3.1. Evaluation of Different Substrates for P. citrinopileatus Cultivation
The final mixtures of SMS and HRL were prepared based on the ratio of C/N (15–36). A variety of C/N ratios have been reported in previous studies for cultivation substrates of
P. citrinopileatus [
48]. The total nitrogen content in the substrates SMS 100% and SMS 90%-HRL 10% were below 1% and consequently, the final C/N ratio was increased. The pH values were also suitable for mushroom cultivation, while the EC values significantly varied among substrates.
The effect of different substrates consisting of SMS or a combination of SMS and HRL, supplemented or not with additives, on
P. citrinopileatus growth was initially examined through the mycelial growth rate (Kr, mm/d). In general, HRL addition led to a faster Kr than the substrates only consisting of SMS (
Figure 1). Specifically, the highest Kr value (6.1 mm/d) for
P. citrinopileatus was recorded on substrates with the highest ratio of HRL (SMS 60%-HRL 40% and SMS 50%-HRL 40%), higher values than the control substrate (WS). Regarding substrates with only SMS, the mycelial growth rate of
P. citrinopileatus increased progressively with the SMS increase, as has already been reported by Wang et al. [
49]. Also, it is noteworthy that the lack of additives in WB and SF did not negatively affect Kr values. In previous studies, faster growth rates (6–8 mm/d) were detected for
P. citrinopileatus cultivated on substrates consisting of the stalks of several grass plants [
48], whereas
P. ostreatus and
P. eryngii achieved lower Kr values (3.1 and 2.6 mm/d, respectively) on commercial SMS [
50] than samples in the present study. As combinations of different ratios of SMS and HRL have not been examined as alternative substrates for new mushroom cultivations, in the subsequent larger-scale experiments (1 kg bags), all the above substrates were tested.
P. citrinopileatus successfully grew and colonized all of the alternative substrates used in this study within a period of 17 to 20 days after inoculation. The earliness period ranged from 26 to 30 days and in most cases, HRL supplementation favored a decrease in earliness (
Table 3). In previous studies, further SMS supplementation had a positive role in reducing the total
P. ostreatus,
P. eryngii and
P. pulmonarius cultivation time [
50,
51]. However, this trend was not observed in the present study for
P. citrinopileatus; the growing period of mushrooms depends on many parameters, such as the type of strain, the selected lignocellulosic cultivation substrate, the biological structure of the substrate and the additive ratio. A similar earliness period (26–30 days) was also detected by Kulshreshtha et al. [
52] when they cultivated
P. citrinopileatus on substrates consisting of WS with cardboard industrial waste and WS with handmade paper industrial waste (1:1
w/
w), while a longer earliness period occurred on the control substrate, consisting of 100% WS (38.6 days). Atila [
53] recorded a longer earliness period for
P. citrinopileatus cultivated on oak sawdust, safflower hay, bean straw and sunflower head residue from 38 to 46 days.
Regarding the biological efficiency (B.E., %), although all values were lower than that on the control substrate, satisfactory values (up to 50%) were detected on substrates consisting of 70–80% SMS and 10–20% HRL (
Table 3). The highest B.E. values were recorded on SMS 70%-HRL 20% (61.0%) among the alternative substrates. On the other hand, the highest ratio of HRL led to the lowest B.E. (39.2%), while the B.E. was increased when the other substrate with the highest ratio of HRL supplemented with additives (SMS 50%-HRL 40%) was used. As has already been mentioned, substrate supplementation with protein-rich additives (wheat bran, soybean flour) increases mushroom B.E. [
51,
54]. In the present study, substrate supplementation led to higher B.E. values than the corresponding substrate without additives. For example, the B.E. value was 41.9% on SMS 90%-HRL 10% and increased to 52.7% after additive supplementation in the cultivation substrate SMS 80%-HRL 10%. However, one of the highest B.E. values, 55.9%, was recorded on SMS 80%-HRL 20%, a substrate without additives, indicating that there are crucial physicochemical parameters other than additives that can affect mushroom B.E., such as the availability of nutrients, the moisture content, the cellulose and lignin contents, the pH and the EC of the cultivation substrate. In a previous study, higher B.E. was recorded for
P. eryngii cultivated on a substrate consisting of SMS and
Pleurotus waste (stipes, mishappen mushrooms) without additives (97.97%) than on the same one with additives (87.80%) [
50].
