Abstract
The purpose of the current study is to describe the prevalence of Pseudomonas aeruginosa (PA)-producing MβL among Brazilian isolates and the frequency of blaSPM-1 in MβL-PA-producing isolates. From January 2009 to August 2023, we carried out an investigation on this subject in the internet databases SciELO, PubMed, Science Direct, and LILACS. A total of 20 papers that met the eligibility requirements were chosen by comprehensive meta-analysis software v2.2 for data retrieval and analysis by one meta-analysis using a fixed-effects model for the two investigations. The prevalence of MβL-producing P. aeruginosa was 35.8% or 0.358 (95% CI = 0.324–0.393). The studies’ differences were significantly different from one another (x2 = 243.15; p < 0.001; I2 = 92.18%), so they were divided into subgroups based on Brazilian regions. There was indication of asymmetry in the meta-analyses’ publishing bias funnel plot; so, a meta-regression was conducted by the study’s publication year. According to the findings of Begg’s test, no discernible publishing bias was found. blaSPM-1 prevalence was estimated at 66.9% or 0.669 in MβL-PA isolates (95% CI = 0.593–0.738). The analysis of this one showed an average heterogeneity (x2 = 90.93; p < 0.001; I2 = 80.20%). According to the results of Begg’s test and a funnel plot, no discernible publishing bias was found. The research showed that MβL-P. aeruginosa and SPM-1 isolates were relatively common among individuals in Brazil. P. aeruginosa and other opportunistic bacteria are spreading quickly and causing severe infections, so efforts are needed to pinpoint risk factors, reservoirs, transmission pathways, and the origin of infection.
1. Introduction
The emergence and active spread of carbapenem-resistant Pseudomonas aeruginosa (CR-PA) strains are a major concern, especially in healthcare settings, as this class of broad-spectrum antibiotics is often used as a last resort to treat infections caused by multidrug-resistant (MDR) or extensive (XDR) isolates [1,2,3]. Cases of poor prognosis, complicated infections, and high mortality rates have been demonstrated to CR-PA infections, such as bloodstream infections (BSIs), ventilator-associated pneumonia (VAP), and severe burns in hospitalized patients, especially among those in intensive care units (ICUs) [4,5,6,7]. Data from the Antimicrobial Testing Leadership and Surveillance program demonstrate that the highest CR rates are in the Middle East, followed by South America, in which Brazil appears with 16.4% among 116 evaluated isolates [8]. Such increasing resistance trends have also been verified in other neighboring countries, including Bolivia and Peru [2].
Several resistance mechanisms lead to non-susceptibility phenotypes in P. aeruginosa strains, including the production of carbapenemases, changes in outer membrane permeability, and overexpression of efflux pumps that can expel the antibiotic from the cell. Such phenomena generally occur in selective pressure environments along with the ability of P. aeruginosa to form biofilms that can provide a protective environment for antibiotic-resistant cells, causing enhance in the transfer of antimicrobial resistance genes (ARGs) among pathogens [9]. As highlighted by separate reports, the production of carbapenemases efficiently prevents the activity of carbapenems and other β-lactams (except monobactams), and are often not inhibited by clinically available β-lactamase inhibitors, including clavulanic acid or tazobactam [10,11,12]. Additionally, bacteria co-expressing serine β-lactamases (SβLs) and metallo-β-lactamases (MβLs), two kinds of enzymes, are usually able to hydrolyze the clinically relevant monobactam, aztreonam [13].
The Ambler classification, which ranges from A to D, can be used to classify carbepenemases. Enzymes with serine in their active sites make up classes A, C, and D, whereas those with zinc in their active sites make up class B. The carbapenemases from K. pneumoniae (KPCs), imipenem-hydrolyzing β-lactamase (IMI), Guyana Extended Spectrumase Carbapenemase (GES), carbapenemase from Serratia fonticola, Serratia marcescens enzyme, and nonmetallo-carbapenemase-A are all examples of class A carbapenemases. Plasmids are where most of the genes for these class A enzymes are found. The genes encoding these enzymes can be found on plasmids, integrons, or even the bacterial chromosome itself in MβLs, which are class B organisms. Class C β-lactamases, like classes A and D, are based on serine and are encoded by genes that are often located on the bacterial chromosome. AmpC, a cephalosporinase, is the most important member of class C. Class D enzymes, sometimes referred to as oxacylinases (OXA), are typically produced by genes that are found on plasmids or the bacterial chromosome. Penicillins have excellent hydrolysis capabilities, whereas more recent generations of cephalosporins, such ceftazidime and cefepime, do not [14,15].
Among carbapenemases, the production of MβL has been associated with at least twelve (12) different types of MβLs, including Verona imipenemase—VIM, imipenemase—IMP, São Paulo MβL—SPM, German imipenemase—GIM, New Delhi MβL—NDM, Dutch imipemenase—DIM, Australian imipenemase—AIM, Seoul imipenemase—SIM, Kyorin University Hospital MβL—KHM, Serratia marcescens MβL—SMB, Tripoli MβL—TMB, and Florence imipenemase—FIM. Indeed, the mobile genetic element containing MβL in P. aeruginosa was originally reported in Japan at the end of the 1980s and has since been described in different parts of the world in association with several variants [16,17,18].
Regarding blaSPM-1, SPM-1 was initially thought to be contained in a plasmid, but further investigations on the gene for blaSPM-1 found that it was actually placed in movable genetic elements on the chromosome of the matching isolates of P. aeruginosa [19,20]. A broad spectrum MβL with a little bias for cephalosporins is SPM-1 [21]. The enzyme has an unusual structure since it lacks the conserved Asp84 and has a considerably shorter L3 loop. Instead, it has a 24-residue insertion in helix α3 that results in an extended helical segment, abutting the active region in a way reminiscent of B2 enzymes. Less than the deletion of the L3 loop in other B1 enzymes, the deletion of this extension of three reduces SPM-1’s catalytic activity by three- to thirty-two-fold [22,23].
Following a worldwide trend, MβL-producing P. aeruginosa isolates began to be described in Brazil in the early 2000s and are responsible for increased levels of resistance to carbapenems. They are similarly as observed in other locations, and CR-PA strains are often detected harboring blaIMP, blaVIM, and blaNDM variants in samples from various sources [24,25,26,27,28,29,30]. Contrastingly, the blaSPM-1 variant gene stands out for its endemic character and wide detection in different regions of the Brazilian territory, and from its first detection in 2002, it has been spread in the south region (in the State of Rio Grande do Sul—RS), the southeast region (in the State of São Paulo—SP), the northeast region (in the State of Sergipe—SE), the nidwest region, the State of Mato Grosso do Sul—MS, and the northern region (State of Pará) [19,31,32,33,34].
Despite the efforts in identifying and reporting cases of MβL-CR-PA in Brazil, a need for more robust studies is still noted in order to provide data that contemplate the large dimension of the Brazilian territory. Therefore, the present study aims to report, through a systematic review and meta-analysis support, the prevalence of MβL-CR-PA strains in Brazilian territory, as well as the prevalence of blaSPM-1 in comparison to other MβL gene variants. blaSPM-1, which is one of the MβL resistance genes distributed in gram-negative pathogens, was particularly chosen for the analysis in this article due to the fact that it is the most prevalent in Brazil and was originally isolated in SP [35].
2. Materials and Methods
2.1. Study Design
This is a systematic review and meta-analysis with the objective of investigating the aforementioned objective based on antimicrobial resistance data available in studies, including the detection of genetic mechanisms (MβLs) on P. aeruginosa isolated in the Brazilian territory. The present study followed the recommendations of the Preferred Report Items of a Systematic Review and Meta-Analyses (PRISMA) Statement 2020 [36]. This ratio-type meta-analysis was performed in this study for two correlated investigations: (I) to identify the prevalence of MβL-PA among Brazilian isolates and (II) to determine the prevalence of the blaSPM-1 variant in comparison to other MβL variants within the analyzed studies and isolates.
2.2. Search Strategy and Eligibility Criteria
To formulate the guiding question of the present systematic review, the POT strategy was used, and it consisted of the following question: “What is the prevalence of MβL-PA among Brazilian samples?” The anagram for its formation was composed of “P” for problem (MβL-CR-PA), “O” for outcome (frequency of MβL-PA in Brazil), and T for study type (original studies). The keywords used for the database search were Medical Subject Headings (MeSH): (((“beta-Lactamases/analysis” [Mesh]) OR (“beta-Lactamases/genetics” [Mesh])) AND “Brazil” [Mesh]) AND “Pseudomonas aeruginosa” [Mesh] [37].
The databases investigated for the search were PUBMED (https://pubmed.ncbi.nlm.nih.gov/ (accessed on 11 August 2023)), SciELO (https://www.scielo.br/ (accessed on 11 August 2023)), Science Direct (https://www.sciencedirect.com/ (accessed on 11 August 2023)), and LILACS (https://lilacs.bvsalud.org/ (accessed on 11 August 2023)), as those databases in the Latin American have weight and count most Brazilian publications. Primary studies published in Portuguese, English, or Spanish, within the time frame from January 2009 to August 2023, and of analytical case–control, cohort, and cross-sectional types, were eligible [38]. Brief/short communications, letters to the editor, editorials, articles unavailable in their complete form, articles outside the stipulated time cut, and the aforementioned inclusion criteria, were excluded.
2.3. Data Extraction and Methodological Quality Assessment
The data were collected in all databases on 11 August 2023. The database search, collection, investigation, tabulation, and extraction of the data were conducted by two authors independently (PASS and MJAS). Any disagreement between the analyses was resolved with help from a third researcher (YCR). The data extracted from the articles were authors, year of publication, title, sourced database(s), methodology, objective(s), sample type, Brazilian city/region, period in which the P. aeruginosa isolates were collected, MβL-PA frequency results, and if applicable, frequency of blaSPM-1 among MβL-PA.
The quality assessment was performed by two investigators (PASS and MJAS) using the Joanna Briggs Institute’s (JBI) Critical Appraisal Checklist for Analytical Cross-Sectional Studies (0–8), the JBI Checklist for Case–Control Studies (0–10), and the JBI Checklist for Cohort Studies (0–11) [39]. Only when the conditioned response was “Yes” were the scores for completing the checklist questions taken into account [40]. A third researcher (YCR) opined in case of disagreement.
2.4. Statistical Analysis
The Comprehensive Meta-Analyses—CMA program, version 2.2 (Biostat, Englewood, NJ, USA), was used on a computer to perform the statistical analyses of the meta-analysis for the two investigations. The fixed effects model estimated the combined frequency of P. aeruginosa with 95% confidence intervals (95% CI). A subgroup analysis was performed to determine the frequency of MβL-PA isolates according to Brazilian regions (northern—composed of seven states, northeast—composed of nine states, midwest—composed of three states, the Federal District, south—composed of four states, and southeast—composed of four states). The Cochrane Q-test and I-squared (I2) measure were used to determine the statistical difference groups (p < 0.05 was considered statistically significant). Begg’s rank correlation test and a funnel plot were used to examine the potential for publication bias (p < 0.05 was considered statistically significant). Sensitivity analysis, meta-regression, and subgroup analysis based on study location were used to assess potential causes of variability, where applicable [41].
