Nanopore MinION Sequencing Generates a White Spot Syndrome Virus Genome from a Pooled Cloacal Swab Sample of Domestic Chickens in South Africa
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Sample Collection
2.3. Nucleic Acid Extraction
2.4. Metagenomic Sequencing
2.4.1. Round A—First-Strand Synthesis with Reverse Transcriptase (RT)
2.4.2. Second-Strand Synthesis with Sequenase Version 2.0 DNA Polymerase
2.4.3. Round B—Random PCR Amplification
2.4.4. Round C—PCR Enrichment and Clean Up
2.4.5. Nanopore MinION Library Preparation and Sequencing
2.4.6. Genome Assembly and Annotation
2.5. Phylogenetic Analysis
2.6. Principal Coordinates Analysis (PCoA)
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, F.; He, J.; Lin, X.; Li, Q.; Pan, D.; Zhang, X.; Xu, X. Complete genome sequence of the shrimp white spot bacilliform virus. J. Virol. 2001, 75, 11811–11820. [Google Scholar] [CrossRef]
- Patil, P.K.; Geetha, R.; Ravisankar, T.; Avunje, S.; Solanki, H.G.; Abraham, T.J.; Vinoth, S.P.; Jithendran, K.P.; Alavandi, S.V.; Vijayan, K.K. Economic loss due to diseases in Indian shrimp farming with special reference to Enterocytozoon hepatopenaei (EHP) and white spot syndrome virus (WSSV). Aquaculture 2021, 533, 736231. [Google Scholar] [CrossRef]
- Zhan, W.-B.; Wang, Y.-H.; Fryer, J.L.; Yu, K.-K.; Fukuda, H.; Meng, Q.-X. White Spot Syndrome Virus Infection of Cultured Shrimp in China. J. Aquat. Anim. Health 1998, 10, 405–410. [Google Scholar] [CrossRef]
- Kono, T.; Savan, R.; Sakai, M.; Itami, T. Detection of white spot syndrome virus in shrimp by loop-mediated isothermal amplification. J. Virol. Methods 2004, 115, 59–65. [Google Scholar] [CrossRef]
- Saravanan, K.; Kumar, P.P.; Praveenraj, J.; Baruah, A.; Sivaramakrishnan, T.; Kumar, T.S.; Kumar, S.P.; Sankar, R.K.; Roy, S.D. Investigation and confirmation of white spot syndrome virus (WSSV) infection in wild caught penaeid shrimps of Andaman and Nicobar Islands, India. Virusdisease 2017, 28, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Paz, A. White spot syndrome virus: An overview on an emergent concern. Vet. Res. 2010, 41, 43. [Google Scholar] [CrossRef]
- Escobedo-Bonilla, C.M.; Wille, M.; Alday Sanz, V.; Sorgeloos, P.; Pensaert, M.B.; Nauwynck, H.J. Pathogenesis of a Thai strain of white spot syndrome virus (WSSV) in juvenile, specific pathogen-free Litopenaeus vannamei. Dis. Aquat. Organ. 2007, 74, 85–94. [Google Scholar] [CrossRef]
- Dey, B.K.; Dugassa, G.H.; Hinzano, S.M.; Bossier, P. Causative agent, diagnosis and management of white spot disease in shrimp: A review. Rev. Aquac. 2020, 12, 822–865. [Google Scholar] [CrossRef]
- Chou, H.Y.; Huang, C.Y.; Lo, C.F.; Kou, G.H. Studies on transmission of white spot syndrome associated baculovirus (WSBV) in Penaeus monodon and P. japonicus via waterborne contact and oral ingestion. Aquaculture 1998, 164, 263–276. [Google Scholar] [CrossRef]
- Chou Hy, H.C.Y.; Wang, C.H.; Chiang, H.C.; Lo, C.F. Pathogenicity of a baculovirus infection causing white spot syndrome in cultured penaeid shrimp in Taiwan. Dis. Aquat. Org. 1995, 23, 165–173. [Google Scholar] [CrossRef]
- Nakano, H.; Koube, H.; Umezawa, S.; Momoyama, K.; Hiraoka, M.; Inouye, K.; Oseko, N. Mass Mortalities of Cultured Kuruma Shrimp, Penaeus japonicus, in Japan in 1993 : Epizootiological Survey and Infection Trials. Fish Pathol. 1994, 29, 135–139. [Google Scholar] [CrossRef]
- Zhu, F.; Twan, W.-H.; Tseng, L.-C.; Peng, S.-H.; Hwang, J.-S. First detection of white spot syndrome virus (WSSV) in the mud shrimp Austinogebia edulis in Taiwan. Sci. Rep. 2019, 9, 18572. [Google Scholar] [CrossRef]
