Abstract
Opportunistic bacteria are often ubiquitous and do not trigger disease in insects unless the conditions are specifically favorable for bacterial development in a suitable host. In this paper, we isolated and identified a bacterium, Pseudomonas aeruginosa, from the larvae of the giant mealworm Zophobas morio and we studied the possible entry routes by challenging larvae with per os injection and subdermal injection. We also evaluated the effect of exposing groups of larvae to P. aeruginosa inoculated in their feed and the effect of exposing wounded larvae to P. aeruginosa. We concluded that the mortality rate of Z. morio larvae is higher when P. aeruginosa gets in direct contact with the hemolymph via intracoelomic injection compared to a situation where the bacterium is force-fed. Larvae with an open wound exposed to P. aeruginosa presented higher mortality rate compared to larvae with a wound that was not exposed to the bacterium. We documented too, that cannibalism and scavenging were more prevalent among larvae in a group, when P. aeruginosa is present compared to when it is absent. We discuss hereby different aspects related with the pathogen’s entry routes to insects the complexity of pathogen´s transmission in high population densities and different ways to prevent and/or control P. aeruginosa in mass rearing systems.
1. Introduction
Insect mass-rearing aims to obtain large numbers of individuals [1] in a limited space under controlled production conditions. Unfavorable production conditions such as high population density, high relative humidity, suboptimal temperature, non-balanced diet, and inbreeding are among the factors that may cause insects to suffer from physiological stress. Such stress increases the chance for disease outbreaks by opportunistic pathogens, and poses a challenge for insect rearing in terms of yield, productivity, and microbiological contamination (particularly in cases when insects are reared for human food or animal feed) [2,3]. Two well-known bacterial pathogens infecting insects, plants, and mammals are Serratia marcescens and Pseudomonas aeruginosa [4,5,6]. Both bacteria are ubiquitous [7], and they inhabit soil, fresh water, and the rhizosphere. Furthermore, they are able to live on other different surfaces. Their success when thriving in several environments is due to their ability to use different substrates and their conversion of many different sources into utilizable energy [8]. Particularly for P. aeruginosa, its prevalence in natural environments is enhanced by its ability to form biofilms that increase the growth surface and protects the bacteria from environmental factors (i.e., UV light, desiccation, etc.) [9]. They are opportunistic insect pathogens since they are usually present in low numbers inside or outside the insects without causing disease, but they may become pathogenic under certain stress conditions for the insects and/or when the insects have a weakened immune system [10]. Serratia marcescens is present as part of the gut microbiota of some sandflies and triatomine species [11]. Examples of S. marcescens, isolated as a pathogen from insects of the orders Coleoptera, Diptera, Orthoptera, and Lepidoptera, have been reported [12,13,14,15,16,17,18]. Similarly, P. aeruginosa has been isolated from different weevil species (the pecan weevil Curculio caryae [14]; the southern pine weevil Dendroctonus brevicomis [12]; the red palm weevil Rynchophorus ferrugineus [19], the tobacco and tomato hornworms Manduca sexta and M. quinquemaculata [20], from Schistocerca gregaria eggs [21] in the wild and from several other insect species. P. aeruginosa epizootic outbreaks have been reported in several insect species in laboratory rearings, but no epizootic event has been recorded in nature. High relative humidity combined with high temperature, are conducive factors for the development of P. aeruginosa in insects [7]. Virulence factors recognized for P. aeruginosa pathogenesis are the production of metalloproteases alkaline proteinase (aeruginolysin), elastase B (pseudolysin), elastase A (staphylolysin), and one serine proteinase. Also, the production of phospholipase C, proteinase IV, pili, flagella, exotoxins A, lipopolysaccharids (LPS), enzymes secreted by the type III secretion system, as well as quorum sensing (with its implications in biofilm formation) are also virulence factors [8,22]. For example, Exotoxin A has been associated with the inhibition of the synthesis of intracellular proteins [23]. A recent case of P. aeruginosa detection in large scale rearing occurred in a giant mealworm (Z. morio) production system in China [24]. Zophobas morio is one of three mealworm species that are being explored and mass reared as an alternative source of protein [25,26], and control of diseases is essential. At the end of 2015, we received in our laboratory a sample of Z. morio larvae from a commercial rearing colony with a high number of insects dying within a few days. We looked for pathogens in the larvae and we detected and isolated the bacterium P. aeruginosa in vitro. Our aim for the current study was to observe and describe the disease symptoms and to evaluate experimentally the different potential entry routes of the pathogen.
2. Materials and Methods
2.1. P. aeruginosa (Isolation, Identification and Preservation)
Infected larvae were obtained from a commercial insect production company. First, hemolymph from symptomatic alive larvae (immobilized prior to dissection for 10 min at −20 °C) and dead larvae (different times post mortem) was observed using light microscopy. Diseased and dead larvae were then surface sterilized (ethanol 70%; water; chlorine 3%, 1 min; water; ethanol 70%; water 2×) and dissected under sterile conditions. A 10 µL loop with hemolymph from individual larvae was streaked on nutrient agar (Standard I nutrient agar, Merck, Darmstad, Germany). Streaked plates were incubated at 32 °C for 48 h and thereafter verification of cell and colony morphology was done. A highly predominant type of colony was detected across the different streaked plates. Such type of colony was observed to be the only one growing particularly in plates streaked with hemolymph of symptomatic larvae and recently dead larvae (i.e., 24–48 h post mortem). Of these colonies, a single one was picked and thereafter inoculated in an Erlenmeyer flask (V = 250 mL) containing 30 mL of nutrient broth (Standard I nutrient broth, Merck, Darmstad, Germany) in an orbital shaker at 180 rpm and 32 °C for 24 h Cryopreservation vials were made by adding 20% v/v glycerol to the bacterial liquid culture. After confirmation of a gram negative bacterium, biochemical characterization was done using an API20E test strip (Biomeurieux, Grenoble, France) for gram negative bacteria. For molecular identification, 500 µL of bacterial liquid culture (cultured as described above) were pelleted at 5000 rpm for 5 min and 5 °C. The pellet was subjected to total DNA extraction. DNA extraction was done using the DNeasy® Plant Mini Kit (Qiagen GmbH, Hilden, Germany). For PCR, 1 µL of DNA solution was added to a mix of 5 µL of each of the primers from the 16sRNA gene in bacteria (27F: AGAGTTTGATCMTGGCTCAG and 806R: GGACTACNNGGGTATCTAAT), 25 µL of premixed dNTp’s (TAKARA BIO, Shiga, Japan)) and Taq polymerase (AmpliTaq Gold polymerase & MgCl2, APPLIED BIOSYSTEMS, TermoFischer Scientific, Waltham, MA, USA) and 15 µL of water. The PCR program included in total 39 cycles of 15 s at 92 °C, 1 min at 55 °C and 50 s at 74 °C. PCR products were visualized on a 1% agarose gel and were purified with a GFX™ PCR DNA and Gel Band Purification Kit (GE Healthcare, Chicago, IL, USA). PCR products sequencing service was performed by Beckman Coulter Genomics (now Genewiz, South Plainfield, NJ, USA), using the same pair of primers used for DNA amplification. Alignment of the obtained sequence (Appendix A) and highly similar sequences (Megablast) with a maximum target of 100 sequences, was performed by using the software BLASTN 2.8.0+ (NCBI, Bethesda, MD, USA) [27].
