Ocean Acidification Induces Changes in Virus–Host Relationships in Mediterranean Benthic Ecosystems
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Areas
2.2. Sediment Sampling Strategy and Storage
2.3. Biochemical Composition of Sediment Organic Matter
2.4. Viral Abundance, Production and Viral-Induced Prokaryotic Mortality
2.5. Prokaryotic Abundance
2.6. Extracellular Enzymatic Activities
2.7. Prokaryotic Diversity
2.8. Statistical Analyses
3. Results
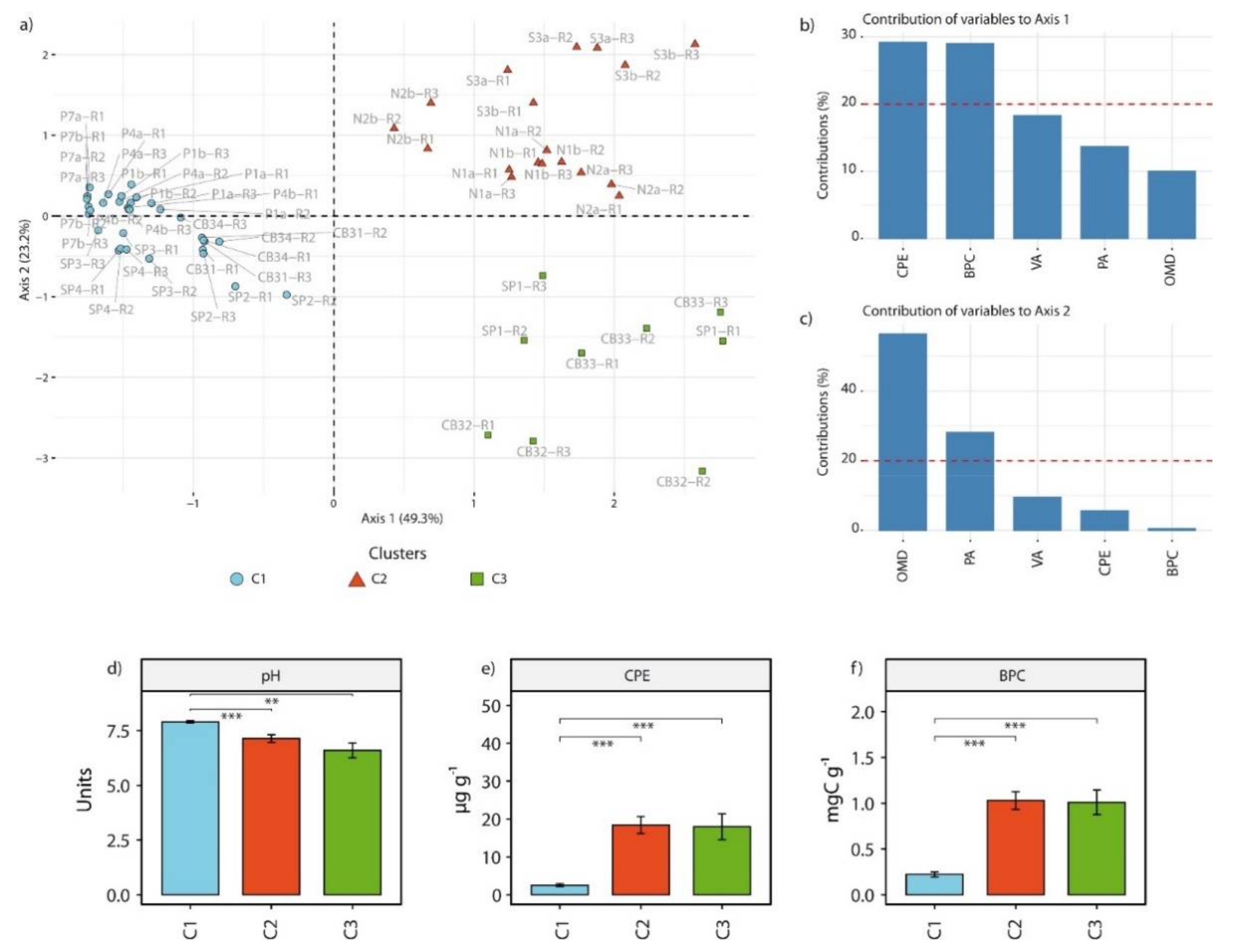
3.1. Environmental Characteristics
3.2. Effects of Acidification on Biological Variables
3.3. Prokaryotic Abundance and Activity
3.4. Viral Abundance and Production and Viral Impact on Prokaryotes
3.5. Microbial Assemblage Diversity and Structure
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Solomon, S.; Plattner, G.-K.; Knutti, R.; Friedlingstein, P. Irreversible climate change due to carbon dioxide emissions. Proc. Natl. Acad. Sci. USA 2009, 106, 1704–1709. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R.; Corinaldesi, C.; Dell’Anno, A.; Fuhrman, J.A.; Middelburg, J.J.; Noble, R.T.; Suttle, C.A. Marine viruses and global climate change. FEMS Microbiol. Rev. 2011, 35, 993–1034. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M.M.B., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; p. 1535. [Google Scholar]
- Chan, I.; Peng, S.H.; Chang, C.F.; Hung, J.J.; Hwang, J.S. Effects of acidified seawater on the skeletal structure of a Scleractinian coral from evidence identified by SEM. Zool. Stud. 2012, 51, 1319–1331. [Google Scholar]
- Hassenrück, C.; Fink, A.; Lichtschlag, A.; Tegetmeyer, H.E.; De Beer, D.; Ramette, A. Quantification of the effects of ocean acidification on sediment microbial communities in the environment: The importance of ecosystem approaches. FEMS Microbiol. Ecol. 2016, 92, fiw027. [Google Scholar] [CrossRef] [PubMed]
- Molari, M.; Guilini, K.; Lott, C.; Weber, M.; De Beer, D.; Meyer, S.; Ramette, A.; Wegener, G.; Wenzhöfer, F.; Martin, D.; et al. CO2 leakage alters biogeochemical and ecological functions of submarine sands. Sci. Adv. 2018, 4, eaao2040. [Google Scholar] [CrossRef]
- Ravaglioli, C.; Bulleri, F.; Rühl, S.; McCoy, S.J.; Findlay, H.S.; Widdicombe, S.; Queirós, A.M. Ocean acidification and hypoxia alter organic carbon fluxes in marine soft sediments. Glob. Chang. Biol. 2019, 25, 4165–4178. [Google Scholar] [CrossRef] [PubMed]
- Amaro, T.; Bertocci, I.; Queirós, A.; Rastelli, E.; Borgersen, G.; Brkljacic, M.; Nunes, J.; Sorensen, K.; Danovaro, R.; Widdicombe, S. Effects of sub-seabed CO2 leakage: Short- and medium-term responses of benthic macrofaunal assemblages. Mar. Pollut. Bull. 2018, 128, 519–526. [Google Scholar] [CrossRef]
