.; Kim, K.S.; Lee, S.J.; Lee, K.H.; Choi, K.Y. Drosophila PI3 kinase and Akt involved in insulin-stimulated proliferation and ERK pathway activation in Schneider cells. Cell. Signal. 2004, 16, 1309–1317. [Google Scholar] [CrossRef]
- Hansen, I.A.; Attardo, G.M.; Roy, S.G.; Raikhel, A.S. Target of rapamycin-dependent activation of S6 kinase is a central step in the transduction of nutritional signals during egg development in a mosquito. J. Biol. Chem. 2005, 280, 20565–20572. [Google Scholar] [CrossRef]
- Wang, X.X.; Geng, S.L.; Zhang, X.S.; Xu, W.H. P-S6K is associated with insect diapause via the ROS/AKT/S6K/CREB/HIF-1 pathway in the cotton bollworm, Helicoverpa armigera. Insect Biochem. Mol. Biol. 2020, 120, 103262. [Google Scholar] [CrossRef]
- Nässel, D.R.; Vanden Broeck, J. Insulin/IGF signaling in Drosophila and other insects: Factors that regulate production, release and post-release action of the insulin-like peptides. Cell. Mol. Life Sci. 2016, 73, 271–290. [Google Scholar] [CrossRef]
- Chen, Q.; Li, G.; Pang, Y. A simple artificial diet for mass rearing of some noctuid species. Entomol. Knowl. 2000, 37, 325–327. [Google Scholar]
- Dong, X.; Zhong, G.; Hu, M.; Yi, X.; Zhao, H.; Wang, H. Molecular cloning and functional identification of an insect odorant receptor gene in Spodoptera litura (F.) for the botanical insecticide rhodojaponin III. J. Insect Physiol. 2013, 59, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Chen, J.; Xu, R.; Li, X.; Wang, Y.; Pan, X.; Zhang, C.; Li, Y.; Wang, F.; Li, C. Molecular identification and lipid mobilization role of adipokinetic hormone receptor in Spodoptera litura (F.). Bull. Entomol. Res. 2022, 18, 1–8. [Google Scholar] [CrossRef]
- Plantevin, G.; De Reggi, M.; Nardon, C. Changes in ecdysteroid and juvenile hormone titers in the hemolymph of Galleria mellonella larvae and pupae. Gen. Comp. Endocrinol. 1984, 56, 218–230. [Google Scholar] [CrossRef]
- Uno, T.; Furutani, M.; Sakamoto, K.; Uno, Y.; Kanamaru, K.; Mizoguchi, A.; Hiragaki, S.; Takeda, M. Localization and functional analysis of the insect-specific RabX4 in the brain of Bombyx mori. Arch. Insect Biochem. Physiol. 2017, 96, 1–13. [Google Scholar] [CrossRef]
- Taniguchi, C.M.; Emanuelli, B.; Kahn, C.R. Critical nodes in signalling pathways: Insights into insulin action. Nat. Rev. Mol. Cell Biol. 2006, 7, 85–96. [Google Scholar] [CrossRef]
- Liu, C.; Kuei, C.; Sutton, S.; Chen, J.; Bonaventure, P.; Wu, J.; Nepomuceno, D.; Kamme, F.; Tran, D.T.; Zhu, J.; et al. INSL5 is a high affinity specific agonist for GPCR142 (GPR100). J. Biol. Chem. 2005, 280, 292–300. [Google Scholar] [CrossRef]
- Yenush, L.; Fernandez, R.; Myers, M.G.; Grammer, T.C., Jr.; Sun, X.J.; Blenis, J.; Pierce, J.H.; Schlessinger, J.; White, M.F. The Drosophila insulin receptor activates multiple signaling pathways but requires insulin receptor substrate proteins for DNA synthesis. Mol. Cell. Biol. 1996, 16, 2509–2517. [Google Scholar] [CrossRef]
- Sheng, Z.; Xu, J.; Bai, H.; Zhu, F.; Palli, S.R. Juvenile hormone regulates vitellogenin gene expression through insulin-like peptide signaling pathway in the red flour beetle, Tribolium castaneum. J. Biol. Chem. 2011, 286, 41924–41936. [Google Scholar] [CrossRef]
- Tatar, M.; Kopelman, A.; Epstein, D.; Tu, M.P.; Yin, C.M.; Garofalo, R.S. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 2001, 292, 107–110. [Google Scholar] [CrossRef]
- Riehle, M.A.; Fan, Y.; Cao, C.; Brown, M.R. Molecular characterization of insulin-like peptides in the yellow fever mosquito, Aedes aegypti: Expression, cellular localization, and phylogeny. Peptides 2006, 27, 2547–2560. [Google Scholar] [CrossRef] [PubMed]
- Richard, D.S.; Rybczynski, R.; Wilson, T.G.; Wang, Y.; Wayne, M.L.; Zhou, Y.; Partridge, L.; Harshman, L.G. Insulin signaling is necessary for vitellogenesis in Drosophila melanogaster independent of the roles of juvenile hormone and ecdysteroids: Female sterility of the chico1 insulin signaling mutation is autonomous to the ovary. J. Insect Physiol. 2005, 51, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.W.; Wu, F.Y.; Ye, Y.; Li, T.; Zhang, Z.Y.; Zhang, H. Small GTPase Rab40C is upregulated by 20-hydroxyecdysone and insulin pathways to regulate ovarian development and fecundity. Insect Sci. 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- Sang, M.; Li, C.; Wu, W.; Li, B. Identification and evolution of two insulin receptor genes involved in Tribolium castaneum development and reproduction. Gene 2016, 585, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Nuss, A.B.; Brown, M.R.; Murty, U.S.; Gulia-Nuss, M. Insulin receptor knockdown blocks filarial parasite development and alters egg production in the southern house mosquito, Culex quinquefasciatus. PLoS Negl. Trop. Dis. 2018, 12, e0006413. [Google Scholar] [CrossRef]
