Electrolyzed Water Treatment for the Control of the Zoonotic Pathogen Vibrio vulnificus in Aquaculture: A One Health Perspective
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Cultures
2.2. Electrolyzed Water (EW) Preparation and Characterizacion
2.3. Bacterial Survival in EW
2.4. Statistical Analysis
3. Results and Discussion
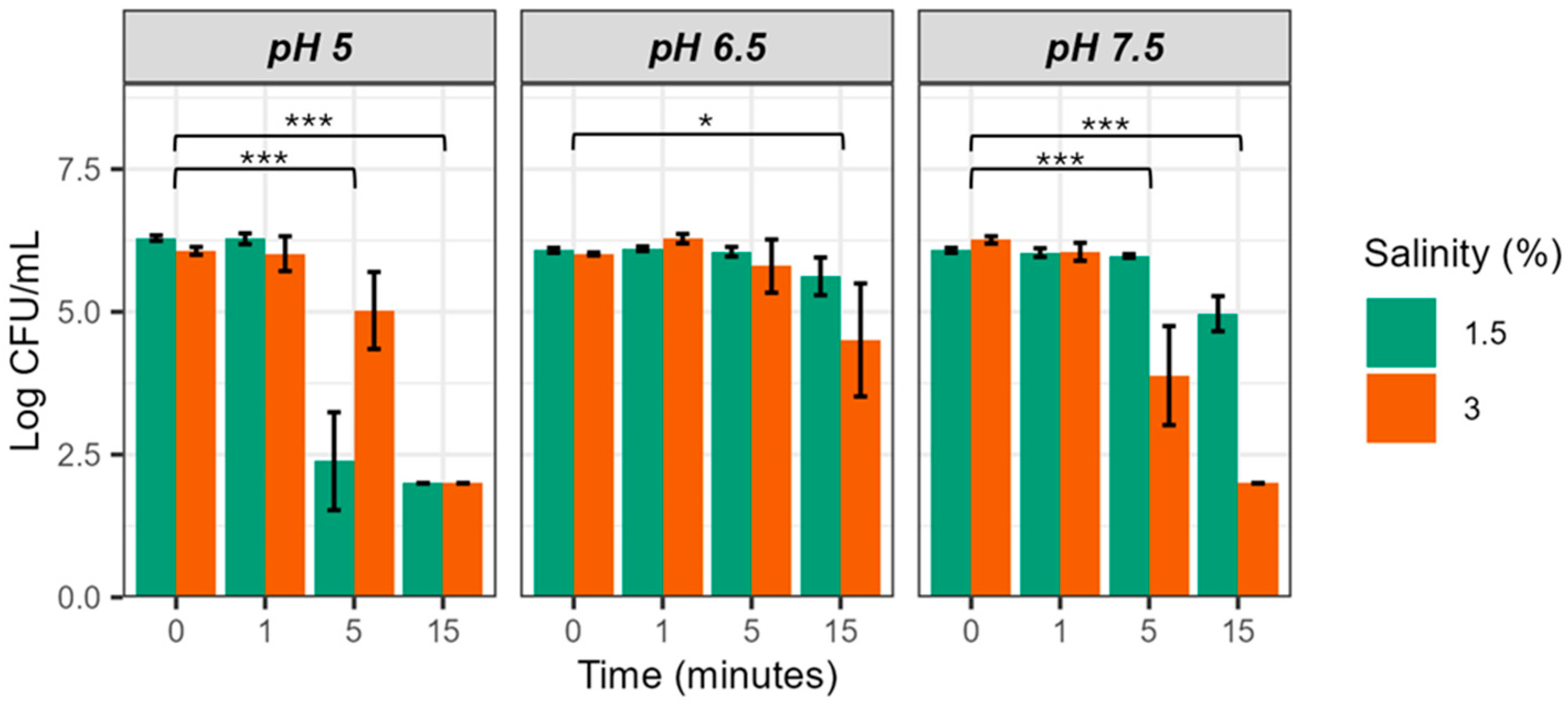
3.1. Influence of Salinity, pH and FAC on the Bactericidal Activity of EW
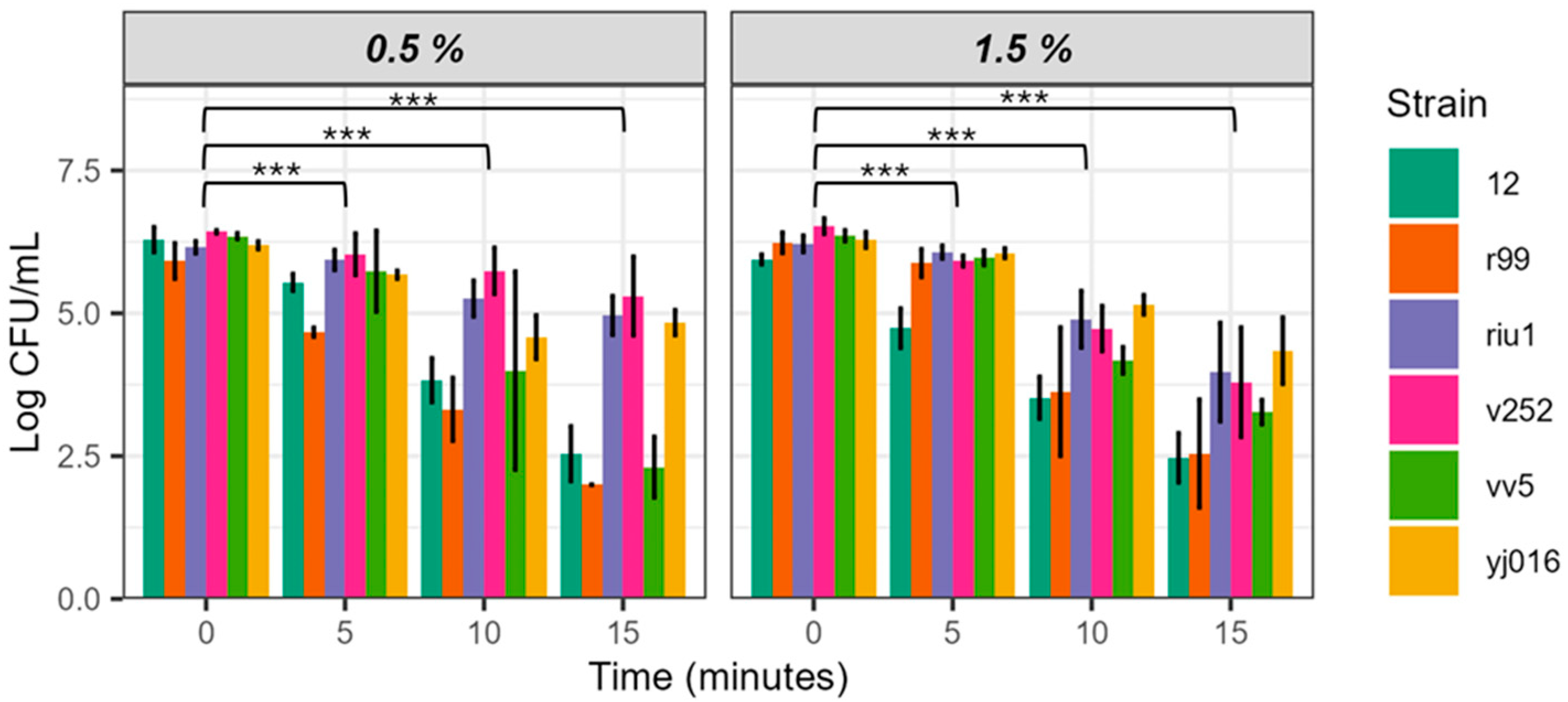
3.2. Evaluation of the Bactericidal Effect on Different Strains of V. vulnificus
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Oliver, J.D. The Biology of Vibrio vulnificus. Microbiol. Spectr. 2015, 3, 3.3.01. [Google Scholar] [CrossRef] [PubMed]
- Baker-Austin, C.; Trinanes, J.; Gonzalez-Escalona, N.; Martinez-Urtaza, J. Non-Cholera Vibrios: The Microbial Barometer of Climate Change. Trends Microbiol. 2017, 25, 76–84. [Google Scholar] [CrossRef]
- Hernández-Cabanyero, C.; Sanjuán, E.; Fouz, B.; Pajuelo, D.; Vallejos-Vidal, E.; Reyes-López, F.E.; Amaro, C. The Effect of the Environmental Temperature on the Adaptation to Host in the Zoonotic Pathogen Vibrio vulnificus. Front. Microbiol. 2020, 11, 489. [Google Scholar] [CrossRef] [PubMed]
- Baker-Austin, C.; Oliver, J.D. Vibrio vulnificus: New Insights into a Deadly Opportunistic Pathogen. Environ. Microbiol. 2018, 20, 423–430. [Google Scholar] [CrossRef]
- Amaro, C.; Sanjuán, E.; Fouz, B.; Pajuelo, D.; Lee, C.-T.; Hor, L.-I.; Barrera, R. The Fish Pathogen Vibrio vulnificus Biotype 2: Epidemiology, Phylogeny, and Virulence Factors Involved in Warm-Water Vibriosis. Microbiol. Spectr. 2015, 3. [Google Scholar] [CrossRef]
