Development of a Lightweight Tungsten Shielding Fiber That Can Be Used for Improving the Performance of Medical Radiation Shields
Abstract
:1. Introduction
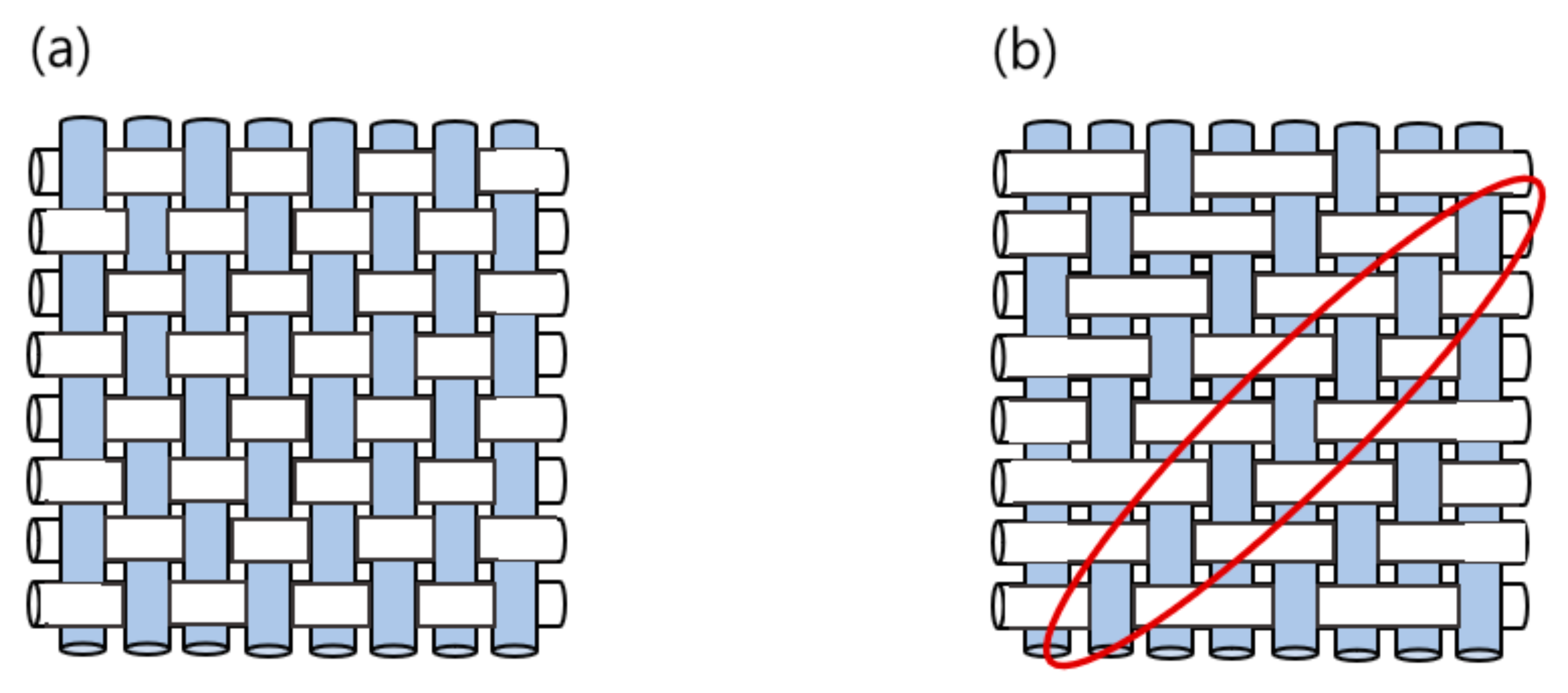
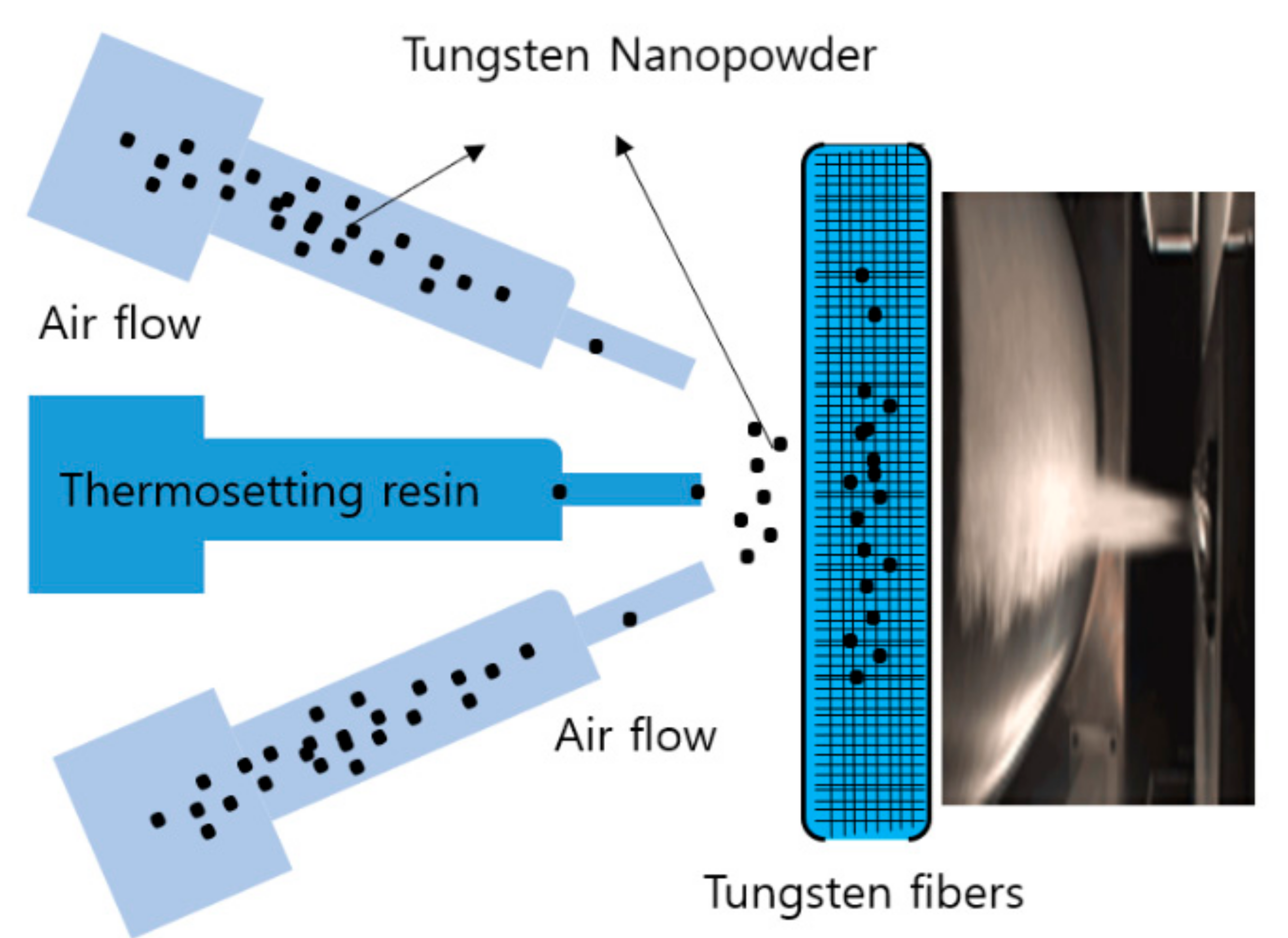
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kalra, M.K.; Maher, M.M.; Toth, T.L.; Hamberg, L.M.; Blake, M.A.; Shepard, J.; Saini, S. Strategies for CT radiation dose optimization. Radiology 2004, 230, 619–628. [Google Scholar] [CrossRef]
- Morishima, Y.; Chida, K.; Katahira, Y. The effectiveness of additional lead-shielding drape and low pulse rate fluoroscopy in protecting staff from scatter radiation during cardiac resynchronization therapy (CRT). Jpn. J. Radiol. 2019, 37, 95–101. [Google Scholar] [CrossRef]
- Hunter, D.; Mauldon, E.; Anderson, N. Cost-containment in hypofractionated radiation therapy: A literature review. J. Med. Radiat. Sci. 2018, 65, 148–157. [Google Scholar] [CrossRef] [Green Version]
- Henry, D.; Royal, M.D. Effects of low-level radiation—What’s new? Semin. Nucl. Med. 2008, 38, 392–402. [Google Scholar] [CrossRef] [Green Version]
- Cooper, J. The 2007 Recommendations of the International Commission on Radiological Protection; ICRP Publication 103; ICPR: Chilton, UK, 2007. [Google Scholar] [CrossRef]
- Forward, H.W.; Cardew, P.; Smith, B.; Clack, L.; McWhirter, K.; Johnson, S.; Wessel, K. A comparison of dose savings of lead and lightweight aprons for shielding of 99m-Technetium radiation. Radiat. Prot. Dosim. 2007, 124, 89–96. [Google Scholar] [CrossRef]
- McCaffrey, J.P.; Tessier, F.; Shen, H. Radiation shielding materials and radiation scatter effects for interventional radiology (IR) physicians. Radiat. Meas. Phys. 2021, 39, 4537–4546. [Google Scholar] [CrossRef]
- Cai, Y.; Hu, H.; Lu, S.; Jia, Q. Optimization of radiation shielding material aiming at compactness, lightweight, and low activation for a vehicle-mounted accelerator-driven DT neutron source. Appl. Radiat. Isot. 2018, 135, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Adlienė, D.; Gilys, L.; Griškonis, E. Development and characterization of new tungsten and tantalum containing composites for radiation shielding in medicine. Nucl. Inst. Methods Phys. Res. B 2020, 467, 21–26. [Google Scholar] [CrossRef]
