Inhibitory Effect of Ophthalmic Solutions against SARS-CoV-2: A Preventive Action to Block the Viral Transmission?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Test Compounds
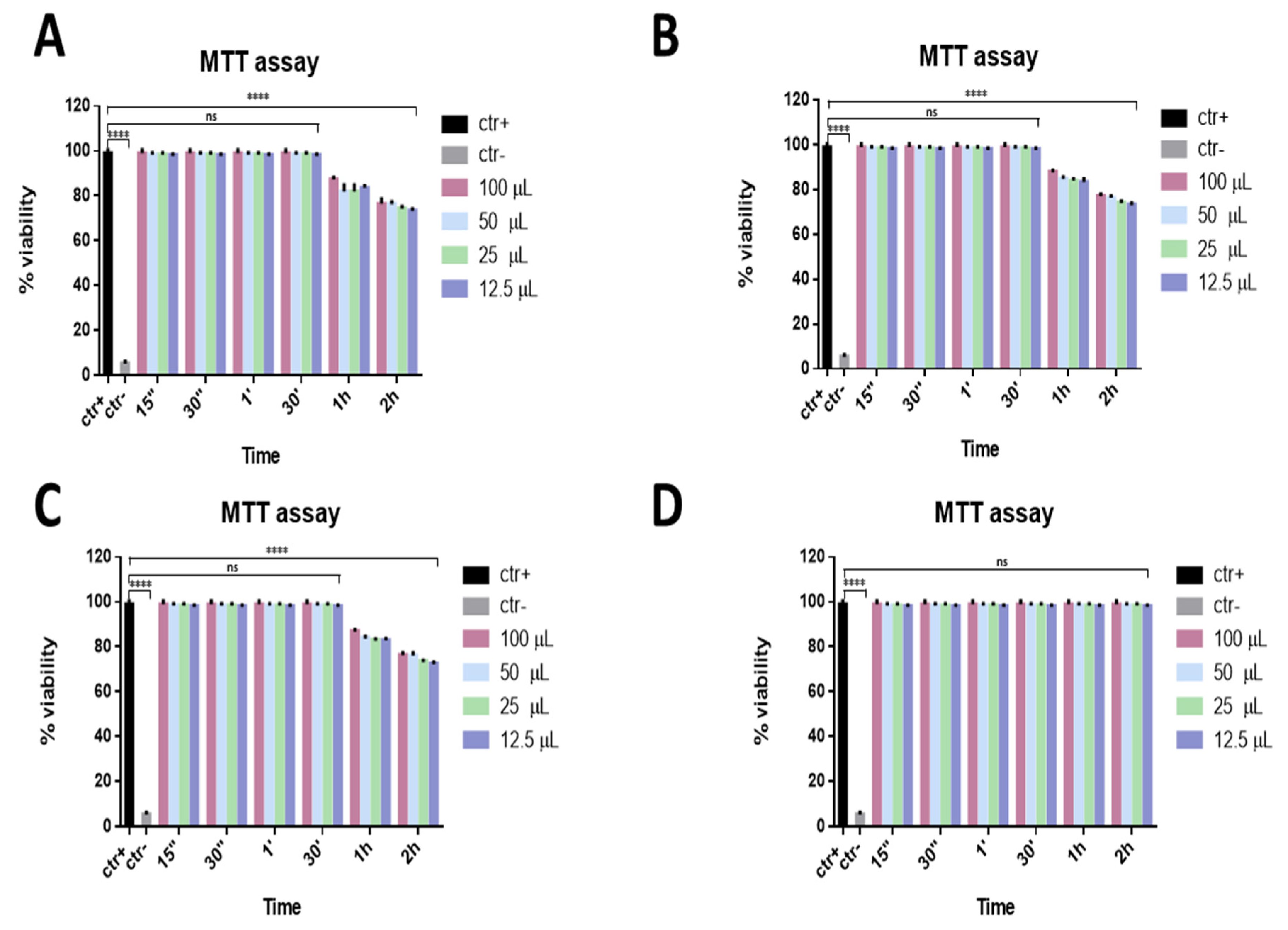
2.2. Cytotoxic Activity
2.3. Viral Strains and Cell Culture Conditions
2.4. Plaque Reduction Assays
2.5. Real-Time PCR
2.6. Statistical Analysis
3. Results
3.1. Cytotoxic Activity
3.2. Antiviral Activity against SARS-CoV-2
3.3. Real-Time PCR
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bidra, A.S.; Pelletier, J.S.; Westover, J.B.; Frank, S.; Brown, S.M.; Tessema, B. Rapid In-Vitro Inactivation of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Using Povidone-Iodine Oral Antiseptic Rinse. J. Prosthodont. 2020, 29, 529–533. [Google Scholar] [CrossRef]
- Li, J.O.; Lam, D.S.C.; Chen, Y.; Ting, D.S.W. Novel Coronavirus disease 2019 (COVID-19): The importance of recognising possible early ocular manifestation and using protective eyewear. Br. J. Ophthalmol. 2020, 104, 297–298. [Google Scholar] [CrossRef] [Green Version]
- Carta, M.G.; Scano, A.; Lindert, J.; Bonanno, S.; Rinaldi, L.; Fais, S.; Orru, G. Association between the spread of COVID-19 and weather-climatic parameters. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 8226–8231. [Google Scholar] [CrossRef]
- Lotfi, M.; Hamblin, M.R.; Rezaei, N. COVID-19: Transmission, prevention, and potential therapeutic opportunities. Clin. Chim. Acta 2020, 508, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.S.; Tessema, B.; Frank, S.; Westover, J.B.; Brown, S.M.; Capriotti, J.A. Efficacy of Povidone-Iodine Nasal and Oral Antiseptic Preparations Against Severe Acute Respiratory Syndrome-Coronavirus 2 (SARS-CoV-2). Ear Nose Throat J. 2021, 100, 192S–196S, Erratum in 2020. [Google Scholar] [CrossRef]
- Dimitriewna, T.N.; Wasilewna, D.L. Organization of preventive health care for children in the USSR. Arztl Jugendkd 1988, 79, 91–96. [Google Scholar] [PubMed]
- Chu, D.K.; Akl, E.A.; Duda, S.; Solo, K.; Yaacoub, S.; Schunemann, H.J.; COVID-19 Systematic Urgent Review Group Effort (SURGE) Study Authors. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: A systematic review and meta-analysis. Lancet 2020, 395, 1973–1987. [Google Scholar] [CrossRef]
- Lai, T.H.T.; Tang, E.W.H.; Chau, S.K.Y.; Li, K.K.W. Reply to ocular manifestation, eye protection, and COVID-19. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 1341. [Google Scholar] [CrossRef] [Green Version]
- Valitutti, F.; Zenzeri, L.; Mauro, A.; Pacifico, R.; Borrelli, M.; Muzzica, S.; Boccia, G.; Tipo, V.; Vajro, P. Effect of Population Lockdown on Pediatric Emergency Room Demands in the Era of COVID-19. Front. Pediatr. 2020, 8, 521. [Google Scholar] [CrossRef]
- Hung, O.; Lehmann, C.; Coonan, T.; Murphy, M.; Stewart, R. Personal protective equipment during the COVID-19 pandemic (Letter #2). Can. J. Anaesth 2020, 67, 1649–1650. [Google Scholar] [CrossRef]
- Feng, Y.; Armenti, S.T.; Mian, S.I. COVID-19 and the Eye: A Comprehensive Review of the Literature. Int. Ophthalmol. Clin. 2021, 61, 1–14. [Google Scholar] [CrossRef]
