Exploiting the Innate Potential of Sorghum/Sorghum–Sudangrass Cover Crops to Improve Soil Microbial Profile That Can Lead to Suppression of Plant-Parasitic Nematodes
Abstract
:1. Introduction
2. Materials and Methods
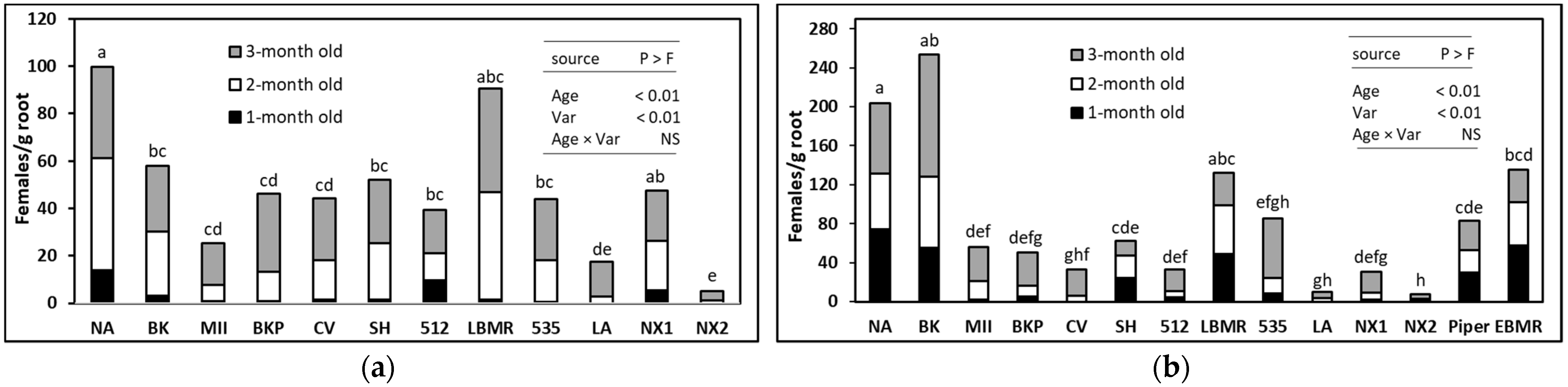
2.1. Effect of SSgH Variety and Age on the Allelopathic Effect against M. incognita
2.2. Effect of SSgH Varieties on Soil Edaphic Factors, Nematode Community, and Microbial Profile
2.2.1. Nematode Community Analysis
2.2.2. Soil Quality Analysis
2.2.3. Soil Microbial Profiling
2.2.4. Eggplant Plant Response
2.2.5. Statistical Analysis
3. Results
3.1. Effect of SSgH Variety and Plant Age on M. incognita Suppression
3.2. Effect of No-Till SSgH Cover Cropping on Soil Edaphic Properties
3.3. Effect of SSgH on Soil Nematode Communities
3.4. Effect of SSgH on Soil Microbial Profile
3.5. Relationships between Soil Health Indicators and Plant-Parasitic Nematode
3.6. Eggplant Growth and Yield
4. Discussion
4.1. Allelopathic Potential of SSgH
4.2. Effects of SSgH Cover Crop on Soil Edaphic Properties and Soil Health Indicators
4.3. Relationship between Nematode Indicators with Soil Edaphic Factors, PPNs, and Crop Yield
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jones, J.T.; Haegeman, A.; Danchin, E.G.J.; Gaur, H.S.; Helder, J.; Jones, M.G.K.; Kikuchi, T.; Manzanilla-López, R.H.; Palomares-Rius, J.E.; Wesemael, W.M.L.; et al. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef]
- Caillaud, M.-C.; Dubreuil, G.; Quentin, M.; Perfus-Barbeoch, L.; Lecomte, P.; De Almeida Engler, J.; Abad, P.; Rosso, M.-N.; Favery, B. Root-knot nematodes manipulate plant cell functions during a compatible interaction. J. Plant Physiol. 2008, 165, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Hooks, C.R.R.; Wang, K.-H.; Ploeg, A.; McSorley, R. Using marigold (Tagetes spp.) as a cover crop to protect crops from plant-parasitic nematodes. Appl. Soil Ecol. 2010, 46, 307–320. [Google Scholar] [CrossRef]
- Martin, F.N. Development of alternative strategies for management of soilborne pathogens currently controlled with methyl bromide. Annu. Rev. Phytopathol. 2003, 41, 325–350. [Google Scholar] [CrossRef] [Green Version]
- Waisen, P.; Wang, K.-H.; Uyeda, J.; Myers, R.Y. Effects of fluopyram and azadirachtin integration with sunn hemp on nematode communities in zucchini, tomato and sweet potato in Hawaii. J. Nematol. 2021, 53, e2021-30. [Google Scholar] [CrossRef]
- Meyer, S.L.F.; Zasada, I.A.; Orisajo, S.B.; Morra, M.J. Mustard seed meal mixtures: Management of Meloidogyne incognita on pepper and potential phytotoxicity. J. Nematol. 2011, 43, 7–15. [Google Scholar] [PubMed]
- Zasada, I.A.; Meyer, S.L.F.; Morra, M.J. Brassicaceous Seed Meals as Soil Amendments to Suppress the Plant-parasitic Nematodes Pratylenchus penetrans and Meloidogyne incognita. J. Nematol. 2009, 41, 221–227. [Google Scholar] [PubMed]
- Dandurand, L.-M.; Morra, M.J.; Zasada, I.A.; Phillips, W.S.; Popova, I.; Harder, C. Control of Globodera spp. Using Brassica juncea Seed Meal and Seed Meal Extract. J. Nematol. 2018, 49, 437–445. [Google Scholar] [CrossRef] [Green Version]
- Scholte, K.; Vos, J. Effects of potential trap crops and planting date on soil infestation with potato cyst nematodes and root-knot nematodes. Ann. Appl. Biol. 2000, 137, 153–164. [Google Scholar] [CrossRef]
- Kirkegaard, J.A.; Sarwar, M. Biofumigation potential of brassicas. Plant Soil 1998, 201, 71–89. [Google Scholar] [CrossRef]
- Dutta, T.K.; Khan, M.R.; Phani, V. Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: Current status and future prospects. Curr. Plant Biol. 2019, 17, 17–32. [Google Scholar] [CrossRef]
- Robson, S. Prussic Acid Poisoning in Livestock; NSW Department of Primary Industries: Albury, NSW, Australia, 2007; Volume 417. [Google Scholar]
- Panter, K.E. Cyanogenic Glycoside–Containing Plants. In Veterinary Toxicology: Basic and Clinical Principles, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 935–940. ISBN 978-0-12-811410-0. [Google Scholar]
