1. Introduction
Protozoa parasites are a highly diverse group of successful organisms globally distributed via a wide range of hosts [
1] and whose role is crucial for ecosystem functioning [
2]. Among the blood protozoa parasites, the phylum Apicomplexa has received much attention for its zoonotic relevance, for example, the vector borne diseases due to
Plasmodium spp. or the
Toxoplasma spp. transmitted in urban cycles that include domestic species and humans. Other protozoan parasites, such as the
Hepatozoon genus represented by 340 species, have also been commonly recorded in peri-urban cycles sustained by domestic species (e.g., cats and dogs) [
3,
4,
5,
6,
7,
8], although no zoonotic relevance has been reported so far [
9,
10]. Further,
Hepatozoon spp. have been detected in amphibians, reptiles, birds that act as intermediate hosts, and mammals, which can be both paratenic and reservoir hosts [
11].
The transmission of
Hepatozoon spp. can occur through different modalities that most often involve the ingestion of infected vectors or hosts. In mammals, for example, animals can become infected through grooming behavior [
12] by eating infected arthropod ectoparasites attached to another individual, such as ticks [
13], fleas [
14], and mosquitoes [
15]. Further, predator–prey food web transmission routes can occur through the ingestion of prey either containing infective meronts or infested by infected vectors [
16]. Additionally, vertical transplacental transmission has also been reported [
17].
Hepatozoon spp. have been found in a diversity of habitats across the globe, such as temperate broad-leaved forests [
18,
19], conifer forests [
20], Mediterranean scrubland [
21], subtropical forests and savannah [
22], and taiga [
23], likely indicating complex transmission cycles that are still largely unknown. However, the occurrence and prevalence of
Hepatozoon spp. in some crucial ecosystems remain unexplored, for example, across altitudinal gradients where the rapid succession of habitats often corresponds to the high biodiversity of partially sympatric hosts. Under global and climate changes, such mountain habitats are particularly exposed to abiotic and biotic variations that favor upward distributional shifts of host mammal species, with local modifications of the communities [
24,
25]. This may result in parasite expansion in terms of geographic distribution and diversity of the host species involved [
26,
27]. The monitoring of the occurrence of a parasite with a very plastic transmission cycle, such as
Hepatozoon spp., is therefore of crucial importance in these habitats undergoing intense dynamics. This is the case for Alpine ecosystems, where
Hepatozoon spp. were identified in wild carnivores [
18,
20] but have never been recorded in alpine small mammals [
28]. The occurrence of the parasite in the small mammal community is of particular interest, as it prevalently depends on arthropods acting as vectors, the vertical transmission route being secondary. The main arthropods transmitting
Hepatozoon spp. to rodents are blood ectoparasites, such as ticks, fleas, mites, and mosquitoes [
29], the occurrence of which is shifting upward in the Alps as the temperature and humidity conditions become milder with the climate change [
30].
In small mammals,
Hepatozoon spp. life cycles involve schizogony, which occurs in various organs, such as muscles, lymph nodes, the spleen, and generally in the liver, while gametogony occurs in lymphocytes, monocytes, or occasionally, in granulocytes [
29]. Further development requires an arthropod vector in which fertilization and sporogony take place [
29]. Several species of the
Hepatozoon genus were detected in European rodents, e.g.,
H. lavieri [
31],
H. erhardovae [
23,
32],
H. sylvatici [
33,
34],
H. griseisciuri [
33], and sporadically
H. muris (formerly known as
H. perniciosum) [
33,
35]. In rodents, the infection of
Hepatozoon spp. may be pathogenic [
36,
37,
38,
39,
40], although generally mild and asymptomatic [
41]. Conversely, in carnivores (i.e., secondary hosts), such as canids [
42], felids [
43], mustelids [
44], and in snakes [
45],
Hepatozoon spp. may compromise the immune status, thus predisposing it toward coinfection by other pathogens.
