Simple Summary
The jaguar spans about 7,000,000 km2 across the Americas. Livestock predation by jaguars often leads to retaliatory killings, but methods to reduce the frequency of these events have been developed and tested. We surveyed 248 livestock operations from northern Mexico to Argentina and evaluated the success of anti-depredation strategies implemented across 194 of those operations. These strategies necessitated varying levels of investment, but all achieved a notable reduction in depredation and, in most cases, were cost-effective. Anti-depredation strategies are effective, can be adapted to local needs, and merit wider application.
Abstract
The current range of the jaguar (Panthera onca) spans sixty degrees of latitude across eighteen countries in the Western Hemisphere and covers approximately 7,000,000 km2. Throughout this geographical breadth, jaguars represent an essential component of native biological diversity, but conflict revolving around real and perceived jaguar depredation on livestock is a factor in jaguar mortality. We developed a structured questionnaire to evaluate the effectiveness of anti-depredation strategies from northern Mexico to Argentina, collecting data from 11 countries and 248 livestock operations, 194 with efficacy metrics, and 24 with benefit–cost ratios (value of the livestock losses averted/cost of the intervention). Using coarse categories, 11 intervention types were tested. Techniques effectively reducing livestock losses were documented across the entire livestock operation size (2–130,000 ha, 5–30,000 head) and biome spectrum. While the techniques varied in complexity and required levels of investment, successful reductions in depredation were achieved at all levels. We conclude that anti-depredation strategies are highly effective, and when benefits are evaluated, they surpass costs, sometimes substantially. Given the proven efficacy and cost-effectiveness of the techniques described in this paper, we advocate for broader application across the species range to increase tolerance towards jaguars and a more effective human–jaguar coexistence.
1. Introduction
Large carnivores have impressed and intimidated human societies for thousands of years, but their survival is now one of our most pressing conservation challenges [1,2]. Human expansion has led to a decline in their populations worldwide due to habitat loss, fragmentation, degradation, and direct killing [3,4,5,6]. Carnivores play a crucial role in ecological processes and ecosystem structure and function by influencing the density and dynamics of prey species, which are typically primary consumer herbivores [2,3,4,5,6,7]. Large felids can play a role in vertebrate and invertebrate diversity and ecosystem services [8,9,10]. Their position at the top of the food chain makes them an important indicator of the status of biodiversity conservation [11]. Anthropogenic habitat fragmentation disrupts the behavior of carnivores with large home ranges [12,13] and can lead to increased conflicts with humans [14]. On a worldwide scale, inadequate livestock management can contribute to such conflicts and thus pose a significant threat to the survival of large felids and an issue to address [2,15].
The jaguar (Panthera onca) is the largest cat in the Americas and fits all the above-described global trends. Despite occupying around 50% of its original range, with sub-populations occurring from the northern Mexico–United States border area to the north of Argentina, the species is overall in decline. Jaguar habitats encompass multiple biomes, ranging from semi-xeric cactus-rich scrub forests to the flooded forests of the Amazon, spanning approximately 60° of latitude [16,17,18]. In 2020, the Jaguar 2030 Roadmap [19] estimated the remaining jaguar habitat at 7 million square kilometers. A team of experts estimated that, in 2023, there was 14% less jaguar habitat in South America than in 2015 and 25% less than in 2000 [20]. These decreases in habitat due to the expansion of human dwellings and agriculture emphasize the need for effective tools for coexistence.
Habitat connectivity for jaguars is still relatively intact in South America’s central Amazonian and Guiana Shield areas. While connectivity is still favorable in those and several other significant areas within the jaguar’s range, even in the most extensive extant blocks of jaguar habitat, fragmentation is increasing rapidly. Outside of the few truly vast areas of wild habitat, most sub-populations are endangered [21], and overall, the range-wide connectivity between fragmented jaguar populations is extremely tenuous and at risk [20]. Despite isolated success stories, the range reduction has been severe in many areas, and the trends show little sign of reversal. The jaguar has been eliminated from 77% of its historical range (or more) in Mesoamerica [22,23,24]. The jaguar is classified as “Near Threatened” by the IUCN Red List [25] and listed on Appendix I of CITES (www.cites.org). This status does not accurately reflect its imperiled status in most range states. On national levels, the jaguar is listed as “Extinct in the Wild” in two countries, “Critically Endangered” in four countries, “Endangered” in seven countries, “Vulnerable” in four countries, and “Near Threatened” in two countries [26,27].
The main threats to jaguar populations are habitat loss [28,29,30], reduced availability of natural prey [30,31,32], and direct killing, often due to conflicts with livestock [4,33,34,35,36]. Another factor contributing to direct fatalities is the trade of body parts, which varies in scale and characteristics across the species’ range [25,37,38]. The interactions of habitat loss, reduced natural prey, and direct killing often combine synergistically to drive local extinctions. For example, 85% of the jaguar habitat in the Atlantic Forest biome has been lost, with only 7% remaining in good condition. Within that, jaguars now only occupy 2.8% of their previous range. The primary causes of jaguar declines in that area were habitat loss and fragmentation, which drove increased proximity of humans, livestock, and jaguars. Direct killing became a critical threat in the remaining habitat [39].
Although habitat loss exerts a prominent role in the decline of the jaguar population, human–jaguar conflicts caused by livestock depredations significantly contributed to the contraction of the historical range of distribution of the species from Mexico to Argentina [40,41]. Human–jaguar conflicts focused on livestock were the primary factor in eradicating jaguars from the northernmost portion of their range in the United States [42], with similar dynamics driving range reduction on the southern edge of their distribution in Argentina [43]. The need for tools for successful coexistence has been urgent for decades.
Scientific observations on the jaguar depredation of livestock started in the 1980s [41,44]. During the first scientific study focused on jaguars and their prey [45,46], which took place on a ranch, the authors were forced to change the study area because ranch workers were killing the study animals. Strategies to reduce these conflicts were introduced in the early 1990s [47]. By the mid-1990s and early 2000s, knowledge and recommendations for lowering human–jaguar conflicts and improving coexistence increased [34,48,49].
Large-scale analyses of the status of jaguars started in 1999 [22]. These were accompanied by wide-ranging information sharing [18,50,51], which encouraged and facilitated discussions about the causes and solutions for human–jaguar conflicts [4,33]. These also include manuals that provide tools to reduce jaguar attacks on livestock [52,53].
Formal efforts to create comprehensive jaguar conservation strategies started in March 1999 when 29 jaguar experts from ten countries joined to evaluate the status of the jaguar and develop a detailed database describing the geographic distribution of jaguars and knowledge about them at the end of the twentieth century [51]. In 2006–2007, the Mesoamerican Jaguar Corridor concept was introduced [54,55]. Another significant milestone in the conservation of jaguars was the inaugural range-wide meeting on jaguar conservation at the United Nations in March 2018 [19]. Delegates from 14 jaguar range states joined the meeting, which marked the beginning of the multi-institutional and multi-governmental Jaguar 2030 conservation initiative, which includes the Jaguar 2030 Roadmap for the Americas [19]. The same momentum facilitated the implementation of the World Wildlife Fund’s Jaguar Strategy 2020–2030 [56]. The Jaguar 2030 initiative currently involves most jaguar range states, various United Nations agencies, several international conventions, numerous NGOs, and local communities. This high-level initiative aims to increase attention on jaguars to create tangible conservation impacts across the jaguar range, including where humans and jaguars coexist.
At the local level, there often needs to be more clarity between how farmers and ranchers perceive the impact of jaguar depredation on livestock and the reality of those impacts [33]. However, actual losses can be severe, especially for small-scale operators. There is no doubt that, historically, direct killing associated with actual or perceived conflict contributed to the decline of the jaguar range and that it continues to do so today. Killing jaguars may be illegal in almost every country in the jaguar’s range [27], but it is happening somewhere, to this very day, and is usually associated with livestock. While we recognize that habitat loss is the most significant threat across the jaguar range, we are also aware that (i) habitat loss generates a synergy that exacerbates conflicts with livestock, (ii) the direct killing of jaguars associated with livestock remains an enormous threat, and (iii) tools to reduce jaguar attacks on livestock have been tried and tested in select locations across much of jaguar range, and they now need to be upscaled.
Tools to reduce jaguar attacks on livestock have been tested in multiple countries, cultures, biomes, and contexts. Those tools may also reduce puma (Puma concolor) attacks on livestock. Building on the opportunities created by large-scale jaguar conservation efforts, we conducted a comprehensive study to assess the effectiveness of anti-depredation strategies (ADS). Our primary focus is jaguar conservation, but the findings also relate to puma. Our study aimed to determine which ADS have been tested, where they have been tested, the specific contexts in which they were tested, and their effectiveness in reducing livestock and financial losses. We also sought to identify the most effective techniques for different contexts. Whereas Khorozyan and Waltert (2019) [57] conducted a global review of the efficacy of interventions used to protect livestock from wild cats, including jaguars, in this study, we examine the tools used across the jaguar range in greater detail and across a broader spectrum of livestock operations. Our study compiles data from practitioners across eleven countries, encompassing various biomes and livestock operations, from small to large. It incorporates information from private, community, and indigenous operations, extending from northern Mexico to Argentina. This marks the first comprehensive evaluation of the effectiveness of jaguar anti-depredation tools at this scale and depth.
2. Materials and Methods
A core team developed a detailed questionnaire to distribute among a large group of jaguar biologists and livestock operators from northern Mexico to northern Argentina who possess field experience in techniques designed to reduce jaguar attacks on livestock. The questionnaire was developed iteratively by the following experts: John Polisar (experience in this subject from 1996 to 2020 across Central America and parts of South America); Rafael Hoogesteijn (experience from 1987 to 2025 in South America and areas of Central America); and Almira Hoogesteijn (1991–2025) with experience in parts of South America and Mexico. Over 90 years of cumulative experience working in and with non-governmental organizations, universities, and particularly within ranches and farms contributed to the original design of the questionnaires, which were then shared with the broader circle of participants. The questionnaire covered 12 pages and included significant details in every subsection. This questionnaire was divided into several sections [(Supplementary Materials S1): Study Participants and Study Area; Site Characteristics; Livestock Characteristics; Pasture Characteristics; Livestock Operation Management; Purpose of Livestock; Characteristics of Attacks; Methods for Deploying Attack-Reducing Strategies; Employed Methods and Their Features; Results (Effectiveness of the Implemented Methods); Discussion and Conclusions Drawn from Participants’ Experiences]. Summaries of site-specific responses are available in the Supplementary Materials S2.
