A 5-Years (2015–2019) Control Activity of an EU Laboratory: Contamination of Histamine in Fish Products and Exposure Assessment
Abstract
:Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Sample Preparation
2.3. ELISA Test Reagents, Equipment and Procedure
2.4. HPLC Reagents, Equipment, and Procedure
2.5. Methods Quality Control
2.6. Data Handling and Statistical Analysis
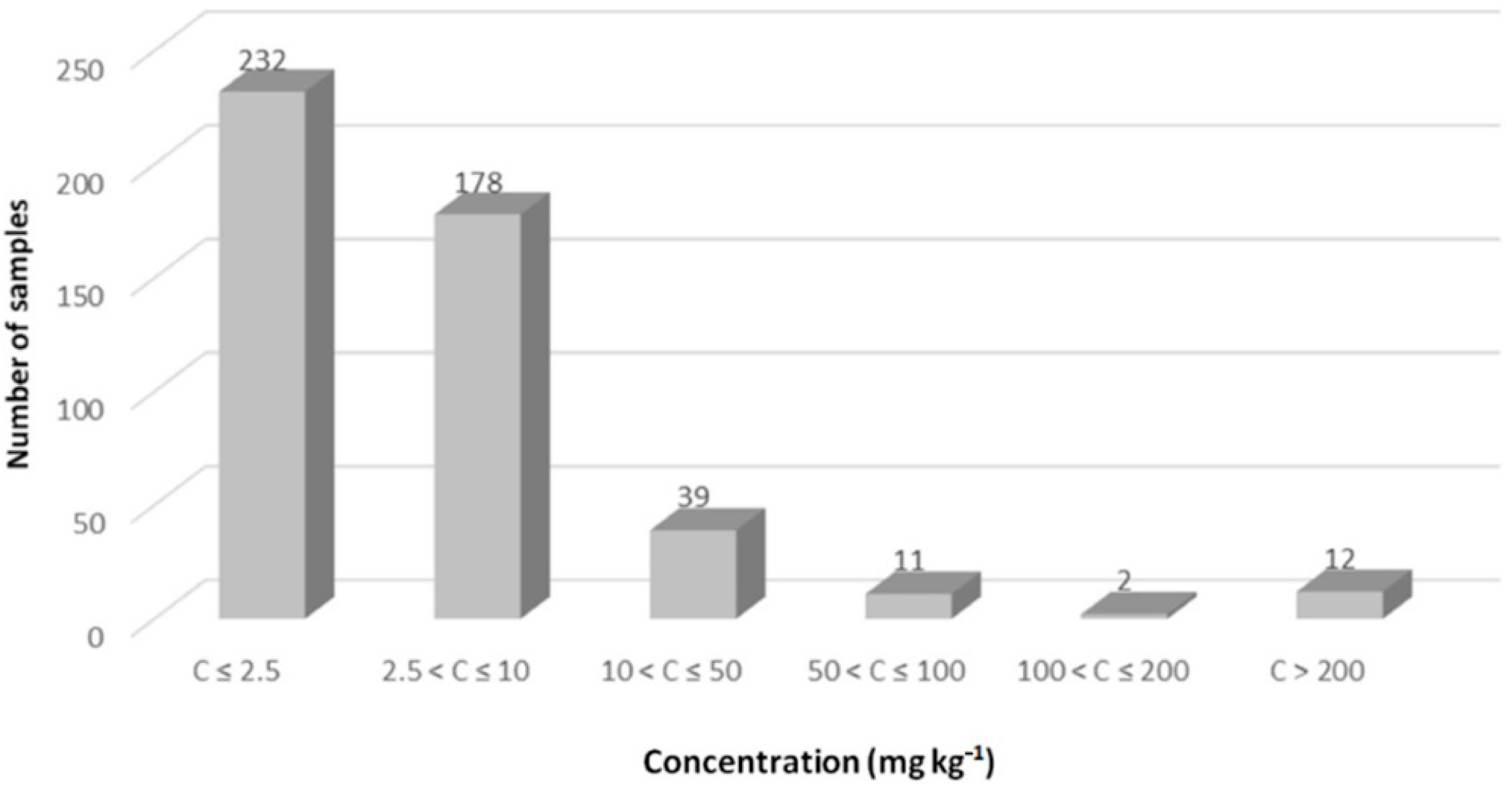
3. Results
4. Discussion
4.1. HIM Levels in Fish Samples
4.1.1. Fresh, Frozen, and Defrosted Fish
4.1.2. Canned Products
4.1.3. Ripened Products
4.2. Risk Exposure
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pesce, Italia Leader in Europa per i Consumi. Available online: https://www.repubblica.it/economia/rapporti/osservaitalia/conad/2018/11/21/news/pesce_italia_leader_in_europa_per_i_consumi-212229828/ (accessed on 25 October 2020).
