An Integrated Process for the Xylitol and Ethanol Production from Oil Palm Empty Fruit Bunch (OPEFB) Using Debaryomyces hansenii and Saccharomyces cerevisiae
Abstract
1. Introduction
2. Materials and Methods
2.1. Tools and Materials
2.2. Autohydrolysis OPEFB Pretreatment
2.3. Hemicellulose Hydrolysis
2.4. Fermentation of OPEFB Hydrolyzate by D. hansenii
2.5. Semi-Simultaneous Saccharification and Fermentation (Semi-SSF) of Ethanol
2.6. Analysis Method
2.7. Data Interpretation
2.8. Statistical Analysis
3. Results
3.1. Characterization of Used Oil Palm Fruit Empty Bunches (OPEFB)
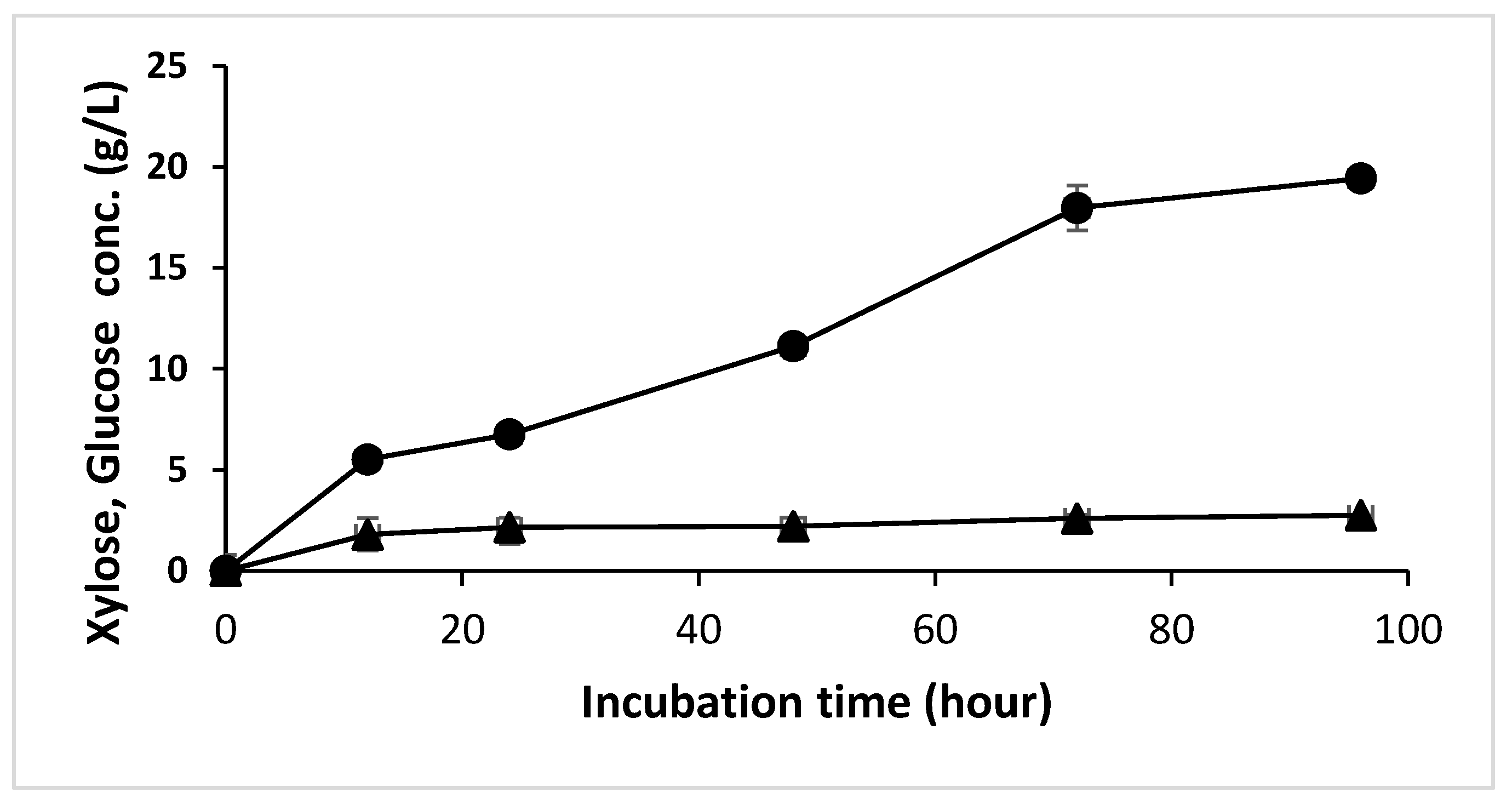
3.2. OPEFB Hydrolysis using Xylanase
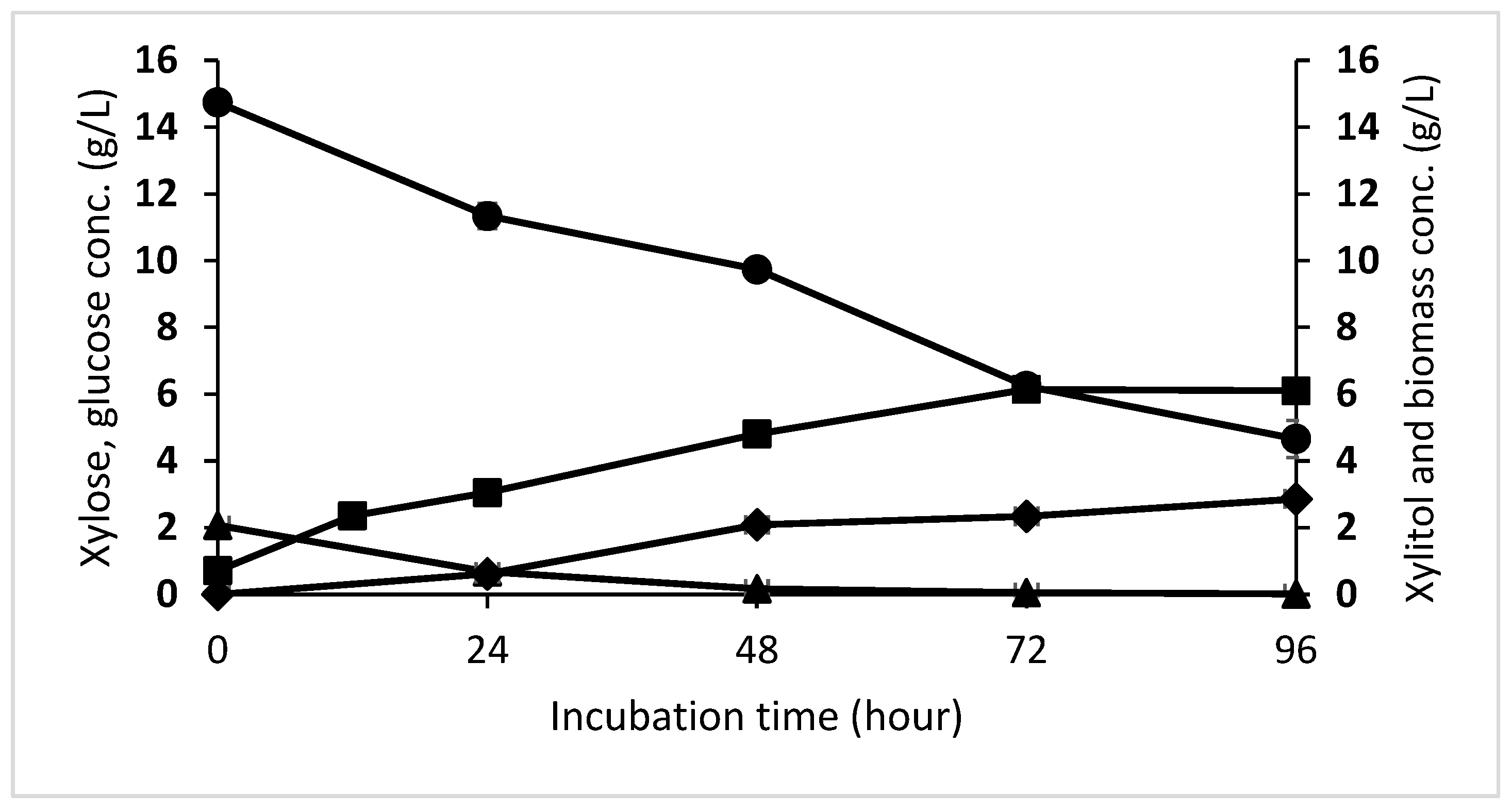
3.3. Growth and Xylitol Fermentation Profile of D. hansenii
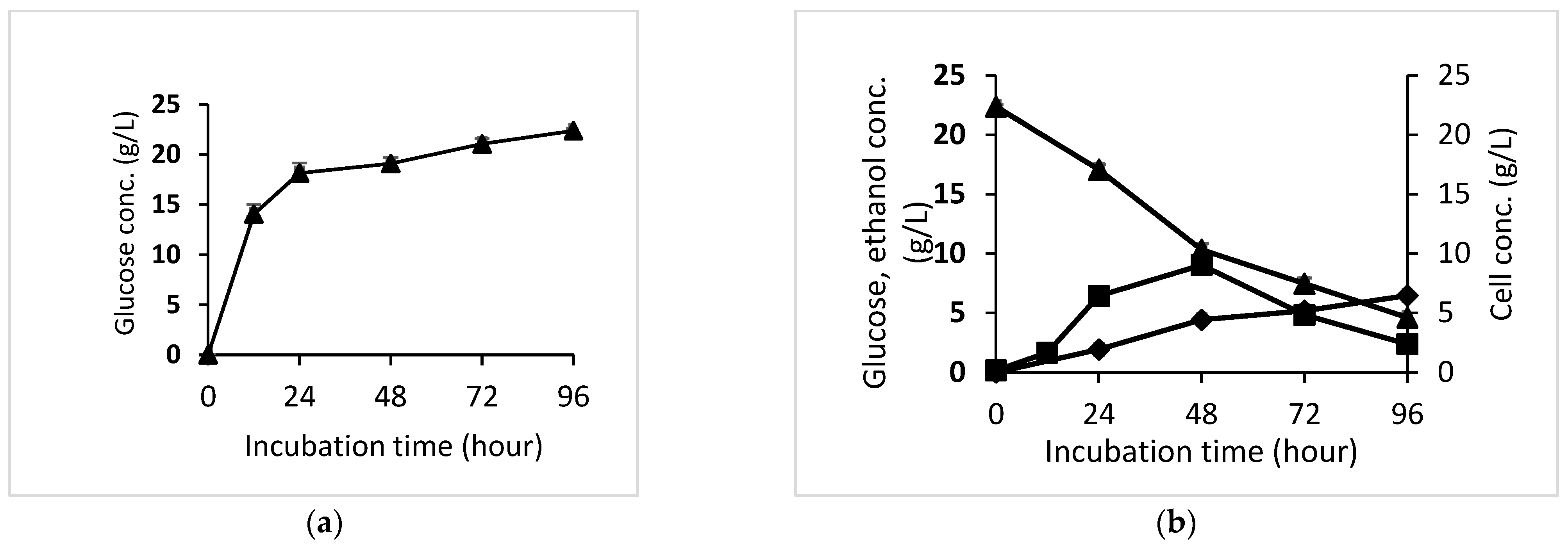
3.4. Bioethanol Production by Semi-SSF Process using S. cerevisiae
3.5. Morphological Changes Occurred in OPEFB during the Reactions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Indriyadi, W. Palm Oil Plantation in Indonesia: A Question of Sustainability. Salus Cult. J. Pembang. Mns. Kebud. 2022, 2, 1–10. [Google Scholar] [CrossRef]
- Kresnowati, M.T.A.P.; Setiadi, T.; Tantra, T.M.; Rusdi, D. Microbial Production of Xylitol from Oil Palm Empty Fruit Bunch Hydrolysate: Effects of Inoculum and PH. J. Eng. Technol. Sci. 2016, 48, 523–533. [Google Scholar] [CrossRef][Green Version]
- Josefa, K.M.; Alejandro, B.; Juan, P.A.; Isabel, O.; Andres, V.; Mario, A. Biotechnological Production of Xylitol from Oil Palm Empty Fruit Bunches Hydrolysate. Adv. J. Food Sci. Technol. 2018, 16, 134–137. [Google Scholar] [CrossRef]
- Omar, R.; Idris, A.; Yunus, R.; Khalid, K.; Aida Isma, M.I. Characterization of Empty Fruit Bunch for Microwave-Assisted Pyrolysis. Fuel 2011, 90, 1536–1544. [Google Scholar] [CrossRef]
- Ngadi, N.; Lani, N.S. Jurnal Teknologi Full Paper Extraction and Characterization of Cellulose from Empty Fruit Bunch (EFB). J. Teknol. 2014, 68, 35–39. [Google Scholar] [CrossRef]
