Abstract
Members of the genus Spiroplasma are Gram-positive bacteria without cell walls. Some Spiroplasma species can cause disease in arthropods such as bees, whereas others provide their host with resistance to pathogens. Ticks also harbour Spiroplasma, but their role has not been elucidated yet. Here, the infection status and genetic diversity of Spiroplasma in ticks were investigated using samples collected from different geographic regions in Japan. A total of 712 ticks were tested for Spiroplasma infection by PCR targeting 16S rDNA, and Spiroplasma species were genetically characterized based on 16S rDNA, ITS, dnaA, and rpoB gene sequences. A total of 109 samples originating from eight tick species were positive for Spiroplasma infection, with infection rates ranging from 0% to 84% depending on the species. A linear mixed model indicated that tick species was the primary factor associated with Spiroplasma infection. Moreover, certain Spiroplasma alleles that are highly adapted to specific tick species may explain the high infection rates in Ixodes ovatus and Haemaphysalis kitaokai. A comparison of the alleles obtained suggests that horizontal transmission between tick species may not be a frequent event. These findings provide clues to understand the transmission cycle of Spiroplasma species in wild tick populations and their roles in host ticks.
1. Introduction
Members of the genus Spiroplasma are Gram-positive bacteria without cell walls. They are known as symbionts of arthropods and plants and are classified into three major clades based on the 16S ribosomal RNA gene (rDNA) sequence: Ixodetis, Citri-Chrysopicola-Mirum (CCM), and Apis [1,2]. Spiroplasma is one of the most common endosymbionts with a wide range of hosts, including insects, arachnids, crustaceans, and plants [3]. It is estimated that 5–10% of insect species harbor this symbiont group [2,4].
Spiroplasma has a wide range of fitness effects and transmission strategies [2,5,6,7,8,9,10,11,12,13,14,15,16,17]. Some Spiroplasma species affect the sex ratio by inducing male killing in hosts such as flies, butterflies, and ladybird beetles [7,8,9,10]. Several Spiroplasma species are known to cause disease in arthropods such as bees and plants [6,17,18]. On the other hand, some flies infected with Spiroplasma can develop resistance to other pathogens [5,10,11,12]. A wide range of symbiotic relationships involving Spiroplasma have been observed [5,7,8,14,15,16]. The rapid spread of Spiroplasma infection in fruit fly natural populations has been reported in some areas of North America, and this phenomenon has been confirmed in laboratory settings [19]. This characteristic of Spiroplasma is not only biologically interesting, but also useful for symbiotic control applications among host individuals [20].
Ticks have long been studied, since they transmit a variety of pathogens to humans and animals. Spiroplasma mirum is the first reported tick-associated Spiroplasma, which was obtained from Haemaphysalis leporispalustris in the United States in 1982 during the search for rickettsiae in ticks [21]. Another species, S. ixodetis, was isolated from Ixodes pacificus in the United States in 1981 [22]. Thus far, these two species are the only validated Spiroplasma species detected in ticks. Nevertheless, several alleles or putative new species of Spiroplasma have been found in various tick species such as I. arboricola, I. frontalis, I. ovatus, I. persulcatus, I. ricinus, I. uriae, Dermacentor marginatus, Rhipicephalus annulatus, R. decoloratus, R. geigyi, and R. pusillus [23,24,25,26,27,28,29,30].
In Japan, 46 tick species belonging to seven genera (Amblyomma, Argas, Dermacentor, Rhipicephalus, Haemaphysalis, Ixodes, and Ornithodoros) have been recorded [11,12]. Several tick-borne diseases such as Lyme disease, relapsing fever, Japanese spotted fever, severe fever with thrombocytopenia syndrome, and tick-borne encephalitis are endemic [31]. Taroura et al. first detected Spiroplasma DNA in questing I. ovatus ticks captured in several prefectures [24]. Subsequently, a microbiome study revealed the presence of Spiroplasma in the salivary glands of I. ovatus and I. persulcatus [23]. More recently, several Spiroplasma isolates were obtained by incubating the homogenates of I. monospinosus, I. persulcatus, and H. kitaokai with tick and mosquito cells [32]. These studies collectively indicate that there is a close relationship between Spiroplasma and ticks in Japan; however, no comprehensive studies have been conducted to determine the genetic diversity and prevalence of tick-associated Spiroplasma.
The aim of this study was to identify and genetically characterize Spiroplasma in different tick species in Japan. A linear mixed model (LMM) was developed to resolve the correlation among several extrinsic and intrinsic factors associated with Spiroplasma infection in ticks.
2. Materials and Methods
2.1. Sample Collection
Ticks were collected by flagging the vegetation during the period of tick activity (between April 2013 and August 2018) at 112 different sampling sites in 19 different prefectures in Japan. The sampling sites were classified into nine geographical blocks: Hokkaido (Hokkaido prefecture), Tohoku (Yamagata and Fukushima prefectures), Kanto (Chiba prefecture), Chubu (Nagano and Shizuoka prefectures), Kinki (Mie, Nara, and Wakayama prefectures), Chugoku (Hiroshima and Shimane prefectures), Shikoku (Kagawa, Ehime, and Kochi prefectures), Kyushu (Nagasaki, Kumamoto, Miyazaki, and Kagoshima prefectures), and Okinawa (Okinawa prefecture). All collected ticks were transferred to Petri dishes and preserved in an incubator at 16 °C until use.
2.2. Identification of Tick Species
Tick species were identified morphologically under a stereomicroscope according to standard morphological keys [33,34]. A total of 712 adult ticks from four genera were examined in this study. These included two species in the genus Amblyomma (A. geoemydae, n = 3; A. testudinarium, n = 26), one species in the genus Dermacentor (D. taiwanensis, n = 9), 10 species in the genus Haemaphysalis (H. concinna, n = 2; H. cornigera, n = 1; H. flava, n = 65; H. formosensis, n = 83; H. hystricis, n = 60; H. japonica, n = 20; H. kitaokai, n = 78; H. longicornis, n = 106; H. megaspinosa, n = 66; H. yeni, n = 1), and seven species in the genus Ixodes (I. monospinosus, n = 21; I. nipponensis, n = 3; I. ovatus, n = 80; I. pavlovsky, n = 26; I. persulcatus, n = 55; I. tanuki, n = 1; I. turdus, n = 6).
2.3. DNA Extraction
The procedures for DNA extraction from individual ticks have been reported previously [35]. In brief, the surface of tick bodies was individually washed with 70% ethanol and sterilized phosphate-buffered solution (PBS). The whole tick bodies were homogenized in 100 μL of high-glucose Dulbecco’s modified Eagle’s medium (Gibco, Life Technologies, Grand Island, NY, USA) using Micro Smash MS100R (TOMY, Tokyo, Japan) for 30 s at 3000 rpm. DNA was extracted from 50 μL of the tick homogenate using the blackPREP Tick DNA/RNA Kit (Analytik Jena, Jena, Germany) according to the manufacturer’s protocol.
2.4. Detection of Spiroplasma in Ticks
To detect Spiroplasma DNA, PCR amplification of a sequence of approximately 1028 bp in the 16S rDNA was performed. The PCR was carried out in a 20 μL reaction mixture containing 10 μL of 2× Gflex PCR Buffer (Mg2+, dNTP plus), 400 nM of Tks Gflex™ DNA Polymerase (Takara Bio, Shiga, Japan), 400 nM of each primer, 1 μL of DNA template, and sterilized water. The reaction was performed at 94 °C for 1 min, followed by 45 cycles at 98 °C for 10 s, 60 °C for 30 s, and 68 °C for 45 s and a final step at 68 °C for 5 min. PCR products were electrophoresed on a 1.0% agarose gel. The DNA of a Spiroplasma species isolated from I. persulcatus in our previous study [23] and sterilized water were included in each PCR run as positive and negative controls, respectively. Primer sets used for each assay are shown in Table 1 [13,36]. The amplified PCR products were purified using ExoSAP-IT Express PCR Cleanup Reagent (Thermo Fisher Scientific, Tokyo, Japan). Sanger sequencing was performed using the BigDye Terminator version 3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA) and the ABI Prism 3130xl Genetic Analyzer according to the manufacturer’ s instructions. Sequence data were assembled using ATGC software version 6.0.4 (GENETYX, Tokyo, Japan).