- Vaseeharan, B.; Jayakumar, R.; Ramasamy, P. PCR-based detection of white spot syndrome virus in cultured and captured crustaceans in India. Lett. Appl. Microbiol. 2003, 37, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Kawato, S.; Shitara, A.; Wang, Y.; Nozaki, R.; Kondo, H.; Hirono, I. Crustacean Genome Exploration Reveals the Evolutionary Origin of White Spot Syndrome Virus. J. Virol. 2019, 93, e01144-18. [Google Scholar] [CrossRef]
- Sahul Hameed, A.S.; Yoganandhan, K.; Sathish, S.; Rasheed, M.; Murugan, V.; Jayaraman, K. White spot syndrome virus (WSSV) in two species of freshwater crabs (Paratelphusa hydrodomous and P. pulvinata). Aquaculture 2001, 201, 179–186. [Google Scholar] [CrossRef]
- Stentiford, G.D.; Lightner, D.V. Cases of White Spot Disease (WSD) in European shrimp farms. Aquaculture 2011, 319, 302–306. [Google Scholar] [CrossRef]
- Oakey, H.J.; Smith, C.S. Complete genome sequence of a white spot syndrome virus associated with a disease incursion in Australia. Aquaculture 2018, 484, 152–159. [Google Scholar] [CrossRef]
- Restrepo, L.; Reyes, A.; Bajaña, L.; Betancourt, I.; Bayot, B. Draft Genome Sequence of a White Spot Syndrome Virus Isolate Obtained in Ecuador. Genome Announc. 2018, 6, e00605-18. [Google Scholar] [CrossRef] [PubMed]
- Muller, I.C.; Andrade, T.P.; Tang-Nelson, K.F.; Marques, M.R.; Lightner, D.V. Genotyping of white spot syndrome virus (WSSV) geographical isolates from Brazil and comparison to other isolates from the Americas. Dis. Aquat. Organ. 2010, 88, 91–98. [Google Scholar] [CrossRef]
- Rodriguez-Anaya, L.Z.; Gonzalez-Galaviz, J.R.; Casillas-Hernandez, R.; Lares-Villa, F.; Estrada, K.; Ibarra-Gamez, J.C.; Sanchez-Flores, A. Draft Genome Sequence of White Spot Syndrome Virus Isolated from Cultured Litopenaeus vannamei in Mexico. Genome Announc. 2016, 4, e01674-15. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Xiao, J.; Liu, L.; Pan, Y.; Yan, S.; Wang, Y. Characterization and prevalence of a novel white spot syndrome viral genotype in naturally infected wild crayfish, Procambarus clarkii, in Shanghai, China. Virus Dis. 2017, 28, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Syed Musthaq, S.; Sudhakaran, R.; Balasubramanian, G.; Sahul Hameed, A.S. Experimental transmission and tissue tropism of white spot syndrome virus (WSSV) in two species of lobsters, Panulirus homarus and Panulirus ornatus. J. Invertebr. Pathol. 2006, 93, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Clark, K.F.; Greenwood, S.J.; Acorn, A.R.; Byrne, P.J. Molecular immune response of the American lobster (Homarus americanus) to the White Spot Syndrome Virus. J. Invertebr. Pathol. 2013, 114, 298–308. [Google Scholar] [CrossRef]
- Shinn, A.; Pratoomyot, J.; Griffiths, D.; Trong, T.; Vu, N.T.; Jiravanichpaisal, P.; Briggs, M. Asian shrimp production and the economic costs of disease. Asian Fish. Sci. 2018, 31, 29–58. [Google Scholar] [CrossRef]
- Miao, W.; Wang, W. Trends of Aquaculture Production and Trade: Carp, Tilapia, and Shrimp. Asian Fish. Sci. 2020, 33, 1–10. [Google Scholar] [CrossRef]
- Desrina; Prayitno, S.B.; Verdegem, M.C.J.; Verreth, J.A.J.; Vlak, J.M. White spot syndrome virus host range and impact on transmission. Rev. Aquac. 2022, 14, 1843–1860. [Google Scholar] [CrossRef]
- Vanpatten, K.A.; Nunan, L.M.; Lightner, D.V. Seabirds as potential vectors of penaeid shrimp viruses and the development of a surrogate laboratory model utilizing domestic chickens. Aquaculture 2004, 241, 31–46. [Google Scholar] [CrossRef]