- Mardawati, E.; Kresnowati, M.T.A.P.; Purwadi, R.; Bindar, Y.; Setiadi, T. Fungal Production of Xylanase from Oil Palm Empty Fruit Bunches via Solid State Cultivation. Int. J. Adv. Sci. Eng. Inf. Technol. 2018, 8, 2539–2546. [Google Scholar] [CrossRef]
- Suhartini, S.; Rohma, N.A.; Mardawati, E.; Kasbawati; Hidayat, N.; Melville, L. Biorefining of Oil Palm Empty Fruit Bunches for Bioethanol and Xylitol Production in Indonesia: A Review. Renew. Sustain. Energy Rev. 2022, 154, 111817. [Google Scholar] [CrossRef]
- Pant, S.; Ritika; Prakash, A.; Kuila, A. Integrated Production of Ethanol and Xylitol from Brassica juncea using Candida sojae JCM 1644. Bioresour. Technol. 2022, 351, 126903. [Google Scholar] [CrossRef]
- Mardawati, E.; Mandra Harahap, B.; Ayu Febrianti, E.; Try Hartono, A.; Putri Siahaan, N.; Wulandari, A.; Yudiastuti, S.; Suhartini, S.; Kasbawati, K. Integrated and Partial Process of Xylitol and Bioethanol Production from Oil Palm Empty Fruit Bunches. Adv. Food Sci. Sustain. Agric. Agroind. Eng. 2022, 5, 49–67. [Google Scholar] [CrossRef]
- Law, K.N.; Daud, W.R.W.; Ghazali, A. Morphological and Chemical Nature of Fiber Strands of Oil Palm Empty-Fruit-Bunch (OPEFB). BioResources 2007, 2, 351–362. [Google Scholar]
- Sudiyani, Y.; Hermiati, E. Utilization of Oil Palm Empty Fruit Bunch (OPEFB) for Bioethanol Production Through Alkali and Dilute Acid Pretreatment and Simultaneous Saccharification and Fermentation. Indones. J. Chem. 2010, 10, 261–267. [Google Scholar] [CrossRef]
- Gírio, F.M.; Fonseca, C.; Carvalheiro, F.; Duarte, L.C.; Marques, S.; Bogel-Łukasik, R. Hemicelluloses for Fuel Ethanol: A Review. Bioresour. Technol. 2010, 101, 4775–4800. [Google Scholar] [CrossRef] [PubMed]
- Laca, A.; Laca, A.; Díaz, M. Hydrolysis: From Cellulose and Hemicellulose to Simple Sugars. In Second and Third Generation of Feedstocks: The Evolution of Biofuels; Basile, A., Dalena, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 1, pp. 213–240. [Google Scholar] [CrossRef]
