Performance Evaluation of Enhanced Bioretention Systems in Removing Dissolved Nutrients in Stormwater Runoff
Abstract
:1. Introduction
2. Materials and Methods
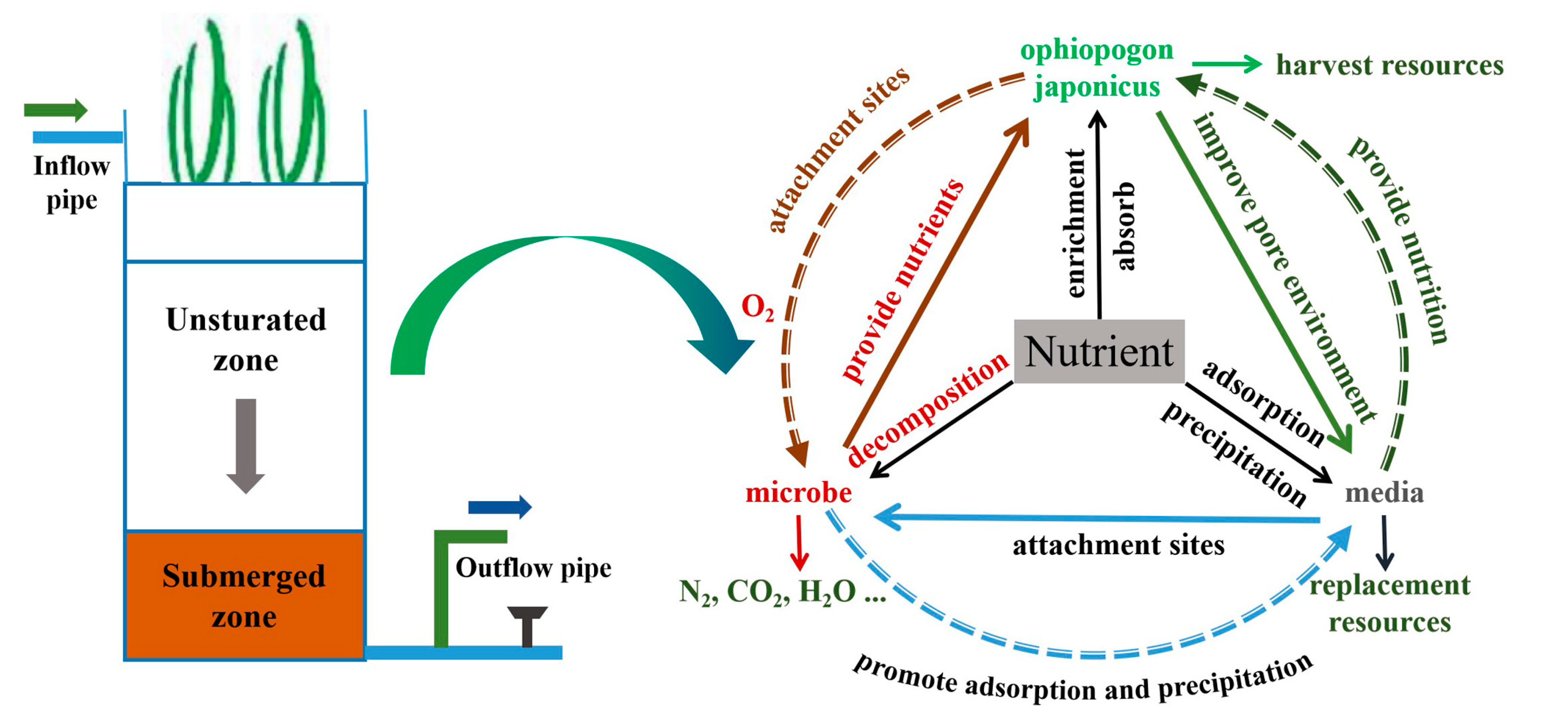
2.1. Set-Up of the Bioretention Columns
2.2. Synthetic Stormwater Runoff
2.3. Experimental Design
2.4. Evaluation of Removal Efficiency by Bioretention
2.5. Surface Characterization and Chemical Compositions
2.6. Water Quality Assessment Based on Fuzzy Water Pollution Index Method
2.6.1. Establishing the Model of the Fuzzy Comprehensive Index (FCI) Method
- (1)
- Identify the factor set U = {u1, u2, ···, un}. Assume that the number of factors related to the evaluated object is n.
- (2)
- Identify the remark set V = {v1, v2, ···, vn}. Assume that the number of all the possible remarks is m.
- (3)
- Process the single factor evaluation, which means evaluating every factor ui {i = 1, 2, ···, n}, and getting the single factor evaluation set ri = {ri1, ri2, ···, rim}. Determine the evaluation matrix by the mapping relationship.
- (4)
- Since every factor’s status is not necessarily equal, there is a need for weighting factors. Assume that the fuzzy set A = {a1, a2, ···, an} represents the weight distribution of every factor and n. The comprehensive evaluation set is: B = A × R = {b1, b2, bi, ···, bm}.
2.6.2. Calculation of the FCI of Water Quality
3. Results and Discussion
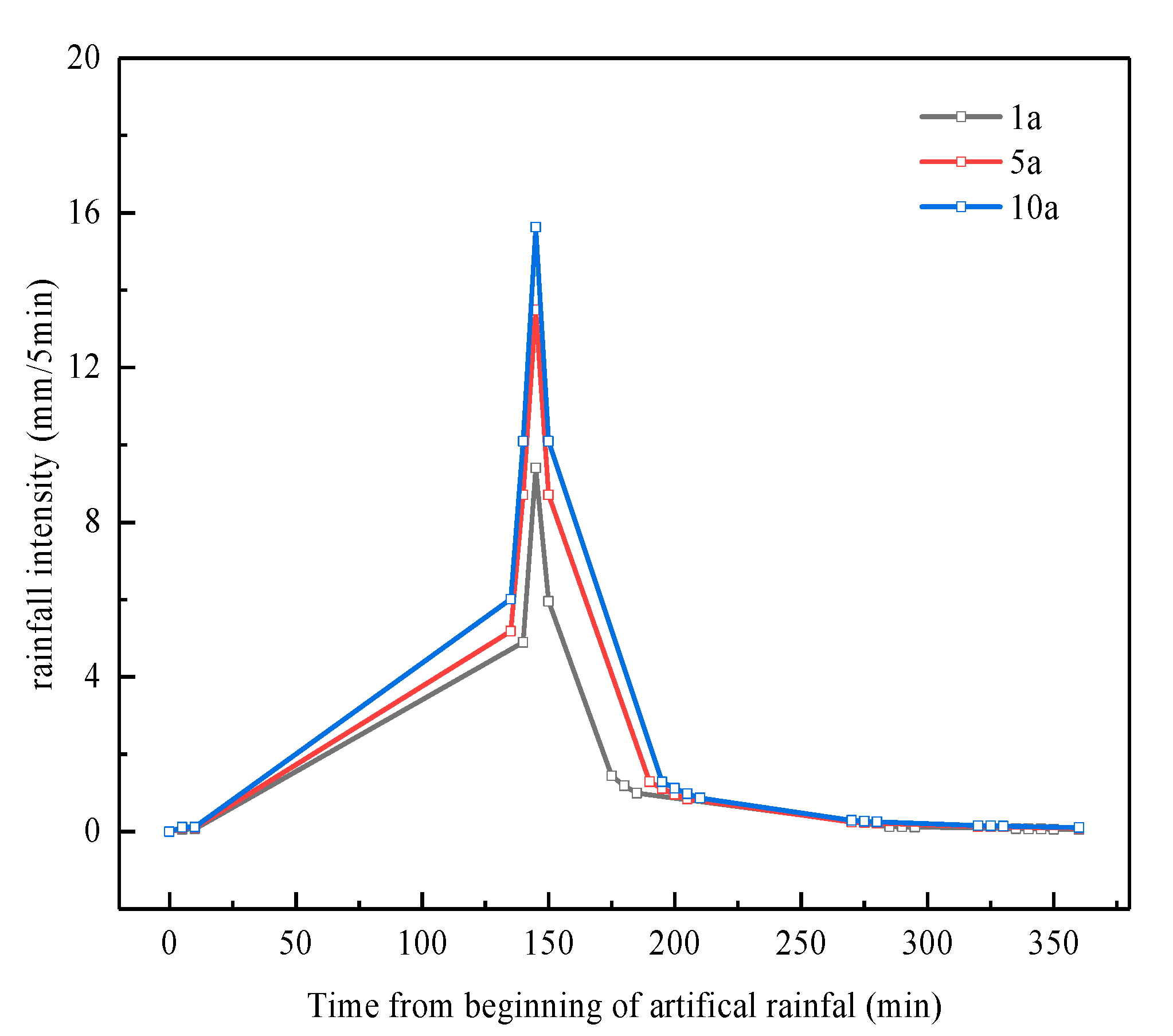
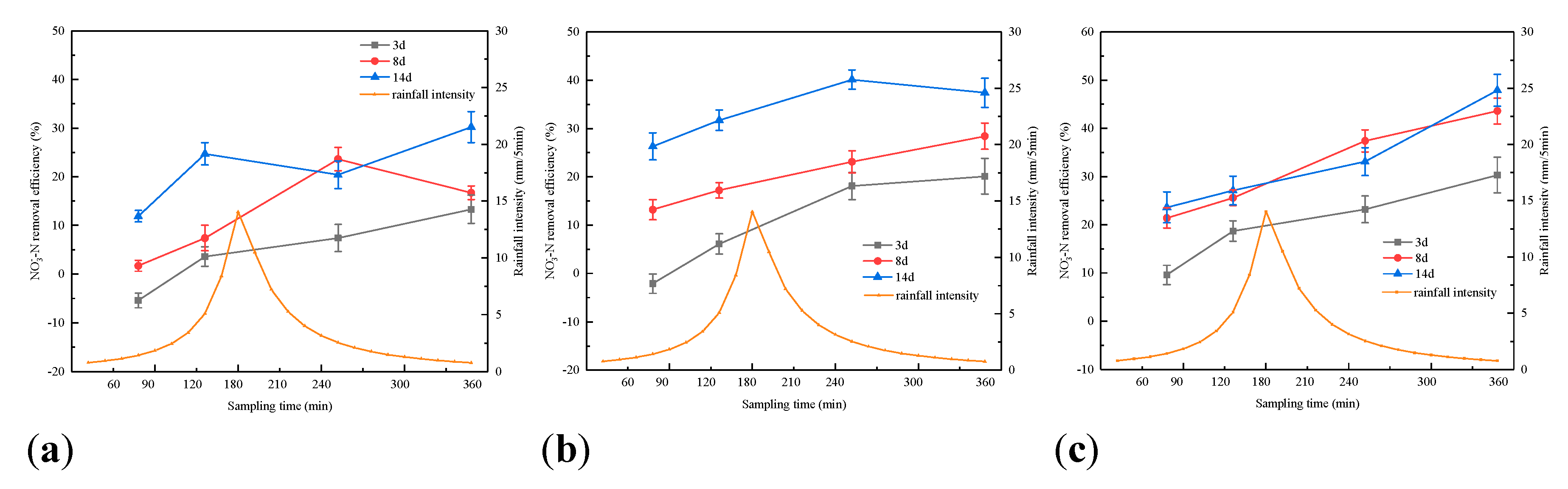
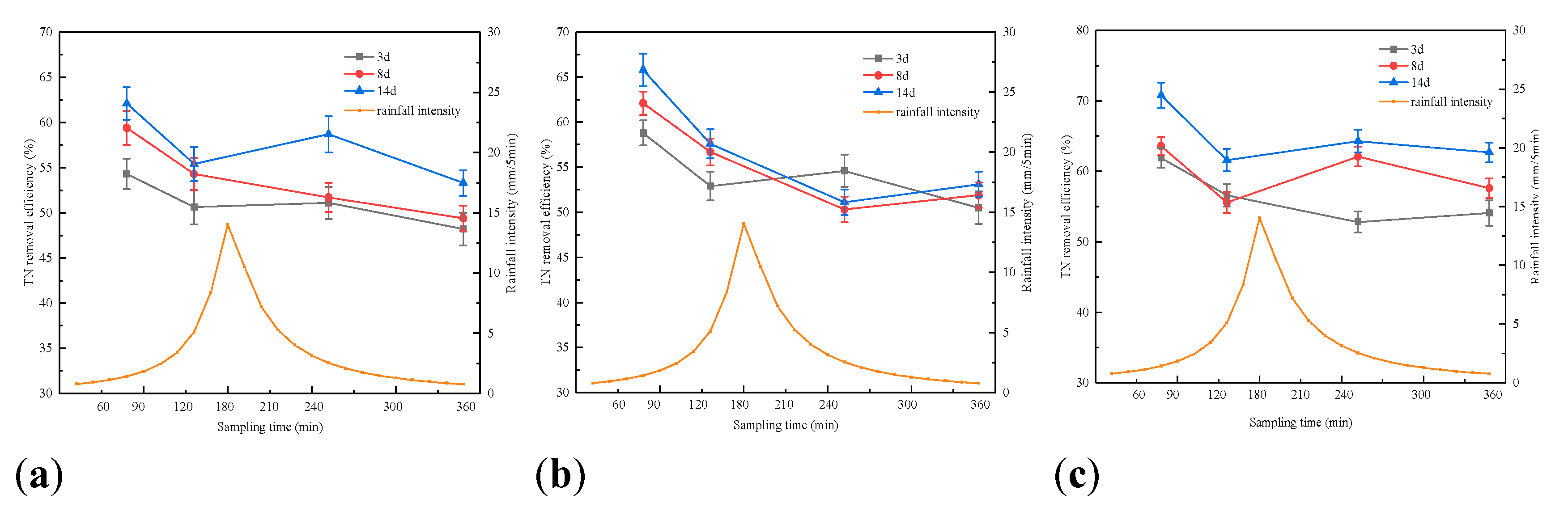
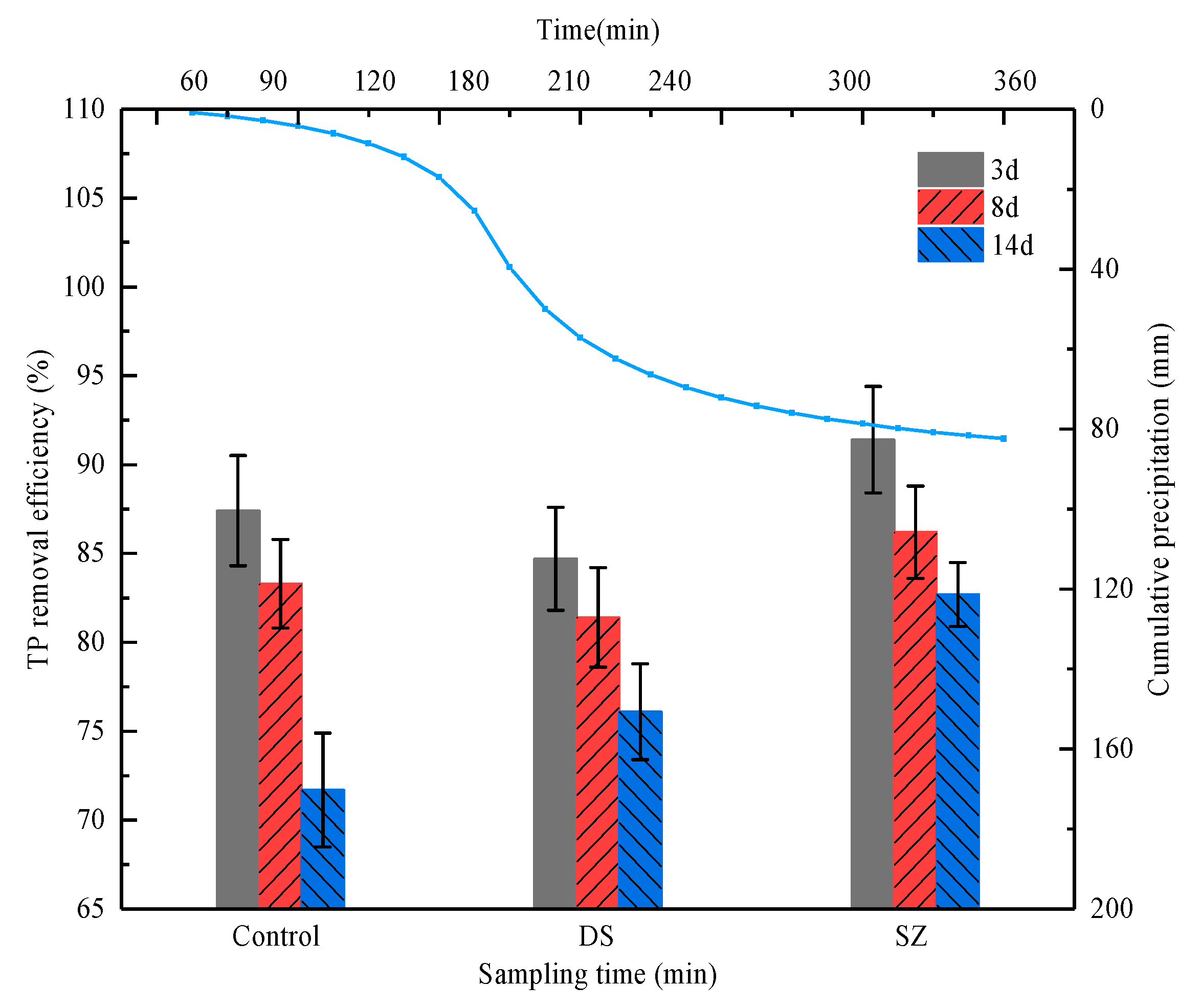
3.1. Effect of Rainfall Intensity on the Retention of Pollutants by Bioretention
3.2. Effect of Antecedent Dry Periods on Pollutants Retention in Bioretention Systems
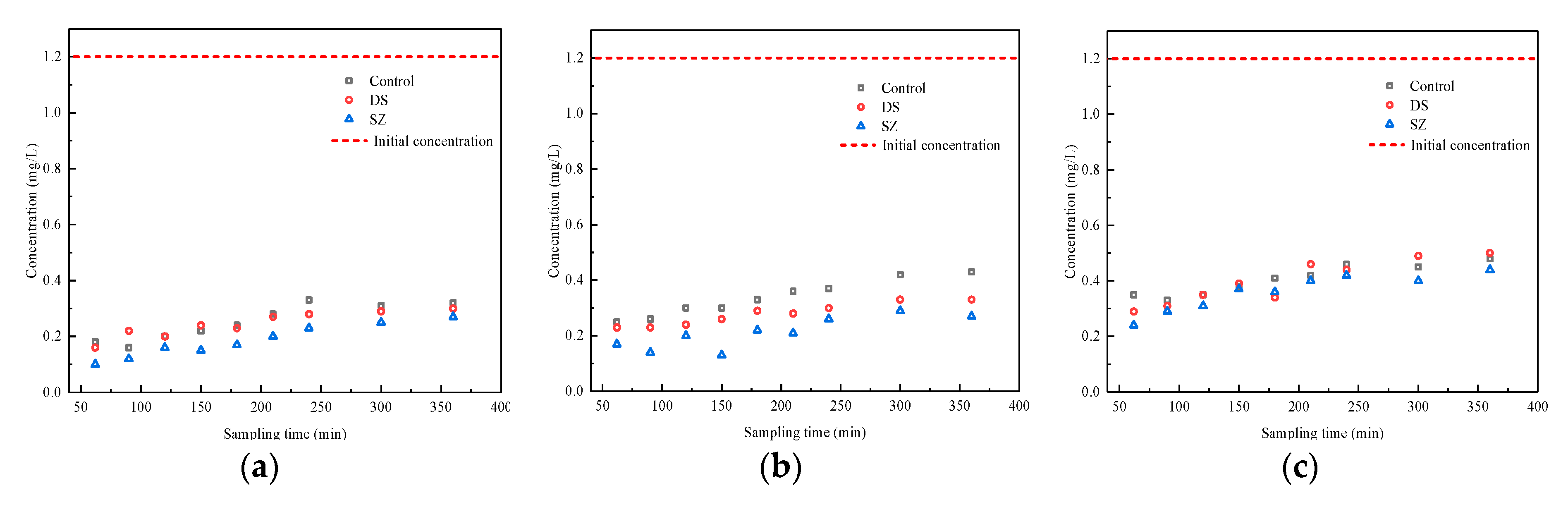
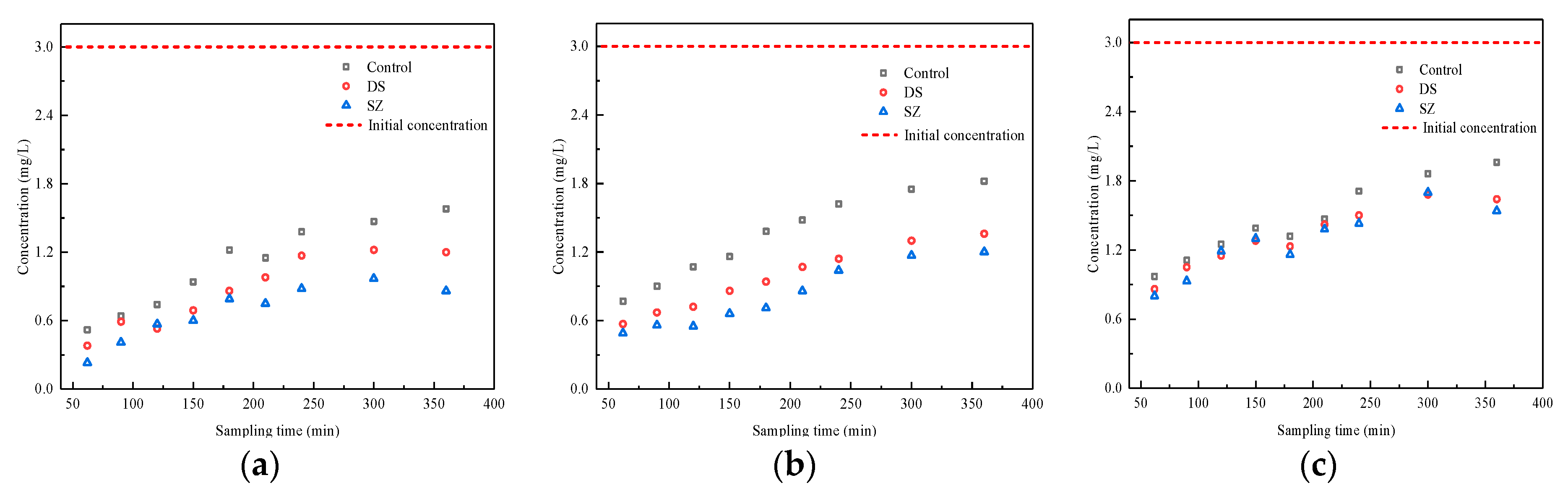
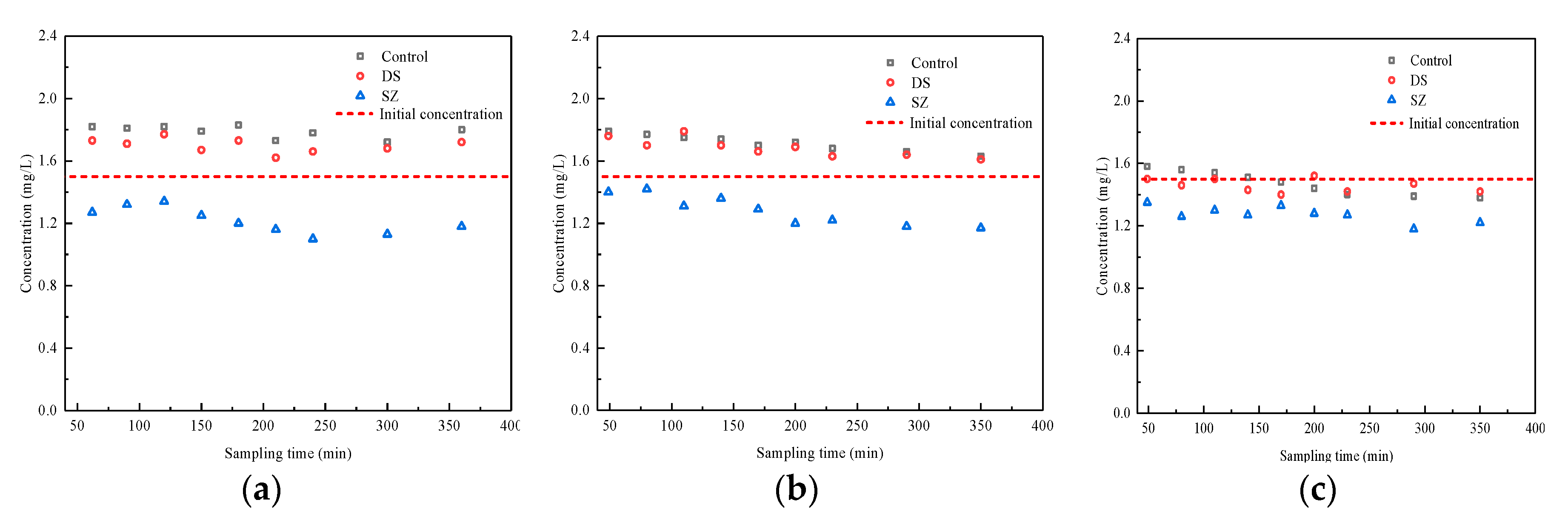
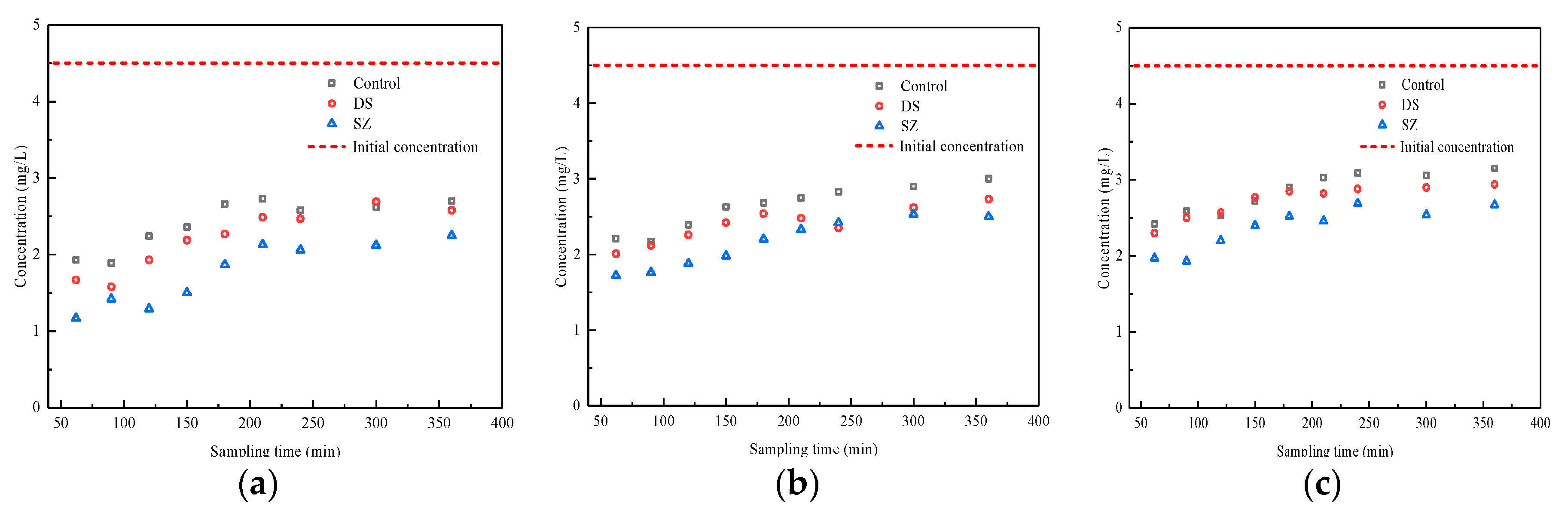
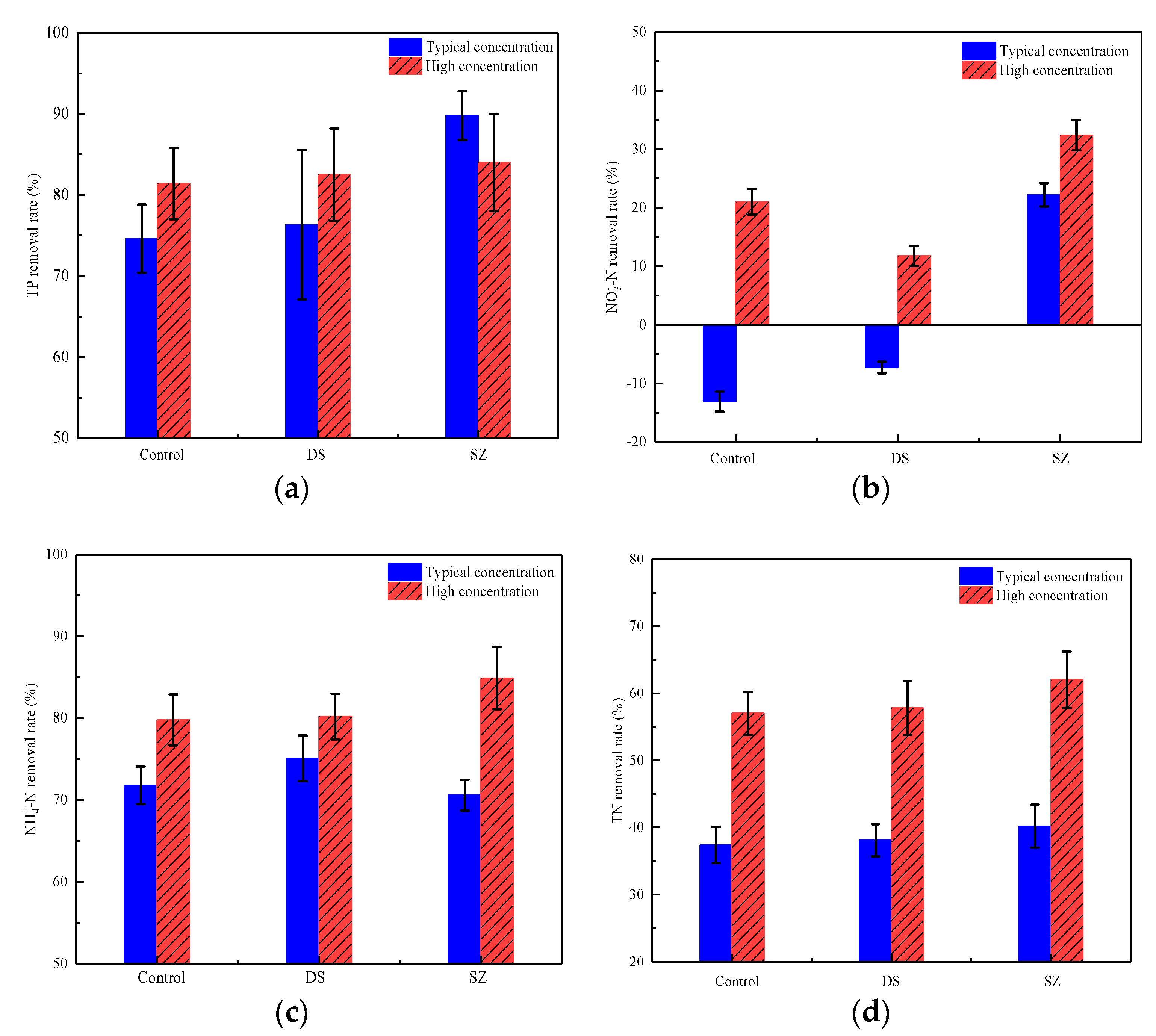
3.3. Effect of Nutrient Concentration on Pollutant Retention by Bioretention Systems
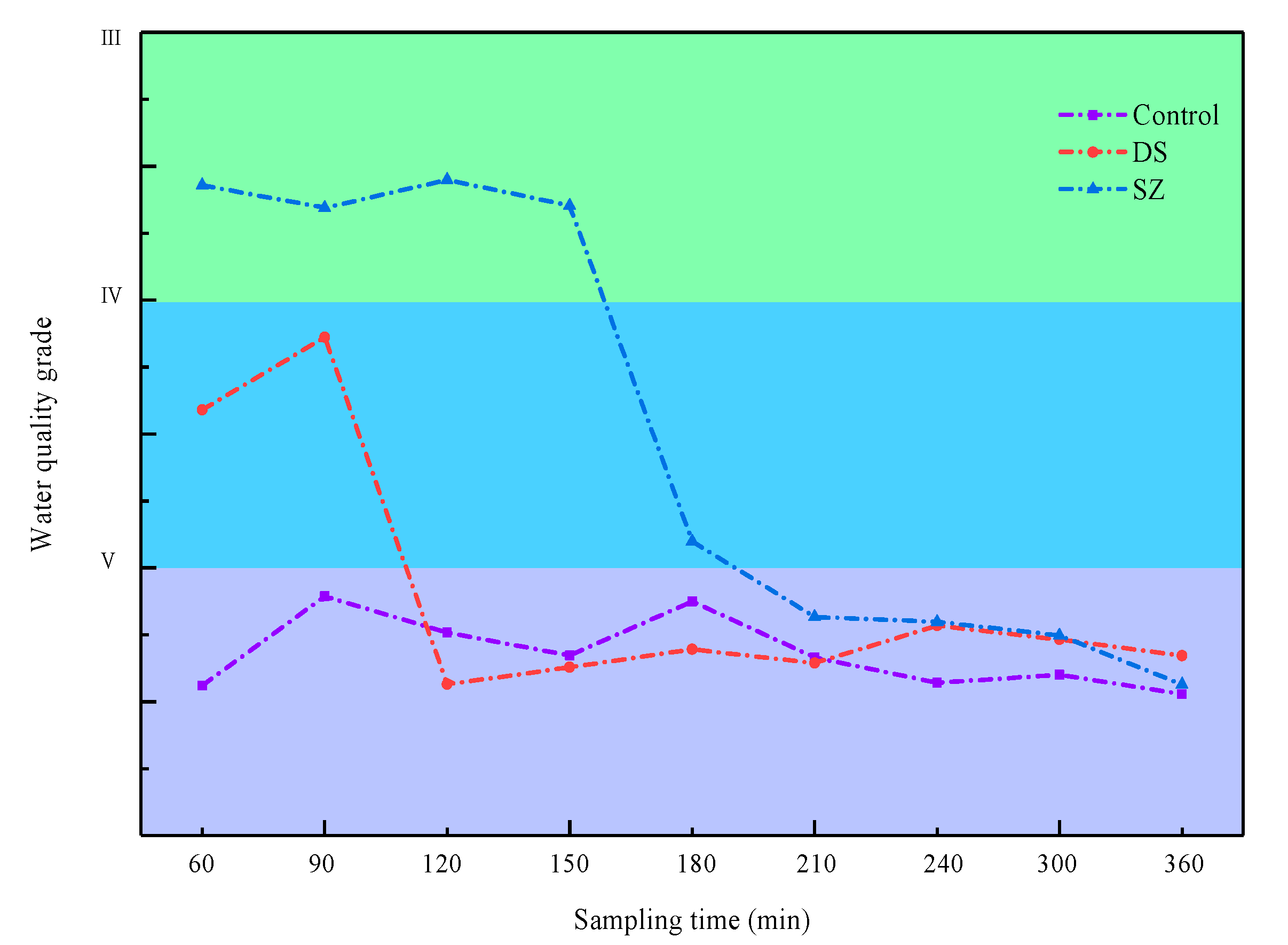
3.4. Evaluation of Water Quality in Bioretention Systems
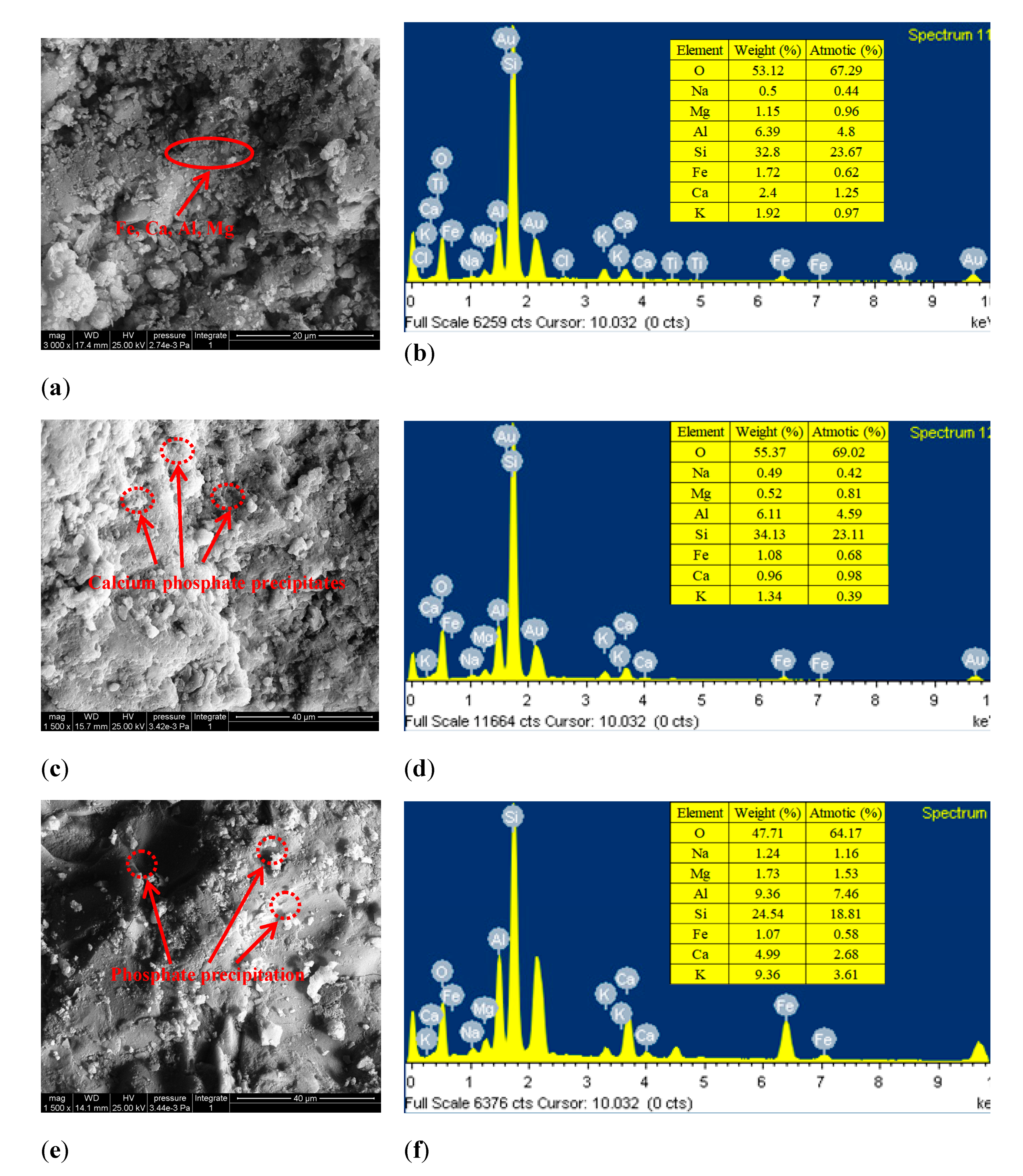
3.5. Study on the Adsorption Mechanism
