Diagnostic Potential of IgG and IgA Responses to Mycobacterium tuberculosis Antigens for Discrimination among Active Tuberculosis, Latent Tuberculosis Infection, and Non-Infected Individuals
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Clinical Samples
2.2. Preparation of Mycobacterium tuberculosis (Mtb) Antigens
2.3. Antibody Enzyme-Linked Immunosorbent Assay (ELISA)
2.4. Data Analysis
2.5. Ethics Statement
3. Results
3.1. Characteristics of the Study Participants
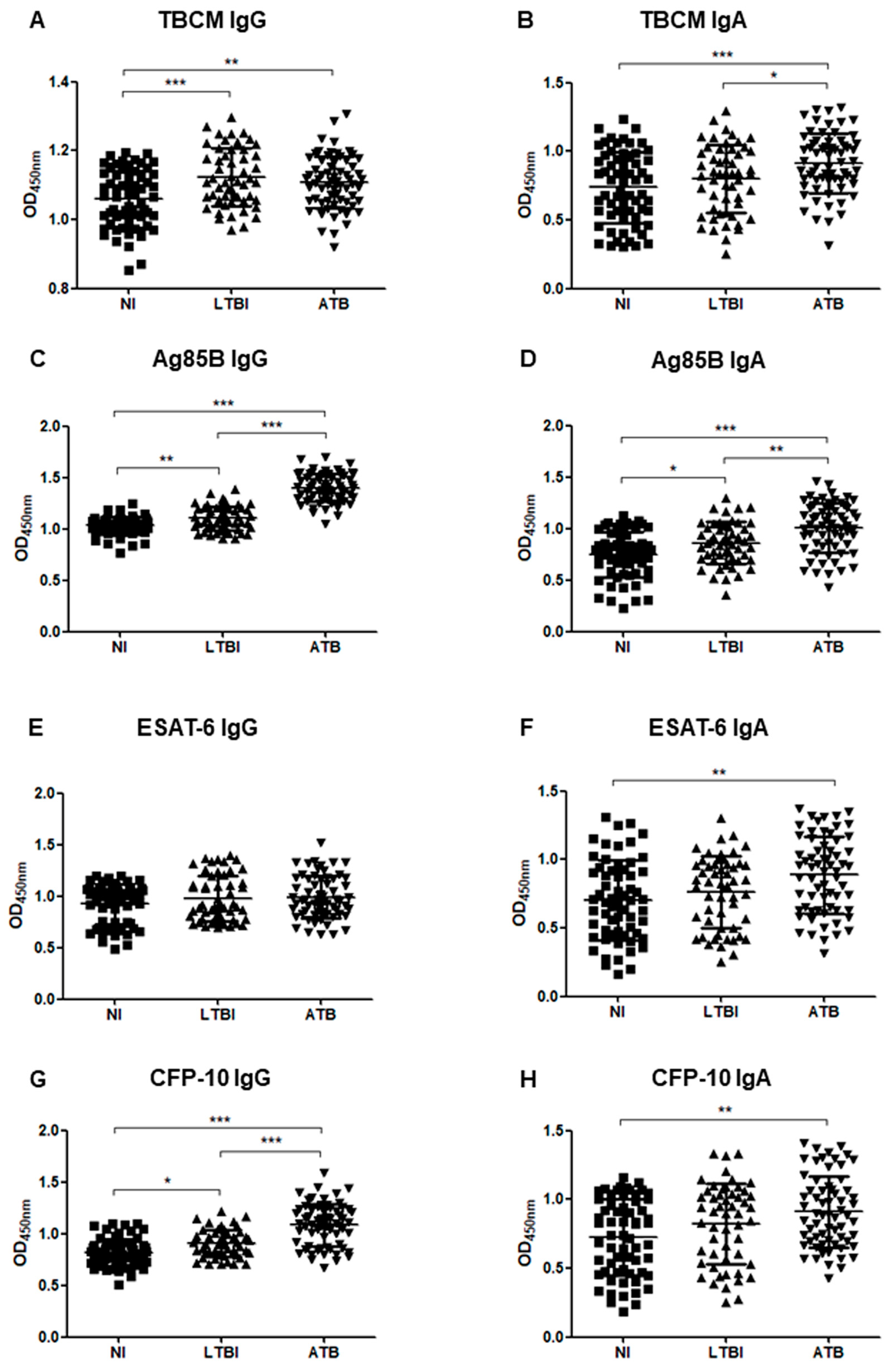
3.2. Serum Levels of Immunoglobulin G (IgG) and Immunoglobulin A (IgA) against Mycobacterium Tuberculosis Chorismate Mutase (TBCM), Antigen 85B (Ag85B), Early Secreted Antigen-6 (ESAT-6), and Culture Filtrate Protein-10 (CFP-10)
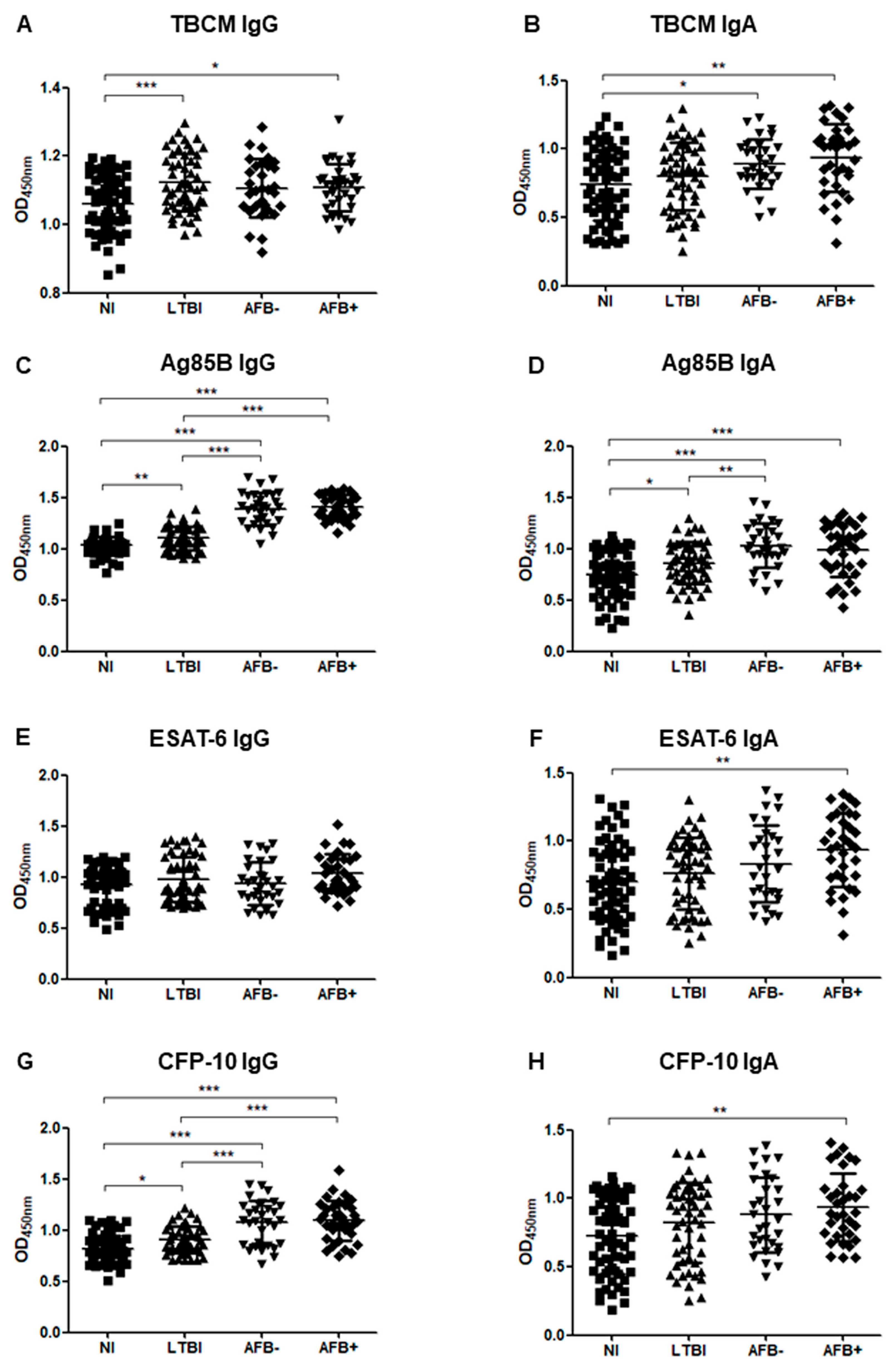
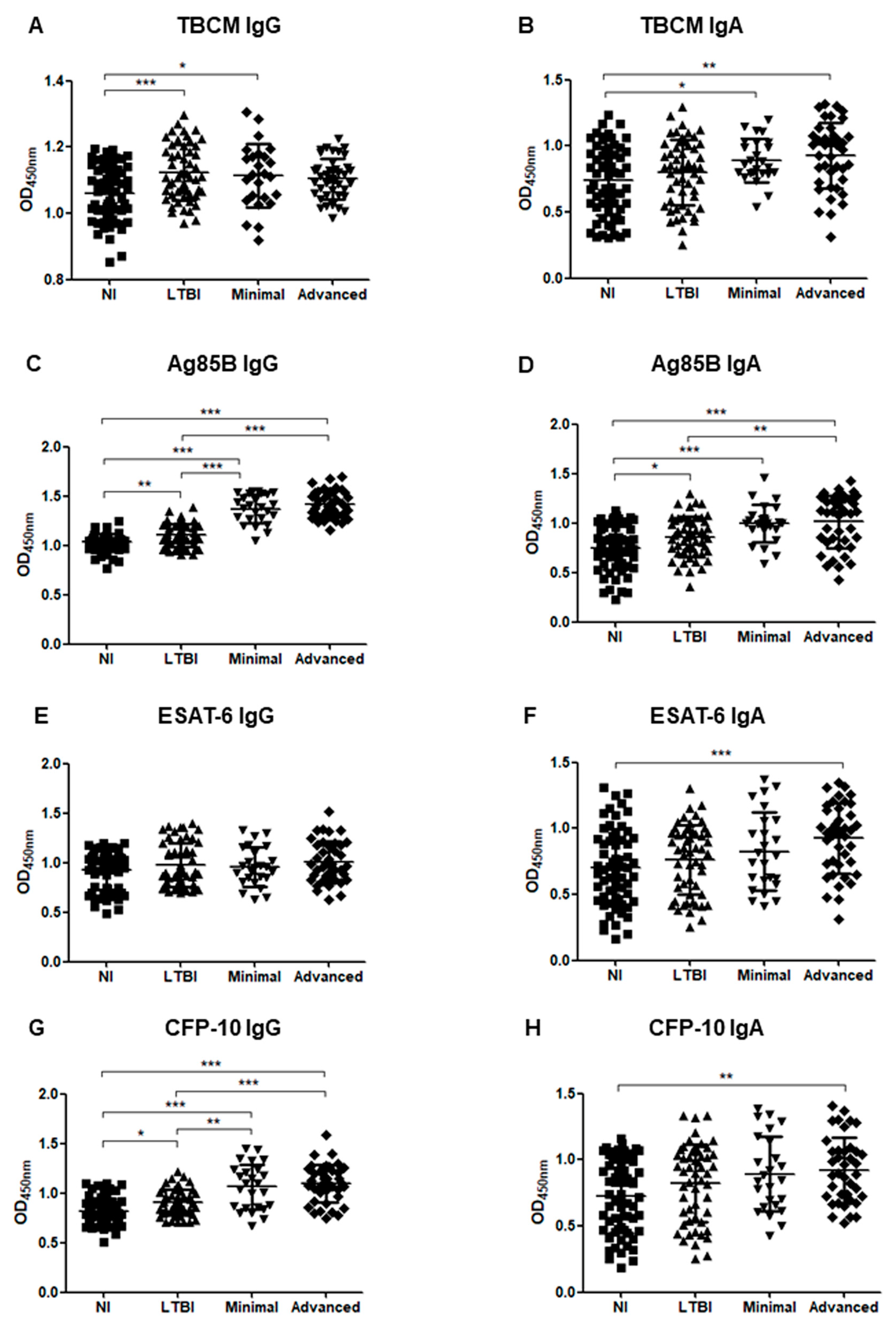