2.2. Zophobas morio
Healthy larvae were purchased from a local retailer. Seventy larvae (~0.8–1 g) were placed individually in acrylic boxes for pupation. They were fed one carrot slice every 4 days and ad libitum diet (DT2) formulated at our laboratory (55.5% wheat bran, 25% oats, 16% brewer yeast, 3.5% casein). After eclosion, 50 non-sexed adults were randomly selected, combined in one container for mating to occur and reared to get a new generation of larvae. From this new generation, larvae of ~0.68–0.73 g (N = 54) were selected for per os injection and subdermal injection; larvae weighing ~0.07 g (N = 14 batches of 5 larvae; average larval weight was calculated) were used for the experiment of group exposure in the feed. Starting one day after injection, Z. morio larvae were fed every third day with a cube (~0.8 g) of agar diet (400 mL distilled water, 15 g DT2 and 10 g bacteriological agar) during and after the injection (per os and subdermal) and during and after the exposure by wounding. For the group exposure bioassay, one cube of agar diet was added on day 10 after the start of the experiment. All the experiments were incubated in darkness at 28 °C and at an average relative humidity of ~70%.
2.3. Infection Experiments
P. aeruginosa was cultured by inoculating 750 µL from a cryopreservation vial (prepared as described above) in an Erlenmeyer flask (V = 250 mL) containing 30 mL of nutrient broth (Standard I nutrient broth). The culture was incubated at 200 rpm for 20 h at 37 °C. Bacterial cells were washed three times (5000 rpm, 7 min, 5 °C) with a 10 mM MgSO2 solution in demineralized water. A bacterial suspension was prepared by adjusting OD600 (BioPhotometer, Eppendorf, Hamburg, Germany) to 0.7, equivalent to a concentration of ≈5.4 × 108 cells/mL. Our bio-assay procedures, including the doses used in each type of experiments, were based on pilot experiments. The doses were chosen in order to address the research aims in the best way.
We conducted two bioassays in which purposeful injection of a specific dose was conducted, either per os (force-feeding) or subdermal. For these two experiments, larvae were treated and incubated individually. We also conducted one experiment in which groups of insects were exposed to P. aeruginosa cells in the feed. This experiment mimicked the natural wounding and scavenging that occurs in reared mealworms due to cannibalism. A last experiment was conducted in which Z. morio larvae were purposefully wounded and individually exposed to bacterial cells. Table 1 summarizes the experiments described above.

Table 1.
Summary of experiments conducted in this work.
Per os injection: Force-feeding was performed by manually injecting 10 µL aliquots of a bacterial suspension to each of 20 larvae by using a blunt 0.32 gauge needle attached to a 100 µL syringe (Hamilton, Reno, NV, USA). Batches of 12 larvae were immobilized for 5 min at −20 °C prior injection. Two doses and a control were included in the experiment: a dose (D1) of approximately 1.1 × 107 UFC/larva, a dose (D2) consisting of approximately half of the cells in D1 (D2 ≈ 5.4 × 106 UFC/larva) and 10 mM MgSO2 solution as the control. Injection bioassays were done in triplicate. Mortality was recorded daily during 15 days.
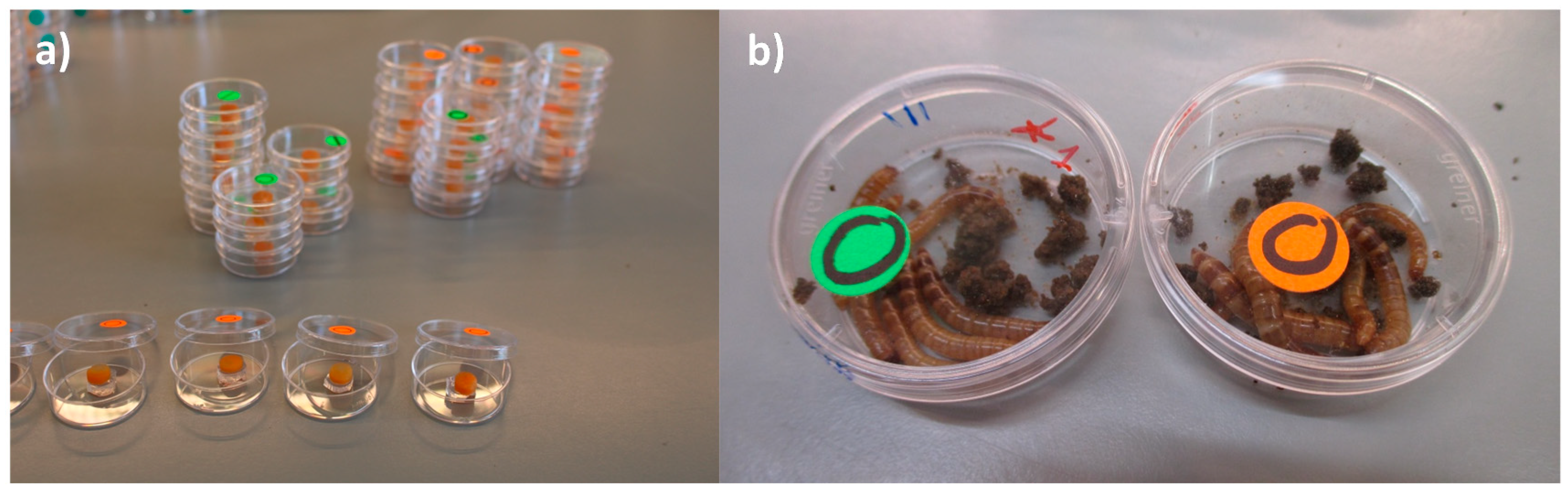
Group exposure (in the feed): Five batches of 6 larvae (each larva weighed ~0.07 g) were placed each in a Petri dish (d = 3 cm, a = 5 cm2). Each batch was exposed as a group to a total load of ≈ 2.16 × 108 (≈3.6 × 107 UFC/larvae). A 24 µL droplet of a bacterial suspension of ≈9.0 × 109 UFC/mL was deposited on a feeding arena made with a carrot disc (d = 0.8 cm; thickness ≈0.3 cm; weight ~0.16 g) that was placed on top of a 10 mM MgSO2 solution-agar disc in the middle of a Petri dish. The larvae of the control treatment were exposed to 24 µL of 10 mM MgSO2 (Figure 1a,b). The number of larvae chosen for this experiment was the maximum number of larvae that fitted in the petri dish area including the carrot-agar arena. We considered this setting to be representative of a high population density. The number of dead (categorized as visibly cannibalized or not) and missing larvae (due to cannibalism) was recorded daily during 15 days. The dead larvae were not removed as our aim was to mimic the densely populated mass-rearing conditions in which normally, removal of sick/dead individuals does not occur. In this experiment, it was not possible to determine the cause of dead for the larvae in each group (i.e., whether a larva died due to injuries caused by cannibalism and subsequent infection via the hemolymph, whether a weak (infected or not) larva was cannibalized before dead or whether a larva died due to P. aeruginosa’s infection and it was afterwards consumed by a conspecific larva). Hence, we expressed all three possible options as cannibalism and reported it as the difference between the total number of insects from the original group and that of the insects that remained at the end of the experiment. The group exposure experiment was done in triplicate.