All participants represented either an individual site or a cluster of sites (some represented many) with relevant information on the study topic. The questionnaire was shared with these experienced experts but did not constitute a classical “expert elicitation” involving opinions, projections, or conjectures. Instead, it served as a tool among experienced experts to gather and organize available information to standardize results. Participation signified consent, which was solicited and granted by all authors. With each 17-site cluster team listed as co-authors, anonymity was not required. An additional four known sites/site clusters were invited but did not participate.
The livestock types and classes that we included in our analyses were bovids (cattle and Asiatic water buffalo (Bubalus bubalis), hereafter buffalo), equids (horses, mules, donkeys), ovids, caprids, and suids (sheep, goats, and pigs, respectively). We did not include domestic dogs or birds. While many domestic dogs are lost to jaguars and some domestic birds, the management techniques to reduce those attacks and losses differ from those used for large grazing and browsing domestic herbivores. In contrast, there is cohesion among the practices deployed for the domestic animals that we focused on.
We extracted methods featuring clear efficacy metrics from the completed questionnaires to reduce encounters with jaguars and pumas from 194 livestock operations, representing 77% of the 248 operations analyzed. The remaining 23% (57) reported on feline attacks and their characteristics but did not provide well-defined metrics to evaluate the success of the implemented strategies. In the 194 operations that detailed the impacts of the interventions applied in the field, we investigated the specific aspects of decreased livestock losses resulting from these methods.
Only 24 livestock operations had sufficiently detailed information to allow for an examination of benefit/cost ratios as a cost-effectiveness test. We used data from six ranches in the Paraguayan Chaco [58], 16 in the Colombian Llanos [59], and two ranches in the Brazilian Pantanal to examine the ratio between the investments that ADS required and the benefits in terms of livestock losses averted. The ratio was obtained by dividing the economic benefit (the value of the livestock not preyed upon) by the intervention cost. In all these cases, the losses averted figures were based upon either before–after data (n = six Paraguayan ranches) or strict controls (n = sixteen ranches in Colombia and two in Brazil). A ratio greater than 1.0 indicated that the intervention was financially advantageous, meaning that the strategy generated an economic benefit that exceeded its cost. The total cost of implementing electric fences and night enclosures (and/or other techniques) was calculated individually for each property. The expenses included materials and labor for installation and maintenance. The economic benefit was assessed based on the value of the livestock that were no longer preyed upon after implementing the strategies. This calculation was made by considering the historical predation rates for those specific areas of each ranch, or control areas that experienced losses, and the market value of the livestock that were protected (not lost to depredation). More generalized evaluations of benefits yielded by interventions were available for the study area in Mexico’s Selva Lacandona [21], Nicaragua’s Mosquitia [60], and Casanare Llanos [59]. This study concentrated strictly on investments made in ADS and the returns yielded in livestock losses averted.
3. Results
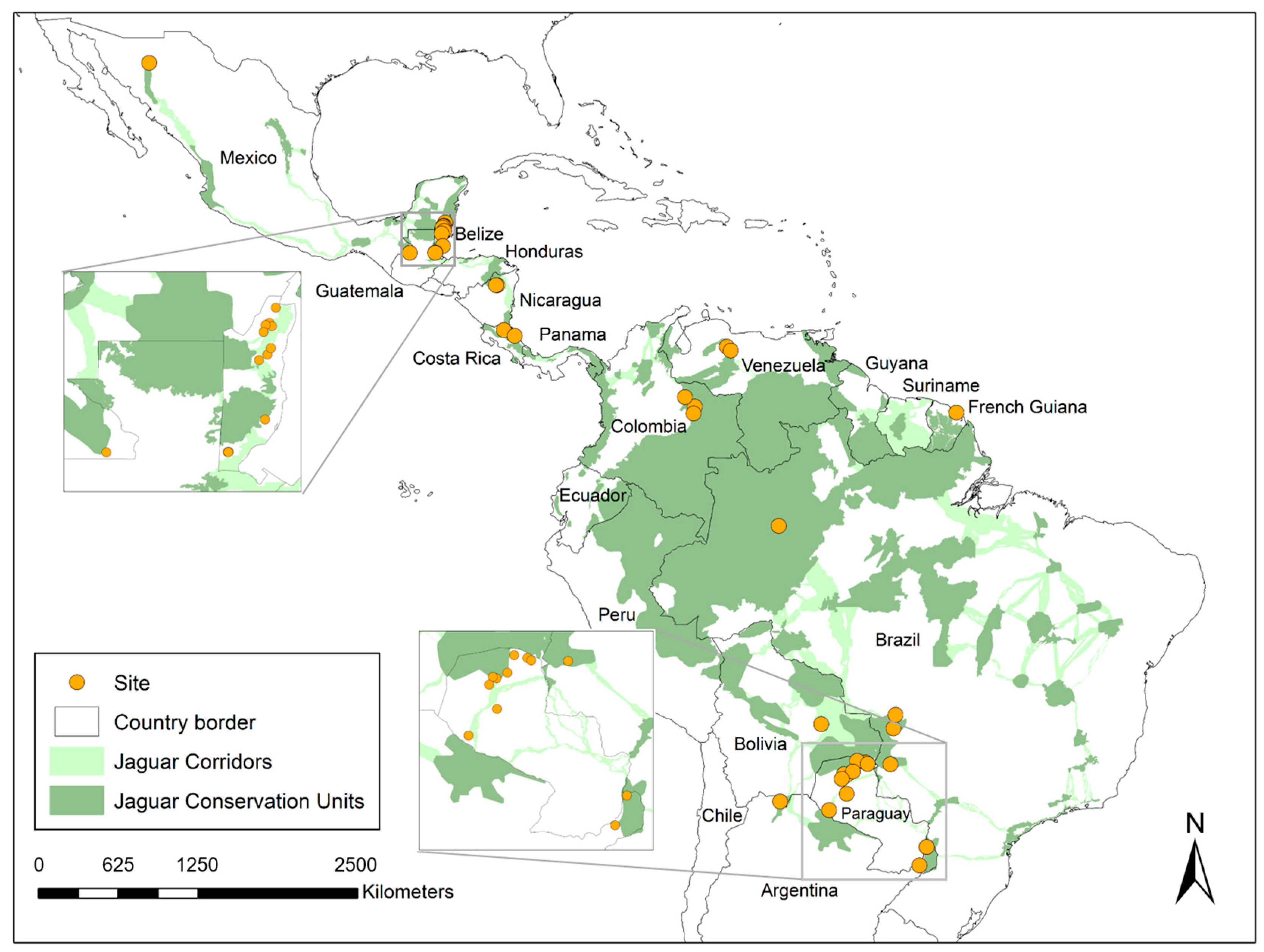
The questionnaires were completed in 11 countries (Figure 1: Mexico, Belize, Nicaragua, Costa Rica, Colombia, Venezuela, French Guiana, Bolivia, Brazil, Paraguay, and Argentina). A total of 45 individuals (including members from 17 site/site cluster teams) completed the questionnaire. Due to space constraints, only 30 individuals are listed as authors, while the remainder are acknowledged separately. Four more sites and site clusters were invited but did not fully engage. This included representatives from non-governmental organizations in Guatemala and Panama and two more ranches in Bolivia. No reasons were provided in those cases, but time constraints and/or data characteristics are inferred as likely reasons for not fully engaging. The livestock operations ranged from 2 hectares (ha) to 130,000 ha and five to 30,000 head of livestock (Figure 2a,b). This included private, Indigenous, and community livestock operations. We sampled several biomes, including the following: semi-arid Sonoran desert in northern Mexico; lowland and montane humid forests in southern Mexico, Belize, Nicaragua, and Costa Rica; Los Llanos (seasonally flooded lowland savanna–forest mosaics) in Colombia and Venezuela; Amazon and Guianan Shield forests (Brazil and French Guiana); Chaco (low stature tropical dry forest) in Paraguay; a Chaco-Chiquitania (higher stature semi-deciduous forest) transition zone in Bolivia; Yungas (montane forests) in Argentina; Atlantic Forest in Argentina; and the Pantanal (a seasonally flooded savanna–forest mosaic) in Brazil. A summary of reports is provided in Supplementary Materials S2. Using coarse categories, 11 intervention types were tested (Figure 3).
Figure 1.
Map of the study area, including the Jaguar Corridor (Jaguar Conservation Units, dark green; Jaguar Corridors, light green) with points indicating the general location of participating livestock operations (n = 248 total). The following eleven countries were represented within the dataset (listed alphabetically): Argentina (n = 26); Belize (n = 12); Bolivia (n = 1); Brazil (n = 86); Colombia (n = 22); Costa Rica (n = 13); French Guiana (n = 4); Mexico (n = 31); Nicaragua (n = 43); Paraguay (n = 9); and Venezuela (n = 1).