- FAO. In Termini Pro Capite il Consumo Mondiale di Pesce Raggiungerà i 21.5 kg nel 2030. Available online: https://www.pesceinrete.com/fao-in-termini-pro-capite-il-consumo-mondiale-di-pesce-raggiungera-i-215-kg-nel-2030/ (accessed on 25 October 2020).
- EFSA Scientific Committee. Statement on the benefits of fish/seafood consumption compared to the risks of methylmercury in fish/seafood. EFSA J. 2015, 13, 3982. [Google Scholar] [CrossRef] [Green Version]
- FAO Fisheries Report. FAO Report of the Joint FAO/WHO Expert Consultation on the Risks and Benefits of Fish Consumption Rome, 25–29 January 2010; FAO: Rome, Italy, 2011; ISBN 978-92-5-106999-8. [Google Scholar]
- Domingo, J.L.; Bocio, A.; Falcó, G.; Llobet, J.M. Benefits and risks of fish consumption Part, I. A quantitative analysis of the intake of omega-3 fatty acids and chemical contaminants. Toxicology 2007, 230, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Sumner, J.; Ross, T. A semi-quantitative seafood safety risk assessment. Int. J. Food Microbiol. 2002, 77, 55–59. [Google Scholar] [CrossRef]
- Miedico, O.; Iammarino, M.; Pompa, C.; Tarallo, M.; Chiaravalle, A.E. Assessment of lead, cadmium and mercury in seafood marketed in Puglia and Basilicata (Italy) by inductively coupled plasma mass spectrometry. Food Addit. Contam. B 2015, 8, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Caspers, H.R. Malcolm love: The chemical biology of fishes. Volume 2: Advances 1968–1977. With a supplementary key to the chemical literature. Int. Revue Gesamten Hydrobiol. Hydrogr. 1981, 66, 910. [Google Scholar] [CrossRef]
- Lehane, L.; Olley, J. Histamine fish poisoning revisited. Int. J. Food Microbiol. 2000, 58, 1–37. [Google Scholar] [CrossRef]
- Kim, S.H.; Price, R.J.; Morrissey, M.T.; Field, K.G.; Wei, C.I.; An, H. Histamine production by morganella morganii in Mackerel, Albacore, Mahi-mahi, and Salmon at Various Storage Temperatures. J. Food Sci. 2002, 67, 1522–1528. [Google Scholar] [CrossRef]
- Takahashi, H.; Ogai, M.; Miya, S.; Kuda, T.; Kimura, B. Effects of environmental factors on histamine production in the psychrophilic histamine-producing bacterium Photobacterium iliopiscarium. Food Control 2015, 52, 39–42. [Google Scholar] [CrossRef]
- Muscarella, M.; Iammarino, M.; Centonze, D.; Palermo, C. Measurement of histamine in seafood by HPLC, CE, and ELISA: Comparison of three techniques. Vet. Res. Commun. 2005, 29, 343–346. [Google Scholar] [CrossRef]
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Fuchs, T.; Kleine-Tebbe, J.; Klimek, L.; Lepp, U.; Niggemann, B.; Saloga, J.; Schäfer, C.; et al. German guideline for the management of adverse reactions to ingested histamine. Allergo J. Int. 2017, 26, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Taylor, S.L. Histamine food poisoning: Toxicology and clinical aspects. Crit. Rev. Toxicol. 1986, 17, 91–128. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (EC) No 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs. Volume L 338, pp. 1–26. Available online: http://www.sice.oas.org/tpd/cacm_eu/Negotiations/IIround_microbiological_e.pdf (accessed on 13 September 2020).