- Petrillo, F.; Pignataro, D.; Lavano, M.A.; Santella, B.; Folliero, V.; Zannella, C.; Astarita, C.; Gagliano, C.; Franci, G.; Avitabile, T.; et al. Current Evidence on the Ocular Surface Microbiota and Related Diseases. Microorganisms 2020, 8, 1033. [Google Scholar] [CrossRef]
- Lawrenson, J.G.; Buckley, R.J. COVID-19 and the eye. Ophthalmic Physiol. Opt. 2020, 40, 383–388. [Google Scholar] [CrossRef]
- Mungmungpuntipantip, R.; Wiwanitkit, V. Ocular manifestation, eye protection, and COVID-19. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 1339. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Y.; Wang, K.; Zhu, Y.; Lyu, D.; Yao, K. COVID-19 and the Eye. J. Infect. 2020, 81, e122–e123. [Google Scholar] [CrossRef]
- Seah, I.; Agrawal, R. Can the Coronavirus Disease 2019 (COVID-19) Affect the Eyes? A Review of Coronaviruses and Ocular Implications in Humans and Animals. Ocul. Immunol. Inflamm. 2020, 28, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Willcox, M.D.; Walsh, K.; Nichols, J.J.; Morgan, P.B.; Jones, L.W. The ocular surface, coronaviruses and COVID-19. Clin. Exp. Optom. 2020, 103, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, M.; Galdiero, M.; Galdiero, M. Inhibition of viral-induced membrane fusion by peptides. Protein Pept. Lett. 2009, 16, 786–793. [Google Scholar] [CrossRef]
- Ho, D.; Low, R.; Tong, L.; Gupta, V.; Veeraraghavan, A.; Agrawal, R. COVID-19 and the Ocular Surface: A Review of Transmission and Manifestations. Ocul. Immunol. Inflamm. 2020, 28, 726–734. [Google Scholar] [CrossRef] [PubMed]
- de Freitas Santoro, D.; de Sousa, L.B.; Camara, N.O.S.; de Freitas, D.; de Oliveira, L.A. SARS-COV-2 and Ocular Surface: From Physiology to Pathology, a Route to Understand Transmission and Disease. Front. Physiol. 2021, 12, 612319. [Google Scholar] [CrossRef] [PubMed]
- Shetty, R.; Lalgudi, V.G.; Khamar, P.; Gupta, K.; Sethu, S.; Nair, A.; Honavar, S.G.; Ghosh, A.; D’Souza, S. Potential ocular and systemic COVID-19 prophylaxis approaches for healthcare professionals. Indian J. Ophthalmol. 2020, 68, 1349–1356. [Google Scholar] [CrossRef]
- Guemes-Villahoz, N.; Burgos-Blasco, B.; Vidal-Villegas, B.; Garcia-Feijoo, J.; Arriola-Villalobos, P.; Martinez-de-la-Casa, J.M.; Diaz-Valle, D.; Konstas, A.G. Novel Insights into the Transmission of SARS-CoV-2 Through the Ocular Surface and its Detection in Tears and Conjunctival Secretions: A Review. Adv. Ther. 2020, 37, 4086–4095. [Google Scholar] [CrossRef] [PubMed]
- Chopra, A.; Sivaraman, K.; Radhakrishnan, R.; Balakrishnan, D.; Narayana, A. Can povidone iodine gargle/mouthrinse inactivate SARS-CoV-2 and decrease the risk of nosocomial and community transmission during the COVID-19 pandemic? An evidence-based update. Jpn. Dent. Sci. Rev. 2021, 57, 39–45. [Google Scholar] [CrossRef]