3.3. Serum Levels of IgG and IgA in Subgroup Analysis
3.4. Correlations among Serum Levels of IgA, IgG, and Interferon-Gamma (IFN-γ)
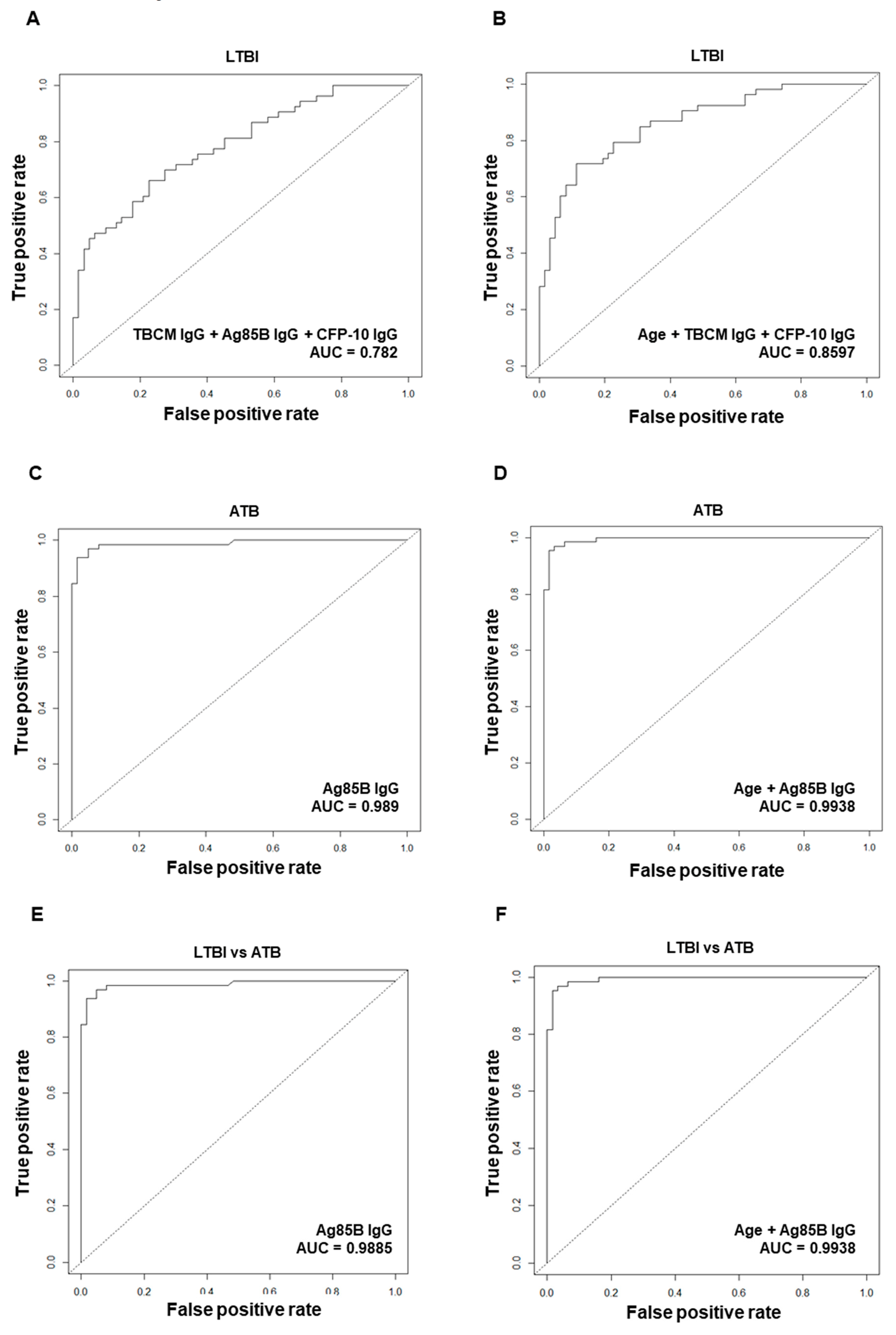
3.5. Diagnostic Performance of Antibodies against Mtb Antigens for Diagnosis of Latent Tuberculosis Infection (LTBI) or Active Tuberculosis (ATB)
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2019; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Kim, H.R.; Shin, J.Y.; Kim, H.A.; Lee, J.E.; Kim, J.H.; Kong, I.S. Notified Tuberculosis in the Republic of Korea, 2018. Public Health Wkly. Rep. 2019, 12, 366–371. [Google Scholar]
- Ministry of Health and Welfare, Korean Institute of Tuberculosis, Korean National Tuberculosis Association. Report on the 7th Tuberculosis Prevalence Survey in Korea; Korean Institute of Tuberculosis: Seoul, Korea, 1996.
