The Impacts and Economic Analysis of Jack Mackerel Meal Inclusion in Low Fish Meal Diets on the Growth and Feed Availability of Juvenile Rockfish (Sebastes schlegeli)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Rearing Conditions of the Feeding Trial
2.2. Experimental Design of the Feeding Trial
| Experimental Diets | |||||||
|---|---|---|---|---|---|---|---|
| Con | CBM25J | CBM50J | MM25J | MM50J | TBM25J | TBM50J | |
| Ingredients (%, DM basis) | |||||||
| Fish meal (FM) a | 55.0 | 19.3 | 5.5 | 19.3 | 5.5 | 19.3 | 5.5 |
| Jack mackerel meal (JMM) b | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 | |
| Chicken by-product meal (CBM) c | 16.5 | 33.0 | |||||
| Meat meal (MM) d | 11.8 | 23.6 | |||||
| Tuna by-product meal (TBM) e | 17.1 | 34.2 | |||||
| Fermented soybean meal f | 12.0 | 12.0 | 12.0 | 12.0 | 12.0 | 12.0 | 12.0 |
| Wheat flour | 21.5 | 19.3 | 16.6 | 24.4 | 27 | 19.8 | 17.6 |
| Fish oil | 4.5 | 4.5 | 4.5 | 4.5 | 4.5 | 4.5 | 4.5 |
| Soybean oil | 4.5 | 3.9 | 3.9 | 3.5 | 2.9 | 2.8 | 1.7 |
| Vitamin premix g | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Mineral premix h | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Choline | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Nutrients (%, DM basis) | |||||||
| Dry matter | 95.0 | 93.5 | 91.8 | 94.2 | 91.5 | 93.2 | 93.9 |
| Crude protein | 51.7 | 51.7 | 51.8 | 51.6 | 51.3 | 51.4 | 51.1 |
| Crude lipid | 14.3 | 14.6 | 14.5 | 14.4 | 14.3 | 14.5 | 14.5 |
| Ash | 10.4 | 11.1 | 11.6 | 9.4 | 7.7 | 11.9 | 12.8 |
| Carbohydrates i | 23.6 | 22.6 | 22.1 | 24.6 | 26.7 | 22.2 | 21.6 |
| Gross energy (kcal/g) j | 4.3 | 4.3 | 4.3 | 4.3 | 4.4 | 4.2 | 4.2 |
2.3. Measurement of the Biological Indices of Rockfish
2.4. Biochemical Composition Analysis of the Experimental Diets and Whole-Body Rockfish
2.5. Plasma and Serum Analysis of Rockfish
2.6. Economic Analysis of the Study
2.7. Statistical Analysis
3. Results
3.1. Amino Acid and Fatty Acid Profiles of the Experimental Diets
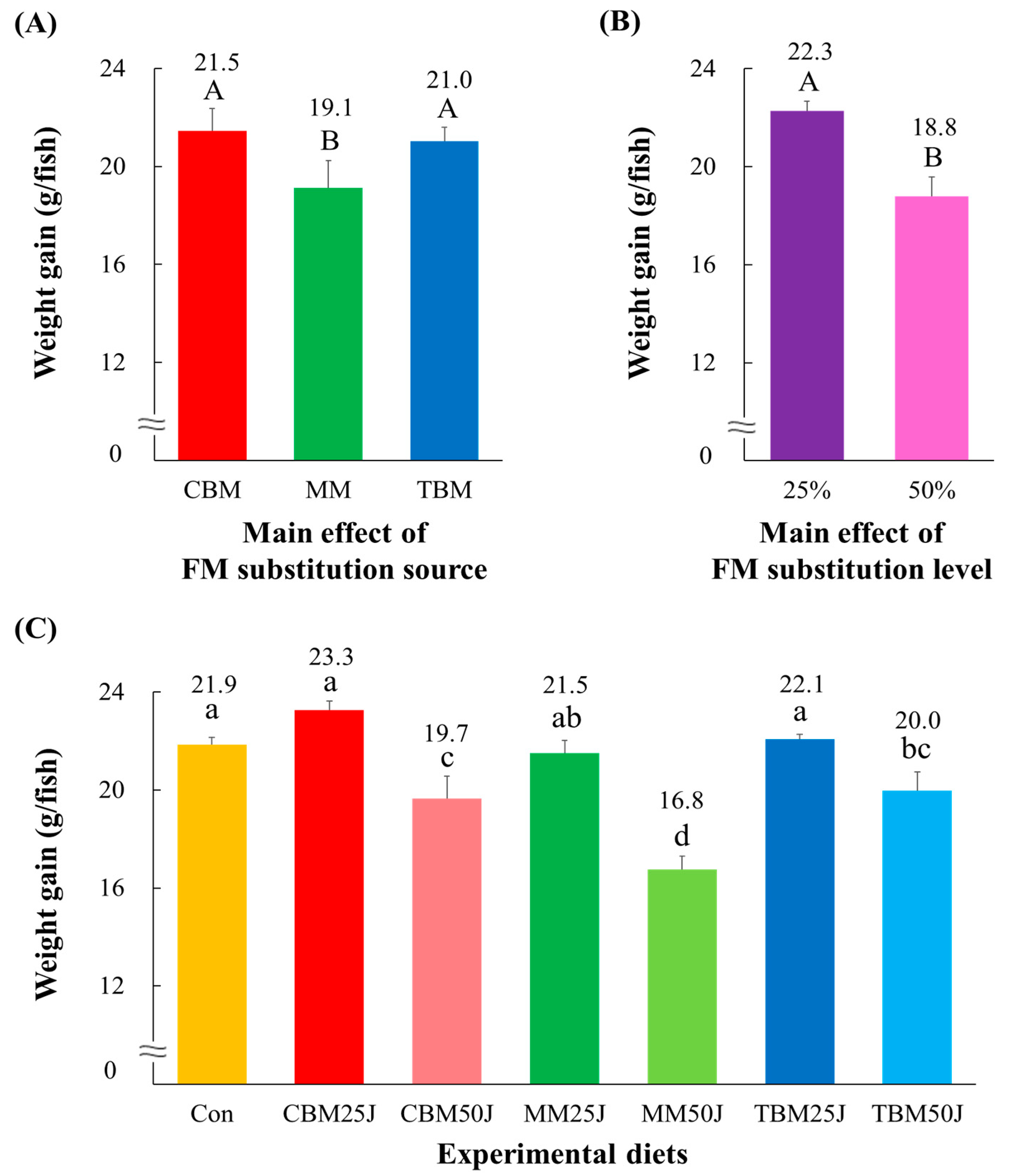
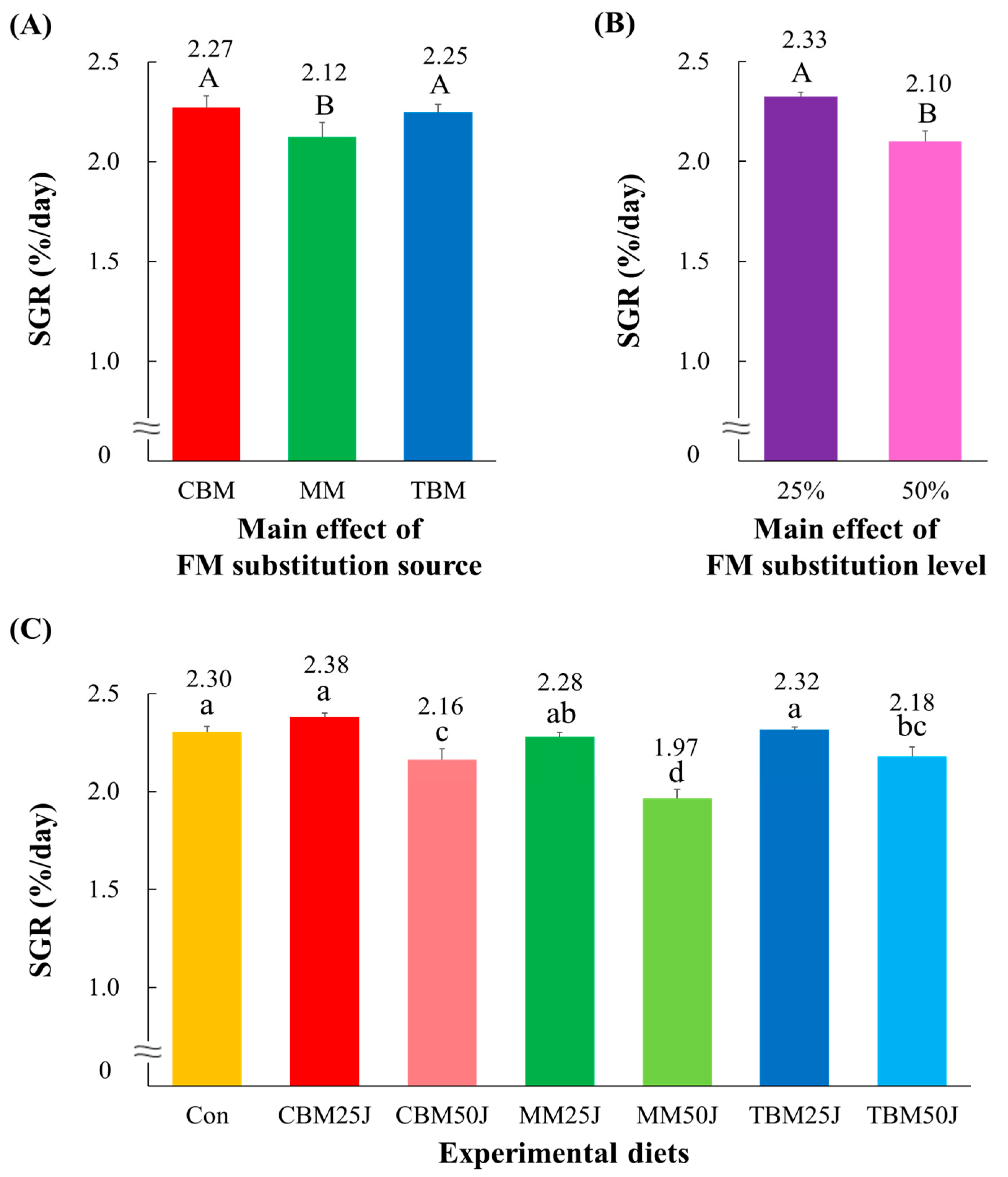
3.2. Survival and Growth Performance of Rockfish
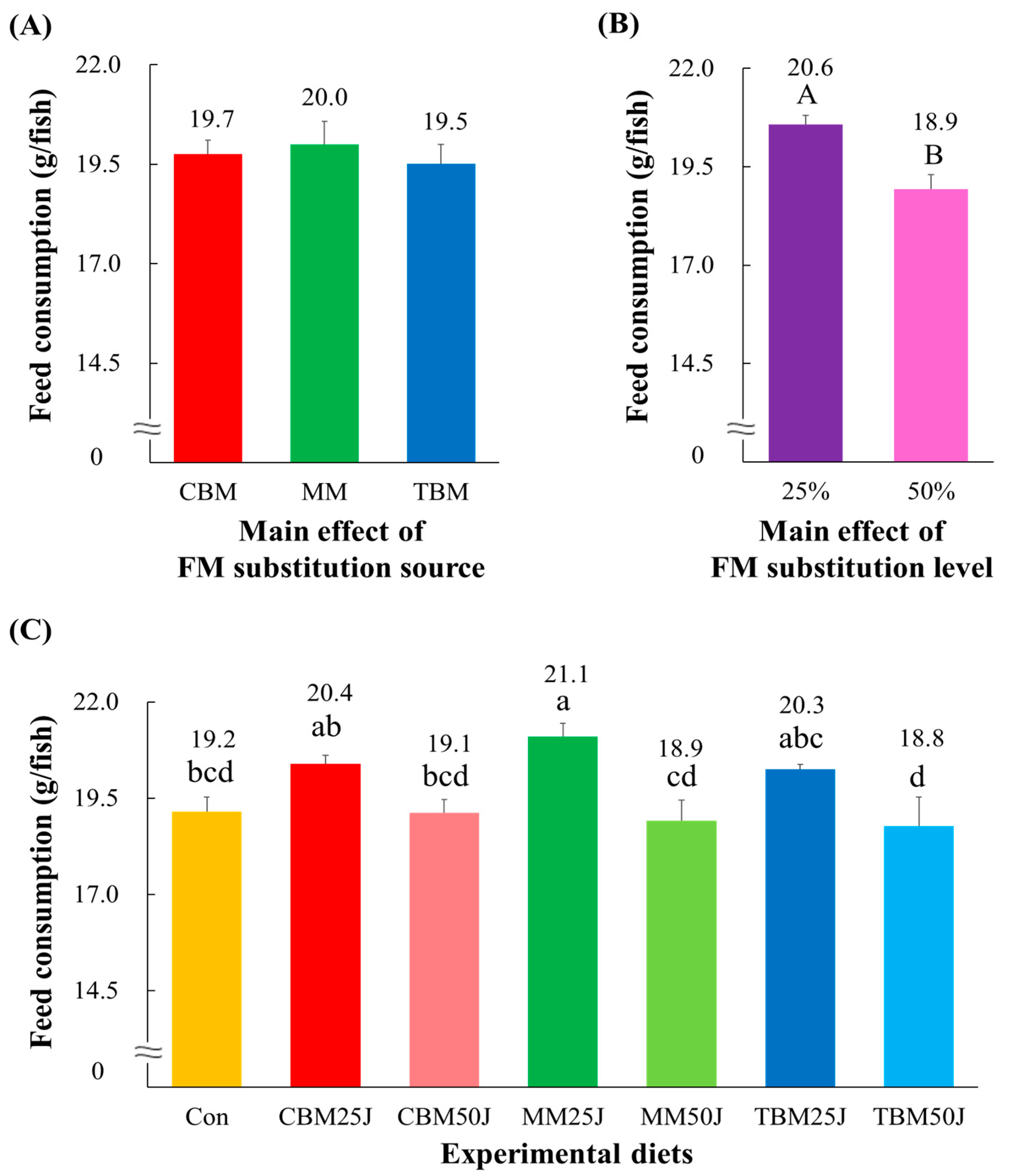
3.3. Feed Availability and Biological Indices of Rockfish
3.4. Biochemical Composition of the Whole Body of Rockfish
3.5. Plasma and Serum Parameters of Rockfish
3.6. Results of Economic Analysis of the Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- KOSIS. Korean Statistical Information Service. 2024. Available online: http://kosis.kr/statisticsList/statisticsListIndex.do/ (accessed on 20 May 2024).
- Glencross, B.D.; Booth, M.; Allan, G.L. A feed is only as good as its ingredients—A review of ingredient evaluation strategies for aquaculture feeds. Aquac. Nutr. 2007, 13, 17–34. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Feed matters: Satisfying the feed demand of aquaculture. Rev. Fish. Sci. Aquac. 2015, 23, 1–10. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Global overview on the use of fish meal and fish oil in industrially compounded aquafeed: Trends and future prospects. Aquaculture 2008, 285, 146–158. [Google Scholar] [CrossRef]
- Tabinda, A.B.; Butt, A. Replacement of fish meal with poultry by-product meal (chicken intestine) as a protein source in grass carp fry diet. Pak. J. Zool. 2012, 44, 1373–1381. [Google Scholar]
- Zhou, Q.; Tan, B.; Mai, K.; Liu, Y. Apparent digestibility of selected feed ingredients for juvenile cobia Rachycentron canadum. Aquaculture 2004, 241, 441–451. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhao, J.; Li, P.; Wang, H.; Wang, L. Evaluation of poultry by-product meal in commercial diets for juvenile cobia (Rachycentron canadum). Aquaculture 2011, 322, 122–127. [Google Scholar] [CrossRef]
- Ha, M.S.; Lee, K.W.; Kim, J.; Yun, A.; Jeong, H.S.; Lee, M.J.; Baek, S.I.; Cho, S.H.; Kim, K.W.; Lim, G.S.; et al. Dietary substitution effect of fish meal with chicken by-product meal on growth, feed utilization, body composition, haematology and non-specific immune responses of olive flounder (Paralichthys olivaceus). Aquac. Nutr. 2021, 27, 315–326. [Google Scholar] [CrossRef]
- El-Sayed, A.F.M. Evaluation of soybean meal, spirulina meal and chicken offal meal as protein sources for silver seabream (Rhabdosargus sarba) fingerlings. Aquaculture 1994, 127, 169–176. [Google Scholar] [CrossRef]
- Nandakumar, S.; Ambasankar, K.; Dayal, J.S.; Raman, C.; Ali, S.R. Fish meal replacement with chicken waste meal in Asian seabass (Lates calcarifer) feeds. Indian J. Fish. 2013, 60, 109–114. [Google Scholar]
- Millamena, O.M. Replacement of fish meal by animal by-product meals in a practical diet for grow-out culture of grouper Epinephelus coioides. Aquaculture 2002, 204, 75–84. [Google Scholar] [CrossRef]
- Sato, T.; Kikuchi, K. Meat meal as a protein source in the diet of juvenile Japanese flounder. Fish. Sci. 1997, 63, 877–880. [Google Scholar] [CrossRef]
- Williams, K.C.; Barlow, C.G.; Rodgers, L.J.; Ruscoe, I. Potential of meat meal to replace fish meal in extruded dry diets for barramundi, Lates calcarifer (Bloch). I. Growth performance. Aquac. Res. 2003, 34, 23–32. [Google Scholar] [CrossRef]
