1. Introduction
The average surface temperature has been increasing by 0.19 °C per decade [
1]. Consequently, the frequency and duration of extreme weather events, especially heatwaves and extreme precipitation, have significantly increased [
2]. As the ruminal fermentation process produces excessive heat, the mammary gland conducts high anabolic activities, and cows have a low surface-area-to-mass ratio, making lactating cows more vulnerable to heat stress [
3,
4]. It has been reported that, when the temperature–humidity index (THI) reached 72 (recent research indicated this value can be as low as 68 or 70), side effects of heat stress on dairy cows notably appeared [
5,
6]. The detrimental impacts of heat stress on dairy cows mainly include reduced feed intake, milk production metrics, milk protein metrics, and the following conception rate [
7], as well as an increased risk of ruminal acidosis and enteric methane (CH4) production [
8]. Therefore, heat stress is an obvious restrictive factor in dairy cow production and will worsen with the continuously increasing global temperature. Thus, determining an effective cooling method to attenuate heat stress in cows is of the utmost importance.
Re-establishing the physiological thermal energy balance between heat acquisition (e.g., maintenance, exercise, growth, lactation, gestation, and feed intake) and dissipation in the environment is critical for attenuating heat stress [
9,
10]. During the production process, several cooling options, such as shading and air fans, can be used to lower temperatures and restore the normal physiology of cows [
11]. Furthermore, nutritional strategies, such as supplementing appropriate energy ingredients, minerals, vitamins, antioxidants, prebiotics, and probiotics, have also been applied to ameliorate heat stress [
12]. Compared with nutritional regulations, directly cooling the environment (e.g., cooling with sprinklers) appears to be the most economical and efficient method for relieving heat stress [
13]. Cooling with sprinklers has been proven to be successful in reducing shell and body temperatures, increasing heat evaporation to a lower respiration rate, enhancing feed intake, and promoting digestibility [
14], which contributed to increased milk and milk protein production [
15,
16,
17,
18]. However, the mechanisms underlying these positive effects in attenuating heat stress have not been elucidated.
Ruminal microbiota is of vital importance for animal health and production performance, as microorganisms can ferment feed ingredients to supply critical metabolites for the host [
19,
20,
21,
22,
23,
24,
25]. For example, ruminal microbial communities can produce nutritional fermentation metabolites, like vitamins and functional fatty acids [
19,
26,
27,
28,
29]. These metabolites can be adsorbed through rumen epithelial cells and then transported through the blood to the target tissues to regulate tissue functions. Recent research has documented that rumen microbial communities are directly regulated by heat stress [
30,
31,
32,
33]. For instance, Zhao et al. (2019) found that heat stress led to ruminal bacterial composition alteration and functional deterioration, increased lactate, and reduced acetate-producing bacterial relative abundance [
30]. Other studies have disclosed that ruminal fiber-degrading bacteria populations (e.g.,
Fibrobacter) decreased, while starch-degrading bacteria populations (e.g.,
Clostridium and
Streptococcus) increased [
34,
35] in response to heat stress. Another study found that live yeast supplementation positively ameliorated heat stress in dairy cows through regulating the microbiota composition and rumen fermentation in the rumen and hindgut [
36]. These studies demonstrate that ruminal microbiota response may be an underlying mechanism of the positive effects on heat stress in ruminal animals.
Therefore, the present study evaluated the effect of an automatic sprinkler on the production performances of cows and elucidated the potential mechanism through considering the ruminal microbiota.