Similar B.E. values were detected for
P. citrinopileatus in various grass plants, from 40 to 65% [
48] and in different agro-residues, from 42.5 to 73.9% [
53]. Kulshreshtha et al. [
52] and Koutrotsios et al. [
55] reported lower B.E. values for
P. citrinopileatus cultivated on WS: 58.2% and 53.7%, respectively. Higher values (88.9–94.5%) were recorded in mixtures of handmade paper and cardboard industrial waste with wheat straw [
52] and in grape marc mixed with wheat straw (78.5%), whereas
P. citrinopileatus B.E. was not favored in olive mill byproducts (26.2%) [
55].
The results of this study revealed the successful bioconversion of alternative substrates, mixtures of SMS and HRL, by P. citrinopileatus, which has not been examined before. In particular, the mushroom grew very well on the SMS 80% substrate and on mixtures of SMS with the lowest supplementation of HRL (SMS 80%-HRL 10%, SMS 80%-HRL 20% and SMS 70%-HRL 30%), indicating that SSF is a possible solution to deal with the mushroom industry’s byproducts and agro-industrial waste.
Concerning the quality of the fruit bodies formed, the average pileus diameter and stipe length of
P. citrinopileatus fruit bodies ranged from 3.7 to 5.2 cm and from 1.9 to 3.2 cm, respectively (
Table 3). In general, no significant differences were noticed in the appearance of
P. citrinopileatus fruit bodies cultivated on different substrates, as has already been mentioned in previous studies [
55,
56]. Jatily et al. [
57] reported a slightly higher stipe length (3.5 cm) and bigger pileus width (7.0 cm) for
P. citrinopileatus cultivated on wheat straw. So, the alternative substrates used in the present study can be utilized to produce high-quality
P. citrinopileatus fruit bodies with commercially desirable morphological characteristics [
58].
3.2. Nutrient Composition of Fruit Bodies/Macronutrients of P. citrinopileatus
The fruit bodies of mushrooms are rich in carbohydrates, proteins and minerals, with very low fat content. Chemical constituents were estimated for fruit bodies produced from the first and the second flushes. The composition reported for
P. citrinopileatus cultivated on different substrates showed that the protein content of fruit bodies from the alternative substrates was significantly higher (25.6 to 34.7% d.w.) compared to the control substrate (24.4% d.w.) (
Table 4). In this study, replacing WS with SMS and adding HRL, which contains more nitrogen, appears to have positively affected the mushroom protein content. Nitrogen is a crucial nutrient for fungal growth, serving as a vital component of amino acids, the building blocks of proteins. So, by increasing the nitrogen content of the substrate, mushroom growers could enhance the protein content of the fruit bodies. This improvement could have a positive impact on the nutritional value and marketability of the mushrooms.
The lipid content was generally low, ranging from 2.6 to 5.1% d.w., in
P. citrinopileatus fruit bodies grown on different substrates. Also, negligible differences were detected in the ash content of
P. citrinopileatus, regardless of the cultivation substrate, with values ranging from 6.7 to 8.4%. The total carbohydrate values ranged between 52.0 and 65.7%, which are considerably high. However, the carbohydrate content estimated by difference includes fiber, as well as some components that are not strictly carbohydrates, e.g., organic acids [
59].
These findings are supported and confirmed by many authors. Singh et al. [
60] reported higher protein content, above 40% and lower total carbohydrates for
P. citrinopileatus cultivated on vegetable waste. Musieba et al. [
19] reported that
P. citrinopileatus contained 22.1% protein, 1.3% crude lipid and 20.78% fiber. Many studies reported that
P. citrinopileatus contained (on a d.w. basis) 16–25% protein, 19–28% carbohydrate and about 9% crude fiber [
61,
62]. Another study by Ahmed et al. [
63] revealed that
Pleurotus species contain 86–90% moisture, 28–31.8% protein, 3.5–4.7% fat and 8.6–12.8% ash on a dry weight basis. Ragunathan [
64] found that mushrooms grown on various agro-residues have a nutrient composition of 41.5–44.4% carbohydrates, 30.1–40.6% proteins and 1.7–2.9% lipids. Overall,
P. citrinopileatus mushrooms emerge as excellent sources of essential nutrients, reaffirming their potential as valuable functional foods.