- Tao, Z.; Sato, M.; Yamaguchi, T.; Nakano, T. Formation and diffusion mechanism of histamine in the muscle of tuna fish. Food Control 2009, 20, 923–926. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No 1019/2013 of 23 October 2013. Amending Annex I to Regulation (EC) No 2073/2005 as Regards Histamine in Fishery Products. Volume L 282, pp. 46–47. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32013R1019&from=FR (accessed on 14 September 2020).
- RASFF Portal. Available online: https://webgate.ec.europa.eu/rasff-window/portal/?event=SearchForm&cleanSearch=1 (accessed on 25 October 2020).
- Fasolato, L.; Mirisola, M.; Tepedino, G.; Balzan, S.; Arcangeli, G.; Rosteghin, M.; Corrain, C.; Manfrin, A.; Berzaghi, P. Mai Più Decongelato per Fresco. Available online: https://www.eurofishmarket.it/b2b/decongelato.pdf (accessed on 25 October 2020).
- Food Fraud Network EU-Coordinated Case. Illegal Treatment of Tuna: From Canning Grade to Sushi Grade; European Commission: Brussels, Belgium, 2017; Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/food-fraud_succ-coop_tuna.pdf (accessed on 25 October 2020).
- Commission Regulation (EU) No 231/2012 of 9 March 2012. Laying down Specifications for Food Additives Listed in Annexes II and III to Regulation (EC) No 1333/2008. Volume L 83, pp. 1–290. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CONSLEG:2012R0231:20121129:EN:PDF (accessed on 16 September 2020).
- Lee, H.S. Exposure estimates of nitrite and nitrate from consumption of cured meat products by the U.S. population. Food Addit. Contam. A 2018, 35, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Studi sui Consumi Alimentari in Italia, 2005–2006. Available online: https://www.crea.gov.it/web/alimenti-e-nutrizione/-/indagine-sui-consumi-alimentari (accessed on 25 October 2020).
- Leclercq, C.; Arcella, D.; Piccinelli, R.; Sette, S.; Le Donne, C.; Turrini, A. INRAN-SCAI 2005-06 study group the italian national food consumption survey INRAN-SCAI 2005-06: Main results in terms of food consumption. Public Health Nutr. 2009, 12, 2504–2532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iammarino, M.; Mentana, A.; Centonze, D.; Palermo, C.; Mangiacotti, M.; Chiaravalle, A.E. Dye use in fresh meat preparations and meat products: A survey by a validated method based on HPLC-UV-diode array detection as a contribution to risk assessment. Int. J. Food Sci. Tech. 2020, 55, 1126–1135. [Google Scholar] [CrossRef]
- Rizzi, A. Inferenza Statistica, 2nd ed.; UTET Università: Rome, Italy, 1992. [Google Scholar]
- European Market Observatory for Fisheries and Aquaculture Products EUMOFA. The EU Fish Market—2016 Edition; European Commission: Brussels, Belgium, 2016; Available online: https://www.eumofa.eu/documents/20178/77960/The+EU+fish+market+-+2016+Edition.pdf (accessed on 22 September 2020).
- Miedico, O.; Pompa, C.; Moscatelli, S.; Chiappinelli, A.; Carosielli, L.; Chiaravalle, A.E. Lead, cadmium and mercury in canned and unprocessed tuna: Six-years monitoring survey, comparison with previous studies and recommended tolerable limits. J. Food Compos. Anal. 2020, 94, 103638. [Google Scholar] [CrossRef]
- European Food Safety Authority, Panel on Biological Hazards (BIOHAZ). Scientific Opinion on risk based control of biogenic amine formation in fermented foods. EFSA J. 2011, 9, 2393. [Google Scholar] [CrossRef] [Green Version]
- Motil, K.J.; Scrimshaw, N.S. The role of exogenous histamine in scombroid poisoning. Toxicol. Lett. 1979, 3, 219–223. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations and World Health Organization. Joint FAO/WHO expert Meeting on the Public Health Risks of Histamine and other Biogenic Amines from Fish and Fishery Products; Meeting Report; FAO/WHO: Rome, Italy, 2012; Available online: http://www.fao.org/fileadmin/user_upload/agns/news_events/Histamine_Final_Report.pdf (accessed on 20 August 2020).