- Wang, Y.; Zhong, R.; Li, Q.; Liao, J.; Liu, N.; Joshi, N.S.; Shi, B.; Liao, X.; Guo, J. Lightweight and Wearable X-Ray Shielding Material with Biological Structure for Low Secondary Radiation and Metabolic Saving Performance. Adv. Mater. Technol. 2020, 5, 1–8. [Google Scholar] [CrossRef]
- Shi, L.; Tashiro, S. Estimation of the effects of medical diagnostic radiation exposure based on DNA damage. J. Radiat. Res. 2018, 59, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aral, N.; Nergis, F.B.; Candan, C. An alternative X-ray shielding material based on coated textiles. Text. Res. J. 2015, 86, 803–811. [Google Scholar] [CrossRef]
- Yang, J.P.; Chen, Z.K.; Yang, G.; Fu, S.Y.; Ye, L. Simultaneous improvements in the cryogenic tensile strength ductility and impact strength of epoxy resins by a hyperbranched polymer. Polymer 2008, 49, 90–96. [Google Scholar] [CrossRef]
- Nadin, J.A.; Natasha, A.M.; Noorfatin, B.A.; Rafidah, Z. Tungsten-based material as promising new lead-free gamma radiation shielding material in nuclear medicine. Phys. Med. 2020, 78, 48–57. [Google Scholar] [CrossRef]
- Hari, P.K. 1—Types and properties of fibres and yarns used in weaving. In Woven Textiles, 2nd ed.; Woodhead Publishing: Cambridge, UK, 2012; pp. 3–34. [Google Scholar] [CrossRef]
- Maghrabi, H.A.; Vijayan, A.; Deb, P.; Wang, L. Bismuth oxide-coated fabrics for X-ray shielding. Text. Res. J. 2015, 86, 649–658. [Google Scholar] [CrossRef]
- Jahan, I. Effect of Fabric Structure on the Mechanical Properties of Woven Fabrics. Adv. Res. Text. Eng. 2017, 2, 1–4. [Google Scholar] [CrossRef]
- Maghrabi, H.A.; Vijayan, A.; Wang, L.; Deb, P. Design of seamless knitted radiation shielding garments with 3D body scanning technology. Procedia Technol. 2015, 20, 123–125. [Google Scholar] [CrossRef] [Green Version]
- Aral, N.; Nerg, F.B.; Candan, C. Investigation of X-ray attenuation and the flex resistance properties of fabrics coated with tungsten and barium sulphate additives. Tekst. Konfeksiyon 2016, 26, 166–171. [Google Scholar]
- Maghrabi, H.A.; Vijayan, A.; Mohaddes, F.; Deb, P.; Wang, L. Evaluation of X-ray radiation shielding performance of barium sulphate-coated fabrics. Fibers Polym. 2016, 17, 2047–2054. [Google Scholar] [CrossRef]