Table 1.
Primers used in the present study.
2.5. Molecular Characterization of Spiroplasma
To further characterize Spiroplasma in ticks, additional PCRs based on the 16S–23S rRNA intergenic transcribed spacer (ITS) region (301 bp), chromosomal replication initiator protein dnaA (dnaA) (515 bp), and RNA polymerase B (rpoB) genes (1703 bp) were performed with primers widely used for the characterization of Spiroplasma in arthropods [2,36]. These PCRs were performed for selected samples using the following criteria: (1) more than three samples (when available) were selected for each 16S rDNA allele; (2) the samples were selected from each tick species when the 16S rDNA allele was obtained from multiple tick species. The PCRs were carried out as described above, except that 56 and 52 °C were used as the annealing temperatures for ITS and dnaA PCRs, respectively. The primer sets used for each assay are shown in Table 1. All PCR amplicons were subjected to Sanger sequencing analysis. The sequences obtained were submitted to the DNA Data Bank of Japan (DDBJ) (http://www.ddbj.nig.ac.jp) under specific accession numbers (16S rDNA: LC592079–LC592113; ITS: LC592139–C592161; dnaA: LC592127–LC592138; rpoB: LC592114–LC592126).
2.6. Phylogenetic Analysis
Phylogenetic trees were constructed based on the partial sequences of 16S rDNA, dnaA, rpoB genes, and ITS region. The nucleotide sequences obtained were aligned with representative sequences of known Spiroplasma species available in GenBank as implemented in MEGA7 [30,37]. The reference sequences of ITS region of S. ixodetis were obtained by de novo assembly of Illumina raw reads of Spiroplasma-infected African monarch butterfly Danaus chrysippus deposited in the sequence read archives (SRA) of the NCBI with accession numbers of SRX3872086 and SRX3872088-SRX3872090 [38] using CLC Genomics Workbench v 20.0.4 (Qiagen, Hilden, Germany). Phylogenetic trees were constructed using maximum likelihood (ML) method with bootstrap tests of 1000 replicates. The sequence data of the evolutionary models were determined using the Akaike information criterion with MEGA7 [37].
2.7. Phylogenetic Analysis
Spiroplasma infection in ticks can be affected by various extrinsic and intrinsic factors. Here, the extrinsic factors included sampling district, city/town, season, month, and year variations, and the intrinsic factors were tick species and sex. First, multicollinearity among the explanatory variables was examined using pairwise correlations and the “VIF” function in R package [39] to determine whether multicollinearity was likely to influence LMM results. A