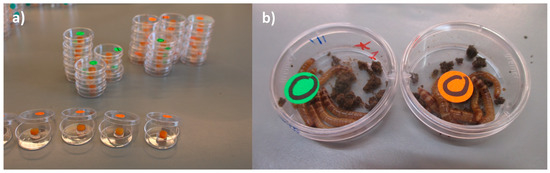
Figure 1.
(a) Set-up of group exposure of larvae to P. aeruginosa (in the feed); (b) control larvae (green label) and larvae exposed to bacterial cells (orange label) 10 dpi.
Subdermal injection: Subdermal infection was performed by manually injecting 20 larvae with 10 µL aliquots of a bacterial suspension in the 2nd sternite (ventral) with a 45° beveled point 0.30 gauze needle attached to a 100 µL syringe (Hamilton, Reno, NV, USA). Two doses and a control were included in the experiment: a dose of approximately 5.4 × 106 UFC/larva (D2: same as for per os injection), a dose which corresponds to approximately one tenth of D2 (D3 ≈ 5.4 × 105 UFC/larva) and 10 mM MgSO2 solution as the control. Injection bioassays were done in triplicate. Mortality was recorded daily during 5 days.
Wounding: Wounding was done by abscission of the left hind leg tarsus of each of 20 larvae with sterile dissection scissors. One 20 µL droplet of a bacterial suspension with approximately 1.1 × 109 UFC/mL (≈2.16 × 107 UFC in total) was placed in the bottom of a Petri dish (d = 6 cm); a 20 µL droplet of a 10 mM MgSO2 solution was used as the control. A purposefully wounded larva was placed with the wound on top of the droplet. The experiment was conducted in duplicate. Mortality was checked daily for 10 days.
2.4. Statistical Analysis of Bioassays
Mortality values for the group of larvae injected per os (corrected by Henderson-Tilton’s formula) were distributed normally and subjected to One Way Analysis of Variance (ANOVA) at a significance level of 0.05, followed by a pairwise multiple comparison by the Student-Newman-Keuls test. Cannibalism rates in groups of larvae exposed (or not exposed) to P. aeruginosa were distributed normally and analyzed with a Student’s t-test at a significance level of 0.05. Data of mortality due to subdermal injection did not conform to the normal distribution so they were analyzed by a Krustal-Wallis test at a significance level of 0.05, followed by a pairwise multiple comparison by the Student-Newman-Keuls test. Mortality values of larvae exposed to P. aeruginosa by purposeful wounding were distributed normally and analyzed with a Welch’s t-test at a significance level of 0.05. All statistical analyses were performed using SigmaPlot v. 14 (Systat Software, San Jose, CA, USA).
3. Results
3.1. Observations on Disease Symptoms in Z. morio Larvae
Diseased larvae received from the insect producer ceased feeding and curved their bodies with a ventral orientation, almost entirely losing their mobility. This position could last for 2–4 days before the insects died, however close to the death the insect body became flaccid most of the times. Molting frequency seemed lower for diseased larvae compared to healthy larvae. Cannibalism of diseased larvae was observed, often in the legs’ region. Melanization started with small stains which increased in size during the 72 h after the first stain appeared. Melanization eventually took over parts of or even most of the body in moribund larvae, as a result of a septicemia and lysis of the larval inner organs and tissues, resulting in the characteristic dark brown colored larvae. Dead larvae turned black, especially towards each end (Figure 2a,b). An unpleasant odor was characteristic for moribund and dead larvae. For larvae where melanization was absent, the gut remained undisrupted. In those where melanization started, the gut was in a phase of disintegration.

Figure 2.
(a) Diseased Z. morio larvae displaying melanized stains before death (white arrows); (b) Z. morio larvae in a process of melanization, 48 h after death.
3.2. Characterization of P. aeruginosa
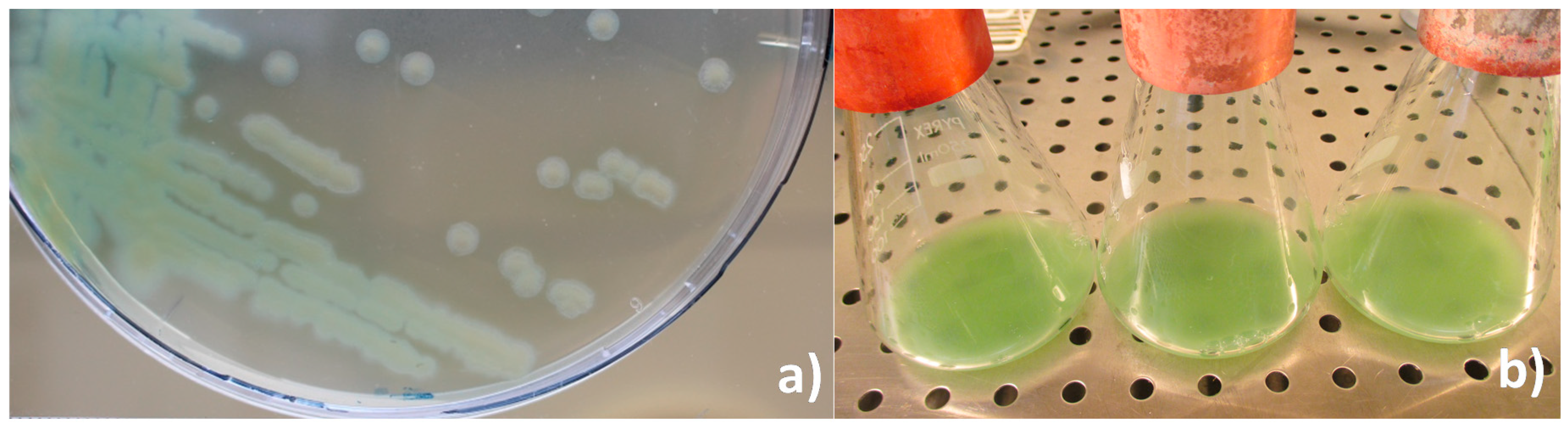
Colony morphology and in vitro cultivation: By direct observation of the hemolymph from diseased larvae, the presence of bacilliform non-sporulated, non crystalliferous bacterial cells was confirmed. In vitro cultivation in nutrient agar resulted in just one type of colonies: greenish, convex but flattened as growth progressed, round with slight irregular borders and slimy (Figure 3a), suggesting a monospecific infection. In vitro liquid cultivation in nutrient broth resulted in a yellow-green pigmented culture that changed color to green-blueish when agitated (Figure 3b).