- Sanda, T.; Hamasaki, K.; Dan, S.; Kitada, S. Expansion of the northern geographical distribution of land hermit crab populations: Colonization and overwintering success of Coenobita purpureus on the Coast of the Boso Peninsula, Japan. Zool. Stud. 2019, 58, e25. [Google Scholar]
- Cerrano, C.; Cardini, U.; Bianchelli, S.; Corinaldesi, C.; Pusceddu, A.; Danovaro, R. Red coral extinction risk enhanced by ocean acidification. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Hofmann, G.E.; Barry, J.P.; Edmunds, P.J.; Gates, R.D.; Hutchins, D.A.; Klinger, T.; Sewell, M.A. The effect of ocean acidification on calcifying organisms in marine ecosystems: An organism-to-ecosystem perspective. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 127–147. [Google Scholar] [CrossRef]
- Koch, M.; Bowes, G.; Ross, C.; Zhang, X.-H. Climate change and ocean acidification effects on seagrasses and marine macroalgae. Glob. Chang. Biol. 2012, 19, 103–132. [Google Scholar] [CrossRef] [PubMed]
- Barry, J.P.; Widdicombe, S. Effects of ocean acidification on marine biodiversity and ecosystem function. In Ocean Acidification; Oxford University Press (OUP): Oxford, UK, 2011; pp. 192–209. [Google Scholar]
- Cunha, A.; Almeida, A.; Coelho, F.J.R.C.; Gomes, N.C.M.; Oliveira, V.; Santos, A.L. Bacterial extracellular enzymatic activity in globally changing aquatic ecosystems. Curr. Res. Technol. Educ. Top. Appl. Microbiol. Microb. Biotechnol. 2010, 1, 124–135. [Google Scholar]
- Sala, M.M.; Aparicio, F.L.; Balague, V.; Boras, J.A.; Borrull, E.; Cardelus, C.; Cros, L.; Gomes, A.; López-Sanz, A.; Malits, A.; et al. Contrasting effects of ocean acidification on the microbial food web under different trophic conditions. ICES J. Mar. Sci. 2016, 73, 670–679. [Google Scholar] [CrossRef]
- Deppeler, S.; Petrou, K.; Schulz, K.G.; Westwood, K.; Pearce, I.; McKinlay, J.; Davidson, A. Ocean acidification of a coastal Antarctic marine microbial community reveals a critical threshold for CO2 tolerance in phytoplankton productivity. Biogeosciences 2018, 15, 209–231. [Google Scholar] [CrossRef]
- Westwood, K.J.; Thomson, P.G.; Van Den Enden, R.L.; Maher, L.E.; Wright, S.W.; Davidson, A.T. Ocean acidification impacts primary and bacterial production in Antarctic coastal waters during austral summer. J. Exp. Mar. Biol. Ecol. 2018, 498, 46–60. [Google Scholar] [CrossRef]
- Tait, K.; Laverock, B.; Shaw, J.; Somerfield, P.J.; Widdicombe, S. Minor impact of ocean acidification to the composition of the active microbial community in an Arctic sediment. Environ. Microbiol. Rep. 2013, 5, 851–860. [Google Scholar] [CrossRef]
- Currie, A.R.; Tait, K.; Parry, H.; De Francisco-Mora, B.; Hicks, N.; Osborn, A.M.; Widdicombe, S.; Stahl, H. Marine microbial gene abundance and community composition in response to ocean acidification and elevated temperature in two contrasting coastal marine sediments. Front. Microbiol. 2017, 8, 1599. [Google Scholar] [CrossRef]