- Gallagher, L.A.; Manoil, C. Pseudomonas aeruginosa PAO1 Kills Caenorhabditis elegans by Cyanide Poisoning. J. Bacteriol. 2001, 183, 6207–6214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, B.R.; Anderson, A.J.; Kim, Y.C. Hydrogen Cyanide Produced by Pseudomonas chlororaphis O6 Exhibits Nematicidal Activity against Meloidogyne hapla. Plant Pathol. J. 2018, 34, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Poulton, J.E.; Thayer, S.S.; Conn, E.E. Tissue Distributions of Dhurrin and of Enzymes Involved in Its Metabolism in Leaves of Sorghum bicolor. Plant Physiol. 1979, 63, 1022–1028. [Google Scholar] [CrossRef] [Green Version]
- Stapleton, J.J.; Summers, C.G.; Mitchell, J.P.; Prather, T.S. Deleterious activity of cultivated grasses (Poaceae) and residues on soilborne fungal, nematode and weed pests. Phytoparasitica 2009, 38, 61–69. [Google Scholar] [CrossRef] [Green Version]
- Nyczepir, A.P.; Rodriguez-Kabana, R. Preplant Biofumigation with Sorghum or Methyl Bromide Compared for Managing Criconemoides xenoplax in a Young Peach Orchard. Plant Dis. 2007, 91, 1607–1611. [Google Scholar] [CrossRef]
- De Brida, A.L.; Castro, B.M.D.C.E.; Zanuncio, J.C.; Serrão, J.E.; Wilcken, S.R.S. Oat, wheat and sorghum cultivars for the management of Meloidogyne enterolobii. Nematology 2018, 20, 169–173. [Google Scholar] [CrossRef]
- Djian-Caporalino, C.; Mateille, T.; Bailly-Bechet, M.; Marteu, N.; Fazari, A.; Bautheac, P.; Raptopoulo, A.; Van Duong, L.; Tavoillot, J.; Martiny, B.; et al. Evaluating sorghums as green manure against root-knot nematodes. Crop Prot. 2019, 122, 142–150. [Google Scholar] [CrossRef]
- Fortnum, B.A.; Currin Iii, R.E. Host suitability of grain sorghum cultivars to Meloidogyne spp. Ann. Appl. Nematol. 1988, 2, 61–64. [Google Scholar]
- Kratochvil, R.J.; Sardanelli, S.; Everts, K.; Gallagher, E. Evaluation of Crop Rotation and Other Cultural Practices for Management of Root-Knot and Lesion Nematodes. Agron. J. 2004, 96, 1419–1428. [Google Scholar] [CrossRef]
- Mcsorley, R.; Dickson, D.W.; De Brito, J.A. Host status of tropical rotation crops to four populations of root-knot nematodes. Nematropica 1994, 24, 45–53. [Google Scholar]
- Murata, G.; Takai, T.; Uesugi, K. A fodder sorghum cultivar, “Kyushuko 3 go”, suppresses root-knot nematode, Meloidogyne incognita. Nematology 2019, 21, 489–496. [Google Scholar] [CrossRef]
- Pattison, A.B.; Versteeg, C.; Akiew, S.; Kirkegaard, J. Resistance of Brassicaceae plants to root-knot nematode (Meloidogyne spp.) in northern Australia. Int. J. Pest Manag. 2006, 52, 53–62. [Google Scholar] [CrossRef]
- Rodriguez-Kabana, R.; Weaver, D.B.; Robertson, D.G.; King, P.S.; Carden, E.L. Sorghum in rotation with soybean for the management of cyst and root knot nematode. Nematropica 1990, 20, 111–119. [Google Scholar]
- Sipes, B.S.; Arakaki, A.S. Root-knot Nematode Management in Dryland Taro with Tropical Cover Crops. J. Nematol. 1997, 29, 721–724. [Google Scholar]
- Ibrahim, I.K.A.; Lewis, S.A.; Harshman, D.C. Host Suitability of Graminaceous Crop Cultivars for Isolates of Meloidogyne arenaria and M. incognita. J. Nematol. 1993, 25, 858–862. [Google Scholar]
- Hurd, K.; Faske, T.R. Reproduction of Meloidogyne incognita and M. graminis on Several Grain Sorghum Hybrids. J. Nematol. 2017, 49, 156–161. [Google Scholar] [CrossRef] [Green Version]
- Xavier-Mis, D.M.; Overstreet, C.; McGawley, E.C.; Doyle, V.P. Susceptibility of grain sorghum cultivars to Meloidogyne incognita in Louisiana, U.S.A. Nematropica 2017, 47, 86–98. [Google Scholar]
- Viaene, N.M.; Abawi, G.S. Management of Meloidogyne hapla on Lettuce in Organic Soil with Sudangrass as a Cover Crop. Plant Dis. 1998, 82, 945–952. [Google Scholar] [CrossRef] [Green Version]