- Chang, Y.S.; Chen, T.C.; Liu, W.J.; Hwang, J.S.; Kou, G.H.; Lo, C.F. Assessment of the roles of copepod Apocyclops royi and bivalve mollusk Meretrix lusoria in white spot syndrome virus transmission. Mar. Biotechnol. 2011, 13, 909–917. [Google Scholar] [CrossRef]
- Vazquez-Boucard, C.; Alvarez-Ruiz, P.; Escobedo-Fregoso, C.; Anguiano-Vega, G.; Duran-Avelar Mde, J.; Pinto, V.S.; Escobedo-Bonilla, C.M. Detection of white spot syndrome virus (WSSV) in the Pacific oyster Crassostrea gigas. J. Invertebr. Pathol. 2010, 104, 245–247. [Google Scholar] [CrossRef]
- Vijayan, K.K.; Stalin Raj, V.; Balasubramanian, C.P.; Alavandi, S.V.; Thillai Sekhar, V.; Santiago, T.C. Polychaete worms--a vector for white spot syndrome virus (WSSV). Dis. Aquat. Organ. 2005, 63, 107–111. [Google Scholar] [CrossRef]
- Desrina; Verreth, J.A.; Prayitno, S.B.; Rombout, J.H.; Vlak, J.M.; Verdegem, M.C. Replication of white spot syndrome virus (WSSV) in the polychaete Dendronereis spp. J. Invertebr. Pathol. 2013, 114, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Haryadi, D.; Verreth, J.A.; Verdegem, M.C.; Vlak, J.M. Transmission of white spot syndrome virus (WSSV) from Dendronereis spp. (Peters) (Nereididae) to penaeid shrimp. J. Fish. Dis. 2015, 38, 419–428. [Google Scholar] [CrossRef]
- Jiang, G. Can white spot syndrome virus be transmitted through the phytoplankton→ rotifer→ artemia→ shrimp pathway? Afr. J. Biotechnol. 2012, 11, 1277–1282. [Google Scholar]
- Liu, B.; Yu, Z.; Song, X.; Guan, Y. Studies on the transmission of WSSV (white spot syndrome virus) in juvenile Marsupenaeus japonicus via marine microalgae. J. Invertebr. Pathol. 2007, 95, 87–92. [Google Scholar] [CrossRef]
- Blagodatski, A.; Trutneva, K.; Glazova, O.; Mityaeva, O.; Shevkova, L.; Kegeles, E.; Onyanov, N.; Fede, K.; Maznina, A.; Khavina, E.; et al. Avian Influenza in Wild Birds and Poultry: Dissemination Pathways, Monitoring Methods, and Virus Ecology. Pathogens 2021, 10, 630. [Google Scholar] [CrossRef]
- McDuie, F.; Matchett, E.L.; Prosser, D.J.; Takekawa, J.Y.; Pitesky, M.E.; Lorenz, A.A.; McCuen, M.M.; T, O.C.; Ackerman, J.T.; De La Cruz, S.E.W.; et al. Pathways for avian influenza virus spread: GPS reveals wild waterfowl in commercial livestock facilities and connectivity with the natural wetland landscape. Transbound. Emerg. Dis. 2022, 69, 2898–2912. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.M.; Douglas, D.C.; Ramey, A.M.; Mullinax, J.M.; Soos, C.; Link, P.; Walther, P.; Prosser, D.J. The spatial–temporal relationship of blue-winged teal to domestic poultry: Movement state modelling of a highly mobile avian influenza host. J. Appl. Ecol. 2021, 58, 2040–2052. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Meng, C.; Sun, Y.; Mahrose, K.M.; Umar, S.; Ding, C.; Munir, M. Pathobiology of Avian avulavirus 1: Special focus on waterfowl. Vet. Res. 2018, 49, 94. [Google Scholar] [CrossRef]
- Reusch, K.; Suárez, N.; Ryan, P.G.; Pichegru, L. Foraging movements of breeding Kelp Gulls in South Africa. Mov Ecol 2020, 8, 36. [Google Scholar] [CrossRef]
- Bussière, E.M.; Underhill, L.G.; Altwegg, R. Patterns of bird migration phenology in South Africa suggest northern hemisphere climate as the most consistent driver of change. Glob. Change Biol. 2015, 21, 2179–2190. [Google Scholar] [CrossRef]
- Szép, T.; Møller, A.P.; Piper, S.; Nuttall, R.; Szabó, Z.D.; Pap, P.L. Searching for potential wintering and migration areas of a Danish Barn Swallow population in South Africa by correlating NDVI with survival estimates. J. Ornithol. 2006, 147, 245–253. [Google Scholar] [CrossRef]