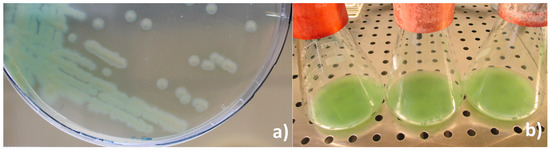
Figure 3.
(a) In vitro solid (nutrient agar) and (b) liquid culture (nutrient broth) of P. aeruginosa isolate from Z. morio larvae.
Biochemical characterization Comparison of the biochemical tests was done using corresponding values for reference strains of P. aeruginosa and Pseudomonas fluorescens/putida reported in the API database (Table 2).

Table 2.
Biochemical characterization of the P. aeruginosa isolate from Z. morio by the API20E test.
Molecular identification: The length of the amplified 16s RNA gene fragment for the our bacterial isolate was 770 bp. Out of 100 hits, 95 sequences produced significant alignments with different accessions of P. aeruginosa, including sequences of the 16S ribosomal RNA gene and also sequences of the whole genome of P. aeruginosa (i.e., NCBI—reference strains PAO1 and PA14; Accession numbers: NC_002516AE00409). The query cover for the alignment was 100% as well as the sequences identity, with a total maximum score of 1423 and a 735 bp effective length of the query.
3.3. Infection Experiments
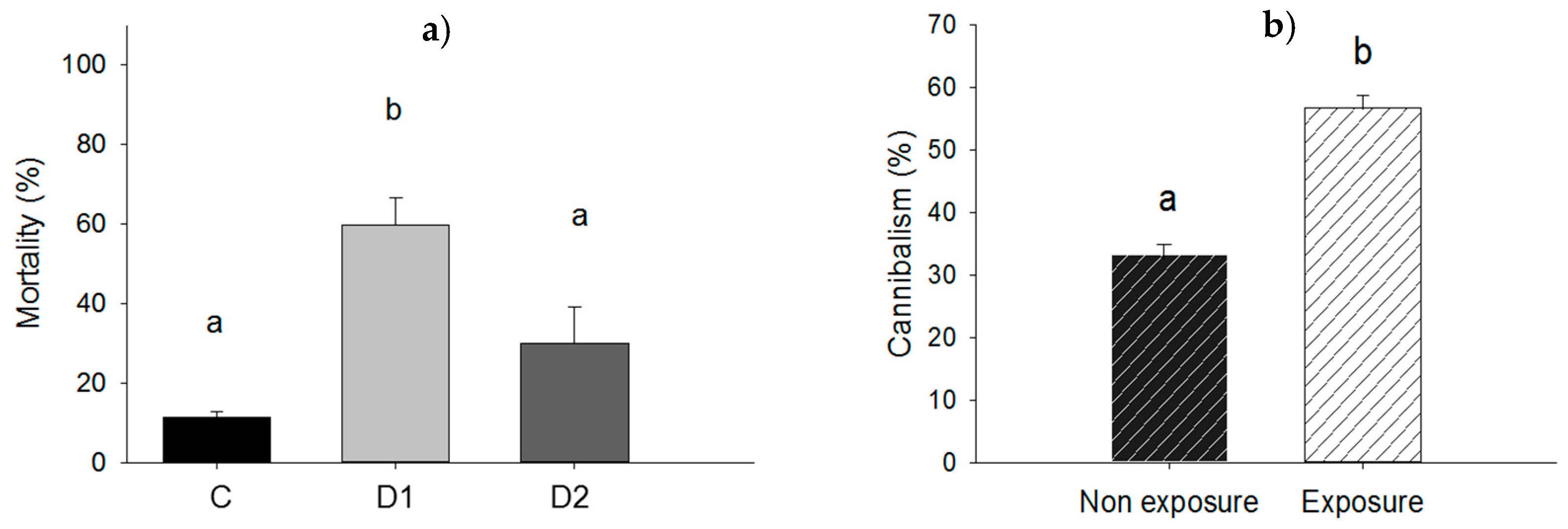
Force-feeding of larvae with P. aeruginosa resulted in higher mortality at the dose D1 when compared to dose D2: 59.6% vs. 30.0%; p = 0.020 and significantly higher than mortality in control (p = 0.005). However, mortality was not significantly different for the control and dose D2 (11.3% vs. 30.0%; p = 0.096) as shown in Figure 4a. For the experiment in which larvae were exposed to P. aeruginosa in groups, there were no dead larvae at the end of the experiment and cannibalism rate was compared in exposed and non-exposed groups of larvae (Figure 5a,b). Amongst larvae exposed in groups to ≈2.2 × 108 UFC, cannibalism rate was 56.6% compared to 33.3% for unexposed control larvae (p = 0.001) (Figure 4b).
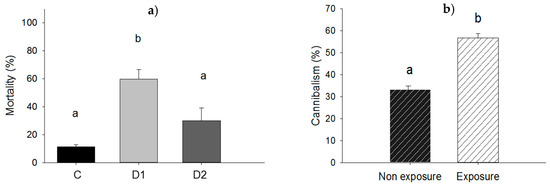
Figure 4.
(a) Mortality rate of larvae force-fed with cells of P. aeruginosa at doses D1 of ≈1.1 × 107 UFC/larva (D1), D2 of ≈5.4 × 106 UFC/larva (D2) or 10 mM MgSO4 solution as a control (C); (b) cannibalism rate among larvae in groups exposed to ≈2.2 × 108 UFC of P. aeruginosa. Bars having no letters in common differ significantly (p ≤ 0.05). Error bars indicate the standard error of the mean.

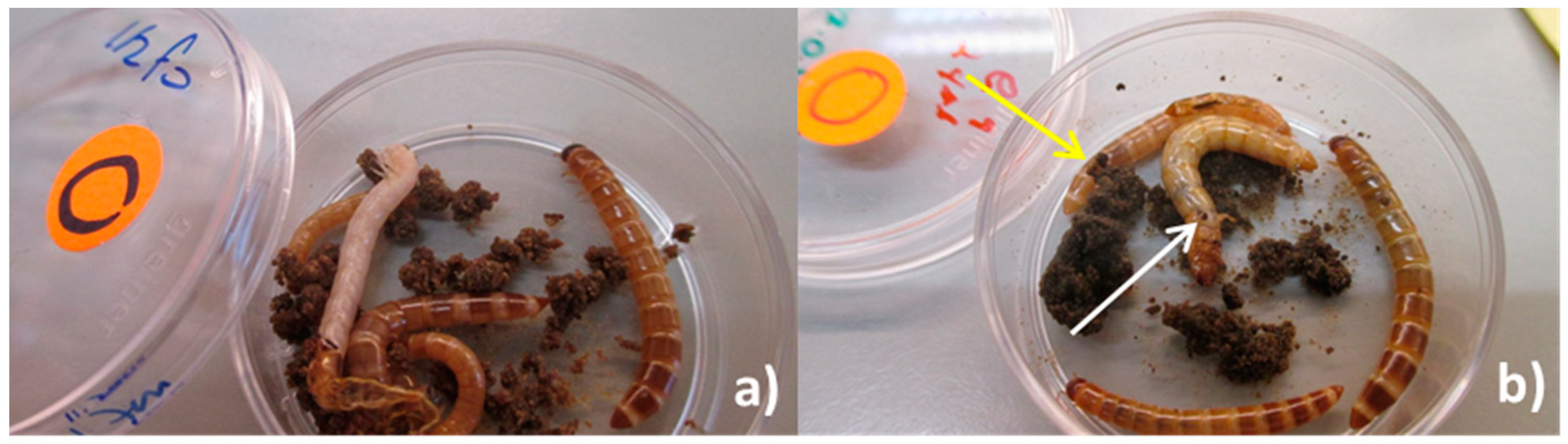
Figure 5.
