Abstract
For cells and tissues, cell–cell and cell–extracellular matrix adhesion is important for proliferation, differentiation, and response to mechanical stimuli. This adhesion is provided by various cell adhesion molecules (CAMs). However, in breast cancer, depending upon the type and stage, this adherence is dysregulated where the expression of these cell adhesion molecules is either overregulated or unregulated, triggering essential oncogenic pathways. Thus, to control the invasiveness of tumor cells, and reduce metastasis, regulating the homophilic and heterophilic interaction of these molecules and controlling the essential cell pathways is important. In this study, we targeted critical CAMs- integrins to regulate their aberrated behavior via siRNAs delivery. However, as due to charge repulsion and propensity to be degraded by nucleases prior to reaching the target site, naked siRNAs are unable to cross plasma membrane, use of a suitable carrier vehicle is essential. Thus, we employed carbonate apatite (CA), to deliver the selected siRNAs targeting integrin αv, α6, β1, β3, β4, β5, and β6 subunits to various breast cancer cell lines and 4T1-breast cancer induced murine model. Delivery of individual integrin siRNAs complexed with CA nanoparticles (NPs) reduced cell viability and caused decrease in tumor burden. To check the gene knockdown effects on phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) and extracellular signal–regulated kinases/mitogen-activated protein kinase (ERK/MAPK) pathways, Western blot analysis was performed, revealing downregulation of the signaling molecules. Thus, CA-facilitated gene therapy targeting various integrins could poise potential therapeutic modality against breast cancer.
1. Introduction
In 2020, about 2.3 million women were diagnosed with breast cancer, of which 685,000 were observed globally [1]. The National Cancer Institute recorded about 3,676,262 breast cancer women in US in 2018, with an estimate of 281,550 new cases in 2021, of which around 43,600 will be estimated deaths [2].
Breast cancer is a group of diseases possessing phenotypic and genotypic heterogeneity. Each type of breast cancer possesses distinct characteristics which differentiate it from the other type/subtype and determine the type of effective treatment regime. Overexpression of human epidermal growth factor receptor 2 (HER2) is an important marker of an aggressive form of tumors [3], while as a type of basal breast cancer, triple negative breast cancer (TNBC) where expression of all the three hormone receptors (estrogen receptor-ER, progesterone receptor-PR, and HER2) is negative represents one of the most aggressive forms which possess distant metastasis patterns and lack targeted therapies [4,5]. Histologically, breast canceris categorized into ductal or lobular, with 80% of invasive carcinomas being ductal in origin. Accounting to one fourth of breast cancers, ductal carcinoma in situ (DCIS) consists of heterogeneous lesions with varied clinical features, morphology, and biological functioning [3].
The underlying mechanism of invasiveness and progression of cancer is yet to be fully understood. However, altercations in cell–cell adhesion, increase in motility, production of metalloproteases (MMPs), invasion of basement membrane, degradation of extracellular membrane, intravastation into blood and lymphatic vessels, extravasation, proliferation, and colonization at distant sites are some of the essential and critical steps involved [6,7,8]. During the primary and secondary cancer development several cell molecules and receptors which are vital for normal cellular development, proliferation, growth, and survival can malfunction. This results in altered gene expression, uncontrolled proliferation, and cellular growth. One such essential category of the molecules is CAMs. CAMs such as integrins mediate interactions between tumor cells and surrounding environment to contribute to cancer progression and metastasis. These molecules associate with receptors and other cell adhesion molecules, triggering cascades of pathways such as ERK/MAPK, PI3K-AKT, wingless/integrated (WNT), c-Jun N-terminal kinase (JNK), and transforming growth factor beta (TGF-β) to initiate, progress, and metastasize the tumors [9,10,11]. Based on the stage of cancer, the upregulation and downregulation of CAMs at transcriptional and translational level cause changes in protein expression, activation of pathways, and modifications in cell adhesion patterns. Altered expression of adhesion complex proteins which help in maintaining the breast epithelia contributes towards tissue disorganization, leading to development of breast cancer [12]. In breast cancer, depending upon type of breast carcinoma and its extent metastasis, these adhesion molecules have been found either under expressed or overexpressed [12,13,14,15].