- Joint Committee for the Revision of Korean Guidelines for Tuberculosis Korea Centers for Disease Control and Prevention. Korean guidelines for tuberculosis, 3rd ed.; Korea Centers for Disease Control and Prevention: Cheongju, Korea, 2017.
- Barry, C.E., 3rd; Boshoff, H.I.; Dartois, V.; Dick, T.; Ehrt, S.; Flynn, J.; Schnappinger, D.; Wilkinson, R.J.; Young, D. The spectrum of latent tuberculosis: Rethinking the biology and intervention strategies. Nat. Rev. Microbiol. 2009, 7, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Canadian Thoracic Society. Canadian Tuberculosis Standards, 7th ed.; The Public Health Agency of Canada: Ottawa, ON, Canada, 2013.
- Pai, M.; Denkinger, C.M.; Kik, S.V.; Rangaka, M.X.; Zwerling, A.; Oxlade, O.; Metcalfe, J.Z.; Cattamanchi, A.; Dowdy, D.W.; Dheda, K.; et al. Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clin. Microbiol. Rev. 2014, 27, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Legesse, M.; Ameni, G.; Medhin, G.; Mamo, G.; Franken, K.L.; Ottenhoff, T.H.; Bjune, G.; Abebe, F. IgA response to ESAT-6/CFP-10 and Rv2031 antigens varies in patients with culture-confirmed pulmonary tuberculosis, healthy Mycobacterium tuberculosis-infected and non-infected individuals in a tuberculosis endemic setting, Ethiopia. Scand. J. Immunol. 2013, 78, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Kunnath-Velayudhan, S.; Davidow, A.L.; Wang, H.Y.; Molina, D.M.; Huynh, V.T.; Salamon, H.; Pine, R.; Michel, G.; Perkins, M.D.; Xiaowu, L.; et al. Proteome-scale antibody responses and outcome of Mycobacterium tuberculosis infection in nonhuman primates and in tuberculosis patients. J. Infect. Dis. 2012, 206, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Berthet, F.X.; Rasmussen, P.B.; Rosenkrands, I.; Andersen, P.; Gicquel, B. A Mycobacterium tuberculosis operon encoding ESAT-6 and a novel low-molecular-mass culture filtrate protein (CFP-10). Microbiology 1998, 144, 3195–3203. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.; Munk, M.E.; Pollock, J.M.; Doherty, T.M. Specific immune-based diagnosis of tuberculosis. Lancet 2000, 356, 1099–1104. [Google Scholar] [CrossRef]
- Williams, A.; Hatch, G.J.; Clark, S.O.; Gooch, K.E.; Hatch, K.A.; Hall, G.A.; Huygen, K.; Ottenhoff, T.H.; Franken, K.L.; Andersen, P.; et al. Evaluation of vaccines in the EU TB Vaccine Cluster using a guinea pig aerosol infection model of tuberculosis. Tuberculosis 2005, 85, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Langermans, J.A.; Doherty, T.M.; Vervenne, R.A.; van der Laan, T.; Lyashchenko, K.; Greenwald, R.; Agger, E.M.; Aagaard, C.; Weiler, H.; van Soolingen, D.; et al. Protection of macaques against Mycobacterium tuberculosis infection by a subunit vaccine based on a fusion protein of antigen 85B and ESAT-6. Vaccine 2005, 23, 2740–2750. [Google Scholar] [CrossRef]
- Belisle, J.T.; Vissa, V.D.; Sievert, T.; Takayama, K.; Brennan, P.J.; Besra, G.S. Role of the major antigen of Mycobacterium tuberculosis in cell wall biogenesis. Science 1997, 276, 1420–1422. [Google Scholar] [CrossRef]