(a) Larvae molting in group exposure experiment; (b) injury due to cannibalism in larva’s proleg (white arrow) and cannibalized dead larva (yellow arrow).
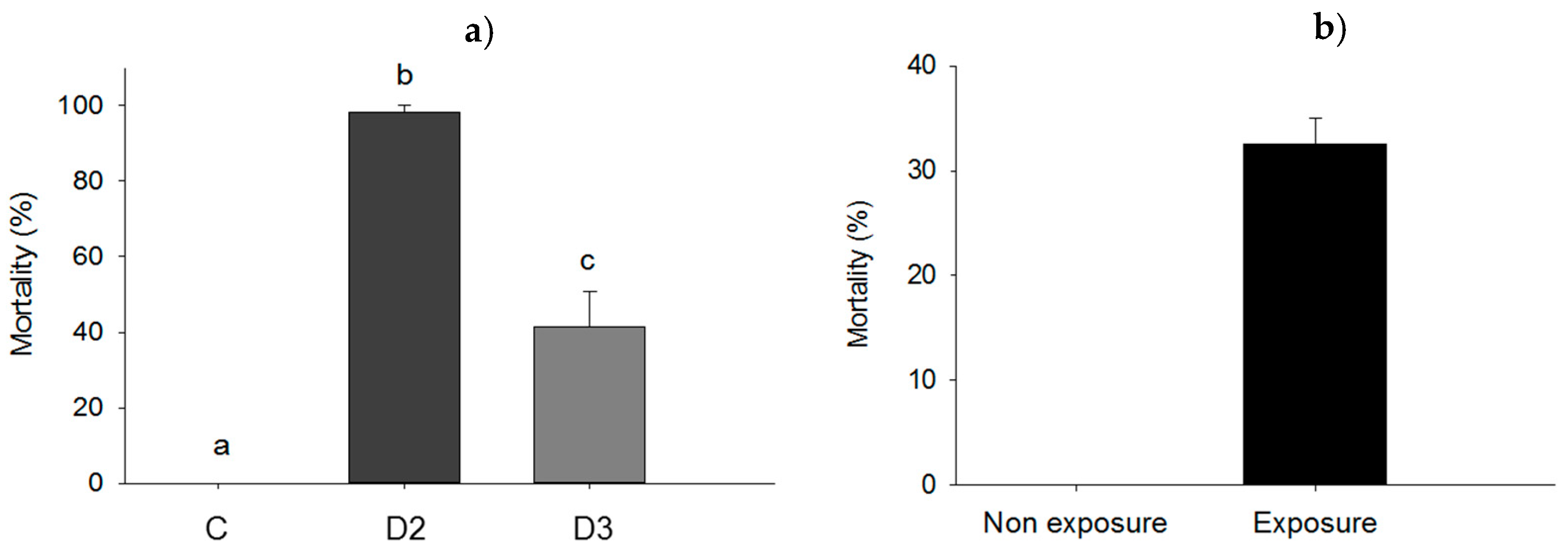
Subdermal injection without bacteria did not affect larval survival (0% mortality rate for control). However, the mortality rate of larvae subjected to subdermal injection with dose 2 (D2) was significantly higher (98.3%; p = 0.05) compared to the mortality rate of larvae injected with dose 3 (D3) (41.3%; p = 0.05) and the control (Figure 6a). Wounding in itself was found not to influence mortality (0% in the control) whereas wounding and exposure to ≈2.2 × 108 cells of P. aeruginosa resulted in a mortality rate of 32.5% (p = 0.049) (Figure 6b).
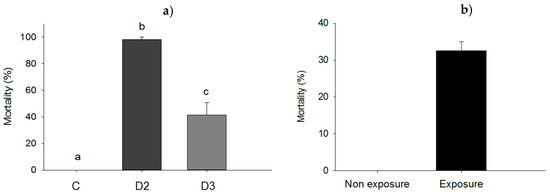
Figure 6.
(a) Mortality rate of larvae injected subdermal with either dose 2 (D2) of ≈5.4 × 106 UFC/larva), dose 3 (D3) of ≈5.4 × 105 UFC/larva) of P. aeruginosa cells, or 10 mM MgSO2 solution as a control (C); (b) mortality of Z. morio larvae subjected to P. aeruginosa exposure by wounding. Bars having no letters in common and differ significantly (p ≤ 0.05). Error bars indicate the standard error of the mean.
4. Discussion and Conclusions
The bacterium isolated from larvae of the giant mealworm Z. morio was identified as P. aeruginosa, based on the following well-known physical and biochemical characteristics (colony morphology, culture medium affinity, culture color) and ultimately by molecular tools. A relevant characteristic was siderophore chromatogenesis, a key feature for members of the Pseudomonas genus [28], also confirmed by the green-yellowish color of the in vitro solid and liquid cultures. Siderophores pyocianin and pyoverdin are essential virulence factors of P. aeruginosa during the infection process in animal species [29,30].
Diseased Z. morio larvae exhibited general symptoms that are described for infection of other insect species with P. aeruginosa: weakness, sluggishness, cease of feeding, melanization, putrid odor, and finally, death [19,21]. Importantly, the first symptoms of infection caused by P. aeruginosa can go unnoticed, and in several cases, only when insects are moribund and start to die, the disease becomes apparent [21,23]. P. aeruginosa is normally present in small numbers and can cause disease if it enters the insect hemocoel by conducive factors, which weaken the insects by means of different physiological stressors. Most bacteria enter the insect hemocoel by an injury or aided by a “support host factor” (i.e., gut pH) [23]. Regardless of the entry route, the wide range of P. aeruginosa virulence factors allows the bacterium to induce disease in insects, if conditions for such infections are favorable.