- Muscarella, M.; Lo Magro, S.; Campaniello, M.; Armentano, A.; Stacchini, P. Survey of histamine levels in fresh fish and fish products collected in Puglia (Italy) by ELISA and HPLC with fluorimetric detection. Food Control 2013, 31, 211–217. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO 19343:2017 Microbiology of the Food Chain—Detection and Quantification of Histamine in Fish and Fishery Products—HPLC Method; ISO: Geneva, Switzerland, 2017; Available online: http://store.uni.com/catalogo/iso-19343-2017?josso_back_to=http://store.uni.com/josso-security-check.php&josso_cmd=login_optional&josso_partnerapp_host=store.uni.com (accessed on 24 September 2020).
- Helsel, D.R. Non Detects and Data Analysis: Statistics for Censored Environmental Data; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Rogers, P.L.; Staruszkiewicz, W.F. Histamine test kit comparison. J. Aquat. Food Prod. Technol. 2000, 9, 5–17. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human ServicesFood and Drug AdministrationCenter for Food Safety and Applied Nutrition. Fish and Fishery Products Hazards and Controls Guidance, 4th ed.; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2020. Available online: https://www.fda.gov/media/80637/download (accessed on 22 September 2020).
- Piersanti, A.; Tavoloni, T.; Lestingi, C.; Galarini, R. High-throughput histamine analysis approach in an official control laboratory: Analytical methods and four years fish products results. Food Chem. 2014, 153, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.; Cammilleri, G.; Galluzzo, F.G.; Calabrese, I.; Pulvirenti, A.; Giangrosso, G.; Cicero, N.; Cumbo, V.; Vella, A.; Macaluso, A.; et al. Histamine in fish products randomly collected in southern Italy: A 6-year study. J. Food Prot. 2020, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, J.; Babić, J.; Jaksic, S.; Kartalovic, B.; Ljubojevic, D.; Cirkovic, M. Fish product–borne histamine intoxication outbreak and survey of imported fish and fish products in Serbia. J. Food Prot. 2016, 79, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Altieri, I.; Semeraro, A.; Scalise, F.; Calderari, I.; Stacchini, P. European official control of food: Determination of histamine in fish products by a HPLC-UV-DAD method. Food Chem. 2016, 211, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Ðordević, Ð.; Buchtová, H.; Borkovcová, I. Estimation of amino acids profile and escolar fish consumption risks due to biogenic amines content fluctuations in vacuum skin packaging/VSP during cold storage. LWT —Food Sci. Technol. 2016, 66, 657–663. [Google Scholar] [CrossRef]
- Ababouch, L.; Alaoui, M.M.; Busta, F.F. Histamine levels in commercially processed fish in Morocco. J. Food Prot. 1986, 49, 904–908. [Google Scholar] [CrossRef]
- Economou, V.; Brett, M.M.; Papadopoulou, C.; Frillingos, S.; Nichols, T. Changes in histamine and microbiological analyses in fresh and frozen tuna muscle during temperature abuse. Food Add. Contam. 2007, 24, 820–832. [Google Scholar] [CrossRef]
- Mancusi, R.; Bini, R.M.; Cecchini, M.; Donne, G.D.; Rosmini, R.; Trevisani, M. Occurance of histamine in fish products on market. It. J. Food Saf. 2012, 1, 35–39. [Google Scholar] [CrossRef]
- Park, J.S.; Lee, C.H.; Kwon, E.Y.; Lee, H.J.; Kim, J.Y.; Kim, S.H. Monitoring the contents of biogenic amines in fish and fish products consumed in Korea. Food Control 2010, 21, 1219–1226. [Google Scholar] [CrossRef]
- Ali, A.; Waheed, K.N.; Hadaiyt, A.; Begum, I.; Hayat, S. Determination of histamine levels by LC-MS/MS in various fish species available in the local markets of Punjab, Pakistan. Int. J. Fish. Aquat. Stud. 2016, 4, 128–132. [Google Scholar]
- Auerswald, L.; Morren, C.; Lopata, A.L. Histamine levels in seventeen species of fresh and processed South African seafood. Food Chem. 2006, 98, 231–239. [Google Scholar] [CrossRef]
- Gessner, B.D.; Hokama, Y.; Isto, S. Scombrotoxicosis-like illness following the ingestion of smoked salmon that demonstrated low histamine levels and high toxicity on mouse bioassay. Clin. Infect. Dis. 1996, 23, 1316–1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smart, D.R. Scombroid poisoning A report of seven cases involving the Western Australian salmon, Arripis truttaceus. Med. J. Aust. 1992, 157, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-C.; Kung, H.-F.; Chen, H.-C.; Lin, C.-S.; Tsai, Y.-H. Determination of histamine and bacterial isolation in swordfish fillets (Xiphias gladius) implicated in a food borne poisoning. Food Control 2008, 19, 16–21. [Google Scholar] [CrossRef]
- Chen, H.C.; Lee, Y.C.; Hwang, D.F.; Chiou, T.K.; Tsai, Y.H. Determination of histamine in mahi-mahi fillets (coryphaena Hippurus) Implicated in a foodborne poisoning. J. Food Saf. 2011, 31, 320–325. [Google Scholar] [CrossRef]
- Colombo, F.M.; Cattaneo, P.; Confalonieri, E.; Bernardi, C. Histamine food poisonings: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1131–1151. [Google Scholar] [CrossRef] [Green Version]
- Cattaneo, P. Sindrome Sgombroide—Intossicazione da Istamina. Sindrome Sgombroide—Intossicazione da Istamina 2001. Available online: https://riviste.unimi.it/index.php/food-in/article/download/1702/2854/ (accessed on 25 October 2020).
