Portland/Sulfoaluminate Cement Blends for the Control of Early Age Hydration and Yield Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization Methods
3. Results
3.1. Chemical Characterization
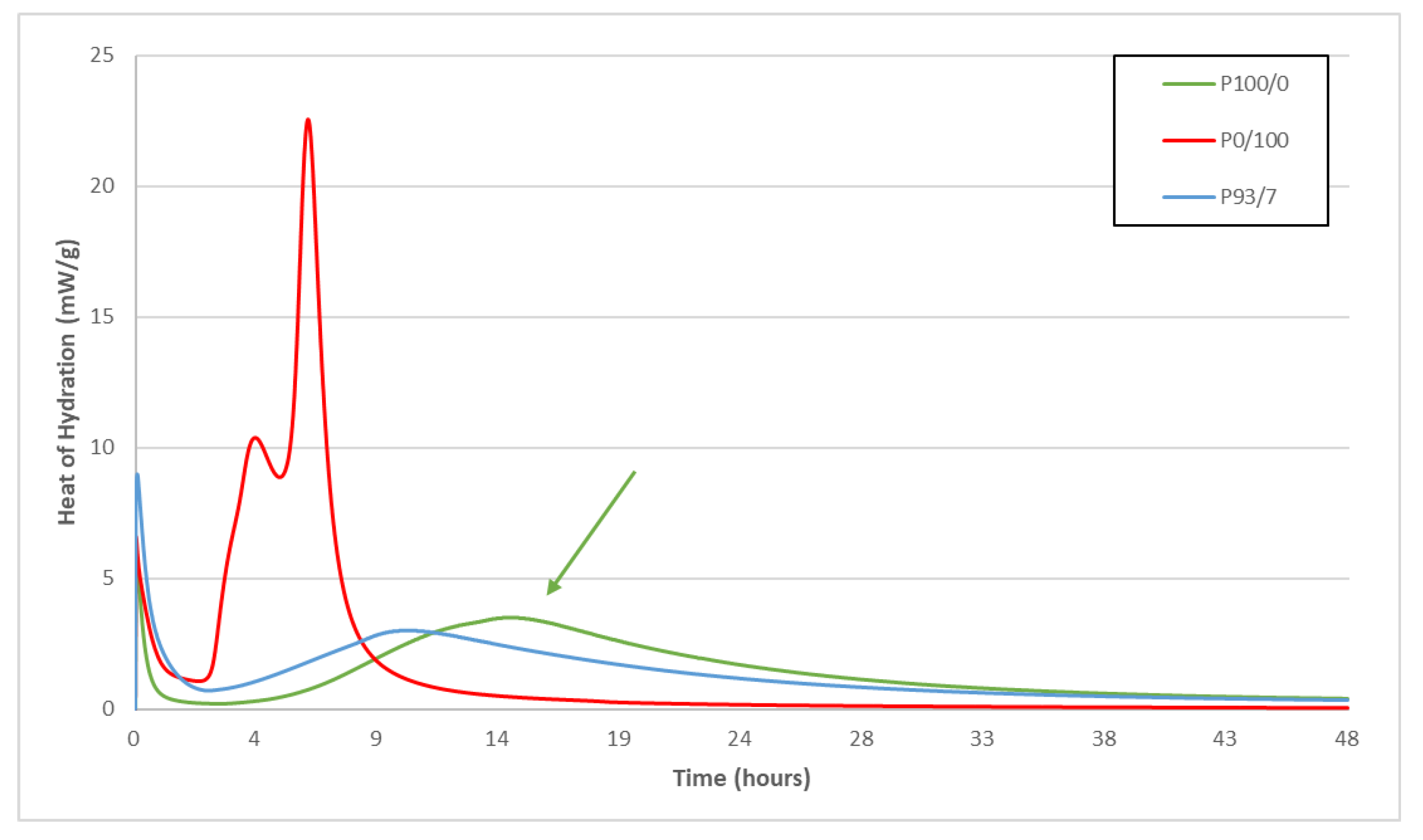
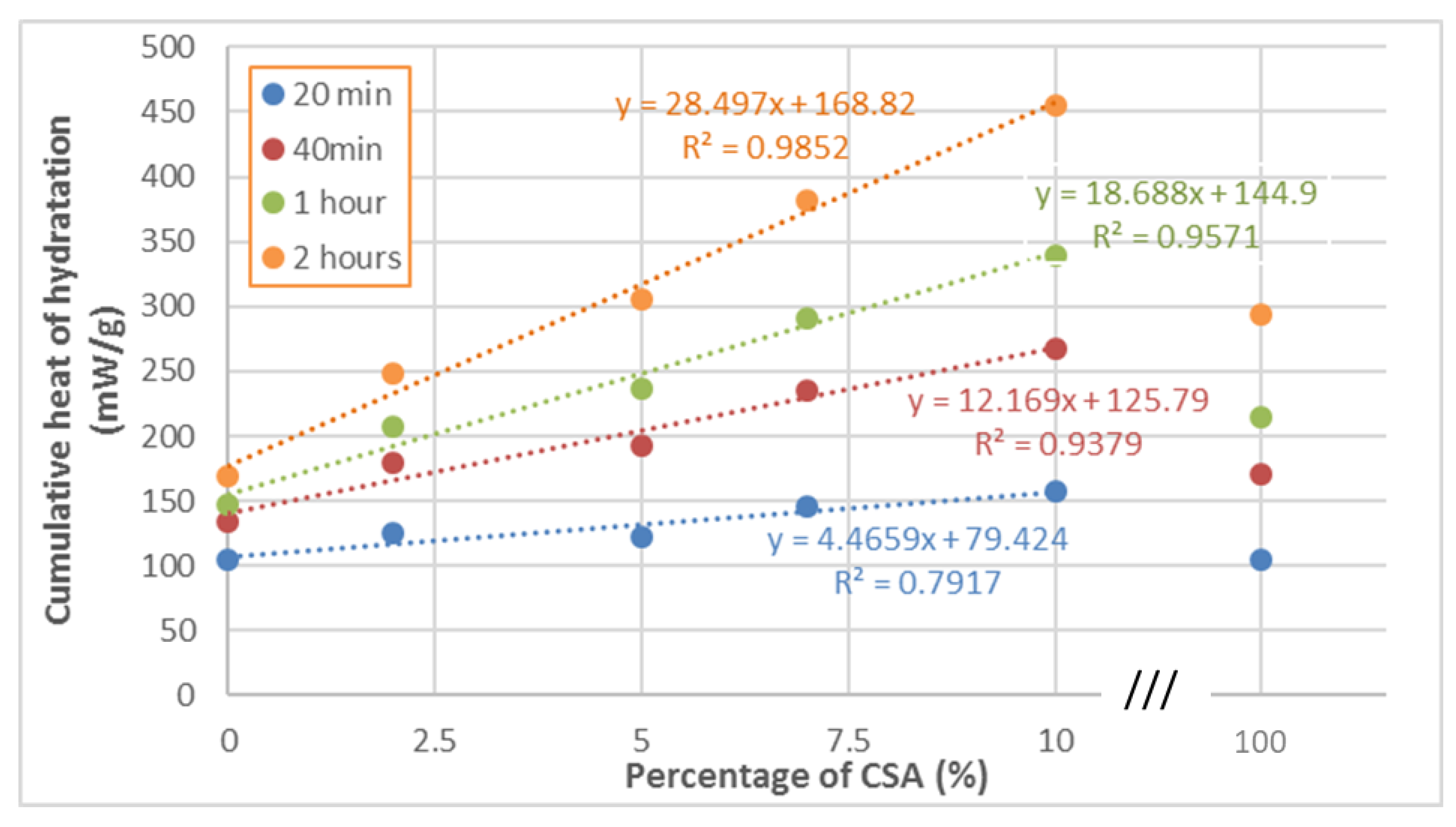
3.1.1. Reactivity of Blends
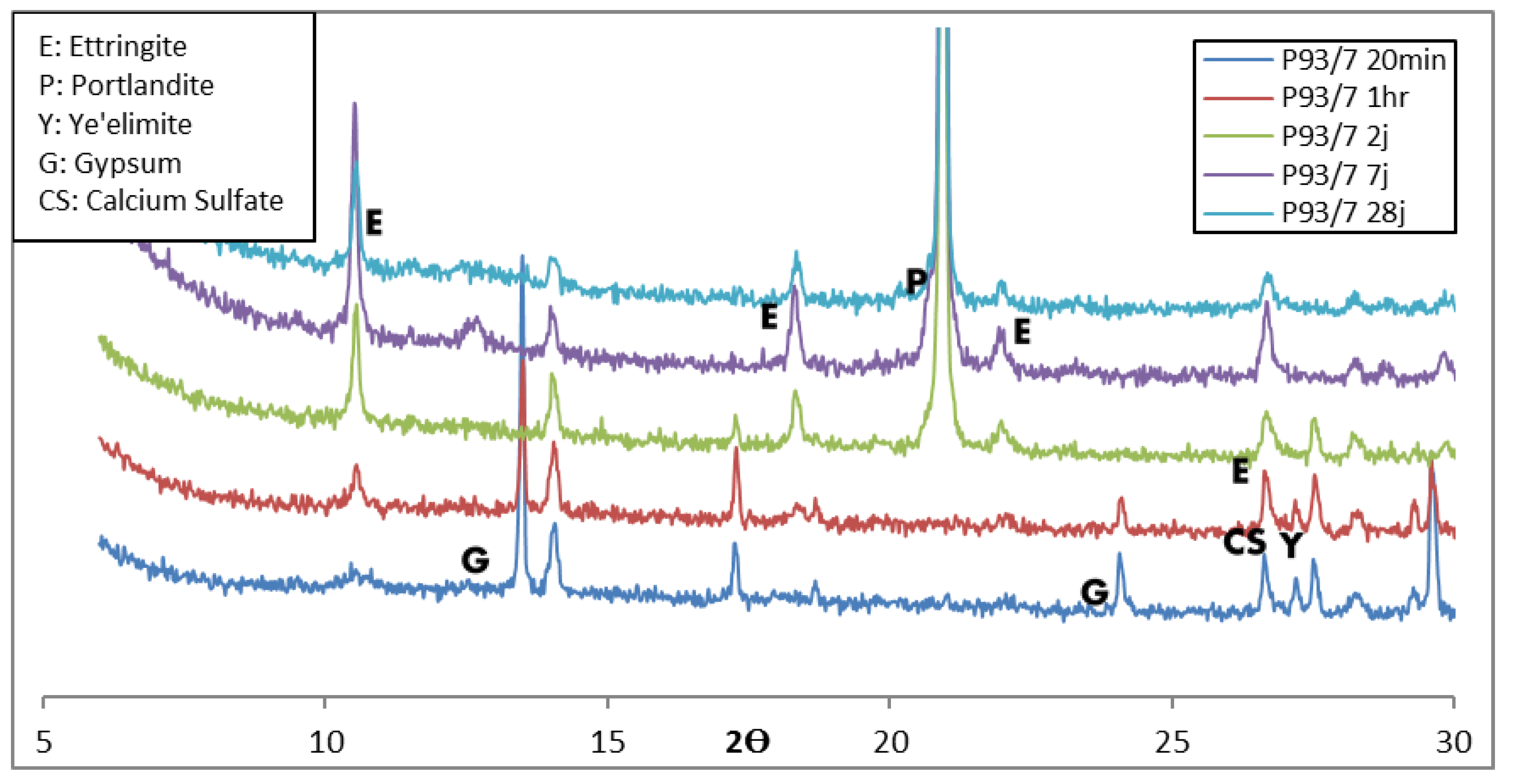
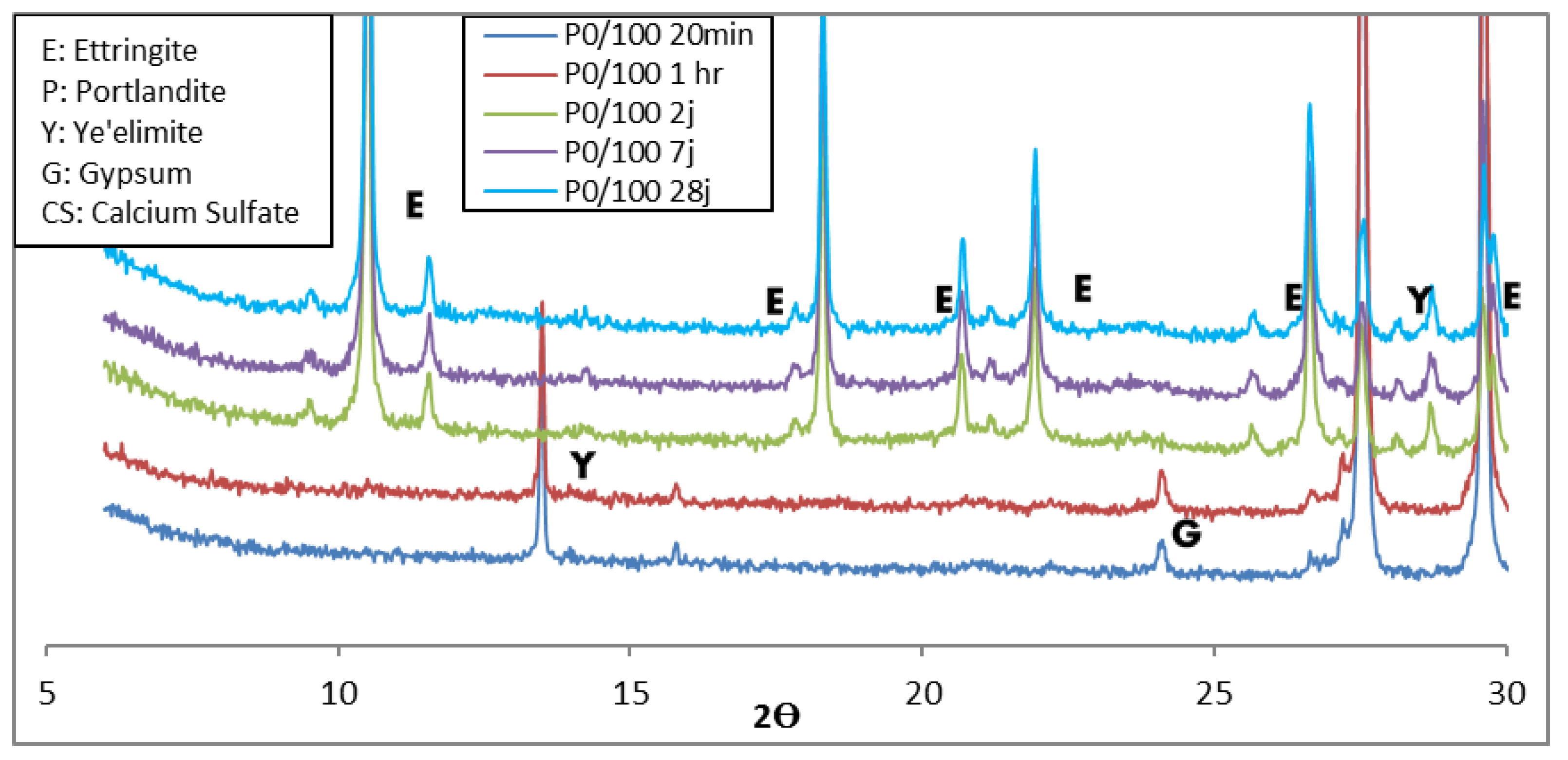
3.1.2. XRD
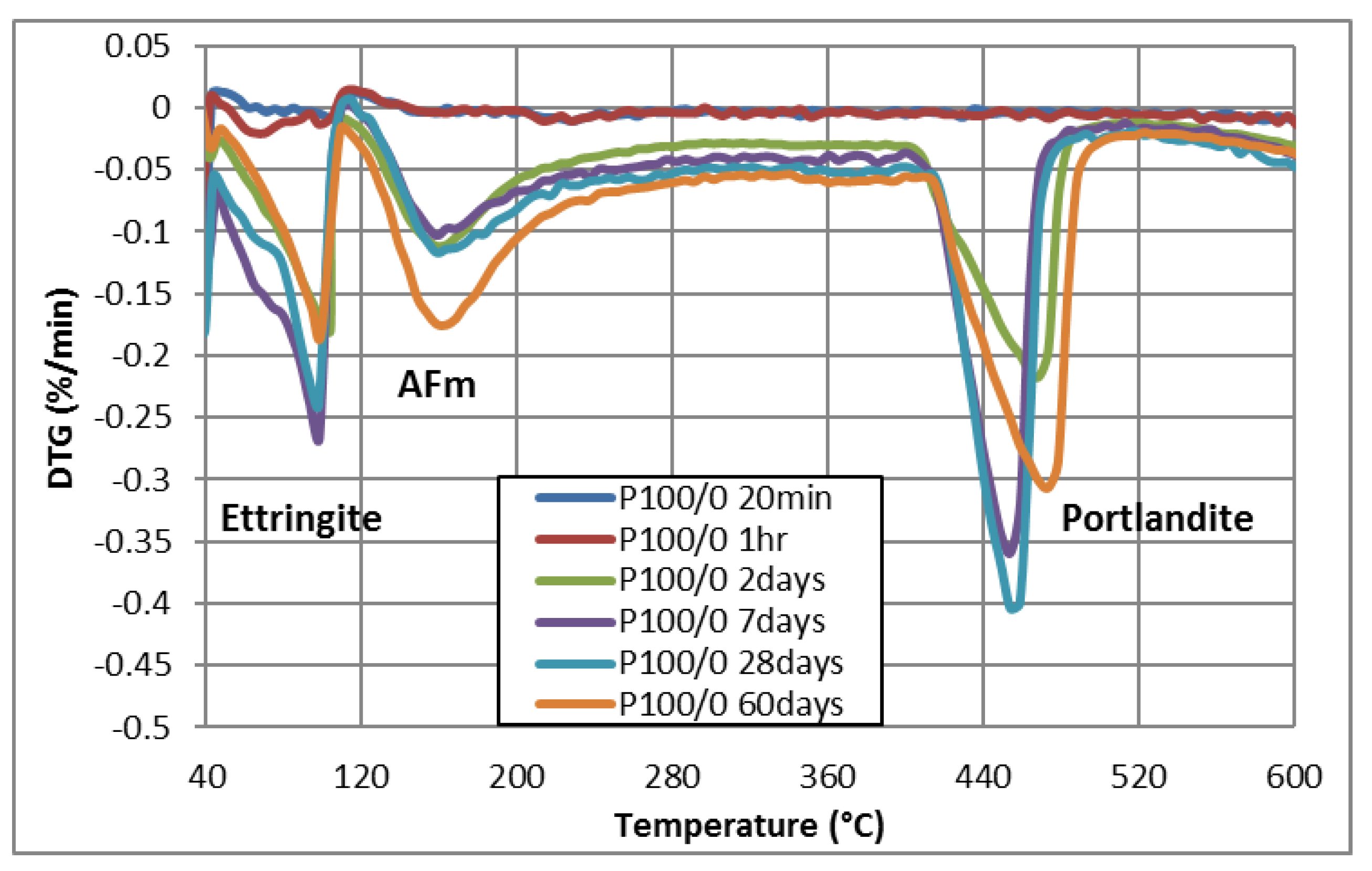
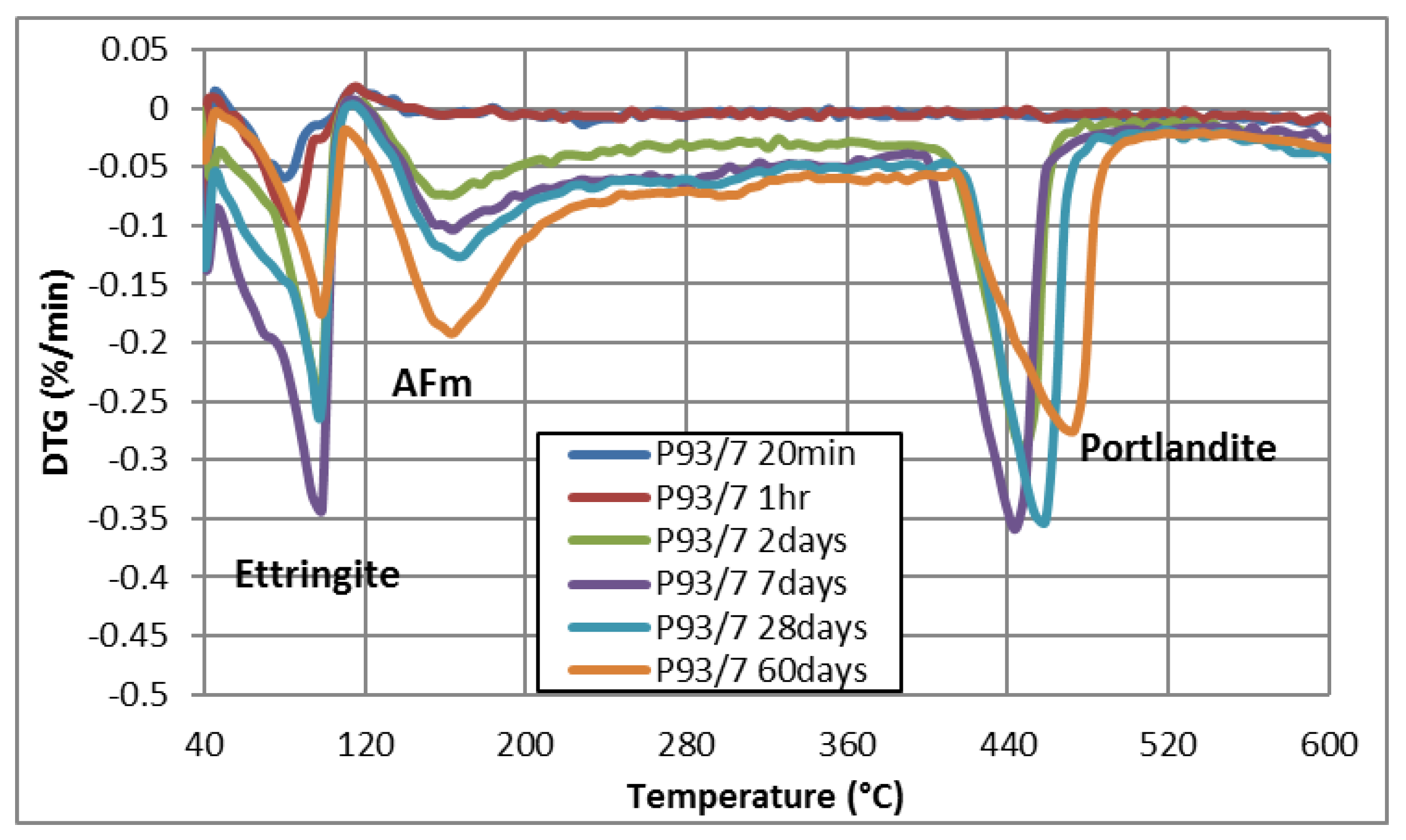
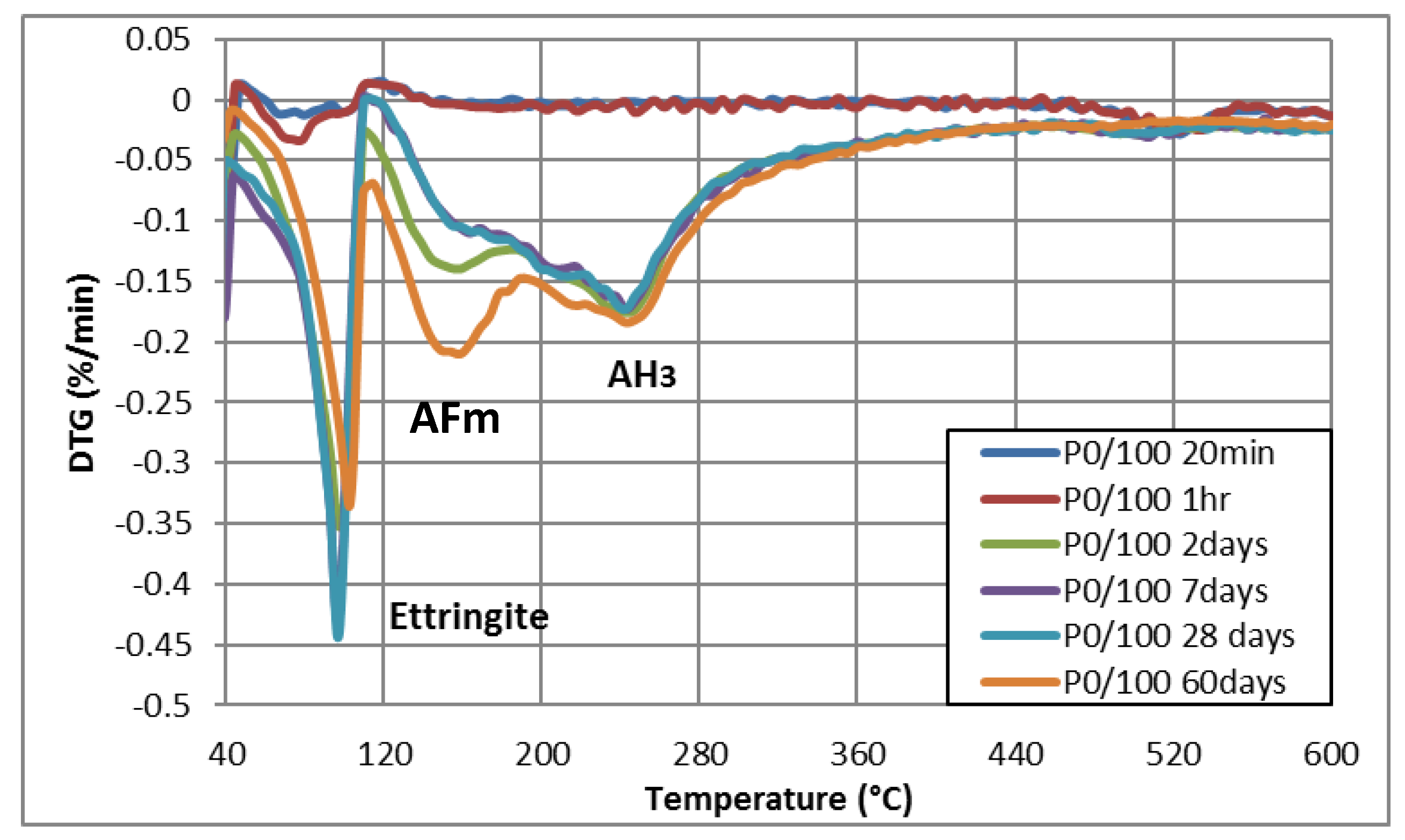
3.1.3. TGA-DTG
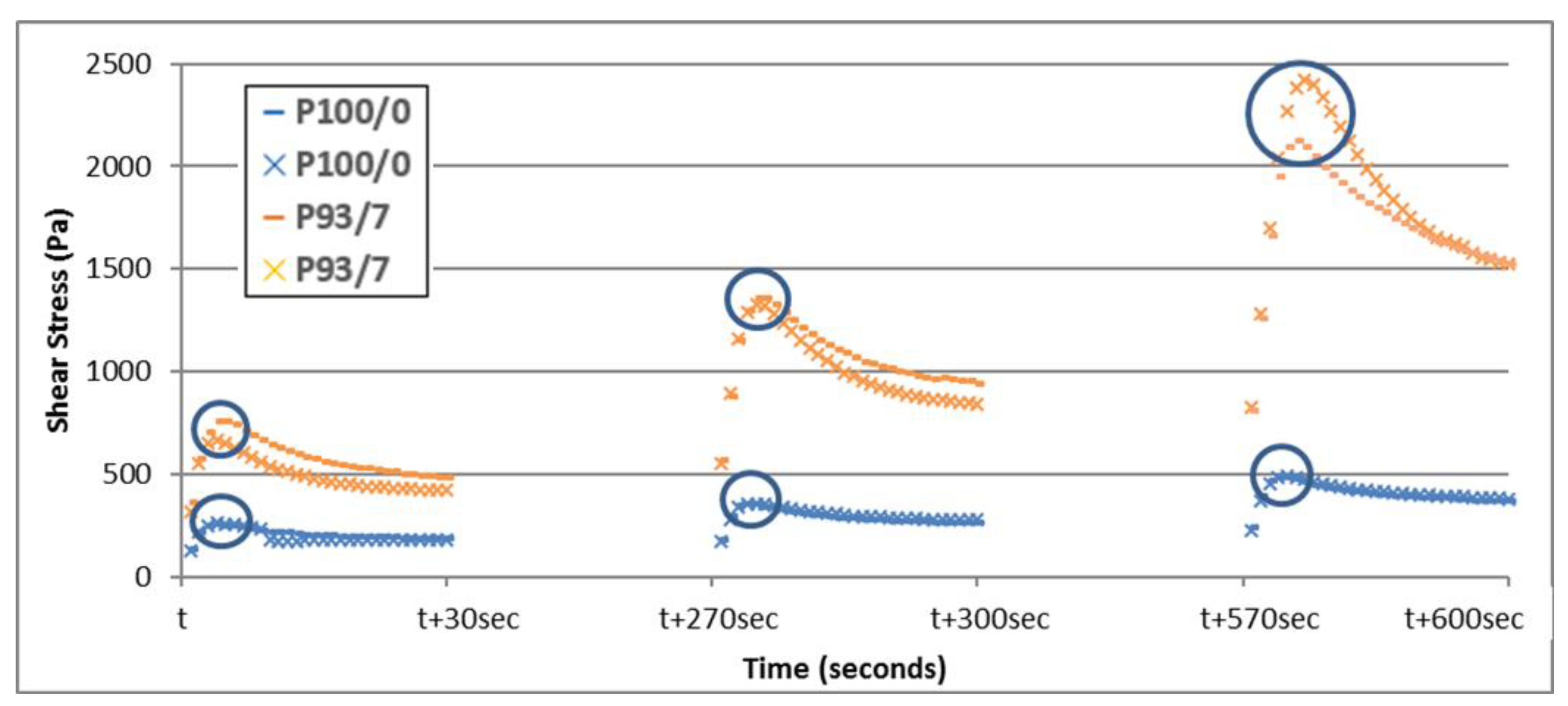
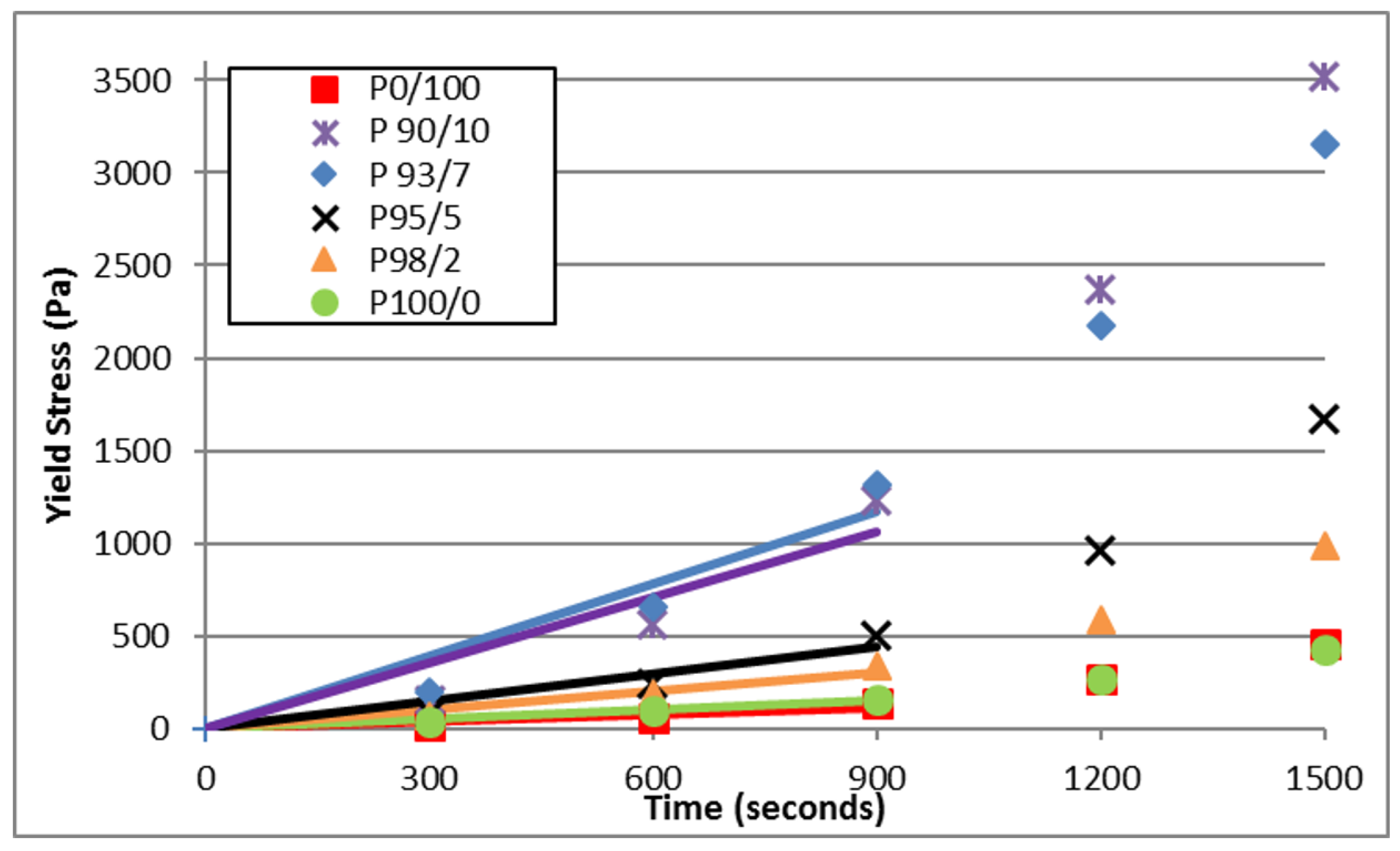
3.2. Yield Stress Evolution
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ludwig, H.-M.; Zhang, W. Research review of cement clinker chemistry. Cem. Concr. Res. 2015, 78, 24–37. [Google Scholar] [CrossRef]
- Péra, J.; Ambroise, J. New applications of calcium sulfoaluminate cement. Cem. Concr. Res. 2004, 34, 671–676. [Google Scholar] [CrossRef]
- Kleib, J.; Aouad, G.; Louis, G.; Zakhour, M.; Boulos, M.; Rousselet, A.; Bulteel, D. The use of calcium sulfoaluminate cement to mitigate the alkali silica reaction in mortars. Constr. Build. Mater. 2018, 184, 295–303. [Google Scholar] [CrossRef]
- Antoun, M.; Becquart, F.; Gerges, N.; Aouad, G. The use of calcium sulfo-aluminate cement as an alternative to Portland Cement for the recycling of municipal solid waste incineration bottom ash in mortar. Waste Manag. Res. 2020, 38, 868–875. [Google Scholar] [CrossRef]