2. Materials and Methods
2.1. Experiment Animals and Management
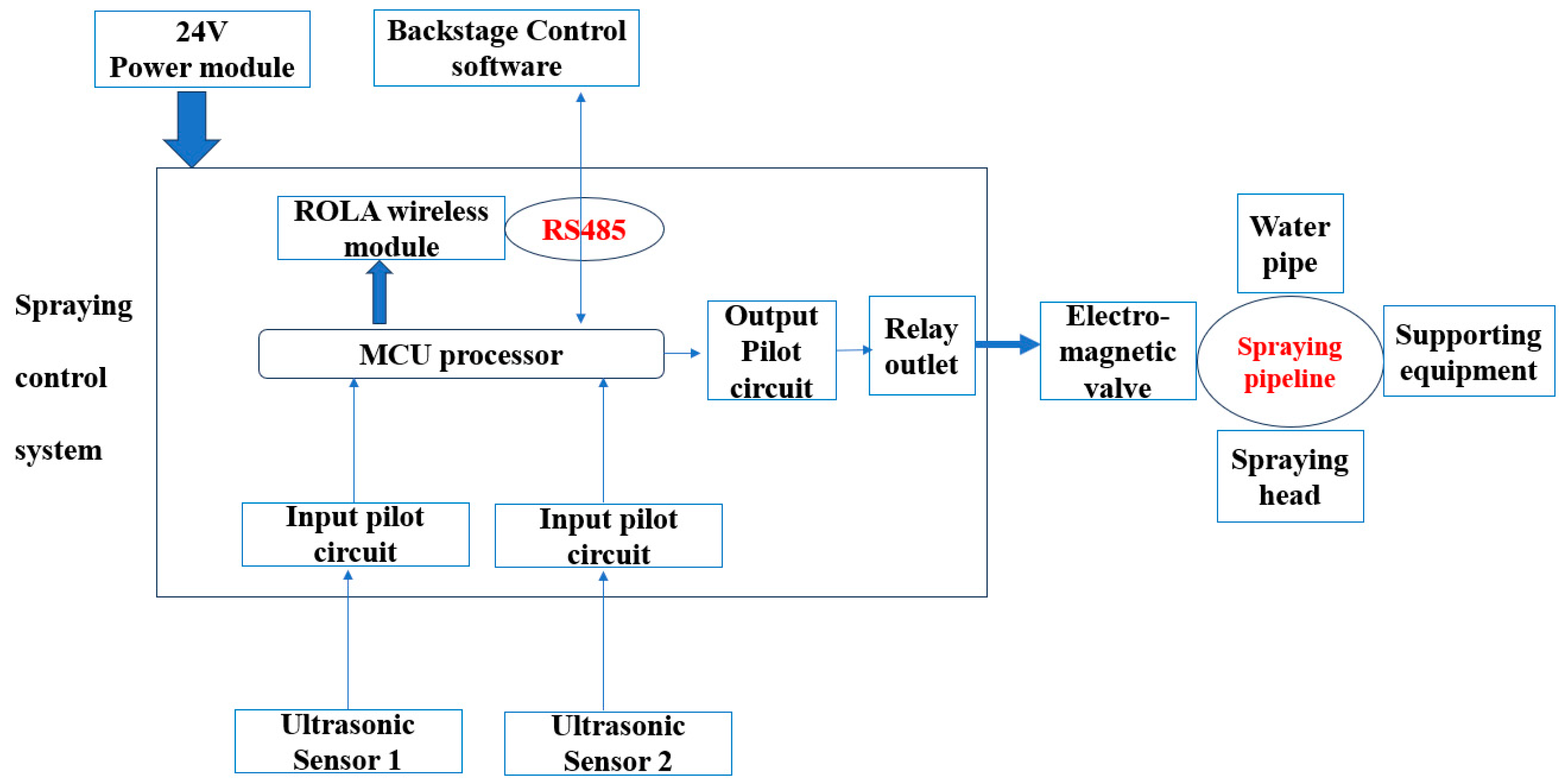
The experiment was conducted in the Bengbu dairy farm, Modern Farming (Wuhe) Co. Ltd., Anhui Province, China (32.92 N, 117.38 E), from 10 June 2023, to 10 August 2023. A total of 1208 multiparous Chinese Holstein dairy cows with an average live weight of 683.6 ± 27.3 kg, lactation of 199.3 ± 16.8 d, and lactating parities of 2.88 ± 0.49 was used and randomly allocated to 6 barns. Three barns were equipped with automatic sprinklers (SPs), and each barn was considered a replicate. The other three barns followed the routine feeding procedure (CON) without sprinklers. The schematic diagram of the sprinkler is shown in
Figure 1.
