Abstract
Nanoparticles of the semiconductor ZnAl2O4 were prepared using a microwave-assisted wet chemistry method in the presence of ethylenediamine and calcination at 250 °C. The material’s crystallinity and purity were verified by X-ray diffraction. The pure phase of the ZnAl2O4 presented a cubic crystalline structure with cell parameters a = 8.087 Å and space group Fd-3m (227). Dynamic tests in propane atmospheres were carried out on pellets (~500 µm in diameter) manufactured with ZnAl2O4 powders. In the tests, the oxide showed variations with time in electrical resistance when injecting air-propane at an operating temperature of 250 °C. The pellets showed good stability, high sensitivity, and an optimal dynamic response as a function of time. On the other hand, a mathematical model was proposed to describe the chemical sensor’s dynamic behavior based on the electrical response and linear systems theory. The sensor’s transient response was obtained with the model by exposing the oxide to air and propane gas; its stability was checked, and the stabilization time was calculated. Subsequently, an operating point was selected, and, with it, a propane gas detector was designed. The sensor operated flawlessly at 250 °C at a concentration of 1000 ppm, with a response time of three seconds. The developed device is inexpensive and easy to implement.
1. Introduction
For some years now, the literature has reported that spinel-type transition metal oxides conform to the general formula XM2O4 [1,2], where X can be the divalent cations Fe2+, Mg2+, Mn2+, Co2+, and Ni2+. M can be substituted for the trivalent cations Cr3+, Ga3+, and Fe3+ [2]. These compounds show interesting physical (such as magnetic and refractory) and chemical properties that make them suitable for several technological applications [3,4,5]. Moreover, they possess high resistance to acids, high melting points, relatively high surface areas, and good catalytic properties [1]. The good properties of these materials are due mainly to the nanometric size of their particles [6]. Wet chemistry methods are employed for obtaining particle sizes of less than 100 nm from spinel-type materials [3], since it allows the control of the physical and chemical parameters involved during the synthesis process [2,3,7].
The ZnAl2O4 is a spinel-type ternary compound [1,2,8], a ceramic semiconductor material with a bandgap of 3.5–3.9 eV [9,10]. According to the literature, ZnAl2O4 can be adjusted to the aluminates’ formula XAl2O4 [5,11], where X is replaced by the divalent cation Zn2+ [3,5]. However, it can also be substituted for the divalent ions Co2+, Ni2+, and Cu2+ [6]. The material typically has a crystalline phase with a cubic-type crystallographic system with space group Fd-3m [3,10,12]. It has been intensively studied due to its attractive properties, such as high thermal stability, low-temperature sinterability, low surface acidity, good diffusion, and high quantum yields [13], which have promoted its technological application in different fields. These applications include optical coatings, photo-electronic devices, display technologies, ceramics, and sensors [1,9,14]. Some reports have stated that the ZnAl2O4 possesses high sensitivity, good linearity, small hysteresis, and short response and recovery times in humid [9] and propane [1] atmospheres, which is attributed to the porosity and the nanostructures (such as nanorods) obtained during the compound’s synthesis [9]. The goal now is to apply new materials (such as the semiconductors) to toxic gas detection systems [15,16,17]. These devices are intended to provide safe zones for humans in C3H8, CO, or CO2 atmospheres, among others [18,19,20].
Our proposed detector consists of two essential parts: the sensor and the electronic circuit. The sensor detects the chemical “signals” (reactions) of the gases. Analog or digital, the electronic circuit transforms the chemical signal into an electrical signal and records it. When the sensor signal is processed with analog circuits, the electronic circuit design is specific because it depends on the signal’s features. For other characteristics, it would not be functional. However, its advantages are easy construction, fast response, low cost, and high sensitivity.
For its application as a detector of toxic gases, our group synthesized powders of the semiconductor oxide ZnAl2O4 at a relatively low temperature applying a wet chemistry process assisted with microwave radiation. Pellets were manufactured from the powders to study the oxide’s ability to detect propane (C3H8) atmospheres in direct current (DC). The oxide showed good thermal stability, high sensitivity, and excellent dynamic response at an operating temperature of 250 °C. We found that the ZnAl2O4 is suitable for being applied as a propane gas sensor. Furthermore, we modeled its electrical response based on the time-invariant linear systems theory. Through the model, the transient response of the sensor was verified by exposing it to air and propane gas, checking its stability (and stabilization time). With that, an operating point was found for the detector’s design, whose features were, in summary, an operating temperature of 250 °C, a concentration of 1000 ppm, size of 10 cm × 10 cm, low cost, applicable to high-temperature processes, and short response times.
2. Materials and Methods
2.1. Synthesis of ZnAl2O4 Powders
For the synthesis of ZnAl2O4 nanoparticles, 3.75 g of Al (NO3)3·9H2O (Sigma-Aldrich, Guadalajara, Mexico), 1.48 g of Zn (NO3)2·6H2O (Jalmek, Guadalajara, Mexico), and 2 mL of ethylenediamine (Sigma-Aldrich, Guadalajara, Mexico) were used. Five milliliters of ethyl alcohol (CTR) were added to each reagent and stirred for 20 min. Later, the solutions were mixed, still stirring, obtaining a suspension (colloidal dispersion) of very fine particles. The solution was left stirring at 375 rpm for 24 h at room temperature. Subsequently, the solvent was evaporated using microwave radiation using a domestic oven (LG, model MS1147 X). The solution was radiated 40 times at 140 W during 70 s/cycle. After evaporation, the precursor material was dried at 200 °C for 8 h and calcined at 250 °C for 5 h, applying a heating ramp of 100 °C/h in air. The material’s calcination process was completed using a programmable control muffle (Novatech, Tlaquepaque, Mexico).
2.2. Gas Sensing Tests
Before the electrical sensing tests, a crystallographic characterization of the ZnAl2O4 powders calcined at 250 °C was carried out by X-ray powder diffraction using a Panalytical Empyren device with CuKα radiation (λ = 1.546 Å). The 2θ continuous scan range was from 10 to 90° with steps of 0.026° at a rate of 30 s/step.
The gas sensing tests were performed on the surface of pellets manufactured with the ZnAl2O4 powders. For this, 0.3 g of the powders were compressed at a pressure of 11 tons for 5 min using a Simplex Ital Equip-25 tons hydraulic equipment. The dimensions of the pellets were 12 mm in diameter and 0.5 mm in thickness. Two ohmic contacts made from colloidal silver paint (Alfa Aesar, 99%, Mexico City, Mexico) were placed on the pellets’ surface so that there was good contact between the surface and the electrons present during the experiments. The pellets were introduced into a small metal box (with a volume of 19 cm3) located inside the measuring chamber (with a capacity of 10−3 torr). For the dynamic tests, the small box had two holes for the inlet and outlet of the test gas. One hole also allowed the introduction of the pellets’ electrodes. The gas discharged from the metal box was evacuated by a system installed in the vacuum chamber. A Leybold TM20 electronic detector monitored the test gas partial pressure. The variations in electrical resistance were recorded with a Keithley 2001 multimeter coupled to a control and data acquisition system using the LabView software (National Instruments, Mexico City, Mexico). Mass flow regulators (Brooks Instruments) with a capacity of 2600 cm3/min (model GF100CXXC-SH452.6L, Mexico City, Mexico) and 10 cm3/min (model GF100CXXC-SH40010C, Mexico City, Mexico) were used to control the propane flows.