Integrins, family of glycoproteins, serve as main extracellular matrix (ECM) receptors, linking ECM and cytoskeleton via ligands. Structurally, integrin receptors consisting of 18 α and 8 β-subunits are linked non-covalently, forming at least 24 heterodimer pairs. While α subunit is larger than β subunit, a distinct similarity has been found within both. Localized at plasma membrane, each integrin binds to various proteins on the intracellular side, and interacts with specific ligands such as fibronectin, fibrinogen, vitronectin, laminin, and collagen on extracellular domain, accounting for bidirectional signaling [16,17,18,19]. Integrin serves as a main ECM receptor of mammary epithelium, where signaling pathways help modulate the cellular responses and control the breast biology. This includes mammary gland functioning, ductal morphogenesis, mammary alveoli, and lumen formation. While, in normal breasts, the histochemical assays show presence of integrins in myoepitheial layer of basement membrane, their concentration and expression pattern are different in breast cancer [12,20,21,22]. Abnormal integrin expression contributes to the migratory properties of tumor cells. This permits them to pass through blood vessels and collagen matrix, invade surrounding cells and tissues, and eventually colonize tumor cells at secondary tissues [20,21]. Various in vitro and in vivo studies show compromised expression of integrin receptors and their associated signaling pathways. Integrins such as αvβ5, αvβ6, α2β1, α6β1, and α6β4 play critical roles in breast cancer growth, invasion, and metastasis [22]. Integrin αv (ITGαv) which forms heterodimer with six β integrin sub is a receptor for various proteins [23]. Each heterodimer of αv-β plays an important part in the regulation of breast cancer and metastasis. For example, integrin αvβ3 causes bone metastasis in breast cancer through enhanced tumor adhesion. While the expression of this integrin is found in both primary tumors and metastatic cells, it is more active in metastatic cells [24,25]. Apart from ITGαv, integrin α6 (ITGα6) also has an important role in invasion, adhesion, migration, and progression of breast cancer cells. Overexpressed in breast cancer cells and tissues, it is associated with poor prognosis and reduced survival rates. Furthermore, studies show that in various breast cancer cells its overexpression alters the processes such as apoptosis, cell cycle arrest, and DNA damage and double-strand break repair mechanism [26]. This integrin subunit can form heterodimers with β1 or β4 subunits, forming α6β1 and α6β4 integrins, with each integrin unit forming receptor for laminins. By participating via different mechanisms, both α6β1 and α6β4 contribute to the progression and survival of breast cancer [27].
While there are 8 subunits of β integrin which play important roles in cellular processes, integrins such as β1, β3, β4, β5, β6, and β8 have vital role in breast cancer. Each type of β subunits plays a distinct role in breast cancer where they are essentially involved in pro-tumorigenic processes such as epithelial mesenchymal transition (EMT), tumor development, progression, survival, angiogenesis, neo-angiogenesis, inflammation, and metastasis [16,28,29,30,31,32]. For example, the role of β1 integrin based on the type of breast cancer varies, whereby its decreased and increased expression plays essential roles in tumorigenesis. Regular expression of this integrin helps in development of epithelial tissues via cellular proliferation and differentiation, but when overexpressed it can participate in angiogenesis, progression, and metastasis of breast cancer in invasive ductal carcinoma (IDC) [33]. Like α subunit, β subunit also associates with various receptor molecules, modulating different signaling pathways in breast cancer. For example, ITGβ5, which is present in primary and metastatic breast cancer cells, activates TGF-β and ERK signaling pathways via focal adhesion molecules such as focal adhesion kinase (FAK) and paxilin, and epidermal growth factor receptor (EGFR) signaling via proto-oncogene tyrosine-protein kinase (Src), causing tumor cell migration, invasion proliferation, and metastasis [28].