- Kim, S.C. Development of air pressure mirroring particle dispersion method for producing high-density tungsten medical radiation shielding film. Sci. Rep. 2021, 11, 485. [Google Scholar] [CrossRef] [PubMed]
- Neto, A.T.N.; Faria, L.O. Construction and calibration of a multipurpose instrument to simultaneously measure dose, voltage and half-value layer in X-ray emission equipment. Radiat. Meas. 2014, 71, 178–182. [Google Scholar] [CrossRef]
- Sakar, E.; Ozpolat, O.F.; Alım, B.; Sayyed, M.I.; Kurudirek, M. Phy-X/PSD: Development of a user-friendly online software for calculation of parameters relevant to radiation shielding and dosimetry. Radiat. Phys. Chem. 2020, 166, 108496. [Google Scholar] [CrossRef]
- al-Dhuhaibat, M.J.R. Study of the shielding properties for some composite materials manufactured from polymer epoxy supported by cement, aluminum, iron and lead against gamma rays of the cobalt radioactive source (Co-60). Int. J. Appl. Innov. Eng. Manag. 2015, 4, 90–98. [Google Scholar]
- Hubbell, J.H. Photon mass attenuation and energy absorption coefficients from 1 keV to 20 MeV. Int. Appl. Radiat. Isot. 1982, 33, 1269–1290. [Google Scholar] [CrossRef]
- Pingale, P.L. Formulation, evaluation or fast dissolving tablet of anti-HIV drugs as fixed dose combination: Use of freeze-dried powder of Annona reticulata and comparison with synthetic super disintegrants. J. Pharm. Sci. Innov. 2019, 8, 38–41. [Google Scholar] [CrossRef]
- Chiang, H.W.; Liu, Y.L.; Chen, T.R.; Chen, C.L.; Chiang, H.J.; Chao, S.Y. Scattered radiation doses absorbed by technicians at different distances from X-ray exposure: Experiments on prosthesis. Bio-Med. Mater. Eng. 2015, 26, 1641–1650. [Google Scholar] [CrossRef] [Green Version]
- Verdun, F.R.; Bochud, F.; Gundinchet, F.; Aroua, A.; Schnyder, P.; Meuli, R. Quality Initiatives Radiation Risk: What You Should Know to Tell Your Patient. Radiographics 2008, 28, 1807–1816. [Google Scholar] [CrossRef] [Green Version]
- Soylu, H.M.; Lambrecht, F.Y.; Ersöz, O.A. Gamma radiation shielding efficiency of a new lead-free composite material. J. Radioanal Nucl. Chem. 2015, 305, 529–534. [Google Scholar] [CrossRef]