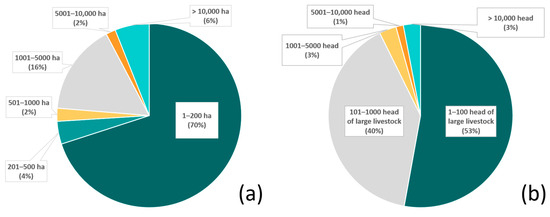
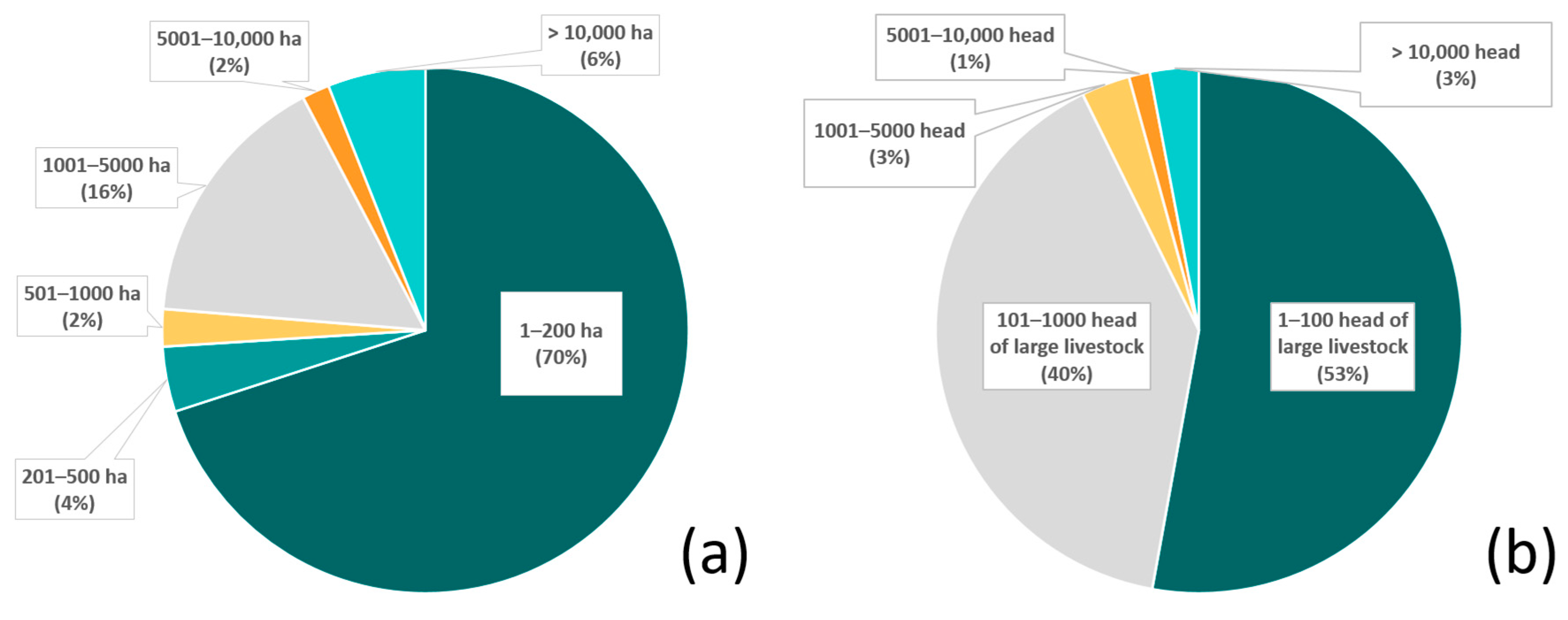
Figure 2.
The percentage of livestock operations included in the study classified by (a) property size (hectares, ha; data available for n = 176 total properties) and (b) herd size, estimated by number of head of large livestock (data available for n = 229 properties).
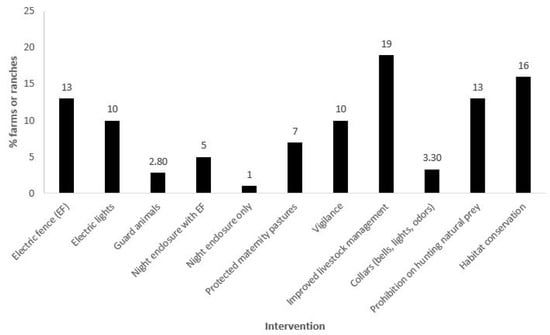
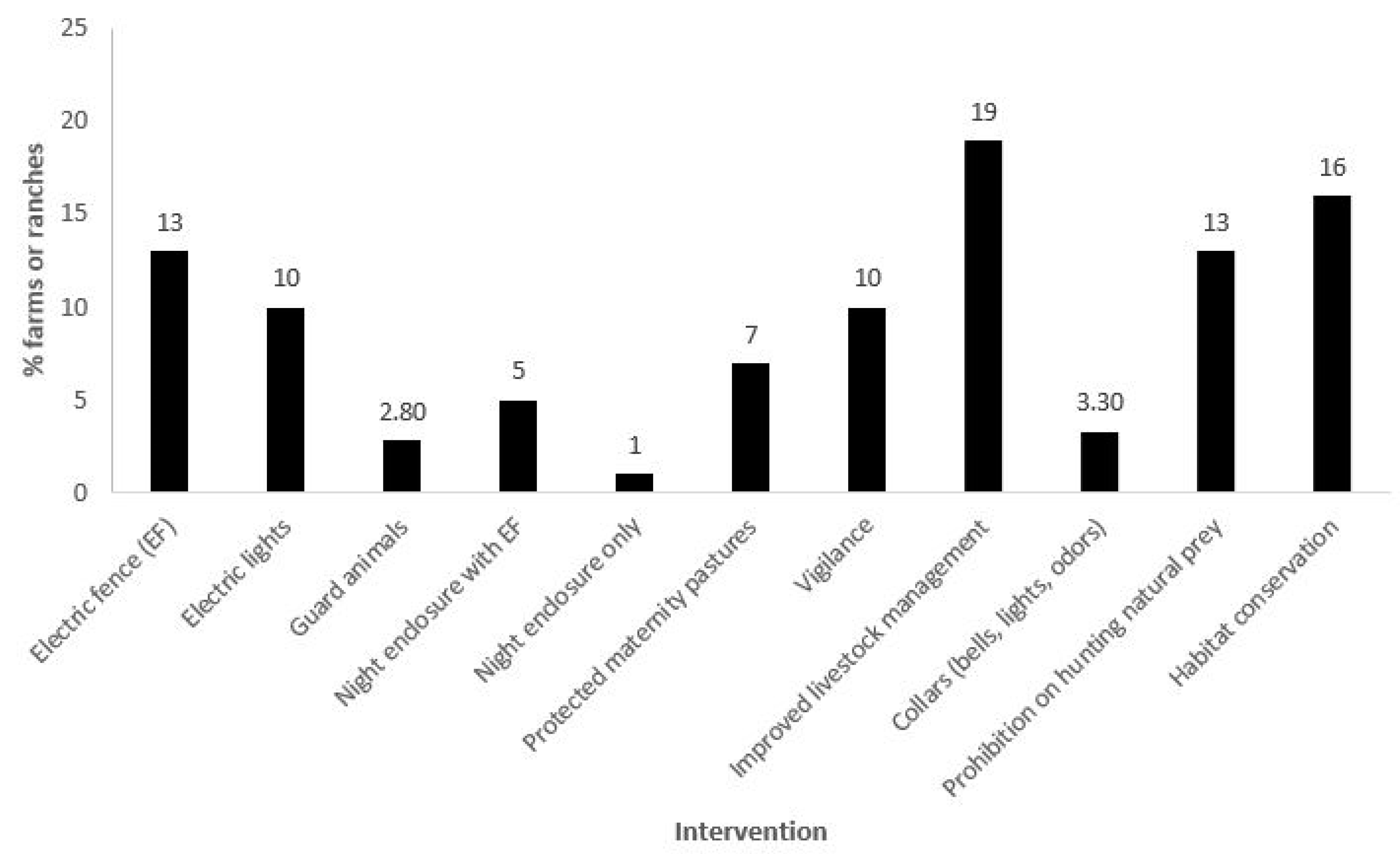
Figure 3.
The percentage of livestock operations (n = 248 total, independent of size and number of animals) included in the study that used at least one of the following 11 intervention types to mitigate jaguar/livestock conflict: electric fencing (EF); electric lights; guard animals (e.g., buffalo, creole cattle, donkeys, dogs); night enclosure with EF; night enclosure only; protected maternity pastures (or pasture for young livestock, near human dwellings); vigilance (day or night); improved livestock management (e.g., improved nutrition or herd health practices, excluding livestock from forested areas); equipping livestock with deterrence collars (e.g., bells, bells and lights, odors); prohibition on hunting of natural prey species; and habitat conservation.
Approximately 92% of the livestock operations were between 2 and 5000 ha in size; within those, 70% were between 2 and 200 ha (Figure 2a). Approximately 93% of the operations had between 5 and 1000 head of livestock, with 53% less than 100 head (Figure 2b). Although some operations utilized a single ADS, it was typical for multiple ADS to be deployed. Methods included tighter livestock management (19% of the livestock operations), bans on hunting of natural prey or restoration of natural prey (13%), electric fences (13%), electric lights (10%), increased vigilance (10%), a variety of special protections for young animals (7%), and night enclosures (6%), all of which totaled 78% of the methods deployed. The protection of natural habitats (which also preserve natural prey bases) was practiced by 16% of the operations. Approximately 6% had experimented with collars (with bells and or lights or odors) and guard animals (Figure 3).
Among the 194 livestock operations that measured the efficacy of the methods deployed, there were examples of successes from both the largest and smallest operations (Table 1). The largest operation in the study (30,000 head of cattle in 130,000 ha in the Brazilian Pantanal) deployed electrified night enclosures for mother cows and calves, electric lights in pastures (mothers and calves), bells on cows, fences that excluded livestock from the forest, a complete ban on hunting, and forested corridors for wildlife. On that ranch, depredation losses in areas without electric fences were 5.92 to 22.8 times greater than those where ADS were deployed. The smallest operations in the study (80% of herds under 25 head) were in remote Indigenous territories in the humid forests of the Nicaraguan Mosquitia. A combination of improved husbandry methods in these operations included improved nutrition/pastures, exclusion from the forest, enhanced vigilance, and a ban on hunting of key prey species; these interventions resulted in a 100% reduction in losses to depredation. In another remote setting with small Indigenous and community operations in Mexico’s Selva Lacandona (averaging 90 cattle and 50 sheep), night enclosures achieved a 97% reduction in losses of sheep, and electric-fenced maternity pastures achieved an 86% reduction in losses of vulnerable cattle classes.

Table 1.
Efficacy impact of anti-depredation strategies.
Techniques effectively reducing losses were documented across the entire operation size and biome spectrum (Table 1). We illustrate this with a few select examples. In the Chaco of Paraguay, combinations of methods (electric lights, electric fence, improved husbandry, exclusion from forests, cleaner pastures, controlled birthing seasons, secure maternity pastures) deployed among ranches ranging from 1000 to 38,627 ha and up to 17,500 cattle resulted in a 100% reduction in losses. In operations ranging from 101 to 500 cattle in the Colombian Llanos, combinations that included maternity paddocks (electric-fenced pastures for mother cows and calves), electric-fenced night enclosures for vulnerable young classes, exclusion from the forest, increased vigilance, strategic placement of pastures and water sources, a partial ban on hunting and deforestation, and creole Sanmartinero bulls introduced to Cebu herds resulted in a 100% reduction of losses in 14 ranches and, in six ranches, a reduction of losses less than 100% but ≥75%. In a large community herd of 1300 cattle across 80,000 ha in Argentina’s Yungas (montane forests), electric fences controlled depredation with 100% effectiveness, as did pastures with electric lights and near houses. In medium-sized operations in the lowland humid forests of Costa Rica (average 520 cattle), electric fences controlled (reduced by 100%) depredation on 12 ranches. In medium-sized operations located in the humid lowland forests of French Guiana, the effective use of electric fences resulted in a 98% reduction in losses (Table 1). In one medium-sized ranch (2994 ha) in the Chaco and Chiquitania ecotone in Bolivia, an increased proportion of buffalo, combined with electric fence, night corrals, and enclosures with electric lights, strategic placement of nutrition supplements, and a ban on hunting, resulted in substantial reductions in losses to jaguar attacks, with the ranch owner attributing a high proportion of the success to the use of buffalo. Electric fences in the Atlantic Forest of Misiones, Argentina, prevented 100% of jaguar depredations in three medium and large cattle properties (200 to 2500 head of cattle, 60–2500 ha) for over a decade [61].