- Cumming, G.S.; Caron, A.; Abolnik, C.; Cattoli, G.; Bruinzeel, L.W.; Burger, C.E.; Cecchettin, K.; Chiweshe, N.; Mochotlhoane, B.; Mutumi, G.L.; et al. The ecology of influenza A viruses in wild birds in southern Africa. Ecohealth 2011, 8, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Froneman, P.W.; Blake, J.D.; Hulley, P. Aspects of population dynamics and feeding by piscivorous birds in the intermittently open Riet River estuary, Eastern Cape, South Africa. Afr. J. Aquat. Sci. 2011, 36, 101–107. [Google Scholar] [CrossRef]
- Elbers, A.R.W.; Gonzales, J.L. Quantification of visits of wild fauna to a commercial free-range layer farm in the Netherlands located in an avian influenza hot-spot area assessed by video-camera monitoring. Transbound. Emerg. Dis. 2020, 67, 661–677. [Google Scholar] [CrossRef]
- Klenner, J.; Kohl, C.; Dabrowski, P.W.; Nitsche, A. Comparing Viral Metagenomic Extraction Methods. Curr. Issues Mol. Biol. 2017, 24, 59–70. [Google Scholar] [CrossRef]
- Ladman, B.S.; Spackman, E.; Gelb, J., Jr. Comparison of pooling 11 or 5 oropharyngeal swabbings for detecting avian influenza virus by real-time reverse transcription-PCR in broiler chickens. Avian Dis. 2012, 56, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Urisman, A.; Liu, Y.T.; Springer, M.; Ksiazek, T.G.; Erdman, D.D.; Mardis, E.R.; Hickenbotham, M.; Magrini, V.; Eldred, J.; et al. Viral discovery and sequence recovery using DNA microarrays. PLoS Biol. 2003, 1, E2. [Google Scholar] [CrossRef] [PubMed]
- Vilsker, M.; Moosa, Y.; Nooij, S.; Fonseca, V.; Ghysens, Y.; Dumon, K.; Pauwels, R.; Alcantara, L.C.; Vanden Eynden, E.; Vandamme, A.-M.; et al. Genome Detective: An automated system for virus identification from high-throughput sequencing data. Bioinformatics 2018, 35, 871–873. [Google Scholar] [CrossRef]
- Deforche, K. An alignment method for nucleic acid sequences against annotated genomes. bioRxiv 2017. bioRxiv:200394. [Google Scholar] [CrossRef]
- Tcherepanov, V.; Ehlers, A.; Upton, C. Genome Annotation Transfer Utility (GATU): Rapid annotation of viral genomes using a closely related reference genome. BMC Genom. 2006, 7, 150. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Asche, F.; Anderson, J.L.; Botta, R.; Kumar, G.; Abrahamsen, E.B.; Nguyen, L.T.; Valderrama, D. The economics of shrimp disease. J. Invertebr. Pathol. 2021, 186, 107397. [Google Scholar] [CrossRef] [PubMed]
- Onihary, A.M.; Razanajatovo, I.M.; Rabetafika, L.; Bastaraud, A.; Heraud, J.M.; Rasolofo, V. Genotype Diversity and Spread of White Spot Syndrome Virus (WSSV) in Madagascar (2012–2016). Viruses 2021, 13, 1713. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.F.; Le Groumellec, M.; Lightner, D.V. Novel, closely related, white spot syndrome virus (WSSV) genotypes from Madagascar, Mozambique and the Kingdom of Saudi Arabia. Dis. Aquat. Organ. 2013, 106, 1–6. [Google Scholar] [CrossRef]
- Baumgartner, W.A.; Hawke, J.P.; Bowles, K.; Varner, P.W.; Hasson, K.W. Primary diagnosis and surveillance of white spot syndrome virus in wild and farmed crawfish (Procambarus clarkii, P. zonangulus) in Louisiana, USA. Dis. Aquat. Organ. 2009, 85, 15–22. [Google Scholar] [CrossRef]
- Chapman, R.W.; Browdy, C.L.; Savin, S.; Prior, S.; Wenner, E. Sampling and evaluation of white spot syndrome virus in commercially important Atlantic penaeid shrimp stocks. Dis. Aquat. Organ. 2004, 59, 179–185. [Google Scholar] [CrossRef]