- Mullet, J.; Morishige, D.; McCormick, R.; Truong, S.; Hilley, J.; Mckinley, B.; Anderson, R.; Olson, S.N.; Rooney, W. Energy Sorghum—a genetic model for the design of C4 grass bioenergy crops. J. Exp. Bot. 2014, 65, 3479–3489. [Google Scholar] [CrossRef] [Green Version]
- Meki, M.N.; Ogoshi, R.M.; Kiniry, J.R.; Crow, S.E.; Youkhana, A.H.; Nakahata, M.H.; Littlejohn, K. Performance evaluation of biomass sorghum in Hawaii and Texas. Ind. Crop. Prod. 2017, 103, 257–266. [Google Scholar] [CrossRef] [Green Version]
- Clark, A. Managing Cover Crops Profitably, 3rd ed.; Clark, A., Ed.; Sustainable Agriculture Network: Beltsville, MD, USA, 2012; ISBN 9781888626124. [Google Scholar]
- Krull, E.S.; Skjemstad, J.O.; Baldock, J.A. Functions of Soil Organic Matter and the Effect on Soil Properties; Cooperative Research Center for Greenhouse Accounting: Canberra, Australia, 2004; ISBN 0957959745. [Google Scholar]
- Wander, M. Soil organic matter fractions and their relevance to soil function. In Soil Organic Matter in Sustainable Agriculture; Magdoff, F., Weil, R.R., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 67–102. ISBN 9780203496374. [Google Scholar]
- Vida, C.; de Vicente, A.; Cazorla, F.M. The role of organic amendments to soil for crop protection: Induction of suppression of soilborne pathogens. Ann. Appl. Biol. 2020, 176, 1–15. [Google Scholar] [CrossRef]
- Mendes, R.; Garbeva, P.; Raaijmakers, J.M. The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol. Rev. 2013, 37, 634–663. [Google Scholar] [CrossRef] [PubMed]
- Bongers, T. The maturity index: An ecological measure of environmental disturbance based on nematode species composition. Oecologia 1990, 83, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Freckman, D.W. Bacterivorous nematodes and organic-matter decomposition. Agric. Ecosyst. Environ. 1988, 24, 195–217. [Google Scholar] [CrossRef]
- Neher, D.A. Role of Nematodes in Soil Health and Their Use as Indicators. J. Nematol. 2001, 33, 161–168. [Google Scholar] [PubMed]
- Bongers, T.; Ferris, H. Nematode community structure as a bioindicator in environmental monitoring. Trends Ecol. Evol. 1999, 14, 224–228. [Google Scholar] [CrossRef]
- Ferris, H.; Bongers, T. Nematode indicators of organic enrichment. J. Nematol. 2006, 38, 3–12. [Google Scholar]
- Ferris, H.; Matute, M.M. Structural and functional succession in the nematode fauna of a soil food web. Appl. Soil Ecol. 2003, 23, 93–110. [Google Scholar] [CrossRef]
- Ferris, H.; Bongers, T.; De Goede, R.G.M. A framework for soil food web diagnostics: Extension of the nematode faunal analysis concept. Appl. Soil Ecol. 2001, 18, 13–29. [Google Scholar] [CrossRef]
- Ingham, R.; Trofymow, J.; Ingham, E.; Coleman, D. Interactions of Bacteria, Fungi, and their Nematode Grazers: Effects on Nutrient Cycling and Plant Growth. Ecol. Monogr. 1985, 55, 119–140. [Google Scholar] [CrossRef]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallenbach, C.M.; Frey, S.D.; Grandy, A.S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 2016, 7, 13630. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, S.M.; Ahmad, M.; Zahir, Z.A.; Javaid, A.; Ashraf, M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol. Adv. 2014, 32, 429–448. [Google Scholar] [CrossRef] [PubMed]
- Philippot, L.; Ritz, K.; Pandard, P.; Hallin, S.; Martin-Laurent, F. Standardisation of methods in soil microbiology: Progress and challenges. FEMS Microbiol. Ecol. 2012, 82, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Olsson, P.A. Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soil. FEMS Microbiol. Ecol. 1999, 29, 303–310. [Google Scholar] [CrossRef]
- Williams, M.A.; Rice, C.W. Seven years of enhanced water availability influences the physiological, structural, and functional attributes of a soil microbial community. Appl. Soil Ecol. 2007, 35, 535–545. [Google Scholar] [CrossRef]
- Geisen, S.; Briones, M.J.I.; Gan, H.; Behan-Pelletier, V.M.; Friman, V.-P.; de Groot, G.A.; Hannula, S.E.; Lindo, Z.; Philippot, L.; Tiunov, A.V.; et al. A methodological framework to embrace soil biodiversity. Soil Biol. Biochem. 2019, 136. [Google Scholar] [CrossRef]