Of 194 livestock operations with metrics on ADS effectiveness, only 24 provided sufficient data to assess the cost-effectiveness of the implemented interventions. Evaluations of the cost-effectiveness of the ADS on these 24 livestock operations across three countries (Paraguay, Brazil, and Colombia) found that benefits in livestock losses averted exceeded the investments required in 88% of the cases (Table 2). In 21% of the livestock operations (five of 24 total), benefits exceeded investment by more than ten times (11.6–22.1 times). In 17% (four of 24 total) of operations, benefits exceeded investments by 3.1 to 10.0 times. The highest proportion (50%) of operations experienced benefits between 1.0 and 3.0 times the investments. In 12% of operations, costs exceeded benefits, which was attributed to failing to apply the ADS consistently, with resulting abnormal losses skewing the results. An additional (non-measurable) benefit is the owners’/community’s ease of mind, knowing that their livestock is protected.

Table 2.
Benefit–cost analysis across three countries and biomes.
4. Discussion
ADS applications vary in complexity and require investment (see Table 3). In areas with challenging access, where roads and electricity are unavailable, the simplest and most effective strategies are enhancing animal husbandry, erecting fences, building night corrals, and maintaining the natural prey population (rows A, B, and C in Table 3). Introducing simple and affordable improvements in livestock management can increase productivity and reduce the chances of jaguar and puma attacks (refer to Table 1 and Table 3); evidence from Nicaragua supports this (discussed below). Efforts to improve livestock management and education at 43 farms, accessible only after two days of river travel, resulted in zero losses to jaguars during the project [60]. This level of simplicity may be suitable—possibly the only feasible choice—for more secluded areas like the Mamirauá Sustainable Development Reserve in the west-central Amazon (S2), as well as parts of the Upper Amazon and remote regions of the Guianan Shield. However, a collaborative process with local communities is essential to align interventions with their practices and priorities. Incorporating community perceptions and attitudes toward jaguars enhances the relevance and sustainability of coexistence strategies while fostering local stewardship. A participatory approach with technical experts and livestock producers working together will address logistical constraints while creating solutions that resonate with community needs and expectations. While improved livestock management can minimize losses, the best results typically arise from integrating various forms of ADS at multiple levels.

Table 3.
Intervention types along the investment and complexity spectrum.
We noted that participating livestock operations that reported fewer losses implemented multiple ADS concurrently. Enhanced livestock management became a central theme, with intensity variations linked to available resources and local expertise. Furthermore, although veterinary care was quite advanced in numerous operations, some still required its assistance. The overall standard of quality in animal husbandry improved through specific ADS. The broader aspect of ADS, quality animal husbandry, was complemented by more specific elements; the combinations differed (see Table 1), but the results were consistently positive throughout (see Table 1 and Table 3). In the analysis of 24 livestock operations regarding benefit-to-cost ratios (refer to Table 2), 38% demonstrated that benefits were more than three times the costs, and 50% had benefits ranging from one to three times the costs. Together, these amount to 88% overall. In instances where costs surpassed benefits, the predominant reason for failure was the inconsistent implementation of ADS, leading to corresponding livestock losses.
Our findings, supported by Table 1 and Table 2, show that ADS are very effective, often exceeding their costs, sometimes by a significant margin, when evaluating financial benefits. De la Torre et al. (2021) [62] found that the benefit–cost ratios for 11 livestock operations in the Selva Lacandona of southern Mexico varied between 1.2 and 26.6. They noted that the benefits were more than double the costs in nine of the 11 operations (82%). The results from de la Torre et al. (2021) [62] were not included in Table 3 because their analysis considered a more comprehensive range of factors that needed to match the more focused scope of our analysis in this paper. A key takeaway from the de la Torre et al. (2021) [62] study is that ADS methods beyond improved husbandry, such as electric fences, could be successfully implemented even in a remote region. The authors attributed the project’s success to a thorough strategy that included outreach, education, robust community engagement, and animal care and livestock management improvements, complemented by persistent supervision and focused ADS techniques. The emphasis on a mixed strategy, advocated by a team working with small farms (where an average flock is 30 sheep and cattle herds average 90), was similarly evident in the largest operation in our sample. This operation, run by Instituto Homem Pantaneiro, managed 130,000 hectares with 30,000 livestock (see Table 1 and Table 2, and Supplementary Materials S2). Although the methods varied by location and operation, 93% of the participating sites indicated using multiple ADS within the same operations. The mixture of tools differed, which is appropriate for this discussion, and within the broader context of biological and carnivore conservation, where the choice of tools and their application intensity must be tailored to the specific characteristics of each site, owner, and situation.
Although tourism was not highlighted as a strategy against depredation—since it is not one—there are instances where it can significantly contribute to human–jaguar coexistence. Some livestock operations featured in this study incorporated tourism into their business models. This situation was observed during two operations in Brazil’s Pantanal, one in Bolivia’s Chiquitania/Chaco, two in Colombia’s Llanos, and one in Brazilian Varzea (seasonally flooded Amazon forest). Tortato et al. (2017) [63] discussed the benefits of jaguar-focused tourism in the Northern Pantanal, while studies have shown that tourism supports jaguar survival in some areas of Venezuela’s Llanos [64,65,66] when tourism emphasizes the importance of jaguars for ranches, communities, or regions, tolerance increases, coexistence improves, and jaguars become assets, instead of being liabilities [64]. In the Mamirauá Reserve in the Brazilian Amazon, community-based tourism and research activities have positively impacted human–jaguar relationships. Interviews conducted between 2020 and 2021 revealed that areas with these initiatives exhibited higher tolerance toward jaguars, more positive attitudes, and lower intentions to kill them [67].
Electric fences have become an effective tool widely used in various situations. While specific features—like length, number of strands, strand height, voltage, and power source—differ by location, 87% of sites that reported on ADS’s effectiveness (Table 1), used electric fences. These fences primarily target pastures and nighttime enclosures containing young animals alongside mother cows and calves, as these livestock areas and age classes are particularly vulnerable to predation [34,44]. A possible barrier to using electric fences is the limited expertise in rural areas and the associated maintenance costs. These issues can be addressed by involving specialized companies to train potential clients, along with support from technicians from government entities and NGOs. Electric fences offer extra advantages, including improved herd management via subdivided grazing areas, which allows for the creation of new paddocks and expanded rotation options. They can also provide energy supply possibilities for low-income households, enabling families to utilize the same energy sources that power the fences [59]. The implementation of electric lights yielded positive outcomes (see Table 1). However, experienced participants observed that jaguars are likely to adapt to the sensory disruptions caused by these lights and recognize that they do not present any genuine physical threat or discomfort. This contrasts sharply with an electric shock’s deterrent and repellent effects. If lights stay on for long durations, then their benefits can diminish, especially in areas with tourism-habituated jaguars. Therefore, it is advisable to utilize them for shorter spans, such as two months (sixty days), which corresponds to the peak vulnerability period for calves [34]. Observations and diagrams of electric fences can be found in Supplementary Materials S3.
We observed maintenance challenges related to electric fences in southern Mexico, French Guiana, Colombia, Venezuela, and Brazil, as discussed in Supplementary Materials S2. Generally, the solution hinges on the scale of electric fence installation, further justifying their use primarily during the most vulnerable phases of livestock. In this research, the most extensive pastures secured by electric fences measured around 100 ha. Cost comparisons between traditional and electric fences revealed some variations. In Colombia, traditional fences are priced at three times the cost of electric fences, while the differences in other areas are negligible.
In contrast, electric fences in French Guiana are more expensive than traditional options. Although there are some maintenance challenges, there is a general consensus that electric fences are effective and preferred deterrents. This strategy has the potential to significantly expand in various areas, as it safeguards at-risk age groups and livestock stages. Additionally, many maternity pastures and night enclosures are situated close to homes, providing an extra deterrent and enabling constant monitoring. This setup is recommended whenever possible.
The use of depredation-resistant livestock, such as criollo cattle and buffalo, has mainly yielded favorable results, particularly in Bolivia, Colombia, and Costa Rica. It is essential for field workers handling criollo cattle, and especially buffalo, to have time to adjust to new husbandry practices. Moreover, a market for these alternatives to traditional beef should be developed, or owners must be aware of related market trends. The low prices of buffalo meat are less of a concern because the performance of the meat carcass and the higher productivity of buffalo offset these lower market prices. While both criollo cattle and buffalo show remarkable resilience against depredation, there are situations in which neither species is entirely safe from jaguar attacks. In the Venezuelan silvopastoral systems, young buffalo were depredated nearly as often as cattle, mainly when the weaned buffaloes were managed without adult guard buffaloes. As herds mature and gain experience, the animals and their owners can adopt effective methods while viable product markets emerge, producing positive results. Buffaloes also present advantages due to their impressive milk production in tropical environments. Items like mozzarella cheese, “dulce de leche” (a dessert created with buffalo milk), and ricotta are greatly valued in local, national, and international markets throughout these countries.
A fundamental principle of ADS is minimizing the interaction between domestic animals and predators, aiming to “keep livestock away from forests and shared water sources.” This strategy includes suids (pigs), sheep and goats, bovids (cows and buffalo), and equids (horses) [16,52,59]. Experiences from Nicaragua highlight the irregular presence of shade trees, varying forage banks, and nitrogen-fixing living fences in silvopastoral systems. In contrast, agroforestry and silvopastoral practices in Venezuela focus on a system that enables cattle and buffalo to graze within forest plantations. Despite the advantages, these systems face unique challenges.