- Mercogliano, R.; Santonicola, S. Scombroid fish poisoning: Factors influencing the production of histamine in tuna supply chain. A review. LWT-Food Sci. Technol. 2019, 114, 108374. [Google Scholar] [CrossRef]
- Sadeghi, N.; Behzad, M.; Jannat, B.; Oveisi, M.; Hajimahmoodi, M.; Mozafzri, M. Determination of histamine in canned tuna fish available in Tehran market by ELISA method. J. Food Safe. Hyg. 2019, 5, 46–50. [Google Scholar] [CrossRef]
- Silva, T.M.; Sabaini, P.S.; Evangelista, W.P.; Gloria, M.B.A. Occurrence of histamine in Brazilian fresh and canned tuna. Food Control 2011, 22, 323–327. [Google Scholar] [CrossRef]
- Yesudhason, P.; Al-Zidjali, M.; Al-Zidjali, A.; Al-Busaidi, M.; Al-Waili, A.; Al-Mazrooei, N.; Al-Habsi, S. Histamine levels in commercially important fresh and processed fish of Oman with reference to international standards. Food Chem. 2013, 140, 777–783. [Google Scholar] [CrossRef]
- Er, B.; Demirhan, B.; Bas, S.Y.; Yentur, G.; Oktem, A.B. Determination of histamine levels in canned tuna fish. Bulg. J. Agric. Sci. 2014, 20, 834–838. [Google Scholar]
- Peivasteh-Roudsari, L.; Rahmani, A.; Shariatifar, N.; Tajdar-Oranj, B.; Mazaheri, M.; Sadighara, P.; Khaneghah, A.M. Occurrence of Histamine in Canned Fish Samples (Tuna, Sardine, Kilka, and Mackerel) from Markets in Tehran. J. Food Prot. 2020, 83, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.-H.; Kung, H.-F.; Lee, T.-M.; Chen, H.-C.; Chou, S.-S.; Wei, C.-I.; Hwang, D.-F. Determination of histamine in canned mackerel implicated in a food borne poisoning. Food Control 2005, 16, 579–585. [Google Scholar] [CrossRef]
- Vosikis, V.; Papageorgopoulou, A.; Economou, V.; Frillingos, S.; Papadopoulou, C. Survey of the histamine content in fish samples randomly selected from the Greek retail market. Food Addit. Contam. B 2008, 1, 122–129. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Decision 2007/642/EC on emergency measures applying to fishery products imported from Albania and intended for human consumption. Off. J. Eur. Union 2007, L260, 21–22. [Google Scholar]
- Rahmani, J.; Miri, A.; Mohseni-Bandpei, A.; Fakhri, Y.; Bjørklund, G.; Keramati, H.; Moradi, B.; Amanidaz, N.; Shariatifar, N.; Khaneghah, A.M. Contamination and prevalence of histamine in canned tuna from Iran: A systematic review, meta-analysis, and health risk assessment. J. Food Prot. 2018, 81, 2019–2027. [Google Scholar] [CrossRef]
- El Hariri, O.; Bouchriti, N.; Bengueddour, R. Risk assessment of histamine in chilled, frozen, canned and semi-preserved fish in Morocco; Implementation of risk ranger and recommendations to risk managers. Foods 2018, 7, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Fish Species | Products | Number of Batches | Histamine Content (mg kg−1) | Non-Compliant Batches | Histamine Content in Non-Compliant Batches (mg kg−1) | ||