correlation between several variables affecting Spiroplasma infection in tsetse flies was reported in a previous study [40]. To identify this possible correlation in ticks, we performed multiple correspondence analysis (MCA) using the “MCA” and “fviz_mca_var” functions in the R packages FactoMineR and Factoextra, respectively [41]. We used an LMM to resolve the correlation among the predictor variables associated with Spiroplasma infection in ticks. We fit the LMM with the predictor variables (sampling season, year, tick sex, and species) as the fixed effects with and without geographic location (district) as the random effect. This was followed by testing in additional LMMs using combinations of the predictor variables with district as the random effect variable and Spiroplasma infection as the response variable. We compared the effectiveness of the tested models with the Chi-square test using the “ANOVA” function in R software. Finally, the “lmer” function in the R package lme4 [42] was used for the selected LMM, with each detected Spiroplasma allele as the response variable.
3. Results
3.1. Infection Rate of Spiroplasma in Different Tick Species
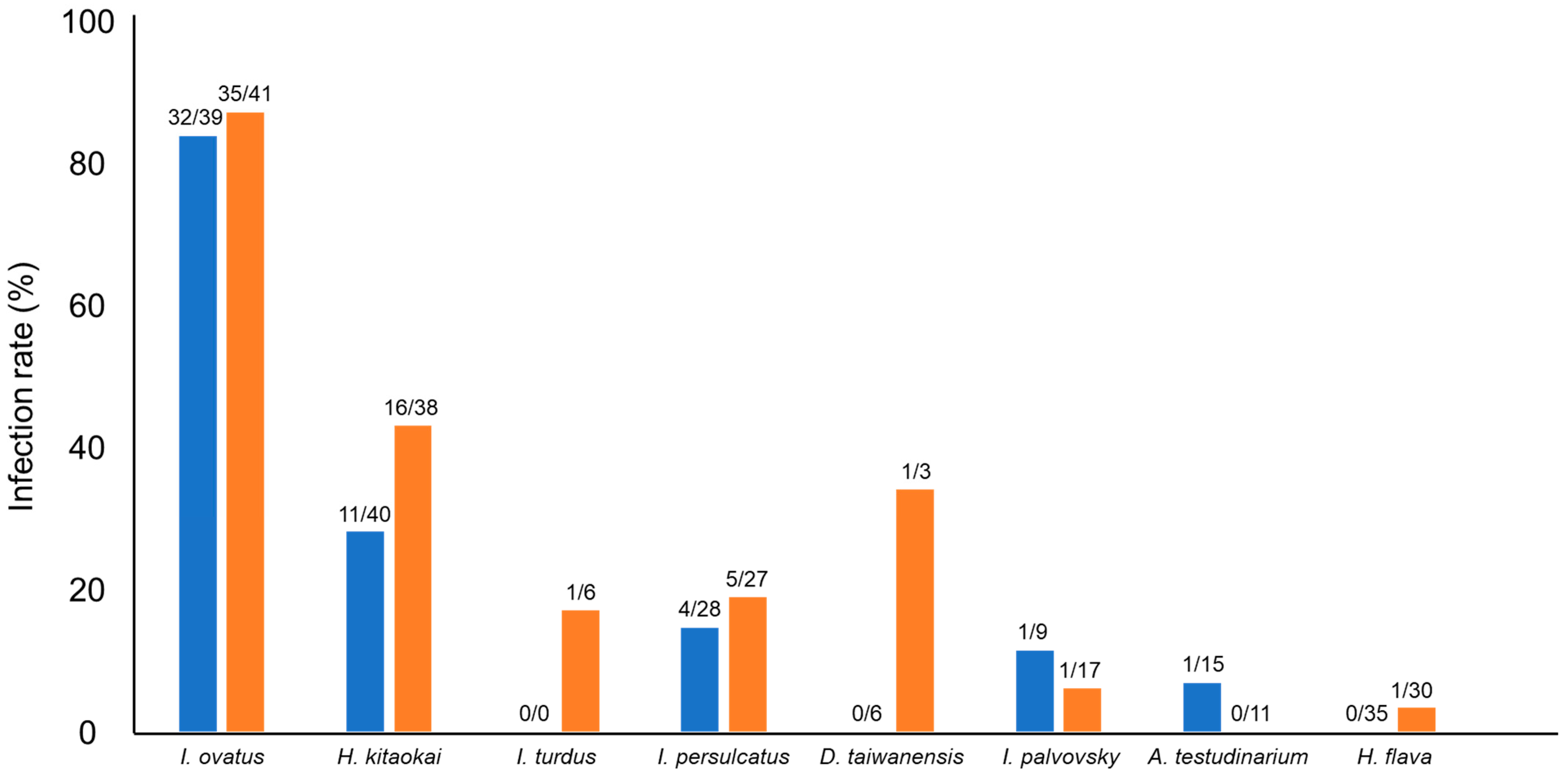
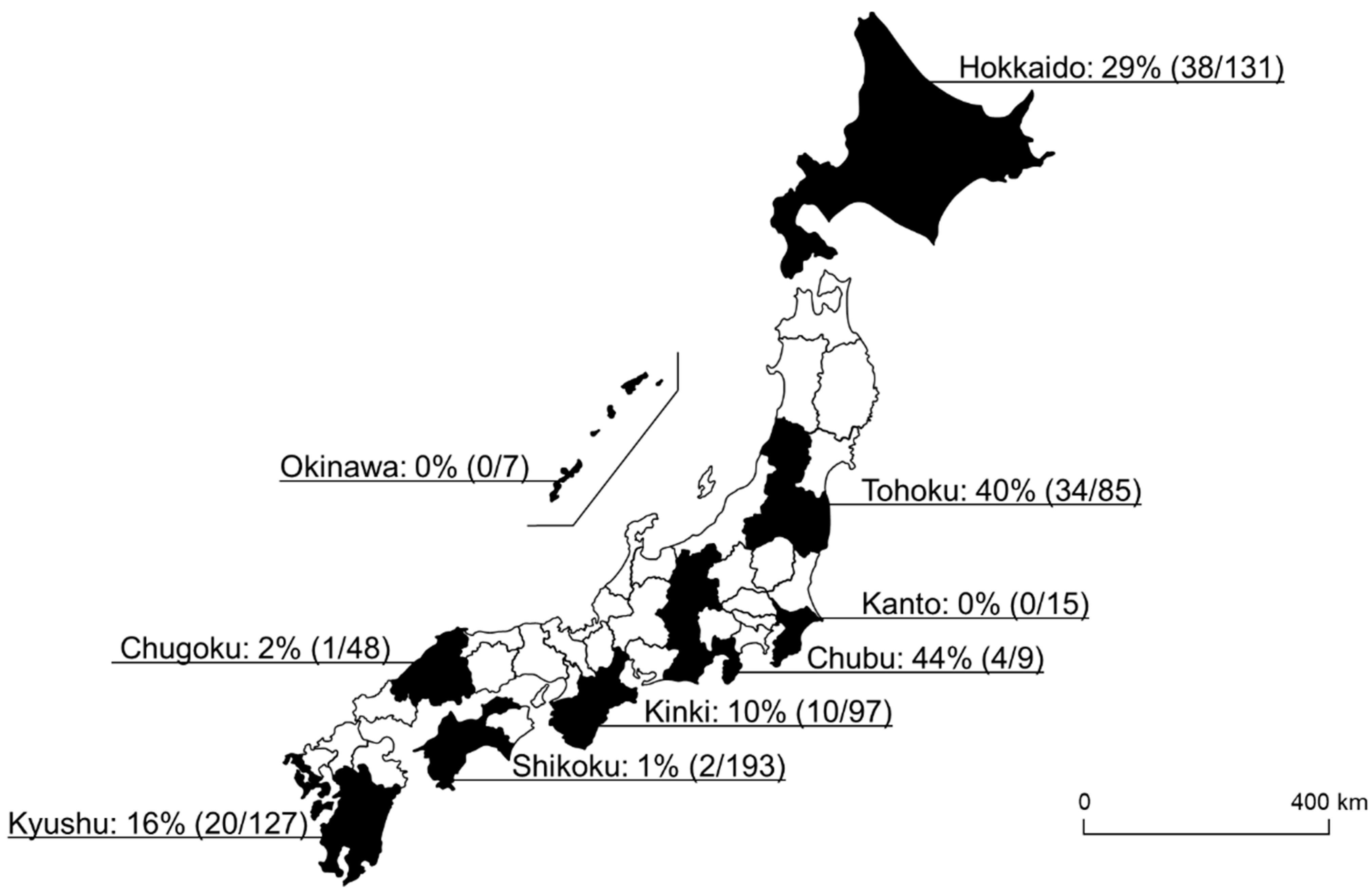
In this study, 109 of 712 samples (15%) were positive for Spiroplasma infection. Among the 20 different tick species, eight tick species were positive for Spiroplasma infection, and the highest infection rate was observed in I. ovatus (84%; 67/80), followed by H. kitaokai (35%; 27/78), I. turdus (17%; 1/6), I. persulcatus (16%; 9/55), D. taiwanensis (11%; 1/9), I. pavlovsky (8%; 2/26), A. testudinarium (4%; 1/26), and H. flava (2%; 1/65) (Figure 1). Only female ticks were positive for the infection in I. turdus, D. taiwanensis, and H. flava, while only one male was positive in A. testudinarium. The difference in Spiroplasma infection rates between male and female ticks was not statistically significant (Fisher’s exact test). Spiroplasma-positive ticks were detected from most of the geographic blocks except for Kanto and Okinawa (Figure 2).
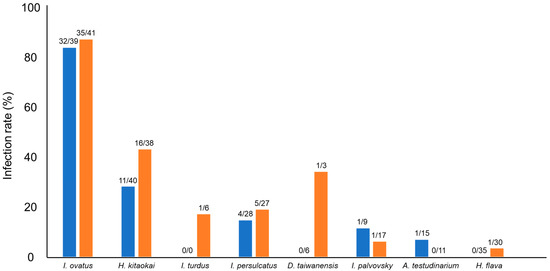
Figure 1.
Spiroplasma-positive rates of different tick species. Blue and orange bars represent male and female ticks, respectively. The numbers at the top of the bars indicate the number of Spiroplasma-positive ticks/number of tested ticks.
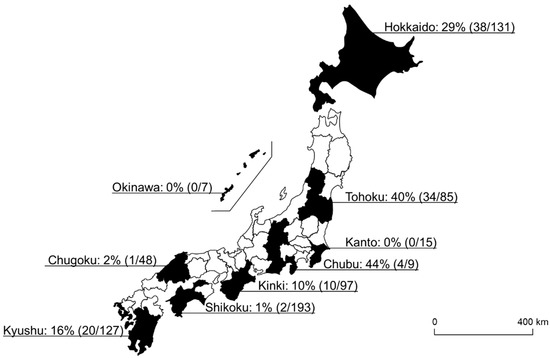
Figure 2.
A map of Japan showing the Spiroplasma-positive rate of each geographical block. The numbers in the parentheses refer to the number of Spiroplasma-positive ticks/number of tested ticks.