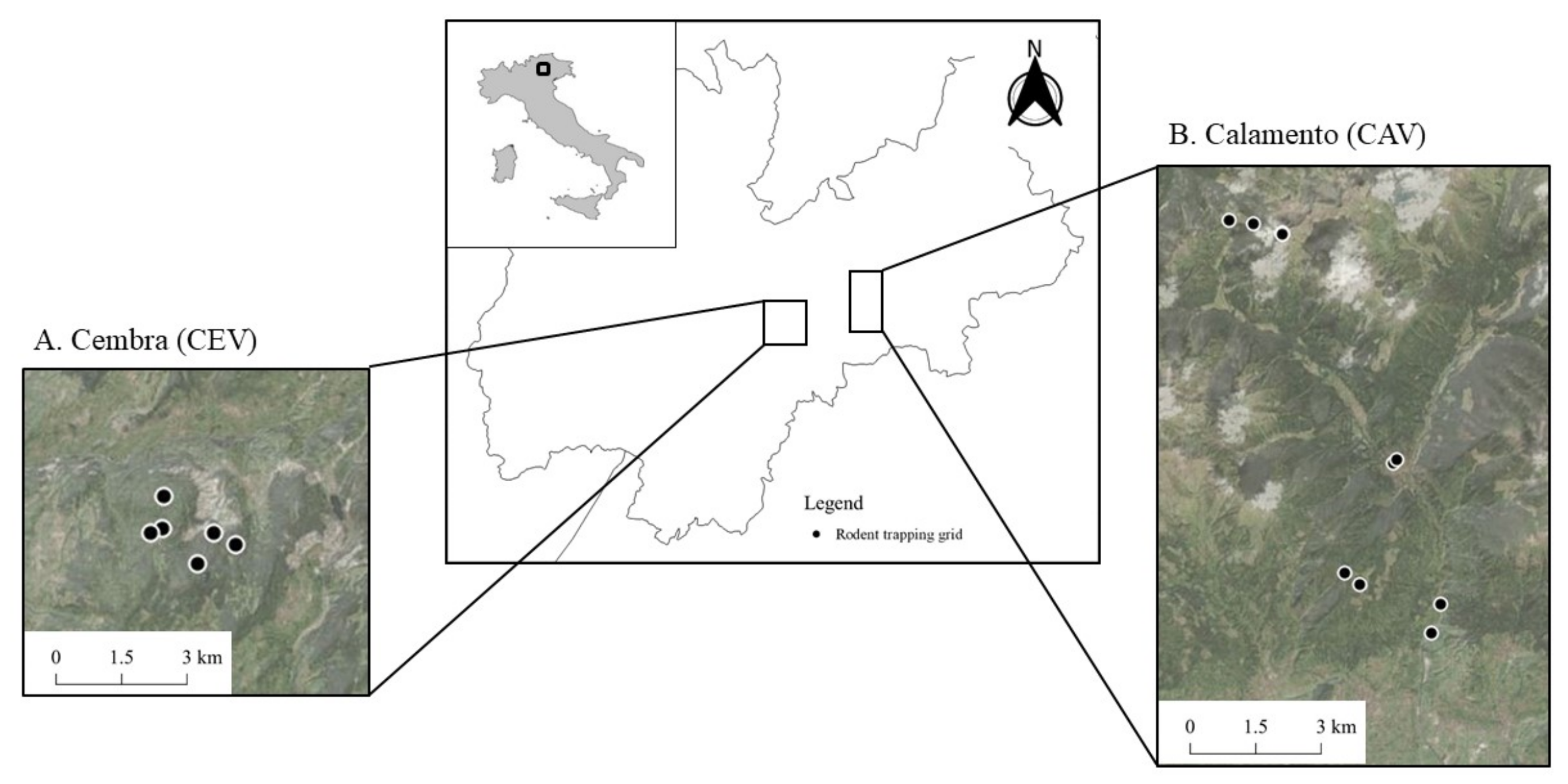
In this work, we conducted a cross-sectional study, i.e., we assessed the parasite prevalence without specifically considering the temporal pattern from 2019 to 2021 in the Italian Alps by intensively live-trapping small mammals from forested habitats up to above the tree line. Our main aim was to evaluate the occurrence of Hepatozoon spp. across the altitudinal gradient of the Alpine habitat also in consideration of the anthropic pressure on such habitat. To the best of our knowledge, this is the first study investigating Hepatozoon spp. occurrence in small mammals, and more generally in rodents in Italy, with a special focus on the Alpine habitat.