- Kresnowati, M.; Mardawati, E.; Setiadi, T. Production of Xylitol from Oil Palm Empty Friuts Bunch: A Case Study on Bioefinery Concept. Mod. Appl. Sci. 2015, 9, 206. [Google Scholar] [CrossRef]
- Lenhart, A.; Chey, W.D. A Systematic Review of the Effects of Polyols on Gastrointestinal Health and Irritable Bowel Syndrome. Adv. Nutr. 2017, 8, 587–596. [Google Scholar] [CrossRef]
- Dasgupta, D.; Bandhu, S.; Adhikari, D.K.; Ghosh, D. Challenges and Prospects of Xylitol Production with Whole Cell Bio-Catalysis: A Review. Microbiol. Res. 2017, 197, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Felipe Hernández-Pérez, A.; de Arruda, P.V.; Sene, L.; da Silva, S.S.; Kumar Chandel, A.; de Almeida Felipe, M.d.G. Xylitol Bioproduction: State-of-the-Art, Industrial Paradigm Shift, and Opportunities for Integrated Biorefineries. Crit. Rev. Biotechnol. 2019, 39, 924–943. [Google Scholar] [CrossRef]
- Yoshitake, J.; Ishizaki, H.; Shimamura, M.; Imai, T. Xylitol Production by an Enterobacter Species. Agric. Biol. Chem. 1973, 37, 2261–2267. [Google Scholar] [CrossRef][Green Version]
- Kang, T.Z.; Mohammad, S.H.; Murad, A.M.A.; Illias, R.M.; Jahim, J.M. Fermentative Production of Xylitol: A First Trial on Xylose Bifurcation. Indian J. Sci. Technol. 2016, 9, 21. [Google Scholar] [CrossRef]
- de Albuquerque, T.L.; Gomes, S.D.L.; Marques, J.E.; da Silva, I.J.; Rocha, M.V.P. Xylitol Production from Cashew Apple Bagasse by Kluyveromyces marxianus CCA510. Catal. Today 2015, 255, 33–40. [Google Scholar] [CrossRef]
- Roseiro, J.C.; Peito, M.A.; Gírio, F.M.; Amaral-Collaço, M.T. The Effects of the Oxygen Transfer Coefficient and Substrate Concentration on the Xylose Fermentation by Debaryomyces hansenii. Arch. Microbiol. 1991, 156, 484–490. [Google Scholar] [CrossRef]