All cows were reared in a 312 m long × 96 m wide shed to ensure the same feeding environment. Diets were formulated, according to NRC (2001) [
37], to meet the energy requirements of Holstein dairy cows, yielding 30 kg of milk/day with 3.5% milk fat and 3.0% true protein. The nutrient level and ingredient composition of the employed diet are shown in
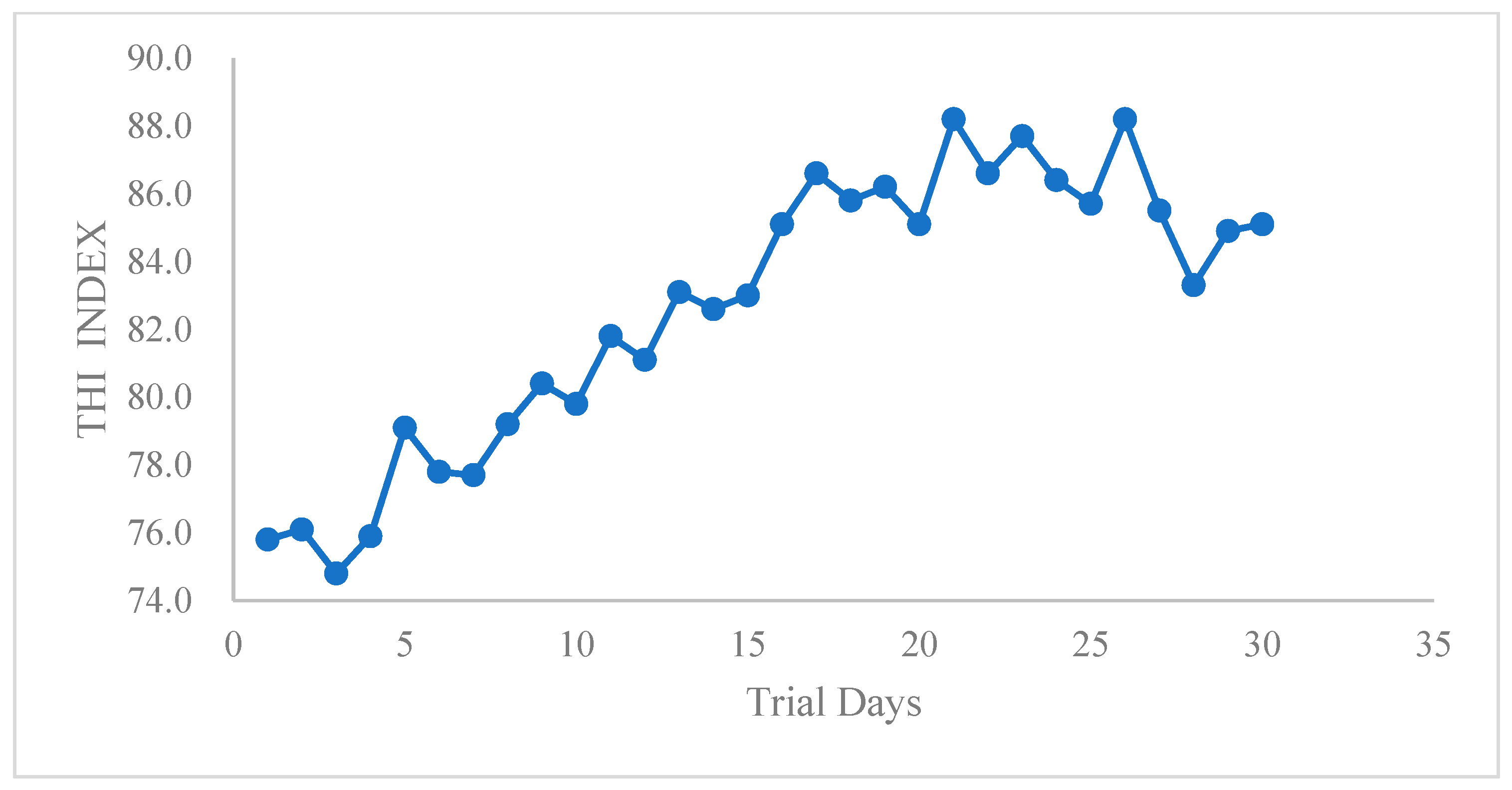
Table 1. Cows were fed three times per day, at 06:00, 13:00, and 21:00. During the experimental period, all cows had free access to food and water. Temperature and humidity were recorded every day, and THIs were calculated using the following equation, as previously reported in [
38]: THI = (1.8 × T + 32) − [(0.55 − 0.0055 × RH) × (1.8 × T −26)], where T = temperature and RH = relative humidity. When the THI ≥ 70, heat stress occurred in high-yielding dairy cows [
6]. Cows were milked three times per day (08:00, 14:00, and 20:00 h) [
39].
2.2. Feed Intake and Composition Analysis
The average daily intake in barns was determined based on the dry matter intake (DMI), which was calculated as the difference between the feed offered and the residues on the dry matter basis. Feed samples were collected from each feeding time and mixed for analysis. Air-dried samples were obtained from the fresh feed and dried using a forced-air oven (GZX-9246MBE, Shanghai Boxunshiye Co., Ltd., Shanghai, China) at 65 °C for 48 h. Then, the absolute dried feed samples were obtained from the air-dried samples and dried further at 105 °C for 3 h using the forced-air oven.