- Bandeira, J.d.T.; Morais, R.S.M.M.d.; Silva, R.P.P.e.; Mendes, E.S.; Silva, S.M.B.C.d.; Santos, F.L.d. First report of white spot syndrome virus in wild crustaceans and mollusks in the Paraíba River, Brazil. Aquac. Res. 2019, 50, 680–684. [Google Scholar] [CrossRef]
- Rajendran, K.; Vijayan, K.; Santiago, T.; Krol, R. Experimental host range and histopathology of white spot syndrome virus (WSSV) infection in shrimp, prawns, crabs and lobsters from India. J. Fish Dis. 1999, 22, 183–191. [Google Scholar] [CrossRef]
- Sánchez-Paz, A.; Terán-Díaz, B.; Enríquez-Espinoza, T.; Encinas-Garcia, T.; Vázquez-Sánchez, I.; Mendoza-Cano, F. The tidepool shrimp, Palaemon ritteri Holmes, constitutes a novel host to the white spot syndrome virus. J. Fish. Dis. 2015, 38, 613–620. [Google Scholar] [CrossRef]
- Hameed, A.S.; Balasubramanian, G.; Musthaq, S.S.; Yoganandhan, K. Experimental infection of twenty species of Indian marine crabs with white spot syndrome virus (WSSV). Dis. Aquat. Organ. 2003, 57, 157–161. [Google Scholar] [CrossRef]
- Ross, E.P.; Behringer, D.C.; Bojko, J. White spot syndrome virus and the Caribbean spiny lobster, Panulirus argus: Susceptibility and behavioral immunity. J. Invertebr. Pathol. 2019, 162, 1–9. [Google Scholar] [CrossRef]
- Swart, J.J.; Brink, D. Rapid communication: The 1998 production and status of aquaculture in South Africa. Water SA 2000, 26, 133–135. [Google Scholar]
- Caliendo, V.; Lewis, N.S.; Pohlmann, A.; Baillie, S.R.; Banyard, A.C.; Beer, M.; Brown, I.H.; Fouchier, R.A.M.; Hansen, R.D.E.; Lameris, T.K.; et al. Transatlantic spread of highly pathogenic avian influenza H5N1 by wild birds from Europe to North America in 2021. Sci. Rep. 2022, 12, 11729. [Google Scholar] [CrossRef] [PubMed]
- Reed, K.D.; Meece, J.K.; Henkel, J.S.; Shukla, S.K. Birds, migration and emerging zoonoses: West nile virus, lyme disease, influenza A and enteropathogens. Clin. Med. Res. 2003, 1, 5–12. [Google Scholar] [CrossRef]
- Global Consortium for H5N8 and Related Influenza Viruses. Role for migratory wild birds in the global spread of avian influenza H5N8. Science 2016, 354, 213–217. [Google Scholar] [CrossRef]
- Verhagen, J.H.; Fouchier, R.A.M.; Lewis, N. Highly Pathogenic Avian Influenza Viruses at the Wild-Domestic Bird Interface in Europe: Future Directions for Research and Surveillance. Viruses 2021, 13, 212. [Google Scholar] [CrossRef] [PubMed]
- Gass, J.D., Jr.; Dusek, R.J.; Hall, J.S.; Hallgrimsson, G.T.; Halldórsson, H.P.; Vignisson, S.R.; Ragnarsdottir, S.B.; Jónsson, J.E.; Krauss, S.; Wong, S.S.; et al. Global dissemination of influenza A virus is driven by wild bird migration through arctic and subarctic zones. Mol. Ecol. 2023, 32, 198–213. [Google Scholar] [CrossRef] [PubMed]
- Fourment, M.; Darling, A.E.; Holmes, E.C. The impact of migratory flyways on the spread of avian influenza virus in North America. BMC Evol. Biol. 2017, 17, 118. [Google Scholar] [CrossRef] [PubMed]
- Buhnerkempe, M.G.; Webb, C.T.; Merton, A.A.; Buhnerkempe, J.E.; Givens, G.H.; Miller, R.S.; Hoeting, J.A. Identification of migratory bird flyways in North America using community detection on biological networks. Ecol. Appl. 2016, 26, 740–751. [Google Scholar] [CrossRef] [PubMed]
- Mine, J.; Uchida, Y.; Sharshov, K.; Sobolev, I.; Shestopalov, A.; Saito, T. Phylogeographic evidence for the inter- and intracontinental dissemination of avian influenza viruses via migration flyways. PLoS ONE 2019, 14, e0218506. [Google Scholar] [CrossRef]