3. Results
3.1. XRD Analysis
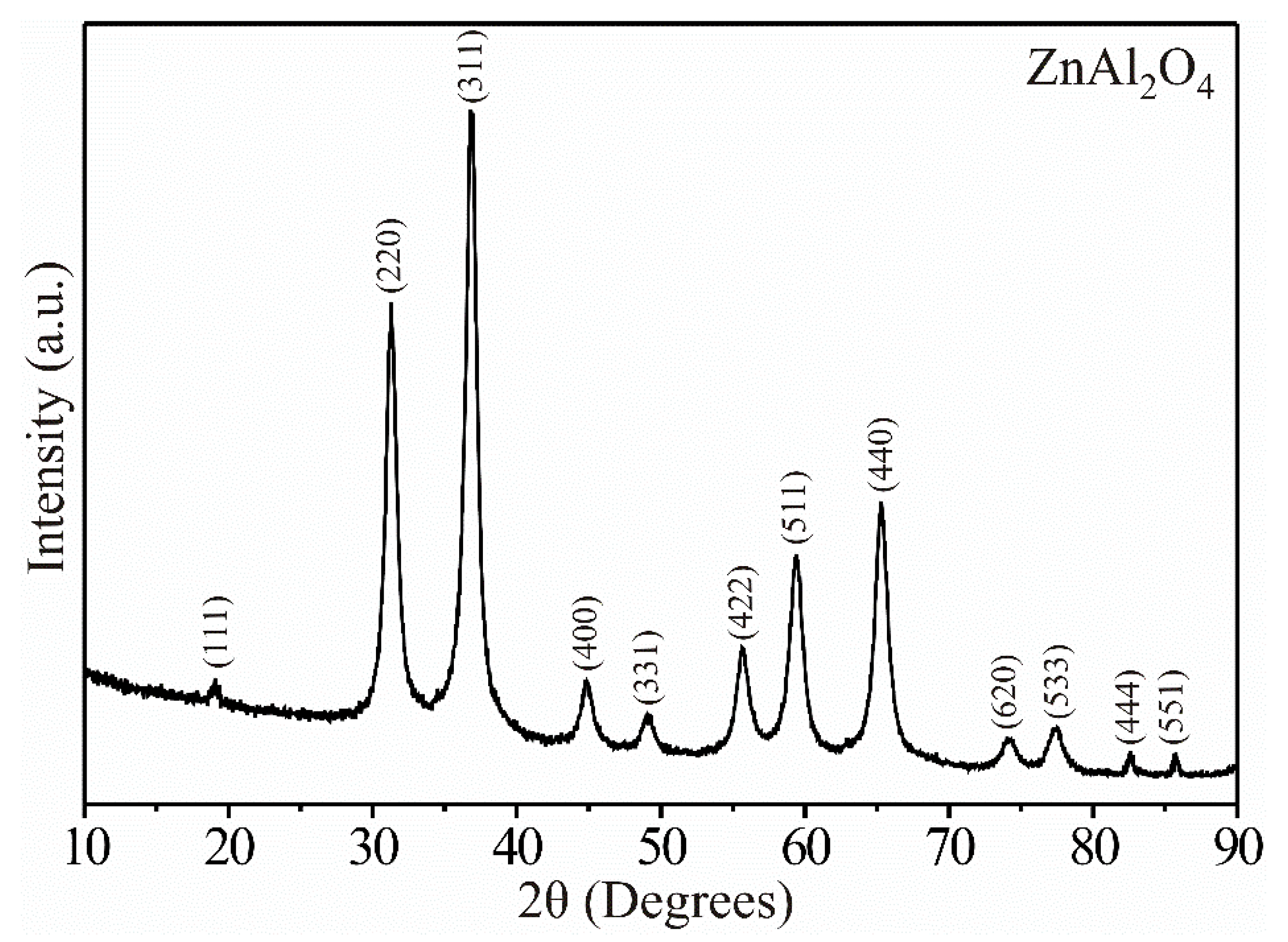
Figure 1 shows XRD results from the precursor material treated at 250 °C. As expected, at that temperature, the ZnAl2O4 showed high-intensity peaks that suggested its high purity and crystallinity. The peaks’ height and width were an indication that the material was made up of nanometric-sized particles [21]. The compound’s pure phase was identified through PDF # 65-3104, indicating that the ZnAl2O4 belongs to the family of spinel-type materials, with a cubic crystalline structure (a = 8.087 Å) and a spatial group Fd-3m (227) [2,3,5]. Our results were consistent with those reported by other research groups, which synthesized the same compound by different processes [5,6,8,9,10].
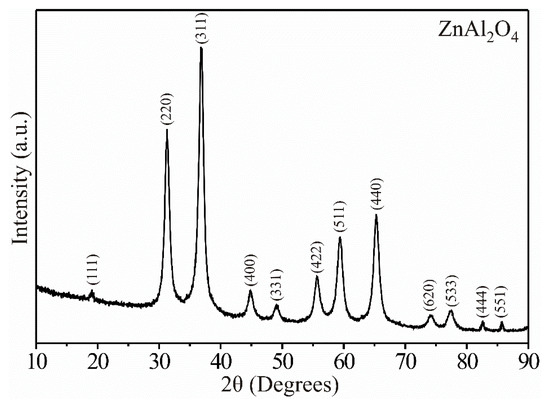
Figure 1.
X-ray diffraction pattern showing the pure phase of the ZnAl2O4 at 250 °C.
Considering Figure 1, the crystal size t was calculated with Scherrer’s equation [22]:
where λ is the wavelength of the radiation (λ = 1518 nm), θ is the Bragg angle, and β is the full width at half maximum (FWHM) of the diffraction peak. In our case, all the diffractograms’ reflections were considered, obtaining an average crystal size of ~14.63 nm.
3.2. Analysis of the Dynamic Response of the ZnAl2O4
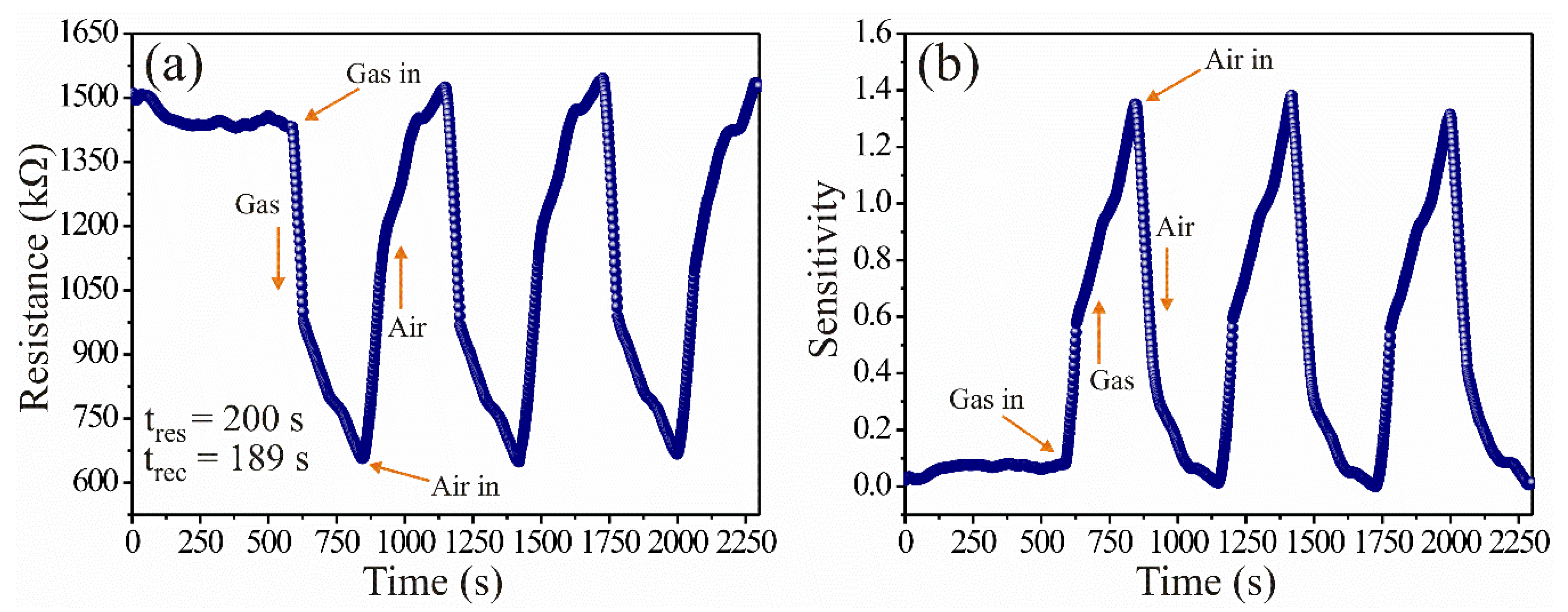
To prove the spinel ZnAl2O4′s ability to detect gas concentrations, dynamic direct current (DC) tests were performed with the prepared pellets (~0.5 mm thick), which were subjected to extra-dry air (500 mL/min) and propane (1000 ppm) flows for measuring the variations in electrical resistance. The experiments were performed in four steps: (1) the pellets were put inside the measurement system placing two electrodes (two-point method) on the ohmic contacts; (2) then, the measuring chamber was heated at a constant temperature of 250 °C, and 500 mL/min of extra-dry air (20% O2 and 80% N2) were injected for five minutes to stabilize the surface of the pellets; (3) after stabilization, 1000 ppm of propane were injected for five minutes, recording variations in the material’s electrical resistance immediately; (4) after that, the propane was removed and extra-dry air was injected into the measuring chamber, which caused the electrical resistance to return to its initial values (when the pellets were stabilized in air, step 1). This process was repeated for several cycles until obtaining the results shown in Figure 2, where electrical resistance (Figure 2a) and sensitivity (Figure 2b) were plotted as a function of time. Using the results of Figure 2a, we calculated the sensitivity of the pellets with the formula , where is the electrical resistance in extra dry air and is the electrical resistance in propane. By considering the reciprocal of the electrical resistance (1/electrical resistance), we calculate [1,23]: , where and are the propane and air conductances, respectively.