- Huang, J.; Zhou, C.; Xu, F.; Luo, X.; Huang, X.; Huang, Z.; Yu, W.; Xun, P.; Wu, Y.; Lin, H. Effects of partial replacement of fish meal with porcine meat meal on growth performance, antioxidant status, intestinal morphology, gut microflora and immune response of juvenile golden pompano (Trachinotus ovatus). Aquaculture 2022, 561, 738646. [Google Scholar] [CrossRef]
- Ha, M.S.; Cho, S.H.; Kim, T. Dietary substitution of fish meal by meat meal: Effects on juvenile olive flounder (Paralichthys olivaceus) growth performance, feed utilization, haematology, biochemical profile and disease resistance against Streptococcus iniae. Aquac. Nutr. 2021, 27, 1888–1902. [Google Scholar] [CrossRef]
- Hernández, C.; Olvera-Novoa, M.A.; Voltolina, D.; Hardy, R.W.; González-Rodriguez, B.; Domínguez-Jímenez, P.; Valverde-Romero, M.; Agramon-Romero, S. Use of tuna industry waste in diets for Nile tilapia, Oreochromis niloticus, fingerlings: Effect on digestibility and growth performance. Lat. Am. J. Aquat. Res. 2013, 41, 468–478. [Google Scholar] [CrossRef]
- Kim, K.; Jang, J.W.; Kim, K.; Lee, B.; Hur, S.W.; Han, H. Tuna by-product meal as a dietary protein source replacing fishmeal in juvenile Korean rockfish Sebastes schlegeli. Fish. Aquat. Sci. 2018, 21, 29. [Google Scholar] [CrossRef]
- Hernández, C.; Hardy, R.W.; Contreras-Rojas, D.; Lopez-Molina, B.; González-Rodríguez, B.; Domínguez-Jiménez, P. Evaluation of tuna by-product meal as a protein source in feeds for juvenile spotted rose snapper Lutjanus guttatus. Aquac. Nutr. 2014, 20, 574–582. [Google Scholar] [CrossRef]
- Saïdi, S.A.; Azaza, M.S.; Abdelmouleh, A.; Pelt, J.V.; Kraïem, M.M.; El-Feki, A. The use of tuna industry waste in the practical diets of juvenile Nile tilapia (Oreochromis niloticus L.): Effect on growth performance, nutrient digestibility and oxidative status. Aquac. Res. 2010, 41, 1875–1886. [Google Scholar] [CrossRef]
- Oncul, F.O.; Aya, F.A.; Hamidoghli, A.; Won, S.; Lee, G.; Han, K.R.; Bai, S. Effects of the dietary fermented tuna by-product meal on growth, blood parameters, nonspecific immune response, and disease resistance in juvenile olive flounder, Paralichthys olivaceus. J. World. Aquac. Soc. 2019, 50, 65–77. [Google Scholar] [CrossRef]
- Uyan, O.; Koshio, S.; Teshima, S.; Ishikawa, M.; Thu, M.; Alam, M.S.; Michael, F.R. Growth and phosphorus loading by partially replacing fishmeal with tuna muscle by-product powder in the diet of juvenile Japanese flounder, Paralichthys olivaceus. Aquaculture 2006, 257, 437–445. [Google Scholar] [CrossRef]
- Ai, Q.; Xie, X. Effects of replacement of fish meal by soybean meal and supplementation of methionine in fish meal/soybean meal-based diets on growth performance of the Southern catfish Silurus meridionalis. J. World. Aquac. Soc. 2005, 36, 498–507. [Google Scholar] [CrossRef]
- Zou, Q.; Huang, Y.; Cao, J.; Zhao, H.; Wang, G.; Li, Y.; Pan, Q. Effects of four feeding stimulants in high plant-based diets on feed intake, growth performance, serum biochemical parameters, digestive enzyme activities and appetite-related genes expression of juvenile GIFT tilapia (Oreochromis sp.). Aquac. Nutr. 2017, 23, 1076–1085. [Google Scholar] [CrossRef]
- Tola, S.; Fukada, H.; Masumoto, T. Effects of natural feeding stimulants and glutamic acid supplementation on the feed intake, growth performance and digestive enzyme activities of red sea bream (Pagrus major) fed fish meal-free soy protein concentrate (SPC)-based diet. Aquac. Res. 2019, 50, 1912–1920. [Google Scholar] [CrossRef]
- Papatryphon, E.; Soares, J.H., Jr. The effect of dietary feeding stimulants on growth performance of striped bass, Morone saxatilis, fed-a-plant feedstuff-based diet. Aquaculture 2000, 185, 329–338. [Google Scholar] [CrossRef]
- Hidaka, I.; Kohbara, J.; Araki, T.; Morishita, T.; Miyajima, T.; Shimizu, S.; Kuriyama, I. Identification of feeding stimulants from a jack mackerel muscle extract for young yellowtail Seriola quinqueradiata. Aquaculture 2000, 181, 115–126. [Google Scholar] [CrossRef]
- Kim, H.S.; Baek, S.I.; Lee, K.W.; Jeong, H.S.; Cho, S.H. Attractiveness of various protein sources to juvenile rockfish (Sebastes schlegeli, Hilgendorf 1880). J. Appl. Aquac. 2020, 32, 205–220. [Google Scholar] [CrossRef]
- Kim, J.; Cho, S.H. Inclusion effect of jack mackerel (Trachurus japonicus) meal in the diet of rockfish (Sebastes schlegeli) on growth, feed utilization, biochemical composition, and innate immune responses. J. World Aquac. Soc. 2023, 54, 1137–1161. [Google Scholar] [CrossRef]
- Yamamoto, T.; Matsunari, H.; Oku, H.; Murashita, K.; Yoshinaga, H.; Furuita, H. Supplemental effects of histidine to fishmeal-based and low-fishmeal diets on the growth performance and tissue free amino acid levels of fingerling yellowtail Seriola quinqueradiata. Aquac. Sci. 2019, 67, 367–375. [Google Scholar]
