Simple Summary
Cyclical droughts negatively impact agriculture, with deficits of water availability for the maintenance of crops destined for human food and animal production. Seasonality of forage quantity and quality is a critical obstacle to support domesticated herds over the year. Elephantgrass (Pennisetum purpureum Schum.) is a tropical forage widely used for feeding ruminants, mainly in the form of cut-and-carry, which has the potential to increase tropical pasture productivity, due to the large amount of roughage produced per unit of area. Research evaluated the response of tall and dwarf elephantgrass genotypes under irrigation considering its potential for complementing ruminant diets. This study showed that irrigation of elephantgrass, particularly during the dry season, may improve the regularity of forage production with good nutritive value.
Abstract
This two-year study evaluated the effect of Pennisetum purpureum genotypes under rainfed or irrigated conditions, during the dry and rainy seasons, on herbage, leaf, and stem dry matter (DM) accumulation rates, nutritive value, and carbohydrate and protein fractionation. Treatments were tall (Iri 381 and Elefante B) or dwarf (Mott and Taiwan A-146 2.37) genotypes under rainfed or irrigated conditions. Taiwan A-146 2.37 (146 kg DM ha per day) showed similar herbage accumulation rate (HAR) to tall genotypes during the rainy season (124 and 150 kg DM/ha per day, respectively). Dwarf genotypes showed differences in leaf accumulation rate (LAR) (66 and 49 kg DM/ha per day). Mott leaf had less neutral detergent fiber (NDF) (589 g/kg DM) than Taiwan A-146 2.37 (598 g/kg DM), and tall genotypes had generally greater NDF (668 g/kg DM) than the dwarf genotypes. Irrigation increased fiber deposition in the leaf. Stems of all genotypes had lower in vitro digestible dry matter (IVDDM) (378 g/kg DM) under rainfed conditions in the rainy season. Leaf from irrigated plots had 23% more carbohydrate C fraction (160 g/kg CHO) than those from rainfed plots (122 g/kg CHO). Dwarf genotypes had generally greater nutritive value than tall genotypes. These genotypes show promise under irrigation to fill forage gaps during dry periods.
1. Introduction
Elephantgrass [Pennisetum purpureum Schum. syn. Cenchrus purpureus (Schumach.) Morrone] is one of the most important forage grasses in the Brazilian forage-livestock production system. This forage grass is grown in tropical, subtropical, and even in semiarid regions worldwide [1], and has been successfully used as cut-and-carry forage, silage, and under grazing conditions [2] due to its potential for herbage accumulation, especially in wetter or irrigated areas [3].
The seasonality of forage production due to seasonal variation in weather conditions affects animal output from forage-dependent livestock systems. The use of irrigation may increase productivity and reduce the seasonality of forage production and, when associated with species with large potential of potential herbage accumulation such as elephant grass, can be an important alternative to mitigate forage gaps during the dry season. Under irrigated conditions, tall elephantgrass genotypes are used, but little information is available comparing the potential of irrigated dwarf and tall elephantgrass types to provide forage with good nutritive value to reduce forage deficits. Tall and dwarf elephantgrass types generally show differences in morphological characteristics, herbage accumulation, and nutritive value of forage [4].
Mott elephantgrass is a dwarf type released as a cultivar in 1989 [5] with high forage nutritive value [6] and has been considered one of the best dwarf elephantgrasses released to date. Taiwan A-146 2.37, also a dwarf type elephantgrass, is adaptable and has stable forage production under varying environmental conditions [7]. Tall genotypes are generally more productive than dwarf types but may show less stable forage production [7] and a more rapid decline in nutritive value with maturity [8].
Tall elephantgrass genotypes show greater rates of stem elongation compared to dwarf types, which may affect canopy composition and contribute to differences in forage nutritive value [9]. However, it is important to evaluate the degree of changes in nutritive value in different plant components (e.g., leaf blade, stem) of tall versus dwarf genotypes under different management practices.
Dwarf (Mott and Taiwan A-146) and tall (Elefante B and Iri 381) elephantgrasses were evaluated by [10] in a cut-and-carry system for sheep production in the humid region of Brazil and reported that the dwarf elephantgrass cultivars Mott and Taiwan A-146 2.37 showed greater nutritive value than tall Elefante B and Iri 381. Four elephantgrass genotypes (Common, Silver, Red, and Dwarf) were evaluated by [11] in the tropical humid zone of Malaysia, who observed lower forage production for dwarf (3358 kg/ha per cut), greater leaf:stem ratio (3.18), and greater nutritive value compared with the tall genotypes.
Productivity and nutritive value of elephantgrass BRS Capiaçu was evaluated by [12] under different regrowth maturity (30, 60, 90, and 120 days) during winter season in a semiarid region of Brazil. They observed that, with increase in age, herbage mass likewise increased (760; 3999; 10,798; and 22,115 kg/ha/year, respectively) while nutritive value declined, characterized by the reduction in non-protein nitrogen (fraction A) (60, 54, 56, and 35%), crude protein (15, 11, 8, and 6%) and increase of NDF concentration (55, 63, 66, and 68%). Harvest intervals of 90 days were recommended during the winter season because the elephantgrass showed the best balance among productivity, efficiency, and nutritional value. Rapid growth, however, can contribute to increasing stem accumulation and deposition of lignified tissues in tropical forages, reducing leaf: stem ratio and forage nutritive value with maturity that can be stimulated by the presence of water [13].
Our hypothesis is that dwarf elephantgrass grown under irrigation could bridge the forage gap during dry periods by maintaining greater forage nutritive value compared to tall genotypes with progressing maturity. The objective of this study was to evaluate the differences in leaf and stem accumulation, nutritive value for ruminants, and carbohydrate and protein fractionation among tall and dwarf elephantgrass genotypes grown under rainfed or irrigated conditions during the dry and rainy seasons.
2. Materials and Methods
2.1. Experimental Site, Treatments and Experimental Design
The trial was conducted in Garanhuns, in the Agreste Meridional Region, Pernambuco, Brazil, at the Experimental Farm of the Federal Rural University of Pernambuco. The experimental site is located at 8°53′ S and 36°29′ W, 896 m above sea level. The climate is classified as tropical Aw’ according to Köppen-Geiger climate classification [14].
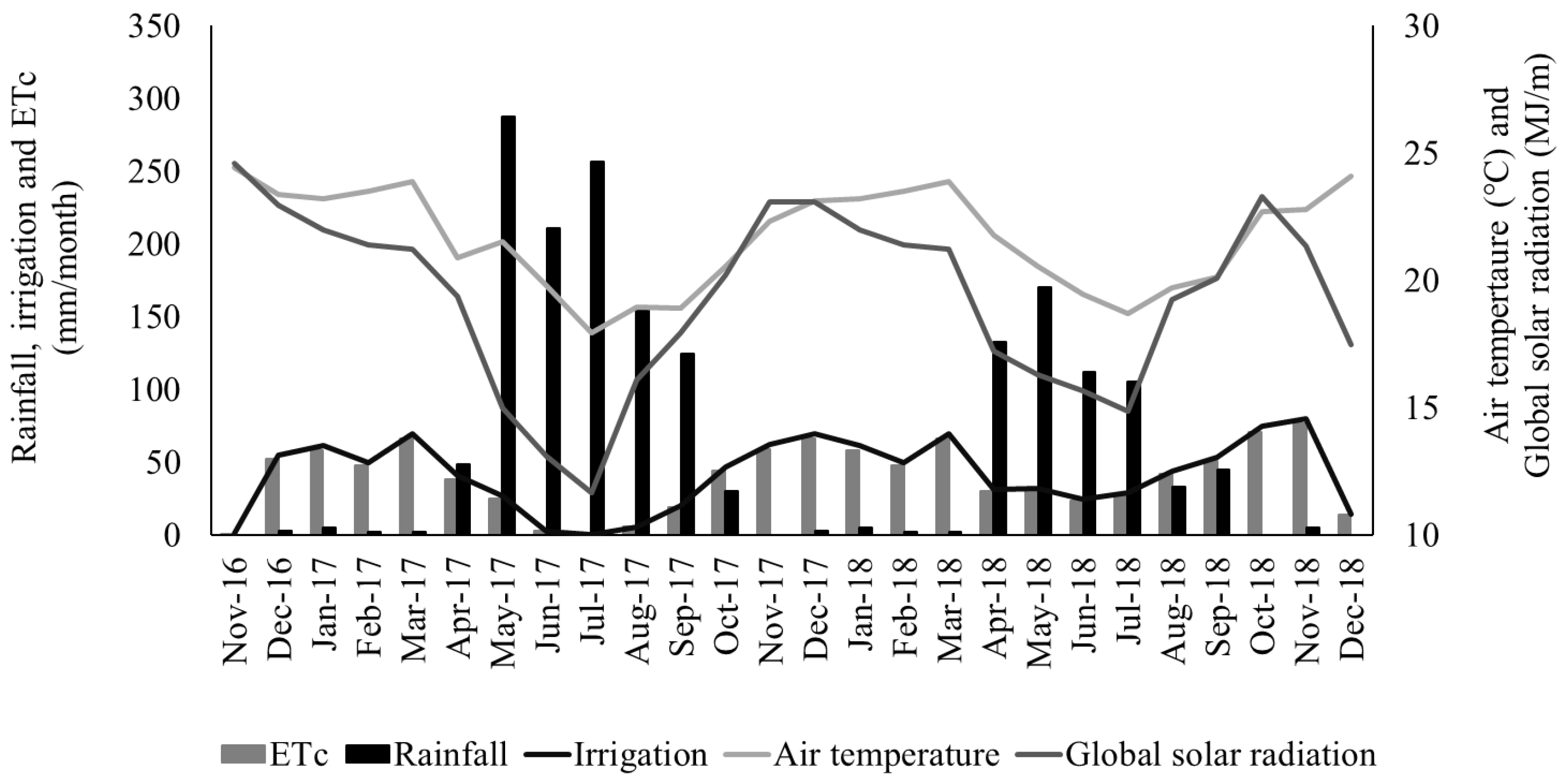
The soil of the experimental area was an Ultisol, and the texture was sandy clay loam. Average soil chemical characteristics were: pH (H2O) = 5.8; P = 4 mg/dm3 (Mehlich I); K = 0.27 cmolc/dm3; Na = 0.03 cmolc/dm3; Al = 0 cmolc/dm3; Ca = 1.8 cmolc/dm3; Mg = 0.4 cmolc/dm3; H+Al (potential acidity) = 2.5 cmolc/dm3; SB (sum by bases) = 2.51 cmolc/dm3; CEC (cation exchange capacity) = 5.01 cmolc/dm3; V (base saturation) = 50.1%; C = 2.1%; m = 0%; OM (organic matter) = 36.4 g/dm3. Weather data for the experimental period are presented in Figure 1.
