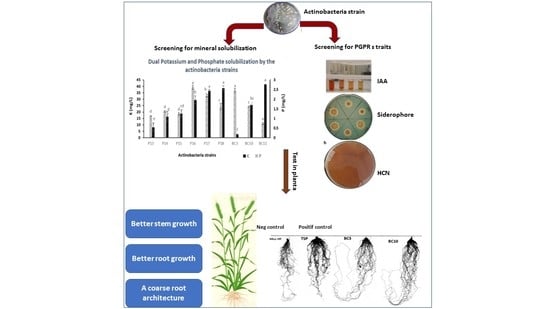
1. Introduction
In many developing countries, agriculture is a key socioeconomic driver and one of the most important sources of employment and income. With roughly 83 million people being added to the world’s population every year, it is estimated that the total demand for food will increase by 40% by 2030 and 70% by 2050 [
1]. One of the most widely grown crops to meet this demand is wheat, which (after rice) is considered in the developing world as the second-most important food source since it provides more calories and proteins (20%) than any other crop [
2]. Wheat demand is expected to increase by 60%. Therefore, achieving such a goal will require the implementation of efficient and sustainable fertilization approaches to improve bioavailability of essential nutrients such as phosphorus (P) and potassium (K). In fact, P and K positively affect plant metabolisms and resistance to biotic and abiotic stresses, while leading to a better soil fertility and crop production [
3,
4,
5]. According to Mitra et al. [
6] P availability also improves other vital plant functions such as cell division, cell enlargement, and transformation of starch and sugars. In this context, the direct application of rock phosphate (RP) and rock potassium (RK) is a promising solution to lower the cost and provide a sustainable complement to conventional fertilization practices, which are not always adapted to the specificities of the African agriculture [
7]. Nonetheless, the low solubility of P and K is a major drawback to their direct application, notably in nonacidic soils [
5,
6]. Currently, converting the insoluble portion of P into soluble fraction is a key objective in sustainable agriculture [
8]. Several techniques and strategies have been recently proposed to increase P solubilization and subsequently increase the P availability. The soil application of phosphate-solubilizing microorganism (PSM)-based inoculum is a promising approach, which takes advantage of the capacity of PSMs to assimilate phosphorus for their own requirement, while making it available for plant uptake [
4,
8,
9,
10]. Actinobacteria or Actinomycetes are considered as plant growth-promoting rhizobacteria (PGPR) and their P solubilization capacity has been well documented [
11,
12,
13,
14]. These filamentous bacteria have the ability to persist in very difficult and competitive environments by producing spores that adhere to soil particles [
15,
16]. Moreover, several interesting properties of these microorganisms have been evidenced such as the production of metabolites that improve plant growth and tolerance to biotic and abiotic stresses [
17,
18,
19], which make them suitable candidates for the production of highly versatile biofertilizers [
14,
20,
21,
22]. Unfortunately, until now Actinobacteria have been scarcely investigated compared to other plant growth-promoting rhizobacteria such as Proteobacteria and Firmicutes [
23,
24,
25]. Consequently, the current study focused on the evaluation of the ability of Actinobacteria strains isolated from contrasting environment to solubilize RK and different RPs, as well as their ability to produce PGP-related compounds such as indole acetic acid (IAA), siderophore, HCN, and ammonia. Overall, the specific objectives of this study are as follows:
- (i)
Assess the impact of the RP type and quality on the ability of Actinobacteria to solubilize P.
- (ii)
Investigate the behavior of RP-solubilizing Actinobacteria under K-bearing minerals such as mica.
- (iii)
Validate the impact of the RP–RK–Actinobacteria combination on plant growth under greenhouse conditions.
- (iv)
Suggest efficient K- and P-solubilizing Actinobacteria strains as biofertilizers for sustainable improvement of plant nutrition and soil fertility.