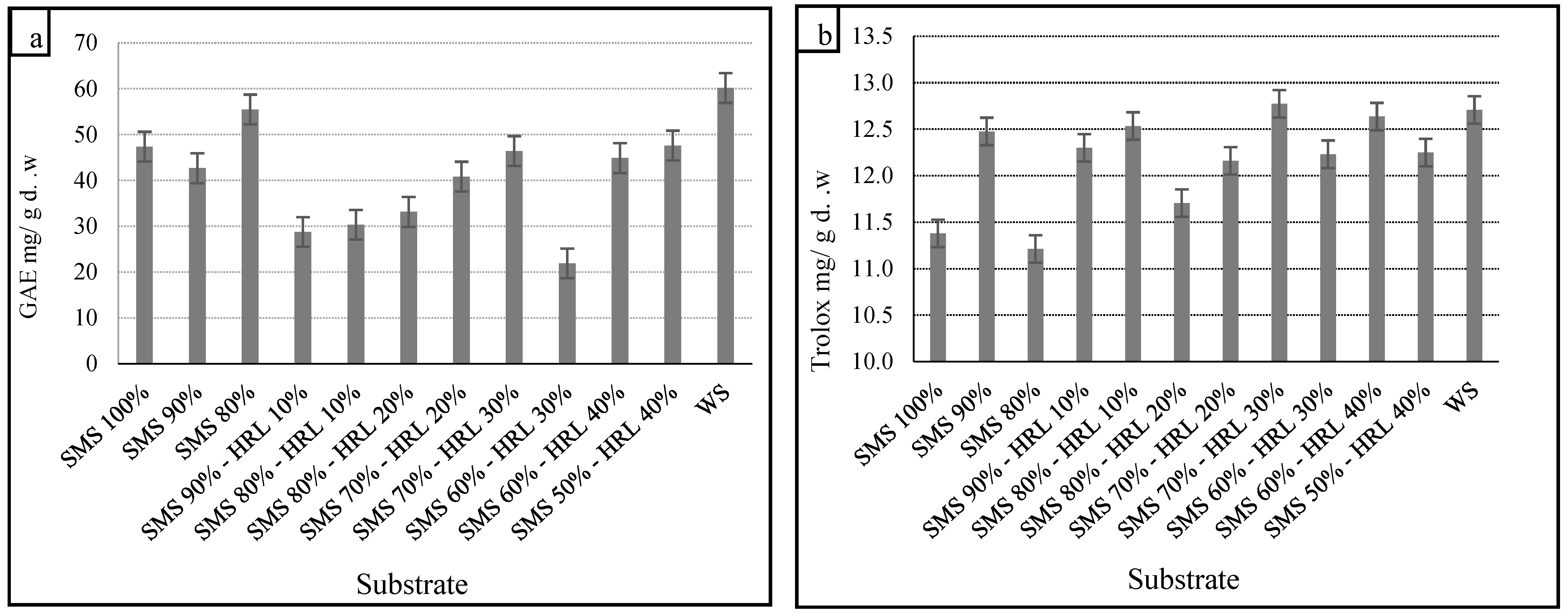
3.3. TPC, TFC, Scavenging Activity of DPPH Radical, Scavenging Activity of ABTS Radical and Ferric Reducing Antioxidant Power (FRAP˙) in Methanol Extracts
Edible mushrooms are known to be a source of phenolic compounds with antioxidant properties [
65]. Our results indicated significant differences in TPCs in the methanol extracts of the mushrooms. Previous studies have reported the impact of the substrate’s chemical composition on the TPCs in mushrooms [
66,
67]. The highest TPC was 60.2 mg GAE/g d.w. on the control substrate, while significantly lower TPC was found on the alternative substrates, with the minimum concentration being 21.9 mg GAE/g d.w. on SMS 60%-HRL 30%. The TPCs in mushroom extracts are shown in
Figure 2a.
The presence of phenolic compounds is an indication of antioxidant activity. Research has shown a strong correlation between antioxidant activity and phenolic content [
68]. Mushroom extracts contain significant quantities of phenolic compounds, which include aromatic rings with hydroxyl groups. These compounds act as free radical scavengers, hydrogen donors, electron donors and metal ion chelators. More hydroxyl groups in phenolics may enhance antioxidant activity [
69]. Most studies show that the DPPH scavenging activities of antioxidants depend on their hydrogen-donating abilities [
70].