- Zhou, Q.; Milestone, N.B.; Hayes, M. An alternative to Portland Cement for waste encapsulation—The calcium sul-foaluminate cement system. J. Hazard. Mater. 2006, 136, 120–129. [Google Scholar] [CrossRef]
- Coumes, C.C.D.; Courtois, S.; Peysson, S.; Ambroise, J.; Pera, J. Calcium sulfoaluminate cement blended with OPC: A potential binder to encapsulate low-level radioactive slurries of complex chemistry. Cem. Concr. Res. 2009, 39, 740–747. [Google Scholar] [CrossRef]
- Khalil, N.; Aouad, G.; El Cheikh, K.; Rémond, S. Use of calcium sulfoaluminate cements for setting control of 3D-printing mortars. Constr. Build. Mater. 2017, 157, 382–391. [Google Scholar] [CrossRef]
- Champenois, J.-B.; Coumes, C.C.D.; Poulesquen, A.; Le Bescop, P.; Damidot, D. Beneficial use of a cell coupling rheometry, conductimetry, and calorimetry to investigate the early age hydration of calcium sulfoaluminate cement. Rheol. Acta 2013, 52, 177–187. [Google Scholar] [CrossRef]
- García-Maté, M.; Santacruz, I.; Ángeles, G.; León-Reina, L.; Aranda, M.A. Rheological and hydration characteri-zation of calcium sulfoaluminate cement pastes. Cem. Concr. Compos. 2012, 34, 684–691. [Google Scholar] [CrossRef]