- Stathis, C.; Victoria, N.; Loomis, K.; Nguyen, S.A.; Eggers, M.; Septimus, E.; Safdar, N. Review of the use of nasal and oral antiseptics during a global pandemic. Future Microbiol. 2021, 16, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.H.; Bleier, B.S. Prophylactic and therapeutic topical povidone-iodine in coronavirus disease 2019 (COVID-19): What is the evidence? Int. Forum Allergy Rhinol. 2020, 10, 1271–1273. [Google Scholar] [CrossRef]
- Frank, S.; Brown, S.M.; Capriotti, J.A.; Westover, J.B.; Pelletier, J.S.; Tessema, B. In Vitro Efficacy of a Povidone-Iodine Nasal Antiseptic for Rapid Inactivation of SARS-CoV-2. JAMA Otolaryngol. Head Neck Surg. 2020, 146, 1054–1058. [Google Scholar] [CrossRef] [PubMed]
- Workman, A.D.; Welling, D.B.; Carter, B.S.; Curry, W.T.; Holbrook, E.H.; Gray, S.T.; Scangas, G.A.; Bleier, B.S. Endonasal instrumentation and aerosolization risk in the era of COVID-19: Simulation, literature review, and proposed mitigation strategies. Int. Forum Allergy Rhinol. 2020, 10, 798–805. [Google Scholar] [CrossRef] [Green Version]
- Mady, L.J.; Kubik, M.W.; Baddour, K.; Snyderman, C.H.; Rowan, N.R. Consideration of povidone-iodine as a public health intervention for COVID-19: Utilization as “Personal Protective Equipment” for frontline providers exposed in high-risk head and neck and skull base oncology care. Oral Oncol. 2020, 105, 104724. [Google Scholar] [CrossRef] [PubMed]
- Accardo, A.; Vitiello, M.; Tesauro, D.; Galdiero, M.; Finamore, E.; Martora, F.; Mansi, R.; Ringhieri, P.; Morelli, G. Self-assembled or mixed peptide amphiphile micelles from Herpes simplex virus glycoproteins as potential immunomodulatory treatment. Int. J. Nanomed. 2014, 9, 2137–2148. [Google Scholar] [CrossRef] [Green Version]
- Bidra, A.S.; Pelletier, J.S.; Westover, J.B.; Frank, S.; Brown, S.M.; Tessema, B. Comparison of In Vitro Inactivation of SARS CoV-2 with Hydrogen Peroxide and Povidone-Iodine Oral Antiseptic Rinses. J. Prosthodont. 2020, 29, 599–603. [Google Scholar] [CrossRef]
- Balakrishnan, K.; Schechtman, S.; Hogikyan, N.D.; Teoh, A.Y.B.; McGrath, B.; Brenner, M.J. COVID-19 Pandemic: What Every Otolaryngologist-Head and Neck Surgeon Needs to Know for Safe Airway Management. Otolaryngol. Head Neck Surg. 2020, 162, 804–808. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kariwa, H.; Fujii, N.; Takashima, I. Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions and chemical reagents. Dermatology 2006, 212 (Suppl. 1), 119–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valdenassi, L.; Franzini, M.; Ricevuti, G.; Rinaldi, L.; Galoforo, A.C.; Tirelli, U. Potential mechanisms by which the oxygen-ozone (O2-O3) therapy could contribute to the treatment against the coronavirus COVID-19. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 4059–4061. [Google Scholar] [CrossRef]
- Singh, M.; Zannella, C.; Folliero, V.; Di Girolamo, R.; Bajardi, F.; Chianese, A.; Altucci, L.; Damasco, A.; Del Sorbo, M.R.; Imperatore, C.; et al. Combating Actions of Green 2D-Materials on Gram Positive and Negative Bacteria and Enveloped Viruses. Front. Bioeng Biotechnol. 2020, 8, 569967. [Google Scholar] [CrossRef]