Nonetheless, using electric fences combined with lighting has significantly reduced livestock losses, even in these difficult situations (Table 1). A fundamental aspect of wildlife conservation is maintaining a robust natural prey population to offer jaguars alternatives to livestock [68]. Game species are intricately linked to jaguar prey [32]. Despite being crucial for reducing depredation [34], only 73% of our sites implemented explicit, partial, or total hunting bans on natural prey. Addressing this issue is vital for creating effective ADS systems despite the challenges.
There is frequently a division between special interests, such as biologists, who focus on forests, carbon capture, mitigating climate change, and biodiversity conservation, and those who do not necessarily see all livestock-rearing practices conflicting with these goals. This divide may arise from personal experiences or entrenched beliefs. People who have primarily lived in areas with mostly uniform forests, suffering significant damage from poorly managed and ever-expanding livestock operations, frequently view all ranching as detrimental to the environment. Conversely, individuals who have participated, resided, and worked within well-managed livestock systems that successfully maintain species diversity, forests, and watersheds understand that achieving a balance is challenging yet feasible, as demonstrated [15,69,70]. For example, in this study, forest cover made up roughly 46% of the land across nine ranches in Paraguay, ranging from 4000 to 45,000 ha, which supported between 1100 and 19,800 head of cattle. In the Llanos of Venezuela, Polisar et al. (2003) [34] noted that forests constituted 50% of the area studied, while in the three Pantanal ranches included in this research, at least 40% of the area consisted of protected riverine forests or forested savannas, with cattle reared chiefly on native grasses.
Approximately 53% of our sites feature specific provisions to conserve forests and habitats, encompassing mechanisms such as conservation agreements. This figure increases to nearly 67% when considering inferred habitat conservation related to loosely enforced state bans or proximity to nationally protected areas. This consideration requires careful balance. Pastures too close to forests, or small pastures within them, can lead to increased depredation rates, as jaguars prefer forest edges and avoid open spaces [33,49,71]. Enhancing the nutrition and health of livestock in a specific area is a way to reduce the need for pasture expansion and subsequent deforestation. Nonetheless, a clear link between higher livestock productivity per unit area—thanks to improved animal husbandry (including nutrition and health)—and a reduction in deforestation due to pasture expansion has been infrequently documented, even though it is feasible [59,60].
Protecting forests and habitats is essential for maintaining healthy prey populations and watersheds. It also provides jaguars with both land and water resources for their diet. The concept is that well-preserved forests and water bodies, devoid of cattle, form ideal habitats for prey, thereby supplying jaguars with the natural nutrition they need. Effectively managed and preserved forests can help to create distinct areas for jaguars away from livestock, reinforcing a previously discussed principle. To boost jaguar conservation, there is a pressing need for enhanced national-, local-, and ranch-level planning that prioritizes forest connectivity and pasture management. Many operations within this region are currently adopting these strategies. When appropriately executed, cattle ranching can contribute to forest conservation efforts [19]. Privately managed ranches and Indigenous lands, frequently well-protected, can sometimes surpass public protected areas in biodiversity conservation, as demonstrated by Devlin et al., 2023 [72].
To grasp the interactions between jaguars and livestock, it is crucial to observe both (1) jaguars to livestock and (2) livestock health on farms, ranches, and community operations. Throughout all study operations, jaguars were monitored along with pumas and sometimes feral dogs, utilizing a blend of camera traps and various signs, such as tracks, scrapes, feces, and kill traits. Moreover, one site implemented linear foot transects in addition to the previous methods. Livestock monitoring showed significant variation, which is anticipated due to the range of operations from around 5 hectares to 130,000 ha and between 30 and 30,000 animals. Yet, even if access and scale pose challenges, livestock monitoring is essential to the ADS suite [52,59]. In this study, nearly all operations conducted regular livestock inventories, although the frequency varied,ranging from weekly or biweekly to monthly or even once or twice a year. Dairy cattle are monitored daily during production. Holding inventories annually or bi-yearly cattle roundups may be insufficient for effectively tracking livestock conditions, including theft, losses, and gains, and supporting adaptive management strategies. One participating operation or group did not have a formal inventory system. Another country noted that only 2 out of 12 (17%) livestock operations performed regular inventories. This practice is an essential first step in monitoring livestock health, identifying the causes of mortality, and pinpointing areas within the operation where mortality rates are elevated, allowing for differentiation between causes such as predation that require attention. Interventions are most effectively evaluated when used alongside regular livestock inventories [73].
In developing this study, we asked about local involvement, the participation of farms and ranches, and methods to expand efforts to affect broader regions of jaguar habitat. We recognize that ADS initiatives are underway and are being assessed in almost all countries where jaguars are found, yet the percentage of land they cover remains likely relatively small. Local project engagement varied by scale. Some authors in this paper focused directly on ranches (Bolivia 1, Brazil 3, Venezuela 1). Other contributors, however, detailed significant involvement at both the local and farm levels, which includes (1) providing direct technical assistance; (2) organizing capacity-building workshops; (3) conducting ongoing communications and evaluations; (4) ensuring timely responses and actions when needed; (5) fostering strong ties with local communities to guarantee that jaguar conservation generates broader local benefits; and (6) encouraging jaguar-focused tourism when feasible, yielding specific local benefits. Practical approaches and cost-efficient interventions at the local level have proven to be the most compelling “advertisement”. In this respect, Costa Rica developed an exemplary national model of an ADS implementation program, The UACFel initiative, between a local NGO and government organizations, as described by Corrales et al. 2016a,b [74,75].
Building and maintaining relationships are crucial for success at every level. Projects that utilize years of collaboration can significantly benefit from these established connections. New initiatives should develop these relationships; nothing strengthens them more than reliable execution that meets commitments. Many participants noted that project performance affected farmers’ and ranchers’ perceptions of predators like jaguars. Improvements in livestock management, resulting in increased productivity, also led to better acceptance and should be integrated into any ADS projects to be carried out.
5. Conclusions
The best approach to improve the uptake of ADS is to showcase proven efficacy. We offer compelling evidence of the effectiveness of interventions across multiple operations and biomes, along with cost-effectiveness data for select interventions. While our summary includes specific examples spanning sixty degrees of latitude, we emphasize the importance of monitoring all ADS interventions to assess their efficiency and cost-effectiveness. Disseminating these findings will encourage broader engagement and overall uptake. We recommend active collaboration with cattlemen’s associations, publishing articles in local and national newspapers and online platforms, speaking at scientific and agricultural conferences, and organizing meetings with government agency staff and elected officials. While accurate information is crucial, it must be communicated effectively to decision-makers at every level—from local farms to the national stage—to drive change. Delivering the message in venues where it will reach the desired audience is crucial, be it a community center or a government office.
Challenges to implementation and evaluations, particularly in remote regions, may include (1) the lack of dependable channels for citizens to report issues and communicate needs to authorities or NGOs; (2) inadequate routine livestock inventories; (3) the absence of standardized reporting formats, or inconsistencies in timeliness, which can obstruct effective changes; (4) a deficiency in both government and cattlemen associations’ readiness, either fully or partially, to address these issues, alongside a lack of awareness about the tools available and inadequate organization to effectively promote and utilize these resources effectively for a broader impact. All these challenges can be overcome. We stress the importance of increasing the government awareness of and promoting these proven techniques. We urge governments within the jaguar range to enhance their understanding and embrace these efficient, cost-effective strategies.
The Jaguar 2030 Roadmap for the Americas [19] aims to improve jaguar conservation across the species’ range through the following four interconnected strategies: (1) coordinating efforts across the range to protect and link jaguars and their conservation landscapes; (2) implementing national jaguar conservation strategies; (3) significantly expanding the use of sustainable development tools in Jaguar Conservation Units (JCUs) and Corridors, including trans-boundary landscapes; and (4) achieving financial sustainability for jaguar conservation efforts. Pathway 3 emphasizes the importance of innovative strategies to reduce human–wildlife conflict, involving local communities and livestock operators through participatory methods, as detailed in this study. CITES Decision 19 has formalized the Jaguar Roadmap, requiring collaboration among Parties and the UN Convention on Migratory Species (CMS) to establish a Joint Program of Work, a Rangewide Monitoring Framework, and an Intergovernmental Platform [76]. Furthermore, jaguars are recognized as indicators of biodiversity, and preserving these apex predators is seen as an effective strategy for range states to fulfill their obligations under the UN Convention on Biological Diversity’s (CBD) Kunming-Montreal Global Biodiversity Framework [77], particularly in achieving Goal A, Target 4. The pragmatic human–jaguar coexistence tools analyzed here can contribute to the long-term persistence of jaguar populations. The benefits generated also aim for a larger scale. Jaguar conservation focuses on an apex predator and preserving the ecosystems it inhabits. Big picture jaguar conservation seeks a balance between human needs and the persistence of the natural world, stabilizing ecological life support systems that include watersheds, climate, and biological diversity. The tools that this study presents can contribute to that balance.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/ani15091247/s1: Structured Questionnaire-011152024, S2: Summarization 1st parts questionnaire 01122025, S3: Observations, diagrams, and comments on anti-depredation electric fences + lights 01122025 Final, S4: Broad acknowledgements Team x Team.