- Peysson, S.; Péra, J.; Chabannet, M. Immobilization of heavy metals by calcium sulfoaluminate cement. Cem. Concr. Res. 2005, 35, 2261–2270. [Google Scholar] [CrossRef]
- Kleib, J.; Aouad, G.; Khalil, N.; Zakhour, M. Incorporation of zinc in calcium sulfoaluminate cement clinker. Adv. Cem. Res. 2021, 33, 311–317. [Google Scholar] [CrossRef]
- Juenger, M.; Winnefeld, F.; Provis, J.; Ideker, J. Advances in alternative cementitious binders. Cem. Concr. Res. 2011, 41, 1232–1243. [Google Scholar] [CrossRef]
- Trauchessec, R.; Mechling, J.-M.; Lecomte, A.; Roux, A.; Le Rolland, B. Hydration of ordinary Portland cement and calcium sulfoaluminate cement blends. Cem. Concr. Compos. 2015, 56, 106–114. [Google Scholar] [CrossRef]
- Le Saoût, G.; Lothenbach, B.; Hori, A.; Higuchi, T.; Winnefeld, F. Hydration of Portland cement with additions of calcium sulfoaluminates. Cem. Concr. Res. 2013, 43, 81–94. [Google Scholar] [CrossRef]
- Perrot, A.; Pierre, A.; Vitaloni, S.; Picandet, V. Prediction of lateral form pressure exerted by concrete at low casting rates. Mater. Struct. 2014, 48, 2315–2322. [Google Scholar] [CrossRef]
- Sun, L.; Pang, X.; Yan, H. Hydration kinetics of oil well cement in the temperature range between 5 and 30 °C. Front. Mater. 2022, 9, 558. [Google Scholar] [CrossRef]
- Lura, P.; Winnefeld, F.; Klemm, S. Simultaneous measurements of heat of hydration and chemical shrinkage on hardening cement pastes. J. Therm. Anal. Calorim. 2009, 101, 925–932. [Google Scholar] [CrossRef]