- Galdiero, M.; Galdiero, M.; Folliero, V.; Zannella, C.; De Filippis, A.; Mali, A.; Rinaldi, L.; Franci, G. SARS-CoV-2 vaccine development: Where are we? Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 2752–2784. [Google Scholar] [CrossRef]
- Celenza, G.; Iorio, R.; Cracchiolo, S.; Petricca, S.; Costagliola, C.; Cinque, B.; Segatore, B.; Amicosante, G.; Bellio, P. Antimycotic Activity of Ozonized Oil in Liposome Eye Drops against Candida spp. Transl. Vis. Sci. Technol. 2020, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Kredics, L.; Narendran, V.; Shobana, C.S.; Vagvolgyi, C.; Manikandan, P.; Indo-Hungarian Fungal Keratitis Working Group. Filamentous fungal infections of the cornea: A global overview of epidemiology and drug sensitivity. Mycoses 2015, 58, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.S.; Fung, S.S.M.; Macleod, D.; Dart, J.K.G.; Tuft, S.J.; Burton, M.J. Altered Patterns of Fungal Keratitis at a London Ophthalmic Referral Hospital: An Eight-Year Retrospective Observational Study. Am. J. Ophthalmol. 2016, 168, 227–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stelitano, D.; Franci, G.; Chianese, A.; Galdiero, S.; Morelli, G.; Galdiero, M. HSV membrane glycoproteins, their function in viral entry and their use in vaccine studies. In Amino Acids, Peptides and Proteins: Volume 43; The Royal Society of Chemistry: London, UK, 2019; Volume 43, pp. 14–43. [Google Scholar]
- Franci, G.; Crudele, V.; Della Rocca, M.T.; Melardo, C.; Chianese, A.; Finamore, E.; Bencivenga, F.; Astorri, R.; Vitiello, M.; Galdiero, E.; et al. Epstein-Barr Virus Seroprevalence and Primary Infection at the University Hospital Luigi Vanvitelli of Naples from 2007 to 2017. Intervirology 2019, 62, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Spadea, L.; Tonti, E.; Spaterna, A.; Marchegiani, A. Use of Ozone-Based Eye Drops: A Series of Cases in Veterinary and Human Spontaneous Ocular Pathologies. Case Rep. Ophthalmol. 2018, 9, 287–298. [Google Scholar] [CrossRef] [Green Version]
- Elvis, A.M.; Ekta, J.S. Ozone therapy: A clinical review. J. Nat. Sci. Biol. Med. 2011, 2, 66–70. [Google Scholar] [CrossRef] [Green Version]
- Sechi, L.A.; Lezcano, I.; Nunez, N.; Espim, M.; Dupre, I.; Pinna, A.; Molicotti, P.; Fadda, G.; Zanetti, S. Antibacterial activity of ozonized sunflower oil (Oleozon). J. Appl. Microbiol. 2001, 90, 279–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santella, B.; Folliero, V.; Pirofalo, G.M.; Serretiello, E.; Zannella, C.; Moccia, G.; Santoro, E.; Sanna, G.; Motta, O.; De Caro, F.; et al. Sepsis-A Retrospective Cohort Study of Bloodstream Infections. Antibiotics 2020, 9, 851. [Google Scholar] [CrossRef]
- Pinna, A.; Donadu, M.G.; Usai, D.; Dore, S.; D’Amico-Ricci, G.; Boscia, F.; Zanetti, S. In vitro antimicrobial activity of a new ophthalmic solution containing povidone-iodine 0.6% (IODIM(R)). Acta Ophthalmol. 2020, 98, e178–e180. [Google Scholar] [CrossRef]