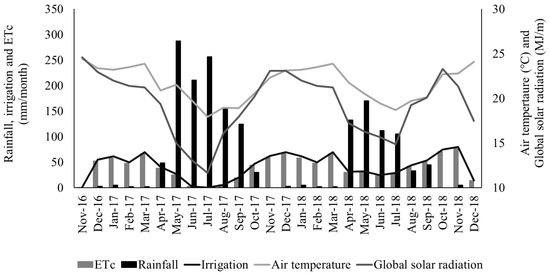
Figure 1.
Rainfall, crop evapotranspiration (ETc), daily irrigation, air temperature, and global solar radiation recorded during the experimental period at Garanhuns, Pernambuco, Brazil.
The experimental design was a randomized complete block with split-plot arrangement and four replications. The main plots consisted of rainfed (non-irrigated) and irrigated, and subplots included four elephantgrass genotypes, tall (Elefante B and Iri-381), and dwarf (Taiwan A-146 2.77 and Mott) types, as subplots.
2.2. Plot Establishment and Management
Before planting, soil sampling was carried out using an auger hole 0–10 and 10–20 cm deep, randomly collected at three points within each subplot, totaling 96 samples. A single composite and representative soil sample of the total area was formed. After preparation of the composite samples (air-dried and sieved), they were analyzed at the Soil Laboratory of the Sugarcane Experimental Station at Carpina of the Federal Rural University of Pernambuco. The pHH2O, P (Mehlich I), K+, Na+, Al3+, Ca2+, Mg2+, H+Al, SB, CEC, V, C, and OM, were determined according to [15]. The need for soil pH correction was identified, which was carried out in accordance with regional fertilizer recommendation for elephantgrass in Pernambuco State [16], applying 500 kg/ha calcitic limestone, followed by plowing and harrowing.
After 90 days of soil amendment, the soil was sampled and chemical analysis was performed again, as described above. Those samples indicated the need to add 44 kg P/ha and 64 kg K/ha using single superphosphate and potassium chloride as sources, respectively, applied to furrows during planting.
Elephantgrass genotypes were established by vegetative propagation with 1-m spacing between rows. Each plot consisted of 546 m2 (91 × 6 m), with spacing of 8 m between main plots. Each subplot had 24 m2 (4 × 6 m), and the sampling area was 15 m2 (3 × 5 m). Annual maintenance soil fertilization was performed only during rainy season in a single application, following the regional fertilizer recommendation for elephantgrass in Pernambuco State [16], consisting of 100 kg N/ha and 64 kg K/ha in the planting furrow. The fertilization sources consisted of ammonium sulfate and potassium chloride, respectively.
In irrigated plots, water was applied using a drip irrigation system with approximately 95% distribution uniformity. Irrigation management distributed the water needed for restoring 100% crop evapotranspiration (ETc) based on the standardized Penman-Monteith method by FAO/56 [17]. The weather data used for ETc calculations was obtained from a weather station from the experimental site. In the first year, 8 and 179 mm of water were applied in the irrigated plots in the rainy and in the dry season, respectively. In the second year, 98 and 168 mm were applied in the irrigated plots in the rainy and dry season, respectively.
In July 2016, 90 days after planting, a staging cut at approximately 5 cm from the ground level was performed, and dead plants were replaced. For the beginning of the experimental period, a second staging cut was performed in November 2016, 120 days after replanting. Thereafter, the genotypes were subjected to successive harvests every 60 days during two consecutive years (2017 to 2018).
2.3. Response Variables
2.3.1. Herbage, Leaf, and Stem Accumulation Rates and Nutritive Value
Two representative harvests (growing cycles) were chosen to evaluate seasonal herbage, leaf, and stem accumulation. The harvest that occurred in August and December represented the rainy and dry season, respectively, over 2 years, giving a total of 4 harvests, and results were considered as the average of harvests in each season. At each harvest, the herbage mass in each subplot was quantified by harvesting the forage at approximately 5 cm from the ground level. The forage harvested from 15 m2 of each subplot was weighed fresh in the field, and a subsample of five randomly selected basal tillers was collected. Each subsample was separated into leaf (blade), stem (stem plus leaf sheath), and dead material, weighed, and subsequently dried in a forced-draft oven at 55 °C until constant weight and then weighed to determine dry matter (DM) concentration. The leaf and stem weight of the subsamples was used to calculate the proportion of each component in the herbage mass, which was then used to calculate the leaf:stem ratio (L:S). Herbage mass was obtained by multiplying fresh weight of forage harvest from 15 m2 of each subplot by the respective DM concentration. Then, the herbage mass was divided by the number of days in the growing cycle (from the harvest of the previous cycle to the harvest of the considered cycle) to obtain the herbage accumulation rate (HAR; kg/ha per day). Leaf and stem accumulation rate (LAR and SAR; kg/ha per day) were obtained by multiplying the herbage accumulation rate by the proportion of each component (leaf and stem) divided by the regrowth duration.
The dried sub-samples were ground in a Wiley mill (MO6666, John Doe Co., Dog city, CA, USA) using a 1-mm sieve. The samples were analyzed for DM at 105 °C (930.15 method), mineral matter (MM) (942.05), ether extract (EE) (920.39), and crude protein (CP) (984.13), according to [18]. Lignin, neutral detergent fiber (NDF), and acid detergent fiber (ADF) adjusted for ash and protein (NDFap) were determined as described by [19], with modifications proposed by [20].
The in vitro digestible dry matter (IVDDM) was estimated according to [21] in a DAISY II Incubator (ANKOM® Technology, Macedon, NY, USA) for 48 h with ruminal fluid and buffer solution (ruminal fermentation stage). After this period, 40 mL of HCl solution (6N) and 8 g pepsin were added, and samples were further incubated for 24 h (chemical digestion stage). The F57 bags containing the digestion residue were oven dried at 105 °C until constant weight and weighed. The experimental procedures were approved by the Ethics committee of Federal Rural University of Pernambuco (License n° 002/2020). A single rumen-fistulated cow was used as a ruminal fluid donor. This animal was fed with elephantgrass silage.