3.2. 16S rDNA Genotyping of Spiroplasma in Ticks
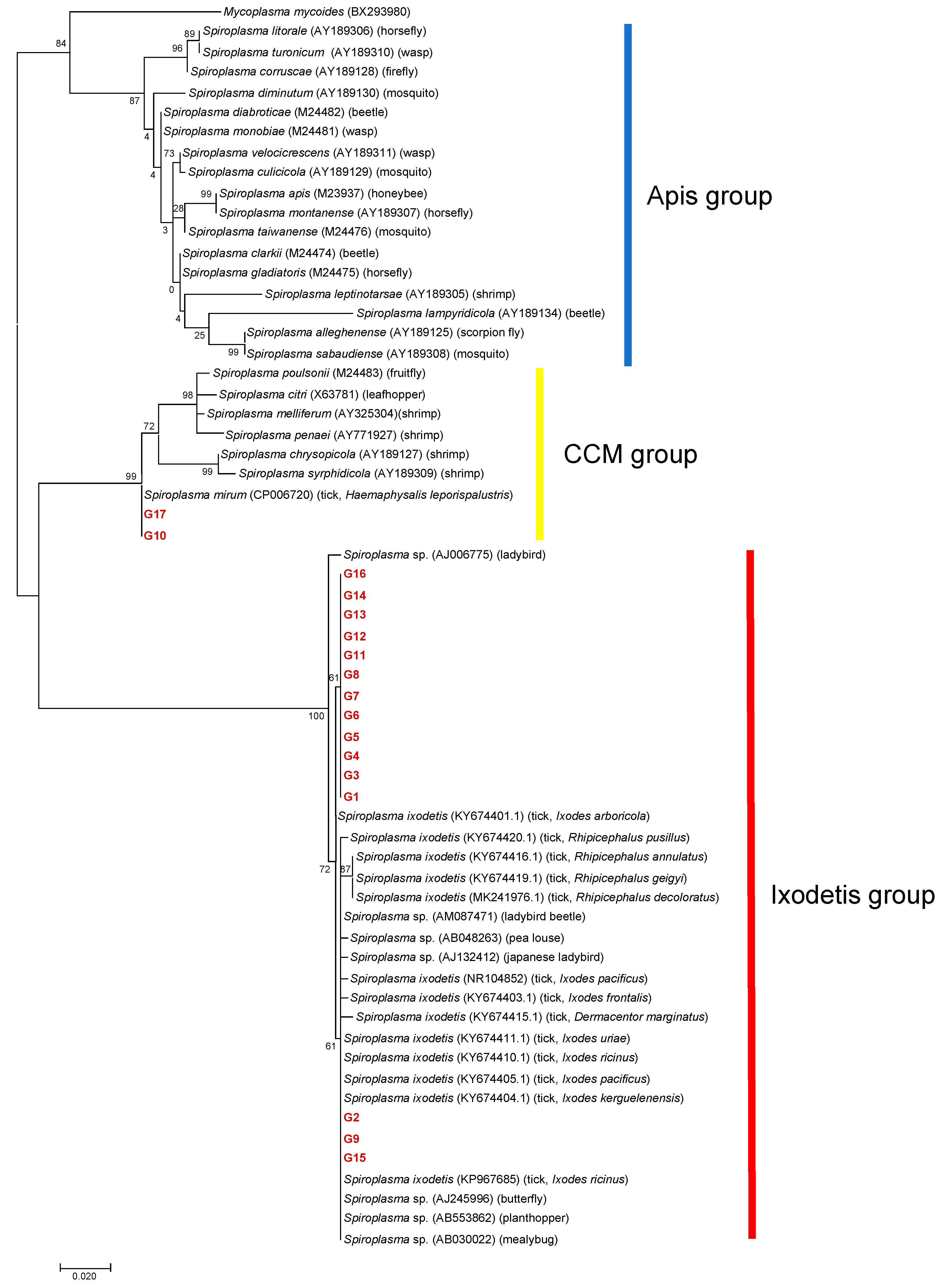
A total of 101 amplicons of 16S rDNA were successfully sequenced, resulting in 17 different 16S rDNA alleles (G1–G17) (Table 2). Eight samples failed in sequencing due to mixed signals. Of the 17 alleles, 13 alleles (G3–G8, and G11–G17) were detected in a single tick species. Two alleles (G1 and G10) were detected in two different tick species: G1 from I. ovatus and I. persulcatus and G10 from A. testudinarium and I. persulcatus. One allele (G2) was detected in three different tick species: I. ovatus, I. persulcatus, and H. kitaokai. Another allele (G9) was observed in four different tick species: I. turdus, I. persulcatus, D. taiwanensis, and H. kitaokai. The detected alleles were classified into the Ixodetis or CCM group in a phylogenetic tree based on the sequences of 16S rDNA (Figure 3). G10 and G17 were clustered with Spiroplasma spp. in the CCM group, whereas other alleles were grouped with members in the Ixodetis group. G10 and G17 showed 99.7% and 99.4% sequence identity, respectively, to S. mirum (CP006720). Alleles in the Ixodetis group formed a cluster with S. ixodetis found in Ixodes, Rhipicephalus, and Dermacentor ticks in other countries and a variety of arthropods such as ladybird, beetle, louse, butterfly, planthopper, and mealybug (Figure 3).

Table 2.
Spiroplasma 16S rDNA alleles and their geographic origins and host tick species.
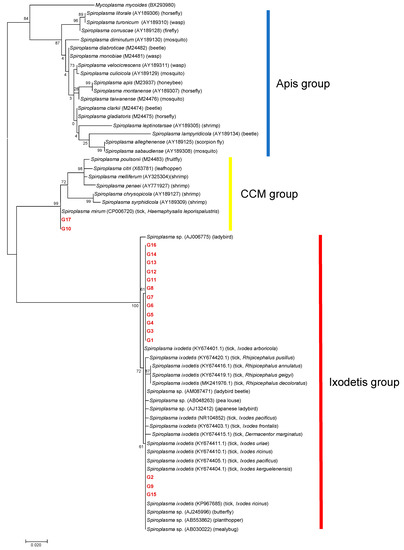
Figure 3.
A phylogenetic tree based on the sequences of 16S rDNA. The analysis was performed using a maximum-likelihood method based on the Hasegawa–Kishino–Yano model with bootstrap tests of 1000 replicates in MEGA7. A discrete Gamma distribution was used to model evolutionary rate differences among sites (five categories (+G, parameter = 0.2496)). The sequences obtained in this study are included with allele names provided in Table 2 and are shown in red. The sequences of other Spiroplasma species were retrieved from GenBank. The host is indicated in the parenthesis for each Spiroplasma sequence.
3.3. Characterization of Spiroplasma Based on the Sequences of ITS Region, dnaA, and rpoB Genes
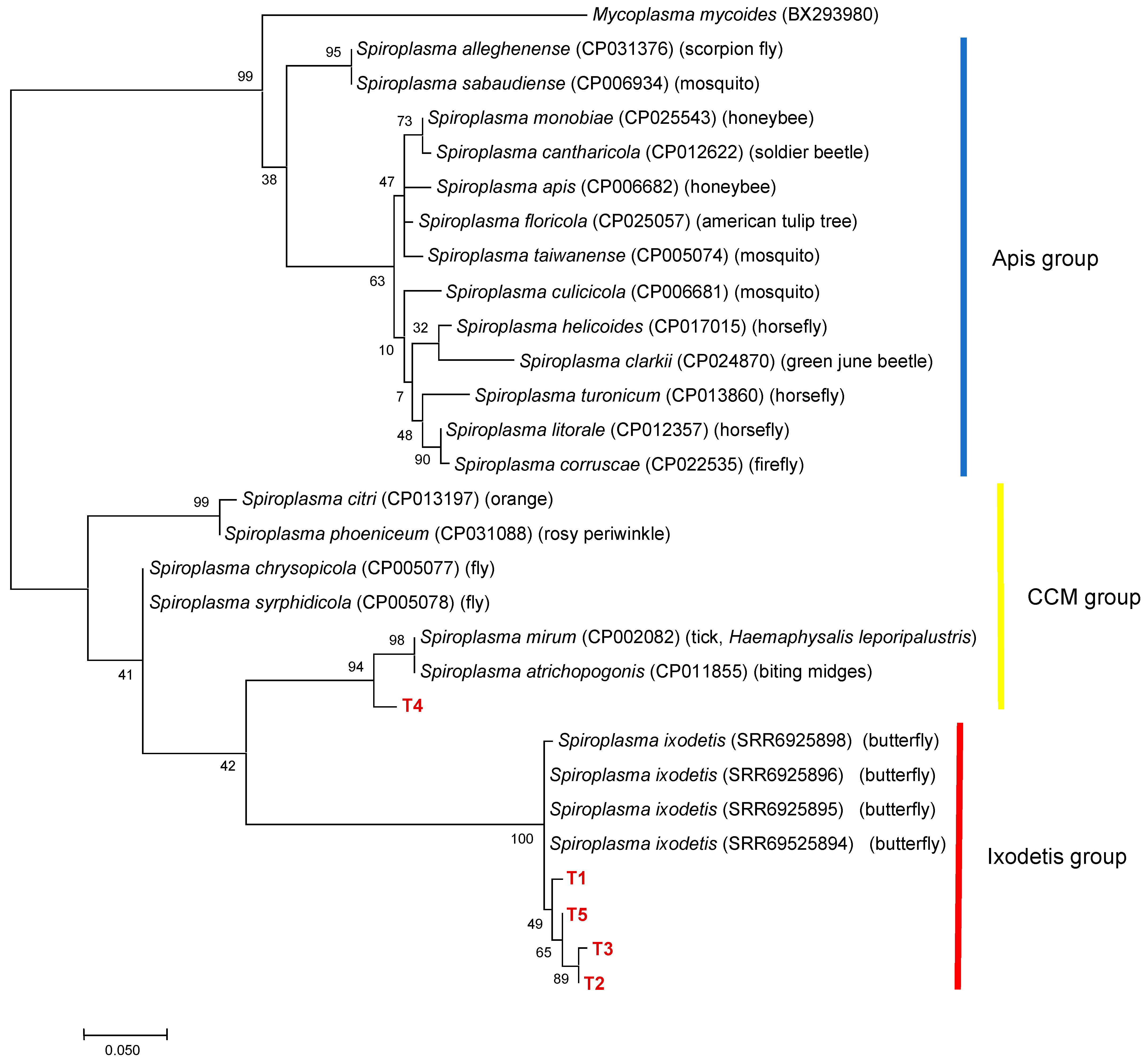
To further characterize Spiroplasma in ticks, 50 Spiroplasma-positive samples were selected based on 16S rDNA genotyping results. The ITS region was amplified in all 16S rDNA alleles, resulting in five different alleles (T1–T5) (Table 3). T1 was the most abundant allele detected in the samples of 10 different 16S rDNA alleles (G1, G2, G4, G8–G10, and G12–G15). Phylogenetic analysis revealed that T4 was clustered with Spiroplasma spp. including S. mirum in the CCM group, whereas T1-T3 and T5 formed a cluster with S. ixodetis reported from butterflies (Figure 4). There was a discrepancy between the 16S rDNA and ITS genotyping results; haplotype SP22 had a 16S rDNA allele (G10) belonging to the CCM group and an ITS allele (T1) belonging to the Ixodetis group. PCR amplification of the dnaA and rpoB genes were only successful for six and seven 16S rRNA alleles, respectively. ML trees based on dnaA and rpoB are shown in Supplementary Figures S3 and S4, respectively.