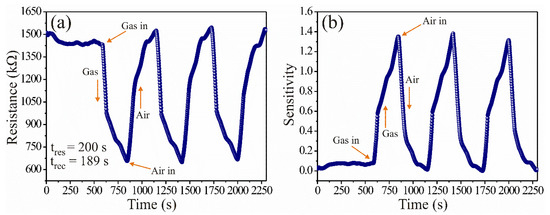
Figure 2.
Dynamic response of the ZnAl2O4 at 250 °C: (a) electrical resistance and (b) sensitivity.
From Figure 2a, when the propane molecules interacted with the surface of the pellets, a decrease in electrical resistance was recorded as the exposure time was extended. That was repeated on several occasions (cycles), which indicated that the pellets maintained a stable process of reversibility and stability in air-propane atmospheres [1]. The excellent response of the ZnAl2O4 was attributed mainly to the reaction that took place on the pellets’ surface between the oxygen and the test gas because of the operating temperature (250 °C) [15,23,24]. The temperature provoked the activation of the charge carriers (electrons) [1], making them move faster on the sensor’s surface [23,25], causing an almost immediate change in electrical resistance and an increase in the sensitivity of the pellets [1]. In addition, the temperature favored the diffusion of the test gas [26], which contributed to an increase in the oxide’s sensitivity (Figure 2b). The variation in electrical resistance (ΔR) was calculated in the range of 1520.04–647.17 kΩ, with an average of 816.85 kΩ (Figure 2a). The sensitivity variations (ΔS) were estimated in the range of 0.11–1.33, with maximum peaks recorded at ~1353 (Figure 2b). The response time (200 s) was estimated considering 90% of the response in propane; the recovery time (189 s) was calculated by considering 10% of the value when the material was exposed to air [1]. The results depicted in Figure 2a,b were consistent with similar semiconductors studied as gas sensors [1,9,26,27,28].
The excellent dynamic response and good sensitivity shown in Figure 2a,b indicate that the oxide is an n-type semiconductor [1]. The oxide’s behavior was due to the reaction between the ionsorbed oxygen [29] on the material’s surface and the propane gas since there was an imbalance between them, leading to the variations in conductivity and, therefore, to the increase in sensitivity. The oxygen species that mostly appeared at 250 °C were the and [30] ionic forms, more reactive than species that predominate below 150 °C [23,30]. That led to the propane’s oxidation on the material’s surface [1], favoring the release of charge carriers that caused the increase in conductivity and sensitivity [1,25,31,32].
The excellent results were attributed to the obtained crystallite size. It has been reported that semiconductors with very fine particles or crystallite sizes smaller than 100 nm showed high electrical variations with short response and recovery times [1,23,25,26,29,31,32]. In agreement with references [1,26], the excellent electrical response and sensitivity of our ZnAl2O4 were due to the rapid and effective propane diffusion over the entire material’s surface, composed of nanometric crystallites (~14.63 nm) that created a favorable porous structure [29]. Therefore, we consider that the ZnAl2O4 is a strong candidate to be applied as a sensor of toxic gases, mainly propane. Comparing the results in Figure 2 with those reported in the references [9,15,23], we found that our sensor ZnAl2O4 had better sensitivity, excellent dynamic response, good reproducibility, shorter response and recovery times at 250 °C. In addition, the curves obtained from the experiments performed at 1000-ppm propane concentrations (see Figure 2), were consistent with those reported in reference [1].
3.3. Theoretical Model
According to the dynamic response of the ZnAl2O4, depicted in Figure 2, the oxide behaved like a first-order system. Then, based on systems theory [33], its dynamic behavior could be described by the following differential equation:
where is the resistance produced by the oxide due to its exposure to air and propane gas, is the shifting time due to the equipment stabilization, is the system’s characteristic time, K is the system’s gain, v(t) is the applied voltage, and t is time. Taking the Laplace Transform of the differential Equation (2),
we calculated:
where is the resistance in the complex plane , is the phase produced by the shift, are the initial conditions of the differential, and is the Laplace transform operator where is a complex number: is the complex rotation operator, is the angular frequency rad/s, and is a real number. From this last expression, we obtained:
and finally, the signal will be