Figure 2b demonstrates DPPH scavenging activity, expressed in mg Trolox/g d.w. The potential of mushrooms to scavenge ABTS radicals ranged from 2.3 to 2.5 mg Trolox/g d.w. (
Figure 2c). Antioxidant activity was also measured using the FRAP method and was observed in all extracts (
Figure 2d). The antioxidant activity of extracts of the dried samples ranged from 13.1 to 15.6 mg Trolox/g d.w. Lee et al. [
71] measured values of 2.3, 15.2 and 16.5 mg/mL for ethanolic, cold water and hot water extracts, respectively, in
P. citrinopileatus fruit bodies. Aqueous extracts of
P. citrinopileatus grown on a substrate mix (38%
w/
w sawdust) showed the highest FRAP values (2.4 mg EAA/g extract) and ABTS radical scavenging activity (7.1 mg EAA/g extract) [
63]. The potential of
P. citrinopileatus to scavenge ABTS radicals ranged from 0.6 to 14.1 mg Trolox/g d.w. for the water extract, while the ethanol extract ranged from 0.4 to 11.8 mg Trolox/g d.w. [
72]. Despite the high antioxidant potential described by Freitas et al. [
63] and Lee et al. [
71], different sample processing methods and result expressions do not allow direct comparisons.
The TFC in all samples was low, ranging from 3.4 to 13.4 mg QE/g (
Figure 2e). Flavonoids represented a very small percentage of TPCs. Arbaayah and Umi [
73] reported high flavonoid contents in ethanol extracts of various
Pleurotus species, with values of 1.4 to 29.8 mg QE/g. Jan-Ying et al. [
74] reported the flavonoid content of ethanolic, cold water and hot water extracts of two
Grifola frondosa strains, with values of 1.1–3.1 and 0.1–0.8 mg QE/g dry sample, respectively. Sudha et al. [
75] reported the flavonoid content of ethyl acetate, methanol and hot water extracts of
Pleurotus eous, with values of 6.4 to 7.8 mg catechin equivalents (CAE)/g extract. However, Gil-Ramírez et al. [
76] reported that mushrooms do not contain flavonoids and those found in hyphae may be due to their ability to absorb nutrients and compounds from substrates or neighboring plants by spreading their hyphae or forming mycorrhizae. Some plants release flavonoids to regulate symbiotic plant–microbe interactions, defining the species that can tolerate growth on their roots. Flavonoids are reported to be antifungal compounds produced by plants to protect against fungal infections, which might negatively affect fungal growth.
The Tr content in sample ethanol extracts (mg/g extract) is presented in
Figure 2f. The content per d.w. (mg/g d.w.) varied depending on the substrate, with the maximum Tr value detected in SMS 80%-HRL 20% (9.8 mg UA/g d.w.). According to researchers [
77,
78], triterpenoids and polysaccharides are valuable biologically active substances in mushrooms. Li et al. [
79] reported that the d.w. of total triterpenoid content in
G. lucidum fruit bodies ranged from 13.6 to 31.5 mg/g, higher than our results. Boh et al. [
80] found that the total triterpenoid content was higher at younger growth stages of Indonesian
Ganoderma applanatum than at older stages. Feng et al. [
81] also demonstrated that the total triterpenoid content at the immature stage was much higher than at the mature stages. In conclusion, edible mushrooms are rich sources of phenolic compounds, which exhibit significant antioxidant properties.