- Cho, J.H.; Kim, M.S.; Rhim, J.D. Comparison of radiation shielding ratios of nano-sized bismuth trioxide and molybdenum. Radiat. Eff. Defects Solids 2015, 170, 651–658. [Google Scholar] [CrossRef]
- Monaco, M.G.L.; Carta, A.; Tamhid, T.; Porru, S. Anti-X apron wearing and musculoskeletal problems among healthcare workers: A systematic scoping review. Int. J. Environ. Res. Public Health 2020, 17, 5877. [Google Scholar] [CrossRef]
- Ross, A.M.; Segal, J.; Borenstein, D.; Jenkins, E.; Cho, S. Prevalence of spinal disc disease among interventional cardiologists. Am. J. Card. 1997, 79, 68–70. [Google Scholar] [CrossRef]
- Kim, S.C. Double-layered fiber for lightweight flexible clothing providing shielding from low-dose natural radiation. Sci. Rep. 2021, 11, 3676. [Google Scholar] [CrossRef] [PubMed]
- Tsapaki, V. Radiation protection in dental radiology—Recent advances and future directions. Phys. Med. 2017, 44, 222–226. [Google Scholar] [CrossRef]
- Bychkov, A.N.; Dzhardimalieva, G.I.; Fetisov, G.P.; Valskiy, V.V.; Golubeva, N.D.; Pomogailo, A.D. Synthesis and characterization of metal–polymer nanocomposites with radiation-protective properties. Russ. Metall. 2016, 13, 1207–1213. [Google Scholar] [CrossRef]
- Lu, H.; Boyd, C.; Dawson, J. Lightweight lead aprons: The emperor’s new clothes in the angiography suite? Eur. J. Vasc. Endovasc. Surg. 2019, 57, 730–739. [Google Scholar] [CrossRef]
- Hobson, J.; Cooper, A. Radiation protection and shielding design—Strengthening the link. Radiat. Prot. Dosim. 2005, 115, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Schueler, B.A. Operator shielding: How and why. Tech. Vasc. Interv. Radiol. 2010, 13, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Roshani, M.; Phan, G.T.; Ali, P.J.M.; Roshani, G.H.; Hanus, R.D. Evaluation of flow pattern recognition and void fraction measurement in two phase flow independent of oil pipeline’s scale layer thickness. Alex. Eng. J. 2021, 60, 1955–1966. [Google Scholar] [CrossRef]
- Roshani, G.H.; Roshani, S. Online measuring density of oil products in annular regime of gas-liquid two phase flows. Measurement 2018, 129, 296–301. [Google Scholar] [CrossRef]
- Meisinger, Q.C.; Stahl, C.M.; Andre, M.P.; Kinney, T.B.; Newton, T.G. Radiation protection for the fluoroscopy operator and staff. Am. J. Roentgenol. 2016, 207, 745–754. [Google Scholar] [CrossRef]