- Winnefeld, F.; Lothenbach, B. Hydration of calcium sulfoaluminate cements—Experimental findings and thermodynamic modelling. Cem. Concr. Res. 2010, 40, 1239–1247. [Google Scholar] [CrossRef]
- Winnefeld, F.; Barlag, S. Calorimetric and thermogravimetric study on the influence of calcium sulfate on the hydration of ye’elimite. J. Therm. Anal. Calorim. 2009, 101, 949–957. [Google Scholar] [CrossRef]
- Telesca, A.; Marroccoli, M.; Pace, M.; Tomasulo, M.; Valenti, G.; Monteiro, P. A hydration study of various calcium sulfoaluminate cements. Cem. Concr. Compos. 2014, 53, 224–232. [Google Scholar] [CrossRef]
- Chaunsali, P.; Mondal, P. Physico-chemical interaction between mineral admixtures and OPC–calcium sulfoaluminate (CSA) cements and its influence on early-age expansion. Cem. Concr. Res. 2016, 80, 10–20. [Google Scholar] [CrossRef]
- Pourchet, S.; Regnaud, L.; Perez, J.; Nonat, A. Early C3A hydration in the presence of different kinds of calcium sulfate. Cem. Concr. Res. 2009, 39, 989–996. [Google Scholar] [CrossRef]
- Martin, L.H.; Winnefeld, F.; Müller, C.J.; Lothenbach, B. Contribution of limestone to the hydration of calcium sul-foaluminate cement. Cem. Concr. Compos. 2015, 62, 204–211. [Google Scholar] [CrossRef]