- Boesen, H.; Jensen, B.N.; Wilcke, T.; Andersen, P. Human T-cell responses to secreted antigen fractions of Mycobacterium tuberculosis. Infect. Immun. 1995, 63, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Klein, M.R.; Malin, A.S.; Sillah, J.; Huygen, K.; Andersen, P.; McAdam, K.P.; Dockrell, H.M. Human CD8(+) T cells specific for Mycobacterium tuberculosis secreted antigens in tuberculosis patients and healthy BCG-vaccinated controls in The Gambia. Infect. Immun. 2000, 68, 7144–7148. [Google Scholar] [CrossRef] [PubMed]
- Khanapur, M.; Alvala, M.; Prabhakar, M.; Shiva Kumar, K.; Edwin, R.K.; Sri Saranya, P.S.; Patel, R.K.; Bulusu, G.; Misra, P.; Pal, M. Mycobacterium tuberculosis chorismate mutase: A potential target for TB. Bioorg. Med. Chem. 2017, 25, 1725–1736. [Google Scholar] [CrossRef] [PubMed]
- Aghababa, H.; Mobarez, A.M.; Behmanesh, M.; Khoramabadi, N.; Mobarhan, M. Production and Purification of Mycolyl Transferase B of Mycobacterium tuberculosis. Tanaffos 2011, 10, 23–30. [Google Scholar]
- Kim, B.J.; Gong, J.R.; Kim, G.N.; Kim, B.R.; Lee, S.Y.; Kook, Y.H.; Kim, B.J. Recombinant Mycobacterium smegmatis with a pMyong2 vector expressing Human Immunodeficiency Virus Type I Gag can induce enhanced virus-specific immune responses. Sci. Rep. 2017, 7, 4477. [Google Scholar] [CrossRef]
- Abebe, F.; Belay, M.; Legesse, M.; KLMC, F.; Ottenhoff, T.H.M. IgA and IgG against Mycobacterium tuberculosis Rv2031 discriminate between pulmonary tuberculosis patients, Mycobacterium tuberculosis-infected and non-infected individuals. PLoS ONE 2018, 13, e0190989. [Google Scholar] [CrossRef]
- Kim, S.K.; Reddy, S.K.; Nelson, B.C.; Vasquez, G.B.; Davis, A.; Howard, A.J.; Patterson, S.; Gilliland, G.L.; Ladner, J.E.; Reddy, P.T. Biochemical and structural characterization of the secreted chorismate mutase (Rv1885c) from Mycobacterium tuberculosis H37Rv: An *AroQ enzyme not regulated by the aromatic amino acids. J. Bacteriol. 2006, 188, 8638–8648. [Google Scholar] [CrossRef]
- Parish, T. Starvation survival response of Mycobacterium tuberculosis. J. Bacteriol. 2003, 185, 6702–6706. [Google Scholar] [CrossRef]
- Betts, J.C.; Lukey, P.T.; Robb, L.C.; McAdam, R.A.; Duncan, K. Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Mol. Microbiol. 2002, 43, 717–731. [Google Scholar] [CrossRef]
- Demissie, A.; Leyten, E.M.; Abebe, M.; Wassie, L.; Aseffa, A.; Abate, G.; Fletcher, H.; Owiafe, P.; Hill, P.C.; Brookes, R.; et al. Recognition of stage-specific mycobacterial antigens differentiates between acute and latent infections with Mycobacterium tuberculosis. Clin. Vaccine Immunol. 2006, 13, 179–186. [Google Scholar] [CrossRef]
- Daniel, T.M.; Janicki, B.W. Mycobacterial antigens: A review of their isolation, chemistry, and immunological properties. Microbiol. Rev. 1978, 42, 84–113. [Google Scholar] [CrossRef] [PubMed]
- Steingart, K.R.; Flores, L.L.; Dendukuri, N.; Schiller, I.; Laal, S.; Ramsay, A.; Hopewell, P.C.; Pai, M. Commercial serological tests for the diagnosis of active pulmonary and extrapulmonary tuberculosis: An updated systematic review and meta-analysis. PLoS Med. 2011, 8, e1001062. [Google Scholar] [CrossRef]