- Cho, S.H.; Kim, H.S.; Myung, S.H.; Jung, W.; Choi, J.; Lee, S. Optimum dietary protein and lipid levels for juvenile rockfish (Sebastes schlegeli, Hilgendorf 1880). Aquac. Res. 2015, 46, 2954–2963. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kang, J.Y.; Lee, S.; Kim, I. Protein requirements of the Korean rockfish Sebastes schlegeli. J. Aquaculture 1993, 6, 13–27. [Google Scholar]
- Garling Jr, D.L.; Wilson, R.P. Optimum dietary protein to energy ratio for channel catfish fingerlings, Ictalurus punctatus. J. Nutr. 1976, 106, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
- AOAC. The Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Guđmundsdottir, B.K.; Magnadottir, B. Humoral immune parameters of cultured Atlantic halibut (Hippoglossus hippoglossus L.). Fish Shellfish Immunol. 2001, 11, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Llorens, S.; Moñino, A.V.; Tomás Vidal, A.; Salvador, V.J.M.; Pla Torres, M.; Jover Cerdá, M. Soybean meal as a protein source in gilthead sea bream (Sparus aurata L.) diets: Effects on growth and nutrient utilization. Aquac. Res. 2007, 38, 82–90. [Google Scholar] [CrossRef]
- Duncan, D.B. Multiple Range and Multiple F Tests. Biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]
- Yan, Q.; Xie, S.; Zhu, X.; Lei, W.; Yang, Y. Dietary lysine requirement for Sebastes schlegeli. Acta Hydrobiolo. Sin. 2006, 30, 459–465. [Google Scholar] [CrossRef]
- Yan, Q.; Xie, S.; Zhu, X.; Lei, W.; Yang, Y. Dietary methionine requirement for juvenile rockfish, Sebastes schlegeli. Aquac. Nutr. 2007, 13, 163–169. [Google Scholar] [CrossRef]
- Hodar, A.R.; Vasava, R.J.; Mahavadiya, D.R.; Joshi, N.H. Fish meal and fish oil replacement for aqua feed formulation by using alternative sources: A review. J. Exp. Zool. India 2020, 23, 13–21. [Google Scholar]
- Luthada-Raswiswi, R.; Mukaratirwa, S.; O’brien, G. Animal protein sources as a substitute for fishmeal in aquaculture diets: A systematic review and meta-analysis. Appl. Sci. 2021, 11, 3854. [Google Scholar] [CrossRef]
- Kim, J.; Cho, S.H.; Kim, T.; Hur, S.W. Substitution effect of fish meal with various sources of animal by-product meals in feed on growth, feed utilization, body composition, haematology and non-specific immune response of olive flounder (Paralichthys olivaceus, Temminck & Schlegel, 1846). Aquac. Res. 2021, 52, 2802–2817. [Google Scholar]
- Jeong, H.S.; Cho, S.H. Inclusion effect of jack mackerel meal as feed stimulants in diets replacing different levels of fish meal with various animal protein sources on growth performance of olive flounder (Paralichthys olivaceus). Aquac. Rep. 2023, 28, 101450. [Google Scholar] [CrossRef]
- Kamalam, B.S.; Medale, F.; Panserat, S. Utilisation of dietary carbohydrates in farmed fishes: New insights on influencing factors, biological limitations and future strategies. Aquaculture 2017, 467, 3–27. [Google Scholar] [CrossRef]
- Panserat, S.; Kaushik, S.; Médale, F. Rainbow trout as a model for nutrition and nutrient metabolism studies. In Trout: From Physiology to Conservation, 1st ed.; Polakof, S., Moon, T.W., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 131–153. ISBN 978-1-62417-009-6. [Google Scholar]
- Ma, H.; Mou, M.; Pu, D.; Lin, S.; Chen, Y.; Luo, L. Effect of dietary starch level on growth, metabolism enzyme and oxidative status of juvenile largemouth bass, Micropterus salmoides. Aquaculture 2019, 498, 482–487. [Google Scholar] [CrossRef]
- Wang, J.; Li, X.; Han, T.; Yang, Y.; Jiang, Y.; Yang, M.; Xu, Y.; Harpaz, S. Effects of different dietary carbohydrate levels on growth, feed utilization and body composition of juvenile grouper Epinephelus akaara. Aquaculture 2016, 459, 143–147. [Google Scholar] [CrossRef]
- Kader, M.A.; Koshio, S.; Ishikawa, M.; Yokoyama, S.; Bulbul, M. Supplemental effects of some crude ingredients in improving nutritive values of low fishmeal diets for red sea bream, Pagrus major. Aquaculture 2010, 308, 136–144. [Google Scholar] [CrossRef]
- Khosravi, S.; Bui, H.T.D.; Herault, M.; Fournier, V.; Kim, K.; Lee, B.; Kim, K.; Lee, K. Supplementation of protein hydrolysates to a low-fishmeal diet improves growth and health status of juvenile olive flounder, Paralichthys olivaceus. J. World. Aquac. Soc. 2018, 49, 897–911. [Google Scholar] [CrossRef]
- Tharaka, K.; Benitez-Santana, T.; Gunathilaka, B.E.; Kim, M.; Lee, C.; Shin, J.; Lee, K. Evaluation of Antarctic krill (Euphausia superba) meal supplementation in diets for olive flounder (Paralichthys olivaceus). Aquac. Res. 2020, 51, 2291–2302. [Google Scholar] [CrossRef]
- Yigit, M.; Erdem, M.; Koshio, S.; Ergün, S.; Türker, A.; Karaali, B. Substituting fish meal with poultry by-product meal in diets for black Sea turbot Psetta maeotica. Aquac. Nutr. 2006, 12, 340–347. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Zhang, C.; Song, K. Effects of substituting fishmeal with soybean meal on growth performance and intestinal morphology in orange-spotted grouper (Epinephelus coioides). Aquac. Rep. 2017, 5, 52–57. [Google Scholar] [CrossRef]