- Suttle, C.A. Viruses in the sea. Nature 2005, 437, 356–361. [Google Scholar] [CrossRef]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef]
- Weinbauer, M.G. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004, 28, 127–181. [Google Scholar] [CrossRef]
- Danovaro, R.; Dell’Anno, A.; Corinaldesi, C.; Magagnini, M.; Noble, R.T.; Tamburini, C.; Weinbauer, M.G. Major viral impact on the functioning of benthic deep-sea ecosystems. Nature 2008, 454, 1084–1087. [Google Scholar] [CrossRef]
- Danovaro, R.; Dell’Anno, A.; Corinaldesi, C.; Rastelli, E.; Cavicchioli, R.; Krupovic, M.; Prangishvili, D. Virus-mediated archaeal hecatomb in the deep seafloor. Sci. Adv. 2016, 2, e1600492. [Google Scholar] [CrossRef]
- Danovaro, R.; Corinaldesi, C.; Filippini, M.; Fischer, U.R.; Gessner, M.O.; Jacquet, S.; Magagnini, M.; Velimirov, B. Viriobenthos in freshwater and marine sediments: A review. Freshw. Biol. 2008, 53, 1186–1213. [Google Scholar] [CrossRef]
- Corinaldesi, C.; Dell’Anno, A.; Danovaro, R. Viral infections stimulate the metabolism and shape prokaryotic assemblages in submarine mud volcanoes. ISME J. 2012, 6, 1250–1259. [Google Scholar] [CrossRef]
- Rastelli, E.; Corinaldesi, C.; Dell’Anno, A.; Amaro, T.; Queirós, A.M.; Widdicombe, S.; Danovaro, R. Impact of CO2 leakage from sub-seabed carbon dioxide capture and storage (CCS) reservoirs on benthic virus–prokaryote interactions and functions. Front. Microbiol. 2015, 6, 935. [Google Scholar] [CrossRef] [PubMed]
- Brussaard, C.P.D.; Noordeloos, A.A.M.; De Witte, H.; Collenteur, M.C.J.; Schulz, K.G.; Ludwig, A.; Riebesell, U. Arctic microbial community dynamics influenced by elevated CO2 levels. Biogeosciences 2013, 10, 719–731. [Google Scholar] [CrossRef]
- Traving, S.J.; Clokie, M.R.; Middelboe, M. Increased acidification has a profound effect on the interactions between the cyanobacterium Synechococcus sp. WH7803 and its viruses. FEMS Microbiol. Ecol. 2014, 87, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R.; Rastelli, E.; Corinaldesi, C.; Tangherlini, M.; Dell’Anno, A. Marine archaea and archaeal viruses under global change. F1000Research 2017, 6, 1241. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R.; Corinaldesi, C.; Dell’Anno, A.; Rastelli, E. Potential impact of global climate change on benthic deep-sea microbes. FEMS Microbiol. Lett. 2017, 364, 214. [Google Scholar] [CrossRef]
- Middelboe, M.; Glud, R.N. Viral activity along a trophic gradient in continental margin sediments off central Chile. Mar. Biol. Res. 2006, 2, 41–51. [Google Scholar] [CrossRef]