The mechanism of P. aeruginosa causing disease after getting in contact with the hemolymph is well known, while the mechanisms by which ingestion of a high number of P. aeruginosa cells can overcome the gut barriers and reach the insect hemocoel are still poorly understood. It is documented that as few as 10 to 20 cells of P. aeruginosa are enough to kill a grasshopper when injected directly into the hemocoel, compared to a wide range of 800 to 29,000 cells when incorporated in the feed (Table 3). In our study, mortality rates due to subdermal infection occurred as early as reported in the literature for other insects, though the doses we used were higher than those used in similar studies (Table 3). Our isolate of P. aeruginosa caused lower mortality of Z. morio larvae at the same time post-infection and overall when injected per os, compared to when injected directly into hemocoel at the same dose. These results are in accordance with studies on mortality rates in other insect species subjected to P. aeruginosa. In such studies, LD50 values and effective doses are much smaller for hemolymph injection compared to force-feeding or incorporation in the feed [23]. It is reported that regardless of the initial dose, P. aeruginosa reaches concentrations of 1 × 108 to 1 × 109 cells just before the host’s death for some insect species such as grasshoppers, the great wax moth Galleria mellonella, and the yellow mealworm Tenebrio molitor [21,23]. Looking at our own results, the overall mortality rate of larvae force-fed with P. aeruginosa was relatively high a very short time post infection (24 h). Hence, we hypothesize that a dose of ≈1.1 × 107 UFC/larva would be high enough to allow bacterial reproduction and toxin synthesis within one day and would allow bacteria to be able to trespass the midgut epithelium and spread into the hemocoel. In this regard, it is reported that the pilli and flagella of P. aeruginosa act as adhesins by binding to a host cell receptor, establishing an essential linkage for the persistence and spread of P. aeruginosa by epithelial cell invasion/infection and cytotoxicity [8]. Thus, the dose of the initial inoculum of P. aeruginosa that enters an insect body is an important determinant of disease development in terms of intensity (post infection bacterial load) and speed (days post infection until death) [23].

Table 3.
Mortality rate of different insect species challenged with P. aeruginosa by different methods.
Our results from the group exposure to P. aeruginosa exemplify how an opportunistic bacterial pathogen may affect production systems where insects are reared at high population densities during part or the whole of their life cycles, such as Z. morio. We found that the cannibalism rate was 23% higher for the groups exposed to P. aeruginosa, compared to the non-exposed groups of larvae, supporting the evidence of cannibalism as a significant enabler for insect pathogens to be transmitted [36,37]. Cannibalism is a common behavior, documented for quite a number of insect species [38]. Cannibalism often affects individuals that are smaller, weaker or more vulnerable, especially, at high population densities where food and space competition are more likely to occur. Molting and pupation are the susceptible stages for cannibalism to occur in mealworms, particularly for Z. morio larvae, for which a higher risk for pathogen transmission rate exists due to the fact that pupation is inhibited by crowding. Molting can be prolonged indefinitely until larvae die of old age if they do not find the chance to disperse as individuals, in order to pupate [39,40]. At the same time, our own experiments showed that ingesting a relatively high amount of bacterial cells (≈1.1 × 107 UFC/larva) killed roughly 60% of the larvae, implying larvae may get weakened after the ingestion of bacteria, becoming more susceptible to be cannibalized upon, becoming a source for subsequent infection of other larvae. In this regard, transmission takes place because besides cannibalism, which occurs to living individuals, there is the added effect of scavenging or conspecific necrophagy, which implies feeding on dead (infected) larvae.
On the same line, our experiment of purposeful wounding (average mortality rate = 32%) suggests that if larvae get accidentally or purposefully injured by a conspecific individual, a pathogen present in the environment may rapidly infect the insects. It has been reported that dead grasshoppers, due to P. aeruginosa infection, harbored a load of 2.4 × 1010 bacterial cells [21], which could potentially contaminate directly the substrate and serve as a concentrated inoculum for progressive bacterial growth on the substrate and frass.
Not every insect species reared under high population density is as susceptible to pathogens [41,42] as mealworms, however, there are other factors that can contribute to infection by opportunistic bacteria such as P. aeruginosa: mixed infections as those occurring to other host-pathogen complexes [43,44,45,46], inbreeding [47,48], nutritional imbalance [49,50], and the relation between temperature and relative humidity [6,51,52,53].
5. Conclusions
We concluded that P. aeruginosa is lethal for Z. morio larvae at specific doses when injected intracoelomically and per os. Mortality rate is higher for wounded larvae that are exposed to P. aeruginosa compared to wounded larvae that are not exposed. We concluded too that the transmission of an opportunistic pathogen as P. aeruginosa at a high host population density is the result of the interaction of different factors. Pseudomonas aeruginosa’s transmission is enhanced by cannibalism, added the effects of scavenging. Insect behaviors that rarely occur in nature for some insect species may therefore be of high significance in commercial insect rearing, and it is a challenge for the insect industry to manage insect pathogens benefiting from cannibalism. P. aeruginosa is an example of one such bacterium whose prevention is needed since it may be detrimental for insect production. Contact with high dosages should be avoided for the staff (from a working environments’ perspective), since the bacterium is opportunistic. To prevent or manage P. aeruginosa, several actions must be taken. First, moribund and dead individuals must be scouted and removed immediately and treated as microbial waste before being discarded (i.e., by pyrolysis). Second, insect feed and the equipment used must be checked for presence of bacterial contamination and sterilized.
Additional actions that should be taken include the following: (a) to verify that the temperature of the water for washing trays and other equipment is around 50 to 60 °C; (b) to include a “soaking step”, where the rearing trays and other plastic materials remain for some minutes in a solution containing an EU approved disinfectant (highly advisable if contains ethylene diamine tetra acetic acid EDTA, due to its ability to reduce the formation of biofilms in surfaces of bacteria like P. aeruginosa); and (c) to reduce the relative humidity in the rearing and in the substrates (below ~60%) as P. aeruginosa thrives well in high relative humidity and it is highly sensitive to desiccation.
As a final remark, it is important to mention that in insect rearing, the production system must be evaluated and eventually redesigned to avoid stressed and weakened insects and to minimize the risk of cannibalism.
Author Contributions
Conceived and designed the experiments: G.M.-V., A.B.J. and J.E. Curated the data: G.M.-V. Performed the experiments: G.M.-V. Analyzed the data/software: G.M.-V. Contributed to project administration/reagents/materials: A.B.J. and J.E. Validation: A.B.J. and J.E. Wrote the paper: G.M.-V., A.B.J. and J.E.
Funding
This article was written as part of the Ph.D. project of Gabriela Maciel-Vergara, supported by the Agricultural Transformation by Innovation (AgTrain) Erasmus Mundus Joint Doctorate Program, funded by the EACEA (Education, Audiovisual and Culture Executive Agency) of the European Commission.
Acknowledgments
We thank Monique van Oers and Joop van Loon for helpful discussions on the experimental set up, results and the way forward for this work. We thank Joop van Loon for reading and commenting on an earlier version of this manuscript. We thank Proti-Farm R&D BV (Netherlands) for providing material for our insect rearing.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, and in the decision to publish the results.