4. Discussion
Poor soil fertility is one of the most important constraint-limiting crop yields in developing countries [
39]. Fortunately, it can be significantly improved by adopting sustainable approaches such as using beneficial rhizobacteria (e.g., PSMs, nitrogen-fixing bacteria) as biofertilizers. Among PSMs, Actinobacteria are known to be eco-friendly and efficient plant growth promoters [
24]. Therefore, the main objective of this study was to focus on Actinobacteria strains’ abilities to dissolve mica and six different RPs. The phosphate solubilization index (SI) was assessed on NBRIP agar containing RP as the sole source of phosphorus. Most of the strains showed a positive SI, except
Streptomyces alboviridis P18 (
Table 2). The highest SI (3.17) was recorded for
Streptomyces anulatus strain P16, which also showed a high RP solubilization rate in a liquid medium. Interestingly,
Streptomyces alboviridis strain P18 showed a strong solubilization capacity in liquid media (
Figure 1) despite the absence of a solubilization halo in solid media, which indicates the occurrence of solubilization mechanisms other than acidification [
40]. In fact, similar findings were reported by Djebaili et al. [
41] regarding P-solubilizing Actinobacteria, as well as by Nautiyal [
26], who isolated two efficient P-solubilizing
Pseudomonas, which could not form the typical halo zone on agar plates. These results suggest that the SI derived from the solubilization halo should not be the sole criterion to take into consideration for screening efficient PSMs [
38,
39].
In liquid media, the relative efficiency of the nine Actinobacteria strains in dissolving the six RPs was related to the type of RP and the bacterial species (
Figure 1). The solubilization rate of RP was directly correlated to RP P
2O
5 content. The capacities of the Actinobacteria strains to solubilize the RPs ranged from 0.1 to 32 mg/L (
Figure 1). These results are in agreement with the findings of Hamdali et al. [
42], who reported a high amount of phosphate-solubilizing activity by
Streptomyces griseus and
Streptomyces cavourensis with 29.67 and 21.43 mg/L, respectively. Similar results were also obtained by Nafis et al. [
43], who reported that two
Streptomyces isolated from the desert and mountain soils are the most efficient phosphate solubilizers (solubilization rate of 12.39 and 8.56 mg/L). Several mechanisms can be involved in microbial P solubilization, with the most common one being via media acidification, as shown by our experiment (
Figure 2). The acidification is usually attributed to the production of several organic acids from the fermentation of organic compounds such as citric, gluconic, lactic, malic, and oxalic acid [
44,
45,
46,
47]. In fact, the decrease in pH enhances RP dissolution by removing Ca from rock phosphate, thus releasing P into the solution [
48]. Several literature reports suggest that the solubilization of mineral phosphate by microorganisms might also be due to the production of chelating substances (e.g., siderophores) that bind with metal cations (aluminum, iron, and calcium), thus preventing phosphorus complexation [
49,
50]. Interestingly, all tested Actinobacteria strains were able to produce siderophores (
Table 3,
Figure 4), which may suggest that a dual solubilization process may be involved, implying both media acidification and siderophore secretion.
K solubilization assays revealed that among the selected strains, only
Streptomyces alboviridis P18,
Streptomyces griseorubens BC3,
Streptomyces griseorubens BC10, and
Nocardiopsis alba BC11 were able to solubilize mica at 3, 11, 12.75, and 17.8 mg/L, respectively (
Figure 3A). Microbial K solubilization has been scarcely investigated, and in our knowledge, only
Arthrobacter sp. 42,
Arthrobacter sp. 4, and
Microbacterium FS-01 were reported to be K solubilizers [
47]. In general, only 5% of potassium-solubilizing bacteria are Actinobacteria [
51,
52]. Similar to P solubilization mechanisms, K solubilization is also attributed to either media acidification or the chelation of cations that usually bind to K [
51]. However, in this study, K solubilization under phosphorus deficiency showed some relevant results since all the Actinobacteria strains were able to solubilize the potassium source (
Figure 3B). These results showed that the deficiency in P stimulates the solubilization of K. For instance, the K solubilization value of
Nocardiopsis alba BC11 was 41.5 mg/L under P deficiency and 17.8 mg/L under P (KH
2PO