3. Results
3.1. Literature Search
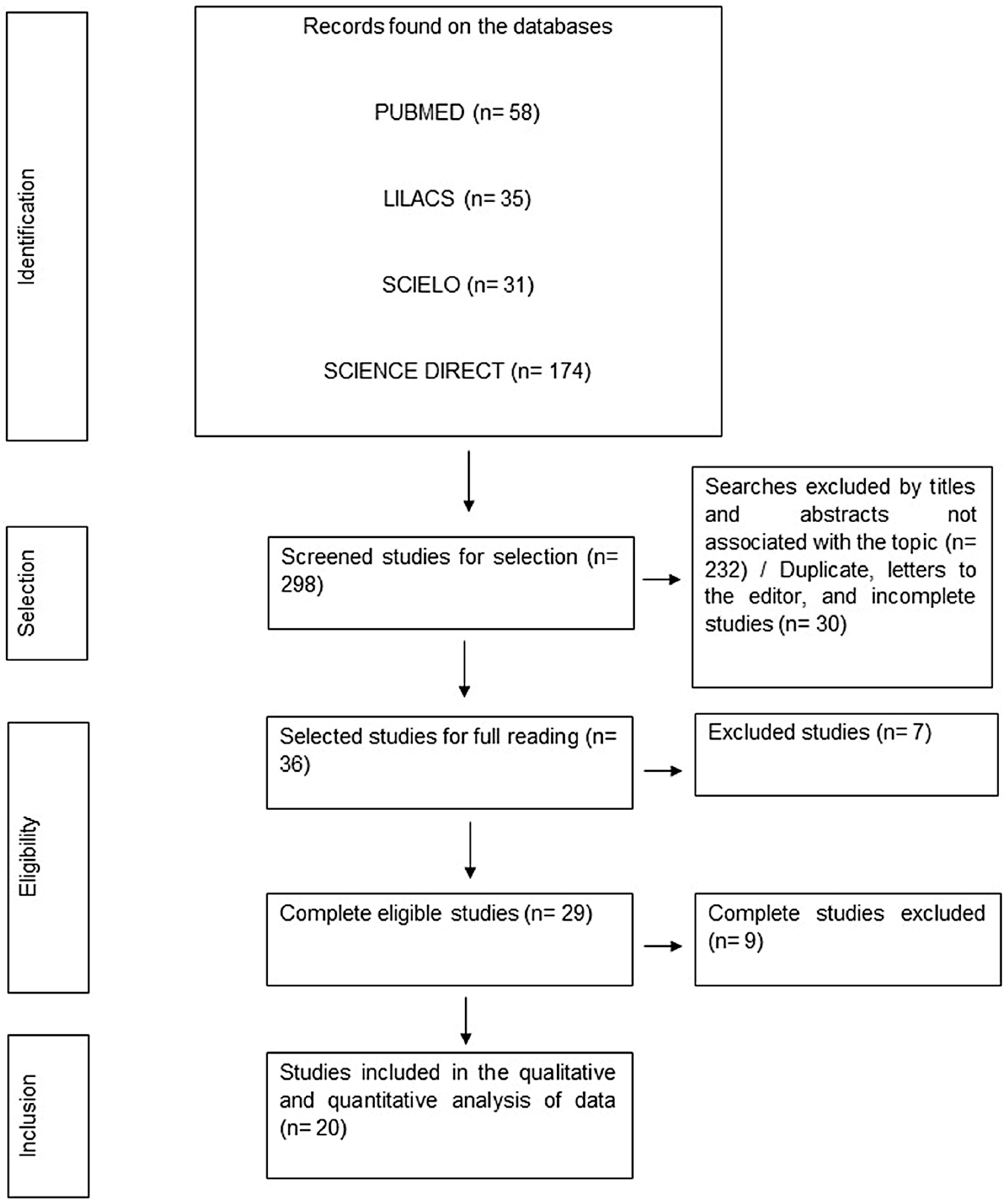
A total of 298 studies were found within the selected databases. After initial screening and application of eligibility and inclusion criteria, 173 were out of the scope proposed by this review, 24 studies were in languages other than those agreed upon in the methodology, 30 studies only had the abstract available, 14 studies were unavailable for full reading, 20 were duplicate studies, 1 was a letter to the editor, and 16 studies through full reading were excluded. The final sample and its stages until final inclusion were described in Figure 1.
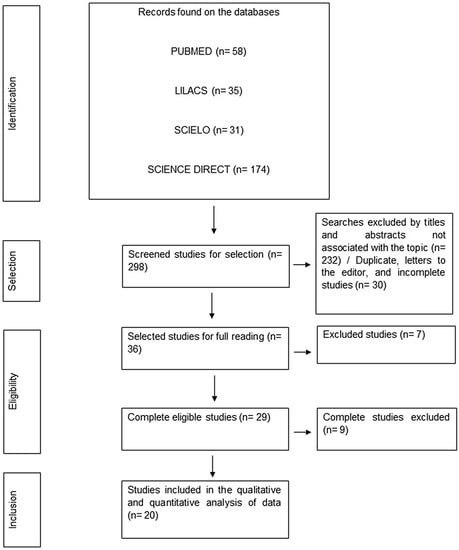
Figure 1.
Flowchart of data collection and screening stages. Belém-PA, Brazil (2023).
3.2. Characterization of Included Studies
A total of 20 studies were included in the final analysis. All were presented in English, found on the PUBMED database, and had who were authors affiliated with Brazilian teaching and research institutions. Most of the studies were characterized as a cohort type (n = 14; 70%), with MβL-PA isolates recovered in a clinical/hospital setting (n = 17; 85%), followed by animal–environmental settings (15%) and a high detection frequency of the blaSPM-1 gene among isolates. The studies were evaluated by the JBI checklists considering each study design investigated and, in general, they showed high methodological quality (Table 1).

Table 1.
Characteristics of the studies included in this systematic review.
3.3. Results and Publication Bias of Meta-Analysis of Proportion of MβL-PA
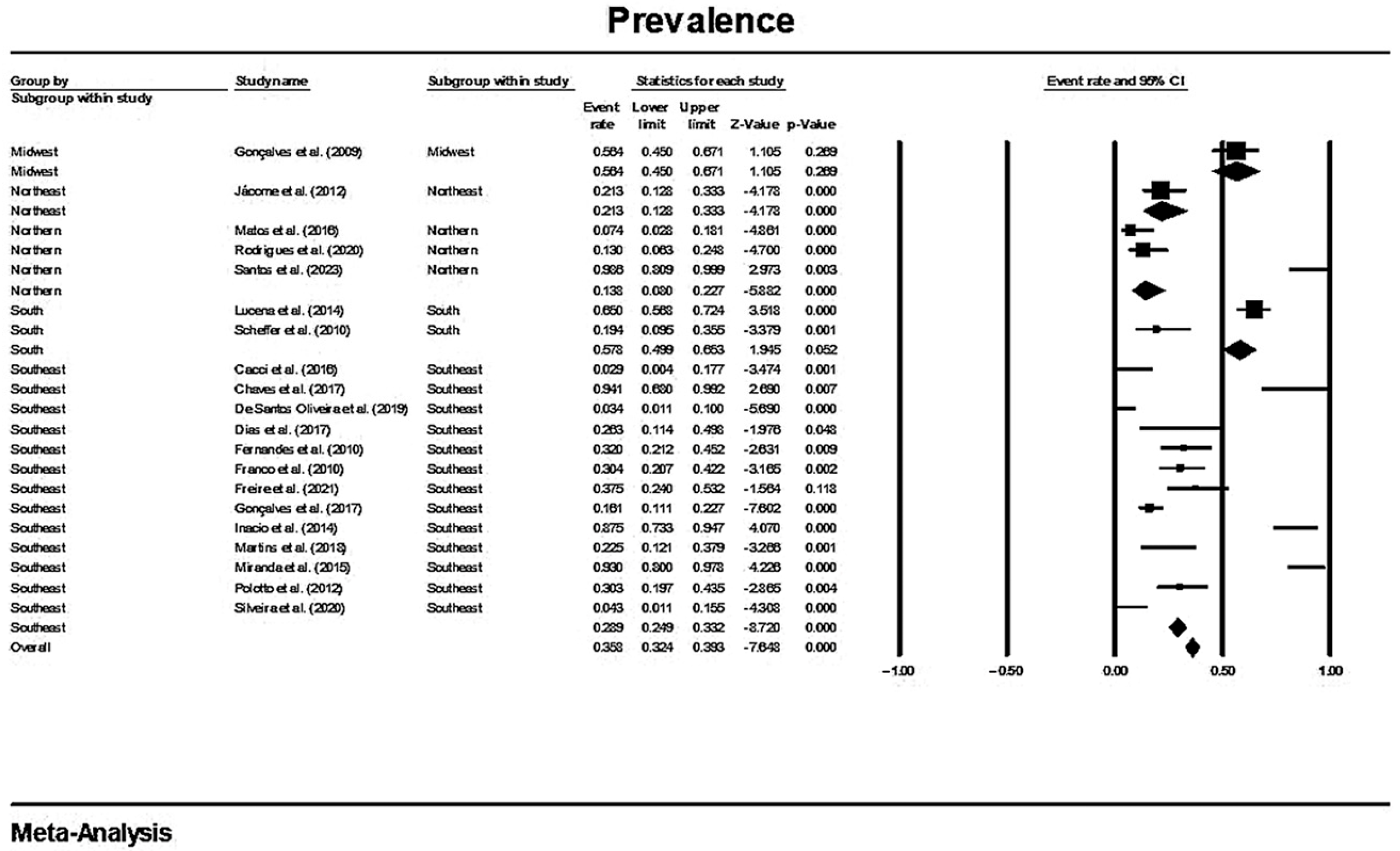
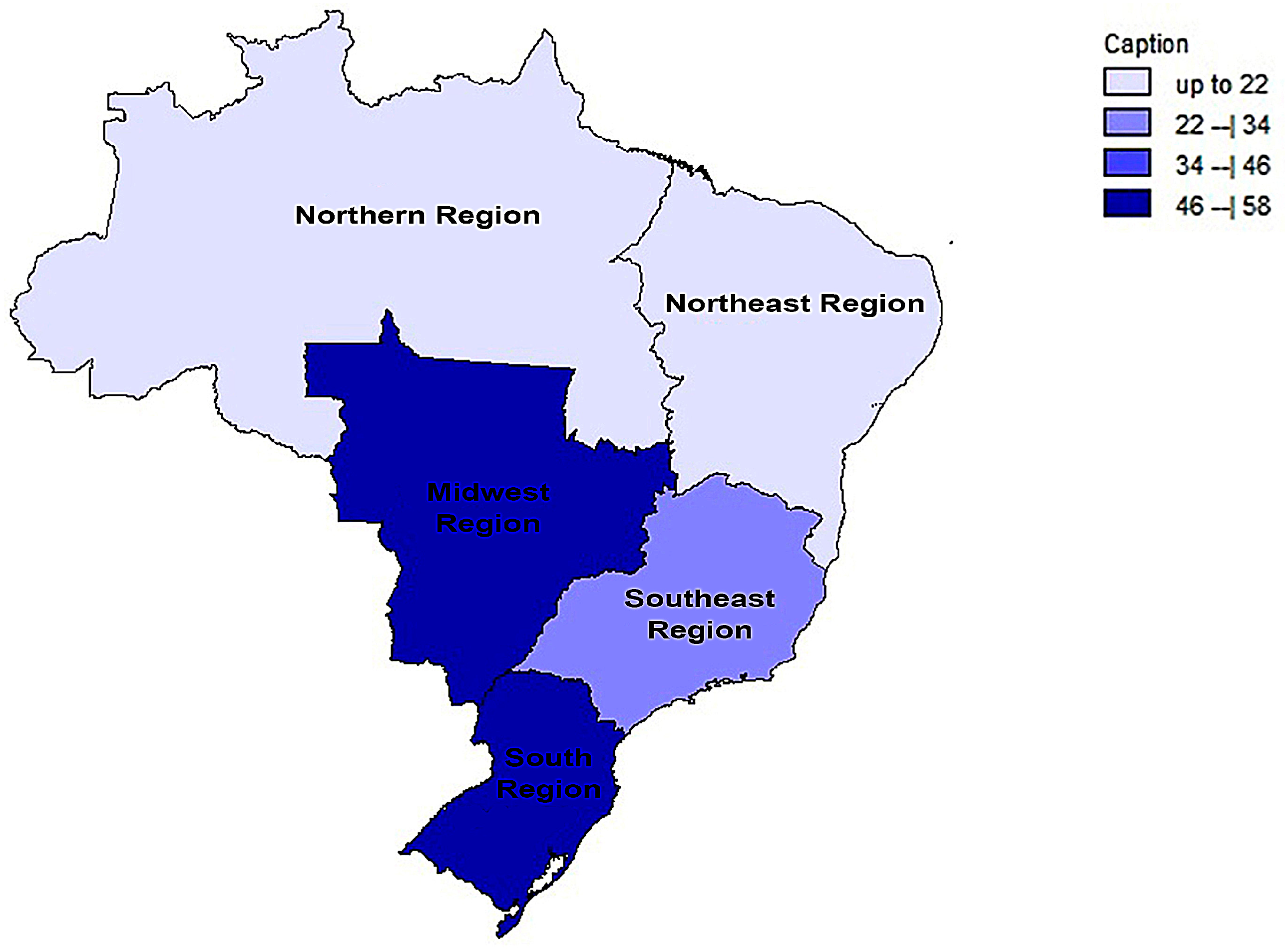
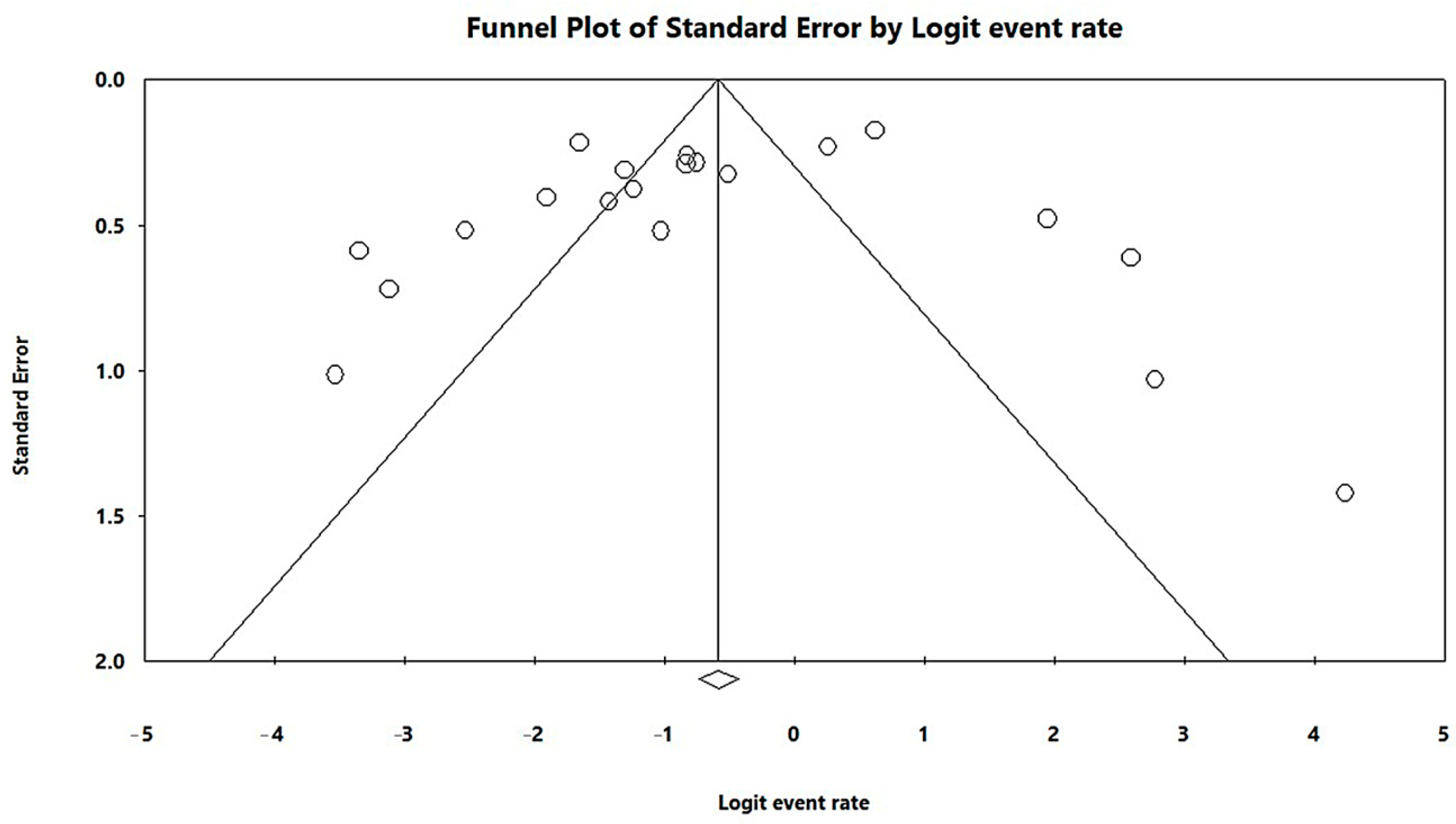
In a generalized analysis of 20 studies, the prevalence rate of MβL-PA isolates was 35.8% or 0.358 (95% CI = 0.324–0.393). The studies’ differences were significantly different from one another (x2 = 243.15; p < 0.001; I2 = 92.18%), so they were divided into subgroups based on Brazilian geographic regions (Figure 2). This analysis demonstrated the northern region, with a frequency of 13.8% or 0.138 (95% CI = 0.080–0.227); the northeast region, with 26.4% or 0.264 (95% CI = 0.167–0.393); the midwest region (although only one study was included in the analysis), with 56.4% (95% CI = 0.450–0.671); the southeast region, with 28.9% or 0.289 (95% CI = 0.249–0.332); and finally the south region (although only two studies were included in the analysis), revealing the highest