- Dusek, R.J.; Hallgrimsson, G.T.; Ip, H.S.; Jónsson, J.E.; Sreevatsan, S.; Nashold, S.W.; TeSlaa, J.L.; Enomoto, S.; Halpin, R.A.; Lin, X.; et al. North Atlantic migratory bird flyways provide routes for intercontinental movement of avian influenza viruses. PLoS ONE 2014, 9, e92075. [Google Scholar] [CrossRef]
- Yong, D.L.; Heim, W.; Chowdhury, S.U.; Choi, C.-Y.; Ktitorov, P.; Kulikova, O.; Kondratyev, A.V.; Round, P.D.; Allen, D.N.S.; Trainor, C.R.; et al. The State of Migratory Landbirds in the East Asian Flyway: Distributions, Threats, and Conservation Needs. Proc. the Front. Ecol. Evol. 2021, 9. [Google Scholar] [CrossRef]
- Takekawa, J.Y.; Prosser, D.J.; Sullivan, J.D.; Yin, S.; Wang, X.; Zhang, G.; Xiao, X. Potential Effects of Habitat Change on Migratory Bird Movements and Avian Influenza Transmission in the East Asian-Australasian Flyway. Diversity 2023, 15, 601. [Google Scholar] [CrossRef]
- Tapia-Harris, C.; Izang, A.; Cresswell, W. Migratory routes, breeding locations and multiple non-breeding sites of Common Whitethroats Curruca communis revealed by geolocators. PLoS ONE 2022, 17, e0274017. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, J.L.; Jones, V.R.; Catry, I.; Beal, M.; Dias, M.P.; Oppel, S.; Vickery, J.A.; Hewson, C.M.; Butchart, S.H.M.; Rodrigues, A.S.L. Connectivity between countries established by landbirds and raptors migrating along the African-Eurasian flyway. Conserv. Biol. 2023, 37, e14002. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, L.; Shittu, I.; Fusaro, A.; Inuwa, B.; Zecchin, B.; Gado, D.; Schivo, A.; Bianco, A.; Laleye, A.; Gobbo, F.; et al. Live Bird Markets in Nigeria: A Potential Reservoir for H9N2 Avian Influenza Viruses. Viruses 2021, 13, 1445. [Google Scholar] [CrossRef]
- Meseko, C.; Milani, A.; Inuwa, B.; Chinyere, C.; Shittu, I.; Ahmed, J.; Giussani, E.; Palumbo, E.; Zecchin, B.; Bonfante, F.; et al. The Evolution of Highly Pathogenic Avian Influenza A (H5) in Poultry in Nigeria, 2021–2022. Viruses 2023, 15, 1387. [Google Scholar] [CrossRef]
- Soliman, A.; Saad, M.; Elassal, E.; Amir, E.; Plathonoff, C.; Bahgat, V.; El-Badry, M.; Ahmed, L.S.; Fouda, M.; Gamaleldin, M.; et al. Surveillance of avian influenza viruses in migratory birds in Egypt, 2003–2009. J. Wildl. Dis. 2012, 48, 669–675. [Google Scholar] [CrossRef]
- Naguib, M.M.; Verhagen, J.H.; Samy, A.; Eriksson, P.; Fife, M.; Lundkvist, Å.; Ellström, P.; Järhult, J.D. Avian influenza viruses at the wild-domestic bird interface in Egypt. Infect. Ecol. Epidemiol. 2019, 9, 1575687. [Google Scholar] [CrossRef]
- Fusaro, A.; Zecchin, B.; Vrancken, B.; Abolnik, C.; Ademun, R.; Alassane, A.; Arafa, A.; Awuni, J.A.; Couacy-Hymann, E.; Coulibaly, M.B.; et al. Disentangling the role of Africa in the global spread of H5 highly pathogenic avian influenza. Nat. Commun. 2019, 10, 5310. [Google Scholar] [CrossRef]
- Abolnik, C.; Phiri, T.P.; van der Zel, G.; Anthony, J.; Daniell, N.; de Boni, L. Wild Bird Surveillance in the Gauteng Province of South Africa during the High-Risk Period for Highly Pathogenic Avian Influenza Virus Introduction. Viruses 2022, 14, 2027. [Google Scholar] [CrossRef]
- Van de Kam, J.; Ens, B.; Piersma, T.; Zwarts, L. Shorebirds: An Illustrated Behavioural Ecology; Brill: Leiden, The Netherlands, 2017. [Google Scholar]
- Davidson, N.C.; Stroud, D.A. African-Western Eurasian Flyways: Current knowledge, population status and future challenges. In Waterbirds around the world; Boere, G.C., Galbraith, C.A., Stroud, D.A., Eds.; The Stationery Office: Edinburgh, UK, 2006; pp. 63–73. [Google Scholar]