According to Equation (6), the resistive response consisted of free and forced parts. The free resistive response depended on the sensor’s initial conditions, and it did not depend on the input signal:
The forced resistive response depended on the sensor’s input signal but it did not depend on the initial conditions:
That is, the total resistive response of the Pellet is formed by the sum of responses and ,
3.3.1. Transient Response
To obtain the sensor’s transient response, we applied input signals [34,35]. The input signal was a step (whose Laplace transform is ). So, we obtained:
where is the displacement time of the resistive signal when the sensor was exposed to air, is the characteristic time, is the gain, and are the initial conditions. Rearranging Equation (10):
The calculation process was as follows: as a first step, we determined the free response in the time domain through
where is the inverse Laplace transform operator; as a second step, we calculated the forced response with
which, solving for , took the form:
where the Heaviside function is due to system drift; as a third step, both responses were added:
Substituting expressions (12) and (14) in (15), we obtained:
In the limit, when time tends to infinity (and the decay term tends to zero), expression (16) can be approximated to:
which corresponded to the resistance behavior when the oxide was exposed to air. On the other hand, to obtain the resistive dynamic response of the ZnAl2O4 due to its exposure to propane gas, we considered the input signal as an impulse (whose Laplace transform is a unit constant). Then, the resistive dynamic behavior was given by:
where is the delay time when the sensor was exposed to propane gas, is the system gain, is the characteristic time, and is the initial condition. Taking its inverse Laplace transform,
we calculated
where, again, the contribution of the exponential term was negligible. Then, Equation (20) could be approximated to
Equation (21) corresponded to the resistance when the oxide was exposed to propane gas. According to Figure 2, the oxide’s dynamic resistive response was periodic because the designed chemical sensor had repeatability. The first half of the period corresponded to the measured air and the second half of the period corresponded to the measured propane gas. Therefore, its total transient response could be expressed as:
From Figure 2a, is the delay time due to the stabilization of the sensor and instruments, is the exposure period of the sensor and , indicates the sensing cycle. For the first period, the time was between 848.48 and 1410.2 s, , , , , , , , , and :
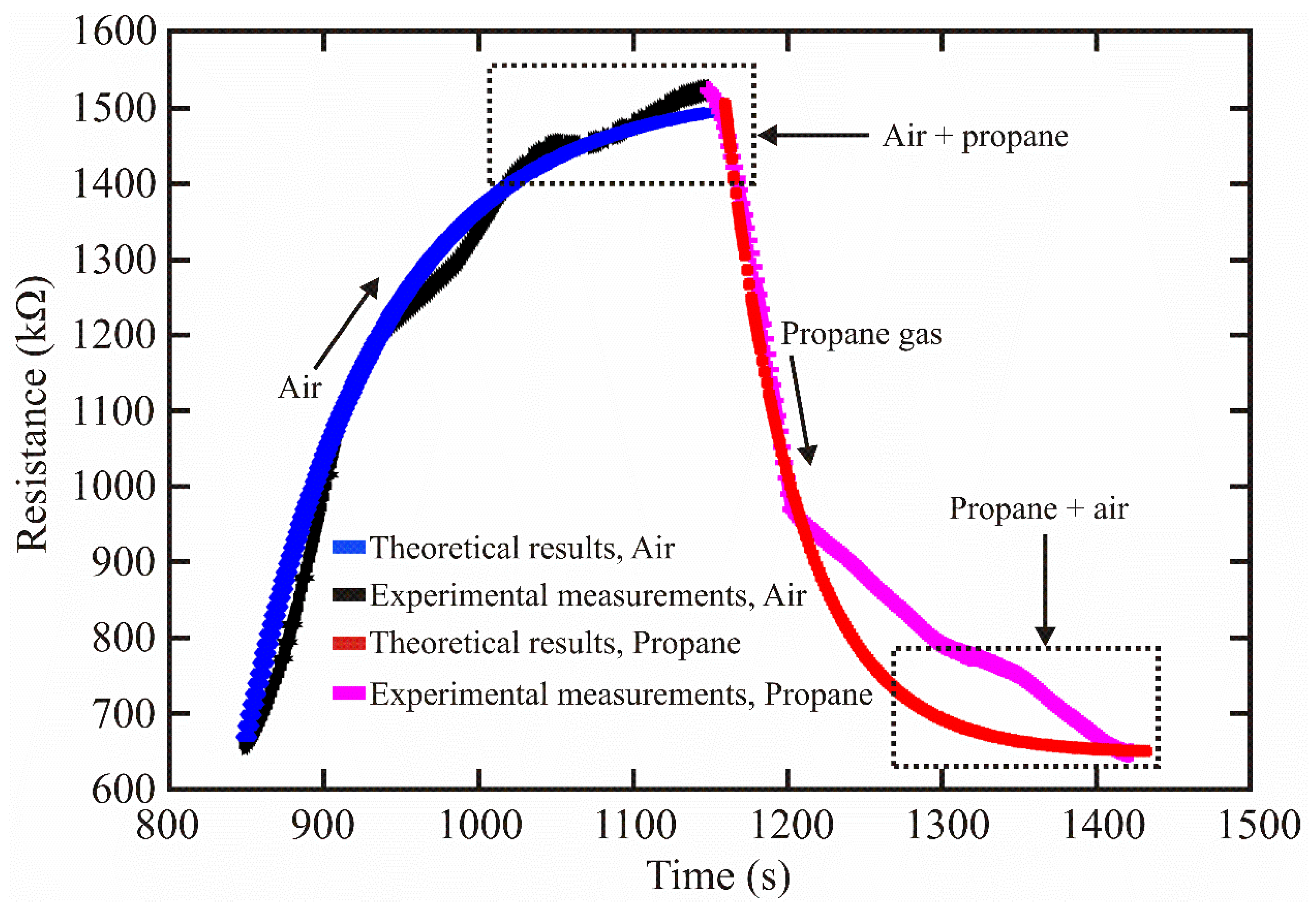
Figure 3 shows a graph of expression (23) where the experimental data were considered.
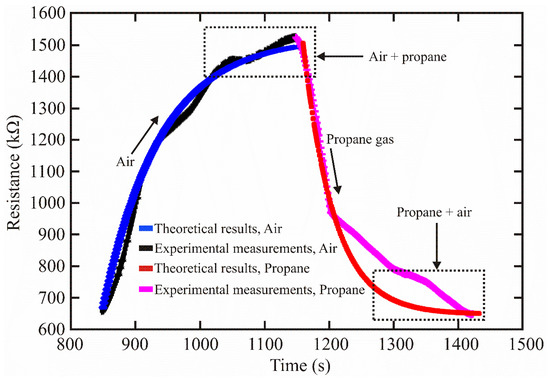
Figure 3.
Theoretical and experimental sensing results of the ZnAl2O4.
From Figure 3, the theoretical result was close to the experimental measurements when the sensor detected air and propane gas. The minor variations can be attributed to experimental errors, system noise, or contamination in the measurement chamber.
3.3.2. Establishment Time
The stability of the ZnAl2O4 sensor could be determined by knowing the position of the poles belonging to the transfer function. The establishment time could be known with the sensor’s “response speed” or its characteristic time.
Since our interest was to detect propane gas atmospheres, we focused on the propane gas transfer function :
Substituting values from the first cycle, we calculated:
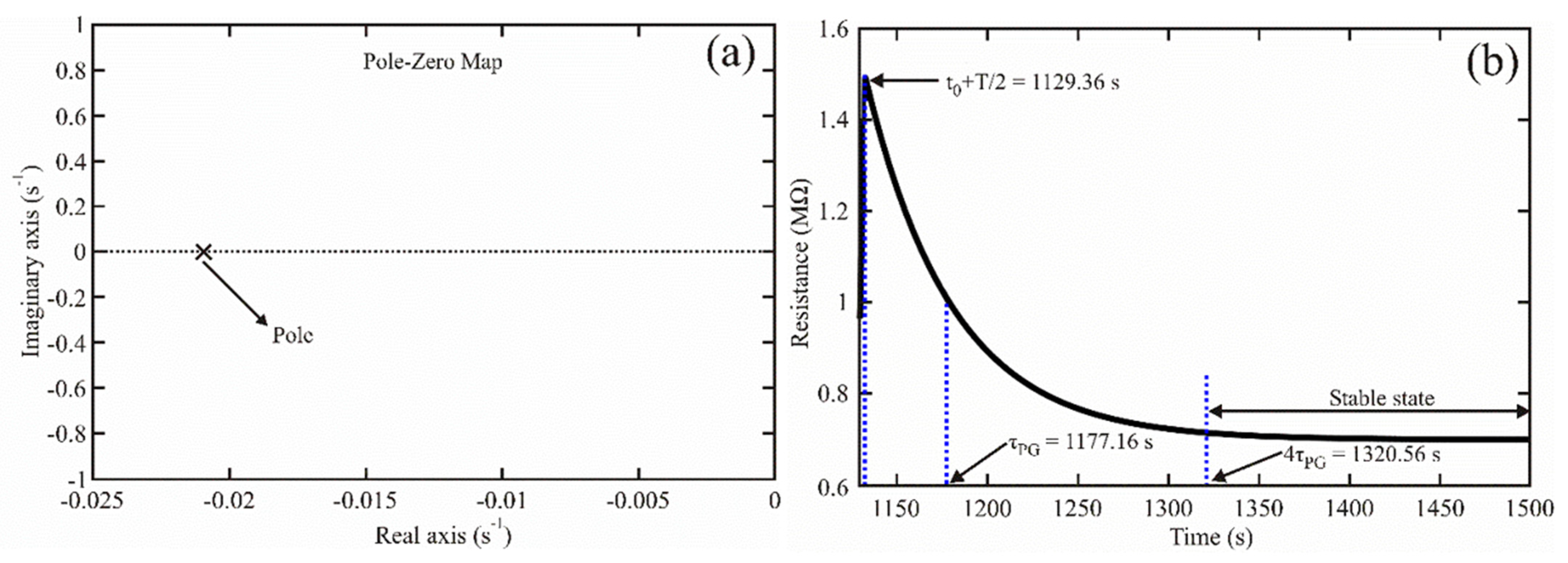
According to the denominator of Equation (25), the sensor’s transfer function had one pole at position , located in the negative half-plane of the complex plane [36,37,38] (see Figure 4a). It indicated that the sensor was stable since taking the inverse Laplace transform of the transfer function, a decaying process was obtained (as Equation (23) shows). If the establishment time is defined as [36], the sensor reached its stability at , with the exit signal at 1.83%. The stability time is shown in Figure 4b.
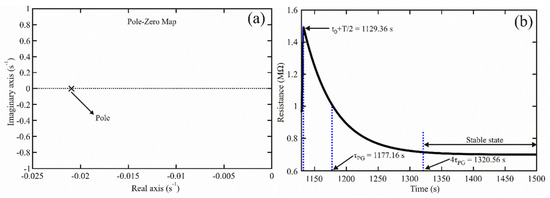
Figure 4.
(a) Pole-zero map obtained for the chemical sensor based on the ZnAl2O4 oxide; (b) Establishment time measured for our chemical sensor.
Based on Figure 4, the sensor was stable, which allowed us to state that the ZnAl2O4 could be safely applied to detect propane gas (for example, to localize explosion-safe zones).