prevalence of MβL-producing P. aeruginosa, with 57.8% or 0.578 (95% CI = 0.499–0.653). A thematic map of these estimated percentual values in Brazilian regions was characterized in Figure 3. Despite the asymmetric distribution of the graph in the funnel plot (Figure 4), as it is a meta-analysis of the proportion type, the Begg test was performed. According to the findings of Begg’s test, no discernible publishing bias was found (z = 0.42; p = 0.67).
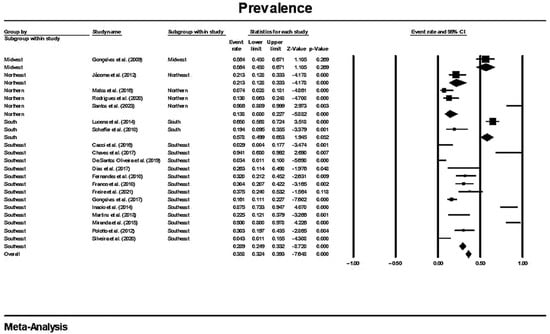
Figure 2.
Forest plot of the prevalence of MβL-PA isolates by sampling PA isolates grouped by Brazilian regional subgroups. The size of each square, which corresponds to the weight of the related research in the meta-analysis, represents the OR of each study on the map. The 95% confidence intervals (CIs) for each study’s OR are shown as horizontal lines. The bold figures indicate the overall OR and the 95% CI, as well as the total frequency of cases and controls [31,32,33,34,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57].
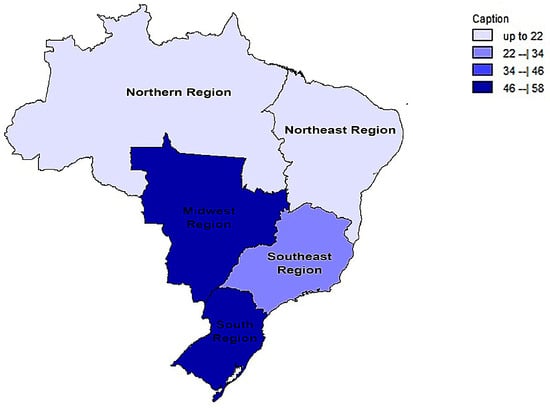
Figure 3.
Thematic map of the estimated percentual value (%) of MβL-producing P. aeruginosa strains in Brazilian national distribution over the years of this review based on official regional groupings.
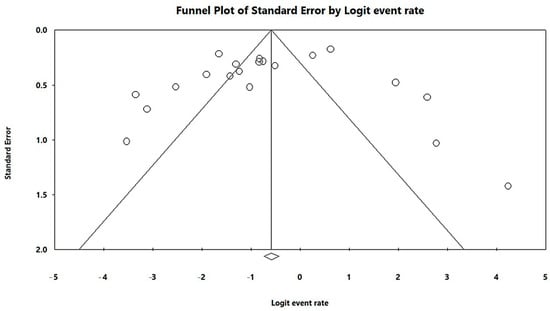
Figure 4.
Funnel plot of the heterogeneity assessment among the studies included in the meta-analysis. The included published studies are represented by circles, which should be evenly spaced around the overall effect to resemble an inverted funnel. Studies that are more precise are in the narrowest region of the funnel and are closest to the real value. The standard error, which is represented on the graph’s Y-axis as a measure of dispersion, is affected by the study’s sample size. The bigger this value, the more inaccurate the study is. The center line of the graph, which is pointed at the X-axis by the diamond, represents the outcome of the effect measure examined in the meta-analysis. The lines that make up the funnel’s outline line up with 95% CI.
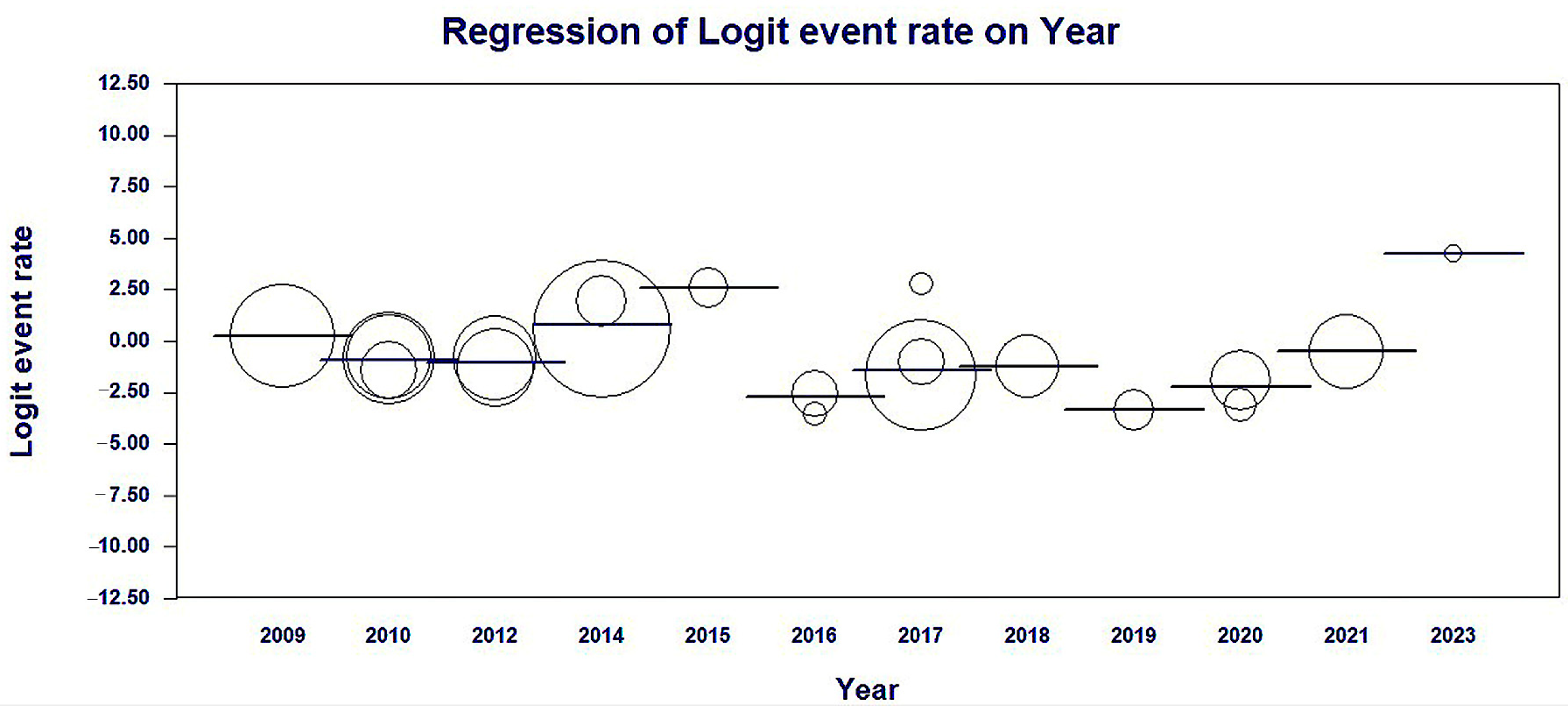
3.4. Meta-Regression of the Studies Included by Year of Publication
Meta-regression results by year of publication of the studies showed a significant association with heterogeneity in findings of the average prevalence rates of MβL-PA among Brazilian isolates in a fixed-effects model; coefficients −0.2574 (95% CI = −0.199–0.7138, p < 0.0001) (Table 2 and Figure 5).