- Djikeng, A.; Halpin, R.; Kuzmickas, R.; DePasse, J.; Feldblyum, J.; Sengamalay, N.; Afonso, C.; Zhang, X.; Anderson, N.G.; Ghedin, E.; et al. Viral genome sequencing by random priming methods. BMC Genom. 2008, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Vinaya Kumar, K.; Shekhar, M.S.; Otta, S.K.; Karthic, K.; Ashok Kumar, J.; Gopikrishna, G.; Vijayan, K.K. First Report of a Complete Genome Sequence of White spot syndrome virus from India. Genome Announc. 2018, 6, e00055-18. [Google Scholar] [CrossRef] [PubMed]
- Dantas, M.D.A.; Teixeira, D.G.; Silva-Portela, R.C.B.; Soares, P.E.T.; Lima, J.P.M.S.; Agnez-Lima, L.F.; Lanza, D.C.F. Direct sequencing of the white spot syndrome virus from Brazil: Genome assembly and new insights on phylogeny. Virus Res. 2018, 245, 52–61. [Google Scholar] [CrossRef] [PubMed]
- van Hulten, M.C.W.; Witteveldt, J.; Peters, S.; Kloosterboer, N.; Tarchini, R.; Fiers, M.; Sandbrink, H.; Lankhorst, R.K.; Vlak, J.M. The White Spot Syndrome Virus DNA Genome Sequence. Virology 2001, 286, 7–22. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Gao, M.; Xu, L.; Yang, F. Comparative genomic analysis of three white spot syndrome virus isolates of different virulence. Virus Genes 2017, 53, 249–258. [Google Scholar] [CrossRef]
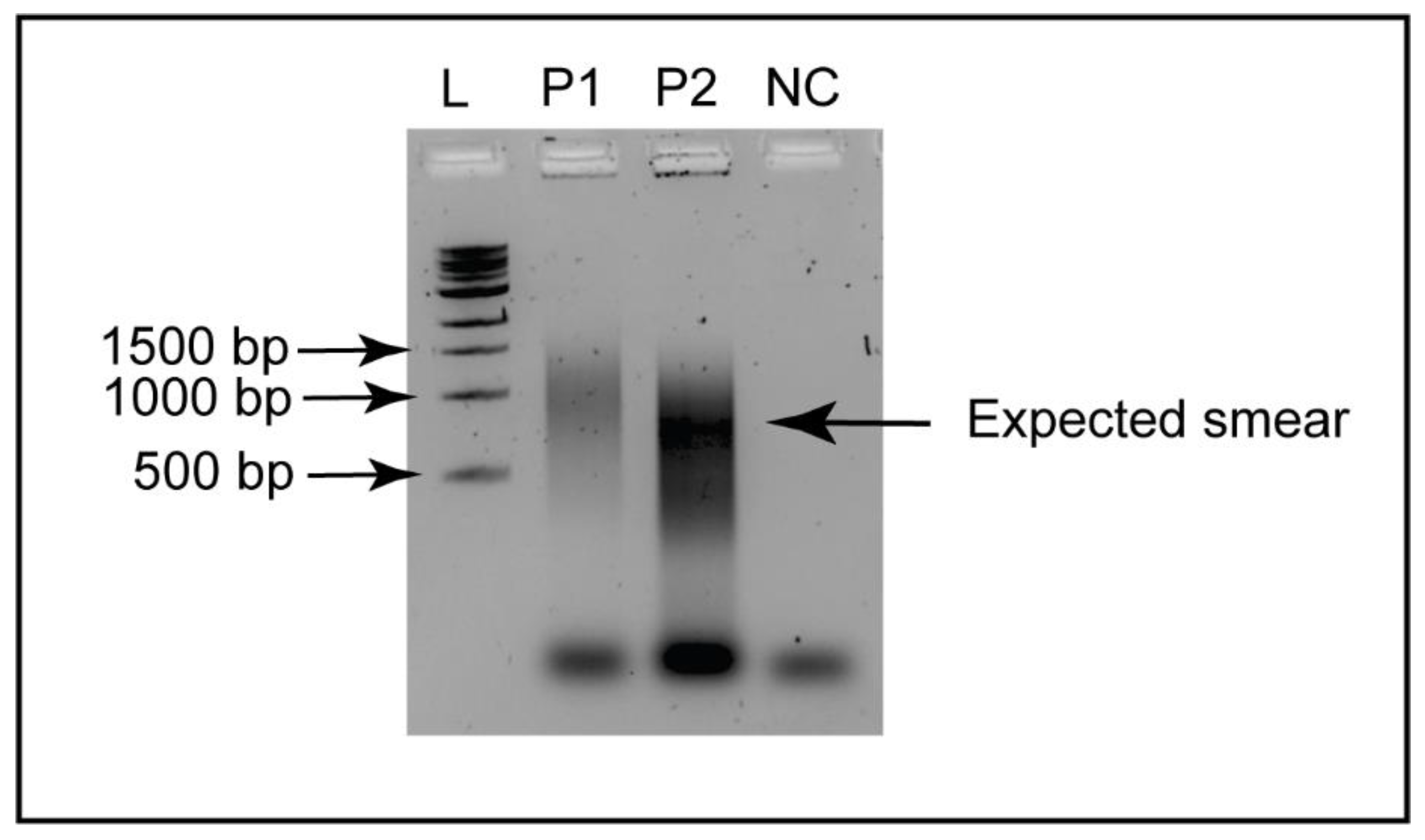

| WSSV–SA (GenBank Accession: OR636681) | Frame | Percent Protein Identity: GenBank Accession (Country) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Protein | Start (bp) | End (bp) | NC_003225.3 (China) | AP027290.1 (Japan) | AF369029.2 (Thailand) | AF440570.1 (Taiwan) | MF768985.1 (Australia) | KU216744.2 (Mexico) | MN840357.1 (USA) | |
| Immediate-early protein | 629 | 1303 | Forward | 100.0 | 99.6 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| dUTPase | 18,927 | 20,312 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 98.7 | 100.0 | 100.0 |
| Envelope protein VP53B | 20,510 | 23,416 | Forward | 100.0 | 100.0 | 99.9 | 100.0 | 100.0 | 100.0 | 100.0 |
| Ribonucleotide reductase large subunit | 57,563 | 60,109 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Ribonucleotide reductase small subunit | 65,794 | 67,035 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 99.8 | 100.0 |
| Envelope protein VP32 | 72,090 | 72,926 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope protein VP187 | 78,128 | 82,948 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 99.8 | 99.9 | 100.0 |
| DNA-binding protein VP15 | 83,105 | 83,290 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 90.9 | 100.0 | 100.0 |
| Envelope protein VP124 | 83,398 | 86,982 | Reverse | 100.0 | 99.9 | 100.0 | 99.9 | 99.2 | 99.9 | 100.0 |
| Capsid protein VP76 | 87,052 | 89,076 | Forward | 100.0 | 100.0 | 99.9 | 99.9 | 98.0 | 100.0 | 100.0 |
| E3 ligase | 89,095 | 91,629 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 99.9 | 99.8 | 100.0 |
| Envelope protein VP41A | 98,576 | 99,454 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | NE* | 100.0 | 100.0 |
| Envelope protein VP52A | 99,491 | 100,951 | Forward | 100.0 | 100.0 | 98.8 | 100.0 | NE* | 100.0 | 100.0 |
| Envelope protein VP41B | 101,003 | 101,905 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | NE* | 100.0 | 100.0 |
| Envelope protein VP52B | 110,480 | 111,634 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 99.7 | 100.0 |
| Envelope protein VP38 | 111,695 | 112,624 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Capsid protein VP136 | 118,623 | 122,282 | Forward | 100.0 | 99.9 | 99.9 | 100.0 | 99.8 | 99.8 | 100.0 |