- Steingart, K.R.; Dendukuri, N.; Henry, M.; Schiller, I.; Nahid, P.; Hopewell, P.C.; Ramsay, A.; Pai, M.; Laal, S. Performance of purified antigens for serodiagnosis of pulmonary tuberculosis: A meta-analysis. Clin. Vaccine Immunol. 2009, 16, 260–276. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Dagur, P.K.; Singh, P.K.; Shankar, H.; Yadav, V.S.; Katoch, V.M.; Bajaj, B.; Gupta, R.; Sengupta, U.; Joshi, B. Serodiagnostic efficacy of Mycobacterium tuberculosis 30/32-kDa mycolyl transferase complex, ESAT-6, and CFP-10 in patients with active tuberculosis. Arch. Immunol. Ther. Exp. 2010, 58, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Raja, A.; Uma Devi, K.R.; Ramalingam, B.; Brennan, P.J. Improved diagnosis of pulmonary tuberculosis by detection of free and immune complex-bound anti-30 kDa antibodies. Diagn. Microbiol. Infect. Dis. 2004, 50, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Song, X.; Zhao, Y.; Zhang, H.; Zhao, S.; Mao, F.; Bai, B.; Wu, S.; Shi, C. Mycobacterium tuberculosis Secreted Proteins as Potential Biomarkers for the Diagnosis of Active Tuberculosis and Latent Tuberculosis Infection. J. Clin. Lab. Anal. 2015, 29, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Yang, X.; Xiu, B.; Qie, S.; Dai, Z.; Chen, K.; Zhao, P.; Zhang, L.; Nicholson, R.A.; Wang, G.; et al. IgG, IgM and IgA antibodies against the novel polyprotein in active tuberculosis. BMC Infect. Dis. 2014, 14, 336. [Google Scholar] [CrossRef]
- Welch, R.J.; Lawless, K.M.; Litwin, C.M. Antituberculosis IgG antibodies as a marker of active Mycobacterium tuberculosis disease. Clin. Vaccine Immunol. 2012, 19, 522–526. [Google Scholar] [CrossRef]
- Broger, T.; Basu Roy, R.; Filomena, A.; Greef, C.H.; Rimmele, S.; Havumaki, J.; Danks, D.; Schneiderhan-Marra, N.; Gray, C.M.; Singh, M.; et al. Diagnostic Performance of Tuberculosis-Specific IgG Antibody Profiles in Patients with Presumptive Tuberculosis from Two Continents. Clin. Infect. Dis. 2017, 64, 947–955. [Google Scholar] [CrossRef]
- Alifano, M.; De Pascalis, R.; Sofia, M.; Faraone, S.; Del Pezzo, M.; Covelli, I. Detection of IgG and IgA against the mycobacterial antigen A60 in patients with extrapulmonary tuberculosis. Thorax 1998, 53, 377–380. [Google Scholar] [CrossRef]
- Julian, E.; Matas, L.; Perez, A.; Alcaide, J.; Laneelle, M.A.; Luquin, M. Serodiagnosis of tuberculosis: Comparison of immunoglobulin A (IgA) response to sulfolipid I with IgG and IgM responses to 2,3-diacyltrehalose, 2,3,6-triacyltrehalose, and cord factor antigens. J. Clin. Microbiol. 2002, 40, 3782–3788. [Google Scholar] [CrossRef]
- Conde, M.B.; Suffys, P.; Lapa, E.S.J.R.; Kritski, A.L.; Dorman, S.E. Immunoglobulin A (IgA) and IgG immune responses against P-90 antigen for diagnosis of pulmonary tuberculosis and screening for Mycobacterium tuberculosis infection. Clin. Diagn. Lab. Immunol. 2004, 11, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Reljic, R.; Williams, A.; Ivanyi, J. Mucosal immunotherapy of tuberculosis: Is there a value in passive IgA? Tuberculosis 2006, 86, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Grange, J.M.; Gibson, J.; Nassau, E.; Kardjito, T. Enzyme-linked immunosorbent assay (ELISA): A study of antibodies to Mycobacterium tuberculosis in the IgG, IgA and IgM classes in tuberculosis, sarcoidosis and Crohn’s disease. Tubercle 1980, 61, 145–152. [Google Scholar] [CrossRef]
- Gupta, S.; Kumari, S.; Banwalikar, J.N.; Gupta, S.K. Diagnostic utility of the estimation of mycobacterial Antigen A60 specific immunoglobulins IgM, IgA and IgG in the sera of cases of adult human tuberculosis. Tuber. Lung Dis. 1995, 76, 418–424. [Google Scholar] [CrossRef]