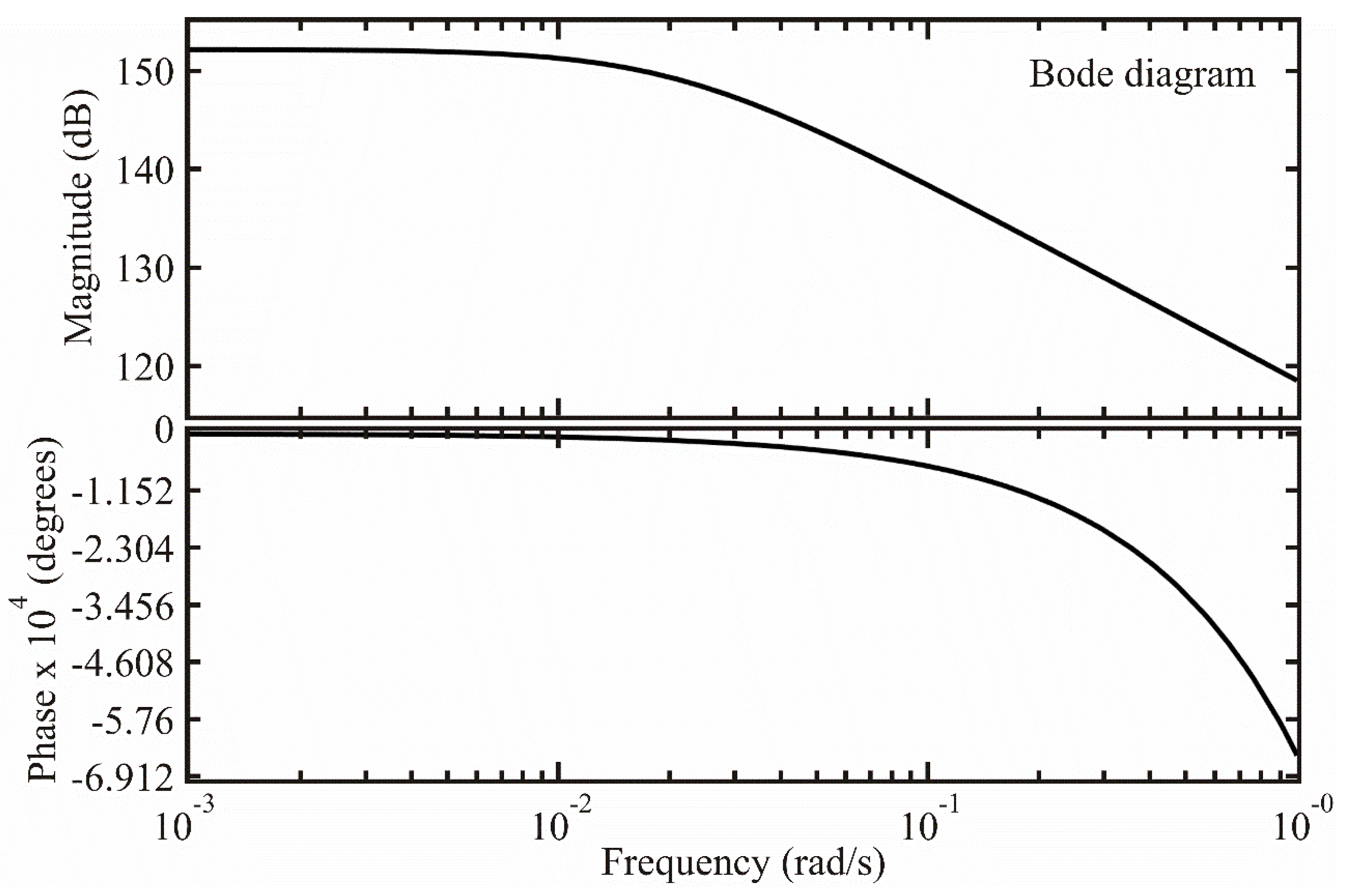
3.3.3. Frequency Response
In this section, using Bode diagrams, a study is made of the behavior at the sensor frequency [36]. With this type of diagram, the behavior of magnitude vs. angular frequency and phase vs. angular frequency and from these graphs it is possible to determine the frequencies where the sensor operates effectively.
First, we performed a change of variable in the transfer function (25) [36,38] and obtained (after some algebraic manipulations):
Removing the complex term from the denominator
We have:
From expression (28), the magnitude was calculated as:
and the phase with
where was the offset angle due to the shift time. The shift time only affected the phase and did not affect the magnitude, as shown in Equations (29) and (30). Figure 5 shows the Bode diagram of the transfer function.
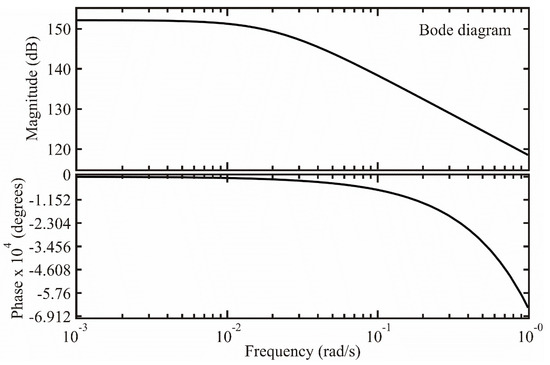
Figure 5.
Frequency behavior of the ZnAl2O4 sensor.
3.3.4. Application to the Detection of Propane Gas
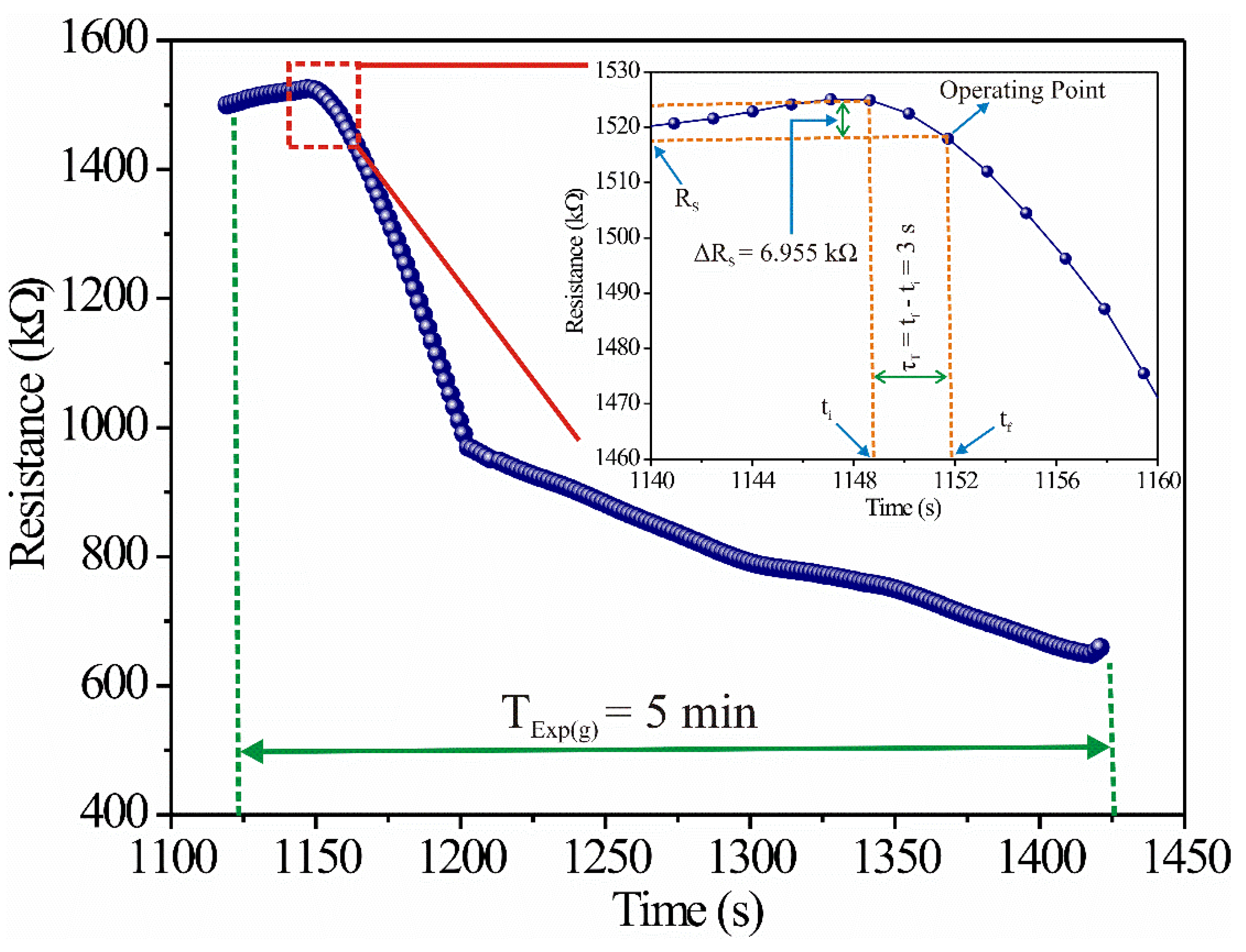
According to Figure 2a and Figure 3, the sensor’s resistance changed as a function of time. Therefore, if our goal was to apply it in a new propane gas detector, the sensor had to operate at a specific point in the dynamic response, which we called the “operating point.” Now, if the sensor’s response time was selected at three seconds, the operating point had the coordinates (≈1151.9 [s] ≈ 1,518,215 [KΩ]) (see Figure 6). Then, we designed the electronic circuit from that operating point.