3.4. Fatty Acid (FA) Composition and Individual Saccharide Profile
From the data presented in
Table 5, it was found that the PUFA concentration ranged from 71.2 to 82.5%,
w/
w, whereas saturated FAs varied from 0.2 to 13.9%,
w/
w, significantly lower concentrations. This agrees with many previous observations that unsaturated FAs predominate over saturated ones in mushroom mycelia and fruit bodies [
31,
82]. All samples showed high linoleic acid (C18:2) concentrations, above 60 g FA/100 gfat, indicating that this is the main FA in the composition of
P. citrinopileatus (63.8–75.2 g FA/100 g fd.w.). The maximum concentration of linoleic acid was detected in fruit bodies cultivated on SMS 60%-HRL 30%. The FA composition of the mushroom species was characterized by much higher contents of mono- and polyunsaturated FAs than saturated FAs. The saturated FA distribution was characterized by palmitic (C16:0) and stearic acids (C18:0). A higher content of palmitic acid was measured in
P. citrinopileatus (10.8–13.9 g FA/100 g d.w.). Interestingly, the concentration of linoleic acid was higher than 63%, but the concentration of oleic acid (C18:1) was the lowest (4.6–9.4 g FA/100 gfat). The FA composition of edible mushrooms contains a wide range of FAs, from lauric acid (C12:0) to docosahexaenoic acid (C22:6), depending on the mushroom species. However, the main FAs typically found are C16:0, C18:1 and C18:2 [
31,
82,
83]. Previous studies on
P. citrinopileatus have reported similar observations regarding fatty acid composition. Linoleic acid (C18:2) was the most abundant FA in mushrooms, ranging from 54.5 to 70.5% of total FAs, followed by palmitic acid (C16:0; 12.3–14.3%) and oleic acid (C18:1; 3.9–4.9%) [
55]. Rodrigues et al. [
10] reported higher levels of palmitic acid (16.5%) and similar levels of oleic acid (5.5%), while Reis et al. [
84,
85] found that the saturated FAs ranged from 17.4 to 25.8%, the monounsaturated fatty acids ranged from 13 to 49% and the PUFAs ranged from 25.2 to 69.4% in wild
Pleurotus spp. To our knowledge, this is the first study describing the FA composition of the edible
P. citrinopileatus; therefore, this work provides valuable data on the nutritional evaluation of such mushroom species.
The carbohydrate composition analysis results are summarized in
Table 6. Fructose was identified as the primary constituent in
P. citrinopileatus fruit bodies when the substrate mix contained less than 80% SMS (up to 70%,
w/
w). Glucose was presented in smaller quantities, around 52%,
w/
w, while mannose was detected in lower concentrations or not at all in some samples. Fructose, commonly known as fruit sugar, is distinctly sweeter than glucose and sucrose, with its perceived sweetness varying based on its concentration and the presence of other compounds. It is essential to consume fructose in moderation as part of a balanced diet [
86,
87]. Generally, differences in sugar composition have been observed between cultivated and wild samples of the same mushroom species, likely due to different cultivation techniques [
88].
3.5. Effect of Protein and Carbohydrate Extracts on Expression of Antioxidant Genes
This study is the first to present substantial evidence regarding the antioxidant-related gene expression induced by the protein extract (PE-D-P3) and polysaccharide extract (CE-D) of P. citrinopileatus following in vitro gastrointestinal digestion. Firstly, the cell viability of both groups of mushroom extracts was tested using activated THP-1 macrophages. A PE-D-P3 concentration of 4 mg/mL (10% PE-D-P3 and 90% RPMI) and a CE-D concentration of 0.34 mg/mL (15% CE-D and 85% RPMI) were found to be nontoxic to THP-1 cells and consequently, these concentrations were selected.
The relative gene expression of key components in antioxidant signaling pathways in response to treatment with digested protein and carbohydrate extracts (PE-D-P3 and CE-D, respectively) was assessed in LPS-challenged THP-1-derived macrophages.
Figure 3 and
Figure 4 show the quantification of transcription levels of a panel of genes associated with antioxidant activity. As shown in
Figure 3, no statistically significant differences were observed in the PE-D-P3 samples either between different substrates or compared to BL-D-P3-treated cells regarding
NFE2L2,
SOD1,
CAT,
HMOX1 and
GSR expression (
p > 0.05). However, CE-D treatments significantly (
p < 0.05) altered the expression of some of the genes mentioned above. Specifically,
NFE2L2 expression was found to be higher (
p < 0.05) in SMS 60%-HRL 40%-treated cells when compared to WS (control). No increase, however, was observed in the other SMS groups, with or without HRL, when compared to BL-D and WS (
p > 0.05;
Figure 4a).