2.3.2. Protein Fractionation
Non-protein nitrogen (fraction A), neutral detergent insoluble nitrogen (NDIN), and acid detergent insoluble nitrogen (ADIN) were analyzed as described by [22] and calculated according to the Cornell Net Carbohydrate and Protein System (CNCPS) as described by [23]. Protein fraction B1 + B2, composed by fractions of rapidly (B1) and intermediate (B2) rates of rumen degradation, was estimated by Equation (1):
B1 + B2 = 100 − (A + B3 + C)
Protein fraction B3 (insoluble fraction in the rumen) was calculated by the difference between NDIN and ADIN, and protein fraction C (insoluble in the rumen and indigestible in the small intestine) was considered as ADIN.
2.3.3. Carbohydrate Fractionation
The carbohydrate fractionation was determined and calculated according to the Equations (2)–(4) described by [22]. Total carbohydrates (CHO) were calculated as noted in Equation (2).
CHO = 100 − (CP + EE + MM)
Carbohydrate fractions A and B1 (which show rapid ruminal fermentation) were calculated as Equation (3):
NFC = 100 − (CP + (NDFap) + EE + MM)
Carbohydrate fraction B2 (carbohydrates of slow ruminal fermentation developed on cell wall) was calculated by the difference between NDFap (ash- and protein-free NDF) and fraction C. Carbohydrate fraction C (indigestible fiber) was calculated as Equation (4):
C = NDF * 0.01 * LIG * 2.4/CHO
2.4. Statistical Analysis
Data were analyzed using the Mixed Procedure of SAS University Edition. Irrigation, genotypes, seasons, and their interactions were considered fixed effects. The effects of blocks, year, and their interaction were considered random. Seasons were considered repeated measures. Averages were compared using the probability of the difference (“pdiff”) adjusted by Tukey test. Treatments were considered different when p ≤ 0.05. The following statistical model was used:
where Yijkl = observation, µ = population mean, Bi = block effect (i = 4–1), Cj = irrigation effect (j = 2–1), Dk = genotype effect (k = 4–1), El = season effect (l = 2–1), CDjk = effect of irrigation × genotype interaction, CEjl = effect of irrigation × season interaction, DEkl = effect of genotype × season interaction, and CDEjkl = effect of irrigation × genotype × season interaction and εijkl = residual error.
Yijkl = µ + Bi + Cj + Dk + El + CDjk + CEjl + DEkl + CDEjkl + εijkl
3. Results
3.1. Herbage, Leaf, and Stem Accumulation Rates and Nutritive Value
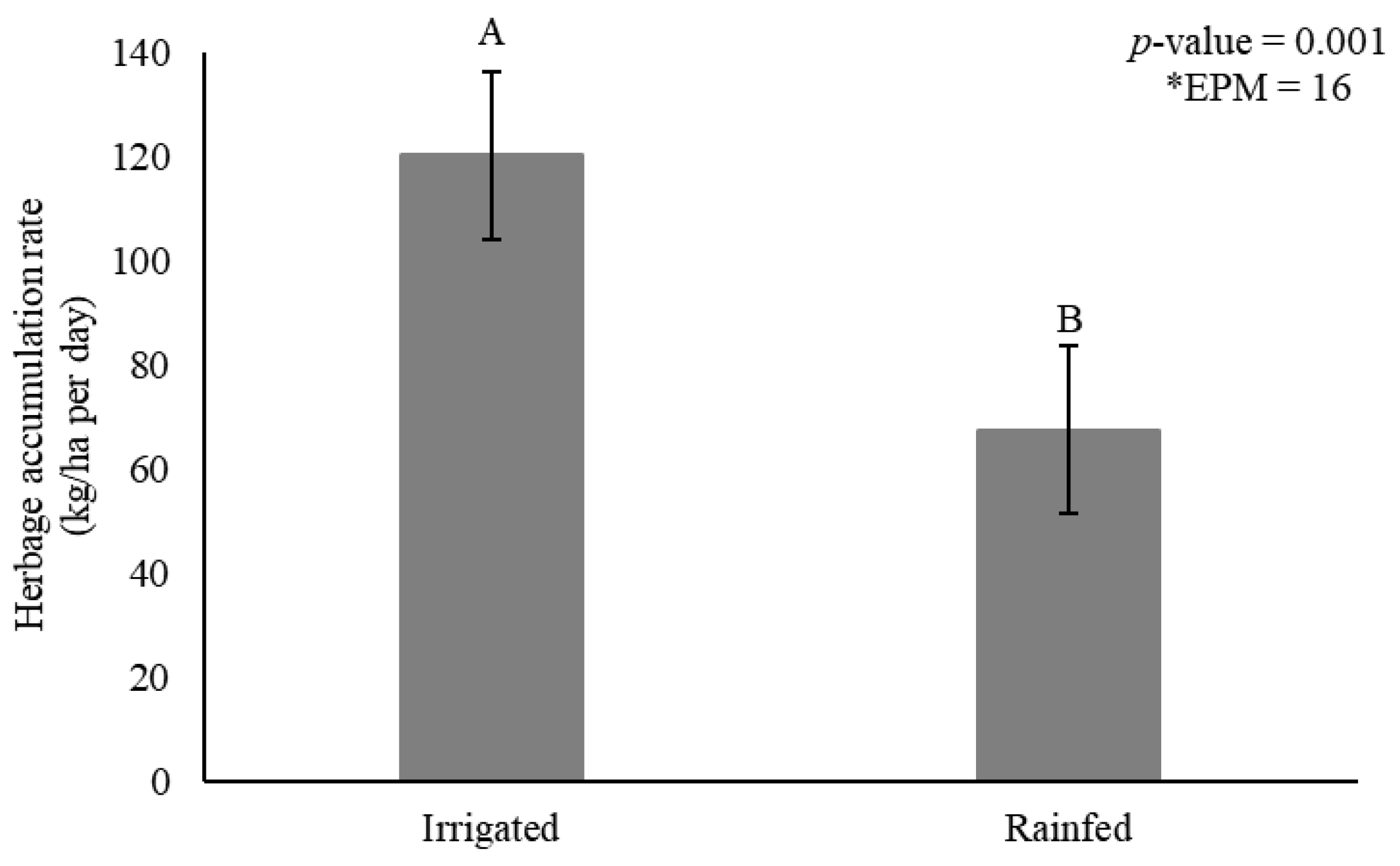
Herbage accumulation rate was affected by irrigation (p = 0.001) (Figure 2) and genotype × season interactions (p = 0.007) (Table 1).
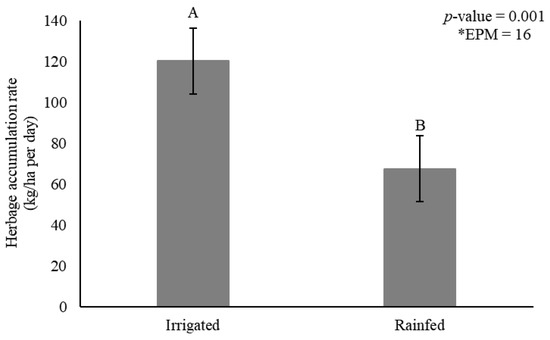
Figure 2.
Herbage accumulation rate (HAR) (kg dry matter/ha per day) as affected by irrigation for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil. *EPM = Standard error of the means.

Table 1.
Herbage accumulation rate (HAR) and stem accumulation rate (SAR; kg dry matter/ha per day), as affected by the interaction between genotype and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
Irrigation increased HAR by 44% compared to rainfed conditions (120 vs. 67 kg DM/ha per day, respectively) (Figure 2). The genotypes showed generally greater HAR during rainy season, and tall genotype Elefante B showed similar HAR compared to the dwarf types. Mott and Taiwan A-146 2.37 in turn did not differ from Iri 381, varying from 122 to 150 kg DM/ha/day (Table 1). Iri 381 showed 19% greater HAR compared to Mott during rainy season.
The SAR was not affected by irrigation (p = 0.19), but it was affected by genotype × season (p = 0.001) interaction (Table 1). The SAR was greater during the rainy season (average within genotypes ~77 kg DM/ha/day) compared to the dry season (~22 kg DM/ha/day) without differences among genotypes in the dry season. During the rainy season, Mott had the lowest SAR, while Taiwan A-146 2.37 SAR did not differ from Elefante B, which in turn did not differ from Iri 381.
There was genotype × irrigation interaction (p = 0.048) for LAR (Table 2) and irrigation × season (p = 0.005) for LAR and L:S (Table 3). It was generally greater under irrigation for all genotypes (~57 kg DM/ha/day), while under rainfed condition, genotypes did not show differences. When irrigated, Mott (66 kg DM/ha per day) had 26% greater LAR than Taiwan A-146 2.37 (49 kg DM/ha per day), but both showed no difference to Elefante B (56 kg DM/ha/day) and Iri 381 (59 kg DM/ha/day) (Table 2).