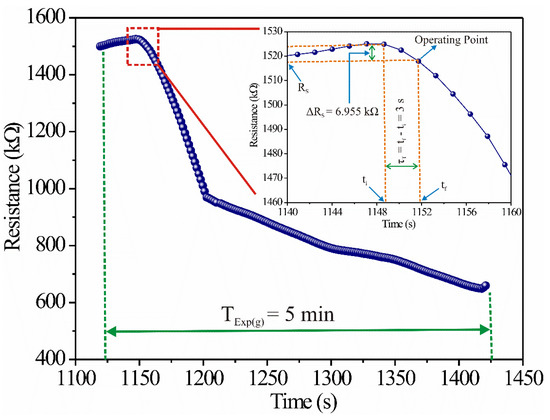
Figure 6.
Graphical representation of the operating point and sensor’s response time (3 s).
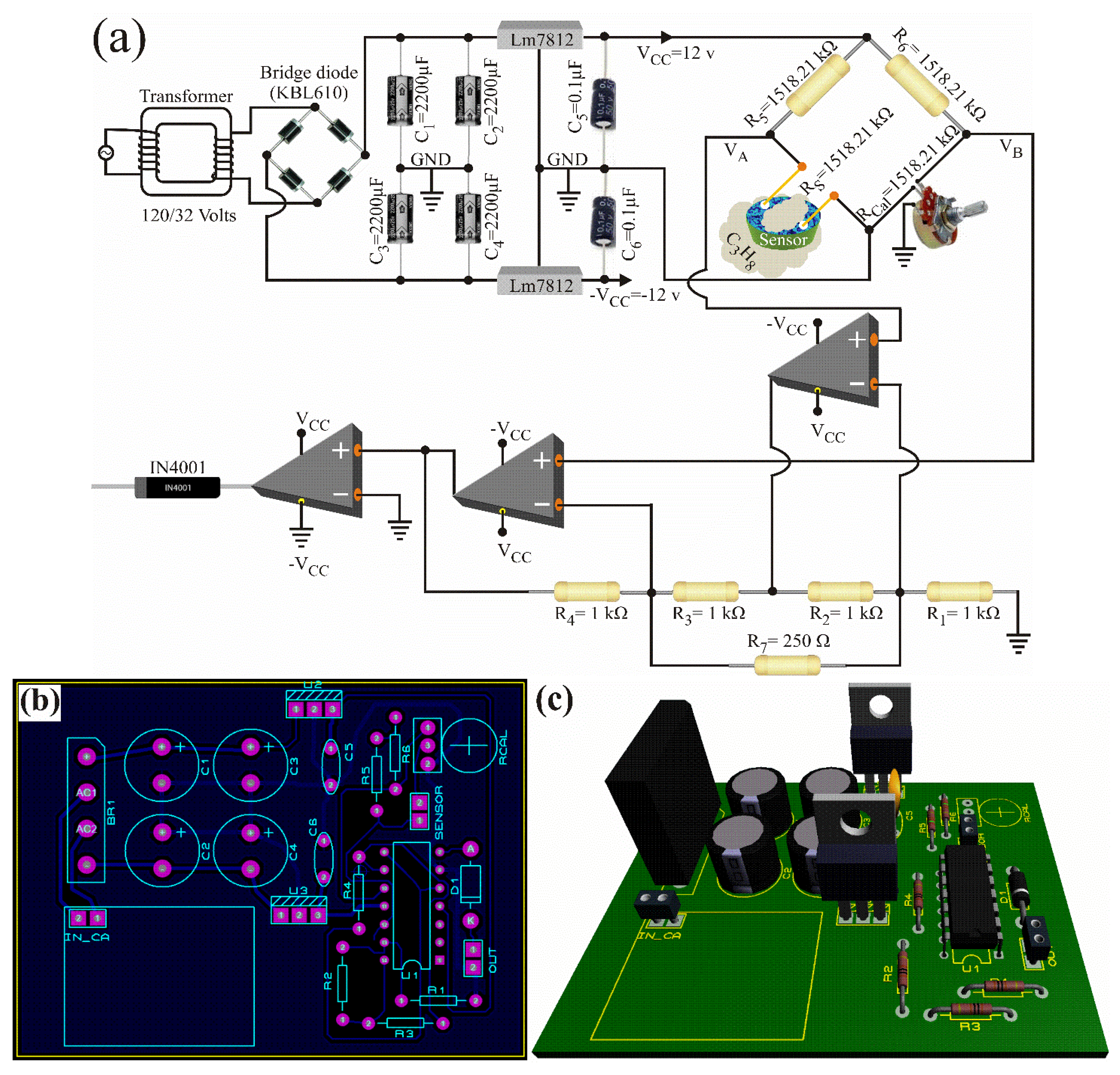
Figure 7a shows the electronic diagram of the propane gas detector. The detector was based on a Wheatstone bridge, an instrumentation amplifier with two operational amplifiers, a comparator circuit, and a voltage source of ±12 Volts. Its operating temperature was 250 °C, it detected concentrations of 1000 ppm, and its response time was three seconds.
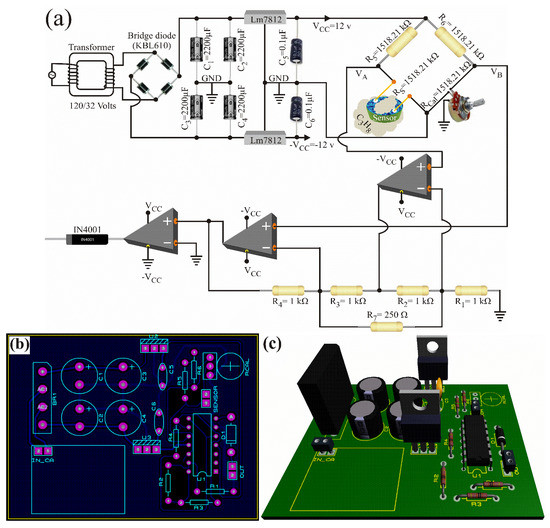
Figure 7.
Propane gas detector: (a) electronic diagram, (b) printed circuit and electronic components’ distribution, and (c) 3D view.
Its working principle consisted of calibration and detection stages [39]. In the calibration stage, the chemical sensor (the ZnAl2O4) was placed in an air atmosphere, its terminals were connected to one arm of the Wheatstone bridge, and the variable resistance was changed until the bridge calibration was achieved, such that the voltage difference (output voltage of the Wheatstone Bridge) was . and were compared with the instrumentation amplifier, and their difference was multiplied by the gain. The output voltage was defined through:
where is the amplifier gain, are precision resistors, and . When was satisfied, the voltage was zero. This voltage may be due to the fact that the comparator circuit had negative saturation and therefore, the diode did not conduct, eliminating any negative signal. Therefore, the alarm signal had a value of zero, . That is, the device would not detect the presence of propane gas in the atmosphere. In the detection stage, when the sensor surface came into contact with propane gas, the sensor had a surface current and suffered a resistance variation. Consequently, the Wheatstone test was unbalanced, and the voltage was less than the voltage . Subsequently, the instrumentation amplifier compared both voltages and amplified 10 times, the difference:
with , , and (see Equation (31) and Figure 7). Since the voltage was greater than zero, the comparator had positive saturation. Therefore, the alarm signal was equal to the positive saturation voltage of the operational amplifier minus the voltage drop of the rectifier diode ( V), . Therefore, the device detected the presence of propane gas in the monitored atmosphere.