- Parajó, J.C.; Domínguez, H.; Domínguez, J.M. Biotechnological Production of Xylitol. Part 2: Operation in Culture Media Made with Commercial Sugars. Bioresour. Technol. 1998, 65, 203–212. [Google Scholar] [CrossRef]
- Mardawati, E.; Putri, A.V.; Yuliana, T.; Rahimah, S.; Nurjanah, S.; Hanidah, I. Effects of Substrate Concentration on Bioethanol Production from Oil Palm Empty Fruit Bunches with Simultaneous Saccharification and Fermentation (SSF). IOP Conf. Ser. Earth Environ. Sci. 2019, 230, 012079. [Google Scholar] [CrossRef]
- Sukhang, S.; Choojit, S.; Reungpeerakul, T.; Sangwichien, C. Bioethanol Production from Oil Palm Empty Fruit Bunch with SSF and SHF Processes Using Kluyveromyces marxianus Yeast. Cellulose 2020, 27, 301–314. [Google Scholar] [CrossRef]
- Harahap, A.F.P.; Panjaitan, J.R.H.; Curie, C.A.; Ramadhan, M.Y.A.; Srinophakun, P.; Gozan, M. Techno-Economic Evaluation of Hand Sanitiser Production Using Oil Palm Empty Fruit Bunch-Based Bioethanol by Simultaneous Saccharification and Fermentation (SSF) Process. Appl. Sci. 2020, 10, 5987. [Google Scholar] [CrossRef]
- Tesfaw, A.; Assefa, F. Current Trends in Bioethanol Production by Saccharomyces cerevisiae: Substrate, Inhibitor Reduction, Growth Variables, Coculture, and Immobilization. Int. Sch. Res. Not. 2014, 2014, 532852. [Google Scholar] [CrossRef]
- Unrean, P.; Ketsub, N. Integrated Lignocellulosic Bioprocess for Co-Production of Ethanol and Xylitol from Sugarcane Bagasse. Ind. Crops Prod. 2018, 123, 238–246. [Google Scholar] [CrossRef]
- Dahnum, D.; Tasum, S.O.; Triwahyuni, E.; Nurdin, M.; Abimanyu, H. Comparison of SHF and SSF Processes Using Enzyme and Dry Yeast for Optimization of Bioethanol Production from Empty Fruit Bunch. Energy Procedia 2015, 68, 107–116. [Google Scholar] [CrossRef]
- Liu, Z.H.; Chen, H.Z. Simultaneous Saccharification and Co-Fermentation for Improving the Xylose Utilization of Steam Exploded Corn Stover at High Solid Loading. Bioresour. Technol. 2016, 201, 15–26. [Google Scholar] [CrossRef]