SOD1 expression, in turn, was higher (
p < 0.05;
Figure 4b) in all SMS groups, with or without the addition of HRL, when compared to WS, but no statistical difference was observed compared to BL-D. On the other hand, exposing THP-1 macrophages to a CE cultivated on a specific substrate, a combination of 60% SMS and 30% HRL, increased the gene expression of
CAT,
HMOX1 and
GSR in activated THP-1 macrophages compared to the WS group. No significant differences were observed between any of the CE digests and BL-D (
p > 0.05;
Figure 4c,
Figure 4d and
Figure 4e, respectively).
Pleurotus sp. consists of a plethora of bioactive compounds, including proteins, polysaccharides like α- and β-glucans, peptides and dietary fiber, among others. These components may offer potential functional and nutritional benefits [
89]. Additionally, fungal polysaccharides have been shown to have a positive effect on human health [
23], with the antioxidant activity of polysaccharides in vivo typically associated with the enhanced activities of oxidative enzymes, including catalase and superoxide dismutase [
90].
The ‘nuclear factor erythroid 2-related factor/heme oxygenase-1’ (Nrf2/HO-1) pathway has recently emerged as a valuable target, acting as a primary cellular sensor of oxidative stress. Nowadays, scientists are actively seeking novel Nrf2 inducers from natural sources [
91]. After its nuclear translocation, Nrf2 binds to antioxidant-responsive elements (AREs), stimulating the transcription of numerous cytoprotective genes. This enhances antioxidant enzymes like SOD and CAT, as well as inducible protective genes such as HO-1. In parallel, Nrf2 is crucial for activating target genes, including HO-1, which is the only highly inducible isoform and a key enzyme in regulating heme catabolism. This process reduces cellular ROS release and promotes various cellular defense mechanisms [
92]. Moreover, evidence suggests that increased Nrf2/HO-1 activity can directly downregulate the nuclear factor-kappa B (NF-κB) pathway, reducing a range of inflammatory responses and demonstrating significant anti-inflammatory properties [
93]. Thus, positively influencing the Nrf2/HO-1 pathway is expected to serve dual purposes. This study indicates that using different substrates, such as SMS or a combination of SMS and HRL, can enhance Nrf2 signaling and the expression of related genes. This research highlights the link between increased nitrogen in the substrate due to HRL addition and the upregulation of the antioxidant genes mentioned only for D-CE. However, there are limited studies on digested food, particularly mushrooms and their impact on the expression of genes like
NFE2L2,
SOD1,
CAT,
HMOX1 and
GSR, which are implicated in the oxidative response in macrophage-like THP1 cells. Some mushroom substances with antioxidant properties act as inducers or cell signals, leading to changes in gene expression that activate enzymes to eliminate ROS [
94].
Antioxidative enzymes, including SOD, CAT and glutathione (GPx), serve as the first line of defense against ROS in organisms [
95]. Xiaoping et al. [
96] showed that
G. lucidum polysaccharides could significantly enhance antioxidant enzyme activities (SOD, CAT and GPx) in rats with cervical cancer. Chuang et al. [
97] evaluated waste mushroom compost, the main byproduct of the cultivation of
Pleurotus eryngii, as a feed supplement for broilers and concluded that all antioxidant-related mRNA levels, including
Nrf2,
HMOX1 and
SOD1, increased significantly following the reuse of mushroom waste compost compared to the control group. Similarly, another study [
98] evaluated the effect of dietary supplementation with
Antrodia cinnamomea mycelia powder on the mRNA expression levels of selected antioxidant genes, particularly those regulated by
NFE2L2. This evaluation was conducted by isolating cells from broilers after they had digested and absorbed the supplemented feed and were then exposed to an LPS challenge. It is noteworthy that the expression of antioxidant genes, including
HMOX1,
SOD1 and
NFE2L2, was significantly elevated compared to the control group. Li et al. [
24] evaluated the antioxidant effects of a polysaccharide–peptide complex from
P. abalonus fruiting bodies and observed that the administration of this extract to aged mice had enhanced the activities of CAT and SOD enzymes with the concurrent upregulation of gene transcripts of
SOD1 and
CAT compared to the control group. Also, a thorough review by Kozarski et al. [
94] covers the antioxidants found in edible mushrooms and shows that a small number of studies have studied the
Pleurotus genus in this context. Still, there have been no studies on the evaluation of different substrates and their possible link to genes related to antioxidant activity in vitro.