| Capsid protein VP190 | 133,055 | 137,752 | Reverse | 100.0 | 99.9 | 99.9 | 98.7 | 99.4 | 99.8 | 100.0 |
| Envelope protein VP14 | 137,706 | 137,999 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | NE* |
| Putative TATA-box bind protein | 141,117 | 143,792 | Forward | 100.0 | 99.9 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope protein VP39A | 143,779 | 145,038 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 91.3 | 100.0 | 100.0 |
| Capsid protein VP51C | 145,063 | 146,463 | Forward | 100.0 | 100.0 | 99.8 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope protein VP26 | 147,361 | 147,975 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope protein VP16 | 152,071 | 152,424 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 95.1 | 100.0 | 100.0 |
| Envelope protein VP56 | 153,372 | 154,769 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 99.8 |
| Envelope protein VP90 | 155,603 | 158,173 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope protein VP11 | 160,758 | 162,059 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | NE* | 99.8 | 100.0 |
| Envelope protein VP39 | 162,082 | 162,933 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | NE* | 100.0 | 100.0 |
| Envelope protein VP31 | 162,937 | 163,722 | Reverse | 100.0 | 99.6 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Capsid protein VP664 | 177,046 | 195,279 | Forward | 100.0 | 100.0 | 99.9 | 99.9 | 99.8 | 100.0 | 100.0 |
| Envelope protein VP12B | 195,419 | 195,625 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 80.0 | 100.0 | 100.0 |
| Anti-apoptosis protein | 198,046 | 199,011 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 99.7 | 100.0 | 100.0 |
| Envelope protein VP19 | 208,379 | 208,744 | Reverse | 100.0 | 100.0 | 99.2 | 100.0 | 100.0 | 100.0 | 100.0 |
| Capsid protein VP60B | 208,882 | 210,516 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 99.1 | 100.0 | 100.0 |
| Envelope protein VP28 | 211,349 | 211,963 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 99.5 | 100.0 | 100.0 |
| Putative protein kinase | 212,058 | 214,250 | Reverse | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Envelope and capsid protein VP95 | 222,182 | 224,584 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 99.9 | 99.8 | 100.0 |
| Immediate-early protein | 240,901 | 244,632 | Reverse | 100.0 | 99.6 | 99.9 | 99.8 | 99.8 | 99.8 | 99.9 |
| DNA polymerase | 265,017 | 272,072 | Forward | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| Structural protein VP55 | 272,381 | 273,727 | Reverse | 100.0 | 100.0 | 100.0 | 99.8 | 99.8 | 99.8 | 100.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chauhan, R.P.; Fogel, R.; Limson, J. Nanopore MinION Sequencing Generates a White Spot Syndrome Virus Genome from a Pooled Cloacal Swab Sample of Domestic Chickens in South Africa. Microorganisms 2023, 11, 2802. https://doi.org/10.3390/microorganisms11112802
Chauhan RP, Fogel R, Limson J. Nanopore MinION Sequencing Generates a White Spot Syndrome Virus Genome from a Pooled Cloacal Swab Sample of Domestic Chickens in South Africa. Microorganisms. 2023; 11(11):2802. https://doi.org/10.3390/microorganisms11112802
Chicago/Turabian StyleChauhan, Ravendra P., Ronen Fogel, and Janice Limson. 2023. "Nanopore MinION Sequencing Generates a White Spot Syndrome Virus Genome from a Pooled Cloacal Swab Sample of Domestic Chickens in South Africa" Microorganisms 11, no. 11: 2802. https://doi.org/10.3390/microorganisms11112802