- Livingstone, R.S.; Varghese, A.; Keshava, S.N. A study on the use of radiation-protective apron among interventionists in radiology. J. Clin. Imaging Sci. 2018, 8, 1–4. [Google Scholar] [CrossRef]
- Hubbert, T.E.; Vucich, J.J.; Armstrong, M.R. Lightweight aprons for protection against scattered radiation during fluoroscopy. Am. J. Roentgenol. 1993, 161, 1079–1084. [Google Scholar] [CrossRef] [PubMed] [Green Version]






| PE-Tungsten Wire Composite Yarn | Shielding Fiber | |||||
|---|---|---|---|---|---|---|
| Fineness | Tensile Strength (g/d) | Elongation at Break (%) | Thread Count (Thread/Inch) | Weight (g/m2) | Thickness (mm) | |
| Warp | Weft | |||||
| 501.1 | 1.45 | 4.2 | 80 | 60 | 112–121 | 0.20–0.21 |
| Radiation Type | Effective X-ray Energy (keV) | Mean of Exposure (µR) | Shielding Rate (%) | |||
|---|---|---|---|---|---|---|
| Nothing | Tungsten Fiber | Tungsten Powder Fiber | Tungsten Fiber | Tungsten Powder Fiber | ||
| X-ray | 24.6 | 106.90 | 72.01 | 55.49 | 32.64 | 48.09 |
| 28.7 | 381.83 | 284.2 | 231.27 | 25.53 | 39.40 | |
| 32.5 | 799.70 | 632.82 | 545.80 | 20.87 | 31.75 | |
| 48.5 | 1318.33 | 1082.01 | 984.00 | 17.96 | 25.36 | |
| 54.9 | 1648.33 | 1407.70 | 1312.67 | 14.60 | 20.36 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.-C. Development of a Lightweight Tungsten Shielding Fiber That Can Be Used for Improving the Performance of Medical Radiation Shields. Appl. Sci. 2021, 11, 6475. https://doi.org/10.3390/app11146475
Kim S-C. Development of a Lightweight Tungsten Shielding Fiber That Can Be Used for Improving the Performance of Medical Radiation Shields. Applied Sciences. 2021; 11(14):6475. https://doi.org/10.3390/app11146475
Chicago/Turabian StyleKim, Seon-Chil. 2021. "Development of a Lightweight Tungsten Shielding Fiber That Can Be Used for Improving the Performance of Medical Radiation Shields" Applied Sciences 11, no. 14: 6475. https://doi.org/10.3390/app11146475
APA StyleKim, S.-C. (2021). Development of a Lightweight Tungsten Shielding Fiber That Can Be Used for Improving the Performance of Medical Radiation Shields. Applied Sciences, 11(14), 6475. https://doi.org/10.3390/app11146475