Author Contributions
Supervision: J.P. Conceptualization, J.P., R.H., A.H. and A.L.D. Data curation, J.P., R.H., D.F.P.V., S.J.C.V., C.V.V., A.L.D., R.J.A.H., M.B., F.B., I.C.G., C.Y.C., D.C.G., J.A.D.l.T., W.D.T.-O., A.K., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., G.E.d.O.P., M.F.P.C., P.R., Y.P.S., C.S.-Z., T.R. and A.Z. Formal analysis, J.P., R.H., A.H., D.F.P.V., S.J.C.V., C.V.V., J.A.D.l.T. and C.S.-Z. Funding acquisition: R.H., D.F.P.V., C.V.V., M.B., I.C.G., D.C.G., J.A.D.l.T., W.D.T.-O., A.K., N.L.O., P.G.P., T.R. and L.V. Investigation, J.P., R.H., A.H., D.F.P.V., S.J.C.V., C.V.V., A.L.D., R.J.A.H., M.B., F.B., I.C.G., C.Y.C., J.A.D.l.T., W.D.T.-O., A.K., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., G.E.d.O.P., M.F.P.C., P.R., Y.P.S., T.R., L.V. and A.Z. Methodology, J.P., R.H., A.H., D.F.P.V., S.J.C.V., C.V.V., A.L.D., R.J.A.H., M.B., F.B., I.C.G., C.Y.C., D.C.G., J.A.D.l.T., W.D.T.-O., A.K., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., G.E.d.O.P., M.F.P.C., P.R., Y.P.S., C.S.-Z., T.R., L.V. and A.Z. Project administration, J.P., R.H., D.F.P.V., C.V.V., R.J.A.H., M.B., J.A.D.l.T., D.L., N.L.O., C.A.M.A., R.D.O.-H., P.G.P., P.R. and L.V. Resources, R.H., D.F.P.V., A.L.D., R.J.A.H., M.B., I.C.G., A.K., F.B., C.Y.C., D.C.G., J.A.D.l.T., W.D.T.-O., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., M.F.P.C., P.R., Y.P.S., T.R., L.V. and A.Z. Validation: J.P., R.H., A.H., D.F.P.V., C.V.V., A.L.D., R.J.A.H., M.B., F.B., I.C.G., C.Y.C., D.C.G., J.A.D.l.T., W.D.T.-O., A.K., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., G.E.d.O.P., M.F.P.C., P.R., Y.P.S., C.S.-Z., T.R., L.V. and A.Z. Visualization, A.H., D.F.P.V., S.J.C.V. and A.L.D. Writing—original draft, J.P., R.H. and A.H. Writing—review and editing, J.P., R.H., A.H., D.F.P.V., S.J.C.V., C.V.V., A.L.D., R.J.A.H., M.B., F.B., I.C.G., C.Y.C., D.C.G., J.A.D.l.T., W.D.T.-O., A.K., D.L., N.L.O., D.M., C.A.M.A., R.D.O.-H., P.G.P., G.E.d.O.P., M.F.P.C., P.R., Y.P.S., C.S.-Z., T.R., L.V. and A.Z. All authors have read and agreed to the published version of the manuscript.
Funding
Argentine Yungas: WildCRU, Born Free Foundation, Robertson Foundation, Ley Nacional de Bosques Nativos (N° 26.331), Small Grants Program of GEF (PPD-GEF-PNUD) and Proyectos de Conservación y Desarrollo con comunidades indígenas (PROFOCI). Belize: Panthera, World Wildlife Fund, Ya’axché Conservation Trust. Brazilian Atlantic Forest/Projeto Onças do Iguaçu: National Geographic Society, WW Brazil, Ron Magil Conservation Endowment, Beauval Nature. Brazilian Pantanal—Fazenda Jofre Velho: Panthera NY, Panthera Brasil, Rainforest Trust, Duncan & Ellen McFarland, Edith McBean, Ross and Patricia Beaty, Larry Westbrook, Datamars, Belgo-Bekaert. Brazilian Pantanal (Instituto Homem Pantaneiro): BRPEC Agropecuária S/A, General Motors South America. Brazilian Várzea (flooded forest): Mamirauá Institute for Sustainable Development, Brazilian Ministry of Technology and Communications, Brazilian National Council for Research and Development (CNPq)—scholarship for Wezddy Del Toro-Orozco. Colombian Llanos: Panthera, U.S. Agency for International Development (USAID)—Programa Riqueza Natural, U.S. Fish and Wildlife Service—USFWS, Action Fund (Tropical Forest Conservation Act—TFCA), The National Learning System (SENA (Spanish acronym)), WebConserva. Costa Rica: Tortuguero and Maquenque: Panthera, I Debt Relief for Nature, EEUU-CR. French Guiana—HISA: French Biodiversity Office (OFB), the Ministry for Ecological Transition and Territorial Cohesion (MTECT), represented by the Prefect of French Guiana and, by delegation, the Directorate-General for Territories and the Sea of French Guiana (DGTM), the Territorial Collectivity of French Guiana (CTG), and the Office for the Development of the Overseas Agricultural Economy (ODEADOM). Mexico north (Sonora): Rufford Foundation (RSG reference 14291-1), Summerlee Foundation, CONACYT, Small Wild Cat Conservation Fund, Pope and Young Club, Sea World and Busch Gardens Conservation Foundation. Mexico south (Selva Lacandona—Jaguares de la Selva Maya Program): National Geographic Society’s Big Cat Initiative Program, Rufford Foundation Small Grants, the Conservation Program for Endangered Species (PROCER-Mexico) of the National Commission of Protected Areas (CONANP-Mexico), Natura y Ecosistemas Mexicanos, Latin American Regional Program of the US Fish & Wildlife Service. Nicaraguan Mosquita: Darwin DEFRA 23-014, Liz Claiborne and Art Ortenberg Foundation, U.S. Fish and Wildlife Service Neotropical Migratory Bird Conservation Fund, American Bird Conservancy. Paraguay: Wildlife Conservation Society (WCS), USAID Project Alliance for Sustainable Development, USFWS “constructing a future for the jaguar in the Paraguayan Chaco”. Venezuelan Llanos: Forestry Development (a private business) San Carlos, S.A. (DEFORSA), the non-governmental organization SEBRABA Project.
Institutional Review Board Statement
This study was approved by following the ethical procedures and protocols by the Zamorano University Department of Environment and Developmenton 27 February 2025.
Informed Consent Statement
Informed consent was obtained from all study authors. At least one author represents every individual area (of seventeen), whether one livestock operation or over 40. All authors understood the responsibilities and implications and provided consent.
Data Availability Statement
None were deposited in an official repository. Data can be provided upon written consent directed to the corresponding author.
Acknowledgments
Many thanks for the significant contributions of Emiliano Ramalho (Brazilian Varzea-Mamiraua); Emma Sanchez (Belize); Roberto Salom (Costa Rica); María Belén Ortiz (Paraguayan Chaco), Flavio Caruso (Argentine Yungas); Carlos Samoyoa and Paulina Arroyo Gerala (Selva Lacandona, México); Joāo Leite de Souza and Santiago José de Jesús Matute Castro (Venezuelan llanos); Thiago Reginato, Patricia Gomez, Yara Barros, Vania Cristina Foster, and Valquíria Marina Nascimento (Brazilian Atlantic Forest-Iguacu); Joao Losano Eubank Campos Jr., Mariana Queiroz, Geovani Vinco Tonolli, and Wener Hugo Arruda Moreno (Brazilian Pantanal). Broader site-specific acknowledgments can be seen in Supplementary Materials S4. This paper is dedicated to all who gave their lives, whether in New York, Idaho, Washington, Montana, Alberta, Kenya, India, Venezuela, or Brazil, on the carnivore conservation trail so the world can be a better place. The trail was cut by remarkable friends and colleagues who are no longer with us. Special thanks go to Peter Crawshaw, Howard Quigley, and Alan Rabinowitz, who lit sparks decades ago that encouraged others to excel. It has been twenty-five years since the first Jaguar Range-wide Priority Setting Exercise was held in México. We know that coexistence with carnivores is possible, especially with jaguars.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Lamb, C.; Ford, A.; McLellan, B.; Proctor, M.; Mowat, G.; Ciarniello, L.; Nielsen, S.; Boutin, S. The ecology of human–carnivore coexistence. Proc. Natl. Acad. Sci. USA 2020, 117, 17876–17883. [Google Scholar] [CrossRef] [PubMed]
- Ripple, W.; Estes, J.; Beschta, R.; Wilmers, C.; Ritchie, E.; Hebblewhite, M.; Berger, J.; Elmhagen, B.; Letnic, M.; Nelson, M.; et al. Status and ecological effects of the world’s largest carnivores. Science 2014, 343, 1241484. [Google Scholar] [CrossRef]
- Acharya, K.; Paudel, P.; Jnawali, S.; Neupane, P.; Köhl, M. Can forest fragmentation and configuration work as indicators of human–wildlife conflict? Evidence from human death and injury by wildlife attacks in Nepal. Ecol. Indic. 2017, 80, 74–83. [Google Scholar] [CrossRef]
- Castaño-Uribe, C.; Lasso, C.; Hoogesteijn, R.; Diaz-Pulido, A.; Payán, E., II. Conflictos Entre Felinos y Humanos en América Latina; Instituto de Investigación de Recursos Biológicos Alexander Von Humboldt (IAvH): Bogotá, Colombia, 2016; 489p. [Google Scholar]
- Dharaiya, N.; Bargali, H.; Sharp, T. Melursus ursinus. In The IUCN Red List of Threatened Species; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2016. [Google Scholar]
- Packer, C.; Shivakumar, S.; Athreya, V.; Craft, M.; Dhanwatey, H.; Dhanwatey, P.; Gurung, B.; Joshi, A.; Kushnir, H.; Linnell, J.; et al. Species-specific spatiotemporal patterns of leopard, lion and tiger attacks on humans. J. Appl. Ecol. 2019, 56, 585–893. [Google Scholar] [CrossRef]
- Terborgh, J.; Estes, J. Trophic Cascades: Predators, Prey, and the Changing Dynamics of Nature; Island Press: Washington, DC, USA, 2010. [Google Scholar]
- Barry, J.; Elbroch, L.; Aiello-Lammens, M.; Sarno, R.; Seelye, L.; Kusler, A.; Quigley, H.; Grigione, M. Pumas as ecosystem engineers: Ungulate carcasses support beetle assemblages in the Greater Yellowstone Ecosystem. Oecologia 2019, 189, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Braczkowski, A.; O’Bryan, C.; Stringer, M.; Watson, J.; Possingham, H.; Beyer, H. Leopards provide public health benefits in Mumbai, India. Front. Ecol. Environ. 2018, 16, 176–182. [Google Scholar] [CrossRef]
- Elbroch, L.; O’Malley, C.; Peziol, M.; Quigley, H. Vertebrate diversity benefitting from carrion provided by pumas, and other subordinate apex felids. Biol. Conserv. 2017, 21, 123–131. [Google Scholar] [CrossRef]
- Polisar, J.; de Thoisy, B.; Rumiz, D.; Díaz-Santos, F.; McNab, R.; Garcia-Anleu, R.; Ponce-Santizo, G.; Arispe, R.; Venegas, C. Using certified timber extraction to benefit jaguar and ecosystem conservation. Ambio 2017, 46, 588–603. [Google Scholar] [CrossRef]
- Sunquist, M.; Sunquist, F. Wild Cats of the World; The University of Chicago Press: Chicago, IL, USA, 2002; 462p. [Google Scholar]
- Woodroffe, R. Predators and People: Using Human Densities to Interpret Declines of Large Carnivores. Anim. Conserv. 2000, 3, 165–173. [Google Scholar] [CrossRef]
- Thirgood, S.; Woodroffe, R.; Rabinowitz, A. The impact of human-wildlife conflict on human lives and livelihoods. In People and Wildlife, Conflict or Coexistence; Woodroffe, R., Thirgood, S., Rabinowitz, A., Eds.; Cambridge University Press: Cambridge, UK, 2005; pp. 13–26. [Google Scholar]
- Hoogesteijn, A.; Febles, J.; Hoogesteijn, R. Seasonally Flooded Savannas of South America: Sustainability and the Cattle-Wildlife Mosaic. In Sustainability and the Rights of Nature in Practice; La Follette, C., Maser, C., Eds.; CRC Press; Taylor & Francis Group: Boca Raton, FL, USA, 2020; pp. 205–238. [Google Scholar]
- Polisar, J.; Matthews, S.; Sollman, R.; Kelly, M.; Beckmann, J.; Sanderson, E.; Fisher, K.; Culver, M.; Núñez, R.; Rosas-Rosas, O.; et al. Protocolo Para las Técnicas de Investigación y Monitoreo de Jaguares. Una Propuesta para el Servicio de Pesca y Vida Silvestre de EEUU como Cumplimiento Parcial del Contrato F13PX01563. 2014, p. 167. Available online: https://library.wcs.org/en-us/Scientific-Research/Research-Publications/Publications-Library/ctl/view/mid/40093/pubid/DMX2816900000 (accessed on 20 April 2025).