Appendix A. FASTA Sequence of P. aeruginosa Isolate from Z. morio
ATGGCTCAGATTGAACGCTGGCGGCAGGCCTAACACATGCAAGTCGAGCGGATGAAGGGAGCTTGCTCCTGGATTCAGCGGCGGACGGGTGAGTAATGCCTAGGAATCTGCCTGGTAGTGGGGGATAACGTCCGGAAACGGGCGCTAATACCGCATACGTCCTGAGGGAGAAAGTGGGGGATCTTCGGACCTCACGCTATCAGATGAGCCTAGGTCGGATTAGCTAGTTGGTGGGGTAAAGGCCTACCAAGGCGACGATCCGTAACTGGTCTGAGAGGATGATCAGTCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAGGCAGCAGTGGGGAATATTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGTCTTCGGATTGTAAAGCACTTTAAGTTGGGAGGAAGGGCAGTAAGTTAATACCTTGCTGTTTTGACGTTACCAACAGAATAAGCACCGGCTAACTTCGTGCCAGCAGCCGCGGTAATACGAAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGCGCGTAGGTGGTTCAGCAAGTTGGATGTGAAATCCCCGGGCTCAACCTGGGAACTGCATCCAAAACTACTGAGCTAGAGTACGGTAGAGGGTGGTGGAATTTCCTGTGTAGCGGTGAAATGCGTAGATATAGGAAGGAACACCAGTGGCGAAGGCGACCACCTGGACTGATACTGACACTGAGGTGCGAAAGCGTGGGGAGCAAACAGGATTAGATA
References
- Boller, E. Behavioral aspects of mass-rearing of insects. Entomophaga 1972, 17, 9–25. [Google Scholar] [CrossRef]
- Fuxa, J.R.; Sun, J.Z.; Weidner, E.H.; LaMotte, L.R. Stressors and rearing diseases of Trichoplusia ni: Evidence of vertical transmission of NPV and CPV. J. Invertebr. Pathol. 1999, 74, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Steinhaus, E.A. Crowding as a possible stress factor in insect disease. Ecology 1958, 39, 503–514. [Google Scholar] [CrossRef]
- Tanada, Y.; Kaya, H. Insect Pathology, 1st ed.; Academic Press: San Diego, CA, USA, 1993; pp. 147–170. ISBN 0-12-683255-2. [Google Scholar]
- Bucher, G.E. Chapter 4: Non sporulating bacterial pathogens. In Insect Pathology, an Advanced Treatise, 1st ed.; Steinhaus, E., Ed.; Academic Press: New York, NY, USA, 1963; Volume 2, pp. 117–149. [Google Scholar]
- Hendrickson, E.L.; Plotnikova, J.; Mahajan-Miklos, S.; Rahme, L.G.; Ausubel, F.M. Differential roles of the Pseudomonas aeruginosa PA14 rpoN gene in pathogenicity in plants, nematodes, insects, and mice. J. Bacteriol. 2001, 183, 7126–7134. [Google Scholar] [CrossRef] [PubMed]
- Jurat-Fuentes, J.L.; Jackson, T.A. Chapter 8: Bacterial entomopathogens. In Insect Pathology, 2nd ed.; Vega, F.E., Kaya, H.K., Eds.; Academic Press: London, UK, 2012; pp. 265–350. [Google Scholar]
- Lyczak, J.B.; Cannon, C.L.; Pier, G.B. Establishment of Pseudomonas aeruginosa infection: Lessons from a versatile opportunist. Microb Infect. 2000, 2, 1051–1060. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the Natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95. [Google Scholar] [CrossRef] [PubMed]
- Sikorowski, P.P.; Lawrence, A.M. Microbial contamination and insect rearing. Am. Entomol. 1994, 40, 240–253. [Google Scholar] [CrossRef]
- Azambuja, P.; García, E.S.; Ratcliffe, N.A. Gut microbiota and parasite transmission by insect vectors. Trends Parasitol. 2005, 21, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Fuxa, J.R.; Ayyappath, R.; Goyer, R.A. Pathogens and Microbial Control of North American Forest Insect Pests; Forest Health Technology Enterprise Team, USDA Forest Service: Washington, DC, USA, 1998; Volume 97.
- Moore, G.E. Isolating entomogenous fungi and bacteria, and tests of fungal isolates against the Southern pine beetle. J. Econ. Entomol. 1970, 63, 1702–1704. [Google Scholar] [CrossRef]
- Sri-Arunotai, S.; Sikorowski, P.P.; Neel, W.W. Study of the pathogens of the pecan weevil larvae. Environ. Entomol. 1975, 4, 790–792. [Google Scholar] [CrossRef]
- Grimont, F.; Grimont, P.A.D. The Genus Serratia. In The Prokaryotes; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 219–244. ISBN 978-0-387-25496-8. [Google Scholar]
- Prinsloo, H.E. The phospholipase and gelatinase activity of gram-negative bacteria isolated from diseased insects, and typing of strains of Serratia marcescens. J. Invertebr. Pathol. 1967, 9, 420–427. [Google Scholar] [CrossRef]
- Scrascia, M.; Pazzani, C.; Valentini, V.; Oliva, M.; Russo, V.; D’Addabbo, P.; Porcelli, F. Identification of pigmented Serratia marcescens symbiotically associated with Rhynchophorus ferrugineus Olivier (Coleoptera: Curculionidae). Microbiol. Open 2016, 5, 883–890. [Google Scholar] [CrossRef] [PubMed]
- Bidari, F.; Shams-Bakhsh, M.; Mehrabadi, M. Isolation and characterization of a Serratia marcescens with insecticidal activity from Polyphylla olivieri (Col.: Scarabaeidae). J. Appl. Entomol. 2018, 142, 162–172. [Google Scholar] [CrossRef]
- Banerjee, A.; Dangar, T. Pseudomonas aeruginosa, a facultative pathogen of red palm weevil, Rhynchophorus ferrugineus. World J. Microbiol. Biotechnol. 1995, 11, 618–620. [Google Scholar] [CrossRef] [PubMed]
- Bucher, G.E. Pathogens of tobacco and tomato hornworms. J. Invertebr. Pathol. 1967, 9, 82–89. [Google Scholar] [CrossRef]
- Bucher, G.E.; Stephens, J.M. A disease of grasshoppers caused by the bacterium Pseudomonas aeruginosa (Schroeter) Migula. Can. J. Microbiol. 1957, 3, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Andrejko, M.; Zdybicka-Barabas, A.; Cytryńska, M. Diverse effects of Galleria mellonella infection with entomopathogenic and clinical strains of Pseudomonas aeruginosa. J. Invertebr. Pathol. 2014, 115, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Lysenko, O. Non-sporeforming bacteria pathogenic to insects: Incidence and mechanisms. Annu. Rev. Microbiol. 1985, 39, 673–695. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Lu, B.; Tang, J.; Wang, C.; Li, H.; Yan, Y.; Zhan, L. The separation and identification and drug resistance analysis of the Pseudomonas Aeruginosa of Zophobas Morio. China Anim. Health 2012, 10, 012. [Google Scholar]
- Van Broekhoven, S.; Oonincx, D.G.A.B.; van Huis, A.; van Loon, J.J.A. Growth performance and feed conversion efficiency of three edible mealworm species (Coleoptera: Tenebrionidae) on diets composed of organic by-products. J. Insect Physiol. 2015, 73, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Muros, M.J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: A review. J. Clean. Prod. 2014, 65, 16–27. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information (NCBI). Available online: https://www.ncbi.nlm.nih.gov/ (accessed on 17 April 2018).