3. Results
We analyzed 830 individual ear samples from 11 species (genetically determined through cytochrome-b [
60]): 562 yellow-necked mice (
Apodemus flavicollis), 10 wood mice (
Apodemus sylvaticus), 24 snow voles (
Chionomys nivalis), 2 common voles (
Microtus arvalis), 2 pine voles (
Microtus subterraneus), 1 short-tailed field vole (
Microtus agrestis), 199 bank voles (
Myodes glareolus), 25 common shrews (
Sorex araneus), 3 alpine shrews (
Sorex alpinus), 1 pygmy shrew (
Sorex minutus), and 1 bicolored-shrew (
Crocidura leucodon) (see
Table S1). Additionally, 1790 ticks (1745 larvae and 45 nymphs) and 3782 ticks (3718 larvae and 64 nymphs) were counted on captured rodents in CEV and CAV, respectively. All sampled ticks belonged to the genus
Ixodes. We also recorded the occurrence of other ectoparasites, i.e., fleas, and mites.
All accidentally caught shrews tested negative for
Hepatozoon spp. and were therefore not included in the following analysis (see
Table 1). The results from the 10 double samples were identical and confirmed the robustness of the PCR method.
Among rodents, for all samples, five species tested positive for
Hepatozoon spp.:
A. flavicollis (28.1%),
A. sylvaticus (30.0%),
C. nivalis (33.3%),
M. arvalis (50%), and
My. glareolus (58.8%) (see
Table 1 for details), with an overall prevalence of 35.9% (see
Appendix B for sequences).
In Cembra Valley (CEV; 506 samples),
Hepatozoon spp. was detected in all three rodent species captured (
Table 1).
Hepatozoon spp. prevalence was higher in
My. glareolus if compared with
A. flavicollis (Z-test,
p-value = 6.855 × 10
−6) (
Figure 2a).
In Calamento Valley (CAV; 294 samples), four rodent species tested positive (
Table 1).
My. glareolus showed a statistically significant higher prevalence compared to
A. flavicollis and
C. nivalis (Z-test,
p-value = 6.075 × 10
−12 and
p-value = 0.027, respectively) (
Figure 2b). Among the species with too few captures to compute prevalence,
Hepatozoon spp. was recorded in
M. arvalis, in one individual out of the two captures and
A. sylvaticus, in three out of eight captures.
The results of the GLMMs showed that rodent species have a crucial role in driving the infection of
Hepatozoon spp. Conversely, both human pressure and the altitudinal gradient were not selected in the best models, indicating their scarce relevance in influencing the probability of becoming infected by
Hepatozoon spp. (
Appendix A). In particular, in the first analysis between study areas at 1000 m a.s.l., the infection probability depended on the species, with
My. glareolus more likely to be infected than
A. flavicollis (β = 1.07 ± 0.21;
p-value = 3.27 × 10
−7; reference category:
A. flavicollis). In the second analysis, the infection of
Hepatozoon spp. depended on the additive effect of the species and breeding status, confirming that
My. glareolus was more prone to becoming infected if compared to
A. flavicollis (β = 1.97 ± 0.30;
p-value = 5.98 × 10
−11; reference category:
A. flavicollis), while no significant difference emerged between
A. flavicollis and
C. nivalis (
Appendix A). In addition, a minor effect of the breeding status emerged, in which juveniles seemed to be marginally more infected compared to adults (β = 1.29 ± 0.70;
p-value = 0.06; reference category: Adults), while we did not detect any significant difference between adults and subadults (
Appendix A).
It is relevant to observe that, after sequencing, two samples (one from
S. araneus and one from
My. glareolus) previously assigned to
Hepatozoon spp. were instead confirmed as
Babesia microti (see
Appendix B).