- Karapanagiotidis, I.T.; Psofakis, P.; Mente, E.; Malandrakis, E.; Golomazou, E. Effect of fishmeal replacement by poultry by-product meal on growth performance, proximate composition, digestive enzyme activity, haematological parameters and gene expression of gilthead seabream (Sparus aurata). Aquac. Nutr. 2019, 25, 3–14. [Google Scholar] [CrossRef]
- Li, P.; Wu, G. Composition of amino acids and related nitrogenous nutrients in feedstuffs for animal diets. Amino Acids 2020, 52, 523–542. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Zhu, X.; Yang, Y.; Han, D.; Xie, S. Feasibility of partial replacement of fishmeal with proteins from different sources in diets of Korean rockfish (Sebastes schlegeli). J. Ocean Univ. China 2014, 13, 1054–1060. [Google Scholar] [CrossRef]
- Yu, Y. Replacement of fishmeal with poultry byproduct meal and meat and bone meal in shrimp, tilapia and trout diets. In Avances en Nutrición Acuicola VII, 1st ed.; Cruz Suárez, L.E., Ricque Marie, D., Nieto López, M.G., Villareal, D., Scholz, U., González, M., Eds.; Memorias del VII Simposium Internacional de Nutrición Acuicola: Hermosillo, Mexico, 2004; pp. 182–201. [Google Scholar]
- Shapawi, R.; Wing, N.; Mustafa, S. Replacement of fish meal with poultry byproduct meal in diets formulated for the humpback grouper, Cromileptes altivelis. Aquaculture 2007, 273, 118–126. [Google Scholar] [CrossRef]
- Joint FAO/WHO Ad Hoc Expert Committee on Energy, Protein Requirements. Energy and Protein Requirements: Report of a Joint FAO/WHO Ad Hoc Expert Committee. In Proceedings of the FAO/WHO Meeting, Rome, Italy, 22 March–2 April 1971. No. 52. [Google Scholar]
- Candebat, C.L.; Booth, M.A.; Pirozzi, I. The sulfur amino acid requirements of juvenile yellowtail kingfish (Seriola lalandi). Aquaculture 2021, 534, 736234. [Google Scholar] [CrossRef]
- Madrid, J.; Pohlenz, C.; Viana, M.T.; Lazo, J.P. Dietary lysine requirement for juvenile, Totoaba macdonaldi. Aquaculture 2019, 500, 92–98. [Google Scholar] [CrossRef]
- Wang, L.; Gao, C.; Wang, B.; Wang, C.; Sagada, G.; Yan, Y. Methionine in fish health and nutrition: Potential mechanisms, affecting factors, and future perspectives. Aquaculture 2023, 568, 739310. [Google Scholar] [CrossRef]
- Feng, H.; Yi, K.; Qian, X.; Niu, X.; Sun, Y.; Ye, J. Growth and metabolic responses of juvenile grouper (Epinephelus coioides) to dietary methionine/cystine ratio at constant sulfur amino acid levels. Aquaculture 2020, 518, 734869. [Google Scholar] [CrossRef]
- Jin, M.; Lu, Y.; Yuan, Y.; Li, Y.; Qiu, H.; Sun, P.; Ma, H.; Ding, L.; Zhou, Q. Regulation of growth, antioxidant capacity, fatty acid profiles, hematological characteristics and expression of lipid related genes by different dietary n-3 highly unsaturated fatty acids in juvenile black seabream (Acanthopagrus schlegelii). Aquaculture 2017, 471, 55–65. [Google Scholar] [CrossRef]
- Zuo, R.; Ai, Q.; Mai, K.; Xu, W.; Wang, J.; Xu, H.; Lihfu, A.; Zhang, Y. Effects of dietary n-3 highly unsaturated fatty acids on growth, nonspecific immunity, expression of some immune related genes and disease resistance of large yellow croaker (Larmichthys crocea) following natural infestation of parasites (Cryptocaryon irritans). Fish Shellfish Immunol. 2012, 32, 249–258. [Google Scholar]
- Zhang, S.; Xie, S.; Zhu, X.; Lei, W.; Yang, Y.; Zhao, M. Meat and bone meal replacement in diets for juvenile gibel carp (Carassius auratus gibelio): Effects on growth performance, phosphorus and nitrogen loading. Aquac. Nutr. 2006, 12, 353–362. [Google Scholar] [CrossRef]
- Kader, M.A.; Koshio, S.; Ishikawa, M.; Yokoyama, S.; Bulbul, M.; Nguyen, B.T.; Gao, J.; Laining, A. Can fermented soybean meal and squid by-product blend be used as fishmeal replacements for Japanese flounder (Paralichthys olivaceus)? Aquac. Res. 2012, 43, 1427–1438. [Google Scholar] [CrossRef]
- Kureshy, N.; Davis, D.A.; Arnold, C.R. Partial replacement of fish meal with meat-and-bone meal, flash-dried poultry by-product meal, and enzyme-digested poultry by-product meal in practical diets for juvenile red drum. N. Am. J. Aquac. 2000, 62, 266–272. [Google Scholar] [CrossRef]
- Dekić, R.; Savić, N.; Manojlović, M.; Golub, D.; Pavličević, J. Condition factor and organosomatic indices of rainbow trout (Onchorhynchus mykiss, Wal.) from different brood stock. Biotechnol. Anim. Husb. 2016, 32, 229–237. [Google Scholar] [CrossRef]
- Alam, M.S.; Liang, X.; Liu, L. Indirect effect of different dietary protein to energy ratio of bait fish mori diets on growth performance, body composition, nitrogen metabolism and relative AMPK & mTOR pathway gene expression of Chinese perch. Aquac. Rep. 2020, 16, 100276. [Google Scholar]
- Takakuwa, F.; Sato, H.; Mineyama, N.; Yamada, S.; Biswas, A.; Tanaka, H. Bioavailability of porcine blood meal as a fish meal substitute in the diet for red sea bream (Pagrus major, Temminck & Schlegel) fingerling. Aquac. Res. 2022, 53, 4616–4626. [Google Scholar]
- Hunter, B.J.; Allan, G.L.; Roberts, D.C.K. Meat meal replacement in diets for silver perch, Bidyanus bidyanus. J. Appl. Aquac. 2000, 10, 51–67. [Google Scholar] [CrossRef]