- Khalil, N.; Rémond, S.; Baz, B.; Aouad, G. Characterization of 3D Printing Mortars Made with OPC/CSA Mixes. In First RILEM International Conference on Concrete and Digital Fabrication—Digital Concrete 2018; Wangler, T., Flatt, R., Eds.; RILEM Bookseries; Springer: Cham, Switzerland, 2018; Volume 19. [Google Scholar] [CrossRef]
- Perrot, A.; Rangeard, D.; Pierre, A. Structural built-up of cement-based materials used for 3D-printing extrusion techniques. Mater. Struct. 2016, 49, 1213–1220. [Google Scholar] [CrossRef]
- Baz, B.; Remond, S.; Aouad, G. Influence of the mix composition on the thixotropy of 3D printable mortars. Mag. Concr. Res. 2022, 74, 271–283. [Google Scholar] [CrossRef]
- Roussel, N. Steady and transient flow behaviour of fresh cement pastes. Cem. Concr. Res. 2005, 35, 1656–1664. [Google Scholar] [CrossRef]
- Ovarlez, G.; Roussel, N. A Physical Model for the Prediction of Lateral Stress Exerted by Self-Compacting Concrete on Formwork. Mater. Struct. 2006, 39, 269–279. [Google Scholar] [CrossRef]
- Assaad, J.; Khayat, K.H. Assessment of Thixotropy of Self-Consolidating Concrete and Concrete-Equivalent-Mortar—Effect of Binder Composition and Content. ACI Mater. J. 2004, 101, 400–408. [Google Scholar]
- García-Maté, M.; De la Torre, A.G.; León-Reina, L.; Losilla, E.R.; Aranda, M.A.; Santacruz, I. Effect of calcium sulfate source on the hydration of calcium sulfoaluminate eco-cement. Cem. Concr. Compos. 2015, 55, 53–61. [Google Scholar] [CrossRef]