Table 2.
Leaf accumulation rate (LAR; kg dry matter/ha per day) as affected by the interaction between genotype and irrigation for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.

Table 3.
Leaf accumulation rate (LAR; kg dry matter/ha per day) and leaf:stem (L:S) ratio, as affected by the interaction between irrigation and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
In the rainy season there was no difference in the LAR between the irrigated and rainfed conditions or between both seasons when irrigation was used (Table 3). Leaf:stem ratio was affected only by the interaction of irrigation and season (p < 0.0001). In the dry season, L:S ratio was 34% greater under rainfed conditions compared to irrigation. During the rainy season, however, similar L:S ratios under both irrigated and rainfed conditions were observed (Table 3).
Leaf nutritive value (Table 4) was affected by genotype × irrigation × season interaction. Greater CP (p = 0.005) concentrations were observed for Mott under rainfed and irrigated treatments during rainy and dry seasons compared to the other genotypes, but Taiwan A-146 2.37 did not differ from Mott, Elefante B, and Iri 381. During the dry season, CP was greater in rainfed compared to irrigated conditions for all genotypes. Mott and Taiwan A-146 2.37 IVDDM (p = 0.0004) was, on average, 14% greater than Elefante B and Iri 381 (Table 4). Mott NDF (p = 0.038) and ADF (p = 0.016) concentrations were generally less. Greater NDF concentrations occurred when elephantgrass genotypes were irrigated during the dry season and under irrigated, and rainfed treatments during rainy season there was no significant difference (Table 4).

Table 4.
Leaf crude protein (CP), neutral detergent fiber (NDF), acid detergent fiber (ADF), and in vitro dry matter digestibility (IVDDM) (g/kg dry matter) as affected by the interaction between irrigation, genotype, and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
During the rainy season there was no difference between irrigated and rainfed treatment forage ADF. Lower concentrations of ADF were observed in Mott and Taiwan A-146 2.37, and Elefante B and Iri 381 did not show a difference in ADF.
Leaf lignin (g/kg DM) concentration was affected by the genotype × irrigation interaction (p = 0.038) (Table 5). Irrigation increased lignin concentration by 29% when compared to rainfed conditions for all genotypes. Taiwan A-146 2.37 did not differ from either Mott or the tall genotypes.

Table 5.
Leaf lignin (g/kg dry matter) as affected by the interaction between irrigation and genotype for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
Stem CP (p < 0.0001), ADF (p = 0.012), and lignin (p = 0.001) concentrations were affected by genotype × irrigation × season interaction (Table 6). Differences between irrigated and rainfed treatments occurred only during the dry season, with greater ADF and lignin concentrations in the stems under irrigated conditions. Taiwan A-146 2.37 showed similar stem CP compared to Elefante B under irrigated and rainfed conditions during rainy season and under irrigation during the dry season, while Iri 381 showed the lowest CP. Under rainfed conditions during dry season, Taiwan A-146 2.37 showed similar CP compared to Mott, and both were greater than Elefante B and Iri 381. Mott always contained less stem ADF and lignin concentrations than other genotypes, regardless of irrigated or rainfed conditions, while Taiwan A-146 2.37 was similar to tall genotypes. The ADF and lignin concentrations were generally less for the genotypes under rainfed conditions compared to irrigated in the dry season.

Table 6.
Stem crude protein (CP), acid detergent fiber (ADF), and lignin concentration (g/kg dry matter), as affected by the interaction between irrigation, genotype, and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
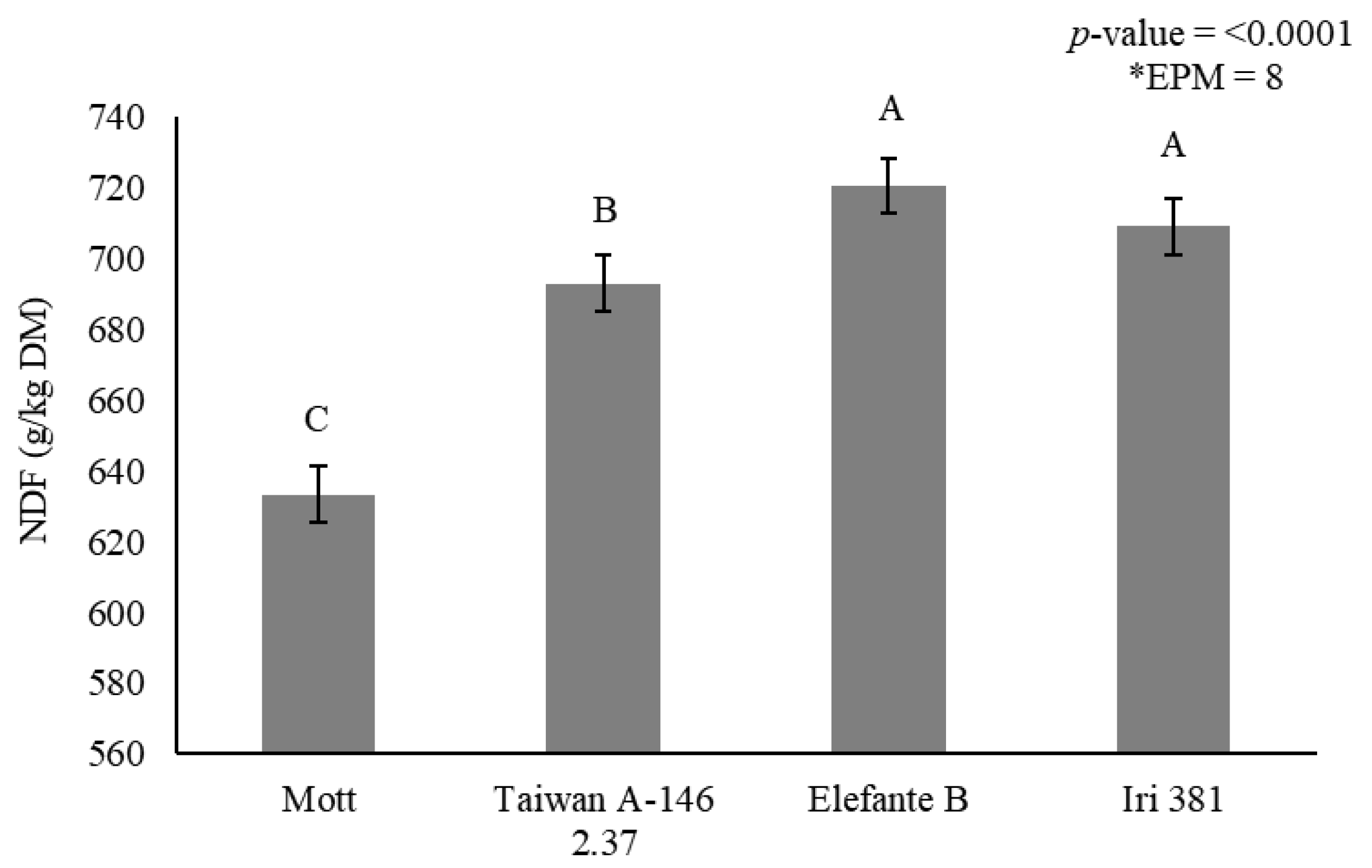
Stem NDF concentration was affected by genotype (p < 0.0001) (Figure 3) and irrigation × season interaction (p < 0.0001) (Table 7). Mott showed the lowest NDF concentration (633 g/kg DM), and Taiwan A-146 2.37 showed intermediary values (693 g/kg DM). Elefante B and Iri 381, on the other hand, showed the greatest stem NDF concentration (720 and 709 g/kg DM), respectively (Figure 3).
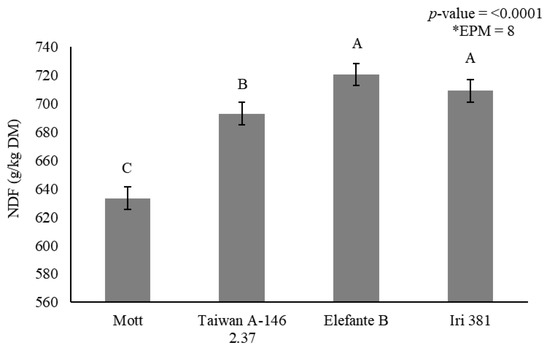
Figure 3.
Stem neutral detergent fiber (NDF) (g/kg dry matter) as affected by genotype for elephantgrass at Garanhuns, Pernambuco, Brazil. *EPM = Standard error of the means.

Table 7.