- Sanderson, E.; Chetkiewicz, C.; Medellin, R.; Rabinowitz, A.; Redford, K.; Robinson, J.; Taber, A. Un análisis geográfico del estado de conservación y distribución de los jaguares a través de su área de distribución. In El Jaguar en el Nuevo Milenio; Medellin, R., Equihua, C., Chetkiewicz, C., Crawshaw, P., Rabinowitz, A., Redford, K., Robinson, J., Sanderson, E., Taber, A., Eds.; Fondo de Cultura Económica: Mexico City, Mexico, 2002; pp. 551–600. [Google Scholar]
- Zeller, K. Jaguars in the New Millennium, Data Set Update: The State of the Jaguar in 2006; Wildlife Conservation Society: Bronx, NY, USA, 2007; 77p, Available online: https://scholar.google.com/citations?view_op=view_citation&hl=en&user=mjtNMbgAAAAJ&citation_for_view=mjtNMbgAAAAJ:u-x6o8ySG0sC (accessed on 20 April 2025).
- Jaguar-2030-Coordination-Committee Jaguar 2030. Conservation Roadmap for the AmericasUNDP, UNEP, UNODC, CITES, CMS, Panthera, WCS, WWF, Jaguar Range States: New York, NY, USA, 17 November 2024. 2022, p. 91. Available online: https://www.internationaljaguarday.org/publication/2020/10/22/jaguar-2030-roadmap-for-the-americas (accessed on 20 April 2025).
- Jędrzejewski, W.; Morato, R.; Negrões, N.; Wallace, R.; Paviolo, A.; de Angelo, C.; Thompson, J.; Paemelaere, E.; Hallett, M.; Berzin, R.; et al. Estimating species distribution changes due to human impacts: The 2020’s status of the jaguar in South America. Cat News Spec. Issue 2023, 16, 44–55. [Google Scholar]
- de-la-Torre, J.; González-Maya, J.; Zarza, H.; Ceballos, G.; Medellín, R. The jaguar’s spots are darker than they appear: Assessing the global conservation status of the jaguar Panthera onca. Oryx 2017, 52, 300–315. [Google Scholar] [CrossRef]
- Sanderson, E.; Redford, K.; Chetkiewicz, C.; Medellin, R.; Rabinowitz, A.; Robinson, J.; Taber, A. Planning to save a species: The jaguar as a model. Conserv. Biol. 2002, 16, 58–72. [Google Scholar] [CrossRef]
- Swank, W.; Teer, J. A proposed program for sustained jaguar populations. In Felinos de Venezuela, Biología, Ecología y Conservación; FUDECI: Caracas, Venezuela, 1992; pp. 95–107. [Google Scholar]
- Wultsch, C.; Caragiulo, A.; Dias-Freedman, I.; Quigley, H.; Rabinowitz, S.; Amato, G. Genetic diversity and population structure of Mesoamerican jaguars (Panthera onca): Implications for conservation and management. PLoS ONE 2016, 11, e0162377. [Google Scholar] [CrossRef]
- Quigley, H.; Foster, R.; Petracca, L.; Payan, E.; Salom, R.; Harmsen, B. Panthera onca (Version Published in 2018). Available online: https://www.iucnredlist.org/species/15953/123791436 (accessed on 18 December 2024).
- CITES. The Illegal Trade in Jaguars (Panthera onca); Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES): Geneva, Switzerland, 2021; 141p. [Google Scholar]
- Kretser, H.; Nuñez-Salas, M.; Polisar, J.; Maffei, L. A Range-wide analysis of legal instruments applicable to jaguar conservation. J. Int. Wildl. Law Policy 2022, 25, 1–61. [Google Scholar] [CrossRef]
- Hansen, M.; Potapov, P.; Moore, R.; Hancher, M.; Turubanova, S.; Tyukavina, A.; Thau, D.; Stehman, S.; Goetz, S.; Loveland, T.; et al. High-resolution global maps of 21st-century forest cover change. Science 2013, 342, 850–853. [Google Scholar] [CrossRef]
- Olsoy, P.; Zeller, K.; Hicke, J.; Quigley, H.; Rabinowitz, A.; Thornton, D. Quantifying the effects of deforestation and fragmentation on a range-wide conservation plan for jaguars. Biol. Conserv. 2016, 203, 8–16. [Google Scholar] [CrossRef]
- Thornton, D.; Zeller, K.; Rondinini, C.; Boitani, L.; Crooks, K.; Burdett, C.; Rabinowitz, A.; Quigley, H. Assessing the umbrella value of a range-wide conservation network for jaguars (Panthera onca). Ecol. Appl. 2016, 26, 1112–1124. [Google Scholar] [CrossRef]
- Espinosa, S.; Celis, G.; Branch, L. When roads appear jaguars decline: Increased access to an Amazonian wilderness area reduces potential for jaguar conservation. PLoS ONE 2016, 13, e0189740. [Google Scholar] [CrossRef]
- Foster, R.; Harmsen, B.; Macdonald, D.; Collins, J.; Urbina, Y.; Garcia, R.; Doncaster, C. Wild meat: A shared resource amongst people and predators. Oryx 2014, 50, 63–75. [Google Scholar] [CrossRef]
- Cavalcanti, S.; Gese, E. Spatial ecology and social interactions of jaguars (Panthera onca) in the Southern Pantanal, Brazil. J. Mammal. 2009, 90, 935–945. [Google Scholar] [CrossRef]
- Polisar, J.; Maxit, I.; Scognamillo, D.; Farrell, L.; Sunquist, M.E.; Eisenberg, J.F. Jaguars, pumas, their prey base, and cattle ranching: Ecological interpretations of a management problem. Biol. Conserv. 2003, 109, 297–310. [Google Scholar] [CrossRef]
- Soto-Shoender, J.; Giuliano, W. Predation on livestock by large carnivores in the tropical lowlands of Guatemala. Oryx 2011, 45, 561–568. [Google Scholar] [CrossRef]
- Tortato, F.; Layme, V.; Crawshaw, P.; Izzo, T. The impact of herd composition and foraging area on livestock predation by big cats in the Pantanal of Brazil. Anim. Conserv. 2015, 18, 539–547. [Google Scholar] [CrossRef]
- Morcatty, T.; Macedo, J.; Nekaris, K.; Ni, Q.; Durigan, C.; Svensson, M.; Nijman, V. Illegal trade in wild cats and its link to Chinese-led development in Central and South America. Conserv. Biol. 2020, 34, 1525–1535. [Google Scholar] [CrossRef] [PubMed]
- Polisar, J.; Davies, C.; Da-Silva, M.; Arais, M.; Morcatty, T.; Lambert, A.; Wallace, R.; Zhang, S.; Oliveira-Da-Costa, M.; Nuñez-Salas, M.; et al. A global perspective on trade in jaguar parts from South America. Cat News 2023, 16, 74. [Google Scholar]
- Paviolo, A.; de-Angelo, C.; Ferraz, K.; Morato, R.; Martinez-Pardo, J.; Srbek-Araujo, A.; Mello-Beisiegel, B.; Lima, F.; Sana, D.; Da-Silva, M.; et al. A biodiversity hotspot losing its top predator: The challenge of jaguar conservation in the Atlantic Forest of South America. Sci. Rep. 2016, 6, 37147. [Google Scholar] [CrossRef]
- Hoogesteijn, R.; Hoogesteijn, A.; Mondolfi, E. Jaguar predation vs. jaguar conservation: Cattle mortality by felines on three ranches in the Venezuelan Llanos. In Mammals as Predators; Dunstone, N., Gorman, M.L., Eds.; Oxford University Press: London, UK, 1993; Volume 65, pp. 391–407. [Google Scholar]
- Rabinowitz, A.; Nottingham, B. Ecology and behavior of the jaguar (Panthera onca) in Belize, Central America. J. Zool. Ser. A 1986, 210, 149–159. [Google Scholar]
- Hausermann, H.; Hutchinson, E.; Walder-Hoge, Z. A spatial analysis border “security” and jaguars in the U.S.-Mexico borderlands. Front. Conserv. Sci. 2024, 5, 1355997. [Google Scholar] [CrossRef]
- Di Bitetti, M.; de-Angelo, C.; Quiroga, V.; Altrichter, M.; Paviolo, A.; Cuyckens, G.; Perovic, P. Estado de conservación del jaguar en Argentina. In El Jaguar en el Siglo XXI; Medellin, R., de-la-Torre, J., Zarza, H., Chávez, C., Ceballos, G., Eds.; Fondo de Cultura Económica: Mexico City, Mexico, 2016; pp. 447–478. [Google Scholar]
- Mondolfi, E.; Hoogesteijn, R. Notes on the biology and status of the Jaguar (Panthera onca) in Venezuela. In Cats of the World: Biology, Conservation and Management; Miller, S., Everett, D., Eds.; National Wildlife Federation: Washington, DC, USA, 1986; pp. 78–89. [Google Scholar]
- Schaller, G. Mammals and their biomass on a Brazilian ranch. Arq. Zool. 1983, 31, 1–36. [Google Scholar] [CrossRef]
- Schaller, G.; Crawshaw, P. Movement patterns of Jaguar. Biotropica 1980, 12, 161–168. [Google Scholar] [CrossRef]
- Quigley, H.; Crawshaw, P. A conservation plan for the Jaguar (Panthera onca) in the Pantanal region of Brazil. Biol. Conserv. 1992, 61, 149–157. [Google Scholar] [CrossRef]
- Azevedo, F.; Murray, D. Evaluation of potential factors predisposing livestock to predation by jaguars. J. Wildl. Manag. 2007, 71, 2379–2386. [Google Scholar] [CrossRef]