The net energy (NE) level of the feed was calculated using the methodology in [
40]. The feed compositions were determined according to the AOAC (2007) method. A Kjeldahl nitrogen analyzer (SKD-1800, Shanghai Peiou Analytical Instrument Co., Ltd., Shanghai, China) was used to determine the crude protein (CP) level. The ether extract (EE) was determined using the Soxhlet extractor and calculated with the following formula:
where m1 = the mass of the sample in grams (g); m2 = the mass of the flask with emery in grams (g); m3 = the mass of the flask containing emery and the obtained dry residue of petroleum ether extraction in grams (g); and f = the unit of the correction factor in grams per kilogram (g/kg) (f = 1000 g/kg).
A semi-automatic fiber analyzer (A200i, ANKOM, Macedon, NY, USA) was used to determine the neutral detergent fiber (NDF) and acid detergent fiber (ADF) levels. Calcium (Ca) and phosphorus (P) levels were determined using the near-infrared spectroscopy (NIRS) method (NIRS DS2500 analyzer, FOSS Co., Ltd., DK-3400 Hilleroed, Denmark).
2.3. Milk Production and Composition
Daily milk yield was automatically recorded through the rotary milking facilities (9JRP-50P2100, Delaval, Israel). Milk samples were collected from each treatment during the last three consecutive days and stored in 100 mL vials with 2-bromo-2-nitropropan-1,3-diol at 4 °C for subsequent analysis.
Milk protein and fat were measured using a near-infrared analyzer (MilkoScanTM 7 RM, Foss Electric, Denmark). The somatic cell count (SCC) was measured using an SCC rapid analyzer (Fossomatic 7/7 DC, FOSS Co., Ltd., DK-3400 Hilleroed, Denmark).
2.4. Body Temperature Measurement
The body temperature of 30 cows in each treatment was measured once a week via the rectal thermometry method using a thermometer (VT 1831; Microlife AG Espenstrasse 139, CH-9443 Widnau, Switzerland).
2.5. Rumen Content Collection and Fermentation Parameter Analysis
Three cows from each barn with similar body weights (693.6 ± 12.3 kg), for a total of nine cows, were selected from the CON and SP groups for sample collection during the second middle lactation period. Three hours after morning feeding, 100 mL of rumen contents from 18 dairy cows was collected using an esophageal tube on the last day of the experiment [
41]. The first 200 mL contents was discarded to avoid potential saliva contamination. All rumen samples were divided into two parts. One part was analyzed for pH, volatile fatty acid (VFA), and ammonia-N (NH
3-N). The pH value of each rumen fluid sample was measured immediately using a portable pH meter (Testo 205, Testo AG, Lenzkirch, Germany). Individual and total VFAs (TVFAs) in the aliquots of ruminal fluid were determined using a gas chromatograph (GC-2010, Shimadzu, Kyoto, Japan). NH3-N concentration was determined using the indophenol method, and the absorbance value was measured using a UV-2600 ultraviolet spectrophotometer (Tianmei Ltd., Chaoyang, Beijing, China) [
42]. The other part was rapidly frozen with liquid nitrogen and then stored at −80 °C for further analysis.
2.6. Rumen Microbial Communities Measurement
Rumen microbial DNA was extracted from approximately 1.0 mL of rumen content using the MagBind® Soil DNA Kit (M5636, Omega, Norcross, GA, USA). DNA concentration, purity, and quality were assessed using a spectrophotometer and agarose gel electrophoresis. The V4 and V3 regions of the 16S rRNA gene were amplified using universal primers (F: ACTCCTACGGGAGGCAGCAG and R: GGACTACHVGGGTWTCTAAT). The PCR product mixture was purified with a Qiagen Gel Extraction Kit (Qiagen, Hilden, Germany). Sequencing was conducted on an Illumina MiSeq PE300 platform /NovaSeq PE250 platform (Illumina, San Diego, CA, USA) in a commercial laboratory. The quality filtering of raw tags was performed under standard filtering conditions to obtain high-quality clean tags, according to the Quantitative Insights into Microbial Ecology (QIIME, V1.7.0, San Diego, CA, USA, V1.7.0) quality control process. Sequences within similarity >97% were assigned to the same operational taxonomic unit (OTU). For each representative sequence, the SILVA classifier algorithm was used to annotate the taxonomic information from the GreenGene Database. Then, the species abundances and α- and β-diversity indices were analyzed at different taxonomic levels.