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, Y.; Engel, B.A.; Flanagan, D.C.; Gitau, M.W.; McMillan, S.K.; Chaubey, I. A review on effectiveness of best management practices in improving hydrology and water quality: Needs and opportunities. Sci. Total Environ. 2017, 601–602, 580–593. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Xu, Z.; Chen, L.; Li, C. How Does Industrial Waste Gas Emission Affect Health Care Expenditure in Different Regions of China: An Application of Bayesian Quantile Regression. Int. J. Environ. Res. Public Health 2019, 16, 2748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, X.; Huang, X.; Huang, J.; Gao, X.; Chen, L. Spatial-Temporal Characteristics of Agriculture Green Total Factor Productivity in China, 1998–2016: Based on More Sophisticated Calculations of Carbon Emissions. Int. J. Environ. Res. Public Health 2019, 16, 3932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Long, C.; Wang, D.; Yang, J. Phytoremediation of cadmium (Cd) and uranium (U) contaminated soils by Brassica juncea L. enhanced with exogenous application of plant growth regulators. Chemosphere 2020, 242, 125112. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, B.A.; Gonsior, M.; Isabelle, L.M.; Luo, W.; Perraud, V.; McIntire, T.M.; Pankow, J.F.; Schmitt-Kopplin, P.; Cooper, W.J.; Simpson, A.J. A regional study of the seasonal variation in the molecular composition of rainwater. Atmos. Environ. 2013, 77, 588–597. [Google Scholar] [CrossRef]
- Pumo, D.; Arnone, E.; Francipane, A.; Caracciolo, D.; Noto, L.V. Potential implications of climate change and urbanization on watershed hydrology. J. Hydrol. 2017, 554, 80–99. [Google Scholar] [CrossRef]
- Kang, A.H.; Mao, H.W.; Li, B.; Kou, C.J.; Xu, X.L.; Jahangiri, B. Investigation of selective filtration characteristics of filter media for pavement runoff treatment. J. Clean. Prod. 2019, 235, 590–602. [Google Scholar] [CrossRef]
- Straffelini, G.; Ciudin, R.; Ciotti, A.; Gialanella, S. Present knowledge and perspectives on the role of copper in brake materials and related environmental issues: A critical assessment. Environ. Pollut. 2015, 207, 211–219. [Google Scholar] [CrossRef]
- Lange, K.; Osterlund, H.; Viklander, M.; Blecken, G.T. Metal speciation in stormwater bioretention: Removal of particulate, colloidal and truly dissolved metals. Sci. Total Environ. 2020, 724, 138121. [Google Scholar] [CrossRef]
- Chen, L.; Wang, Z.; Jing, Z.; Cao, S.; Yu, T. Accumulation and risk of triclosan in surface sediments near the outfalls of municipal wastewater treatment plants. Bull. Environ. Contam. Toxicol. 2015, 95, 525–529. [Google Scholar] [CrossRef]
- Gao, Y.; Jia, Y.L.; Yu, G.R.; He, N.P.; Zhang, L.; Zhu, B.; Wang, Y.F. Anthropogenic reactive nitrogen deposition and associated nutrient limitation effect on gross primary productivity in inland water of China. J. Clean. Prod. 2019, 208, 530–540. [Google Scholar] [CrossRef]