Table 2.
Main results for model 1, fixed effect, Z-distribution, and logit event rate.
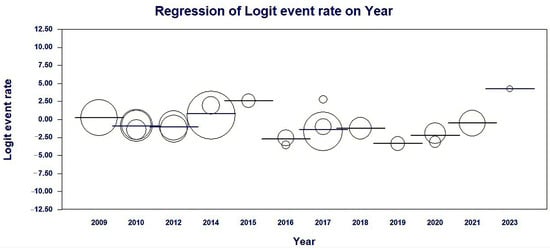
Figure 5.
Scatterplot of the meta-regression of the logit event rate (MβL-PA/PA) by study publication year covariate. The size of the bubble is inversely related to the variance of the study. The solid line represents the linear regression (continent as the meta-independent variable).
3.5. Results and Publication Bias of the Meta-Analysis of blaSPM-1 within MβL-PA Isolates
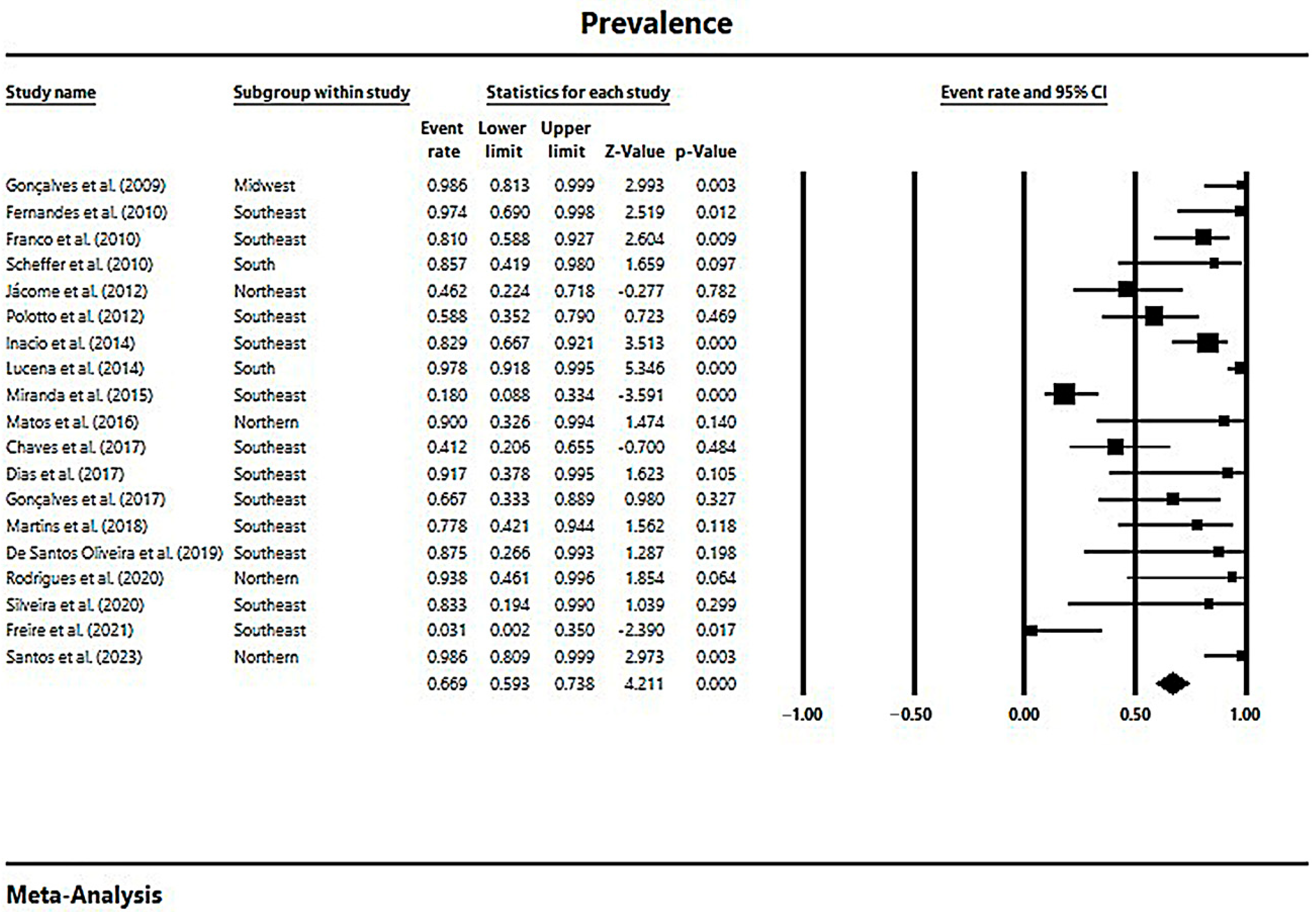
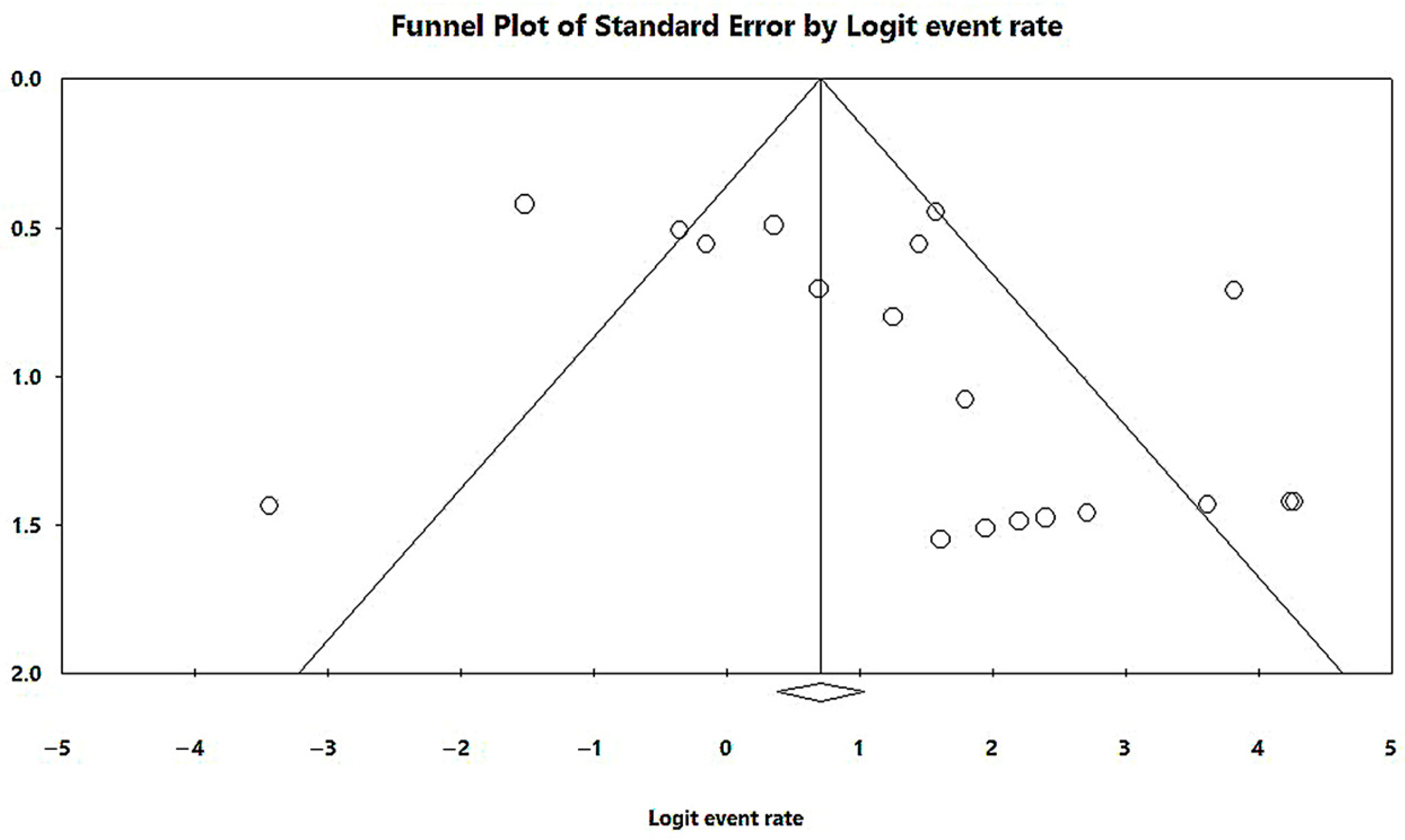
In a generalized analysis of 19 studies, the prevalence rate of MβL-PA isolates in the presence of the blaSPM-1 was 66.9%, ranging from 59.3% to 73.8%, with a 95% CI (Figure 6). Only one study was excluded in this analysis, as only one MβL-PA isolate was found, and the sample size for comparison has to be higher than or equal to two [48]. The heterogeneity of the results among the included studies was considered average (x2 = 90.93; p < 0.001; I2 = 80.20%). The distribution of the studies in the graph in Figure 6 was symmetrical, and Begg’s test reported non-significance (z = 0.48; p = 0.62), demonstrating no publication bias for this meta-analysis (Figure 7).
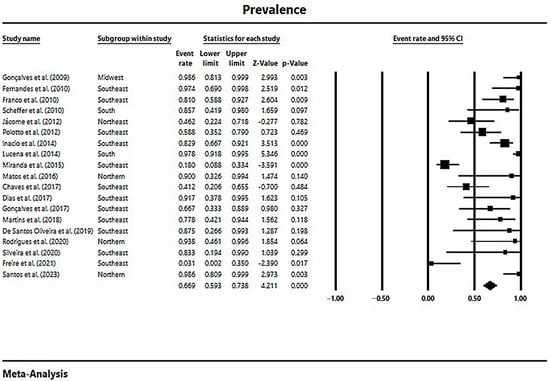
Figure 6.
Forest plot of the prevalence of blaSPM-1 among MβL-PA isolates in the Brazilian territory. The size of each square, which corresponds to the weight of the related research in the meta-analysis, represents the OR of each study on the map. The 95% confidence intervals (CIs) for each study’s OR are shown as horizontal lines. The bold figures indicate the overall OR and the 95% CI, as well as the total frequency of cases and controls [31,32,33,34,42,43,44,45,46,47,49,50,51,52,53,54,55,56,57].
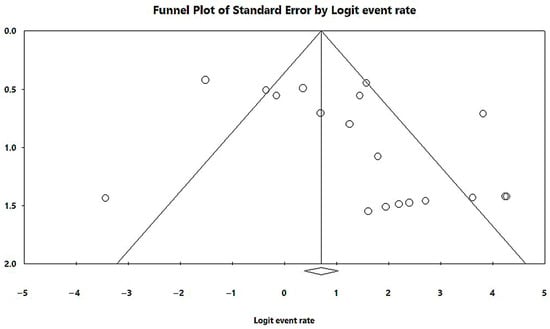
Figure 7.
Funnel plot of the heterogeneity assessment among the studies included for the analysis of the blaSPM-1. The included published studies are represented by circles, which should be evenly spaced around the overall effect to resemble an inverted funnel. Studies that are more precise are in the narrowest region of the funnel and are closest to the real value. The standard error, which is rep-resented on the graph’s Y-axis as a measure of dispersion, is affected by the study’s sample size. The bigger this value, the more inaccurate the study is. The center line of the graph, which is pointed at the X-axis by the diamond, represents the outcome of the effect measure examined in the meta-analysis. The lines that make up the funnel’s outline line up with 95% CI.