Table 3.
Multi-locus sequence typing of Spiroplasma in ticks.
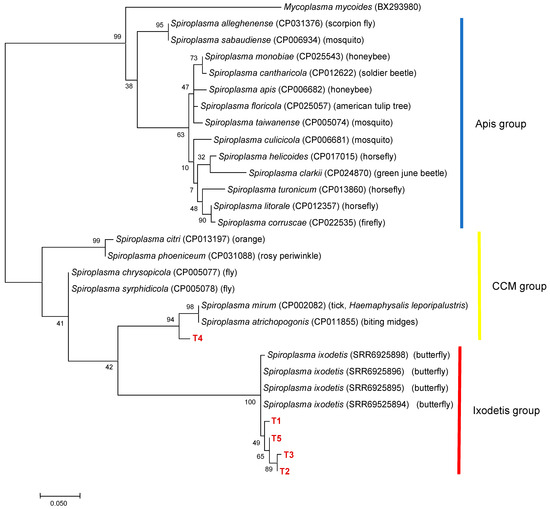
Figure 4.
A phylogenetic tree based on the sequences of ITS region. The analysis was performed using a maximum-likelihood method based on the Tamura 3-parameter model. A discrete Gamma distribution was used to model evolutionary rate differences among sites (five categories (+G, parameter = 0.2599)) with bootstrap tests of 1000 replicates in MEGA7. The sequences obtained in this study are included with allele names provided in Table 3 and are shown in red. The sequences of other Spiroplasma species were retrieved from GenBank.
3.4. Effect of the Genetic Background on Spiroplasma Infection
Based on the estimation of multicollinearity using VIF, the number of degrees of freedom (Df) was more than 1 for all variables except the year; thus, we calculated the generalized variance inflation factors (GVIFs). The Df is equal to the number of associated coefficients for a GVIF. Therefore, we used GVIF1∕2Df to make GVIF values comparable among those with different numbers of Df. High collinearity is usually indicated by VIF > 20. However, multicollinearity analysis using VIF indicated low multicollinearity with all variables (VIF < 5), suggesting that linear regression models would not be influenced by a combination of these variables. Multicollinearity analysis showed that there was a moderate correlation between the predictor variables (season and month; district and city/town) (Table S1). Both month and city/town variables were excluded from further analysis. Then, MCA was performed to identify associations between the predictor variables. The strongest association was detected between district, species, and season (Figure S1). LMM analysis using the predictor variables (season, year, sex, and species) revealed that the introduction of district as the random effect variable improved the models significantly (p ≦ 0.001) (Table 4). Moreover, when tick species was used as the principal predictor, the model for testing Spiroplasma infection in ticks was improved (p ≦ 1.73 × 10−75; Table 5).

Table 4.
LMM to test the correlation between each predictor with Spiroplasma infection using district as the random effect variable.

Table 5.
Effect of several variables on the probability of Spiroplasma infection in the LMM.
The association between Spiroplasma 16S rDNA alleles and host tick species was estimated separately using the best-fit LMM. This analysis was applicable to six alleles (G1–G3, G6, G9, and G11). However, the analysis was not appropriate for the other 11 alleles due to the small sample size (less than five). The analysis revealed that the probability of infection with G1 and G11 was significantly associated with I. ovatus; however, compared with other tick species, H. kitaokai had a significantly higher probability of infection with G9 (Table 6 and Table S2–S4).

Table 6.
Association between Spiroplasma 16S rDNA alleles and tick species.
4. Discussion
Prior to this study, there was only limited information available on the prevalence and genetic diversity of tick-associated Spiroplasma in Japan. In addition to three tick species (H. kitaokai, I. ovatus, and I. persulcatus) that were previously revealed to harbour Spiroplasma [24,32], five additional species, i.e., A. testudinarium, D. taiwanensis, H. flava, I. pavlovsky, and I. turdus, were found to be infected with Spiroplasma, thus expanding our knowledge of the host range of tick-associated Spiroplasma in Japan.
The infection rate of Spiroplasma ranged from 0% to 84% depending on the tick species. To investigate whether this difference in infection rate is determined by the tick species or other factors, LMM analysis was performed. The results indicated that Spiroplasma infection was mainly influenced by the species of ticks but less likely to be influenced by temporal and seasonal factors (Table 5). Although the prevalence of Spiroplasma in tick populations has not been well understood, several previous studies reported that the Spiroplasma infection rates are variable between populations such as in I. arboricola, I. ricinus, and R. decoloratus [28,43]. A study investigating Spiroplasma infection rates in natural Drosophila populations in the southwestern United States and northwestern Mexico observed varying infection rates depending on the fly species [44]. In the same study, there was a difference in Spiroplasma infection rates in two fly species between the two collection sites. Similarly, in our LMM analysis, the introduction of district as the random effect variable improved the models significantly (Table 4), indicating that the Spiroplasma infection status in ticks may be partially influenced by the sampling location.
The highest infection rate was observed in I. ovatus; 82% (32/39) of males and 85% (35/41) of females were positive based on PCR amplification of Spiroplasma 16S rDNA (Figure 1). Sequencing analysis of PCR amplicons identified 11 Spiroplasma alleles in this tick species (Table 3). Furthermore, H. kitaokai, the second most infected species (28% (11/40) of males and 42% (16/38) of females), had four different Spiroplasma alleles. The association between specific 16S rDNA alleles (G1, G9, and G11) and their host tick species was statistically confirmed (Table 6). The presence of these alleles resulted in the high overall infection rates in I. ovatus and H. kitaokai. These Spiroplasma alleles may have adapted to the tick environment, which is important for symbionts [45]. The transmission of symbionts occurs mainly through the vertical or horizontal route. Vertical transmission involves the dispersal of symbionts and occurs primarily from the mother to offspring. Horizontal transmission occurs via host-to-host contact and acquisition from the environment [45]. The high infection rates observed in I. ovatus and H. kitaokai suggest the vertical transmission of Spiroplasma in these tick species. Symbionts can positively affect the nutrition, reproduction, and defence of their hosts. These positive effects may promote the coexistence or coevolution of symbionts and their hosts [45]. Therefore, it is of particular interest to investigate whether Spiroplasma affects tick fitness, as it may help understand the close association between Spiroplasma and ticks.