- Kou, C.J.; Kang, A.H.; Xiao, P.; Mikhailenko, P.; Baaj, H.; Sun, L.; Wu, Z.G. A source pollution control measure based on spatial-temporal distribution characteristic of the runoff pollutants at urban pavement sites. Appl. Sci. 2018, 8, 1802. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Zhou, S.Y.; Zhao, C.; Valeo, C. The influence of geotextile type and position in a porous asphalt pavement system on Pb (II) removal from stormwater. Water 2018, 10, 1205. [Google Scholar] [CrossRef] [Green Version]
- Beza, B.B.; Zeunert, J.; Hanson, F. The role of WSUD in contributing to sustainable urban settings. In Approaches to Water Sensitive Urban Design, Sharma, A.K., Gardner, T., Begbie, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 367–380. [Google Scholar]
- Li, Q.; Wang, F.; Yu, Y.; Huang, Z.C.; Li, M.T.; Guan, Y.T. Comprehensive performance evaluation of LID practices for the sponge city construction: A case study in Guangxi, China. J. Environ. Manag. 2019, 231, 10–20. [Google Scholar] [CrossRef]
- He, B.J.; Zhu, J.; Zhao, D.X.; Gou, Z.H.; Qi, J.D.; Wang, J.S. Co-benefits approach: Opportunities for implementing sponge city and urban heat island mitigation. Land Use Policy 2019, 86, 147–157. [Google Scholar] [CrossRef]
- Wang, J.S.; Meng, Q.L.; Zhang, L.; Zhang, Y.; He, B.J.; Zheng, S.L.; Santamouris, M. Impacts of the water absorption capability on the evaporative cooling effect of pervious paving materials. Build. Environ. 2019, 151, 187–197. [Google Scholar] [CrossRef]
- Davis, A.P.; Stagge, J.H.; Jamil, E.; Kim, H. Hydraulic performance of grass swales for managing highway runoff. Water Res. 2012, 46, 6775–6786. [Google Scholar] [CrossRef]
- Jia, H.F.; Lu, Y.W.; Yu, S.L.; Chen, Y.R. Planning of LID-BMPs for urban runoff control: The case of Beijing Olympic Village. Sep. Purif. Technol. 2012, 84, 112–119. [Google Scholar] [CrossRef]
- Grebel, J.E.; Mohanty, S.K.; Torkelson, A.A.; Boehm, A.B.; Higgins, C.P.; Maxwell, R.M.; Nelson, K.L.; Sedlak, D.L. Engineered infiltration systems for urban stormwater reclamation. Environ. Eng. Sci. 2013, 30, 437–454. [Google Scholar] [CrossRef]
- Boelee, N.C.; Temmink, H.; Janssen, M.; Buisman, C.J.N.; Wijffels, R.H. Nitrogen and phosphorus removal from municipal wastewater effluent using microalgal biofilms. Water Res. 2011, 45, 5925–5933. [Google Scholar] [CrossRef]