- Hussey, R.S.; Barker, K.R. A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis. Rep. 1973, 57, 1025–1028. [Google Scholar]
- Jenkins, W.R. A rapid centrifugal-flotation technique for separating nematodes from soil. Plant Dis. Report. 1964, 48, 692. [Google Scholar]
- McSorley, R.; Frederick, J.J. Extraction Efficiency of Belonolaimus longicaudatus from Sandy Soil. J. Nematol. 1991, 23, 511–518. [Google Scholar]
- Bybd, D.W.; Kirkpatrick, T.; Barker, K.R. An Improved Technique for Clearing and Staining Plant Tissues for Detection of Nematodes. J. Nematol. 1983, 15, 142–143. [Google Scholar] [PubMed]
- Byrd, D.W.; Barker, K.R.; Ferris, H.; Nusbaum, C.J.; Griffin, W.E.; Small, R.H.; Stone, C.A. Two semi-automatic elutriators for extracting nematodes and certain fungi from soil. J. Nematol. 1976, 8, 206–212. [Google Scholar]
- Yeates, G.W.; Bongers, T.; De Goede, R.G.M.; Freckman, D.W.; Georgieva, S.S. Feeding habits in soil nematode families and genera-an outline for soil ecologists. J. Nematol. 1993, 25, 315–331. [Google Scholar] [PubMed]
- Simpson, E.H. Measurement of Diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Fares, A.; Abbas, F.; Ahmad, A.; Deenik, J.L.; Safeeq, M. Response of Selected Soil Physical and Hydrologic Properties to Manure Amendment Rates, Levels, Andtypes. Soil Sci. 2008, 173, 522–533. [Google Scholar] [CrossRef]
- Netscher, C.; Sikora, R.A. Others Nematode parasites of vegetables. In Plant Parasitic Nematodes in Subtropical and Tropical Agriculture; Luc, M., Sikora, R., Bridge, J., Eds.; CAB International: Wallingford, UK, 2005; pp. 319–392. ISBN 9781845931445. [Google Scholar]
- Ter Braak, C.; Smilauer, P. Canoco Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5)—Research@WUR. Available online: https://research.wur.nl/en/publications/canoco-reference-manual-and-canodraw-for-windows-users-guide-soft (accessed on 15 July 2021).
- Akazawa, T.; Miljanich, P.; Conn, E.E. Studies on Cyanogenic Glycoside of Sorghum Vulgare. Plant Physiol. 1960, 35, 535–538. [Google Scholar] [CrossRef]
- Waisen, P.; Cheng, Z.; Sipes, B.S.; De Frank, J.; Marahatta, S.P.; Wang, K.-H. Effects of biofumigant crop termination methods on suppression of plant-parasitic nematodes. Appl. Soil Ecol. 2020, 154. [Google Scholar] [CrossRef]
- Widmer, T.L.; Abawi, G.S. Relationship Between Levels of Cyanide in Sudangrass Hybrids Incorporated into Soil and Suppression of Meloidogyne hapla. J. Nematol. 2002, 34, 16–22. [Google Scholar]
- Balota, E.L.; Colozzi-Filho, A.; Andrade, D.; Dick, R.P. Microbial biomass in soils under different tillage and crop rotation systems. Biol. Fertil. Soils 2003, 38, 15–20. [Google Scholar] [CrossRef]
- Arunachalam, A.; Arunachalam, K. Influence of gap size and soil properties on microbial biomass in a subtropical humid forest of north-east India. Plant Soil 2000, 223, 187–195. [Google Scholar] [CrossRef]
- Kumar Patel, A.; Kujur, M. Quantifying the contribution of different soil properties on microbial biomass carbon, nitrogen and phosphorous in dry tropical ecosystem. Int. J. Environ. Sci. 2012, 2, 2272–2284. [Google Scholar] [CrossRef]
- Bergmann, J.; Verbruggen, E.; Heinze, J.; Xiang, D.; Chen, B.; Joshi, J.; Rillig, M.C. The interplay between soil structure, roots, and microbiota as a determinant of plant-soil feedback. Ecol. Evol. 2016, 6, 7633–7644. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Chaudhary, A.; Kaur, A.; Choudhary, R.; Kaushik, R. Phospholipid fatty acid—A bioindicator of environment monitoring and assessment in soil ecosystem. Curr. Sci. 2005, 89, 1103–1112. [Google Scholar]
- Bardgett, R.D.; Leemans, D.K.; Cook, R.; Hobbs, P.J. Seasonality of the soil biota of grazed and ungrazed hill grasslands. Soil Biol. Biochem. 1997, 29, 1285–1294. [Google Scholar] [CrossRef]