Among the three Spiroplasma clades, tick-associated Spiroplasma has only been identified in the Ixodetis and CCM groups. In the present study, most of the samples were classified as belonging to the Ixodetis group (n = 98), and only three samples were classified as belonging to the CCM group (Figure 3). Considering that most of the Spiroplasma species from ticks identified in previous studies belong to the Ixodetis group [21,22,24,25,29,30,43,46], this group of Spiroplasma may be widely distributed in the world. On the other hand, there is a lack of information on the geographic distribution and host range of tick-associated Spiroplasma in the CCM group. The alleles G10 and G17 obtained in the present study showed high sequence identities (99.7% and 99.4%, respectively) to S. mirum, which has been found to cause persistent infection in the mouse brain [47] and neurological deterioration and spongiform encephalopathy in suckling rats [48,49]. Furthermore, several ruminants such as deer, sheep, and goats developed spongiform encephalopathy in a dose-dependent manner when experimentally inoculated with S. mirum in their brains [50]. The alleles G10 and G17 were obtained from A. testudinarium, I. pavlovsky, and I. persulcatus, whose primary hosts include domestic and wild ruminants such as cattle and sika deer in Japan [51,52]. Furthermore, A. testudinarium and I. persulcatus are human-biting species that serve as main vectors for human tick-borne diseases [53,54]. Hence, it is important to investigate the potential of these Spiroplasma alleles as agents of human and animal diseases.
The 16S rDNA-based genotyping of 101 Spiroplasma-positive samples identified 17 alleles, some of which were observed in more than two different tick species (Table 2). However, further characterization by sequencing additional genes (ITS, dnaA, and rpoB) divided them into 31 haplotypes, and only one of them (SP24) was observed in two tick species (A. testudinarium and I. persulcatus) (Table 3). A previous study suggested the possible horizontal transmission of Spiroplasma between different ticks and other arthropods, considering that tick-derived S. ixodetis did not form a tick species-specific clade [30]. Our results indicated that horizontal transmission among tick species is not common, at least among the tested tick species. However, the fact that certain alleles (G2, G9, and G15) in the Ixodetis group were more related to Spiroplasma found in other arthropods than other alleles found in ticks highlights the important role of horizontal transmission between arthropods in the spread of Spiroplasma in ticks, as suggested previously [30].
The genes dnaA and rpoB are frequently used in the detection and characterization of Spiroplasma alleles in various arthropods [1,29,36,40,46,55]. In this study, dnaA and rpoB were not amplified in nearly half of the haplotypes tested (Table 3). This may be attributed to nucleotide mismatches in the primer annealing sites. To understand the genetic diversity of Spiroplasma and clarify the mode of horizontal transmission in ticks, further assays using different gene targets and primer sets are necessary. A previous study developed a multi-locus sequence typing method based on five genes (16S rDNA, rpoB, dnaK, gyrA, and EpsG) by referring the daft genome of S. ixodetis Y32 type [30]. Considering high PCR success rates reported for ticks and other arthropods, the method might be useful to genotype Spiroplasma in ticks.
Some species of Spiroplasma are known to affect host reproductive systems through mechanisms such as male killing [7,8,9,10]. For instance, Spiroplasma kills Drosophila males by inducing male X chromosome-specific DNA damage and activating p53-dependent abnormal apoptosis in male embryos [56]. In this study, 49 male ticks and 60 female ticks were infected with Spiroplasma, and there was no statistically significant difference for any of the tested tick species (Figure 1). This result is consistent with that of LLM analysis, where sex was not selected as a variable to improve the model of Spiroplasma infection in ticks (Table 4). Similarly, two previous studies targeting wild populations of R. decoloratus and wild and laboratory populations of I. arboricola did not find any association between sex and Spiroplasma infection [27,30].
In a previous study, Spiroplasma was highly abundant in the salivary glands of I. ovatus [23]. It is known that S. citri, a plant pathogenic Spiroplasma, propagates in the salivary glands of arthropod hosts such as leafhoppers and is released along with the saliva into a new plant during feeding, which leads to transmission from an infected plant to new arthropod hosts [57,58]. Similarly, the presence of Spiroplasma in the tick salivary glands may cause horizontal transmission via feeding to unidentified hosts. One recent study reported that the salivary protein components of Wolbachia/Spiroplasma-infected spider mites differed from those of uninfected mites [59]. Tick saliva is an important biological material for various processes such as combating host defences, accelerating blood-feeding processes, and facilitating the transmission of pathogens to hosts [60]. Therefore, the effects of Spiroplasma on tick physiology and pathogen transmission involving the tick salivary glands should be clarified in future experimental studies.
5. Conclusions
Spiroplasma is one of the most common symbionts in arthropods; however, only limited data are available on species that infect ticks. This study expanded our knowledge of the host range of tick-associated Spiroplasma in Japan. Modelling analysis using tick samples with different infection rates indicated that the host tick species was the primary factor associated with Spiroplasma infection. Moreover, the presence of certain alleles that are highly adapted to specific tick species may explain the high infection rates in I. ovatus and H. kitaokai. A comparison of the alleles suggests that the horizontal transmission of Spiroplasma between tick species may not be a frequent event. Further studies are required to understand the transmission cycle of Spiroplasma species in wild tick populations and their roles in ticks.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-2607/9/2/333/s1: Table S1: Multicollinearity analysis of predictive variables, Table S2: A summary for the Linear mixed models (LMM) testing the probability of infection of Spiroplasma allele G1, Table S3: A summary for the Linear mixed models (LMM) testing the probability of infection of Spiroplasma allele G9, Table S4: A summary for the Linear mixed models (LMM) testing the probability of infection of Spiroplasma allele G11, Figure S1: The results of multiple correspondence analysis, Figure S2: Multicollinearity analysis of predictive variables, Figure S3: A phylogenetic tree based on the sequences of rpoB gene, Figure S4: A phylogenetic tree based on the sequences of dnaA gene.
Author Contributions
Conceptualization: S.O. and R.N.; methodology: S.O.; formal analysis: S.O., W.M.A.M., and M.A.M.M.; investigation: S.O., K.K. (Kodai Kusakisako), M.J.T., K.M., K.K. (Ken Katakura), N.N., and Y.Q.; writing—original draft preparation: S.O.; writing—review and editing: S.O. and R.N.; supervision: R.N.; funding acquisition: R.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by KAKENHI, grant numbers 16H06429, 16K21723, 16H06431, 19H03118, 19F19097, 20K21358, and 20KK0151.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in the DNA Data Bank of Japan (DDBJ) (http://www.ddbj.nig.ac.jp) and the accession numbers are available in the text.