Stem neutral detergent fiber (NDF) and in vitro digestible dry matter (IVDDM) (g/kg dry matter), as affected by the interaction between irrigation and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
There was no difference in stem NDF concentration between rainy and dry season when irrigation was used, while rainfed treatments had lower stem NDF concentration during the dry season when compared to the rainy season (Table 7).
Stem IVDDM was affected by irrigation × season (p = 0.0002) (Table 7) and genotype × season interactions (p < 0.0001) (Table 8). During the rainy season, IVDDM did not show a difference between irrigated and rainfed conditions, when compared to dry season (Table 7). There was a decrease in IVDDM during the rainy season compared to the dry season (Table 8). Mott and Taiwan A-146 2.37 showed greater IVDDM compared to other genotypes and did not differ from each other (Table 8).

Table 8.
Stem in vitro dry matter digestibility (g/kg dry matter) as affected by the interaction between genotype and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
3.2. Protein Fractionation
There was an irrigation × genotype × season interaction for all protein fractionation variables from leaves and stems (Table 9 and Table 10). Leaf from Mott and Taiwan A-146 2.37 had greater proportion of N fraction A (p = 0.002) (average of irrigated and rainfed in rainy and dry season ~153 g/kg CP) than the other genotypes. Greater concentrations of this protein fraction (A) were observed in dry season forage in rainfed conditions, for all genotypes. When comparing rainy and dry season, lower concentrations of this fraction were observed for all genotypes during rainy season under rainfed conditions (Table 9).

Table 9.
Leaf protein fractionation (g/kg crude protein) as affected by the interaction between irrigation, genotype, and season for elephantgrass at Garanhuns, Pernambuco, Brazil.

Table 10.
Stem protein fractionations (g/kg crude protein) as affected by the interaction between irrigation, genotype, and season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
No difference was observed for the B1 + B2 (p < 0.0001) fraction between the genotypes leaf in all conditions analyzed. Less fraction B3 (p < 0.0001) was observed in Mott leaf compared to other genotypes. The irrigated plots accumulated greater fraction B3 in leaf during dry season. Less indigestible N (p = 0.0006) fraction in leaf was observed during dry season when no irrigation was applied (Table 9).
The proportion of fraction A of CP compounds in stems (p = 0.001) was similar during rainy and dry season for all genotypes when irrigation was used. During the dry season, fraction A in stems was lower in irrigated conditions. Fraction B1 + B2 (p < 0.0001) in stems was 24% greater in rainfed conditions during dry season compared, to irrigated plants. Fraction B3 (p < 0.0001) of Elefante B and Iri 381 were 19% greater than Mott and Taiwan A-146 2.37 in irrigated and rainfed treatments during both seasons (Table 10). Fractions B3 and C showed lower concentrations under rainfed conditions during dry season, when compared to rainy season. Mott contained less fraction C (p = 0.001) proportion in stems under rainfed conditions during the rainy and dry seasons. Taiwan A-146 2.37 had similar proportions to Elefante B and Iri 381 under rainfed conditions, during dry season (Table 10).
3.3. Carbohydrate Fractionation
There was an interaction between irrigation × genotype for all leaf carbohydrate fractionation variables (Table 11). In general, the greatest soluble carbohydrate (A + B1) (p = 0.001) concentrations were detected in plants under rainfed conditions for all genotypes. Less fraction B2 (p < 0.0001) was observed for the Mott genotype compared to other genotypes. Fraction C (p < 0.0001) represents the indigestible fiber fraction, which did not differ among genotypes and increased by 23% under irrigation conditions (Table 11).

Table 11.
Leaf carbohydrate fractionation (g/kg carbohydrate) as affected by the interaction between genotype and irrigation for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
Stem A + B1 was affected only by the interaction between irrigation × genotype (p < 0.0001) (Table 12). Mott showed greater carbohydrate concentration for fraction A + B1 in stems under irrigation and rainfed treatments, when compared with other genotypes. Carbohydrate fraction B2 in stems was affected by genotype × irrigation interaction (p < 0.0001) (Table 12) and season (p < 0.0001) (Figure 4).

Table 12.
Stem carbohydrate fractions A + B1 and B2 (g/kg carbohydrate) as affected by the interaction between genotype and irrigation for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil.
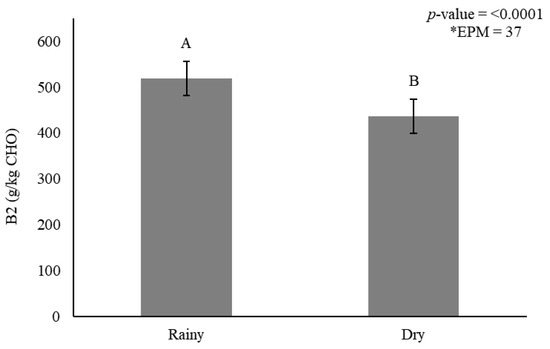
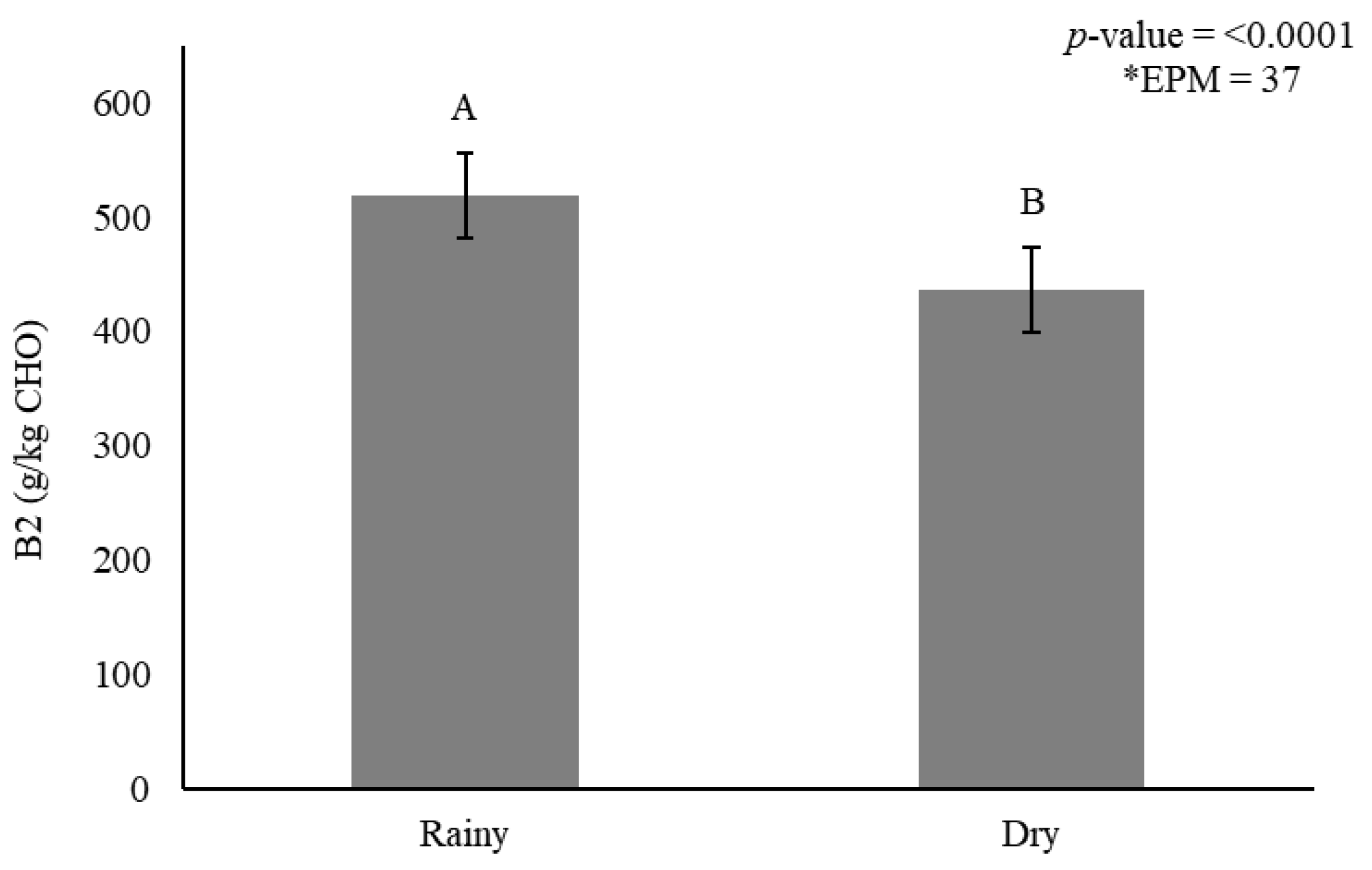
Figure 4.