- Sahu, S.; Havlica, J.; Tomková, V.; Majling, J. Hydration behaviour of sulphoaluminate belite cement in the presence op various calcium sulphates. Thermochim. Acta 1991, 175, 45–52. [Google Scholar] [CrossRef]
- Péra, J.; Ambroise, J.; Holard, E.; Beauvent, G. Influence of the type of calciumsulfate on the properties of calcium sul-foaluminate cement. Int. Congr. Chem. Cem. Durb. South Afr. 2003, 3, 1129–1135. [Google Scholar]
- Prince, W.; Espagne, M.; Aïtcin, P.-C. Ettringite formation: A crucial step in cement superplasticizer compatibility. Cem. Concr. Res. 2003, 33, 635–641. [Google Scholar] [CrossRef]

| Composition | Na2O eq. | Fire Loss 950 °C | MgO | SiO2 | Al2O3 | Fe2O3 | CaO | SO3 | Cl− |
|---|---|---|---|---|---|---|---|---|---|
| OPC | 0.46 | 1.20 | 0.91 | 20.96 | 3.65 | 5.05 | 64.02 | 2.24 | 0.06 |
| CSA | 0.16 | 3.8 | nd | 8.16 | 18.22 | 7.64 | 43.60 | 15.24 | 0.05 |
| Composition | C3S | C4A3Š | C2S | C3MS2 | C3A | C3FT | C4AF | CŠ | Free Lime |
|---|---|---|---|---|---|---|---|---|---|
| OPC | 62.6 | -- | 16.5 | -- | 1.5 | -- | 13.1 | 3.8 | 0.5 |
| CSA | -- | 54.3 | 29.1 | 4.5 | -- | 9.3 | -- | 0.4 | 0.2 |
| Paste | Time | Ye’elimite | Gypsum | Calcium Sulfate (Anhydrite) | Ettringite | Portlandite |
|---|---|---|---|---|---|---|
| P100/0 | 20 min | _ | √ | _ | _ | _ |
| 1 h | _ | √ | _ | _ | _ | |
| 2 days | _ | _ | _ | √ | √ | |