2.7. Statistical Analysis
A normal distribution test was first conducted on the production performances, milk quality, rumen fermentable parameters, and relative abundances of rumen microbial communities using the SAS (Statistics Analysis System, version 9.2, SAS Institute Inc., Cary, NC, USA) procedure, denoted as “proc univariate data=test normal”. Data were presented as mean ± SE. Further differential analysis on the above-mentioned parameters was performed using an unpaired two-tailed Student’s T-test. p-value < 0.05 was significant, and 0.05 ≤ p ≤ 0.10 indicated a trend.
4. Discussion
Heat stress detrimentally influences dairy production. When heat stress occurs, dairy cows present reduced feed intake, dairy production, and milk quality; increased evaporated water loss; and metabolic disturbance. During the experiments, the THI remained over 72, indicating that cows were all heat stressed. Under heat stress conditions, high environmental temperatures improve body temperatures, which extends the feed duration in rumen and, in turn, activates the rumen sensor as the stomach expands [
43]. This alteration impacted the hypothalamic anorexia center, resulting in a lowered appetite and feed intake [
44]. As a result, DMI and energy intake also reduced. The automatic spray cooling method reduced cow body temperatures and SCC; increased DMI, milk yield and fat, and ICR; and enhanced rumen fermentation functions, effectively alleviating heat stress in cows.
Rumen microbial communities are critical to dairy health and production performances. One important function of rumen microorganisms is to provide the host with energy and functional metabolites through the metabolism of nutrient residues. Carbohydrates are usually converted to pyruvate and acetyl-CoA in the rumen via microorganisms through the glycolytic and pentose phosphate pathways, and finally metabolized to VFAs—especially acetate and propionate—to provide energy for production. In our present study, carbohydrate-degrading bacteria, such as
Pseudobutyrivibrio, Bifidobacterium, and
Succinivibrio, significantly increased after SP treatment, which may further indicate a higher energy provision for milk production and more physiological activities. It has been reported that approximately 70% to 80% of the energy absorbed by cows is provided via rumen fermentation processes [
19]. Acetate was further transported into the mammary gland and synthesized into milk fat, while butyrate was utilized in the intestinal tract for epithelial development. In the present study, VFAs, including acetate, propionate, and butyrate, significantly increased in rumen fluids under SP treatment. TVFAs, especially the VFA composition and proportions, reflect energy balance and utilization in cows and act as effective indicators for rumen fermentation capacity [
45]. In particular, acetate undergoes lipid synthesis in adipose and the mammary tissues of ruminants with a primary carbon source [
46], which partially accounts for the increased milk fat level. In addition, heat stress triggered Toll-like receptor pathways and caused inflammatory responses. Deteriorated inflammation will cause mastitis and lead to increased milk SCC [
47]. Butyrate serves as critical immune regulator, helpful in inhibiting mastitis [
48], which may explain the reduced SCC in milk when the butyrate content increased. It is worth noting that, although rumen fermentation was enhanced via automatic spraying, the rumen acetate-to-propionate ratio did not differ between the SP and CON groups, suggesting that the rumen fermentation pattern remained unchanged. Therefore, further investigation is needed.
Ruminal homeostasis provided an ideal environment for nutrient digestion and transportation, energy generation, and microbial proliferation [
49,
50]. The rumen microbial composition and function significantly altered under heat-stressed environments, potentially disrupting primary homeostasis and further causing reduced feed intake and milk production. Rumen microbial richness and composition were modulated via automatic spraying, indicating the rumen microbial may contribute to the positive effects of automatic spraying under heat stress conditions. Zhao et al. (2019) and Uyeno et al. (2010) reported that the relative abundances of
Spirochaeta,
Streptococcus, and
Ruminobacter increased and acetic acid decreased in the heat stress condition [
30,
51]. In agreement with these data, we found that the relevant abundance of
Streptococcus decreased and the rumen acetic acid level increased. Under ruminal conditions, acetate was transported through the rumen epithelium and then utilized by mammary gland cells as the key substrate for milk fat synthesis. Therefore, milk fat content increased after SP treatment. In addition, some functional microorganism abundances were modulated via the automatic spraying treatment. For example, the relative abundances of
Prevotella and
Pseudobutyrivibrio increased, where
Prevotella is important in starch and
Pseudobutyrivibrio is a stressor-related genera. Indeed, these changes reduced fiber degradation and enhanced starch degradation [
34,
35], which ensured the abundant energy supply for body production and thus enhanced yield production.