Stem carbohydrate fraction B2 (g/kg carbohydrate) as affected by season for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil. *EPM = Standard error of the means.
When irrigation was used, B2 fractions increased by 16%. Among the genotypes, Mott and Taiwan A-146 2.37 had fewer B2 fractions (Table 12).
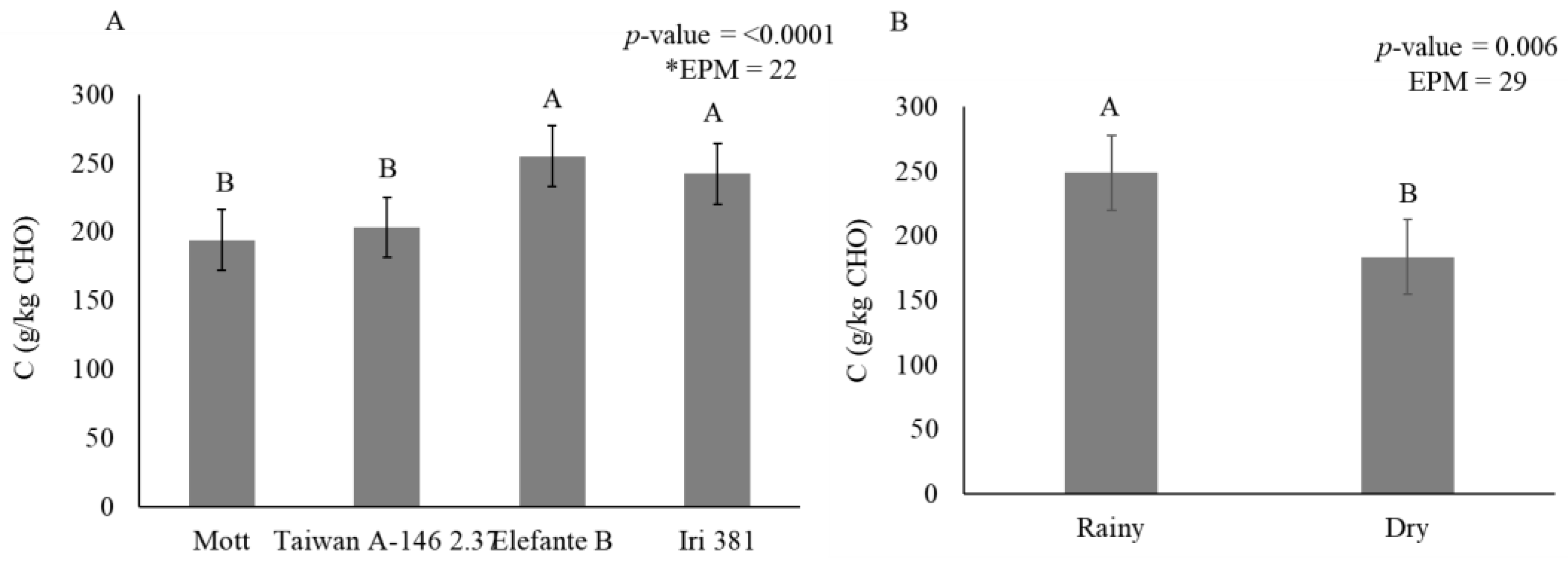
Smaller B2 fractions accumulated in stems during the dry season (436 g/kg CHO) when compared with the rainy season (519 g/kg CHO) (Figure 4). The indigestible carbohydrate fraction (C) in stems was affected by genotype (p < 0.0001) (Figure 5A) and season (p = 0.006) (Figure 5B).
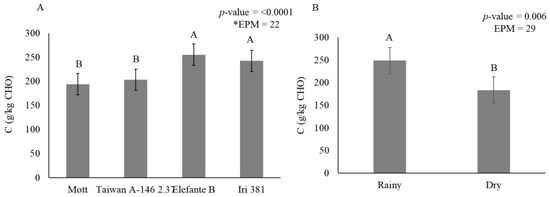
Figure 5.
Stem carbohydrate fraction C (g/kg carbohydrate) as affected by genotype (A) and season (B) for elephantgrass genotypes at Garanhuns, Pernambuco, Brazil. *EPM = Standard error of the means.
Mott (193 g/kg CHO) and Taiwan A-146 2.37 (203 g/kg CHO) contained a lower indigestible carbohydrate fraction than Elefante B (254 g/kg CHO) and Iri 381 (242 g/kg CHO) (Figure 5A). During the rainy season (248 g/kg CHO), there was an increase of 26% in the indigestible carbohydrate fraction compared to the dry season (183 g/kg CHO) (Figure 5B).
4. Discussion
4.1. Herbage, Leaf, and Stem Accumulation Rates and Nutritive Value
Irrigation contributed to soil-water deficit reduction during the dry season and possible water stress during rainy season due to rainfall distribution, increasing herbage accumulation rate of elephantgrass. However, when the availability of light and temperature to plant growth is reduced, the impact of irrigation can be limited [24]. This shows the influence of ambient climate on the nutritive value and morphological aspects of forage [25]. Our study suggests that when soil water availability is a limiting factor, irrigation has the potential to decrease seasonality of forage accumulation, reducing forage gap. However, during the rainy season, irrigation showed little effect on variables. This probably occurred due to favorable rainfall distribution during the rainy season, which reduced water stress.
Thus, using forage grasses with greater potential for forage accumulation, such as elephantgrass, can help to increase the efficiency of forage-based livestock systems. Additionally, during the rainy season the differences in HAR and LAR among dwarf and tall genotypes declined, indicating the potential for dwarf types with greater forage nutritive value to fill forage gaps, in line with the results found by [10] using the same genotypes in this study to feed sheep. They concluded that dwarf genotypes are recommended for cut-and-carry system, due to greater nutritive value.
Water contributes to cell elongation and plant growth [26]. Of the processes related to plant growth, cell formation (mainly cell expansion and differentiation) is the most sensitive to cell turgor because cells become turgid when water enters and cell size increases [27]. Genetic differences among genotypes contribute to differences in leaf and stem accumulation rate and affect forage nutritive value. In our study, water availability was the main factor in declining L:S ratio, due to advancing phenological phases and consequent plant maturity, that happened much faster under irrigation [28].
Similar reduction in nutritive value to our study, featured by CP and IVDDM in the dry season for the tall elephantgrass cv. ‘Roxo’ under irrigation, was reported by [3]. Reduction in CP and increase in NDF concentrations of tropical grasses in the rainy season under irrigation conditions were also reported by [29]. According to the authors, irrigation contributed to changes in grass growth, increasing SAR and reducing leaf:stem ratio. Irrigation contributed to a greater cell wall deposition, since turgor pressure increases the mechanical rigidity of cells and tissues of leaf and stems, increasing lignin concentration, which has a negative impact on IVDDM.
Water stress affects leaf and stem elongation due to increased stomatal closure and decrease in carbon dioxide and photosynthetic rate, contributing to disturbances in amino acid and carbohydrate metabolism. Thus, carbohydrates and protein metabolites such as proline and glycine accumulate in leaves and stems, in line with our finding of increased CP and soluble carbohydrates with rainfed conditions, favoring the growth and development of the ruminal microbiota [28]. Additionally, during rainy season or irrigation conditions, the high activity of meristematic regions drain most of the available assimilates, increasing stem tracheid maturation even in thin cell walls. This also contributes to secondary thickening and lignification [26], thereby influencing NDF, ADF, and lignin accumulation in elephantgrass genotypes.
According to [30], Taiwan A-146 2.37 has a spiky and erect arrangement of leaves, which probably requires more fiber to maintain, even for stems that show greater elongation capacity than Mott [9], likewise explained by the higher support requirement mentioned above. According to [31], Mott and Taiwan A-146 2.37 dwarf elephantgrasses exhibit a greater distance between the epidermis and vascular bundles, possessing a larger parenchymatic tissue area. This tissue is generally easily digested by ruminal microorganisms and probably contributed to the similar IVDDM for Mott and Taiwan A-146 2.37, despite Taiwan A-146 2.37 presenting the greater fiber concentration.
4.2. Protein and Carbohydrate Fractionation
Variations in CHO and CP contents were reported by [32]. The greater proportion of fraction A contributes to increased animal performance, because it is a source of readily available N important to carbohydrate fermentation and incorporation into carbon skeletons by microorganisms [33]. According to [34], grass development occurs at the expense of non-fiber carbohydrates, reducing potentially digestible nutrients and affecting forage nutritive value; it was observed in our experiment the greater that grass development under irrigated conditions was associated with increases in plant cell wall, which was probably a result of increase the thickness as the cell content (potentially digestible nutrients) decreases with the stimulation of plant growth [23,35,36], thereby reducing animal performance [33].