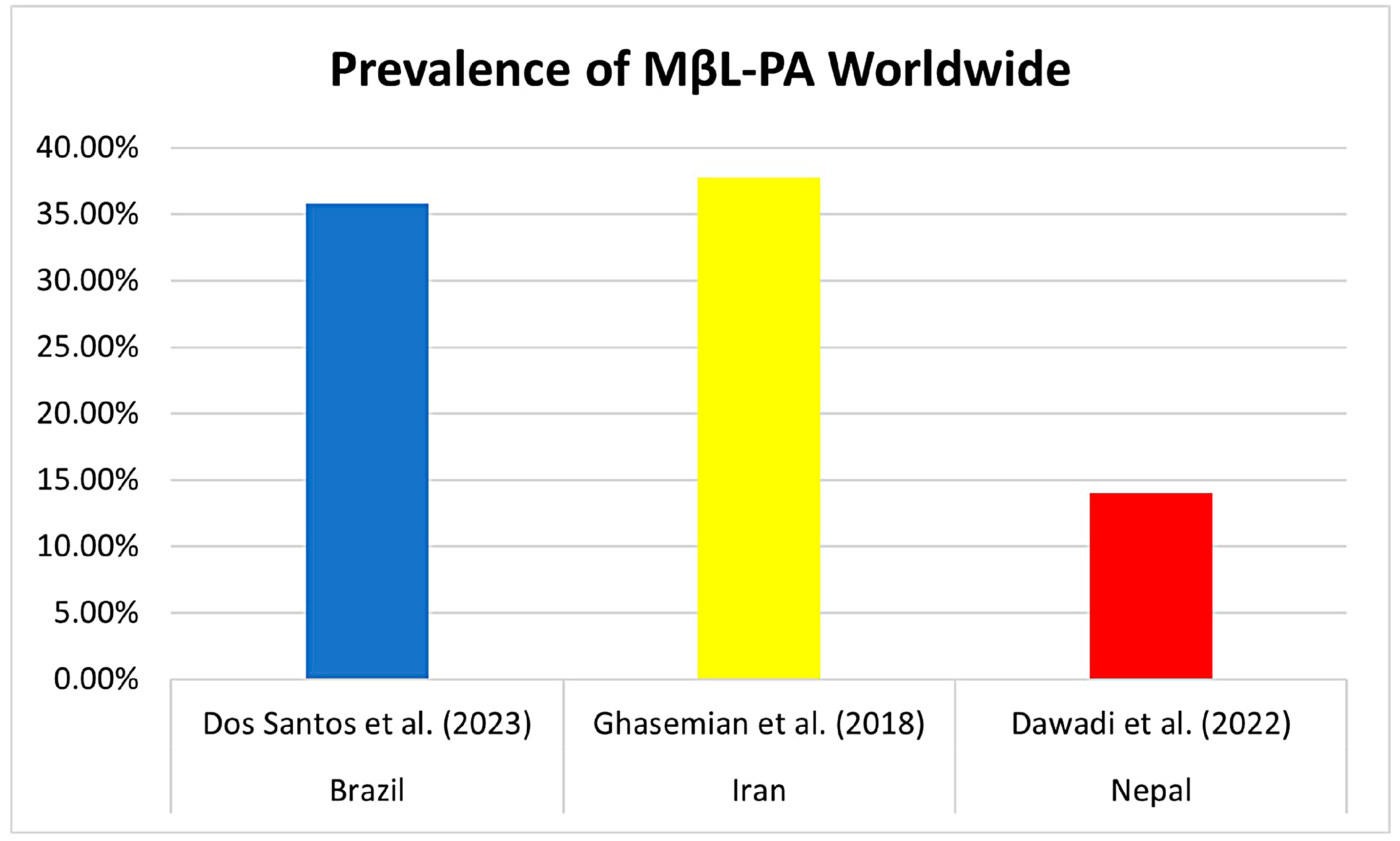
3.6. Comparison between the Results of this Brazilian Meta-Analysis and Prevalence Worldwide
Previous reviews have already reported the prevalence of MβL-PA in hospital isolates globally, especially in Asia. Ghasemian et al. (2018) in Iran composed a review of 36 studies in 2018 and reported an incidence of 37.72% MβL-PA, ranging from 16.68% to 100%, with no reports of isolates harboring blaNDM-1 and blaSPM-1 [58]. Another meta-analysis by Dawadi et al. (2022), conducted in Nepal, found a frequency of 14% MβL-PA (95% CI = 0.10–0.19) for data pooled from studies published until 2021 in Nepal, and MDR-PA isolates were about 42% in Nepal [59] (Figure 8).
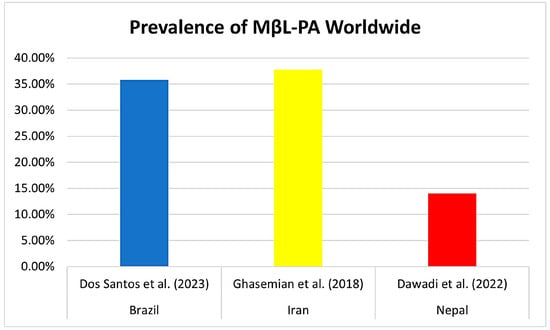
Figure 8.
Comparison of the prevalence (%) of MβL-PA isolates already reported in the literature in countries around the world. The first study by Dos Santos et al. (2023) cited in the graphic is about the results of this meta-analysis [58,59].
4. Discussion
Although they are known as “carbapenemases”, many of these enzymes recognize nearly all hydrolyzable β-lactams, and most are resistant to inhibition by all commercially viable β-lactamase inhibitors. With the exception of the SPM type enzyme, most MβL genes reside in various integron cassette compositions attached to mobile elements, which is a condition that facilitates their dissemination among different bacterial species through horizontal gene transfer [60,61]. This scenario is in association with the poor consequences of unrestricted antimicrobial usage and heterogeny in the social–economic context worldwide. CR-PA has proved to be a public and healthcare emergency [62,63,64,65,66]. Hence, the present study aims to provide robust data on the prevalence of MβL-PA isolates in Brazil, as well as the prevalence of the endemic blaSPM-1 resistance gene.
In the global antimicrobial resistance epidemiological scenario, CR-PA has been reported to contain a wide variety of carbapenemases. In Latin America, the Arabian Peninsula, and the United States of America, this includes KPC, GES, IMP, VIM, NDM, and SPM [67,68,69,70]. Approximately 49.4% of CR-PA from Spain tested positive for MβL, in which the blaVIM-2 was detected in almost all isolates. In addition, 75% tested positive for integron class 1 [71]. Class 1 and 2 integrons were found among 60% of the carbapenem-resistant isolates in Colombia that were VIM-positive but were all IMP- and NDM-negative [72]. Germany, Switzerland, Brazil, and Italy reported GIM-1-, SPM-1-, and FIM-1-producing strains, respectively [73,74,75,76]. The predominant MβL-encoding gene in the Middle East and the majority of other Asian nations is the blaVIM and its variants. Recently, the emergence of the NDM-1-producing P. aeruginosa is quite concerning because it first appeared in nations other than India [77].
Brazil also has proven to be an important ally in the detection of MDR/XDR CR-MβL-PA strains, as highlighted by data from the present study, in which IMP-, VIM-, and SPM-producing isolates have been mostly detected [17,78,79,80]. Worryingly, there is a recent report of CR-PA harboring the blaNDM-1 gene along with other MβL [29]. Indeed, the occurrence of isolates with the co-production of MBLs has been verified, as reported in some studies included in this meta-analysis [31,47]. As opposed to to other MβL found in lower frequency, the endemic feature is key to the blaSPM-1 gene, as there are scarce reposts outside Brazil, Europe [73], Iran [81,82], the UK [83], Chile [84], and Egypt [85]. This fact must be related to the clonal dissemination of MβL-PA in association outbreaks by the ST277, as described in several studies in different Brazilian regions, and the chromosomal nature of this variant, which may hinder its dissemination [42,50,56,86].
This is a pioneer review and meta-analysis study involving the prevalence of MβL-PA isolates in Brazil and providing significant data on the endemic blaSPM-1 gene frequency. Previous data from the two studies cited in Section 3.6 of this article highlight a similar profile of results presented in this meta-analysis based on Brazilian territory isolates. This is an emerging problem worldwide [58,59].
Other meta-analyses present in the literature, such as those conducted by Ul Ain et al. (2020) [87] and Osei Sekyere et al. (2021) [88], performed a broader and less specific analysis of P. aeruginosa in Pakistan by compiling a brief description and, for the first time, a report of the resistant carbepenemases rate (RC) for different gram-negative bacteria, including P. aeruginosa. In the first study, the prevalence of MβLs from different geographical regions of Pakistan showed concerning high rates of RCs and incidences of MβLs; while the second evaluated the global distribution of the rate of carbapenem resistance in gram-negative bacterial strains in pregnant hosts and children in comparison with polymyxin resistance and death rate in the countries, in which Brazil had a CR rate of approximately 54.5% and death rate higher than 10%. Vaez et al. (2018), in a meta-analysis in Iran in the period up to 2018, determined a prevalence of about 32.4% of MDR-PA, and the most reported MβL genes were blaVIM and blaIMP, with frequencies of 19% (95% CI: 0.15–0.23) and 11% (95% CI: 0.08–0.14) [89], respectively.
Nationally, only one meta-analysis published by our research group includes isolates from the Brazilian population, which under a time cut between 2006 and 2016, investigated the mortality rate related to MDR-PA and SPM-1-producing strains. The results demonstrated a higher mortality rate among MDR-PA-infected patients (44.6%, 363/813) than those with a non-MDR-PA infection (24.8%, 593/2. 388) (OR = 2.39, 95% CI = 1.70–3.36, p < 0.000017) [7].
Our study demonstrated that most of the samples are from clinical/hospital settings, in which CR-PA is one of the main pathogens involved in infections, colonization, and invading things like respiratory treatment machines, disinfectants, sinks, distilled water, central venous catheters, urine catheters, and even the hands of medical staff [90]. Humans also expel resistant bacteria into the effluent, which may spread to other environmental compartments. The selection of resistant bacteria, such as those related to some Brazilian regions of this study, is influenced by the selective pressure applied by antibiotics often found in hospital effluent [91].
Depending on the type of geographic region and the type of methodology applied in the primary study, the results tend to be different. This is due to the socioeconomic, environmental, and zooanthropological characteristics of the investigated region, in addition to the type of sample analyzed, such as the prevalence of this etiological agent in surgical and respiratory wounds or in urinary and surgical infections (where there is usually a higher occurrence) [92]. In this case, this meta-analysis shows that there is a large number of studies covering the southeast region that may come from the suggestion of several factors that still need to be better investigated together, such as the hospital environments themselves and the outbreaks of this pathogen, which present a relative frequency of MβL-PA that still constitutes a relevant public health problem. The south region of Brazil has been identified as the region with the highest incidence rate of MΒL-producing strains. In contrast, numerous other evaluated studies have indicated that the rate of MβL-P. aeruginosa varies. In the northern region, our research group conducted molecular epidemiology studies providing significant data on SPM-1 clinical impact and the role of high-risk clone ST277 on the spread of this variant in the region [32,49]. This heterogeneity may be related to several elements, including antibiotic availability and use, variations between nosocomial surveys, and techniques to identify MβL producers [59].
The limitations of this present systematic review and meta-analysis conducted in Brazil lie in terms of the search methodology applied (due to the use of specific keywords for the topic) and the inclusion of studies focusing on evaluating MβL detection by molecular methods, even though some include data on phenotypical detection. Also, in this meta-analysis, only original articles were included; however, it was observed that there are articles, mainly in a short communication format, which were excluded, which reported a high frequency of MβL and SPM-1 gene-producing strains in the Brazilian regions, which demonstrates that the estimated prevalence of MβL and SPM-1 may still be underestimated in their epidemiological natures [44,93,94,95]. Even though other sources may have been excluded from this review due to their in-depth profile or lack of high-quality evidence, this article denotes a good standard of methodological and scientific evidence and provides a basis for one-health strategies and new studies on this topic.