- Ugazio, E.; Tullio, V.; Binello, A.; Tagliapietra, S.; Dosio, F. Ozonated Oils as Antimicrobial Systems in Topical Applications. Their Characterization, Current Applications, and Advances in Improved Delivery Techniques. Molecules 2020, 25, 334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabracos, L.; Romanou, S.; Dontas, I.; Coulocheri, S.; Ploumidou, K.; Perrea, D. The in vitro effective antiviral action of povidone-iodine (PVP-I) may also have therapeutic potential by its intravenous administration diluted with Ringer’s solution. Med. Hypotheses 2007, 68, 272–274. [Google Scholar] [CrossRef] [PubMed]
- Sriwilaijaroen, N.; Wilairat, P.; Hiramatsu, H.; Takahashi, T.; Suzuki, T.; Ito, M.; Ito, Y.; Tashiro, M.; Suzuki, Y. Mechanisms of the action of povidone-iodine against human and avian influenza A viruses: Its effects on hemagglutination and sialidase activities. Virol. J. 2009, 6, 124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, S.; Sawant, O.B.; Mian, S.I.; Kumar, A. Povidone-Iodine Attenuates Viral Replication in Ocular Cells: Implications for Ocular Transmission of RNA Viruses. Biomolecules 2021, 11, 753. [Google Scholar] [CrossRef]
- Caruso, C.; Eletto, D.; Rinaldi, M.; Pacente, L.; Troisi, S.; Semeraro, F.; dell’Omo, R.; Costagliola, C. Effectiveness and Safety of Topical Chlorhexidine and Vitamin E TPGS in the Treatment of Acanthamoeba Keratitis: A Survey on 29 Cases. J. Clin. Med. 2020, 9, 3775. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, D.; Schiff, G.; Echler, G.; Prince, A.; Feller, M.; Briner, W. In vitro virucidal effectiveness of a 0.12%-chlorhexidine gluconate mouthrinse. J. Dent. Res. 1990, 69, 874–876. [Google Scholar] [CrossRef]
- Pelletier, J.S.; Miller, D.; Liang, B.; Capriotti, J.A. In vitro efficacy of a povidone-iodine 0.4% and dexamethasone 0.1% suspension against ocular pathogens. J. Cataract. Refract. Surg. 2011, 37, 763–766. [Google Scholar] [CrossRef] [PubMed]
- Pinna, A.; Usai, D.; Sechi, L.A.; Molicotti, P.; Zanetti, S.; Carta, A. Detection of virulence factors in Pseudomonas aeruginosa strains isolated from contact lens-associated corneal ulcers. Cornea 2008, 27, 320–326. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrillo, F.; Chianese, A.; De Bernardo, M.; Zannella, C.; Galdiero, M.; Reibaldi, M.; Avitabile, T.; Boccia, G.; Galdiero, M.; Rosa, N.; et al. Inhibitory Effect of Ophthalmic Solutions against SARS-CoV-2: A Preventive Action to Block the Viral Transmission? Microorganisms 2021, 9, 1550. https://doi.org/10.3390/microorganisms9081550
Petrillo F, Chianese A, De Bernardo M, Zannella C, Galdiero M, Reibaldi M, Avitabile T, Boccia G, Galdiero M, Rosa N, et al. Inhibitory Effect of Ophthalmic Solutions against SARS-CoV-2: A Preventive Action to Block the Viral Transmission? Microorganisms. 2021; 9(8):1550. https://doi.org/10.3390/microorganisms9081550
Chicago/Turabian StylePetrillo, Francesco, Annalisa Chianese, Maddalena De Bernardo, Carla Zannella, Marilena Galdiero, Michele Reibaldi, Teresio Avitabile, Giovanni Boccia, Massimiliano Galdiero, Nicola Rosa, and et al. 2021. "Inhibitory Effect of Ophthalmic Solutions against SARS-CoV-2: A Preventive Action to Block the Viral Transmission?" Microorganisms 9, no. 8: 1550. https://doi.org/10.3390/microorganisms9081550