- Blaise, D.; Velmourougane, K.; Santosh, S.; Manikandan, A. Intercrop mulch affects soil biology and microbial diversity in rainfed transgenic Bt cotton hybrids. Sci. Total. Environ. 2021, 794, 148787. [Google Scholar] [CrossRef]
- Six, J.; Paustian, K. Aggregate-associated soil organic matter as an ecosystem property and a measurement tool. Soil Biol. Biochem. 2014, 68, A4–A9. [Google Scholar] [CrossRef]
- Wilpiszeski, R.L.; Aufrecht, J.A.; Retterer, S.T.; Sullivan, M.B.; Graham, D.E.; Pierce, E.M.; Zablocki, O.D.; Palumbo, A.V.; Elias, D.A. Soil Aggregate Microbial Communities: Towards Understanding Microbiome Interactions at Biologically Relevant Scales. Appl. Environ. Microbiol. 2019, 85. [Google Scholar] [CrossRef] [Green Version]
- Fanin, N.; Kardol, P.; Farrell, M.; Nilsson, M.-C.; Gundale, M.J.; Wardle, D. The ratio of Gram-positive to Gram-negative bacterial PLFA markers as an indicator of carbon availability in organic soils. Soil Biol. Biochem. 2019, 128, 111–114. [Google Scholar] [CrossRef]
- Fierer, N.; Bradford, M.A.; Jackson, R.B. Toward an Ecological Classification of Soil Bacteria. Ecology 2007, 88, 1354–1364. [Google Scholar] [CrossRef]
- Gao, Z.; Karlsson, I.; Geisen, S.; Kowalchuk, G.; Jousset, A. Protists: Puppet Masters of the Rhizosphere Microbiome. Trends Plant Sci. 2019, 24, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Geisen, S.; Lara, E.; Mitchell, E.A.D.; Völcker, E.; Krashevska, V. Soil protist life matters! Soil Org. 2020, 92, 189–196. [Google Scholar] [CrossRef]
- Haubert, D.; Birkhofer, K.; Fliessbach, A.; Gehre, M.; Scheu, S.; Ruess, L. Trophic structure and major trophic links in conventional versus organic farming systems as indicated by carbon stable isotope ratios of fatty acids. Oikos 2009, 118, 1579–1589. [Google Scholar] [CrossRef]
- Li, C.; Fultz, L.M.; Moore-Kucera, J.; Acosta-Martínez, V.; Kakarla, M.; Weindorf, D. Soil microbial community restoration in Conservation Reserve Program semi-arid grasslands. Soil Biol. Biochem. 2018, 118, 166–177. [Google Scholar] [CrossRef]
- Trögl, J.; Jirková, I.; Kuráň, P.; Akhmetshina, E.; Brovdyová, T.; Sirotkin, A.; Kirilina, T. Phospholipid Fatty Acids as Physiological Indicators of Paracoccus denitrificans Encapsulated in Silica Sol-Gel Hydrogels. Sensors 2015, 15, 3426–3434. [Google Scholar] [CrossRef] [Green Version]
- Hinds, J.; Wang, K.-H.; Marahatta, S.P.; Meyer, S.L.F.; Hooks, C.R.R. Sunn Hemp Cover Cropping and Organic Fertilizer Effects on the Nematode Community Under Temperate Growing Conditions. J. Nematol. 2013, 45, 265–271. [Google Scholar]
- Wang, K.-H.; Sipes, B.; Hooks, C. Sunn Hemp Cover Cropping and Solarization as Alternatives to Soil Fumigants for Pineapple Production. Acta Hortic. 2011, 221–232. [Google Scholar] [CrossRef]
- Wang, K.-H.; Myers, R.Y.; Srivastava, A.; Sipes, B.S. Evaluating the predatory potential of carnivorous nematodes against Rotylenchulus reniformis and Meloidogyne incognita. Biol. Control 2015, 88, 54–60. [Google Scholar] [CrossRef]
- Becker, J.O. Effects of Rhizobacteria on Root-Knot Nematodes and Gall Formation. Phytopathology 1988, 78, 1466. [Google Scholar] [CrossRef]
- Ciancio, A. Density-Dependent Parasitism of Xiphinema diversicaudatum by Pasteuria penetrans in a Naturally Infested Field. Phytopathology 1995, 85, 144–149. [Google Scholar] [CrossRef]
- Pittelkow, C.M.; Linquist, B.A.; Lundy, M.E.; Liang, X.; van Groenigen, K.J.; Lee, J.; van Gestel, N.; Six, J.; Venterea, R.T.; van Kessel, C. When does no-till yield more? A global meta-analysis. Field Crops Res. 2015, 183, 156–168. [Google Scholar] [CrossRef] [Green Version]




 indicates two weeks after cover crop termination and eggplant planting time.
indicates two weeks after cover crop termination and eggplant planting time.
 indicates two weeks after cover crop termination and eggplant planting time.
indicates two weeks after cover crop termination and eggplant planting time.