The carbohydrate A + B1 fraction is particularly important because it has rapid rates of rumen fermentation contributing to a greater IVDDM, because it provides the primary energy source for rumen microorganism multiplication [37].
Carbohydrate fraction C is represented by lignin, an indigestible plant fiber fraction [38]. Stems generally contain a greater proportion of vascular bundles with different proportions of lignin-rich sclerenchyma rings [39]. Resistant tissues present in the stems support plant structures (e.g., leaves and inflorescences), especially under greater water supply. Greater content of fraction C, however, contributes to reducing potentially degradable fiber fraction, positively affecting animal intake by the rumen filling effect [40]. In our study, presence of water contributed to faster plant growth and consequently increased C.
However, more research should be carried on indigestible fractions of carbohydrates, considering the advances in research and methodologies regarding the measurement of indigestible NDF. Lignin type and linkages of phenolic acids may explain a reasonable portion of the variation in indigestible NDF, but they may differ among varieties and stages of maturity, indicating that use of the same factors to explain digestibility in all forages across all agronomic conditions will likely lead to erroneous characterizations. This indicates that the laboratory detergent system and the chemical composition of the plant may not correctly describe the rate and extent of digestibility at all conditions since the linkages among fiber fractions explain only a portion of the digestion behavior [41].
Fiber accounts for most of the total carbohydrates contained in forage grasses [40], and elephantgrass is considered one of the best ruminant feed sources in warm climates [42]. Carbohydrate fractionation of various tropical grasses was evaluated by [34]. They reported greater forage accumulation rate during the rainy season, with a more rapid increase of stem fraction in the total forage mass, contributing to increases of NDF, ADF, carbohydrate fraction C, and lignin. An increase in fraction C is usually followed by the reduction in A + B1 fraction, contributing to reduced available energy for rumen microorganisms responsible for fiber carbohydrates fermentation, lowering ruminant performance.
5. Conclusions
Irrigation increases HAR, LAR, and SAR of elephantgrass genotypes, with the potential to minimize seasonal deficits in forage production. Taiwan A-146 2.37, one of the dwarf cultivars in our study, had similar leaf:stem ratios as tall genotypes, while dwarf type Mott, with a greater leaf:stem ratio, had the greatest forage nutritive value under irrigated and rainfed conditions. Most CHO and CP soluble fractions accumulated in leaves. Elefante B and Iri 381 had a greater proportion of C fraction, which contributed to a reduction in forage nutritive value.
Under irrigation, the advantage of the generally greater forage nutritive value of Mott dwarf elephantgrass was not limited, while Taiwan A-146 2.37 showed an intermediary nutritive value and did not differ from Mott and tall genotypes. The optimization of elephantgrass forage production potential through irrigation promoted the greatest growth rates, notably in tall genotypes. Adjustments in harvest frequency by physiological type must therefore be made in order to obtain a forage with consistent nutritive value for ruminants.
Author Contributions
R.T.d.A.S.: Investigation, Methodology, Data curation, Writing—Original draft; M.V.F.d.S.: Conceptualization, Validation, Resources, Project administration; M.V.d.C.: Methodology, Data curation, Validation, Supervision; G.D.G.: Writing—Review and Editing, Visualization, Supervision; V.J.d.S.: Writing—Review and Editing; A.C.L.d.M.: Validation, Resources, Writing—Review and Editing, Project administration; J.P.M.: Writing—Review and Editing; R.E.P.R.: Investigation, Methodology, Writing—Review; J.C.B.D.J.: Writing—Review and Editing, Supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by CAPES—Coordination for the Improvement of Higher Level Personnel—Brazil—Financing Code 001, FACEPE—Science and Technology Support Foundation of Pernambuco State, and CNPq—National Council of Scientific and Technological Development—Grants.
Institutional Review Board Statement
Ethics approval—The Ethics committee of Federal Rural University of Pernambuco approved the study (License n° 002/2020).
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pereira, A.V.; Lédo, F.J.S.; Machado, J.C. BRS Kurumi and BRS Capiaçu-New elephant grass cultivars for grazing and cut-and-carry system. Crop Breed. Appl. Biotechnol. 2017, 17, 59–62. [Google Scholar] [CrossRef]
- Viana, B.L.; Mello, A.C.L.; Lira, M.A.; Guim, A.; Dubeux Júnior, J.C.B.; Santos, M.V.F.; Cunha, M.V. Adaptability and stability analysis for selection of elephantgrass clones under grazing. Crop Sci. 2015, 55, 250–957. [Google Scholar] [CrossRef]
- Carvalho, A.P.S.; Arruda, R.M.; Abreu, J.G.; Souza, A.L.; Rodrigues, R.C.; Lima, L.R.; Cabral, L.S.; Neto, A.B. Agronomic features of elephant grass (Pennisetum purpureum Schum) cv. Roxo under irrigation. Semin. Ciênc. Agrár. 2018, 39, 275–286. [Google Scholar] [CrossRef]
- Silva, P.H.F.; Sales, T.B.; Lemos, M.F.; Silva, M.C.; Ribeiro, R.E.P.; Santos, M.V.F.; Mello, A.C.L.; Cunha, M.V. Tall and short-sized elephant grass genotypes: Morphological aspects cut-and-carry, and grazing management. Cienc. Rural 2021, 51, e20200848. [Google Scholar] [CrossRef]
- Sollenberger, L.E.; Prine, G.M.; Ocumpaugh, W.R.; Hanna, W.W.; Jones, C.S., Jr.; Schank, S.C.; Kalmbacher, R.S. Registration of ‘Mott’ dwarf elephantgrass. Crop Sci. 1989, 29, 827–828. [Google Scholar] [CrossRef]
- Turano, B.; Tiwari, U.P.; Jha, R. Growth and nutritional evaluation of napier grass hybrids as forage for ruminants. Trop. Grassl. Forrajes Trop. 2016, 4, 168–178. [Google Scholar] [CrossRef]
- Cunha, M.V.; Lira, M.A.; Santos, M.V.F.; Dubeux, J.C.B., Jr.; Mello, A.C.L.; Freitas, E.V. Adaptability and stability of the forage yield by different methods in the selection of Pennisetum spp. clones. Rev. Bras. Cienc. Agrar. 2013, 8, 681–686. [Google Scholar] [CrossRef][Green Version]
- Garcia, C.S.; Fernandes, A.M.; Fontes, C.A.A.; Vieira, R.A.M.; Sant’ana, N.F.; Pimentel, V.A. Performance of steers grazed on elephant grass and Mombaça grass pastures. Rev. Bras. Zootech. 2011, 40, 403–410. [Google Scholar] [CrossRef]
- Cunha, M.V.; Lira, M.A.; Santos, M.V.F.; Freitas, E.V.; Dubeux, J.C.B., Jr.; Mello, A.C.L.; Martins, K.G.R.M. Association between the morphological and productive characteristics in the selection of elephant grass clones. Rev. Bras. Zootech. 2011, 40, 482–488. [Google Scholar] [CrossRef][Green Version]
- Silva, J.K.B.; Cunha, M.V.; Santos, M.V.F.; Magalhães, A.L.R.; Mello, A.C.L.; Silva, J.R.C.; Souza, C.I.R.; Carvalho, A.L.; Souza, E.J.O. Dwarf versus tall elephant grass in sheep fed: Which one is the most recommendes for cut-and-carry? Trop. Anim. Health Prod. 2021, 53, 93. [Google Scholar] [CrossRef] [PubMed]
- Zailan, M.Z.; Yaakub, H.; Jusoh, S. Yield and nutritive quality of napier (Pennisetum purpureum) cultivars as fresh and ensiled fodder. J. Anim. Plant. Sci. 2018, 28, 63–72. [Google Scholar]