- Saha, B.C.; Nichols, N.N.; Qureshi, N.; Cotta, M.A. Comparison of Separate Hydrolysis and Fermentation and Simultaneous Saccharification and Fermentation Processes for Ethanol Production from Wheat Straw by Recombinant Escherichia coli Strain FBR5. Appl. Microbiol. Biotechnol. 2011, 92, 865–874. [Google Scholar] [CrossRef]
- Cheng, K.K.; Zhang, J.A.; Chavez, E.; Li, J.P. Integrated Production of Xylitol and Ethanol Using Corncob. Appl. Microbiol. Biotechnol. 2010, 87, 411–417. [Google Scholar] [CrossRef]
- Xavier, F.D.; Bezerra, G.S.; Santos, S.F.M.; Oliveira, L.S.C.; Silva, F.L.H.; Silva, A.J.O.; Conceição, M.M. Evaluation of the Simultaneous Production of Xylitol and Ethanol from Sisal Fiber. Biomolecules 2018, 8, 2. [Google Scholar] [CrossRef]
- Shankar, K.; Kulkarni, N.S.; Sajjanshetty, R.; Jayalakshmi, S.K.; Sreeramulu, K. Co-Production of Xylitol and Ethanol by the Fermentation of the Lignocellulosic Hydrolysates of Banana and Water Hyacinth Leaves by Individual Yeast Strains. Ind. Crops Prod. 2020, 155, 112809. [Google Scholar] [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Tamburini, E.; Costa, S.; Marchetti, M.G.; Pedrini, P. Optimized Production of Xylitol from Xylose Using a Hyper-Acidophilic Candida tropicalis. Biomolecules 2015, 5, 1979–1989. [Google Scholar] [CrossRef]
- Krishnan, M.S.; Ho, N.W.Y.; Tsao, G.T. Fermentation Kinetics of Ethanol Production from Glucose and Xylose by Recombinant Saccharomyces 1400(PLNH33). Appl. Biochem. Biotechnol.—Part A Enzym. Eng. Biotechnol. 1999, 77–79, 373–388. [Google Scholar] [CrossRef]
- Maulana Hidayatullah, I.; Setiadi, T.; Kresnowati, M.T.A.P.; Boopathy, R. Xylanase Inhibition by the Derivatives of Lignocellulosic Material. Bioresour. Technol. 2020, 300, 122740. [Google Scholar] [CrossRef]