- Bassin, J.P.; Pronk, M.; Kraan, R.; Kleerebezem, R.; van Loosdrecht, M.C.M. Ammonium adsorption in aerobic granular sludge, activated sludge and anammox granules. Water Res. 2011, 45, 5257–5265. [Google Scholar] [CrossRef] [PubMed]
- Norton, R.A.; Harrison, J.A.; Keller, C.K.; Moffett, K.B. Effects of storm size and frequency on nitrogen retention, denitrification, and N2O production in bioretention swale mesocosms. Biogeochemistry 2017, 134, 353–370. [Google Scholar] [CrossRef]
- Chen, X.L.; Peltier, E.; Sturm, B.S.M.; Young, C.B. Nitrogen removal and nitrifying and denitrifying bacteria quantification in a stormwater bioretention system. Water Res. 2013, 47, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.B.; Li, J.K.; Li, H.E.; Li, Y.J. Remediation and accumulation characteristics of dissolved pollutants for stormwater in improved bioretention basins. Sci. Total Environ. 2019, 685, 763–771. [Google Scholar] [CrossRef]
- Fan, G.D.; Li, Z.S.; Wang, S.M.; Huang, K.S.; Luo, J. Migration and transformation of nitrogen in bioretention system during rainfall runoff. Chemosphere 2019, 232, 54–62. [Google Scholar] [CrossRef]
- Li, J.K.; Davis, A.P. A unified look at phosphorus treatment using bioretention. Water Res. 2016, 90, 141–155. [Google Scholar] [CrossRef]
- Wang, Z.; Zhong, M.G.; Chen, L. Coal-based granular activated carbon loaded with MnO2 as an efficient adsorbent for removing formaldehyde from aqueous solution. Desalin. Water Treat. 2016, 57, 13225–13235. [Google Scholar] [CrossRef]
- Wang, R.D.; Peng, Y.Z.; Cheng, Z.L.; Ren, N.Q. Understanding the role of extracellular polymeric substances in an enhanced biological phosphorus removal granular sludge system. Bioresour. Technol. 2014, 169, 307–312. [Google Scholar] [CrossRef]
- You, Z.Y.; Zhang, L.; Pan, S.Y.; Chiang, P.C.; Pei, S.L.; Zhang, S.J. Performance evaluation of modified bioretention systems with alkaline solid wastes for enhanced nutrient removal from stormwater runoff. Water Res. 2019, 161, 61–73. [Google Scholar] [CrossRef]
- Luo, Y.; Yue, X.; Duan, Y.; Zhou, A.; Gao, Y.; Zhang, X. A bilayer media bioretention system for enhanced nitrogen removal from road runoff. Sci. Total Environ. 2020, 705, 135893. [Google Scholar] [CrossRef]
- Mangum, K.R.; Yan, Q.; Ostrom, T.K.; Davis, A.P. Nutrient leaching from green waste compost addition to stormwater submerged gravel wetland mesocosms. J. Environ. Eng. 2020, 146, 04019128. [Google Scholar] [CrossRef]