Figure 7 shows the propane gas detector’s design obtained with proteus® software. The detector was 10 cm × 10 cm, and its main features were low construction cost, high working temperatures, short response time, high sensitivity, easy repair, and a chemical sensor based on the oxide ZnAl2O4.
In particular, we performed a comparison of our results obtained from the dynamic response and the designed device of the ZnAl2O4 with other semiconducting oxides that have been investigated as potential gas sensors. For example, in references [1,9,27] they report changes in electrical resistance as a function of time, concentration of propane atmospheres and sensitivity humidity on the ZnAl2O4 and ZnO. According to these authors, its optimum temperature to detect propane is from 200 to 300 °C. On the other hand, in reference [26] they acquired the detection of CO2 and O2 over the CoSb2O6 applying an operating temperature of 400 °C. In this work, it was found that the ZnAl2O4 shows excellent dynamic response and sensitivity at 250 °C (1000 ppm of propane), which is a lower temperature than those reported in the previously cited references. In addition, we found that the electrical response of our material was uniform and stable. This is reflected in the number of cycles obtained, which show the thermal stability, and reversibility of the test gas detection process. These parameters and the excellent electrical response, as well as the dynamic sensitivity obtained in the conditions applied during the measurements (250 °C y 1000 ppm of propane) are positive. Theoretical and experimental models were developed for the electronic design of the device based on ZnAl2O4 in dynamic propane atmospheres. Therefore, with the obtained results and the operating conditions of our sensor, these are the optimal parameters to obtain a sensitive, efficient, reliable, easy to implement sensor that presents a prompt response in conditions of propane contamination in the atmosphere.
4. Discussion
Our experimental results for propane detection were consistent with those reported in references [9,26,27,28]. The characteristics of the designed propane gas detector were:
- Operating temperature: 250 °C.
- Working concentration of 1000 ppm.
- 11.3 V alarm voltage due to the presence of propane gas.
- Short response times when detecting the presence of propane gas (e.g., three seconds).
- Applicable to high-temperature processes.
- Low fabrication cost.
- Easy repair.
- High sensitivity.
- Supply voltage of 120 Volts.
- The proposed device finds practical application in boiler safety systems where high temperatures and high concentrations are frequent.
- If the device is placed in an atmosphere with different conditions, the chemical sensor based on the oxide ZnAl2O6 will have a different electrical response, causing the Wheatstone bridge to decalibrate, and therefore, the device will send an erroneous alarm signal. To solve such a situation, the Wheatstone bridge must be calibrated for the new operating conditions.
Our proposal has a wide range of application where there is interest in detecting areas with a high risk of explosion due to fuel leaks, its construction is economical, and it has excellent functional characteristics. Our future work is to develop gas detectors applying programmable electronic devices and develop an error analysis.
5. Conclusions
The semiconductor oxide ZnAl2O4 was synthesized by a wet chemistry method and characterized by X-ray diffraction, identifying its pure phase and crystalline structure. For electrical characterization, pellets were made to record their electrical resistance as a function of time, obtaining excellent results.
A mathematical model based on linear systems was proposed, considering the chemical sensor as a first-order system with shift and initial conditions. We obtained the sensor’s transient response when exposed to air and propane gas by combining the mathematical model and the experimental results. With the models, its stabilization time was calculated, and its frequency response was verified. We selected an operating point, and a novel device was proposed for working as a propane gas detector. Our proposal has many applications, especially detecting areas with a high explosion risk due to fuel leaks. Its construction is economical and has excellent functional features. Our next aim is to develop gas detectors applying programmable electronic devices.
Author Contributions
H.G.-B., and J.T.G.-B., were the supervisors and designed the research; investigation H.G.-B., J.T.G.-B., V.M.R.-B., O.B.-A., M.J.-R., J.A.R.-O., E.H.-P., and A.G.-B.; formal analysis A.G.-B., J.T.G.-B., M.E.S.-M., and H.G.-B.; preparation of materials for gas sensors E.H.-P., A.G.-B., J.A.R.-O., M.J.-R., and M.E.S.-M., writing–review and editing J.T.G.-B., V.M.R.-B., A.G.-B., O.B.-A., and H.G.-B. All the authors participated in the conception and design of the work, meeting the authorship conditions, and have approved the final version. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding authors upon request.
Acknowledgments
The authors thank Mexico’s National Council of Science and Technology (CONACyT) and the University of Guadalajara for their support. Likewise, we thank Juan Reyes Gómez for their technical assistance. This investigation was carried out following the lines of research “Nanostructured Semiconductor Oxides” of the academic group UDG-CA-895 “Nanostructured Semiconductors” of CUCEI, University of Guadalajara. J. T. Guillen Bonilla expresses his gratitude to PRODEP 2020, project No. F-PROMEP-18/Rev-03 SEP-23-003-A (186769).
Conflicts of Interest
The authors declare that there are no conflicts of interest regarding the publication of this article.
References
- Huízar-Padilla, E.; Guillén-Bonilla, H.; Guillén-Bonilla, A.; Rodríguez-Betancourtt, V.-M.; Sánchez-Martínez, A.; Guillén-Bonilla, J.T.; Gildo-Ortiz, L.; Reyes-Gómez, J. Synthesis of ZnAl2O4 and evaluation of the response in propane atmospheres of pellets and thick films manufactured with powders of the oxide. Sensors 2021, 21, 2362. [Google Scholar] [CrossRef]
- Tielens, F.; Calatayud, M.; Franco, R.; Recio, J.M.; Pérez-Ramírez, J.; Minot, C. Periodic DFT study of the structural and electronic properties of bulk CoAl2O4 spinel. J. Phys. Chem. B 2006, 110, 988–995. [Google Scholar] [CrossRef]
- Confalonieri, G.; Rotiroti, N.; Bernasconi, A.; Dapiaggi, M. Structural study of nano-sized gahnite (ZnAl2O4): From the average to the local scale. Nanomaterials 2020, 10, 824. [Google Scholar] [CrossRef] [PubMed]
- Okal, J.; Zawadzki, M. Combustion of propane over novel zinc aluminate-supported ruthenium catalysts. Appl. Catal. B Environ. 2011, 105, 182–190. [Google Scholar] [CrossRef]
- Kapse, S.D.; Raghuwanshi, F.C.; Kapse, V.D.; Patil, D.R. Characteristics of high sensitivity ethanol gas sensors based on nanostructured spinel Zn1−xCoxAl2O4. Curr. Appl. Phys. 2012, 12, 307–312. [Google Scholar] [CrossRef]
- Dhak, D.; Pramanik, P. Particle size comparison of soft-chemically prepared transition metal (Co, Ni, Cu, Zn) aluminate spinels. J. Am. Ceram. Soc. 2006, 89, 1014–1021. [Google Scholar] [CrossRef]
- Quirino, M.R.; Oliveira, M.J.C.; Keyson, D.; Lucena, G.L.; Oliveira, J.B.L.; Gama, L. Synthesis of zinc aluminate with high surface area by microwave hydrothermal method applied in the transesterification of soybean oil (biodiesel). Mater. Res. Bull. 2016, 74, 124–128. [Google Scholar] [CrossRef]