- Mardawati, E.; Werner, A.; Bley, T.; Kresnowati, M.T.A.P.; Setiadi, T. The Enzymatic Hydrolysis of Oil Palm Empty Fruit Bunches to Xylose. J. Japan Inst. Energy 2014, 93, 973–978. [Google Scholar] [CrossRef]
- Qin, L.; Zhao, X.; Li, W.C.; Zhu, J.Q.; Liu, L.; Li, B.Z.; Yuan, Y.J. Process Analysis and Optimization of Simultaneous Saccharification and Co-Fermentation of Ethylenediamine-Pretreated Corn Stover for Ethanol Production. Biotechnol. Biofuels 2018, 11, 118. [Google Scholar] [CrossRef]
- Isroi; Ishola, M.M.; Millati, R.; Syamsiah, S.; Cahyanto, M.N.; Niklasson, C.; Taherzadeh, M.J. Structural Changes of Oil Palm Empty Fruit Bunch (OPEFB) after Fungal and Phosphoric Acid Pretreatment. Molecules 2012, 17, 14995–15012. [Google Scholar] [CrossRef]
- Nieves, D.C.; Karimi, K.; Horváth, I.S. Improvement of Biogas Production from Oil Palm Empty Fruit Bunches (OPEFB). Ind. Crops Prod. 2011, 34, 1097–1101. [Google Scholar] [CrossRef]
- Siti Sabrina, S.; Roshanida, A.R.; Norzita, N. Pretreatment of Oil Palm Fronds for Improving Hemicelluloses Content for Higher Recovery of Xylose. J. Teknol. Sci. Eng. 2013, 62, 39–42. [Google Scholar] [CrossRef]
- Harahap, B.M. Degradation Techniques of Hemicellulose Fraction from Biomass Feedstock for Optimum Xylose Production: A Review. J. Keteknikan Pertan. Trop. Biosist. 2020, 8, 107–124. [Google Scholar] [CrossRef]
- Mardawati, E.; Maharani, N.; Wira, D.W.; Harahap, B.M.; Yuliana, T.; Sukarminah, E. Xylitol Production from Oil Palm Empty Fruit Bunches (OPEFB) Via Simultaneous Enzymatic Hydrolysis and Fermentation Process. J. Ind. Inf. Technol. Agric. 2020, 2, 29–36. [Google Scholar] [CrossRef]
- Branco, R.F.; Santos, J.C.; Murakami, L.Y.; Mussatto, S.I.; Dragone, G.; Silva, S.S. Xylitol Production in a Bubble Column Bioreactor: Influence of the Aeration Rate and Immobilized System Concentration. Process Biochem. 2007, 42, 258–262. [Google Scholar] [CrossRef]