| Cultivar | Type | Characteristics |
|---|---|---|
| Big Kahuna Plus BMR (BKP) | Forage sorghum | Brown mid-rib trait with less lignin for increased digestibility in cattle, photoperiod sensitive, 2.4–2.7 m tall, late maturing. |
| Cow Vittles II (CV) | Forage sorghum | High yield potential, early seedling vigor. |
| Bundle King (BK) | Forage sorghum | Male sterile (no grain head), exceptional sweetness, large stems (less lodging), 2.4–2.7 m tall. |
| Latte (LA) | Sorghum–sudan hybrid | Late maturing, drought resistance, 2.1–2.4 m tall. |
| 512 × 14 (512) | Sorghum–sudan hybrid | Conventional, excellent anti-lodging ability, hay grazing silage, long season. |
| NX 4264 (NX1) | Energy sorghum | Large biomass, long season hybrid, 4.5–6.1 m tall, dry stalk at the time of harvest. |
| NX-D-61 (NX2) | Energy sorghum | Large biomass, photoperiod neutral, 3–3.9 m tall, medium early maturing. |
| Age | Trial I | Trial II |
|---|---|---|
| 1 month old | 3.26 ± 0.93 a z | 22.28 ± 6.01a |
| 2 month old | 19.40 ± 3.14 b | 24.53 ± 3.96 b |
| 3 month old | 24.78 ± 2.24 c | 37.22 ± 6.95 c |
| Parameters | Treatments | |||||||
|---|---|---|---|---|---|---|---|---|
| BG | 512 | LA | BK | BKP | CV | NX1 | NX2 | |
| Abundance | -----------250 cm3----------- | |||||||
| Root-knot | 447 ± 153 ab y | 966 ± 374 bc | 1556 ± 570 a | 957 ± 312 a | 966 ± 361 ab | 923 ± 330 c | 812 ± 276 a | 725 ± 232 ab |
| Reniform | 800 ± 193 a | 666 ± 86 a | 595 ± 90 a | 376 ± 43 a | 568 ± 115 a | 709 ± 118 a | 466 ± 64 a | 489 ± 82 a |
| Bacterivores | 164 ± 30 b | 293 ± 57 a2 | 206 ± 45 ab | 179 ± 34 b | 224 ± 50 b | 325 ± 98 ab | 220 ± 46 ab | 310 ± 66 ab |
| Fungivores | 69 ± 8 a | 134 ± 22 a | 82 ± 12 a | 99 ± 17 a | 90 ± 11 a | 105 ± 15 a | 117 ± 22 a | 148 ± 28 a |
| Herbivores | 1249 ± 234 a | 1636 ± 415 a | 2161 ± 594 a | 1335 ± 330 a | 1534 ± 389 a | 1642 ± 391 a | 466 ± 64 a | 489 ± 82 a |
| Omnivores | 4 ± 1 b | 16 ± 6 a | 15 ± 5 ab | 16 ± 5 a | 18 ± 6 a | 8 ± 2 ab | 11 ± 4 ab | 19 ± 6 a |
| Indices | ||||||||
| Richness | 8 ± 0 c | 10 ± 1 a | 9 ± 0 ab | 10 ± 1 ab | 9 ± 1 bc | 9 ± 1 bc | 10 ± 1 ab | 10 ± 1 a |
| Diversity | 2.57 ± 0.26 a | 2.84 ± 0.27 a | 2.48 ± 0.25 a | 2.77 ± 0.27 a | 2.78 ± 0.20 a | 2.41 ± 0.20 a | 3.03 ± 0.29 a | 2.99 ± 0.26 a |
| EI (%) z | 1.96 ± 0.03 a | 2.02 ± 0.05 a | 2 ± 0.04 a | 2.02 ± 0.05 a | 2 ± 0.04 a | 1.97 ± 0.05 a | 1.96 ± 0.04 a | 1.99 ± 0.06 a |
| SI (%) | 45.58 ± 2.72 a | 47.19 ± 3.47 a | 48.52 ± 3.04 a | 50.75 ± 3.39 a | 50.51 ± 3.97 a | 53.02 ± 3.74 a | 54.81 ± 3.02 a | 52.18 ± 4.05 a |