- Lidbury, I.; Johnson, V.; Hall-Spencer, J.M.; Munn, C.B.; Cunliffe, M. Community-level response of coastal microbial biofilms to ocean acidification in a natural carbon dioxide vent ecosystem. Mar. Pollut. Bull. 2012, 64, 1063–1066. [Google Scholar] [CrossRef]
- Kerfahi, D.; Hall-Spencer, J.M.; Tripathi, B.M.; Milazzo, M.; Lee, J.; Adams, J.M. Shallow water marine sediment bacterial community shifts along a natural CO2 gradient in the Mediterranean Sea off Vulcano, Italy. Microb. Ecol. 2014, 67, 819–828. [Google Scholar] [CrossRef]
- Hall-Spencer, J.M.; Rodolfo-Metalpa, R.; Martin, S.; Ransome, E.; Fine, M.; Turner, S.M.; Rowley, S.J.; Tedesco, D.; Buia, M.-C. Volcanic carbon dioxide vents show ecosystem effects of ocean acidification. Nature 2008, 454, 96–99. [Google Scholar] [CrossRef]
- Caldeira, K.; Wickett, M.E. Anthropogenic carbon and ocean pH. Nature 2003, 425, 365. [Google Scholar] [CrossRef]
- Espa, S.; Caramanna, G.; Bouché, V. Field study and laboratory experiments of bubble plumes in shallow seas as analogues of sub-seabed CO2 leakages. Appl. Geochem. 2010, 25, 696–704. [Google Scholar] [CrossRef]
- Esposito, V.; Andaloro, F.; Canese, S.; Bortoluzzi, G.; Bo, M.; Di Bella, M.; Italiano, F.; Sabatino, G.; Battaglia, P.; Consoli, P.; et al. Exceptional discovery of a shallow-water hydrothermal site in the SW area of Basiluzzo islet (Aeolian archipelago, South Tyrrhenian Sea): An environment to preserve. PLoS ONE 2018, 13, e0190710. [Google Scholar] [CrossRef] [PubMed]
- Danovaro, R. Methods for the Study of Deep-Sea Sediments, their Functioning and Biodiversity; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Fabiano, M.; Danovaro, R.; Fraschetti, S. A three-year time series of elemental and biochemical composition of organic matter in subtidal sandy sediments of the Ligurian Sea (northwestern Mediterranean). Cont. Shelf Res. 1995, 15, 1453–1469. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2012, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 2016, 4, e2584. [Google Scholar] [CrossRef]
- Janssen, S.; McDonald, D.; Gonzalez, A.; Navas-Molina, J.A.; Jiang, L.; Xu, Z.Z.; Winker, K.; Kado, D.M.; Orwoll, E.; Manary, M.; et al. Phylogenetic placement of exact amplicon sequences improves associations with clinical information. mSystems 2018, 3, e00021-18. [Google Scholar] [CrossRef]
- Tangherlini, M.; Corinaldesi, C.; Rastelli, E.; Musco, L.; Armiento, G.; Danovaro, R.; Dell’Anno, A. Chemical contamination can promote turnover diversity of benthic prokaryotic assemblages: The case study of the Bagnoli-Coroglio bay (Southern Tyrrhenian Sea). Mar. Environ. Res. 2020, 160, 105040. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Kassambara, A.; Mundt, F. Package ‘Factoextra’; Extract and visualize the results of multivariate data analyses. 2017. Available online: https://rpkgs.datanovia.com/factoextra/ (accessed on 4 March 2021).