- Kader, M.A.; Koshio, S. Effect of composite mixture of seafood by-products and soybean proteins in replacement of fishmeal on the performance of red sea bream, Pagrus major. Aquaculture 2012, 368–369, 95–102. [Google Scholar] [CrossRef]
- Zou, Q.; Huang, Y.; Cao, J.; Zhao, H.; Wang, G.; Li, Y.; Pan, Q. Effects of supplemental nucleotides, taurine, and squid liver paste on feed intake, growth performance, serum biochemical parameters, and digestive enzyme activities of juvenile GIFT tilapia (Oreochromis sp.) fed low fishmeal diets. Isr. J. Aquac. 2016, 68, 20820. [Google Scholar]
- Yamamoto, T.; Unuma, T.; Akiyama, T. The influence of dietary protein and fat levels on tissue free amino acid levels of fingerling rainbow trout (Oncorhynchus mykiss). Aquaculture 2000, 182, 353–372. [Google Scholar] [CrossRef]
- Hernández, C.; Gonzalez-Santos, A.; Valverde-Romero, M.; Gonzalez-Rodriguez, B.; Domínguez-Jiménez, P. Partial replacement of fishmeal with meat and bone meal and tuna byproducts meal in practical diets for juvenile spotted rose snapper Lutjanus guttatus. Lat. Am. J. Aquat. Res. 2016, 44, 56–64. [Google Scholar] [CrossRef]
- Yones, A.M.M.; Metwalli, A.A. Effects of fish meal substitution with poultry by-product meal on growth performance, nutrients utilization and blood contents of juvenile Nile Tilapia (Oreochromis niloticus). J. Aquac. Res. Development 2015, 7, 1000389. [Google Scholar]
- Saurabh, S.; Sahoo, P.K. Lysozyme: An important defence molecule of fish innate immune system. Aquac. Res. 2008, 39, 223–239. [Google Scholar] [CrossRef]
- Gutteridge, J.M.; Halliwell, B. Free radicals and antioxidants in the year 2000: A historical look to the future. Ann. N. Y. Acad. Sci. 2000, 899, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Trenzado, C.; Hidalgo, M.C.; García-Gallego, M.; Morales, A.E.; Furné, M.; Domezain, A.; Domezain, J.; Sanz, A. Antioxidant enzymes and lipid peroxidation in sturgeon Acipenser naccarii and trout Oncorhynchus mykiss. A comparative study. Aquaculture 2006, 254, 758–767. [Google Scholar] [CrossRef]
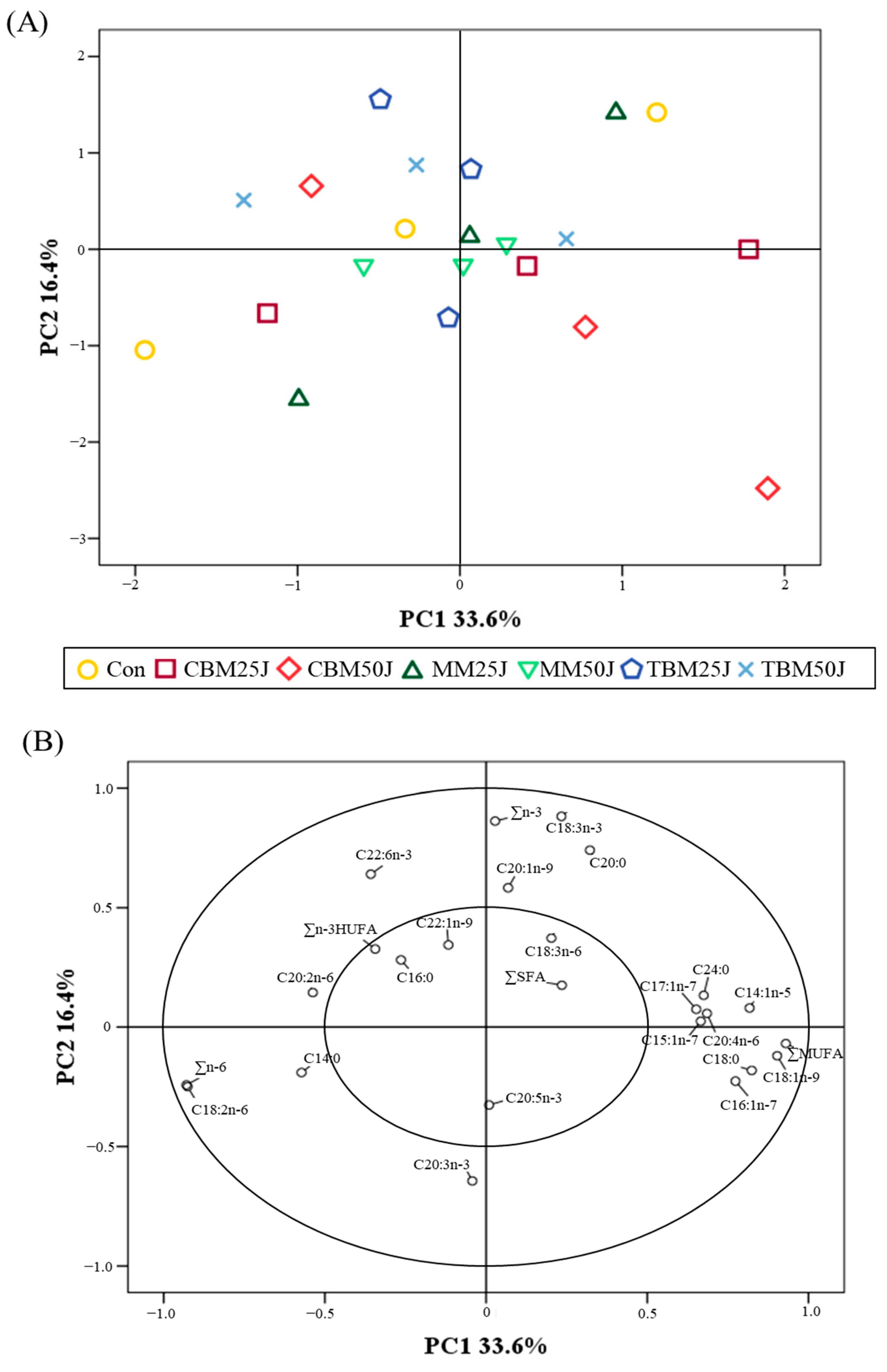
| Main Ingredients | Experimental Diets | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FM | JMM | CBM | MM | TBM | Requirement | Con | CBM25J | CBM50J | MM25J | MM50J | TBM25J | TBM50J | |
| Essential amino acids (EAAs, %) | |||||||||||||
| Arginine | 4.02 | 4.19 | 4.14 | 5.71 | 3.53 | 2.91 | 3.08 | 3.12 | 3.15 | 3.22 | 2.96 | 2.99 | |
| Histidine | 1.61 | 3.03 | 1.29 | 1.61 | 1.88 | 1.21 | 1.34 | 1.31 | 1.33 | 1.24 | 1.45 | 1.48 | |
| Isoleucine | 2.48 | 2.90 | 1.95 | 1.99 | 2.41 | 1.60 | 1.71 | 1.66 | 1.66 | 1.55 | 1.84 | 2.01 | |
| Leucine | 5.23 | 5.29 | 4.08 | 4.42 | 4.35 | 3.71 | 3.46 | 3.48 | 3.28 | 3.15 | 3.70 | 3.65 | |
| Lysine | 5.53 | 5.96 | 3.92 | 4.13 | 4.54 | 2.99 a | 3.81 | 3.25 | 3.13 | 3.17 | 3.10 | 3.87 | 3.72 |
| Methionine | 2.01 | 2.10 | 1.27 | 1.27 | 1.39 | 1.37 b | 1.61 | 1.24 | 1.19 | 1.20 | 1.11 | 1.43 | 1.30 |
| Phenylalanine | 2.77 | 2.86 | 2.22 | 2.50 | 2.38 | 2.06 | 1.95 | 1.84 | 1.87 | 1.81 | 2.11 | 2.20 | |
| Threonine | 3.14 | 3.28 | 2.43 | 2.42 | 2.57 | 2.24 | 2.06 | 1.97 | 1.94 | 1.79 | 2.10 | 2.06 | |
| Tryptophan | 0.45 | 1.22 | 0.47 | 0.38 | 0.46 | 0.31 | 0.42 | 0.46 | 0.36 | 0.32 | 0.41 | 0.44 | |
| Valine | 3.04 | 3.54 | 2.58 | 2.99 | 2.99 | 2.17 | 2.33 | 2.28 | 2.26 | 2.15 | 2.47 | 2.53 | |
| ∑EAAs c | 30.28 | 34.37 | 24.35 | 27.42 | 26.50 | 21.63 | 20.84 | 20.44 | 20.22 | 19.44 | 22.34 | 22.38 | |
| Non-essential amino acids (NEAAs, %) | |||||||||||||
| Alanine | 4.53 | 4.57 | 4.31 | 6.46 | 3.97 | 3.10 | 3.12 | 3.25 | 3.24 | 3.38 | 3.13 | 3.19 | |
| Aspartic acid | 6.40 | 6.69 | 4.87 | 5.57 | 5.21 | 4.76 | 4.36 | 4.25 | 4.09 | 3.89 | 4.56 | 4.42 | |
| Cysteine | 0.86 | 0.90 | 0.75 | 0.61 | 0.65 | 0.12 b | 0.82 | 0.81 | 0.82 | 0.69 | 0.60 | 0.74 | 0.71 |
| Glutamic acid | 8.95 | 9.28 | 8.22 | 9.73 | 7.30 | 7.09 | 7.19 | 7.34 | 6.99 | 6.91 | 7.08 | 6.98 | |
| Glycine | 3.83 | 4.39 | 6.05 | 11.94 | 4.25 | 2.65 | 3.51 | 3.84 | 4.15 | 4.83 | 3.06 | 3.17 | |
| Proline | 2.82 | 3.05 | 3.94 | 7.38 | 2.84 | 2.26 | 2.58 | 2.77 | 2.89 | 3.49 | 2.30 | 2.37 | |
| Serine | 2.94 | 3.05 | 2.46 | 2.90 | 2.36 | 2.25 | 2.15 | 2.10 | 2.04 | 2.01 | 2.13 | 2.10 | |
| Tyrosine | 2.06 | 2.04 | 1.44 | 1.44 | 1.49 | 1.40 | 1.16 | 1.12 | 1.14 | 1.06 | 1.19 | 1.16 | |
| ∑NEAAs d | 32.39 | 33.97 | 32.04 | 46.03 | 28.07 | 24.33 | 24.88 | 25.49 | 25.23 | 26.17 | 24.19 | 24.30 | |
| ∑TAAs e | 62.67 | 68.34 | 56.39 | 73.45 | 54.57 | 45.96 | 45.72 | 45.93 | 45.45 | 45.61 | 46.53 | 46.68 | |
| ∑EAAs/∑TAAs | 0.48 | 0.50 | 0.43 | 0.37 | 0.49 | 0.47 | 0.46 | 0.45 | 0.44 | 0.43 | 0.48 | 0.48 | |
| ∑EAAs/∑NEAAs | 0.93 | 1.01 | 0.76 | 0.60 | 0.94 | 0.89 | 0.84 | 0.80 | 0.80 | 0.74 | 0.92 | 0.92 | |
| Main Ingredients | Experimental Diets | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fatty Acid (%) | FM | JMM | CBM | MM | TBM | Con | CBM25J | CBM50J | MM25J | MM50J | TBM25J | TBM50J |
| C14:0 | 6.41 | 5.01 | 0.97 | 2.43 | 4.22 | 1.69 | 1.39 | 1.36 | 1.50 | 1.31 | 1.65 | 1.62 |
| C16:0 | 23.34 | 21.42 | 28.12 | 28.08 | 27.36 | 15.19 | 17.05 | 17.74 | 17.01 | 17.69 | 16.84 | 16.98 |