- Skorobogatov, A.; He, J.; Chu, A.; Valeo, C.; Duin, B. The impact of media, plants and their interactions on bioretention performance: A review. Sci. Total Environ. 2020, 715, 136918. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.F.; Li, T.; Li, H. Characteristics of surface runoff pollution of Shanghai urban area. Environ. Sci. 2007, 28, 1430–1434. [Google Scholar]
- Li, L.Q.; Yin, C.Q. Transport and sources of runoff pollution from urban area with combined sewer system. Environ. Sci. 2009, 30, 368–375. [Google Scholar]
- Li, L.Q.; Shan, B.Q.; Zhao, J.W.; Guo, S.G.; Gao, Y. Research on stormwater runoff quality of mountain city by source area monitoring. Environ. Sci. 2012, 33, 3397–3403. [Google Scholar]
- Tirpak, R.A.; Hathaway, J.M.; Franklin, J.A. Investigating the hydrologic and water quality performance of trees in bioretention mesocosms. J. Hydrol. 2019, 576, 65–71. [Google Scholar] [CrossRef]
- Guo, X.F.; Guo, Z.H.; Zhuang, Q. Environmental assessment and evolution analysis of water quality in the Xiamen West Sea based on fuzzy mathematics. Fresen. Environ. Bull. 2019, 28, 10052–10057. [Google Scholar]
- Xiao, Y.; Huang, S.L.; Zhou, J.G.; Kong, F.Q.; Liu, M.Z.; Li, Y. Risk assessment of upper-middle reaches of Luanhe River Basin in sudden water pollution incidents based on control units of water function areas. Water 2018, 10, 1268. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Qiu, Z.Z.; Zhang, J.D.; Liu, C.Y.; Cai, Y.; Xiao, M.S.; Zhu, L.Y. Temporal variation of major nutrients and probabilistic eutrophication evaluation based on stochastic-fuzzy method in Honghu Lake, Middle China. Sci. China Technol. Sci. 2019, 62, 417–426. [Google Scholar] [CrossRef]
- Liu, B.; Huang, J.J.; McBean, E.; Li, Y. Risk assessment of hybrid rain harvesting system and other small drinking water supply systems by game theory and fuzzy logic modeling. Sci. Total Environ. 2020, 708, 134436. [Google Scholar] [CrossRef]
- Singh, S.; Ghosh, N.C.; Gurjar, S.; Krishan, G.; Kumar, S.; Berwal, P. Index-based assessment of suitability of water quality for irrigation purpose under Indian conditions. Environ. Monit. Assess. 2018, 190, 29. [Google Scholar] [CrossRef] [PubMed]
- Lucas, W.C.; Greenway, M. Nutrient retention in vegetated and nonvegetated bioretention mesocosms. J. Irrig. Drain. Eng. 2008, 134, 613–623. [Google Scholar] [CrossRef]
- Davis, A.P.; Shokouhian, M.; Sharma, H.; Minami, C. Water quality improvement through bioretention media: Nitrogen and phosphorus removal. Water Environ. Res. 2006, 78, 284–293. [Google Scholar] [CrossRef]