The overexpression of these integrins provides resistance to chemotherapy and radiations, resulting in a decrease in the survival rate of breast cancer patients. Hence, modulating their expressions can help in limiting the growth and invasion of tumor cells, thereby increasing the survival rates. This could be achieved by silencing the gene expression of overregulated genes using small interfering RNAs (siRNAs). siRNAs are short double stranded RNA sequences that impede the expression of over expressed genes by degrading the mRNA sequences in cell cytoplasm. In addition to curbing the upregulated gene expression, siRNA gene silencing can also help in chemosensitizing the tumor cells for various drugs, resulting in increased chemotherapy efficacy [34,35,36]. However, targeted delivery of siRNAs to the cancer cells is a major challenge because of inability to pass the plasma membrane, and sensitiveness to nuclease-based breakdown [34,36]. Therefore, it is necessary to use a delivery vehicle to carry nucleic acids and/or chemotherapy drugs to the target site. Currently nanoparticles serve as one of the efficient carrier vehicles. While several nanoparticles such as lipid based carriers, polymers, carbon nanotubes, and inorganic nanoparticles are being used at various pre-clinical and clinical stages, pH receptive inorganic CA is also gaining science based evidence for a carrier vehicle [37,38,39]. With the help of cation (Ca2+)-rich domains, CA is capable of electrostatically binding a nucleic acid sequence possessing an anionic phosphate backbone. The presence of carbonate as a core constituent of CA notably helps in preventing particle aggregation and thus maintaining the size within few hundred nanometers—a size range potentially beneficial not only in increasing loading of nucleic acids, but also in accelerating passive tumor accumulation by enhanced permeability and retention (EPR) effect. While CA is very stable at physiological pH, it is highly sensitive to dissolution at endosomal acidic pH—a fascinating property that facilitates fast release of payload (DNA or siRNA) after the particles are endocytosed by the target cells. The particle dissolution might also assist in endosomal destabilization through development of osmotic pressure across the endosomal membrane due to accumulation of Ca2+, phosphate, and carbonate ions—the core constituents of CA, thus enabling release of bound DNA or siRNA from both the carrier and cellular endosomes to cytosol. This in turn results in targeted therapy whereby expression of a desirable protein or specific cleavage of a mRNA transcript is achieved, without causing any toxicity to healthy cells [40,41,42,43]. Therefore, by targeting altered integrin genes via CA-mediated intracellular delivery of specific siRNAs, the reduction in proliferation and survival of breast cancer cells could be achievable. Apart from the regular treatments, focus on the cell adhesion biomarkers could enhance the survival rate in breast cancer patients with poor prognosis. In this study we delivered individual siRNAs targeting integrin αv, α6, β1, β3, β4, β5, and β6 via CA nanoparticles in breast cancer cell lines as well as murine metastatic breast cancer model to evaluate the effect on tumor cell proliferation and growth.
2. Materials and Methods
2.1. Reagents
Cell lines: Human breast cancer cells—Adenocarcinoma MCF-7; Triple negative breast cancer cells—MDA-MB-231; and murine metastatic breast cancer cells—T1.