- Kassambara, A.; Kassambara, M.A. Package ‘ggpubr’. 2020. Available online: https://rpkgs.datanovia.com/ggpubr/ (accessed on 4 March 2021).
- Gloor, G.B.; Macklaim, J.M.; Pawlowsky-Glahn, V.; Egozcue, J.J. Microbiome datasets are compositional: And this is not optional. Front. Microbiol. 2017, 8, 2224. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.T.; Marotz, C.; Washburne, A.; Silverman, J.; Zaramela, L.S.; Edlund, A.; Knight, R. Establishing microbial composition measurement standards with reference frames. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fedarko, M.W.; Martino, C.; Morton, J.T.; González, A.; Rahman, G.; Marotz, C.A.; Minich, J.J.; Allen, E.E.; Knight, R. Visualizing ’omic feature rankings and log-ratios using Qurro. NAR Genom. Bioinform. 2020, 2, lqaa023. [Google Scholar] [CrossRef] [PubMed]
- Kerrison, P.; Hall-Spencer, J.M.; Suggett, D.J.; Hepburn, L.J.; Steinke, M. Assessment of pH variability at a coastal CO2 vent for ocean acidification studies. Estuarine Coast. Shelf Sci. 2011, 94, 129–137. [Google Scholar] [CrossRef]
- Chan, B.K.K.; Wang, T.-W.; Chen, P.-C.; Lin, C.-W.; Chan, T.-Y.; Tsang, L.M. Community structure of macrobiota and environmental parameters in shallow water hydrothermal vents off Kueishan Island, Taiwan. PLoS ONE 2016, 11, e0148675. [Google Scholar] [CrossRef] [PubMed]
- Burrell, T.J.; Maas, E.W.; Hulston, D.A.; Law, C.S. Bacterial abundance, processes and diversity responses to acidification at a coastal CO2vent. FEMS Microbiol. Lett. 2015, 362, fnv154. [Google Scholar] [CrossRef] [PubMed]
- Celussi, M.; Malfatti, F.; Annalisa, F.; Gazeau, F.; Giannakourou, A.; Pitta, P.; Tsiola, A.; Del Negro, P. Ocean acidification effect on prokaryotic metabolism tested in two diverse trophic regimes in the Mediterranean Sea. Estuarine Coast. Shelf Sci. 2017, 186, 125–138. [Google Scholar] [CrossRef]
- Gonzalez-Delgado, S.; Hernández, J.C. The importance of natural acidified systems in the study of ocean acidification: What have we learned? Adv. Mar. Biol. 2018, 80, 57–99. [Google Scholar] [PubMed]
- Orr, J.C.; Fabry, V.J.; Aumont, O.; Bopp, L.; Doney, S.C.; Feely, R.A.; Gnanadesikan, A.; Gruber, N.; Ishida, A.; Joos, F.; et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 2005, 437, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Thingstad, T.F. Elements of a theory for the mechanisms controlling abundance, diversity, and biogeochemical role of lytic bacterial viruses in aquatic systems. Limnol. Oceanogr. 2000, 45, 1320–1328. [Google Scholar] [CrossRef]
- Morris, R.M.; Rappé, M.S.; Connon, S.A.; Vergin, K.L.; Siebold, W.A.; Carlson, C.A.; Giovannoni, S.J. SAR11 clade dominates ocean surface bacterioplankton communities. Nature 2002, 420, 806–810. [Google Scholar] [CrossRef]
- Morris, R.M.; Cain, K.R.; Hvorecny, K.L.; Kollman, J.M. Lysogenic host–virus interactions in SAR11 marine bacteria. Nat. Microbiol. 2020, 5, 1011–1015. [Google Scholar] [CrossRef]
- Clifton, S.M.; Whitaker, R.J.; Rapti, Z. Temperate and chronic virus competition leads to low lysogen frequency. arXiv 2019, arXiv:1911.07233. [Google Scholar]
- Beman, J.M.; Chow, C.-E.; King, A.L.; Feng, Y.; Fuhrman, J.A.; Andersson, A.; Bates, N.R.; Popp, B.N.; Hutchins, D.A. Global declines in oceanic nitrification rates as a consequence of ocean acidification. Proc. Natl. Acad. Sci. USA 2010, 108, 208–213. [Google Scholar] [CrossRef]