- Rosenquist, S.E.; Hession, W.C.; Eick, M.J.; Vaughan, D.H. Variability in adsorptive phosphorus removal by structural stormwater best management practices. Ecol. Eng. 2010, 36, 664–671. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.J.; Guo, G.Y.; Sun, S.Q.; Hu, C.W.; Liu, J. Co-pelletization of microalgae and fungi for efficient nutrient purification and biogas upgrading. Bioresour. Technol. 2019, 289, 121656. [Google Scholar] [CrossRef] [PubMed]
- Okochi, N.C. Phosphorus Removal from Stormwater Using Electric ARC Furnace Steel Slag. Ph.D. Thesis, University of Regina, Regina, SK, Canada, 2013. [Google Scholar]
- Huang, H.M.; Xiao, D.; Pang, R.; Han, C.C.; Ding, L. Simultaneous removal of nutrients from simulated swine wastewater by adsorption of modified zeolite combined with struvite crystallization. Chem. Eng. J. 2014, 256, 431–438. [Google Scholar] [CrossRef]



| Media Layer | Filling Format | Height | ||
|---|---|---|---|---|
| Control | DS | SZ | ||
| Water-retention layer | Local soil (20%) mixed with sand (80%) | 10 cm | ||
| Adsorption layer | Zeolite: Volcanic rock: Sponge iron = 1:1:1 (volume ratio), layered filling, grain size of 1–2 mm | Zeolite: Volcanic rock: Sponge iron = 1:1:1 (volume ratio), mixed filling, grain size of 1–2 mm | 60 cm | |
| Filter layer | A non-woven permeable geotextile membrane | / | ||
| Catchment layer | Crushed limestone gravel (grain size of 5–10 mm, void space of 34%) | 5 cm | ||
| Project | Components | |||||
|---|---|---|---|---|---|---|
| TP (mg/L) | NH4+-N (mg/L) | NO3−-N (mg/L) | COD (mg/L) | pH | Temp (°C) | |
| Typical concentration (normal event) | 1.2 ± 0.1 | 3.0 ± 0.3 | 1.5 ± 0.1 | 100.0 ± 3.0 | 7.0 ± 0.3 | 22.0 ± 2.0 |
| High concentration (extreme event) | 2.4 ± 0.1 | 6.0 ± 0.3 | 3.0 ± 0.4 | 200.0 ± 6.0 | 7.0 ± 0.3 | 22.0 ± 2.0 |
| Source | KH2PO4 | NH4Cl | KNO3 | NaNO2 | NaOH/HCl | / |
| Recurrence Interval (Year) | Percent Chance of Occurrence in Any Year | Mean Rainfall Intensity (mm/min) | Rain Intensity of Six Hours (L/s·ha) | Rainfall in Six Hours (mm) |
|---|---|---|---|---|
| 1a | 100 | 0.1532 | 25.5436 | 55.1689 |
| 5a | 20 | 0.2429 | 40.4865 | 87.4333 |
| 10a | 10 | 0.2815 | 46.9206 | 101.3289 |
| Sampling Time (min) | Grade | Control | DS | SZ | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| NH4+-N | TN | TP | NH4+-N | TN | TP | NH4+-N | TN | TP | ||
| 60 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0.3948 | 0 | 0 |