- Carmona-Salido, H.; Fouz, B.; Sanjuán, E.; Carda, M.; Delannoy, C.M.J.; García-González, N.; González-Candelas, F.; Amaro, C. The Widespread Presence of a Family of Fish Virulence Plasmids in Vibrio vulnificus Stresses Its Relevance as a Zoonotic Pathogen Linked to Fish Farms. Emerg. Microbes Infect. 2021, 10, 2128–2140. [Google Scholar] [CrossRef]
- Baker-Austin, C.; McArthur, J.V.; Lindell, A.H.; Wright, M.S.; Tuckfield, R.C.; Gooch, J.; Warner, L.; Oliver, J.; Stepanauskas, R. Multi-Site Analysis Reveals Widespread Antibiotic Resistance in the Marine Pathogen Vibrio vulnificus. Microb. Ecol. 2009, 57, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Elmahdi, S.; DaSilva, L.V.; Parveen, S. Antibiotic Resistance of Vibrio parahaemolyticus and Vibrio vulnificus in Various Countries: A Review. Food Microbiol. 2016, 57, 128–134. [Google Scholar] [CrossRef]
- Heng, S.-P.; Letchumanan, V.; Deng, C.-Y.; Ab Mutalib, N.-S.; Khan, T.M.; Chuah, L.-H.; Chan, K.-G.; Goh, B.-H.; Pusparajah, P.; Lee, L.-H. Vibrio vulnificus: An Environmental and Clinical Burden. Front. Microbiol. 2017, 8, 997. [Google Scholar] [CrossRef]
- Singh, A.K.; Bhunia, A.K. Animal-Use Antibiotics Induce Cross-Resistance in Bacterial Pathogens to Human Therapeutic Antibiotics. Curr. Microbiol. 2019, 76, 1112–1117. [Google Scholar] [CrossRef]
- Li, H.; Cui, Z.; Cui, H.; Bai, Y.; Yin, Z.; Qu, K. A Review of Influencing Factors on a Recirculating Aquaculture System: Environmental Conditions, Feeding Strategies, and Disinfection Methods. J. World Aquac. Soc. 2023, 54, 566–602. [Google Scholar] [CrossRef]
- Kim, C.; Hung, Y.-C.; Brackett, R.E. Efficacy of Electrolyzed Oxidizing (EO) and Chemically Modified Water on Different Types of Foodborne Pathogens. Int. J. Food Microbiol. 2000, 61, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-J.; Alexander, E.; Taylor, G.A.; Costa, R.; Kang, D.-H. Effect of Electrolyzed Water for Reduction of Foodborne Pathogens on Lettuce and Spinach. J. Food Sci. 2008, 73, M268–M272. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Daliri, E.B.-M.; Oh, D.-H. New Clinical Applications of Electrolyzed Water: A Review. Microorganisms 2021, 9, 136. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.-K.; Wang, C.-K. Electrolyzed Water and Its Pharmacological Activities: A Mini-Review. Molecules 2022, 27, 1222. [Google Scholar] [CrossRef]
- Goh, C.F.; Ming, L.C.; Wong, L.C. Dermatologic Reactions to Disinfectant Use during the COVID-19 Pandemic. Clin. Dermatol. 2021, 39, 314–322. [Google Scholar] [CrossRef]
- Venkitanarayanan, K.S.; Ezeike, G.O.; Hung, Y.-C.; Doyle, M.P. Efficacy of Electrolyzed Oxidizing Water for Inactivating Escherichia coli O157:H7, Salmonella enteritidis, and Listeria monocytogenes. Appl. Environ. Microbiol. 1999, 65, 4276–4279. [Google Scholar] [CrossRef]
- Amaro, C.; Biosca, E.G. Vibrio vulnificus Biotype 2, Pathogenic for Eels, Is Also an Opportunistic Pathogen for Humans. Appl. Environ. Microbiol. 1996, 62, 1454–1457. [Google Scholar] [CrossRef]
- Hoben, H.J.; Somasegaran, P. Comparison of the Pour, Spread, and Drop Plate Methods for Enumeration of Rhizobium Spp. in Inoculants Made from Presterilized Peat. Appl. Environ. Microbiol. 1982, 44, 1246–1247. [Google Scholar] [CrossRef]
- Deeb, R.; Tufford, D.; Scott, G.I.; Moore, J.G.; Dow, K. Impact of Climate Change on Vibrio vulnificus Abundance and Exposure Risk. Estuaries Coasts 2018, 41, 2289–2303. [Google Scholar] [CrossRef]
- Kasai, H.; Kawana, K.; Labaiden, M.; Namba, K.; Yoshimizu, M. Elimination of Escherichia coli from Oysters Using Electrolyzed Seawater. Aquaculture 2011, 319, 315–318. [Google Scholar] [CrossRef]
- Katayose, M.; Yoshida, K.; Achiwa, N.; Eguchi, M. Safety of Electrolyzed Seawater for Use in Aquaculture. Aquaculture 2007, 264, 119–129. [Google Scholar] [CrossRef]