- Narisetty, V.; Castro, E.; Durgapal, S.; Coulon, F.; Jacob, S.; Kumar, D.; Kumar Awasthi, M.; Kishore Pant, K.; Parameswaran, B.; Kumar, V. High Level Xylitol Production by Pichia Fermentans Using Non-Detoxified Xylose-Rich Sugarcane Bagasse and Olive Pits Hydrolysates. Bioresour. Technol. 2021, 342, 126005. [Google Scholar] [CrossRef] [PubMed]
- Mardawati, E.; Wira, D.W.; Kresnowati, M.; Purwadi, R.; Setiadi, T. Microbial Production of Xylitol from Oil Palm Empty Fruit Bunches Hydrolysate: The Effect of Glucose Concentration. J. Japan Inst. Energy 2015, 94, 769–774. [Google Scholar] [CrossRef]
- Zhang, Q.; Wu, D.; Lin, Y.; Wang, X.; Kong, H.; Tanaka, S. Substrate and Product Inhibition on Yeast Performance in Ethanol Fermentation. Energy and Fuels 2015, 29, 1019–1027. [Google Scholar] [CrossRef]
- Apweiler, E.; Sameith, K.; Margaritis, T.; Brabers, N.; van de Pasch, L.; Bakker, L.V.; van Leenen, D.; Holstege, F.C.P.; Kemmeren, P. Yeast Glucose Pathways Converge on the Transcriptional Regulation of Trehalose Biosynthesis. BMC Genomics 2012, 13, 239. [Google Scholar] [CrossRef]
- Gibson, B.R.; Lawrence, S.J.; Leclaire, J.P.R.; Powell, C.D.; Smart, K.A. Yeast Responses to Stresses Associated with Industrial Brewery Handling. FEMS Microbiol. 2007, 31, 535–569. [Google Scholar] [CrossRef]
- Zhao, X.Q.; Bai, F.W. Mechanisms of Yeast Stress Tolerance and Its Manipulation for Efficient Fuel Ethanol Production. J. Biotechnol. 2009, 144, 23–30. [Google Scholar] [CrossRef]
- Wyman, C.E.; Dale, B.E.; Elander, R.T.; Holtzapple, M.; Ladisch, M.R.; Lee, Y.Y. Coordinated Development of Leading Biomass Pretreatment Technologies. Bioresour. Technol. 2005, 96, 1959–1966. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, M.J.; Alonso, J.L.; Domínguez, H.; Parajó, J.C. Production of Xylose-Containing Fermentation Media by Enzymatic Post-Hydrolysis of Oligomers Produced by Corn Cob Autohydrolysis. World J. Microbiol. Biotechnol. 2001, 17, 817–822. [Google Scholar] [CrossRef]