4. Discussion
This study reports for the first time the occurrence and prevalence of Hepatozoon spp. in wild rodents in the Italian Alps. In particular, we found a high prevalence in the most common woodland rodent hosts, i.e., My. glareolus and A. flavicollis, as well as in two other alpine species: the sympatric A. sylvaticus and C. nivalis at high altitudes.
This study is part of a wider project investigating tick-borne pathogen circulation in small mammals in the Italian Alps. In this context, the detection of rodent infection by
Hepatozoon spp. represented a bycatch of the assessment of
Babesia spp. For this reason,
Babesia-specific primers were used for the screening, and the positive samples were further identified through sequencing. The identification of two positive samples of
Babesia microti underlines the risk of mismatch when
Babesia-specific primers are used for detecting
Hepatozoon spp., as already underlined by other studies [
47]. This finding suggests that follow-up sequencing is a compulsory step to discriminate this parasite by other protozoa when their detection is due to nonspecific protocols. For these reasons, we were not able to further identify
Hepatozoon spp. at the species level due to the low quality of the obtained sequences. The identification could be refined by using
Hepatozoon-specific primers (HEPF/HEPR [
47]).
European studies on
Hepatozoon spp. in wild rodents have been generally based on blood [
32,
47,
61,
62], organs [
14,
22,
23], and, more rarely, on skin [
13,
63] samples, which are often collected through invasive sampling methods that, in many cases, require the suppression of the animals. In live rodents,
Hepatozoon spp. can be identified only through blood sampling, to the best of our knowledge [
31,
32]. This comes with the drawback that traditional microscopy based on a blood smear can underestimate
Hepatozoon spp. prevalence, especially with low intensity of infection [
47]. On the contrary, PCR-based assays, such as those that we implemented using ear biopsy samples collected from live-trapped rodents, are considered more sensitive and robust. Coupling molecular screening approaches with less invasive sampling methods may accomplish conservation issues in cases of endangered and vulnerable species, e.g.,
C. nivalis [
64], limiting the impact on the studied species. Within this framework, we believe that our contribution may provide a useful methodological approach. Although the comparison of past studies based on different protocols or matrices might not be feasible, the comparison between different procedures to assess
Hepatozoon spp., e.g., by comparing ear tissue samples with skin ones as those collected in studies [
13,
63], could be an interesting direction of research.
The number of species of the small mammal community in which
Hepatozoon spp. was detected, its first identification in
C. nivalis (33.3%), and its occurrence at 2000 and 2500 m a.s.l., the highest elevation at which these parasites have ever been recorded, represent key findings to speculate on the transmission pathways of
Hepatozoon spp. in the Alpine range. Additionally, this is the first record of
Hepatozoon spp. in
A. sylvaticus (30.0%) using molecular approaches [
34,
65]. Since small mammals can become infected from
Hepatozoon spp. mainly though the ingestion of infected vectors, it is reasonable to consider
Hepatozoon spp. as a vector-borne infectious agent for small mammal hosts and specifically in our case, although we did not assess the infective status and transmission dynamics of arthropod vectors. Our results suggest that
Hepatozoon spp. are common in alpine environments, showing a high prevalence in a broad spectrum of alpine small mammal species. In particular, we did not detect any relevant difference in
Hepatozoon spp. prevalence between anthropic and wild systems, implying a stable and ubiquitous presence of
Hepatozoon spp. in the analyzed small mammal species. This is interesting, as
Hepatozoon spp. have been previously studied mainly in domestic species [
3,
4,
5,
6,
7,
8]. The low host specificity displayed by
Hepatozoon spp. [
23,
29] may indicate that these parasites persist in the environment sustained by competent and reservoir hosts, such as small mammals [
66], both in wild and more anthropic settings. These conditions may increase the risk of spillover events among wildlife and domestic animals [
67,
68]. For example,
A. sylvaticus generally shares food resources, part of the habitats and vectors, with other rodent competitor species, especially in case of high rodent and vector density [
69], as it may occur in permissive environmental conditions (low elevations, in our case). The high density of reservoir hosts and of potentially infected vectors may amplify parasite circulation and spreading within the ecosystem, facilitating also the infection of less abundant host species [
70]. This mechanism may have also favored the transition of the parasite to high altitudes, especially under increasingly milder climatic conditions. The less-limiting abiotic conditions along the altitudinal gradient occurring under climate change may promote an upward distributional shift of opportunistic small mammal host species (e.g.,
My. glareolus and
Apodemus spp.) and, in turn, the survival and development of arthropod vectors (e.g., mites, fleas, and ticks) [
27,
71,
72]. The combination of abiotic (i.e., climate) and biotic (i.e., interspecific interactions) conditions that permit host and vector persistence, together with the ability of a species to colonize favorable habitats, may therefore alter the local alpine communities, leading to novel host–vector species pairings and, thus, to the emergence of vector-borne diseases in new environments [
73,
74,
75,
76]. The occurrence of
Hepatozoon spp. in
C. nivalis and high altitudes that we recorded is compatible with these dynamics, although there is no track record of previous investigations at the same altitudes in small mammals (see Reference [
28]).