| C18:0 | 5.01 | 7.44 | 8.71 | 14.99 | 8.06 | 4.23 | 4.97 | 5.02 | 5.93 | 6.68 | 4.83 | 5.09 |
| C20:0 | 0.32 | 0.32 | 0.17 | 0.23 | 0.69 | 0.26 | 0.34 | 0.34 | 0.27 | 0.35 | 0.30 | 0.32 |
| C24:0 | 2.56 | 3.31 | 0.04 | 0.03 | 1.26 | 0.66 | 0.61 | 0.47 | 0.59 | 0.39 | 0.65 | 0.57 |
| ∑SFA a | 37.64 | 37.50 | 38.01 | 45.76 | 41.59 | 22.03 | 24.36 | 24.93 | 25.30 | 26.42 | 24.27 | 24.58 |
| C14:1n-5 | 0.13 | 0.14 | 0.24 | 0.05 | 0.05 | 0.01 | 0.04 | 0.07 | 0.04 | 0.05 | 0.02 | 0.04 |
| C15:1n-7 | 0.07 | 0.07 | 0.27 | 0.06 | 0.13 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.03 | 0.04 |
| C16:1n-7 | 6.73 | 6.74 | 5.78 | 3.05 | 5.65 | 2.26 | 2.24 | 2.18 | 2.18 | 2.04 | 2.22 | 2.19 |
| C17:1n-7 | 0.81 | 0.86 | 0.19 | 0.45 | 0.75 | 0.12 | 0.16 | 0.12 | 0.17 | 0.16 | 0.16 | 0.14 |
| C18:1n-9 | 15.23 | 20.36 | 48.02 | 45.46 | 23.47 | 22.38 | 28.21 | 30.51 | 27.79 | 29.06 | 25.85 | 26.38 |
| C20:1n-9 | 4.17 | 1.80 | 1.05 | 1.36 | 2.30 | 2.11 | 0.88 | 0.85 | 1.03 | 0.94 | 1.21 | 1.05 |
| C22:1n-9 | 0.45 | 0.83 | 0.06 | 0.03 | 2.16 | 0.28 | 0.24 | 0.22 | 0.25 | 0.25 | 0.29 | 0.31 |
| ∑MUFA b | 27.59 | 30.80 | 55.61 | 50.46 | 34.51 | 27.18 | 31.79 | 33.97 | 31.48 | 32.52 | 29.78 | 30.15 |
| C18:2n-6 | 4.17 | 1.73 | 4.86 | 2.30 | 2.37 | 35.72 | 31.81 | 31.88 | 31.68 | 31.02 | 31.75 | 31.68 |
| C18:3n-3 | 0.57 | 0.76 | 0.06 | 0.03 | 0.80 | 4.43 | 3.67 | 2.07 | 3.59 | 3.54 | 4.52 | 4.42 |
| C18:3n-6 | 0.23 | 0.10 | 0.02 | 0.04 | 0.32 | 0.06 | 0.06 | 0.04 | 0.05 | 0.05 | 0.06 | 0.06 |
| C20:2n-6 | 0.08 | 0.17 | 0.08 | 0.09 | 0.29 | 0.13 | 0.12 | 0.12 | 0.12 | 0.11 | 0.14 | 0.15 |
| C20:3n-3 | 0.08 | 0.03 | 0.03 | 0.03 | 0.04 | 0.03 | 0.04 | 0.05 | ||||
| C20:4n-6 | 1.14 | 0.69 | 0.36 | 0.15 | 1.31 | 0.36 | 0.33 | 0.31 | 0.30 | 0.24 | 0.42 | 0.44 |
| C20:5n-3 | 10.54 | 10.73 | 0.13 | 0.06 | 4.58 | 4.53 | 3.59 | 3.02 | 3.49 | 2.76 | 3.86 | 3.36 |
| C22:6n-3 | 13.30 | 15.40 | 0.37 | 0.02 | 10.48 | 4.61 | 3.52 | 3.01 | 3.24 | 2.68 | 4.17 | 4.07 |
| ∑n-3 HUFA c | 23.92 | 26.13 | 0.50 | 0.08 | 15.06 | 9.17 | 7.14 | 6.06 | 6.77 | 5.47 | 8.07 | 7.48 |
| ∑n-3 d | 24.49 | 26.89 | 0.56 | 0.11 | 15.86 | 13.60 | 10.81 | 8.13 | 10.36 | 9.01 | 12.59 | 11.90 |
| ∑n-6 e | 5.62 | 2.69 | 5.32 | 2.58 | 4.29 | 36.27 | 32.32 | 32.35 | 32.15 | 31.42 | 32.37 | 32.33 |
| ∑n-3/∑n-6 | 4.36 | 10.00 | 0.11 | 0.04 | 3.70 | 0.37 | 0.33 | 0.25 | 0.32 | 0.29 | 0.39 | 0.37 |
| Unknown | 4.66 | 2.12 | 0.50 | 1.09 | 3.75 | 0.92 | 0.72 | 0.62 | 0.71 | 0.63 | 0.99 | 1.04 |
| Experimental Diets | Initial Weight (g/Fish) | Final Weight (g/Fish) | Survival (%) | FE a | PER b | PR c (%) | CF d | VSI e (%) | HIS f (%) |
|---|---|---|---|---|---|---|---|---|---|
| Con | 8.3 ± 0.07 | 30.1 ± 0.25 ab | 100.0 ± 0.00 | 1.14 ± 0.006 a | 2.21 ± 0.012 a | 41.03 ± 1.811 a | 1.65 ± 0.032 | 9.86 ± 0.271 | 2.79 ± 0.118 |
| CBM25J | 8.3 ± 0.05 | 31.6 ± 0.39 a | 98.7 ± 1.33 | 1.15 ± 0.035 a | 2.21 ± 0.058 a | 38.41 ± 1.346 ab | 1.62 ± 0.030 | 10.20 ± 0.309 | 2.89 ± 0.021 |
| CBM50J | 8.3 ± 0.09 | 28.0 ± 0.96 c | 98.7 ± 1.33 | 1.03 ± 0.032 b | 1.98 ± 0.063 b | 35.75 ± 0.889 b | 1.60 ± 0.040 | 9.46 ± 0.179 | 2.57 ± 0.148 |
| MM25J | 8.3 ± 0.05 | 29.8 ± 0.56 ab | 96.0 ± 2.31 | 1.03 ± 0.043 b | 1.98 ± 0.080 b | 34.44 ± 1.494 bc | 1.63 ± 0.050 | 9.47 ± 0.112 | 2.84 ± 0.113 |
| MM50J | 8.3 ± 0.05 | 25.1 ± 0.48 d | 100.0 ± 0.00 | 0.89 ± 0.050 c | 1.73 ± 0.097 c | 30.60 ± 2.074 c | 1.54 ± 0.016 | 9.65 ± 0.161 | 3.10 ± 0.030 |
| TBM25J | 8.3 ± 0.07 | 30.4 ± 0.26 a | 100.0 ± 0.00 | 1.09 ± 0.010 ab | 2.12 ± 0.020 ab | 36.39 ± 1.155 b | 1.60 ± 0.009 | 9.77 ± 0.423 | 2.51 ± 0.251 |
| TBM50J | 8.4 ± 0.03 | 28.3 ± 0.73 bc | 97.3 ± 2.67 | 1.09 ± 0.025 ab | 2.08 ± 0.043 ab | 36.51 ± 0.241 b | 1.63 ± 0.018 | 9.98 ± 0.448 | 2.69 ± 0.191 |
| p-value | p < 0.0001 | p > 0.4 | p < 0.001 | p < 0.001 | p < 0.005 | p > 0.3 | p > 0.5 | p > 0.1 | |
| Main effect: substitution source | |||||||||
| CBM | 29.8 ± 0.93 A | 98.7 ± 0.84 | 1.09 ± 0.034 A | 2.10 ± 0.063 A | 37.08 ± 0.935 A | 1.61 ± 0.023 | 9.83 ± 0.229 | 2.73 ± 0.097 | |
| MM | 27.5 ± 1.10 B | 98.0 ± 1.37 | 0.96 ± 0.043 B | 1.86 ± 0.079 B | 32.52 ± 1.430 B | 1.59 ± 0.030 | 9.56 ± 0.097 | 2.97 ± 0.078 | |
| TBM | 29.4 ± 0.57 A | 98.7 ± 1.33 | 1.09 ± 0.012 A | 2.10 ± 0.023 A | 36.45 ± 0.529 A | 1.61 ± 0.013 | 9.87 ± 0.279 | 2.60 ± 0.147 | |
| Main effect: substitution level | |||||||||
| 25% | 30.6 ± 0.41 A | 98.2 ± 1.19 | 1.09 ± 0.029 A | 2.10 ± 0.054 A | 36.41 ± 1.080 | 1.61 ± 0.022 | 9.81 ± 0.229 | 2.74 ± 0.122 | |
| 50% | 27.1 ± 0.78 B | 98.7 ± 1.16 | 1.00 ± 0.043 B | 1.93 ± 0.077 B | 34.28 ± 1.391 | 1.59 ± 0.023 | 9.70 ± 0.202 | 2.79 ± 0.129 | |
| Two-way ANOVA | |||||||||
| Substitution source | p < 0.005 | p > 0.8 | p < 0.004 | p < 0.003 | p < 0.01 | p > 0.5 | p > 0.7 | p > 0.08 | |
| Substitution level | p < 0.0001 | p > 0.7 | p < 0.02 | p < 0.008 | p > 0.07 | p > 0.3 | p > 0.7 | p > 0.6 | |
| Interaction | p > 0.1 | p > 0.1 | p > 0.1 | p > 0.2 | p > 0.3 | p > 0.1 | p > 0.4 | p > 0.2 | |
| Experimental Diets | Moisture | Crude Protein | Crude Lipid | Ash |
|---|---|---|---|---|