- Scognamillo, D.; Maxit, I.; Sunquist, M.; Polisar, J. Coexistence of jaguar (Panthera onca) and puma (Puma concolor) in a mosaic landscape in the Venezuelan llanos. J. Zool. 2003, 259, 269–279. [Google Scholar] [CrossRef]
- Medellin, R.; de-la-Torre, J.; Zarza, H.; Chavez, C.; Ceballos, G. El Jaguar en el Siglo XXI la Perspectiva Continental; Fondo de Cultura Económica: Ciudad de México, Mexico, 2016; 543p. [Google Scholar]
- Medellin, R.; Equihua, C.; Chetkiewicz, C.; Crawshaw, P.; Rabinowitz, A.; Redford, K.; Robinson, J.; Sanderson, E.; Taber, A. El Jaguar en el Nuevo Milenio; Fondo de Cultura Económica: Mexico City, Mexico, 2002. [Google Scholar]
- Hoogesteijn, R.; Hoogesteijn, A. Anti-Predation Strategies for Cattle Ranching in Latin America: A Guide, 1st ed.; Panthera: Campo Grande, Brazil, 2014; 63p. [Google Scholar]
- Valderrama-Vásquez, C.; Hoogesteijn, R.; Payán-Garrido, E. GRECO: Manual de Campo Para el Manejo del Conflicto Entre Humanos y Felinos; Panthera, USFWS, Fernando Peña Editores: Cali, Colombia, 2016; 81p. [Google Scholar]
- Rabinowitz, A.; Zeller, K. A range-wide model of landscape connectivity and conservation for the jaguar, Panthera onca. Biol. Conserv. 2010, 143, 939–945. [Google Scholar] [CrossRef]
- WCS-JCP. Path of the Jaguar; Wildlife Conservation Society, Jaguar Conservation Program: New York, NY, USA, 2007; 21p. [Google Scholar]
- WWF. Living Planet Report 2020—Bending the Curve of Biodiversity Loss; WWF: Gland, Switzerland, 2020. [Google Scholar]
- Khorozyan, I.; Waltert, M. A global view on evidence-based effectiveness of interventions used to protect livestock from wild cats. Conserv. Sci. Pract. 2021, 3, e317. [Google Scholar] [CrossRef]
- Villalba, L.; Maffei, L.; Fleytas, M.; Polisar, J. Primeras experiencias de mitigación de conflictos entre ganaderos y grandes felinos en estancias de Paraguay. In II. Conflictos entre felinos y humanos en América Latina; Castaño-Uribe, C., Lasso, C., Hoogesteijn, R., Díaz-Pulido, A., Payán-Garrido, E., Eds.; Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Fundación Herencia Ambiental Caribe y Fundación Panthera 2016: Bogotá, Colombia, 2016; pp. 227–236. [Google Scholar]
- Valderrama-Vásquez, C.; Hoogesteijn, R.; Payán-Garrido, E.; Quigley, H.; Hoogesteijn, A. Predator-friendly ranching, use of electric fences, and creole cattle in the Colombian savannas. Eur. J. Wildl. Res. 2024, 70, 1. [Google Scholar] [CrossRef]
- Diaz-Santos, F.; Polisar, J. Improving Livestock Management for Economic-Environmental Stability in Mesoamerica’s Mosquitia; Darwin DEFRA Project 23-014; Wildlife Conservation Society: New York, NY, USA, 2021; 39p. [Google Scholar]
- Lodeiro-Ocampo, N.; Gantchoff, M.; Nigro, N.; Palaia, J.; Gnatiuk, D. Prevención de depredación de yaguareté Panthera onca a ganado vacuno mediante cercas electrificadas en Misiones, Argentina. Rev. Mex. Mastozoología Nueva época 2021, 11, 1–10. [Google Scholar] [CrossRef]
- de-la-Torre, J.; Camacho, G.; Arroyo-Gerala, P.; Cassiagne, I.; Rivero, M.; Campos-Arceiz, A. A cost-effective approach to mitigate conflict between ranchers and large predators: A case study with jaguars in the Mayan Forest. Biol. Conserv. 2021, 256, 109066. [Google Scholar] [CrossRef]
- Tortato, F.; Izzo, T.; Hoogesteijn, R.; Peres, C. The numbers of the beast: Valuation of jaguar (Panthera onca) tourism and cattle depredation in the Brazilian Pantanal. Glob. Ecol. Conserv. 2017, 11, 106–114. [Google Scholar] [CrossRef]
- Hoogesteijn, R.; Hoogesteijn, A.; Tortato, F.; Rampim, L.; Vilas-Boas-Concone, H.; May- Junior, J.; Sartorello, L. Conservación de jaguares (Panthera onca) fuera de áreas protegidas: Turismo de observación de jaguares en propiedades privadas del Pantanal, Brasil. In I. Conservacion de Grandes Vertebrados en Areas no Protegidas de Colombia, Venezuela y Brasil; Payan-Garrido, E., Lasso, C., Castano-Uribe, C., Eds.; Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH): Bogotá, Colombia, 2015; pp. 259–274. [Google Scholar]
- Jędrzejewski, W.; Puerto, M.; Goldberg, J.; Hebblewhite, M.; Abarca, M.; Gamarra, G.; Calderón, L.; Romero, J.; Viloria, A.; Carreño, R.; et al. Density and population structure of the jaguar (Panthera onca) in a protected area of Los Llanos, Venezuela, from 1 year of camera trap monitoring. Mamm. Res. 2016, 62, 9–19. [Google Scholar] [CrossRef]
- Polisar, J.; Hoogesteijn, A.; Perera-Romero, L.; Puerta-Castillo, F.; Isasi-Catalá, M.; Jedrzejewski, W.; Hoogesteijn, R. The rich tradition of jaguar research and conservation in Venezuela and its impact on human-jaguar coexistence throughout the species’ range. Anartia 2023, 34, 75–95. [Google Scholar]
- Monteiro, M.; Marchini, S.; Del-Toro-Orozco, W.; Ramalho, E. The Impact of community-based tourism on human-jaguar interactions in central Amazonia. Hum. Dimens. Wildl. 2024, 1–17. [Google Scholar] [CrossRef]
- Khorozyan, I.; Ghoddousi, A.; Soofi, M.; Waltert, M. Big cats kill more livestock when wild prey reaches a minimum threshold. Biol. Conserv. 2015, 192, 268–275. [Google Scholar] [CrossRef]
- Hoogesteijn, A.; Hoogesteijn, R. Cattle ranching and biodiversity conservation as allies in South America’s flooded savannas. Great Plains Res. 2010, 20, 37–50. [Google Scholar]
- Polisar, J. Humans and Jaguars Can Live Together—Here Is How. The Revelator, 19 November 2021. [Google Scholar]
- Cavalcanti, S. Predator-Prey Relationships and Spatial Ecology of Jaguars in the Southern Pantanal, Brazil: Implications for Conservation and Management. Ph.D. Thesis, Utah State University, Logan, UT, USA, 2008. [Google Scholar]
- Devlin, A.; Frair, J.; Crawshaw, P.; Hunter, L.; Tortato, F.; Hoogesteijn, R.; Robinson, N.; Robinson, H.; Quigley, H. Drivers of large carnivore density in non-hunted, multi-use landscapes. Conserv. Sci. Pract. 2023, 5, e12745. [Google Scholar] [CrossRef]
- Hoogesteijn, R.; Payán-Garrido, E.; Valderrama-Vásquez, C.; Hoogesteijn, A.; Tortato, F.; Salom-Pérez, R.; Corrales-Gutiérrez, D.; Quigley, H. Protocolo Para a Resolução de Conflito de Felinos e a Pecuaria na América Latina; Secção I: Diretrizes de Diagnóstico e Operação; Panthera: Campo Grande, Brazil, 2024; Volume 1, 131p. [Google Scholar]
- Corrales-Gutiérrez, D.; Salom-Pérez, R.; Hoogesteijn, R. Implementación de estrategias anti-depredatorias en fincas ganaderas ubicadas dentro de dos importantes corredores biológicos de Costa Rica. In II. Conflicto entre Felinos y Humanos en América Latina; Serie Editorial Fauna Silvestre Neotropical; Castaño-Uribe, C., Lasso, C., Hoogesteijn, R., Diaz-Pulido, A., Payán-Garrido, A., Eds.; Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Fundación Herencia Ambiental Caribe, Panthera: Bogotá, Colombia, 2016; pp. 152–167. [Google Scholar]
- Corrales-Gutiérrez, D.; Salom-Pérez, R.; Hoogesteijn, R. Convenio entre el gobierno de Costa Rica y Panthera: Unidad de Atención de Conflictos con Felinos (UACFel). In II. Conflicto entre Felinos y Humanos en América Latina; Serie Editorial Fauna Silvestre Neotropical; Castaño-Uribe, C., Lasso, C., Hoogesteijn, R., Diaz-Pulido, A., Payán-Garrido, A., Eds.; Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Fundación Herencia Ambiental Caribe, Panthera: Bogotá, Colombia, 2016; pp. 169–180. [Google Scholar]
- CITES Decision 19.110 to 19.114 Jaguars (Panthera onca). Decisions of the Conference of the Parties to CITES in Effect After the 19th Meeting. Available online: https://cites.org/eng/dec/index.php/44279 (accessed on 17 November 2024).
- CBD Kunming-Montreal Global Biodiversity Framework. Decision 15/4 After the 15th Meeting. Available online: https://www.cbd.int/doc/decisions/cop-15/cop-15-dec-04-en.pdf (accessed on 17 November 2024).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).