The high rate of infection that we observed in different rodent species across a diversity of Alpine habitats may induce relevant cascading effects across the food web. In particular, being carnivore-competent hosts for
Hepatozoon spp., a broad spectrum of both domestic (e.g., shepherd dogs [
77]) and wild animals (e.g., snakes [
78], meso- and large carnivores [
20,
79], and birds of prey [
80]) may become infected through the ingestion of parasitized rodent preys. Specifically, small mammals are paratenic hosts for
Hepatozoon spp., meaning that, despite not being necessary for parasite development, they help in maintaining its life cycle in the environment by supporting the cystozoite stages [
40] infective for predators via ingestion [
81]. The parasite can affect the immune response of predator secondary hosts, leading to population declines [
39]. However, at the same time, since vector competence in becoming infected, replicating and transmitting the parasite varies across hosts [
81], predators may represent dead end hosts for
Hepatozoon spp. so indirectly reducing parasites transmission across ecosystem (i.e., dilution effect). This aspect of the transmission pathway of
Hepatozoon spp. needs further investigation.
The prevalence of
Hepatozoon spp. that we recorded in the most common rodent hosts, i.e.,
My. glareolus and
A. flavicollis, is in partial accordance with other studies in Europe. In particular, we found that
My. glareolus showed the highest prevalence of
Hepatozoon spp. among all captured rodent species (58.8%), as was also observed in other studies [
13,
32,
47,
63], although with very variable levels (from 3.7% to 87.5% [
13,
23,
47,
48,
62,
82]). Conversely, the prevalence we found for
A. flavicollis (28.1%) is higher than all previously published estimates that were below 10% [
13,
47,
48,
63] and may support a high prevalence of
Hepatozoon spp. infection at lower elevations. The small sample size of
Microtus spp. voles does not allow conclusive indications on the rate of infection of this genus (one positive
M. arvalis out of five in total), as was the case also for other studies [
23,
31,
32,
47].
In conclusion, Apicomplexa remains one of the most poorly investigated groups among protozoa, so that accurate identification (via PCR-based methods coupled with sequencing) of
Hepatozoon spp. in new wild hosts and habitats importantly contributes to the understanding of the ecological role of these parasites, especially under the current global change. Collectively, by detecting
Hepatozoon spp. in a broad range of rodent hosts throughout the altitudinal gradient of Alpine habitats, we indicated the widespread occurrence of this parasite as likely supported by an expanding availability of arthropod vectors, due to variations in the temperature and moisture linked to climate change [
75]. Therefore, this study represents a crucial starting point for future research combining ecological, epidemiological, and molecular analysis to evaluate the trophic transmission route of
Hepatozoon spp. across the food chain in habitats particularly exposed to climate change, such as the Alpine range. In particular, studies investigating vector competence and distribution, as well as host ecology and their role in maintaining and transmitting
Hepatozoon spp., are needed to clarify the function of this group of parasites in such changing ecosystems.