4) sufficiency. Such difference could be explained by the fact that molecules involved in P solubilization may also solubilize the insoluble source of K (mica) and, therefore, trigger more efficient K solubilization. Similar results were found by Abou-el-Seoud and Abdel-Megeed [
53], who reported that co-inoculation of P- and K-dissolving bacteria under RP fertilization increased P and K availability and uptake. Besides their capacity to solubilize RP and K, the Actinobacteria strains were screened for plant growth promoting (PGP) factors, which are considered an effective tool in the investigation of microorganisms that can be used as biofertilizers. Such assays are of high importance as they enable the selection of elite strains that have the best agronomic potential [
54]. All the tested strains showed notable PGP activities, such IAA, HCN, and ammonia production. The production of phytohormones such as auxin (IAA) plays an important role in stimulating the development of the rooting, and they act as signaling molecules involved in the production of secondary metabolites and Actinobacteria sporulation [
55,
56,
57]. In the present investigation, all the Actinobacteria strains were able to produce IAA at concentrations varying between 2.75 and 128.44 mg/L (
Table 3). After 11 days of incubation,
Streptomyces griseorubens BC10 was found to be the best strain for IAA production (128.44 mg/L). These results exceeded the ones previously reported by Nafis et al. [
43] (75.54 mg/L obtained by
Streptomyces sp. MNC-1 isolated from the Merzouga Moroccan desert). The lowest IAA production was observed for
Streptomyces anulatus strain P13 with 2.75 mg/L (
Table 3). These results were in agreement with those reported by Doumbou et al. [
58]. On the other hand, all the Actinobacteria strains produced ammonia and hydrogen cyanide at different levels (
Table 3). These two compounds play a crucial role in the suppression of plant diseases. HCN is a volatile compound with antifungal properties, while ammonia has been reported to have a direct role in alleviating biotic stress [
59,
60].
Most of the evaluated strains had a positive effect on the germination and growth traits of wheat seedlings (
Table 4,
Figure 5 and
Figure 6), which is in agreement with the work of Sharma et al. [
60], who demonstrated that the use of PSB inoculants (
P. fluorescens and
B. megaterium) improves the radicle and plumule lengths by 59.7% and 56.4%, respectively, compared to non-inoculated treatments. Under greenhouse conditions, the selected Actinobacteria strains (
Streptomyces alboviridis P18,
Streptomyces griseorubens BC3,
Streptomyces griseorubens BC10, and
Nocardiopsis alba BC11) significantly enhanced several wheat growth parameters, including root length (1.75–23.84%), shoot length (8.92–23.56%), root volume (41.57–71.46%), root dry weight (46.89–162.41%), and shoot dry weight (2.56–65.68%), over the uninoculated control (
Table 6,
Figure 7)
. The maximum root length was recorded with the treatments inoculated with
Streptomyces griseorubens BC10 and
Nocardiopsis alba BC11 (19.35 and 23.84%, respectively). Those results support our in vitro evaluation of the tested Actinobacteria strains in which a high amount of IAA was positively correlated with the improvement of plant growth parameters. In fact, these phytohormones are known to stimulate wheat germination, initiate root formation, and accelerate plant growth by enhancing root length and growth, thus enabling the plant to have greater access to soil nutrients and water [
61]. Consequently, the enhancement in root length might be due to the combined effect of the higher amount of produced IAA (128.44 mg/L) by
Streptomyces griseorubens BC10 and the mobilization of P and K from RP and mica, respectively. Interestingly, these elite strains (
Streptomyces griseorubens BC10 and
Nocardiopsis alba BC11) were equally efficient in improving both wheat and maize plant growth development [
62]. Our findings are also in concordance with the investigation of Sreevidya et al. [
63], who reported an increase in chickpea root length (17%) and shoot length (3%) following
Streptomyces inoculation. In fact, with their abilities to produce various PGP-related molecules,
Streptomyces are well documented in the literature [
44,
64]. As a hypothesis, the enhancement of the morphological parameters of wheat seeds by
Streptomyces griseorubens BC10 and
Nocardiopsis alba BC11 is plausibly due to their PGP traits, including IAA and siderophore production [
65].