| II | 0.6195 | 0 | 0 | 0.8675 | 0 | 0 | 0.6052 | 0 | 0.6027 | |
| III | 0.3805 | 0 | 0.6856 | 0.1325 | 0 | 0.9124 | 0 | 0.169 | 0.3973 | |
| IV | 0 | 0 | 0.3144 | 0 | 0.152 | 0.0876 | 0 | 0.831 | 0 | |
| V | 0 | 0 | 0 | 0 | 0.848 | 0 | 0 | 0 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 90 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0.2621 | 0 | 0 | 0.3861 | 0 | 0 | 0.8019 | 0 | 0.2807 | |
| III | 0.7379 | 0 | 0.8998 | 0.6139 | 0 | 0.3202 | 0.1981 | 0 | 0.7193 | |
| IV | 0 | 0 | 0.1002 | 0 | 0.341 | 0.6798 | 0 | 0.656 | 0 | |
| V | 0 | 0 | 0 | 0 | 0.659 | 0 | 0 | 0.344 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 120 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0.0287 | 0 | 0 | 0.5174 | 0 | 0 | 0.4226 | 0 | 0 | |
| III | 0.9713 | 0 | 0.5218 | 0.4826 | 0 | 0.5218 | 0.5774 | 0 | 0.9124 | |
| IV | 0 | 0 | 0.4782 | 0 | 0 | 0.4782 | 0 | 0.917 | 0.0876 | |
| V | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.083 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |
| 150 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0.1308 | 0 | 0 | 0.3424 | 0 | 0.0007 | |
| III | 0.6276 | 0 | 0.2698 | 0.8692 | 0 | 0.0556 | 0.6576 | 0 | 0.9993 | |
| IV | 0.3724 | 0 | 0.7302 | 0 | 0 | 0.9444 | 0 | 0.503 | 0 | |
| V | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0.497 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |
| 180 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| III | 0.0572 | 0 | 0.0556 | 0.7826 | 0 | 0.1564 | 0.9252 | 0 | 0.7612 | |
| IV | 0.9428 | 0 | 0.9444 | 0.2174 | 0 | 0.8436 | 0.0748 | 0 | 0.2388 | |
| V | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
| 210 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| III | 0.1936 | 0 | 0 | 0.5408 | 0 | 0 | 0.9996 | 0 | 0.4588 | |
| IV | 0.8064 | 0 | 0.6524 | 0.4592 | 0 | 0.8036 | 0.0004 | 0 | 0.5412 | |
| V | 0 | 0 | 0.3476 | 0 | 0 | 0.1964 | 0 | 0 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
| 240 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| III | 0 | 0 | 0 | 0.1502 | 0 | 0 | 0.7454 | 0 | 0.2446 | |
| IV | 0.741 | 0 | 0.161 | 0.8498 | 0 | 0.6524 | 0.2546 | 0 | 0.7554 | |
| V | 0.259 | 0 | 0.839 | 0 | 0 | 0.3476 | 0 | 0 | 0 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
| 300 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| III | 0 | 0 | 0 | 0.051 | 0 | 0 | 0.5656 | 0 | 0 | |
| IV | 0.5674 | 0 | 0.4256 | 0.949 | 0 | 0.5642 | 0.4344 | 0 | 0.9548 | |