5. Conclusions
MβL production by P. aeruginosa has been proven as complicating factor, not only for the worsening of infections but also for the successful treatment of infected patients, thus making it a public health problem, especially in underdeveloped and emerging countries, such as Brazil, where hospitals lack better structures, the health system is defective, and available resources are limited, especially for the poor population. According to our research results, the prevalence of MβL-PA is alarmingly high in most Brazilian hospitals and clinical settings, along with the detection in animal–environmental interfaces, similar to other places in the world, such as Iran, making it necessary to develop effective dissemination control strategies. It was also possible to verify, through data analysis, that the high prevalence of the resistance gene blaSPM-1 compares with the high dissemination rate of other MΒL genes already detected in Brazil. Finally, we highlight the need for new studies for the better characterization of CR-MβL-PA in the distinct Brazilian regions with a focus on molecular epidemiology-based studies supported by new genomics approaches and technology, which aim to investigate outbreaks in endemic regions and the presence of such strains in non-clinical settings. New insights into the resistome features may be provided.
Author Contributions
Conceptualization, P.A.S.D.S. and Y.C.R.; methodology, M.J.A.S.; software and resources, M.J.A.S.; formal analysis, M.J.A.S., M.I.M.G. and P.A.S.D.S.; data curation, P.A.S.D.S., M.I.M.G. and M.J.A.S.; investigation, M.J.A.S. and P.A.S.D.S.; validation, P.A.S.D.S., M.J.A.S. and Y.C.R., writing—original draft and editing, P.A.S.D.S., M.J.A.S. and Y.C.R.; writing—review and editing, P.A.S.D.S., M.J.A.S., A.J.P.G.Q., D.M.B., K.V.B.L., L.N.G.C.L. and P.D.L.D.L.; and Y.C.R.; resources, A.J.P.G.Q., D.M.B., K.V.B.L. and L.N.G.C.L., supervision, K.V.B.L. and D.M.B.; project administration, K.V.B.L., D.M.B. and Y.C.R. All authors have read and agreed to the published version of the manuscript.
Funding
The APC was funded by Evandro Chaga Institute (IEC/PA). Yan Corrêa Rodrigues scholarship is funded by PDPG—Pós-Doutorado Estratégico (PDPG-POSDOC), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Edital 16/2022). Pabllo Antonny Silva Dos Santos is funded by CAPES.
Data Availability Statement
The original contributions of the study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mustafai, M.M.; Hafeez, M.; Munawar, S.; Basha, S.; Rabaan, A.A.; Halwani, M.A.; Alawfi, A.; Alshengeti, A.; Najim, M.A.; Alwarthan, S.; et al. Prevalence of Carbapenemase and Extended-Spectrum β-Lactamase Producing Enterobacteriaceae: A Cross-Sectional Study. Antibiotics 2023, 12, 148. [Google Scholar] [CrossRef] [PubMed]
- Allel, K.; Day, L.; Hamilton, A.; Lin, L.; Furuya-Kanamori, L.; Moore, C.E.; Van Boeckel, T.; Laxminarayan, R.; Yakob, L. Global Antimicrobial-Resistance Drivers: An Ecological Country-Level Study at the Human-Animal Interface. Lancet Planet. Health 2023, 7, e291–e303. [Google Scholar] [CrossRef] [PubMed]
- Büchler, A.C.; Shahab, S.N.; Severin, J.A.; Vos, M.C.; Voor in ‘t holt, A.F. Outbreak Investigations after Identifying Carbapenem-Resistant Pseudomonas aeruginosa: A Systematic Review. Antimicrob. Resist. Infect. Control 2023, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Li, L.; Li, M.; Liang, H.; He, Y.; Li, S. Risk Factors and Outcomes of Patients with Carbapenem-Resistant Pseudomonas aeruginosa Bloodstream Infection. Infect. Drug Resist. 2023, 16, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Falcone, M.; Tiseo, G.; Carbonara, S.; Marino, A.; Di Caprio, G.; Carretta, A.; Mularoni, A.; Mariani, M.F.; Maraolo, A.E.; Scotto, R.; et al. Mortality Attributable to Bloodstream Infections Caused by Different Carbapenem-Resistant Gram-Negative Bacilli: Results From a Nationwide Study in Italy (ALARICO Network). Clin. Infect. Dis. 2023, 76, 2059–2069. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wei, X.; Xu, G.; Zhang, X.; Wang, X. Carbapenem-Resistant Pseudomonas aeruginosa Infections in Critically Ill Children: Prevalence, Risk Factors, and Impact on Outcome in a Large Tertiary Pediatric Hospital of China. Front. Public Health 2023, 11, 1088262. [Google Scholar] [CrossRef] [PubMed]
- de Matos, E.C.O.; Andriolo, R.B.; Rodrigues, Y.C.; de Lima, P.D.L.; Carneiro, I.C.d.R.S.; Lima, K.V.B. Mortality in Patients with Multidrug-Resistant Pseudomonas aeruginosa Infections: A Meta-Analysis. Rev. Soc. Bras. Med. Trop. 2018, 51, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Yoon, E.-J.; Jeong, S.H. Mobile Carbapenemase Genes in Pseudomonas aeruginosa. Front. Microbiol. 2021, 12, 614058. [Google Scholar] [CrossRef]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.-J.; Cheng, Z. Antibiotic Resistance in Pseudomonas aeruginosa: Mechanisms and Alternative Therapeutic Strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef]
- Meletis, G. Carbapenem Resistance: Overview of the Problem and Future Perspectives. Ther. Adv. Infect. Dis. 2016, 3, 15–21. [Google Scholar] [CrossRef]
- Codjoe, F.S.; Donkor, E.S. Carbapenem Resistance: A Review. Med. Sci. 2017, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Aurilio, C.; Sansone, P.; Barbarisi, M.; Pota, V.; Giaccari, L.G.; Coppolino, F.; Barbarisi, A.; Passavanti, M.B.; Pace, M.C. Mechanisms of Action of Carbapenem Resistance. Antibiotics 2022, 11, 421. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Poirel, L.; Walsh, T.R.; Livermore, D.M. The Emerging NDM Carbapenemases. Trends Microbiol. 2011, 19, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Elshamy, A.A.; Aboshanab, K.M. A Review on Bacterial Resistance to Carbapenems: Epidemiology, Detection and Treatment Options. Future Sci. OA 2020, 6, FSO438. [Google Scholar] [CrossRef] [PubMed]
- Bahr, G.; González, L.J.; Vila, A.J. Metallo-β-Lactamases in the Age of Multidrug Resistance: From Structure and Mechanism to Evolution, Dissemination and Inhibitor Design. Chem. Rev. 2021, 121, 7957–8094. [Google Scholar] [CrossRef] [PubMed]
- Hammoudi Halat, D.; Ayoub Moubareck, C. The Intriguing Carbapenemases of Pseudomonas aeruginosa: Current Status, Genetic Profile, and Global Epidemiology. Yale J. Biol. Med. 2022, 95, 507–515. [Google Scholar] [PubMed]
- Hong, D.J.; Bae, I.K.; Jang, I.-H.; Jeong, S.H.; Kang, H.-K.; Lee, K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect. Chemother. 2015, 47, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Iyobe, S.; Inoue, M.; Mitsuhashi, S. Transferable Imipenem Resistance in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 1991, 35, 147–151. [Google Scholar] [CrossRef]
- Toleman, M.A. Molecular Characterization of SPM-1, a Novel Metallo-Beta-Lactamase Isolated in Latin America: Report from the SENTRY Antimicrobial Surveillance Programme. J. Antimicrob. Chemother. 2002, 50, 673–679. [Google Scholar] [CrossRef]
- Fonseca, E.L.; Marin, M.A.; Encinas, F.; Vicente, A.C.P. Full Characterization of the Integrative and Conjugative Element Carrying the Metallo-β-Lactamase Bla SPM-1 and Bicyclomycin Bcr1 Resistance Genes Found in the Pandemic Pseudomonas aeruginosa Clone SP/ST277. J. Antimicrob. Chemother. 2015, 70, 2547–2550. [Google Scholar] [CrossRef][Green Version]
- Murphy, T.A.; Simm, A.M.; Toleman, M.A.; Jones, R.N.; Walsh, T.R. Biochemical Characterization of the Acquired Metallo-β-Lactamase SPM-1 from Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2003, 47, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Brem, J.; Struwe, W.B.; Rydzik, A.M.; Tarhonskaya, H.; Pfeffer, I.; Flashman, E.; van Berkel, S.S.; Spencer, J.; Claridge, T.D.; McDonough, M.A. Studying the Active-Site Loop Movement of the São Paolo Metallo-β-Lactamase-1. Chem. Sci. 2015, 6, 956–963. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murphy, T.A.; Catto, L.E.; Halford, S.E.; Hadfield, A.T.; Minor, W.; Walsh, T.R.; Spencer, J. Crystal Structure of Pseudomonas aeruginosa SPM-1 Provides Insights into Variable Zinc Affinity of Metallo-β-Lactamases. J. Mol. Biol. 2006, 357, 890–903. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Santos, J.V.; da Costa Júnior, S.D.; de Fátima Ramos dos Santos Medeiros, S.M.; Cavalcanti, I.D.L.; de Souza, J.B.; Coriolano, D.L.; da Silva, W.R.C.; Alves, M.H.M.E.; Cavalcanti, I.M.F. Panorama of Bacterial Infections Caused by Epidemic Resistant Strains. Curr. Microbiol. 2022, 79, 175. [Google Scholar] [CrossRef] [PubMed]
- García-Betancur, J.C.; Appel, T.M.; Esparza, G.; Gales, A.C.; Levy-Hara, G.; Cornistein, W.; Vega, S.; Nuñez, D.; Cuellar, L.; Bavestrello, L.; et al. Update on the Epidemiology of Carbapenemases in Latin America and the Caribbean. Expert Rev. Anti-Infect. Ther. 2021, 19, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.F.; Zavascki, A.P.; Gaspareto, P.B.; Barth, A.L. Dissemination of Pseudomonas aeruginosa Producing SPM-1-like and IMP-1-like Metallo-β-Lactamases in Hospitals from Southern Brazil. Infection 2007, 35, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.R.; Sellera, F.P.; Moura, Q.; Carvalho, M.P.N.; Rosato, P.N.; Cerdeira, L.; Lincopan, N. Zooanthroponotic Transmission of Drug-Resistant Pseudomonas aeruginosa, Brazil. Emerg. Infect. Dis. 2018, 24, 1160–1162. [Google Scholar] [CrossRef]
- Neves, P.R.; Perdigão Neto, L.V.; Ruedas Martins, R.C.; Ramos, J.F.; Leite, G.; Rossi, F.; Sanabani, S.S.; Rocha, V.; Batista, M.V.; Guimaraes, T.; et al. Carbapenem-Resistant Pseudomonas aeruginosa Carrying BlaVIM-36 Assigned to ST308: Indicated Non-Virulence in a Galleria Mellonella Model. J. Glob. Antimicrob. Resist. 2019, 16, 92–97. [Google Scholar] [CrossRef]