|---|---|---|---|---|---|---|---|
| Mean | Min * | Max | |||||
| Tuna | Fresh/frozen/defrosted | 111 | 197 a | 2.6 | 5542 | 9 | 393–4895–695–2250–443–887–5542–1628–4532 |
| Canned | 100 ** | 4.9 b,c | 2.6 | 38 | 0 | - | |
| Anchovies | Fresh/frozen | 62 | 20 b | 2.6 | 559 | 2 | 328–559 |
| Ripened | 57 | 14 a,b | 2.7 | 132 | 0 | - | |
| Mackerel | Fresh/frozen | 45 | 7.8 a,b | 2.6 | 68 | 0 | - |
| Canned | 12 | 11 a,b | 3.0 | 105 *** | 0 | - | |
| Sardines and herrings | Fresh/frozen | 14 | 2.7 a,b | 2.8 | 5.6 | 0 | - |
| Ripened | 4 | 19 **** | 3.7 | 55 | 0 | - | |
| Other fresh fish | - | 62 | 3.1 b,c | 2.6 | 3.2 | 0 | - |
| Other canned products | - | 6 | 2.7 **** | 4.6 | 6.4 | 0 | - |
| Fresh and Frozen Seafood * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 3.7 (7.4%) | 2.5 (5.0%) | 3.2 (6.4%) | 5.3 (10.6%) | 4.2 (8.4%) | 4.7 (9.5%) |
| Adolescents (10–17 a) | 3.9 (7.7%) | 3.5 (7.1%) | 3.9 (7.9%) | 6.0 (12.1%) | 5.5 (10.9%) | 5.7 (11.4%) |
| Adults (18–64 a) | 3.9 (7.7%) | 3.1 (6.3%) | 3.7 (7.4%) | 5.6 (11.1%) | 5.2 (10.5%) | 5.4 (10.8%) |
| Elderly (65–97 a) | 3.9 (7.7%) | 2.3 (4.7%) | 3.1 (6.3%) | 5.4 (10.8%) | 4.5 (9.0%) | 4.9 (9.8%) |
| Preserved Seafood * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 0.03 (0.05%) | 0.03 (0.07%) | 0.03 (0.05%) | 0.09 (0.19%) | 0.09 (0.19%) | 0.09 (0.19%) |
| Adolescents (10–17 a) | 0.03 (0.07%) | 0.04 (0.09%) | 0.04 (0.09%) | 0.15 (0.29%) | 0.13 (0.26%) | 0.14 (0.28%) |
| Adults (18–64 a) | 0.06 (0.12%) | 0.05 (0.10%) | 0.06 (0.12%) | 0.16 (0.33%) | 0.15 (0.31%) | 0.16 (0.33%) |
| Elderly (65–97 a) | 0.05 (0.10%) | 0.03 (0.07%) | 0.04 (0.09%) | 0.16 (0.33%) | 0.15 (0.29%) | 0.15 (0.31%) |
| Seafood—Overall * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 2.7 (5.3%) | 2.0 (4.0%) | 2.3 (4.6%) | 3.8 (7.6%) | 3.0 (6.0%) | 3.4 (6.8%) |
| Adolescents (10–17 a) | 2.8 (5.5%) | 2.8 (5.7%) | 2.8 (5.7%) | 4.3 (8.7%) | 3.9 (7.9%) | 4.1 (8.2%) |
| Adults (18–64 a) | 2.8 (5.5%) | 2.6 (5.2%) | 2.7 (5.3%) | 4.0 (8.0%) | 3.8 (7.5%) | 3.9 (7.7%) |
| Elderly (65–97 a) | 2.8 (5.5%) | 2.9 (3.8%) | 2.3 (4.5%) | 3.9 (7.7%) | 3.2 (6.5%) | 3.5 (7.1%) |
| Fresh and Frozen Seafood * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 0.24 (0.48%) | 0.18 (0.36%) | 0.21 (0.42%) | 0.34 (0.69%) | 0.27 (0.54%) | 0.31 (0.61%) |
| Adolescents (10–17 a) | 0.25 (0.50%) | 0.25 (0.51%) | 0.25 (0.51%) | 0.39 (0.78%) | 0.35 (0.71%) | 0.37 (0.74%) |