| V | 0.4326 | 0 | 0.5744 | 0 | 0 | 0.4358 | 0 | 0 | 0.0452 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
| 360 | I | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| II | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| III | 0 | 0 | 0 | 0 | 0 | 0 | 0.7702 | 0 | 0 | |
| IV | 0.3318 | 0 | 0.2996 | 0.6666 | 0 | 0.5138 | 0.2298 | 0 | 0.7532 | |
| V | 0.6682 | 0 | 0.7004 | 0.3334 | 0 | 0.4862 | 0 | 0 | 0.2468 | |
| Worse V | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
| Sampling Time (min) | Control | DS | SZ | ||||||
|---|---|---|---|---|---|---|---|---|---|
| NH4+-N | TN | TP | NH4+-N | TN | TP | NH4+-N | TN | TP | |
| 60 | 0.1468 | 0.5768 | 0.2764 | 0.1342 | 0.5836 | 0.2822 | 0.1214 | 0.6192 | 0.2594 |
| 90 | 0.1927 | 0.5635 | 0.2438 | 0.1802 | 0.4812 | 0.3386 | 0.1672 | 0.5755 | 0.2573 |
| 120 | 0.1867 | 0.5605 | 0.2528 | 0.1543 | 0.5551 | 0.2907 | 0.2152 | 0.4825 | 0.3024 |
| 150 | 0.2129 | 0.5311 | 0.2561 | 0.1694 | 0.5295 | 0.3011 | 0.2124 | 0.5215 | 0.2660 |
| 180 | 0.2398 | 0.5179 | 0.2423 | 0.2000 | 0.5243 | 0.2757 | 0.2237 | 0.5268 | 0.2495 |
| 210 | 0.2179 | 0.5104 | 0.2717 | 0.2038 | 0.5129 | 0.2833 | 0.1929 | 0.5421 | 0.2650 |
| 240 | 0.2453 | 0.4549 | 0.2998 | 0.2324 | 0.4831 | 0.2844 | 0.2162 | 0.5031 | 0.2807 |
| 300 | 0.2611 | 0.4626 | 0.2764 | 0.2281 | 0.4957 | 0.2762 | 0.2108 | 0.5091 | 0.2801 |
| 360 | 0.2699 | 0.4548 | 0.2753 | 0.2584 | 0.4666 | 0.2750 | 0.1844 | 0.5198 | 0.2957 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, H.; Guan, L.; Jing, Z.; He, B.; Cao, X.; Zhang, Z.; Tao, M. Performance Evaluation of Enhanced Bioretention Systems in Removing Dissolved Nutrients in Stormwater Runoff. Appl. Sci. 2020, 10, 3148. https://doi.org/10.3390/app10093148
Luo H, Guan L, Jing Z, He B, Cao X, Zhang Z, Tao M. Performance Evaluation of Enhanced Bioretention Systems in Removing Dissolved Nutrients in Stormwater Runoff. Applied Sciences. 2020; 10(9):3148. https://doi.org/10.3390/app10093148
Chicago/Turabian StyleLuo, Hui, Lin Guan, Zhaoqian Jing, BaoJie He, Xinyue Cao, Zeyu Zhang, and Mengni Tao. 2020. "Performance Evaluation of Enhanced Bioretention Systems in Removing Dissolved Nutrients in Stormwater Runoff" Applied Sciences 10, no. 9: 3148. https://doi.org/10.3390/app10093148
APA StyleLuo, H., Guan, L., Jing, Z., He, B., Cao, X., Zhang, Z., & Tao, M. (2020). Performance Evaluation of Enhanced Bioretention Systems in Removing Dissolved Nutrients in Stormwater Runoff. Applied Sciences, 10(9), 3148. https://doi.org/10.3390/app10093148