- Scavuzzi, A.M.L.; Beltrão, E.M.B.; Firmo, E.F.; de Oliveira, É.M.; Beserra, F.G.; de Souza Lopes, A.C. Emergence of BlaVIM-2, BlaNDM-1, BlaIMP-7 and BlaGES-1 in BlaKPC-2-Harbouring Pseudomonas aeruginosa Isolates in Brazil. J. Glob. Antimicrob. Resist. 2019, 19, 181–182. [Google Scholar] [CrossRef]
- de Bertoncheli, C.M.; Hörner, R. Uma revisão sobre metalo-β-lactamases. Rev. Bras. Cienc. Farm. 2008, 44, 577–599. [Google Scholar] [CrossRef]
- Inacio, H.S.; Bomfim, M.R.Q.; França, R.O.; Farias, L.M.; Carvalho, M.A.R.; Serufo, J.C.; Santos, S.G. Phenotypic and Genotypic Diversity of Multidrug-Resistant Pseudomonas aeruginosa Isolates from Bloodstream Infections Recovered in the Hospitals of Belo Horizonte, Brazil. Chemotherapy 2014, 60, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, Y.C.; Furlaneto, I.P.; Maciel, A.H.P.; Quaresma, A.J.P.G.; de Matos, E.C.O.; Conceição, M.L.; da Vieira, M.C.S.; da Brabo, G.L.C.; do Sarges, E.S.N.F.; Lima, L.N.G.C.; et al. High Prevalence of Atypical Virulotype and Genetically Diverse Background among Pseudomonas aeruginosa Isolates from a Referral Hospital in the Brazilian Amazon. PLoS ONE 2020, 15, e0238741. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.R.G.; Caiaffa-Filho, H.H.; Burattini, M.N.; Rossi, F. Metallo-Beta-Lactamases among Imipenem-Resistant Pseudomonas aeruginosa in a Brazilian University Hospital. Clinics 2010, 65, 825–829. [Google Scholar] [CrossRef] [PubMed]
- de Jácome, P.R.L.A.; Alves, L.R.; Cabral, A.B.; Lopes, A.C.S.; Maciel, M.A.V. Phenotypic and Molecular Characterization of Antimicrobial Resistance and Virulence Factors in Pseudomonas aeruginosa Clinical Isolates from Recife, State of Pernambuco, Brazil. Rev. Soc. Bras. Med. Trop. 2012, 45, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Gräf, T.; Fuentefria, D.B.; Corção, G. Ocorrência de Cepas de Pseudomonas aeruginosa Multirresistentes Produtoras de Metalo-β-Lactamase BlaSPM-1 Em Amostras Clínicas. Rev. Soc. Bras. Med. Trop. 2008, 41, 306–308. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- da Santos, C.M.C.; de Pimenta, C.A.M.; Nobre, M.R.C. The PICO Strategy for the Research Question Construction and Evidence Search. Rev. Lat.-Am. Enferm. 2007, 15, 508–511. [Google Scholar] [CrossRef]
- Kar, B.; Sharma, M.; Peter, A.; Chetia, P.; Neog, B.; Borah, A.; Pati, S.; Bhattacharya, D. Prevalence and Molecular Characterization of β-Lactamase Producers and Fluoroquinolone Resistant Clinical Isolates from North East India. J. Infect. Public Health 2021, 14, 628–637. [Google Scholar] [CrossRef]
- Aromataris, E.; Munn, Z. Joanna Briggs Institute Reviewer’s Manual. Joanna Briggs Inst. 2017, 2017. [Google Scholar] [CrossRef]
- Munn, Z.; Aromataris, E.; Tufanaru, C.; Stern, C.; Porritt, K.; Farrow, J.; Lockwood, C.; Stephenson, M.; Moola, S.; Lizarondo, L.; et al. The Development of Software to Support Multiple Systematic Review Types: The Joanna Briggs Institute System for the Unified Management, Assessment and Review of Information (JBI SUMARI). Int. J. Evid. Based Healthc. 2019, 17, 36–43. [Google Scholar] [CrossRef]
- Foumani, A.A.; Kalurazi, T.Y.; Rostami, F.M.; Ebrahim-Saraie, H.S.; Nazari-Alam, A.; Halaji, M. Epidemiology of Pseudomonas aeruginosa in Cystic Fibrosis Patients in Iran: A Systematic Review and Meta-Analysis. Infez. Med. 2020, 28, 314–321. [Google Scholar]
- Gonçalves, D.C.P.S.; Lima, A.B.M.; de Leão, L.S.N.O.; do Carmo Filho, J.R.; Pimenta, F.C.; Vieira, J.D.G. Detecção de Metalo-Beta-Lactamase Em Pseudomonas aeruginosa Isoladas de Pacientes Hospitalizados Em Goiânia, Estado de Goiás. Rev. Soc. Bras. Med. Trop. 2009, 42, 411–414. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fernandes, T.Á.; Pereira, C.A.P.; Petrili, A.S.; Pignatari, A.C.C. Caracterização Molecular de Pseudomonas aeruginosa Resistentes a Carbapenêmicos e Produtoras de Metalo-β-Lactamase Isoladas Em Hemoculturas de Crianças e Adolescentes Com Câncer. Rev. Soc. Bras. Med. Trop. 2010, 43, 372–376. [Google Scholar] [CrossRef]
- Scheffer, M.C.; Bazzo, M.L.; Steindel, M.; Darini, A.L.; Clímaco, E.; Dalla-Costa, L.M. Intrahospital Spread of Carbapenem-Resistant Pseudomonas aeruginosa in a University Hospital in Florianópolis, Santa Catarina, Brazil. Rev. Soc. Bras. Med. Trop. 2010, 43, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Polotto, M.; Casella, T.; de Lucca Oliveira, M.G.; Rúbio, F.G.; Nogueira, M.L.; de Almeida, M.T.; Nogueira, M.C. Detection of P. aeruginosa Harboring Bla CTX-M-2, Bla GES-1 and Bla GES-5, Bla IMP-1 and Bla SPM-1 Causing Infections in Brazilian Tertiary-Care Hospital. BMC Infect. Dis. 2012, 12, 176. [Google Scholar] [CrossRef] [PubMed]
- Lucena, A.; Dalla Costa, L.M.; Nogueira, K.S.; Matos, A.P.; Gales, A.C.; Paganini, M.C.; Castro, M.E.S.; Raboni, S.M. Nosocomial Infections with Metallo-Beta-Lactamase-Producing Pseudomonas aeruginosa: Molecular Epidemiology, Risk Factors, Clinical Features and Outcomes. J. Hosp. Infect. 2014, 87, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.C.; De Filippis, I.; Pinto, L.H.; Coelho-Souza, T.; Bianco, K.; Cacci, L.C.; Picão, R.C.; Clementino, M.M. Genotypic Characteristics of Multidrug-Resistant Pseudomonas aeruginosa from Hospital Wastewater Treatment Plant in Rio de Janeiro, Brazil. J. Appl. Microbiol. 2015, 118, 1276–1286. [Google Scholar] [CrossRef]
- Cacci, L.C.; Chuster, S.G.; Martins, N.; do Carmo, P.R.; de Girão, V.B.C.; Nouér, S.A.; de Freitas, W.V.; de Matos, J.A.; de Magalhães, A.C.G.; Ferreira, A.L.P. Mechanisms of Carbapenem Resistance in Endemic Pseudomonas aeruginosa Isolates after an SPM-1 Metallo-β-Lactamase Producing Strain Subsided in an Intensive Care Unit of a Teaching Hospital in Brazil. Memórias Inst. Oswaldo Cruz 2016, 111, 551–558. [Google Scholar] [CrossRef]
- De Matos, E.C.O.; de Matos, H.J.; Conceição, M.L.; Rodrigues, Y.C.; do Carneiro, I.C.R.S.; Lima, K.V.B. Clinical and Microbiological Features of Infections Caused by Pseudomonas aeruginosa in Patients Hospitalized in Intensive Care Units. Rev. Soc. Bras. Med. Trop. 2016, 49, 305–311. [Google Scholar] [CrossRef]
- Chaves, L.; Tomich, L.M.; Salomão, M.; Leite, G.C.; Ramos, J.; Martins, R.R.; Rizek, C.; Neves, P.; Batista, M.V.; Amigo, U.; et al. High Mortality of Bloodstream Infection Outbreak Caused by Carbapenem-Resistant P. aeruginosa Producing SPM-1 in a Bone Marrow Transplant Unit. J. Med. Microbiol. 2017, 66, 1722–1729. [Google Scholar] [CrossRef]
- Dias, V.C.; Resende, J.A.; Bastos, A.N.; De Andrade Bastos, L.Q.; De Andrade Bastos, V.Q.; Bastos, R.V.; Diniz, C.G.; Da Silva, V.L. Epidemiological, Physiological, and Molecular Characteristics of a Brazilian Collection of Carbapenem-Resistant Acinetobacter baumannii and Pseudomonas aeruginosa. Microb. Drug Resist. 2017, 23, 852–863. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, I.R.; Dantas, R.C.C.; Ferreira, M.L.; da Batistão, D.W.F.; Gontijo-Filho, P.P.; Ribas, R.M. Carbapenem-Resistant Pseudomonas aeruginosa: Association with Virulence Genes and Biofilm Formation. Braz. J. Microbiol. 2017, 48, 211–217. [Google Scholar] [CrossRef]
- Martins, W.M.B.S.; Narciso, A.C.; Cayô, R.; Santos, S.V.; Fehlberg, L.C.C.; Ramos, P.L.; Da Cruz, J.B.; Gales, A.C. SPM-1-Producing Pseudomonas aeruginosa ST277 Clone Recovered from Microbiota of Migratory Birds. Diagn. Microbiol. Infect. Dis. 2018, 90, 221–227. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Santos, I.C.; Pereira De Andrade, N.F.; Da Conceição Neto, O.C.; Da Costa, B.S.; De Andrade Marques, E.; Rocha-de-Souza, C.M.; Asensi, M.D.; D’Alincourt Carvalho-Assef, A.P. Epidemiology and Antibiotic Resistance Trends in Clinical Isolates of Pseudomonas aeruginosa from Rio de Janeiro—Brazil: Importance of Mutational Mechanisms over the Years (1995–2015). Infect. Genet. Evol. 2019, 73, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Silveira, M.C.; Rocha-de-Souza, C.M.; Albano, R.M.; De Oliveira Santos, I.C.; Carvalho-Assef, A.P.D. Exploring the Success of Brazilian Endemic Clone Pseudomonas aeruginosa ST277 and Its Association with the CRISPR-Cas System Type I-C. BMC Genom. 2020, 21, 255. [Google Scholar] [CrossRef] [PubMed]
- Freire, M.P.; Camargo, C.H.; Yamada, A.Y.; Nagamori, F.O.; Reusing Junior, J.O.; Spadão, F.; Cury, A.P.; Rossi, F.; Nahas, W.C.; David-Neto, E.; et al. Critical Points and Potential Pitfalls of Outbreak of IMP-1-Producing Carbapenem-Resistant Pseudomonas aeruginosa among Kidney Transplant Recipients: A Case–Control Study. J. Hosp. Infect. 2021, 115, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, P.A.S.; Rodrigues, Y.C.; Marcon, D.J.; Lobato, A.R.F.; Cazuza, T.B.; Gouveia, M.I.M.; Silva, M.J.A.; Souza, A.B.; Lima, L.N.G.C.; Quaresma, A.J.P.G.; et al. Endemic High-Risk Clone ST277 Is Related to the Spread of SPM-1-Producing Pseudomonas aeruginosa during the COVID-19 Pandemic Period in Northern Brazil. Microorganisms 2023, 11, 2069. [Google Scholar] [CrossRef]
- Ghasemian, A.; Rizi, K.S.; Vardanjani, H.R.; Nojoomi, F. Prevalence of Clinically Isolated Metallo-Beta-Lactamase-Producing Pseudomonas aeruginosa, Coding Genes, and Possible Risk Factors in Iran. Iran. J. Pathol. 2018, 13, 1. [Google Scholar]
- Dawadi, P.; Khadka, C.; Shyaula, M.; Syangtan, G.; Joshi, T.P.; Pepper, S.H.; Kanel, S.R.; Pokhrel, L.R. Prevalence of Metallo-β-Lactamases as a Correlate of Multidrug Resistance among Clinical Pseudomonas aeruginosa Isolates in Nepal. Sci. Total Environ. 2022, 850, 157975. [Google Scholar] [CrossRef]