| Adults (18–64 a) | 0.25 (0.50%) | 0.23 (0.47%) | 0.24 (0.48%) | 0.36 (0.72%) | 0.34 (0.68%) | 0.35 (0.70%) |
| Elderly (65–97 a) | 0.25 (0.50%) | 0.17 (0.34%) | 0.20 (0.41%) | 0.35 (0.70%) | 0.29 (0.58%) | 0.32 (0.63%) |
| Preserved Seafood * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 0.02 (0.03%) | 0.02 (0.04%) | 0.02 (0.03%) | 0.06 (0.12%) | 0.06 (0.12%) | 0.06 (0.12%) |
| Adolescents (10–17 a) | 0.02 (0.04%) | 0.03 (0.06%) | 0.03 (0.06%) | 0.09 (0.19%) | 0.08 (0.17%) | 0.09 (0.18%) |
| Adults (18–64 a) | 0.04 (0.08%) | 0.03 (0.07%) | 0.04 (0.08%) | 0.10 (0.21%) | 0.10 (0.20%) | 0.10 (0.21%) |
| Elderly (65–97 a) | 0.03 (0.07%) | 0.02 (0.04%) | 0.03 (0.06%) | 0.10 (0.21%) | 0.09 (0.19%) | 0.10 (0.20%) |
| Seafood—Overall * | ||||||
| Population Group | Global Population g Die−1 (% of NOAEL) | Only Consumers g Die−1 (% of NOAEL) | ||||
| Male | Female | Male + Female | Male | Female | Male + Female | |
| Children (3–9 a) | 0.25 (0.49%) | 0.19 (0.38%) | 0.21 (0.43%) | 0.35 (0.71%) | 0.28 (0.56%) | 0.32 (0.63%) |
| Adolescents (10–17 a) | 0.26 (0.51%) | 0.26 (0.53%) | 0.26 (0.53%) | 0.40 (0.80%) | 0.36 (0.73%) | 0.38 (0.76%) |
| Adults (18–64 a) | 0.26 (0.51%) | 0.24 (0.48%) | 0.25 (0.49%) | 0.37 (0.74%) | 0.35 (0.70%) | 0.36 (0.72%) |
| Elderly (65–97 a) | 0.26 (0.51%) | 0.18 (0.35%) | 0.21 (0.42%) | 0.36 (0.72%) | 0.30 (0.60%) | 0.33 (0.65%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo Magro, S.; Summa, S.; Iammarino, M.; D’Antini, P.; Marchesani, G.; Chiaravalle, A.E.; Muscarella, M. A 5-Years (2015–2019) Control Activity of an EU Laboratory: Contamination of Histamine in Fish Products and Exposure Assessment. Appl. Sci. 2020, 10, 8693. https://doi.org/10.3390/app10238693
Lo Magro S, Summa S, Iammarino M, D’Antini P, Marchesani G, Chiaravalle AE, Muscarella M. A 5-Years (2015–2019) Control Activity of an EU Laboratory: Contamination of Histamine in Fish Products and Exposure Assessment. Applied Sciences. 2020; 10(23):8693. https://doi.org/10.3390/app10238693
Chicago/Turabian StyleLo Magro, Sonia, Simona Summa, Marco Iammarino, Pasquale D’Antini, Giuliana Marchesani, Antonio Eugenio Chiaravalle, and Marilena Muscarella. 2020. "A 5-Years (2015–2019) Control Activity of an EU Laboratory: Contamination of Histamine in Fish Products and Exposure Assessment" Applied Sciences 10, no. 23: 8693. https://doi.org/10.3390/app10238693
APA StyleLo Magro, S., Summa, S., Iammarino, M., D’Antini, P., Marchesani, G., Chiaravalle, A. E., & Muscarella, M. (2020). A 5-Years (2015–2019) Control Activity of an EU Laboratory: Contamination of Histamine in Fish Products and Exposure Assessment. Applied Sciences, 10(23), 8693. https://doi.org/10.3390/app10238693