- Ianoş, R.; Băbuţă, R.; Păcurariu, C.; Lazău, R.; Istratie, R.; Butaciu, C. Combustion synthesis of ZnAl2O4 powders with tuned surface area. Ceram. Int. 2017, 43, 8975–8981. [Google Scholar] [CrossRef]
- Cheng, B.; Ouyang, Z.; Tian, B.; Xiao, Y.; Lei, S. Porous ZnAl2O4 spinel nanorods: High sensitivity humidity sensors. Ceram. Int. 2013, 39, 7379–7386. [Google Scholar] [CrossRef]
- Fernández-Osorio, A.; Rivera, C.E.; Vazquez-Olmos, A.; Chávez, J. Luminescent ceramic nano-pigments based on terbium-doped zinc aluminate: Synthesis, properties and performance. Dyes Pigm. 2015, 119, 22–29. [Google Scholar] [CrossRef]
- El Habra, N.; Crociani, L.; Sada, C.; Zanella, P.; Casarin, M.; Rossetto, G.; Carta, G.; Paolucci, G. MOCVD of CoAl2O4 Thin films from {Co[Al(OiC3H7)4]2} as precursor. Chem. Mater. 2007, 19, 3381–3386. [Google Scholar] [CrossRef]
- Marconato Stringhini, F.; Foletto, E.L.; Sallet, D.; Assumpcao Bertuol, D.; Chiavone-Filho, O.; Oller do Nascimento, C.A. Synthesis of porous zinc aluminate spinel (ZnAl2O4) by metal-chitosan complexation method. J. Alloys Compd. 2014, 588, 305–309. [Google Scholar] [CrossRef]
- Ragupathi, C.; Judith Vijaya, J.; Narayanan, S.; John Kennedy, L.; Ramakrishna, S. Catalytic properties of nanosized zinc aluminates prepared by green process using opuntia dilenii haw plant extract. Chin. J. Catal. 2013, 34, 1951–1958. [Google Scholar] [CrossRef]
- Kumar, M.; Mohapatra, M. A case study of energy transfer mechanism from uranium to europium in ZnAl2O4 spinel host by photoluminescence spectroscopy. Spectrochim. Acta A 2016, 159, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Guillen Bonilla, J.T.; Guillen Bonilla, H.; Rodríguez-Betancourtt, V.M.; Guillen Bonilla, A.; Casillas Zamora, A.; Blanco Alonso, O.; Ramírez Ortega, J.A. A gas sensor for application as a propane leak detector. Sensors 2021, 2021, 8871166. [Google Scholar] [CrossRef]
- Basyooni, M.A.; Zaki, S.E.; Ertugrul, S.; Yilmaz, M.; Eker, Y.R. Fast response of CO2 room base donra gas sensor ase don mixed-valence phases in molybdenum and tungsten oxide nanostructured thin films. Ceram. Int. 2020, 46, 9839–9853. [Google Scholar] [CrossRef]
- Chang, J.; Meng, H.; Li, C.; Gao, J.; Chen, S.; Hu, Q.; Li, H.; Feng, L. Aweareble toxic gas-monitoring device based on triboelectric nanogenerator for self-powered aniline early warning. Adv. Mater. Technol. 2020, 5, 1901087. [Google Scholar] [CrossRef]
- Zuo, J.; Tavakoli, S.; Mathavakrishnan, D.; Ma, T.; Lim, M.; Rotondo, B.; Pauzauskie, P.; Pavanatto, F.; Mackenzie, D. Additive manufacturing of a flexible Carbon monoxide sensor based on a SnO2-graphene nanoink. Chemosensors 2020, 8, 36. [Google Scholar] [CrossRef]
- Steinhauer, S. Gas sensors based on copper oxide nanomaterials: A review. Chemosensors 2021, 9, 51. [Google Scholar] [CrossRef]
- Shellaiah, M.; Wen Sun, K. Review on sensing application of perovskite nanomaterials. Chemosensors 2020, 8, 55. [Google Scholar] [CrossRef]
- Delgado, E.; Michel, C.R. CO2 and O2 sensing behavior of nanostructured barium-doped SmCoO3. Mater. Lett. 2006, 60, 1613–1616. [Google Scholar] [CrossRef]
- Imaduddin, I.S.; Majid, S.R.; Aziz, S.B.; Brevik, I.; Yusuf, S.N.F.; Brza, M.A.; Saeed, S.R.; Kadir, M.F.Z.A. Fabrication of Co3O4 from Cobalt/2,6-Napthalenedicarboxylic acid metal-organic framework as electrode for supercapacitor application. Materials 2021, 14, 573. [Google Scholar] [CrossRef] [PubMed]
- Casillas-Zamora, A.; Guillén-Bonilla, J.T.; Guillén-Bonilla, A.; Rodríguez Betancourtt, M.; Casallas-Moreno, Y.L.; Gildo Ortiz, L.; Olvera-Amador, M.L.; Tomás, S.A.; Guillén-Bonilla, H. Synthesis of MnSb2O6 powders through a simple low-temperature method and their test as a gas sensor. J. Mater. Sci. Mater. Electron. 2019, 31, 7359–7372. [Google Scholar] [CrossRef]
- Chang, S.C. Oxygen chemisorption on tin oxide: Correlation between electrical conductivity and EPR measurements. J. Vac. Sci. Technol. 1979, 17, 366–369. [Google Scholar] [CrossRef]
- Alrammouz, R.; Podlecki, J.; Abboud, P.; Sorli, B.; Habchi, R. A review on flexible gas sensors: From materials to devices. Sens. Actuators A 2018, 284, 209–231. [Google Scholar] [CrossRef]
- Michel, C.R.; Guillén-Bonilla, H.; Martínez-Preciado, A.H.; Morán-Lázaro, J.P. Synthesis and gas sensing properties of nanostructured CoSb2O6 microspheres. Sens. Actuators B Chem. 2009, 143, 278–285. [Google Scholar] [CrossRef]
- Gómez-Pozos, H.; Karthik, T.V.K.; de la Olvera, M.L.; García Barrientos, A.; Pérez Cortés, O.; Vega-Pérez, J.; Maldonado, A.; Pérez-Hernández, R.; Rodríguez-Lugo, V. ZnO thin films as propane sensors: Band structure models to explicate the dependence between the structural and morphological properties on gas sensitivity. J. Phys. Chem. Solids. 2017, 106, 16–28. [Google Scholar] [CrossRef]
- Kita, J.; Engelbrecht, A.; Schubert, F.; Gro, A.; Rettig, F.; Moos, R. Some practical points to consider with respect to thermal conductivity and electrical resistivity of ceramic substrates for high-temperature gas sensors. Sens. Actuators B Chem. 2015, 213, 541–546. [Google Scholar] [CrossRef]
- Dey, A. Semiconductor metal oxide gas sensors: A review. Mater. Sci. Eng. B 2018, 229, 206–217. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, T. An overview: Facet-dependent metal oxide semiconductor gas sensors. Sens. Actuators B Chem. 2018, 277, 604–633. [Google Scholar] [CrossRef]
- Wang, C.; Yin, L.; Zhang, L.; Xiang, D.; Gao, R. Metal oxide gas sensors: Sensitivity and influencing factors. Sensors 2010, 10, 2088. [Google Scholar] [CrossRef] [Green Version]
- Fine, G.F.; Cavanagh, L.M.; Afonja, A.; Binions, R. Metal oxide semi-conductor gas sensors in environmental monitoring. Sensors 2010, 10, 5469. [Google Scholar] [CrossRef] [Green Version]
- Campi, M.C.; Garatti, S.; Prandini, M. The scenario approach for systems and control design. Annu. Rev. Control 2009, 33, 149–157. [Google Scholar] [CrossRef]
- Khalil, H.K. Nonlinear Systems; Prentice-Hall: Upper Saddle River, NJ, USA, 2002. [Google Scholar]
- Guillen Bonilla, A.; Rodríguez Betancourtt, V.M.; Guillen Bonilla, H.; Gildo Ortíz, L.; Blanco Alonso, O.; Franco Rodríguez, N.E.; Reyes Gómez, J.; Guillen Bonilla, J.T. A new detection system based on the trirutile-type CoSb2O6 oxide. J. Mater. Sci. Mater. Electron. 2018, 29, 15741–15753. [Google Scholar] [CrossRef]
- Smith, C.A.; Corripio, A. Principles and Practice of Automatic Process Control, 2nd ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1997. [Google Scholar]
- Bašić, M.; Vukadinović, D.; Petrović, G. Dynamic and pole-zero analysis of self-excited induction generator using a novel model with iron losses. Electr. Power Energy Syst. 2012, 42, 105. [Google Scholar] [CrossRef]
- Guillen Bonilla, J.T.; Guillen Bonilla, H.; Rodríguez Betancourtt, V.M.; Sánchez Morales, M.E.; Casillas Zamora, A.; Guillen Bonilla, A. Low-finesse Fabry-Pérot interferometers applied in the study of the relation between the optical path difference and the poles location. Sensors 2020, 20, 453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guillen Bonilla, J.T.; Guillen Bonilla, H.; Rodríguez Betancourtt, V.M.; Casillas Zamora, A.; Ramírez Ortega, J.A.; Gildo Ortiz, L.; Sánchez Morales, M.E.; Blanco Alonso, O.; Guillen Bonilla, A. Carbon Monoxide (CO) detection device based on the nickel antimonate oxide and a DC electronic circuit. Appl. Sci. 2019, 9, 3799. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).