| 7 days | _ | _ | _ | √ | √ | |
| 28 days | _ | _ | _ | √ | √ | |
| 60 days | _ | _ | _ | √ | √ | |
| P93/7 | 20 min | √ | √ | √ | √ | _ |
| 1 h | √ | √ | √ | √ | _ | |
| 2 days | √ | _ | _ | √ | √ | |
| 7 days | _ | _ | _ | √ | √ | |
| 28 days | _ | _ | _ | √ | √ | |
| 60 days | _ | _ | _ | √ | √ | |
| P0/100 | 20 min | √ | √ | √ | _ | _ |
| 1 h | √ | √ | √ | _ | _ | |
| 2 days | √ | _ | _ | √ | _ | |
| 7 days | √ | _ | _ | √ | _ | |
| 28 days | √ | _ | _ | √ | _ | |
| 60 days | √ | _ | _ | √ | _ |
| Composition | Weight Loss Percentage (%) | |||||
|---|---|---|---|---|---|---|
| 20 min | 1 h | |||||
| P100/0 | P93/7 | P0/100 | P100/0 | P93/7 | P0/100 | |
| AFt (40 °C–110 °C) | 0.861 | 1.356 | 0.930 | 0.977 | 1.960 | 1.199 |
| AFm (110 °C–250 °C) | 0.110 | 0.168 | 0.015 | 0.110 | 0.127 | 0.049 |
| CH (390 °C–500 °C) | 0.137 | 0.229 | 0.222 | 0.189 | 0.192 | 0.240 |
| AH3 (200 °C–350 °C) | 0.149 | 0.213 | ||||
| 2 days | 7 days | |||||
| P100/0 | P93/7 | P0/100 | P100/0 | P93/7 | P0/100 | |
| AFt (40 °C–110 °C) | 4.706 | 5.607 | 12.593 | 7.821 | 9.171 | 15.896 |
| AFm (110 °C–250 °C) | 2.689 | 2.029 | 3.142 | 2.843 | 3.038 | 2.484 |
| CH (390 °C–500 °C) | 3.739 | 3.316 | 0.939 | 4.516 | 4.636 | 0.911 |
| AH3 (200 °C–350 °C) | 5.069 | 5.005 | ||||
| 28 days | 60 days | |||||
| P100/0 | P93/7 | P0/100 | P100/0 | P93/7 | P0/100 | |