Acknowledgments
We would like to thank to all collaborators who supported in collection of ticks in each prefecture.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Haselkorn, T.S.; Markow, T.A.; Moran, N.A. Multiple introductions of the Spiroplasma bacterial endosymbiont into Drosophila. Mol. Ecol. 2009, 18, 1294–1305. [Google Scholar] [CrossRef]
- Goryacheva, I.; Blekhman, A.; Andrianov, B.; Romanov, D.; Zakharov, I. Spiroplasma infection in Harmonia axyridis—Diversity and multiple infection. PLoS ONE 2018, 13, e0198190. [Google Scholar] [CrossRef] [PubMed]
- Gasparich, G.E.; Whitcomb, R.F.; Dodge, D.; French, F.E.; Glass, J.; Williamson, D.L. The genus Spiroplasma and its non-helical descendants: Phylogenetic classification, correlation with phenotype and roots of the Mycoplasma mycoides clade. Int. J. Syst. Evol. Microbiol. 2004, 54, 893–918. [Google Scholar] [CrossRef]
- Duron, O.; Bouchon, D.; Boutin, S.; Bellamy, L.; Zhou, L.; Engelstädter, J.; Hurst, G.D. The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol. 2008, 12, 1–12. [Google Scholar] [CrossRef]
- Hamilton, P.T.; Leong, J.S.; Koop, B.F.; Perlman, S.J. Transcriptional responses in a Drosophila defensive symbiosis. Mol. Ecol. 2014, 23, 1558–1570. [Google Scholar] [CrossRef]
- Whitcomb, R.F.; Chen, T.A.; Williamson, D.L.; Liao, C.; Tully, J.G.; Bové, J.M.; Mouches, C.; Rose, D.L.; Coan, M.E.; Clark, T.B. Spiroplasma kunkelii sp. nov.: Characterization of the Etiological Agent of Corn Stunt Disease. Int. J. Syst. Bacteriol. 1986, 36, 170–178. [Google Scholar] [CrossRef]
- Anbutsu, H.; Fukatsu, T. Tissue-specific infection dynamics of male-killing and nonmale-killing spiroplasmas in Drosophila melanogaster. FEMS Microbiol. Ecol. 2006, 57, 40–46. [Google Scholar] [CrossRef][Green Version]
- Montenegro, H.; Solferini, V.N.; Klaczko, L.B.; Hurst, G.D.D. Male-killing Spiroplasma naturally infecting Drosophila melanogaster. Insect Mol. Biol. 2005, 14, 281–287. [Google Scholar] [CrossRef]
- Jiggins, F.M.; Hurst, G.D.D.; Jiggins, C.D.; Schulenburg, J.H.G.v.d.; Majerus, M.E.N. The butterfly Danaus chrysippus is infected by a male- killing Spiroplasma bacterium. Parasitology 2000, 120, 439–446. [Google Scholar] [CrossRef]
- Majerus, T.M.O.; Graf Von Der Schulenburg, J.H.; Majerus, M.E.N.; Hurst, G.D.D. Molecular identification of a male-killing agent in the ladybird Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). Insect Mol. Biol. 1999, 8, 551–555. [Google Scholar] [CrossRef]
- Yamauchi, T.; Takada, A. Illustrations of Common Adult Ticks in the Mainland Japan. Bull. Hoshizaki Green Found 2015, 18, 287–305. (In Japanese) [Google Scholar]
- Kwak, M.L. A checklist of the ticks (Acari: Argasidae, Ixodidae) of Japan. Exp. Appl. Acarol. 2018, 75, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Sanada-Morimura, S.; Matsumura, M.; Noda, H. Male killing caused by a Spiroplasma symbiont in the small brown planthopper, Laodelphax striatellus. J. Hered. 2013, 104, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, M.J.; Perlman, S.J. Generality of toxins in defensive symbiosis: Ribosome-inactivating proteins and defense against parasitic wasps in Drosophila. PLoS Pathog. 2017, 13, e1006431. [Google Scholar] [CrossRef]
- Lukasik, P.; Guo, H.; van Asch, M.; Ferrari, J.; Godfray, H.C.J. Protection against a fungal pathogen conferred by the aphid facultative endosymbionts Rickettsia and Spiroplasma is expressed in multiple host genotypes and species and is not influenced by co-infection with another symbiont. J. Evol. Biol. 2013, 26, 2654–2661. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Vilchez, I.; Mateos, M. Spiroplasma bacteria enhance survival of Drosophila hydei attacked by the parasitic wasp Leptopilina heterotoma. PLoS ONE 2010, 5, e12149. [Google Scholar] [CrossRef]
- Bastian, F.O.; Elzer, P.H.; Wu, X. Spiroplasma spp. biofilm formation is instrumental for their role in the pathogenesis of plant, insect and animal diseases. Exp. Mol. Pathol. 2012, 93, 116–128. [Google Scholar] [CrossRef]
- Mouches, C.; Bové, J.M.; Albisetti, J.; Clark, T.B.; Tully, J.G. A Spiroplasma of serogroup IV causes a May-disease-like disorder of honeybees in Southwestern France. Microb. Ecol. 1982, 8, 387–399. [Google Scholar] [CrossRef]
- Cockburn, S.N.; Haselkorn, T.S.; Hamilton, P.T.; Landzberg, E.; Jaenike, J.; Perlman, S.J. Dynamics of the continent-wide spread of a Drosophila defensive symbiont. Ecol. Lett. 2013, 16, 609–616. [Google Scholar] [CrossRef]
- Jaenike, J.; Brekke, T.D. Defensive endosymbionts: A cryptic trophic level in community ecology. Ecol. Lett. 2011, 14, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Tully, J.G.; Whitcomb, R.F.; Rose, D.L.; Bove, J.M. Spiroplasma mirum, a new species from the rabbit tick (Haemaphysalis leporispalustris). Int. J. Syst. Bacteriol. 1982, 32, 92–100. [Google Scholar] [CrossRef]
- Tully, J.G.; Rose, D.L.; Yunker, C.E.; Cory, J.; Whitcomb, R.F.; Williamson, D.L. Helical Mycoplasmas (Spiroplasmas) from Ixodes Ticks. Science 1981, 212, 1043–1045. [Google Scholar] [CrossRef]
- Qiu, Y.; Nakao, R.; Ohnuma, A.; Kawamori, F.; Sugimoto, C. Microbial population analysis of the salivary glands of ticks; a possible strategy for the surveillance of bacterial pathogens. PLoS ONE 2014, 9, e103961. [Google Scholar] [CrossRef] [PubMed]
- Taroura, S.; Shimada, Y.; SAKATA, Y.; Miyama, T.; Hiraoka, H.; Watanabe, M.; Itamoto, K.; Okuda, M.; Inokuma, H. Detection of DNA of ‘Candidatus Mycoplasma haemominutum’ and Spiroplasma sp. in Unfed Ticks Collected from Vegetation in Japan. J. Vet. Med. Sci. 2006, 67, 1277–1279. [Google Scholar] [CrossRef] [PubMed]
- Henning, K.; Greiner-Fischer, S.; Hotzel, H.; Ebsen, M.; Theegarten, D. Isolation of Spiroplasma sp. from an Ixodes tick. Int. J. Med. Microbiol. 2006, 296, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Hornok, S.; Meli, M.L.; Perreten, A.; Farkas, R.; Willi, B.; Beugnet, F.; Lutz, H.; Hofmann-Lehmann, R. Molecular investigation of hard ticks (Acari: Ixodidae) and fleas (Siphonaptera: Pulicidae) as potential vectors of rickettsial and mycoplasmal agents. Vet. Microbiol. 2010, 140, 98–104. [Google Scholar] [CrossRef]
- van Oosten, A.R.; Duron, O.; Heylen, D.J.A. Ticks and Tick-borne Diseases Sex ratios of the tick Ixodes arboricola are strongly female-biased, but there are no indications of sex-distorting bacteria. Ticks Tick Borne Dis. 2018, 9, 307–313. [Google Scholar] [CrossRef]
- Duron, O.; Binetruy, F.; Noël, V.; Cremaschi, J.; McCoy, K.D.; Arnathau, C.; Plantard, O.; Goolsby, J.; Pérez de León, A.A.; Heylen, D.J.A.; et al. Evolutionary changes in symbiont community structure in ticks. Mol. Ecol. 2017, 26, 2905–2921. [Google Scholar] [CrossRef]
- Bell-Sakyi, L.; Palomar, A.M.; Kazimirova, M. Isolation and propagation of a Spiroplasma sp. from Slovakian Ixodes ricinus ticks in Ixodes spp. cell lines. Ticks Tick Borne Dis. 2015, 6, 601–606. [Google Scholar] [CrossRef]