- Pettis, G.S.; Mukerji, A.S. Structure, Function, and Regulation of the Essential Virulence Factor Capsular Polysaccharide of Vibrio Vulnificus. Int. J. Mol. Sci. 2020, 21, 3259. [Google Scholar] [CrossRef]
- Han, Q.; Song, X.; Zhang, Z.; Fu, J.; Wang, X.; Malakar, P.K.; Liu, H.; Pan, Y.; Zhao, Y. Removal of Foodborne Pathogen Biofilms by Acidic Electrolyzed Water. Front. Microbiol. 2017, 8, 988. [Google Scholar] [CrossRef] [PubMed]

| Strain 1 | Source | Geographic Location | Year of Isolation | Lineage 2 |
|---|---|---|---|---|
| CECT 4999 | Diseased farmed eel | Spain | 1999 | L2 |
| YJ016 | Human blood | Taiwan | 1993 | L1 |
| Riu1 | Seawater | Western Mediterranean Sea | 2003 | L4 |
| V252 | Human blood | Israel | 2004 | L5 |
| VV12 | Human blood | Israel | 1996 | L3 |
| VV5 | Farmed diseased tilapia | Eastern Mediterranean Sea | 2016 | L1 |
| pH | FAC (ppm) | ORP (mV) | %NaCl | Conductivity (mS/cm) |
|---|---|---|---|---|
| 7.5 | 5 | 828 | 1.5 | 26.2 |
| 900 | 3 | 47.5 | ||
| 25 | 889 | 1.5 | 25.7 | |
| 937 | 3 | 48.1 | ||
| 125 | 943 | 1.5 | 25.9 | |
| 960 | 3 | 49.3 | ||
| 6.5 | 5 | 832 | 1.5 | 26.0 |
| 902 | 3 | 47.4 | ||
| 25 | 892 | 1.5 | 25.9 | |
| 933 | 3 | 47.9 | ||
| 125 | 947 | 1.5 | 26.1 | |
| 962 | 3 | 49.5 | ||
| 5 | 5 | 828 | 1.5 | 26.3 |
| 902 | 3 | 47.3 | ||
| 15 | 835 | 1.5 | 26.1 | |
| 901 | 3 | 48.2 | ||
| 20 | 890 | 1.5 | 26.2 | |
| 940 | 3 | 49.3 | ||
| 25 | 890 | 1.5 | 26 | |
| 940 | 3 | 49.3 | ||
| 125 | 950 | 1.5 | 26.2 | |
| 964 | 3 | 48.9 |
| Source of Variation | Df 1 | F 1 | p-Value 1 |
|---|---|---|---|
| Salinity | 1 | 8.15 | 6.35 × 10−4 (**) |
| pH | 2 | 6.52 | 3.13 × 10−4 (**) |
| Time | 3 | 67.29 | <2.2 × 10−16 (***) |
| Salinity X pH | 2 | 2.56 | 0.087 |
| Salinity X Time | 3 | 2.07 | 0.117 |
| pH X Time | 6 | 8.69 | 2.04 × 10−6 (***) |
| Source of Variation | Df 1 | F 1 | p-Value 1 |