| AFt (40 °C–110 °C) | 7.336 | 7.815 | 15.258 | 5.280 | 5.241 | 3.468 |
| AFm (110 °C–250 °C) | 3.287 | 3.501 | 2.552 | 4.578 | 4.906 | 13.386 |
| CH (390 °C–500 °C) | 5.183 | 4.531 | 0.945 | 5.529 | 4.984 | 0.920 |
| AH3 (200 °C–350 °C) | 5.079 | 6.086 | ||||
| Cement Paste | Athix (Pa/s) | R2 |
|---|---|---|
| P0/100 | 0.128 | 0.8202 |
| P90/10 | 1.183 | 0.848 |
| P93/7 | 1.302 | 0.883 |
| P95/5 | 0.496 | 0.876 |
| P98/2 | 0.341 | 0.935 |
| P100/0 | 0.167 | 0.964 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, N.; Aouad, G.; Kleib, J.; Rémond, S. Portland/Sulfoaluminate Cement Blends for the Control of Early Age Hydration and Yield Stress. Buildings 2023, 13, 409. https://doi.org/10.3390/buildings13020409
Khalil N, Aouad G, Kleib J, Rémond S. Portland/Sulfoaluminate Cement Blends for the Control of Early Age Hydration and Yield Stress. Buildings. 2023; 13(2):409. https://doi.org/10.3390/buildings13020409
Chicago/Turabian StyleKhalil, Noura, Georges Aouad, Joelle Kleib, and Sebastien Rémond. 2023. "Portland/Sulfoaluminate Cement Blends for the Control of Early Age Hydration and Yield Stress" Buildings 13, no. 2: 409. https://doi.org/10.3390/buildings13020409
APA StyleKhalil, N., Aouad, G., Kleib, J., & Rémond, S. (2023). Portland/Sulfoaluminate Cement Blends for the Control of Early Age Hydration and Yield Stress. Buildings, 13(2), 409. https://doi.org/10.3390/buildings13020409