- O’Connor, J.E.; Snyder, I.S. Kinetics and immunoglobulin nature of the antibody response to Mycobacterium tuberculosis. J. Bacteriol. 1967, 94, 815–820. [Google Scholar] [CrossRef]
- Alarcon-Segovia, D.; Fishbein, E. Serum immunoglobulins in pulmonary tuberculosis. Chest 1971, 60, 133–136. [Google Scholar] [CrossRef]



| NI (n = 62) | LTBI (n = 53) | ATB (n = 65) | |
|---|---|---|---|
| Demographics | |||
| Age (years), median (IQR) | 33 (29–33) | 49 (33–57) | 60 (51–67) |
| Sex | |||
| Male | 21 (33.9%) | 21 (39.6%) | 56 (86.2%) |
| Female | 41 (66.1%) | 32 (60.4%) | 9 (13.8%) |
| Body-mass index (kg/m2), median (IQR) | 22.4 (20.2–24.9) | 23.7 (21.4–26.7) | 20 (17.0–21.7) |
| Comorbidities | |||
| Diabetes | 3 (4.8%) | 6 (11.3%) | 14 (21.5%) |
| Chronic alcoholics | 0 (0%) | 1 (1.9%) | 9 (13.8%) |
| Cancer | 0 (0%) | 5 (9.4%) | 4 (6.2%) |
| Liver disease | 0 (0%) | 4 (7.5%) | 7 (10.8%) |
| Chronic kidney disease | 0 (0%) | 2 (3.8%) | 2 (3.1%) |
| Heart disease | 0 (0%) | 3 (5.7%) | 3 (4.6%) |
| Previous history of TB treatment | n/a | n/a | |
| New patients | 34 (52.3%) | ||
| Retreatment | 27 (41.5%) | ||
| Unknown previous TB treatment history | 4 (6.2%) | ||
| Bacteriological examinations | |||
| Acid-fast staining of sputum | n/a | n/a | |
| Negative | 31 (47.7%) | ||
| 1+ | 16 (24.6%) | ||
| 2+ | 7 (10.8%) | ||
| 3+ | 5 (7.7%) | ||
| 4+ | 6 (9.2%) | ||
| Positive Mtb culture of sputum | n/a | n/a | |
| Liquid media | 46 (70.8%) | ||
| Solid media | 39 (60.0%) | ||
| Positive Xpert MTB/RIF assay | n/a | n/a | 59 (90.8%) |
| Drug resistance | n/a | n/a | 14 (21.5%) |
| Radiographical examinations | |||
| Normal | 61 (98.4%) | 51 (96.2%) | n/a |
| Previously healed TB * | 1 (1.6%) | 2 (3.8%) | n/a |
| Radiologic severity of ATB | n/a | n/a | |
| Minimal | 25 (38.5%) | ||
| Advanced | 40 (61.5%) | ||
| Presence of cavity | n/a | n/a | 31 (47.7%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.Y.; Kim, B.-J.; Koo, H.-K.; Kim, J.; Kim, J.-m.; Kook, Y.-H.; Kim, B.-J. Diagnostic Potential of IgG and IgA Responses to Mycobacterium tuberculosis Antigens for Discrimination among Active Tuberculosis, Latent Tuberculosis Infection, and Non-Infected Individuals. Microorganisms 2020, 8, 979. https://doi.org/10.3390/microorganisms8070979
Lee JY, Kim B-J, Koo H-K, Kim J, Kim J-m, Kook Y-H, Kim B-J. Diagnostic Potential of IgG and IgA Responses to Mycobacterium tuberculosis Antigens for Discrimination among Active Tuberculosis, Latent Tuberculosis Infection, and Non-Infected Individuals. Microorganisms. 2020; 8(7):979. https://doi.org/10.3390/microorganisms8070979
Chicago/Turabian StyleLee, Ji Yeon, Byoung-Jun Kim, Hyeon-Kyoung Koo, Junghyun Kim, Jee-min Kim, Yoon-Hoh Kook, and Bum-Joon Kim. 2020. "Diagnostic Potential of IgG and IgA Responses to Mycobacterium tuberculosis Antigens for Discrimination among Active Tuberculosis, Latent Tuberculosis Infection, and Non-Infected Individuals" Microorganisms 8, no. 7: 979. https://doi.org/10.3390/microorganisms8070979
APA StyleLee, J. Y., Kim, B.-J., Koo, H.-K., Kim, J., Kim, J.-m., Kook, Y.-H., & Kim, B.-J. (2020). Diagnostic Potential of IgG and IgA Responses to Mycobacterium tuberculosis Antigens for Discrimination among Active Tuberculosis, Latent Tuberculosis Infection, and Non-Infected Individuals. Microorganisms, 8(7), 979. https://doi.org/10.3390/microorganisms8070979