Chemicals: Dulbecco’s modified eagle medium (DMEM) powder, fetal bovine serum (FBS), penicillin streptomycin, trypLE Express with no phenol red 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), and ethylenediamine tetraacetate (EDTA) purchased from Gibco BRL (Carlsbad, CA, USA). DMEM, calcium chloride dihydrate (CaCl2·2H2O), sodium bicarbonate (NaHCO3), potassium phosphate monobasic (KH₂PO₄), sodium phosphate dibasic heptahydrate (H15Na2O11P), bovine serum albumin (BSA), skim milk powder, Tris (hydroxmethyl)amino-methane, phosphate inhibitor cocktail 2, EDTA-free protease inhibitor tablets, dimethyl sulphoxide (DMSO), thiazolyl blue tetrazolium bromide (MTT), methanol, and trypan blue solution (0.4%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Sodium chloride (NaCl) and potassium chloride (KCl) were purchased from Fisher Scientific (Loughborough, Leicestershire, UK). Sodium dodecyl sulfate (SDS) and Restore PLUS Western blot stripping buffer were purchased from Thermo Fisher Scientific (Rockford, IL, USA). Triton X-100, glycine, blotting-grade blocker, Clarity Western ECL substrate, tween-20, bromophenol blue, quickstart Bradford 1X dye reagent, quickstart bovine serum albumin (BSA) standard set, dithiothreitol (DTT), and mini-protean TGX gels were purchased from Bio Rad (Hercules, CA, USA). Cell signaling primary and secondary antibodies and all the validated siRNAs were purchased from Qiagen (Valencia, CA, USA).
2.2. Cell Culture
MCF-7, MDA-MB-231, and 4T1 cell lines were grown in 75 cm2 tissue flasks (Nunc, Orlando, FL, USA). DMEM with 10% FBS, 50 μg/mL penicillin, 50 μg/mL streptomycin and 1% HEPES was used as complete growth media and the flasks were cultured at 37 °C in 5% CO2 incubator.
2.3. siRNA Design and Sequence
First, 1 nmol lyophilized siRNA was reconstituted with RNase-free water to obtain a stock solution of 10 μM, which was aliquoted and stored at −20 °C. All the siRNAs used were experimentally verified by Qiagen via real-time RT-PCR, with >70% knockdown efficiency against Hela cell line. siRNAs with high knockdown efficiency were chosen for the study. Table 1 summarizes the siRNAs used with their gene sequence and knockdown efficiency against Hela cell line.

Table 1.
siRNAs and their sequences used in this study.
2.4. Generation of CA Nanoparticle Loaded siRNAs Complex
For in vitro study, NPs were prepared as described earlier [43]. Briefly, 1 nM concentration of various siRNAs (listed in Table 1) were individually added in DMEM powder media with 44 mM bircabonate at pH 7.4, with 4 μL of CaCl2, and subjected to 30 min incubation at 37 °C to allow complex formation. Post incubation, particle generation was stopped using 10% FBS and the complex was used for in vitro treatments.
2.5. Cell Proliferation Assay by 3-(4, -Dimethylthiazaol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT)
MTT assay was used to assess CA nanoparticle cytotoxicity on MCF-7, MDA-MB-231, and 4T1 cell lines. In brief, cells from the exponential growth phase were seeded in 24 well plates at 50,000 cell/well density (approximately) in DMEM media containing 10% FBS at 37 °C, 5% CO2 for 24 h. After 24 h, the cells were exposed to various treatments of single integrin siRNAs complexed with CA nanoparticles for 48 h. After 48 h 50 μL of MTT solution (5 mg/mL) was added in each well and plates were further incubated for 4 h at 37 °C 5% CO2. Media was removed after 4 h and DMSO (300 μL) was pipetted in each well to dissolve the purple formazan crystals. The plates were incubated with DMSO for 5 min to ensure complete dissolution of crystals, followed by absorbance reading using microplate reader (Bio-Rad) at 595 nm optical density (OD) and 630 nm reference wavelength. MTT data were represented as mean ± SD. Cell viability in the treated wells was shown as percentage and MTT based absorbance values were used for calculation using following formula: % of cell viability = (OD treated − OD reference/OD untreated − OD reference) × 10F.