- Pri-Tal, B.M.; Brown, J.M.; Riehle, M.A. Identification and characterization of the catalytic subunit of phosphatidylinositol 3-kinase in the yellow fever mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2008, 38, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, R.; Palli, S.R. Molecular analysis of nutritional and hormonal regulation of female reproduction in the red flour beetle, Tribolium castaneum. Insect Biochem. Mol. Biol. 2011, 41, 294–305. [Google Scholar] [CrossRef]
- Leyria, J.; Orchard, I.; Lange, A.B. Transcriptomic analysis of regulatory pathways involved in female reproductive physiology of Rhodnius prolixus under different nutritional states. Sci. Rep. 2020, 10, 11431. [Google Scholar] [CrossRef]
- Zhai, Y.; Sun, Z.; Zhang, J.; Kang, K.; Chen, J.; Zhang, W. Activation of the TOR Signalling Pathway by Glutamine Regulates Insect Fecundity. Sci. Rep. 2015, 5, 10694. [Google Scholar] [CrossRef]
- Dong, Y.; Chen, W.; Kang, K.; Pang, R.; Dong, Y.; Liu, K.; Zhang, W. FoxO directly regulates the expression of TOR/S6K and vitellogenin to modulate the fecundity of the brown planthopper. Sci. China Life Sci. 2021, 64, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Dittmer, N.T.; Sun, G.; Wang, S.F.; Raikhel, A.S. CREB isoform represses yolk protein gene expression in the mosquito fat body. Mol. Cell. Endocrinol. 2003, 210, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Süren-Castillo, S.; Abrisqueta, M.; Maestro, J.L. FoxO inhibits juvenile hormone biosynthesis and vitellogenin production in the German cockroach. Insect Biochem. Mol. Biol. 2012, 42, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.K.; Yang, W.J.; Tian, Y.; Wu, Y.B.; Wang, J.J. Insulin signaling pathway in the oriental fruit fly: The role of insulin receptor substrate in ovarian development. Gen. Comp. Endocrinol. 2015, 216, 125–133. [Google Scholar] [CrossRef]
- Goodman, M.H.; Potter, M.F.; Haynes, K.F. Effects of juvenile hormone analog formulations on development and reproduction in the bed bug Cimex lectularius (Hemiptera: Cimicidae). Pest Manag. Sci. 2013, 69, 240–244. [Google Scholar] [CrossRef]
- Sim, C.; Denlinger, D.L. Insulin signaling and FOXO regulate the overwintering diapause of the mosquito Culex pipiens. Proc. Natl. Acad. Sci. USA 2008, 105, 6777–6781. [Google Scholar] [CrossRef]
- Shiao, S.H.; Hansen, I.A.; Zhu, J.; Sieglaff, D.H.; Raikhel, A.S. Juvenile hormone connects larval nutrition with target of rapamycin signaling in the mosquito Aedes aegypti. J. Insect Physiol. 2008, 54, 231–239. [Google Scholar] [CrossRef]
- Abrisqueta, M.; Süren-Castillo, S.; Maestro, J.L. Insulin receptor-mediated nutritional signalling regulates juvenile hormone biosynthesis and vitellogenin production in the German cockroach. Insect Biochem. Mol. Biol. 2014, 49, 14–23. [Google Scholar] [CrossRef]
- Lu, K.; Chen, X.; Liu, W.T.; Zhang, X.Y.; Chen, M.X.; Zhou, Q. Nutritional Signaling Regulates Vitellogenin Synthesis and Egg Development through Juvenile Hormone in Nilaparvata lugens (Stål). Int. J. Mol. Sci. 2016, 17, 269. [Google Scholar] [CrossRef]
- Wheeler, D. The role of nourishment in oogenesis. Annu. Rev. Entomol. 1996, 41, 407–431. [Google Scholar] [CrossRef]
- Tufail, M.; Elmogy, M.; Ali Fouda, M.M.; Elgendy, A.M.; Bembenek, J.; Trang, L.T.; Shao, Q.M.; Takeda, M. Molecular cloning, characterization, expression pattern and cellular distribution of an ovarian lipophorin receptor in the cockroach, Leucophaea maderae. Insect Mol. Biol. 2009, 18, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Chase, J.; Sevala, V.L.; Schal, C. Lipophorin-facilitated hydrocarbon uptake by oocytes in the German cockroach Blattella germanica (L.). J. Exp. Biol. 2002, 205 Pt 6, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Fruttero, L.L.; Frede, S.; Rubiolo, E.R.; Canavoso, L.E. The storage of nutritional resources during vitellogenesis of Panstrongylus megistus (Hemiptera: Reduviidae): The pathways of lipophorin in lipid delivery to developing oocytes. J. Insect Physiol. 2011, 57, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Wang, Y.; Chen, X.; Zhang, X.; Li, W.; Cheng, Y.; Li, Y.; Zhou, J.; You, K.; Song, Y.; et al. Adipokinetic Hormone Receptor Mediates Trehalose Homeostasis to Promote Vitellogenin Uptake by Oocytes in Nilaparvata lugens. Front. Physiol. 2018, 9, 1904. [Google Scholar] [CrossRef]
- Ziegler, R.; Van Antwerpen, R. Lipid uptake by insect oocytes. Insect Biochem. Mol. Biol. 2006, 36, 264–272. [Google Scholar] [CrossRef]
- Fox, C.W.; Czesak, M.E. Evolutionary ecology of progeny size in arthropods. Annu. Rev. Entomol. 2000, 45, 341–369. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).