- Sugiharto, Y.E.C.; Harimawan, A.; Kresnowati, M.T.A.P.; Purwadi, R.; Mariyana, R.; Andry; Fitriana, H.N.; Hosen, H.F. Enzyme Feeding Strategies for Better Fed-Batch Enzymatic Hydrolysis of Empty Fruit Bunch. Bioresour. Technol. 2016, 207, 175–179. [Google Scholar] [CrossRef] [PubMed]

| Composition | Composition (%) |
|---|---|
| NDF 1 | 68.49 ± 1.53 |
| Hemicellulose | 22.55 ± 1.65 |
| ADF 2 | 45.94 ± 0.83 |
| Cellulose | 34.77 ± 1.20 |
| Lignin | 10.58 ± 0.64 |
| Silica | 0.6 ± 0.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mardawati, E.; Febrianti, E.A.; Fitriana, H.N.; Yuliana, T.; Putriana, N.A.; Suhartini, S.; Kasbawati. An Integrated Process for the Xylitol and Ethanol Production from Oil Palm Empty Fruit Bunch (OPEFB) Using Debaryomyces hansenii and Saccharomyces cerevisiae. Microorganisms 2022, 10, 2036. https://doi.org/10.3390/microorganisms10102036
Mardawati E, Febrianti EA, Fitriana HN, Yuliana T, Putriana NA, Suhartini S, Kasbawati. An Integrated Process for the Xylitol and Ethanol Production from Oil Palm Empty Fruit Bunch (OPEFB) Using Debaryomyces hansenii and Saccharomyces cerevisiae. Microorganisms. 2022; 10(10):2036. https://doi.org/10.3390/microorganisms10102036
Chicago/Turabian StyleMardawati, Efri, Emilda Ayu Febrianti, Hana Nur Fitriana, Tri Yuliana, Norisca Aliza Putriana, Sri Suhartini, and Kasbawati. 2022. "An Integrated Process for the Xylitol and Ethanol Production from Oil Palm Empty Fruit Bunch (OPEFB) Using Debaryomyces hansenii and Saccharomyces cerevisiae" Microorganisms 10, no. 10: 2036. https://doi.org/10.3390/microorganisms10102036
APA StyleMardawati, E., Febrianti, E. A., Fitriana, H. N., Yuliana, T., Putriana, N. A., Suhartini, S., & Kasbawati. (2022). An Integrated Process for the Xylitol and Ethanol Production from Oil Palm Empty Fruit Bunch (OPEFB) Using Debaryomyces hansenii and Saccharomyces cerevisiae. Microorganisms, 10(10), 2036. https://doi.org/10.3390/microorganisms10102036