The following formula was used for cell death and actual cytotoxicity:
Cell death (%) = 100 − Cell viability (%)
Actual cytotoxicity (of siRNA) (%) = CA (%) + siRNA cytotoxicity − CA cytotoxicity (%)
2.6. In Vivo Tumor Regression Study Post Intravenous Delivery of CA Loaded siRNA Complex
All the in vivo work was done upon the approval by Monash University Animal Ethics Committee (MARP/2016/126). Female Balb/c mice (6–8 weeks old) of 15–20 g of body weight (obtained from School of Medicine and Health Science Animal Facility, Monash University) were maintained in 12:12 light:dark condition and provided with ab libitum and water. During the treatment period, the body weight and size of the tumor were measured at regular intervals. The maximum duration for which the experiments were run was 4 weeks. During this period there was no significant tumor burden for the mice to develop pain/distress since the tumor volume would not exceed 1 cm3 within the 4 weeks. Furthermore, there was no drop in the body weight of the mice during the experimenting period. Additionally, there was no labored or irregular breathing observed. To reduce the pain during injections, as a local anaesthetic, Emla cream was applied topically on the injection site before proceeding for intravenous injections. At the end, all the animals were humanely euthanized by cervical dislocation.
Subcutaneous injection of around 1 × 105 4T1 cells dissolved in 100 µL PBS the mammary pad was done. When the tumor reached at palpable stage, mice were randomly arranged, 5 per group. The tumor area was shaved with Veet shaving cream, to locate the tumor and accurately measure the volume. Complexes loaded with CA nanoparticles were formed with 8 μL of CaCl2 mixed in 200 μL of DMEM supplied with bicarbonate (44 mM) and selected single integrin siRNAs. Incubation at 37 °C for 30 min was done for the variables and complex generated was immediately put on ice to stop formation of particles and slow the aggregation. Intravenous injection of 100 μL treatment complexes at right/left caudal vein was done. Second or subsequent doses were administered two days later. Tumor volume and tumor weight was monitored frequently. Tumors were collected and weighed at the end of study and images of isolated tumors were taken. The following formula was used to calculated tumor volume:
Tumor volume (in mm3) = Length × (width)2/2
2.7. SDS-PAGE and Western Blotting
Cells seeded and treated with integrin loaded CA complexes were lysed after 48 h using 150 μL lysis buffer which consisted of 20 mM Tris-HCl (at pH 7.5), 150 mM NaCl, 50 nM NaF, 1 mM EDTA, 1 mM EGTA, phosphate inhibitor cocktail 2, 50 mM beta-glycerolphosphate, complete EDTA-free protease inhibitor, and 1% Triton X-100. After protein lysis was performed, the samples were centrifuged at for 13,000× g rpm for 8 min, followed by collection of supernatant. The concentration of proteins in the lysates was determined using Quick Start Bradford Protein Assay kit. Standard curve was prepared using the BSA standards available. Equal amounts of proteins (9 μg/well) were loaded in precast gels, and the SDS-PAGE was run, followed by transfer of proteins from gel to nitrocellulose membrane using turbo transfer. Transferred proteins on nitrocellulose membrane were blocked with 5% non-fat dry milk prepared in 1X tris buffered saline with tween (TBST) (at pH 7.4, with 0.1% Tween 20) for 1 h at room temperature. The blots were then probed with various primary antibodies for overnight at 4 °C. After overnight probing, blots were washed with 1X TBST to remove excess primary antibody. This was followed by probing the blots for 1 h at room temperature, with secondary antibody conjugated with horse radish peroxide, followed by removing of excess secondary antibody with 1X TBST washing. Clarity Western Enhanced Chemiluminescence (ECL) substrate was applied onto the membrane for 5 min and Gel Document System (Bio Rad) was used for membrane visualization. To re-probe the same membrane with different primary antibody, the membrane was rinsed with 1X TBST to remove excess ECL substrate. Restore™ Plus Western blot stripping buffer was used to strip the membrane of the previously probed antibodies. After removing the antibodies, the nitrocellulose membrane was again blocked with 5% non-fat dry milk in 1X TBST. Different primary antibodies were used, repeating the same procedure as mentioned above.