- Bayer, B.; Vojvoda, J.; Offre, P.; Alves, R.J.E.; Elisabeth, N.H.; Al Garcia, J.; Volland, J.-M.; Srivastava, A.; Schleper, C.; Herndl, G.J. Physiological and genomic characterization of two novel marine thaumarchaeal strains indicates niche differentiation. ISME J. 2016, 10, 1051–1063. [Google Scholar] [CrossRef]
- Perevalova, A.A.; Svetlichny, V.A.; Kublanov, I.V.; Chernyh, N.A.; Kostrikina, N.A.; Tourova, T.P.; Bonch-Osmolovskaya, E.A. Desulfurococcus fermentans sp. nov., a novel hyperthermophilic archaeon from a Kamchatka hot spring, and emended description of the genus Desulfurococcus. Int. J. Syst. Evol. Microbiol. 2005, 55, 995–999. [Google Scholar] [CrossRef]
- Eichorst, S.A.; Trojan, D.; Roux, S.; Herbold, C.; Rattei, T.; Woebken, D. Genomic insights into the Acidobacteria reveal strategies for their success in terrestrial environments. Environ. Microbiol. 2018, 20, 1041–1063. [Google Scholar] [CrossRef] [PubMed]
- Castelle, C.J.; Brown, C.T.; Anantharaman, K.; Probst, A.J.; Huang, R.H.; Banfield, J.F. Biosynthetic capacity, metabolic variety and unusual biology in the CPR and DPANN radiations. Nat. Rev. Genet. 2018, 16, 629–645. [Google Scholar] [CrossRef] [PubMed]





| Site | Area | Latitude (N) | Longitude (E) |
|---|---|---|---|
| P1 | Presidiana | 38.038236° | 14.032400° |
| P4 | Presidiana | 38.037050° | 14.032194° |
| P7 | Presidiana | 38.041211° | 14.025622° |
| N2 | Ischia | 40.732217° | 13.964150° |
| N3 | Ischia | 40.732167° | 13.963733° |
| S3 | Ischia | 40.730883° | 13.963017° |
| SP1 | Panarea (Secca dei Pesci) | 38.594667° | 15.115617° |
| SP2 | Panarea (Secca dei Pesci) | 38.593883° | 15.111633° |
| SP3 | Panarea (Secca dei Pesci) | 38.628250° | 15.048667° |
| SP4 | Panarea (Secca dei Pesci) | 38.637117° | 15.050883° |
| CB31 | Panarea (Basiluzzo) | 38.650883° | 15.102550° |
| CB32 | Panarea (Basiluzzo) | 38.647550° | 15.108500° |
| CB33 | Panarea (Basiluzzo) | 38.650967° | 15.093333° |
| CB34 | Panarea (Basiluzzo) | 38.655950° | 15.088150° |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tangherlini, M.; Corinaldesi, C.; Ape, F.; Greco, S.; Romeo, T.; Andaloro, F.; Danovaro, R. Ocean Acidification Induces Changes in Virus–Host Relationships in Mediterranean Benthic Ecosystems. Microorganisms 2021, 9, 769. https://doi.org/10.3390/microorganisms9040769
Tangherlini M, Corinaldesi C, Ape F, Greco S, Romeo T, Andaloro F, Danovaro R. Ocean Acidification Induces Changes in Virus–Host Relationships in Mediterranean Benthic Ecosystems. Microorganisms. 2021; 9(4):769. https://doi.org/10.3390/microorganisms9040769
Chicago/Turabian StyleTangherlini, Michael, Cinzia Corinaldesi, Francesca Ape, Silvestro Greco, Teresa Romeo, Franco Andaloro, and Roberto Danovaro. 2021. "Ocean Acidification Induces Changes in Virus–Host Relationships in Mediterranean Benthic Ecosystems" Microorganisms 9, no. 4: 769. https://doi.org/10.3390/microorganisms9040769
APA StyleTangherlini, M., Corinaldesi, C., Ape, F., Greco, S., Romeo, T., Andaloro, F., & Danovaro, R. (2021). Ocean Acidification Induces Changes in Virus–Host Relationships in Mediterranean Benthic Ecosystems. Microorganisms, 9(4), 769. https://doi.org/10.3390/microorganisms9040769