- Partridge, S.R. Resistance Mechanisms in Enterobacteriaceae. Pathology 2015, 47, 276–284. [Google Scholar] [CrossRef]
- Edelstein, M.V.; Skleenova, E.N.; Shevchenko, O.V.; D’souza, J.W.; Tapalski, D.V.; Azizov, I.S.; Sukhorukova, M.V.; Pavlukov, R.A.; Kozlov, R.S.; Toleman, M.A.; et al. Spread of Extensively Resistant VIM-2-Positive ST235 Pseudomonas aeruginosa in Belarus, Kazakhstan, and Russia: A Longitudinal Epidemiological and Clinical Study. Lancet Infect. Dis. 2013, 13, 867–876. [Google Scholar] [CrossRef]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, Research, and Development of New Antibiotics: The WHO Priority List of Antibiotic-Resistant Bacteria and Tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Collignon, P.; Beggs, J.J.; Walsh, T.R.; Gandra, S.; Laxminarayan, R. Anthropological and Socioeconomic Factors Contributing to Global Antimicrobial Resistance: A Univariate and Multivariable Analysis. Lancet Planet. Health 2018, 2, e398–e405. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, R.R.; Moreira, B.M.; Picão, R.C. Antimicrobial Resistance among Enterobacteriaceae in South America: History, Current Dissemination Status and Associated Socioeconomic Factors. Drug Resist. Updates 2014, 17, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Planta, M.B. The Role of Poverty in Antimicrobial Resistance. J. Am. Board. Fam. Med. 2007, 20, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Tenover, F.C.; Nicolau, D.P.; Gill, C.M. Carbapenemase-Producing Pseudomonas aeruginosa—An Emerging Challenge. Emerg. Microbes Infect. 2022, 11, 811–814. [Google Scholar] [CrossRef]
- Escandón-Vargas, K.; Reyes, S.; Gutiérrez, S.; Villegas, M.V. The Epidemiology of Carbapenemases in Latin America and the Caribbean. Expert Rev. Anti-Infect. Ther. 2017, 15, 277–297. [Google Scholar] [CrossRef]
- Zowawi, H.M.; Balkhy, H.H.; Walsh, T.R.; Paterson, D.L. β-Lactamase Production in Key Gram-Negative Pathogen Isolates from the Arabian Peninsula. Clin. Microbiol. Rev. 2013, 26, 361–380. [Google Scholar] [CrossRef]
- Poirel, L.; Nordmann, P.; Lagrutta, E.; Cleary, T.; Munoz-Price, L.S. Emergence of KPC-Producing Pseudomonas aeruginosa in the United States. Antimicrob. Agents Chemother. 2010, 54, 3072. [Google Scholar] [CrossRef]
- Walters, M.S.; Grass, J.E.; Bulens, S.N.; Hancock, E.B.; Phipps, E.C.; Muleta, D.; Mounsey, J.; Kainer, M.A.; Concannon, C.; Dumyati, G. Carbapenem-Resistant Pseudomonas aeruginosa at US Emerging Infections Program Sites, 2015. Emerg. Infect. Dis. 2019, 25, 1281. [Google Scholar] [CrossRef]
- Rojo-Bezares, B.; Estepa, V.; Cebollada, R.; de Toro, M.; Somalo, S.; Seral, C.; Castillo, F.J.; Torres, C.; Sáenz, Y. Carbapenem-Resistant Pseudomonas aeruginosa Strains from a Spanish Hospital: Characterization of Metallo-Beta-Lactamases, Porin OprD and Integrons. Int. J. Med. Microbiol. 2014, 304, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Martinez, E.; Pérez, J.E.; Buelvas, F.; Tovar, C.; Vanegas, N.; Stokes, H.W. Establishment and Multi Drug Resistance Evolution of ST235 Pseudomonas aeruginosa Strains in the Intensive Care Unit of a Colombian Hospital. Res. Microbiol. 2014, 165, 852–856. [Google Scholar] [CrossRef] [PubMed]
- El Salabi, A.; Toleman, M.A.; Weeks, J.; Bruderer, T.; Frei, R.; Walsh, T.R. First Report of the Metallo-β-Lactamase SPM-1 in Europe. Antimicrob. Agents Chemother. 2010, 54, 582. [Google Scholar] [CrossRef] [PubMed]
- Wendel, A.F.; Brodner, A.H.B.; Wydra, S.; Ressina, S.; Henrich, B.; Pfeffer, K.; Toleman, M.A.; MacKenzie, C.R. Genetic Characterization and Emergence of the Metallo-β-Lactamase GIM-1 in Pseudomonas Spp. and Enterobacteriaceae during a Long-Term Outbreak. Antimicrob. Agents Chemother. 2013, 57, 5162–5165. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, M.; Deshpande, L.M.; Costello, A.; Davies, T.A.; Jones, R.N. Epidemiology and Carbapenem Resistance Mechanisms of Carbapenem-Non-Susceptible Pseudomonas aeruginosa Collected during 2009-11 in 14 European and Mediterranean Countries. J. Antimicrob. Chemother. 2014, 69, 1804–1814. [Google Scholar] [CrossRef] [PubMed]
- Pollini, S.; Maradei, S.; Pecile, P.; Olivo, G.; Luzzaro, F.; Docquier, J.-D.; Rossolini, G.M. FIM-1, a New Acquired Metallo-β-Lactamase from a Pseudomonas aeruginosa Clinical Isolate from Italy. Antimicrob. Agents Chemother. 2013, 57, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.H.; De, A.S.; Baveja, S.M.; Gore, M.A. Prevalence and Risk Factors of Metallo β-Lactamase Producing Pseudomonas aeruginosa and Acinetobacter Species in Burns and Surgical Wards in a Tertiary Care Hospital. J. Lab. Physicians 2012, 4, 39–42. [Google Scholar] [CrossRef] [PubMed]
- El-Domany, R.A.; Emara, M.; El-Magd, M.A.; Moustafa, W.H.; Abdeltwab, N.M. Emergence of Imipenem-Resistant Pseudomonas aeruginosa Clinical Isolates from Egypt Coharboring VIM and IMP Carbapenemases. Microb. Drug Resist. 2017, 23, 682–686. [Google Scholar] [CrossRef]
- Khosravi, A.D.; Mihani, F. Detection of Metallo-β-Lactamase–Producing Pseudomonas aeruginosa Strains Isolated from Burn Patients in Ahwaz, Iran. Diagn. Microbiol. Infect. Dis. 2008, 60, 125–128. [Google Scholar] [CrossRef]
- Malkoçoğlu, G.; Aktaş, E.; Bayraktar, B.; Otlu, B.; Bulut, M.E. VIM-1, VIM-2, and GES-5 Carbapenemases among Pseudomonas aeruginosa Isolates at a Tertiary Hospital in Istanbul, Turkey. Microb. Drug Resist. 2017, 23, 328–334. [Google Scholar] [CrossRef]
- Ghamgosha, M.; Shahrekizahedani, S.; Kafilzadeh, F.; Bameri, Z.; Taheri, R.A.; Farnoosh, G. Metallo-Beta-Lactamase VIM-1, SPM-1, and IMP-1 Genes Among Clinical Pseudomonas aeruginosa Species Isolated in Zahedan, Iran. Jundishapur J. Microbiol. 2015, 8, e17489. [Google Scholar] [CrossRef] [PubMed]
- Moosavian, M.; Rahimzadeh, M. Molecular Detection of Metallo-β-Lactamase Genes, BlaIMP-1, BlaVIM-2 and BlaSPM-1 in Imipenem Resistant Pseudomonas aeruginosa Isolated from Clinical Specimens in Teaching Hospitals of Ahvaz, Iran. Iran. J. Microbiol. 2015, 7, 2–6. [Google Scholar] [PubMed]
- Hopkins, K.L.; Meunier, D.; Findlay, J.; Mustafa, N.; Parsons, H.; Pike, R.; Wright, L.; Woodford, N. SPM-1 Metallo-β-Lactamase-Producing Pseudomonas aeruginosa ST277 in the UK. J. Med. Microbiol. 2016, 65, 696–697. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, A.; Rodríguez, N.; Alcalde-Rico, M.; Castillo, C.; García, P. Primer Aislado de Pseudomonas aeruginosa Productora de Sao Paulo Metalo-β-Lactamasa (SPM-1) En Un Paciente Chileno. Rev. Chil. Infectol. 2021, 38, 724–726. [Google Scholar] [CrossRef]
- Abdelaziz, N.A. Phenotype-Genotype Correlations among Carbapenem-Resistant Enterobacterales Recovered from Four Egyptian Hospitals with the Report of SPM Carbapenemase. Antimicrob. Resist. Infect. Control 2022, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- Figueredo, A.C.F.; Freitas, N.L.D.; Dalmolin, T.V.; Brandão, F. Pseudomonas aeruginosa: Panorama do perfil de resistência aos carbapenêmicos no brasil/Pseudomonas aeruginosa: Overview of the profile of resistance to carbapenems in brazil. BJD 2021, 7, 9661–9672. [Google Scholar] [CrossRef]
- ul Ain, N.; Abrar, S.; Sherwani, R.A.K.; Hannan, A.; Imran, N.; Riaz, S. Systematic Surveillance and Meta-Analysis on the Prevalence of Metallo-β-Lactamase Producers among Carbapenem Resistant Clinical Isolates in Pakistan. J. Glob. Antimicrob. Resist. 2020, 23, 55–63. [Google Scholar] [CrossRef]
- Osei Sekyere, J.; Reta, M.A. Genomic and Resistance Epidemiology of Gram-Negative Bacteria in Africa: A Systematic Review and Phylogenomic Analyses from a One Health Perspective. Msystems 2020, 5, e00897-20. [Google Scholar] [CrossRef]
- Vaez, H.; Salehi-Abargouei, A.; Ghalehnoo, Z.R.; Khademi, F. Multidrug Resistant Pseudomonas aeruginosa in Iran: A Systematic Review and Metaanalysis. J. Glob. Infect. Dis. 2018, 10, 212. [Google Scholar]
- Pisani, B.; Simões, M.; Prandi, M.A.G.; Rocha, M.M.; Gonçalves, C.R.; Vaz, T.M.; Irino, K. Surto de Bacteriemia Por Pseudomonas aeruginosa Na Unidade de Hemodiálise de Um Hospital de Campinas, São Paulo, Brasil. Rev. Inst. Adolfo Lutz 2000, 59, 51–56. [Google Scholar]
- Kümmerer, K.; Henninger, A. Promoting Resistance by the Emission of Antibiotics from Hospitals and Households into Effluent. Clin. Microbiol. Infect. 2003, 9, 1203–1214. [Google Scholar] [CrossRef]
- Kunin, C.M. Resistance to Antimicrobial Drugs—A Worldwide Calamity. Ann. Intern. Med. 1993, 118, 557. [Google Scholar] [CrossRef]
- Laranjeira, V.D.S.; Marchetti, D.P.; Steyer, J.R.; Corção, G.; Picoli, S.U. Pesquisa de Acinetobacter Sp e Pseudomonas aeruginosa Produtores de Metalo-β-Lactamase Em Hospital de Emergência de Porto Alegre, Estado Do Rio Grande Do Sul, Brasil. Rev. Soc. Bras. Med. Trop. 2010, 43, 462–464. [Google Scholar] [CrossRef]
- Camargo, C.H.; Bruder-Nascimento, A.; Mondelli, A.L.; Montelli, A.C.; Sadatsune, T. Detection of SPM and IMP Metallo-β-Lactamases in Clinical Specimens of Pseudomonas aeruginosa from a Brazilian Public Tertiary Hospital. Braz. J. Infect. Dis. 2011, 15, 478–481. [Google Scholar] [CrossRef]
- de Cavalcanti, F.L.S.; Almeida, A.C.S.; Vilela, M.A.; de Morais, M.M.C.; de Morais Junior, M.A. Changing the Epidemiology of Carbapenem-Resistant Pseudomonas aeruginosa in a Brazilian Teaching Hospital: The Replacement of São Paulo Metallo-β-Lactamase-Producing Isolates. Mem. Inst. Oswaldo Cruz 2012, 107, 420–423. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).