- Monção, F.P.; Costa, M.A.M.S.; Rigueira, J.P.S.; de Sales, E.C.J.; Leal, D.B.; da Silva, M.F.P.; Gomes, V.M.; Chamone, J.M.A.; Alves, D.D.; Carvalho, C.D.C.S.; et al. Productivity and nutritional value of BRS capiaçu grass (Pennisetum purpureum) managed at four regrowth ages in a semiarid region. Trop. Anim. Health Prod. 2020, 52, 235–241. [Google Scholar] [CrossRef]
- Voltolini, T.V.; Silva, J.G.; Silva, W.E.L.; Nascimento, J.M.L.; Queiroz, M.A.A.; Oliveira, A.R. Nutritive value of cultivars of cane sugar under irrigation. Rev. Bras. Saúde Prod. Anim. 2012, 13, 894–901. [Google Scholar] [CrossRef]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.L.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z. 2013, 22, 711–728. [Google Scholar] [CrossRef]
- EMBRAPA. Manual De Métodos De Análise De Solo, 2nd ed.; Centro Nacional de Pesquisa de Solos, Embrapa: Rio de Janeiro, Brazil, 1997; 212p. [Google Scholar]
- Cavalcanti, F.J.A. Recomendações De Adubação Para O Estado De Pernambuco: Segunda Aproximação; Instituto Agronômico de Pernambuco, IPA: Recife, Brazil, 2008; 212p. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration. In FAO Irrigation and Drainage Paper No. 56; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998; Volume 56, p. 300. [Google Scholar]
- AOAC. Official Methods of Analysis, 15th ed.; Association of Analytical Chemists: Washington, DC, USA, 2000. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and non-starch polysaccharides in relation to animal nutrition. Symposium: Carbohydrate methodology, metabolism, and nutritional implications in dairy cattle. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Senger, C.C.D.; Kozloski, G.V.; Sanchez, L.M.B.; Mesquita, F.R.; Alves, T.P.; Castagnino, D.S. Evaluation of autoclave procedures for fibre analysis in forage and concentrate feedstuffs. Anim. Feed Sci. Technol. 2008, 146, 169–174. [Google Scholar] [CrossRef]
- Tilley, J.M.A.; Terry, R.A. A two-stage technique for the in vitro digestion of forage crops. Grass For. Sci. 1963, 18, 104–111. [Google Scholar] [CrossRef]
- Licitra, G.; Hernandez, T.M.; Van Soest, P.J. Standardization of procedures for nitrogen fractionation of ruminant feeds. Anim. Feed Sci. Technol. 1996, 57, 347–358. [Google Scholar] [CrossRef]
- Sniffen, C.J.; O’Connor, J.D.; Van Soest, P.J.; Fox, D.G.; Russell, J.B. A net carbohydrate and protein system for evaluating cattle diets: II. Carbohydrate and protein availability. J. Anim. Sci. 1992, 70, 3562–3577. [Google Scholar] [CrossRef]
- Habermann, E.; Oliveira, E.A.D.; Contin, D.R.; Delvecchio, G.; Viciedo, D.O.; Moraes, M.A.; Orado, R.M.; Costa, K.A.P.; Braga, M.R.; Martinez, C.A. Warming and water deficit impact leaf photosynthesis and decrease forage quality and digestibility of a C4 tropical grass. Physiol. Plant. 2019, 165, 383–402. [Google Scholar] [CrossRef]
- Moyo, M.; Nsahlai, I. Consequences of increases in ambiente temperature and effect of climate type on digestibility of forage by ruminants: A meta-analysis in relation to global warming. Animals 2021, 11, 172. [Google Scholar] [CrossRef]
- Cosgrove, D.J. Plant cell wall extensibility: Connecting plant cell growth with cell wall structure, mechanics, and the action of wall-modifying enzymes. J. Exp. Bot. 2016, 67, 463–476. [Google Scholar] [CrossRef]
- Körner, C. Paradigm shift in plant growth control. Curr. Opin. Plant Biol. 2015, 25, 107–114. [Google Scholar] [CrossRef]
- Islam, M.R.; Garcia, S.C.; Horadagoda, A. Effects of irrigation and rates and timing of nitrogen fertilizer on dry matter yield, proportions of plant fractions of maize and nutritive value and in vitro gas production characteristics of whole crop maize silage. Anim. Feed Sci. Technol. 2012, 172, 125–135. [Google Scholar] [CrossRef]
- Pequeno, D.N.L.; Pedreira, C.G.S.; Sollenberger, L.E.; Faria, A.F.G.; Silva, L.S. Forage accumulation and nutritive value of Brachiariagrasses and Tifton 85 Bermudagrass as affected by harvest frequency and irrigation. Agron. J. 2015, 107, 1741–1749. [Google Scholar] [CrossRef]
- Silva, S.H.B.; Santos, M.V.F.; Lira, M.A.; Dubeux, J.C.B., Jr.; Freitas, E.V.; Ferreira, R.L.C. Use of morphological descriptors and character heritability in small-size elephant grass clones. Rev. Bras. Zootech. 2009, 38, 1451–1459. [Google Scholar] [CrossRef]
- Peixoto, T.V.F.R. Nutritive Value and Anatomical Aspects of Pennisetum purpureum Schum Clones of Different Size. Master’s Thesis, Universidade Federal Rural de Pernambuco, Recife, Brazil, 2018. [Google Scholar]
- Berça, A.S.; Cardoso, A.S.; Longhini, V.Z.; Tedeschi, L.O.; Boddey, R.M.; Reis, R.A.; Ruggieri, A.C. Protein and Carbohydrate Fractions in Warm-Season Pastures: Effects of Nitrogen Management Strategies. Agronomy 2021, 11, 847. [Google Scholar] [CrossRef]
- Brandstetter, E.V.; Costa, K.A.P.; Silva, M.A.P.; Araújo Neto, F.R.; Silva, V.R.; Neves, R.B.S.; Souza, W.F.; Oliveira, I.P. Production of Jiggs bermudagrass and the impact of quality milk production and quality of Holstein dairy cows under an intermittent grazing system. Can. J. Anim. Sci. 2018, 98, 279–288. [Google Scholar] [CrossRef]
- Velásquez, P.A.T.; Berchielli, T.T.; Reis, R.A.; Rivera, A.R.; Dian, P.H.M.; Teixeira, I.A.M.A. Chemical composition, carbohydrate and protein fractionation and in vitro digestibility of tropical forages at different cutting ages. Rev. Bras. Zootech. 2010, 39, 1206–1213. [Google Scholar] [CrossRef]
- Gallardo, I.; Bárcena, R.; Pinos-Rodríguez, J.M.; Ortega, M.E. Influence of exogenous fibrolytic enzymes on in vitro and in sacco degradation of forages for ruminants. Ital. J. Anim. Sci. 2010, 9, 35–38. [Google Scholar] [CrossRef]
- Bureenok, S.; Langsoumechai, S.; Pitiwittayakul, N.; Yuangklang, C.; Vasupen, K.; Saenmahayak, B.; Schonewille, J.T. Effects of fibrolytic enzymes and lactic acid bacteria on fermentation quality and in vitro digestibility of Napier grass silage. Ital. J. Anim. Sci. 2019, 18, 1438–1444. [Google Scholar] [CrossRef]
- Singh, S.; Kushwaha, B.P.; Nag, S.K.; Mishra, A.K.; Singh, A.; Anele, U.Y. In vitro ruminal fermentation, protein and carbohydrate fractionation, methane production and prediction of twelve commonly used Indian green forages. Anim. Feed Sci. Technol. 2012, 178, 2–11. [Google Scholar] [CrossRef]
- Li, Q.; Xie, S.; Serem, W.K.; Naik, M.T.; Liu, L.; Yuan, J.S. Quality carbon fibers from fractionated lignin. Green Chem. 2017, 19, 1628–1634. [Google Scholar] [CrossRef]
- Ferreira, G.D.G.; Cunha, M.V.; Silva, D.K.A.; Santos, M.V.F.; Magalhães, A.L.R.; Lira, M.A.; Dubeux, J.C.B., Jr. Stem histology of elephant grass clones in Agreste of Pernambuco. Rev. Bras. Saúde Prod. Anim. 2010, 11, 36–47. [Google Scholar]
- Brandstetter, E.V.; Costa, K.A.D.P.; Santos, D.D.C.; Souza, W.F.D.; Silva, V.C.; Dias, M.B.D.C. Protein and carbohydrate fractionation of Jiggs Bermudagrass in different seasons and under intermittent grazing by Holstein cows. Acta Sci. Anim. Sci. 2019, 41, e43363. [Google Scholar] [CrossRef]
- Raffrenato, E.; Fievisohn, R.; Cotanch, K.W.; Grant, R.J.; Chase, L.E.; Van Amburgh, M.E. Effect of lignin linkages with other plant cell wall componentes on in vitro and in vivo neutral detergent fiber digestibility and rate of digestion of grass forages. J. Dairy Sci. 2017, 100, 8119–8131. [Google Scholar] [CrossRef]
- Das, L.K.; Kundu, S.S.; Kumar, D.; Datt, C. Fractionation of carbohydrate and protein content of some forage feeds of ruminants for nutritive evaluation. Vet. World 2015, 8, 197–202. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).