| MI (%) | 0.36 ± 0.04 a | 0.33 ± 0.03 a | 0.37 ± 0.04 a | 0.38 ± 0.04 a | 0.39 ± 0.05 a | 0.4 ± 0.04 a | 0.39 ± 0.04 a | 0.37 ± 0.04 a |
| CI (%) | 11.39 ± 3.87 b | 23.97 ± 4.29 ab | 20.52 ± 4.13 ab | 24.34 ± 4.33 a | 24.49 ± 4.27 a | 22.33 ± 4.34 ab | 20.31 ± 4.79 ab | 25.42 ± 4.43 a |
| Parameters | Treatments | |||||||
|---|---|---|---|---|---|---|---|---|
| BG | 512 | LA | BK | BKP | CV | NX1 | NX2 | |
| ---27 August 2020--- | ||||||||
| Abundance | ||||||||
| TPLFA(nmole/g) z | 40.7 ± 4.8 c y | 55.6 ± 4.6 b | 78.1 ± 3.5 a | 55.1 ± 6.2 b | 60.4 ± 7.6 b | 60.1 ± 3.6 b | 53.4 ± 2.5 bc | 65.4 ± 7.5 ab |
| GN (%) | 34.6 ± 0.4 b | 36.3 ± 0.8 ab | 36.7 ± 0.5 ab | 36.8 ± 1 ab | 35.2 ± 1.5 b | 34.7 ± 0.7 b | 38 ± 0.9 a | 36.1 ± 1.2 ab |
| GP (%) | 38.7 ± 0.7 a | 36.9 ± 1.2 a | 36.8 ± 0.2 a | 39.4 ± 2.9 a | 38 ± 0.8 a | 37.1 ± 0.4 a | 38.5 ± 2.9 a | 35.5 ± 0.7 a |
| AMF (%) | 3.9 ± 0.1 a | 3.9 ± 0.1 a | 4.2 ± 0.2 a | 4 ± 0.2 a | 4.1 ± 0.3 a | 3.9 ± 0.2 a | 4.2 ± 0.1 a | 4 ± 0.1 a |
| non-AMF (%) | 2.9 ± 0.7 b | 3.9 ± 0.8 ab | 4 ± 0.2 ab | 3.7 ± 0.4 ab | 4.1 ± 0.8 ab | 5.4 ± 0.9 a | 3.7 ± 0.5 ab | 5.4 ± 1.2 a |
| EUK (%) | 0.9 ± 0.3 b | 1.4 ± 0.3 ab | 2.3 ± 0.4 ab | 0.9 ± 0.1 b | 1.6 ± 0.3 ab | 1.7 ± 0.2 ab | 1.4 ± 0.3 ab | 2.5 ± 0.6 a |
| Ratios | ||||||||
| GP/GN | 1.5 ± 0 a | 1.3 ± 0 b | 1.2 ± 0 b | 1.3 ± 0 b | 1.3 ± 0 b | 1.3 ± 0 b | 1.2 ± 0 b | 1.2 ± 0 b |
| F/B | 0.09 ± 0.01 b | 0.11 ± 0.01 ab | 0.11 ± 0 ab | 0.11 ± 0.11 ab | 0.12 ± 0.01 ab | 0.13 ± 0.01 a | 0.11 ±0.01 ab | 0.13 ± 0.02 a |
| S/U | 1.7 ± 0.1 a | 1.4 ± 0.1 ab | 1.3 ± 0 b | 1.5 ± 0 ab | 1.4 ± 0 b | 1.4 ± 0 ab | 1.4 ± 0 ab | 1.4 ± 0.1 b |
| M/P | 12.1 ± 3 a | 7.2 ± 1.2 ab | 5.8 ± 0.5 b | 7.6 ± 1.1 ab | 6.7 ± 1.1 ab | 5.1 ± 0.7 b | 7.3 ± 1.1 ab | 4.9 ± 0.7 b |
| PD/PR | 0.01 ± 0.01 a | 0.02 ± 0.01 a | 0.04 ± 0 a | 0.02 ± 0 a | 0.03 ± 0 a | 0.03 ± 0 a | 0.02 ± 0 a | 0.04 ± 0.01 a |
| ---10 December 2020--- | ||||||||
| Abundance | ||||||||
| TPLFA(nmole/g) | 41.5 ± 2.9 b | 51.7± 10.7 ab | 77.6 ± 18.2 a | 52.6 ± 6.1 abc | 54.2 ± 12.3 abc | 60.1± 11.2 ab | 34.8 ± 3.5 c | 57 ± 10.7 abc |
| GN (%) | 33.6 ± 0.4 a | 33.2 ± 1.2 a | 33.7 ± 1.5 a | 33.3 ± 0.7 a | 33.7 ± 1.1 a | 33.9 ± 0.7 a | 32.7 ± 0.1 a | 35.6 ± 0.4 a |
| GP (%) | 47.4 ± 0.5 a | 39 ± 1.5 d | 35 ± 1.8 e | 39.7 ± 2 cd | 39.4 ± 2.9 d | 37.4 ± 0.9 de | 45.8 ± 0.7 ab | 43.2 ± 1.4 bc |
| AMF (%) | 4.9 ± 0.2 a | 2.8 ± 0.5 bcd | 3.6 ± 0.4 ab | 1.6 ± 0.2 d | 3.1 ± 0.6 bc | 3.3 ± 0.5 bc | 3.8 ± 0.7 ab | 2.1 ± 0.7 cd |
| non-AMF (%) | 9.7 ± 1.3 a | 4.5 ± 0.8 a | 6.1 ± 0.9 a | 3.5 ± 1.1 a | 5.2 ± 1.3 a | 4.6 ± 0.8 a | 17.2 ± 10.4 a | 3.5 ± 2.1 b |