|---|---|---|---|
| Salinity | 1 | 5.596 | 2.2 × 10−3 (*) |
| FAC | 2 | 68.75 | 8.12 × 10−15 (***) |
| Time | 3 | 93.69 | <2.2 × 10−16 (***) |
| Salinity X FAC | 2 | 11.53 | 8.15 × 10−5 (***) |
| Salinity X Time | 3 | 7.52 | 3.17 × 10−5 (***) |
| FAC X Time | 6 | 20.17 | 1.31 × 10−11 (***) |
| Source of Variation | Df 1 | F 1 | p-Value 1 |
|---|---|---|---|
| Salinity | 1 | 2.31 | 0.13 |
| Strain | 5 | 18.19 | 1.16 × 10−12 (***) |
| Time | 3 | 157.69 | <2.2 × 10−16 (***) |
| Strain X Salinity | 5 | 8.74 | 7.33 × 10−7 (***) |
| Salinity X Time | 3 | 4.89 | 3.3 × 10−4 (**) |
| Strain X Time | 15 | 6.46 | 2.76 × 10−9 (***) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibányez-Payá, P.; Blasco, A.; Ros-Lis, J.V.; Fouz, B.; Amaro, C. Electrolyzed Water Treatment for the Control of the Zoonotic Pathogen Vibrio vulnificus in Aquaculture: A One Health Perspective. Microorganisms 2024, 12, 1992. https://doi.org/10.3390/microorganisms12101992
Ibányez-Payá P, Blasco A, Ros-Lis JV, Fouz B, Amaro C. Electrolyzed Water Treatment for the Control of the Zoonotic Pathogen Vibrio vulnificus in Aquaculture: A One Health Perspective. Microorganisms. 2024; 12(10):1992. https://doi.org/10.3390/microorganisms12101992
Chicago/Turabian StyleIbányez-Payá, Pablo, Adolfo Blasco, José V. Ros-Lis, Belén Fouz, and Carmen Amaro. 2024. "Electrolyzed Water Treatment for the Control of the Zoonotic Pathogen Vibrio vulnificus in Aquaculture: A One Health Perspective" Microorganisms 12, no. 10: 1992. https://doi.org/10.3390/microorganisms12101992
APA StyleIbányez-Payá, P., Blasco, A., Ros-Lis, J. V., Fouz, B., & Amaro, C. (2024). Electrolyzed Water Treatment for the Control of the Zoonotic Pathogen Vibrio vulnificus in Aquaculture: A One Health Perspective. Microorganisms, 12(10), 1992. https://doi.org/10.3390/microorganisms12101992