| Con | 69.9 ± 1.30 | 18.0 ± 0.62 | 8.6 ± 0.29 | 4.4 ± 0.12 |
| CBM25J | 69.7 ± 0.67 | 17.1 ± 0.12 | 7.9 ± 0.37 | 4.5 ± 0.24 |
| CBM50J | 70.8 ± 1.22 | 17.5 ± 0.37 | 6.4 ± 0.68 | 4.8 ± 0.15 |
| MM25J | 70.6 ± 1.63 | 17.1 ± 0.08 | 7.9 ± 0.68 | 4.6 ± 0.11 |
| MM50J | 70.6 ± 2.28 | 17.2 ± 0.25 | 7.0 ± 0.30 | 4.7 ± 0.27 |
| TBM25J | 70.4 ± 1.04 | 16.9 ± 0.38 | 7.4 ± 0.61 | 4.4 ± 0.44 |
| TBM50J | 68.3 ± 2.55 | 17.2 ± 0.23 | 7.9 ± 0.05 | 4.6 ± 0.47 |
| p-value | p > 0.9 | p > 0.4 | p > 0.1 | p > 0.9 |
| Main effect: substitution source | ||||
| CBM | 70.2 ± 0.67 | 17.3 ± 0.20 | 7.1 ± 0.48 | 4.6 ± 0.14 |
| MM | 70.6 ± 1.25 | 17.2 ± 0.12 | 7.5 ± 0.39 | 4.6 ± 0.13 |
| TBM | 69.4 ± 1.32 | 17.0 ± 0.21 | 7.7 ± 0.29 | 4.5 ± 0.29 |
| Main effect: substitution level | ||||
| 25% | 70.3 ± 0.74 | 17.1 ± 0.15 | 7.7 ± 0.36 | 4.5 ± 0.18 |
| 50% | 69.9 ± 1.37 | 17.3 ± 0.19 | 7.1 ± 0.38 | 4.7 ± 0.20 |
| Two-way ANOVA | ||||
| Substitution source | p > 0.7 | p > 0.6 | p > 0.6 | p > 0.8 |
| Substitution level | p > 0.7 | p > 0.2 | p > 0.1 | p > 0.4 |
| Interaction | p > 0.6 | p > 0.8 | p > 0.1 | p > 0.9 |
| Experimental Diets | Main Effect: Substitution Source | Main Effect: Substitution Level | Two-Way ANOVA | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Con | CBM25J | CBM50J | MM25J | MM50J | TBM25J | TBM50J | p-Value | CBM | MM | TBM | 25% | 50% | Substitution Source | Substitution Level | Interaction | |
| Essential amino acids (%) | ||||||||||||||||
| Arginine | 1.1 ± 0.03 | 1.1 ± 0.08 | 1.1 ± 0.02 | 1.1 ± 0.04 | 1.1 ± 0.06 | 1.1 ± 0.04 | 1.0 ± 0.01 | p > 0.5 | 1.1 ± 0.04 | 1.1 ± 0.04 | 1.0 ± 0.03 | 1.1 ± 0.04 | 1.1 ± 0.03 | p > 0.4 | p > 0.4 | p > 0.3 |
| Histidine | 0.4 ± 0.17 | 0.4 ± 0.06 | 0.4 ± 0.02 | 0.4 ± 0.07 | 0.4 ± 0.07 | 0.4 ± 0.06 | 0.4 ± 0.08 | p > 0.9 | 0.4 ± 0.03 | 0.4 ± 0.04 | 0.4 ± 0.05 | 0.4 ± 0.04 | 0.4 ± 0.04 | p > 0.9 | p > 0.6 | p > 0.9 |
| Isoleucine | 0.8 ± 0.12 | 0.7 ± 0.11 | 0.7 ± 0.08 | 0.7 ± 0.10 | 0.7 ± 0.10 | 0.7 ± 0.11 | 0.7 ± 0.07 | p > 0.9 | 0.7 ± 0.06 | 0.7 ± 0.06 | 0.7 ± 0.06 | 0.7 ± 0.07 | 0.7 ± 0.05 | p > 0.9 | p > 0.5 | p > 0.8 |
| Leucine | 1.3 ± 0.10 | 1.3 ± 0.10 | 1.2 ± 0.06 | 1.2 ± 0.06 | 1.2 ± 0.08 | 1.3 ± 0.10 | 1.1 ± 0.01 | p > 0.8 | 1.2 ± 0.05 | 1.2 ± 0.04 | 1.2 ± 0.05 | 1.3 ± 0.05 | 1.2 ± 0.04 | p > 0.8 | p > 0.3 | p > 0.5 |
| Lysine | 1.4 ± 0.10 | 1.5 ± 0.12 | 1.4 ± 0.08 | 1.4 ± 0.08 | 1.4 ± 0.11 | 1.5 ± 0.11 | 1.3 ± 0.01 | p > 0.7 | 1.4 ± 0.07 | 1.4 ± 0.06 | 1.4 ± 0.06 | 1.4 ± 0.06 | 1.4 ± 0.06 | p > 0.8 | p > 0.3 | p > 0.5 |
| Methionine | 0.6 ± 0.05 | 0.6 ± 0.05 | 0.6 ± 0.04 | 0.5 ± 0.02 | 0.6 ± 0.07 | 0.6 ± 0.05 | 0.6 ± 0.03 | p > 0.7 | 0.6 ± 0.03 | 0.6 ± 0.04 | 0.6 ± 0.03 | 0.6 ± 0.03 | 0.6 ± 0.03 | p > 0.9 | p > 0.9 | p > 0.2 |
| Phenylalanine | 0.7 ± 0.08 | 0.7 ± 0.04 | 0.7 ± 0.05 | 0.7 ± 0.03 | 0.7 ± 0.05 | 0.7 ± 0.05 | 0.7 ± 0.03 | p > 0.9 | 0.7 ± 0.03 | 0.7 ± 0.03 | 0.7 ± 0.03 | 0.7 ± 0.03 | 0.7 ± 0.03 | p > 0.9 | p > 0.7 | p > 0.7 |
| Threonine | 0.8 ± 0.04 | 0.8 ± 0.05 | 0.8 ± 0.04 | 0.8 ± 0.04 | 0.8 ± 0.06 | 0.8 ± 0.06 | 0.7 ± 0.02 | p > 0.8 | 0.8 ± 0.03 | 0.8 ± 0.03 | 0.7 ± 0.03 | 0.8 ± 0.03 | 0.8 ± 0.03 | p > 0.5 | p > 0.5 | p > 0.6 |
| Tryptophan | 0.1 ± 0.01 | 0.1 ± 0.02 | 0.1 ± 0.02 | 0.1 ± 0.01 | 0.1 ± 0.02 | 0.1 ± 0.02 | 0.1 ± 0.03 | p > 0.9 | 0.1 ± 0.01 | 0.1 ± 0.01 | 0.1 ± 0.02 | 0.1 ± 0.01 | 0.1 ± 0.01 | p > 0.8 | p > 0.4 | p > 0.8 |
| Valine | 0.9 ± 0.10 | 0.8 ± 0.01 | 0.8 ± 0.05 | 0.8 ± 0.04 | 0.8 ± 0.07 | 0.9 ± 0.10 | 0.7 ± 0.06 | p > 0.8 | 0.8 ± 0.03 | 0.8 ± 0.04 | 0.8 ± 0.06 | 0.8 ± 0.04 | 0.8 ± 0.04 | p > 0.9 | p > 0.3 | p > 0.6 |
| Non-essential amino acids (%) | ||||||||||||||||
| Alanine | 1.2 ± 0.06 | 1.2 ± 0.07 | 1.2 ± 0.04 | 1.2 ± 0.03 | 1.3 ± 0.08 | 1.2 ± 0.05 | 1.1 ± 0.01 | p > 0.6 | 1.2 ± 0.04 | 1.2 ± 0.04 | 1.2 ± 0.03 | 1.2 ± 0.04 | 1.2 ± 0.04 | p > 0.6 | p > 0.7 | p > 0.2 |
| Aspartic acid | 1.7 ± 0.12 | 1.7 ± 0.13 | 1.6 ± 0.08 | 1.7 ± 0.08 | 1.7 ± 0.11 | 1.7 ± 0.12 | 1.5 ± 0.02 | p > 0.7 | 1.7 ± 0.07 | 1.7 ± 0.06 | 1.6 ± 0.07 | 1.7 ± 0.04 | 1.6 ± 0.06 | p > 0.7 | p > 0.4 | p > 0.4 |
| Cysteine | 0.3 ± 0.03 | 0.2 ± 0.02 | 0.3 ± 0.03 | 0.2 ± 0.02 | 0.3 ± 0.03 | 0.2 ± 0.02 | 0.2 ± 0.01 | p > 0.9 | 0.2 ± 0.02 | 0.2 ± 0.02 | 0.2 ± 0.01 | 0.2 ± 0.01 | 0.2 ± 0.02 | p > 0.7 | p > 0.9 | p > 0.5 |
| Glutamic acid | 2.4 ± 0.12 | 2.4 ± 0.14 | 2.3 ± 0.12 | 2.4 ± 0.13 | 2.4 ± 0.17 | 2.4 ± 0.15 | 2.2 ± 0.22 | p > 0.8 | 2.4 ± 0.09 | 2.4 ± 0.10 | 2.3 ± 0.13 | 2.4 ± 0.09 | 2.3 ± 0.11 | p > 0.8 | p > 0.4 | p > 0.6 |
| Glycine | 1.5 ± 0.05 | 1.6 ± 0.12 | 1.5 ± 0.04 | 1.4 ± 0.08 | 1.6 ± 0.12 | 1.5 ± 0.04 | 1.5 ± 0.06 | p > 0.6 | 1.5 ± 0.06 | 1.5 ± 0.07 | 1.5 ± 0.03 | 1.5 ± 0.06 | 1.5 ± 0.05 | p > 0.7 | p > 0.7 | p > 0.2 |
| Proline | 0.8 ± 0.06 | 0.9 ± 0.10 | 0.8 ± 0.06 | 0.8 ± 0.05 | 0.9 ± 0.10 | 0.8 ± 0.04 | 0.8 ± 0.04 | p > 0.9 | 0.8 ± 0.06 | 0.8 ± 0.05 | 0.8 ± 0.03 | 0.8 ± 0.05 | 0.8 ± 0.05 | p > 0.7 | p > 0.9 | p > 0.6 |
| Serine | 0.7 ± 0.06 | 0.8 ± 0.07 | 0.8 ± 0.04 | 0.8 ± 0.05 | 0.8 ± 0.05 | 0.7 ± 0.04 | 0.7 ± 0.05 | p > 0.3 | 0.8 ± 0.04 | 0.8 ± 0.04 | 0.7 ± 0.03 | 0.8 ± 0.04 | 0.8 ± 0.04 | p > 0.1 | p > 0.8 | p > 0.4 |