- Binetruy, F.; Bailly, X.; Chevillon, C.; Martin, O.Y.; Bernasconi, M.V.; Duron, O. Phylogenetics of the Spiroplasma ixodetis endosymbiont reveals past transfers between ticks and other arthropods. Ticks Tick Borne Dis. 2019, 10, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, K.; Aonuma, H.; Kanuka, H. Distribution of tick-borne diseases in Japan: Past patterns and implications for the future. J. Infect. Chemother. 2018, 24, 499–504. [Google Scholar] [CrossRef]
- Thu, M.J.; Qiu, Y.; Kataoka-Nakamura, C.; Sugimoto, C.; Katakura, K.; Isoda, N.; Nakao, R. Isolation of Rickettsia, Rickettsiella, and Spiroplasma from Questing Ticks in Japan Using Arthropod Cells. Vector Borne Zoonotic Dis. 2019, 19, 474–485. [Google Scholar] [CrossRef]
- Yamaguti, N.; Tipton, V.J.; Keegan, H.L.; Toshioka, S. Ticks of Japan, Korea, and the Ryukyu Islands. Brigham Young Univ. Sci. Bull. Biol. Ser. 1971, 15, 1–226. [Google Scholar]
- Nakao, M.; Miyamoto, K.; Kitaoka, S. A new record of Ixodes pavlovskyi Pomerantzev from Hokkaido, Japan (Acari: Ixodidae). Med. Entomol. Zool. 1992, 43, 229–234. [Google Scholar] [CrossRef]
- Thu, M.J.; Qiu, Y.; Matsuno, K.; Kajihara, M.; Mori-Kajihara, A.; Omori, R.; Monma, N.; Chiba, K.; Seto, J.; Gokuden, M.; et al. Diversity of spotted fever group rickettsiae and their association with host ticks in Japan. Sci. Rep. 2019, 9, 1500. [Google Scholar] [CrossRef] [PubMed]
- Doudoumis, V.; Blow, F.; Saridaki, A.; Augustinos, A.; Dyer, N.A.; Goodhead, I.; Solano, P.; Rayaisse, J.B.; Takac, P.; Mekonnen, S.; et al. Challenging the Wigglesworthia, Sodalis, Wolbachia symbiosis dogma in tsetse flies: Spiroplasma is present in both laboratory and natural populations. Sci. Rep. 2017, 7, 4699. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.H.; Singh, K.S.; Gordon, I.J.; Omufwoko, K.S.; Collins, S.; Warren, I.A.; Munby, H.; Brattstrom, O.; Traut, W.; Dino, J.; et al. Whole-chromosome hitchhiking driven by a male-killing endosymbiont. PLoS Biol. 2020, 18, e3000610. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 3rd ed.; SAGE: Thousando Oaks, CA, USA, 2019. [Google Scholar]
- Schneider, D.I.; Saarman, N.; Onyango, M.G.; Hyseni, C.; Opiro, R.; Echodu, R.; O’Neill, M.; Bloch, D.; Vigneron, A.; Johnson, T.J.; et al. Spatio-temporal distribution of Spiroplasma infections in the tsetse fly (Glossina fuscipes fuscipes) in northern Uganda. PLoS Negl. Trop. Dis. 2019, 13, e0007340. [Google Scholar] [CrossRef]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 82599. [Google Scholar] [CrossRef]
- Klubal, R.; Kopecky, J.; Nesvorna, M.; Sparagano, O.A.; Thomayerova, J.; Hubert, J. Prevalence of pathogenic bacteria in Ixodes ricinus ticks in Central Bohemia. Exp. Appl. Acarol. 2016, 68, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Watts, T.; Haselkorn, T.S.; Moran, N.A.; Markow, T.A. Variable incidence of Spiroplasma infections in natural populations of Drosophila species. PLoS ONE 2009, 4, e5703. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.J.; Mihaljevic, J.R.; Des Marteaux, L.; Hrček, J. Metacommunity theory for transmission of heritable symbionts within insect communities. Ecol. Evol. 2019, 10, 1703–1721. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, G.; Sekeyova, Z.; Raoult, D.; Mediannikov, O. Multiple tick-associated bacteria in Ixodes ricinus from Slovakia. Ticks Tick Borne Dis. 2012, 3, 406–410. [Google Scholar] [CrossRef]
- Clark, H.F. The suckling mouse cataract agent (SMCA). A slow mycoplasma-like agent. Prog. Med. Virol. 1974, 18, 307–322. [Google Scholar]
- Bastian, F.O.; Purnell, D.M.; Tully, J.G. Neuropathology of Spiroplasma infection in the rat brain. Am. J. Pathol. 1984, 114, 496–514. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tully, J.G.; Bastian, F.O.; Rose, D.L. Localization and persistence of spiroplasmas in an experimental brain infection in suckling rats. Ann. Microbiol. 1984, 135, 111–117. [Google Scholar] [CrossRef]
- Bastian, F.O.; Sanders, D.E.; Forbes, W.A.; Hagius, S.D.; Walker, J.V.; Henk, W.G.; Enright, F.M.; Elzer, P.H. Spiroplasma spp. from transmissible spongiform encephalopathy brains or ticks induce spongiform encephalopathy in ruminants. J. Med. Microbiol. 2007, 56, 1235–1242. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Isogai, E.; Isogai, H.; Masuzawa, T.; Postic, D.; Baranton, G.; Kamewaka, Y.; Kimura, K.; Nishikawa, T.; Fuji, N.; Ishii, N.; et al. Borrelia burgdorferi sensu lato in an endemic environment: Wild sika deer (Cervus nippon yesoensis) with infected ticks and antibodies. Microbiol. Immunol. 1996, 40, 13–19. [Google Scholar] [CrossRef]
- Nakao, R.; Shinjo, K.; Sakiyama, T.; Ogata, S.; Kusakisako, K.; Kinoshita, G.; Naguib, D.; Chatanga, E.; Mohamed, W.M.A.; Moustafa, M.A.M.; et al. Amblyomma testudinarium infestation on a brown bear (Ursus arctos yesoensis) captured in Hokkaido, a northern island of Japan. Parasitol. Int. 2021, 80, 102209. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Yamagishi, T.; Shimada, T.; Matsui, T.; Shimojima, M.; Saijo, M.; Oishi, K. Epidemiological and clinical features of severe fever with thrombocytopenia syndrome in Japan, 2013-2014. PLoS ONE 2016, 11, e0165207. [Google Scholar] [CrossRef] [PubMed]
- Takano, A.; Toyomane, K.; Konnai, S.; Ohashi, K.; Nakao, M.; Ito, T.; Andoh, M.; Maeda, K.; Watarai, M.; Sato, K.; et al. Tick surveillance for relapsing fever spirochete Borrelia miyamotoi in Hokkaido, Japan. PLoS ONE 2014, 9, e104532. [Google Scholar] [CrossRef]
- Mathé-Hubert, H.; Kaech, H.; Ganesanandamoorthy, P.; Vorburger, C. Evolutionary costs and benefits of infection with diverse strains of Spiroplasma in pea aphids. Evolution 2019, 73, 1466–1481. [Google Scholar] [CrossRef] [PubMed]
- Harumoto, T.; Fukatsu, T.; Lemaitre, B. Common and unique strategies of male killing evolved in two distinct Drosophila symbionts. Proc. Biol. Sci. 2018, 285, 20172167. [Google Scholar] [CrossRef] [PubMed]
- Markham, P.G. Spiroplasmas in Leafhoppers: A review. Yale J. Biol. Med. 1983, 56, 745–751. [Google Scholar]
- Killiny, N.; Batailler, B.; Foissac, X.; Saillard, C. Identification of a Spiroplasma citri hydrophilic protein associated with insect transmissibility. Microbiology 2006, 152, 1221–1230. [Google Scholar] [CrossRef][Green Version]
- Zhu, Y.X.; Song, Y.L.; Huang, H.J.; Zhao, D.S.; Xia, X.; Yang, K.; Lu, Y.J.; Hong, X.Y. Comparative analyses of salivary proteins from the facultative symbiont-infected and uninfected Tetranychus truncatus. Syst. Appl. Acarol. 2018, 23, 1027–1042. [Google Scholar] [CrossRef]
- Šimo, L.; Kazimirova, M.; Richardson, J.; Bonnet, S.I. The essential role of tick salivary glands and saliva in tick feeding and pathogen transmission. Front. Cell. Infect. Microbiol. 2017, 7, 281. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).