- Budzikiewicz, H. Siderophores of the Pseudomonadaceae sensu stricto (Fluorescent and Non-Fluorescent Pseudomonas spp.). Fortschr. Chem. Org. Naturst. 2004, 87, 81–237. [Google Scholar] [PubMed]
- Meyer, J.M.; Neely, A.; Stintzi, A.; Georges, C.; Holder, I.A. Pyoverdin is essential for virulence of Pseudomonas aeruginosa. Infect. Immun. 1996, 64, 518–523. [Google Scholar] [PubMed]
- Lau, G.W.; Hassett, D.J.; Ran, H.; Kong, F. The role of pyocyanin in Pseudomonas aeruginosa infection. Trends Mol. Med. 2004, 10, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Jander, G.; Rahme, L.G.; Ausubel, F.M. Positive correlation between virulence of Pseudomonas aeruginosa mutants in mice and insects. J. Bacteriol. 2000, 182, 3843–3845. [Google Scholar] [CrossRef] [PubMed]
- Chieda, Y.; Iiyama, K.; Yasunaga-Aoki, C.; Lee, J.M.; Kusakabe, T.; Shimizu, S. Pathogenicity of gacA mutant of Pseudomonas aeruginosa PA01 in the silkworm, Bombyx mori. FEMS Microbiol. Lett. 2005, 244, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, S.H.; Zuberi, R.I.; Hafiz, S. Occurrence of Pseudomonas aeruginosa (Schroeter) Migula as a pathogenic bacterium of the desert locust, Schistocerca gregaria (Forskål). J. Invertebr. Pathol. 1965, 7, 189–191. [Google Scholar] [CrossRef]
- Pimenta, A.L.; Di Martino, P.; Blight, M.A. Positive correlation between in vivo and in vitro assays for the evaluation of Pseudomonas virulence. Res. Microbiol. 2006, 157, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.W.; Britigan, B.E.; Hassett, D.J. Pseudomonas aeruginosa OxyR is required for full virulence in rodent and insect models of infection and for resistance to human neutrophils. Infect. Immun. 2005, 73, 2550–2553. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A.; Northfield, T.D.; Rosenheim, J.A. The epidemiology and evolution of parasite transmission through cannibalism. Ecology 2016, 97, 2003–2011. [Google Scholar] [CrossRef] [PubMed]
- Williams, T. Natural invertebrate hosts to iridoviruses (Iridoviridae). Neotrop. Entomol. 2008, 37, 615–632. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.L.; Mitchell, R.F.; Reagel, P.F.; Hanks, L.M. Causes and consequences of cannibalism in noncarnivorous insects. Annu. Rev. Entomol. 2010, 55, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Tschinkel, W.R.; Willaon, C.D. Inhibition of pupation due to crowding in some tenebrionid beetles. J. Exp. Zool. 1971, 176, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Tschinkel, W.R. Larval dispersal and cannibalism in a natural population of Zophobas atratus (Coleoptera: Tenebrionidae). Anim. Behav. 1981, 29, 990–996. [Google Scholar] [CrossRef]
- Adamo, S.A. Estimating disease resistance in insects: Phenoloxidase and lysozyme-like activity and disease resistance in the cricket Gryllus texensis. J. Insect Physiol. 2004, 50, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Piesk, M.; Karl, I.; Franke, K.; Fischer, K. High larval density does not induce a prophylactic immune response in a butterfly. Ecol. Entomol. 2013, 38, 346–354. [Google Scholar] [CrossRef]
- Jakubowska, A.K.; Jakubowska, A.K.; D’Angiolo, M.; González-Martínez, R.M.; Millán-Leiva, A.; Carballo, A.; Murillo, R.; Caballero, P.; Herrero, S. Simultaneous occurrence of covert infections with small RNA viruses in the lepidopteran Spodoptera exigua. J. Invertebr. Pathol. 2014, 121, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Jakubowska, A.K.; Murillo, R.; Carballo, A.; Williams, T.; van Lent, J.W.M.; Caballero, P.; Herrero, S. Iflavirus increases its infectivity and physical stability in association with baculovirus. PeerJ 2016, 4, e1687. [Google Scholar] [CrossRef] [PubMed]
- Kemp, E.M.; Woodward, D.T.; Cory, J.S. Detection of single and mixed covert baculovirus infections in eastern spruce budworm, Choristoneura fumiferana populations. J. Invertebr. Pathol. 2011, 107, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.B.; Watson, E.L.; Valverde-Garcia, P. Mixed infections and insect–pathogen interactions. Ecol. Lett. 2003, 6, 183–188. [Google Scholar] [CrossRef]
- Calleri, D.V.; McGrail, R.E.; Rosengaus, R.B.; Vargo, E.L.; Traniello, J.F.A. Inbreeding and disease resistance in a social insect: Effects of heterozygosity on immunocompetence in the termite Zootermopsis angusticollis. Proc. R. Soc. Lon. B 2006, 273, 2633–2640. [Google Scholar] [CrossRef] [PubMed]
- William, O.H.H.; Boomsma, J.J. Genetic Diversity and Disease Resistance in Leaf-Cutting Ant Societies. Evolution 2004, 58, 1251–1260. [Google Scholar]
- Miller, C.V.L.; Cotter, S.C.; Ardia, D. Resistance and tolerance: The role of nutrients on pathogen dynamics and infection outcomes in an insect host. J. Anim. Ecol. 2018, 87, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Krams, I.; Kecko, S.; Kangassalo, K.; Moore, F.R.; Jankevics, E.; Inashkina, I.; Krama, T.; Lietuvietis, V.; Meija, L.; Rantala, M.J. Effects of food quality on trade-offs among growth, immunity and survival in the greater wax moth Galleria mellonella. Insect Sci. 2015, 22, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Krieg, A. Chapter 11: Diseases caused by bacteria and other procaryotes. In Epizootiology of Insect Diseases; Fuxa, J.R., Tanada, Y., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 1987; pp. 323–356. [Google Scholar]
- Stephens, J.M. Chapter 9: Immunity in insects. In Insect Pathology, An Advanced Treatise, 1st ed.; Steinhaus, E., Ed.; Academic Press: New York, NY, USA, 1963; Volume 1, pp. 273–298. ISBN 9-78-032314352-3. [Google Scholar]
- Greany, P.D.; Allen, G.E.; Webb, J.C.; Sharp, J.L.; Chambers, D.L. Stress-induced septicemia as an impediment to laboratory rearing of the fruit fly parasitoid Biosteres (opius) longicaudatus (Hymenoptera: Braconidae) and the Caribbean fruit fly Anastrepha suspensa (Diptera: Tephritidae). J. Invertebr. Pathol. 1977, 29, 153–161. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).