| Tyrosine | 0.3 ± 0.03 | 0.4 ± 0.09 | 0.4 ± 0.05 | 0.4 ± 0.10 | 0.4 ± 0.09 | 0.4 ± 0.03 | 0.3 ± 0.04 | p > 0.7 | 0.4 ± 0.04 | 0.4 ± 0.06 | 0.3 ± 0.02 | 0.4 ± 0.05 | 0.4 ± 0.04 | p > 0.5 | p > 0.7 | p > 0.9 |
Experimental Diets | Plasma Parameters | Serum Parameters | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AST (U/L) | ALT (U/L) | ALP (U/L) | T-BIL (mg/dL) | T-CHO (mg/dL) | TG (mg/dL) | TP (g/dL) | ALB (g/dL) | Lysozyme Activity (U/mL) | SOD (ng/mL) | |
| Con | 154.0 ± 8.89 | 29.7 ± 1.67 | 293.0 ± 14.29 | 1.1 ± 0.30 | 299.0 ± 21.08 | 358.7 ± 23.41 | 5.4 ± 0.20 | 1.5 ± 0.10 | 502.0 ± 24.04 | 3.6 ± 0.18 |
| CBM25J | 133.3 ± 12.45 | 33.0 ± 12.77 | 229.0 ± 50.90 | 0.7 ± 0.06 | 285.7 ± 24.70 | 407.0 ± 55.19 | 4.8 ± 0.21 | 1.3 ± 0.19 | 386.7 ± 43.81 | 3.6 ± 0.24 |
| CBM50J | 138.7 ± 10.33 | 23.3 ± 2.60 | 230.0 ± 39.72 | 0.9 ± 0.17 | 254.7 ± 23.82 | 393.3 ± 18.50 | 4.8 ± 0.17 | 1.4 ± 0.21 | 408.9 ± 51.14 | 3.8 ± 0.41 |
| MM25J | 154.7 ± 16.75 | 28.3 ± 4.33 | 361.3 ± 40.96 | 2.0 ± 0.78 | 323.3 ± 20.34 | 351.0 ± 30.53 | 5.9 ± 0.47 | 2.0 ± 0.21 | 398.3 ± 31.93 | 4.0 ± 0.25 |
| MM50J | 183.3 ± 5.36 | 34.3 ± 5.78 | 246.3 ± 36.09 | 1.5 ± 0.18 | 276.3 ± 9.96 | 436.7 ± 21.88 | 5.1 ± 0.26 | 1.5 ± 0.09 | 445.0 ± 25.66 | 4.0 ± 0.06 |
| TBM25J | 152.3 ± 4.98 | 34.0 ± 2.31 | 260.0 ± 27.06 | 0.9 ± 0.37 | 281.7 ± 17.90 | 466.3 ± 33.67 | 5.0 ± 0.27 | 1.3 ± 0.15 | 411.7 ± 79.91 | 3.7 ± 0.32 |
| TBM50J | 171.7 ± 9.39 | 29.0 ± 8.08 | 228.7 ± 27.72 | 1.8 ± 0.88 | 256.3 ± 12.88 | 472.3 ± 27.67 | 5.0 ± 0.77 | 1.5 ± 0.38 | 415.0 ± 31.22 | 3.8 ± 0.25 |
| p-value | p > 0.05 | p > 0.8 | p > 0.1 | p > 0.4 | p > 0.2 | p > 0.09 | p > 0.5 | p > 0.3 | p > 0.7 | p > 0.9 |
| Main effect: substitution source | ||||||||||
| CBM | 136.0 ± 7.33 B | 28.2 ± 6.22 | 229.5 ± 28.87 | 0.8 ± 0.09 | 270.2 ± 16.84 | 400.2 ± 26.21 | 4.8 ± 0.12 | 1.4 ± 0.13 | 421.7 ± 21.08 | 3.7 ± 0.31 |
| MM | 169.0 ± 10.15 A | 31.3 ± 3.50 | 303.8 ± 35.46 | 1.8 ± 0.37 | 299.8 ± 14.60 | 393.7 ± 25.48 | 5.5 ± 0.29 | 1.7 ± 0.16 | 413.3 ± 38.38 | 3.8 ± 0.26 |
| TBM | 162.0 ± 6.42 A | 31.5 ± 3.92 | 244.3 ± 18.69 | 1.3 ± 0.47 | 269.0 ± 11.37 | 469.32 ± 19.53 | 5.0 ± 0.37 | 1.4 ± 0.19 | 437.3 ± 37.63 | 3.8 ± 0.20 |
| Main effect: substitution level | ||||||||||
| 25% | 146.8 ± 8.64 | 31.8 ± 4.95 | 283.4 ± 34.98 | 1.2 ± 0.40 | 296.9 ± 15.30 A | 408.1 ± 32.48 | 5.2 ± 0.29 | 1.6 ± 0.18 | 413.3 ± 38.39 | 3.6 ± 0.21 |
| 50% | 164.6 ± 9.75 | 28.9 ± 4.12 | 235.0 ± 21.64 | 1.4 ± 0.36 | 262.4 ± 11.05 B | 434.1 ± 19.85 | 5.0 ± 0.30 | 1.5 ± 0.16 | 434.9 ± 24.55 | 3.9 ± 0.28 |
| Two-way ANOVA | ||||||||||
| Substitution source | p < 0.03 | p > 0.8 | p > 0.1 | p > 0.1 | p > 0.2 | p > 0.08 | p > 0.2 | p > 0.2 | p > 0.8 | p > 0.9 |
| Substitution level | p > 0.06 | p > 0.6 | p > 0.1 | p > 0.6 | p < 0.05 | p > 0.3 | p > 0.7 | p > 0.6 | p > 0.6 | p > 0.4 |
| Interaction | p > 0.5 | p > 0.5 | p > 0.3 | p > 0.4 | p > 0.8 | p > 0.3 | p > 0.6 | p > 0.2 | p > 0.9 | p > 0.9 |
| Experimental Diets | Diet Price (USD/kg) | ECR (USD/kg) a | EPI (USD/Fish) b |
|---|---|---|---|
| Con | 1.63 | 1.43 ± 0.008 bc | 0.97 ± 0.008 ab |
| CBM25J | 1.59 | 1.40 ± 0.037 c | 1.02 ± 0.013 a |
| CBM50J | 1.42 | 1.39 ± 0.043 c | 0.90 ± 0.032 c |
| MM25J | 1.60 | 1.57 ± 0.063 ab | 0.96 ± 0.019 abc |
| MM50J | 1.44 | 1.63 ± 0.087 a | 0.81 ± 0.016 d |
| TBM25J | 1.64 | 1.50 ± 0.014 abc | 0.98 ± 0.009 ab |
| TBM50J | 1.52 | 1.42 ± 0.029 bc | 0.92 ± 0.023 bc |
| p-value | p < 0.02 | p < 0.0001 | |
| Main effect: substitution source | |||
| CBM | 1.39 ± 0.025 B | 0.96 ± 0.030 A | |
| MM | 1.60 ± 0.049 A | 0.88 ± 0.035 B | |
| TBM | 1.46 ± 0.023 B | 0.95 ± 0.018 A | |
| Main effect: substitution level | |||
| 25% | 1.49 ± 0.041 | 0.99 ± 0.014 A | |
| 50% | 1.48 ± 0.058 | 0.88 ± 0.026 B | |
| Two-way ANOVA | |||
| Substitution source | p < 0.004 | p < 0.006 | |
| Substitution level | p > 0.7 | p < 0.0001 | |
| Interaction | p > 0.4 | p > 0.1 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sim, Y.J.; Kim, J.; Cho, S.H. The Impacts and Economic Analysis of Jack Mackerel Meal Inclusion in Low Fish Meal Diets on the Growth and Feed Availability of Juvenile Rockfish (Sebastes schlegeli). Animals 2025, 15, 62. https://doi.org/10.3390/ani15010062
Sim YJ, Kim J, Cho SH. The Impacts and Economic Analysis of Jack Mackerel Meal Inclusion in Low Fish Meal Diets on the Growth and Feed Availability of Juvenile Rockfish (Sebastes schlegeli). Animals. 2025; 15(1):62. https://doi.org/10.3390/ani15010062
Chicago/Turabian StyleSim, Yu Jin, June Kim, and Sung Hwoan Cho. 2025. "The Impacts and Economic Analysis of Jack Mackerel Meal Inclusion in Low Fish Meal Diets on the Growth and Feed Availability of Juvenile Rockfish (Sebastes schlegeli)" Animals 15, no. 1: 62. https://doi.org/10.3390/ani15010062
APA StyleSim, Y. J., Kim, J., & Cho, S. H. (2025). The Impacts and Economic Analysis of Jack Mackerel Meal Inclusion in Low Fish Meal Diets on the Growth and Feed Availability of Juvenile Rockfish (Sebastes schlegeli). Animals, 15(1), 62. https://doi.org/10.3390/ani15010062