| EUK (%) | 1.8 ± 0.3 c | 4 ± 0.6 abc | 8 ± 3.3 a | 6.1 ± 1.3 ab | 5.5 ± 1.3 ab | 4.4 ± 0.4 abc | 1.1 ± 0.4 d | 3.3 ± 0.7 bc |
| Ratios | ||||||||
| GP/GN | 1.9 ± 0 a | 1.5 ± 0.2 bc | 1.3 ± 0 c | 1.6 ± 0.1 b | 1.6 ± 0.2 b | 1.5 ± 0.1 bc | 2 ± 0a | 1.6 ± 0.1 b |
| F/B | 0.2 ± 0 a | 0.1 ± 0 abc | 0.1 ± 0 ab | 0.1 ± 0 bc | 0.1 ± 0 abc | 0.1 ± 0 abc | 0.4 ± 0.3 a | 0.1 ± 0 c |
| S/U | 7.8 ± 0.8 ab | 2.4 ± 0.6 de | 1.5 ± 0.2 e | 2.9 ± 0.6 cd | 4.9 ± 2.8 cd | 2 ± 0.3 de | 9 ± 0.7 a | 5.4 ± 1.9 bc |
| M/P | 6.6 ± 0.8 b | 3.1 ± 0.2 bc | 2.5 ± 0.5 c | 2.5 ± 0.6 c | 2.7 ± 0.4 c | 3.1 ± 0.2 bc | 10.6 ± 2.4 a | 4.6 ± 1.2 bc |
| PD/PR | 0.02 ± 0 b | 0.1 ± 0 ab | 0.1 ± 0.07 a | 0.1 ± 0 a | 0.1 ± 0 a | 0.1 ± 0 ab | 0.02 ± 0 c | 0.1 ± 0 ab |
| Treatments | ||||||||
|---|---|---|---|---|---|---|---|---|
| Parameters | BG | 512 | LA | BK | BKP | CV | NX1 | NX2 |
| Plant height (cm) | 11.7 ± 1.91 a y | 16 ± 1.23 a | 14.02 ± 1.5 a | 13.9 ± 1.75 a | 13.71 ± 0.86 a | 14.34 ± 1.74 a | 14.5 ± 2.01 a | 14.45 ± 0.83 a |
| Total fruit wt (kg) | 2.47 ± 0.33 a | 3.64 ± 0.44 a | 2.15 ± 0.23 a | 3.4 ± 0.76 a | 2.45 ± 0.52 a | 3.63 ± 0.43 a | 2.6 ± 0.61 a | 3.63 ± 0.53 a |
| Total fruit no. | 27 ± 3.72 a | 31 ± 1.89 a | 25 ± 2.1 a | 28 ± 6.28 a | 30 ± 4.7 a | 38 ± 5.68 a | 27 ± 4.56 a | 29 ± 2.32 a |
| Root wt (kg) | 0.07 ± 0.01 b | 0.1 ± 0.01 ab | 0.09 ± 0.01 ab | 0.11 ± 0.01 a | 0.08 ± 0.01 ab | 0.10 ± 0.01 ab | 0.11 ± 0.01 ab | 0.1 ± 0.01 ab |
| RGI (0–10) z | 5.43 ± 0.43 a | 5.50 ± 0.23 a | 6.13 ± 0.55 a | 6.73 ± 0.22 a | 5.33 ± 0.35 a | 5.50 ± 0.51 a | 5.98 ± 0.38 a | 6.10 ± 0.41 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paudel, R.; Waisen, P.; Wang, K.-H. Exploiting the Innate Potential of Sorghum/Sorghum–Sudangrass Cover Crops to Improve Soil Microbial Profile That Can Lead to Suppression of Plant-Parasitic Nematodes. Microorganisms 2021, 9, 1831. https://doi.org/10.3390/microorganisms9091831
Paudel R, Waisen P, Wang K-H. Exploiting the Innate Potential of Sorghum/Sorghum–Sudangrass Cover Crops to Improve Soil Microbial Profile That Can Lead to Suppression of Plant-Parasitic Nematodes. Microorganisms. 2021; 9(9):1831. https://doi.org/10.3390/microorganisms9091831
Chicago/Turabian StylePaudel, Roshan, Philip Waisen, and Koon-Hui Wang. 2021. "Exploiting the Innate Potential of Sorghum/Sorghum–Sudangrass Cover Crops to Improve Soil Microbial Profile That Can Lead to Suppression of Plant-Parasitic Nematodes" Microorganisms 9, no. 9: 1831. https://doi.org/10.3390/microorganisms9091831