3. Statistical Analysis
Statistical analysis was performed using SPSS (version 17 for windows) for in vivo tumor regression results. Analysis and comparison of the significant difference between differently treated groups was done by LSD post-hoc test for one-way ANOVA. Data were statistically significant with p < 0.05 (indicated by *).
4. Results
4.1. Intracellular Delivery of CA Assisted Single Integrin siRNAs in Breast Cancer Cells
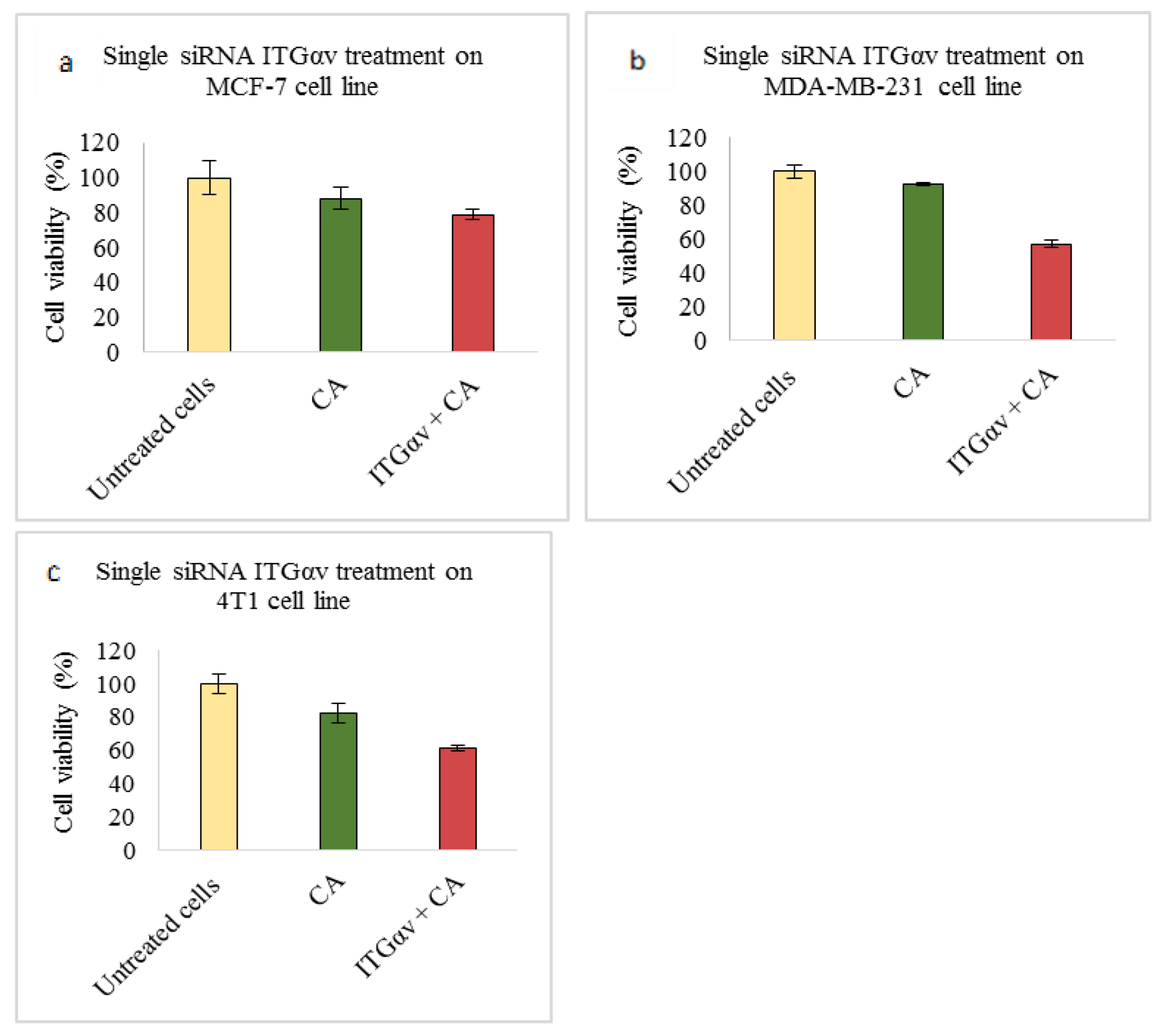
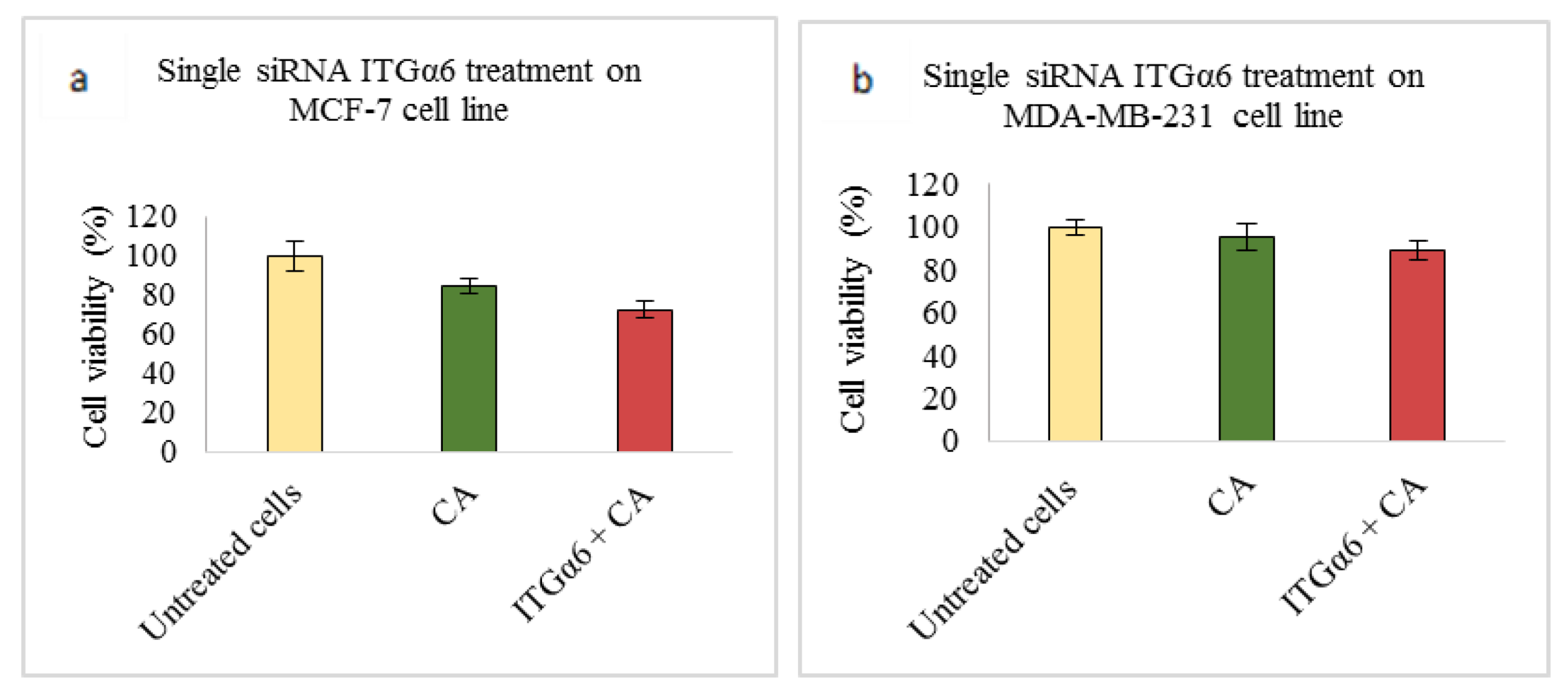
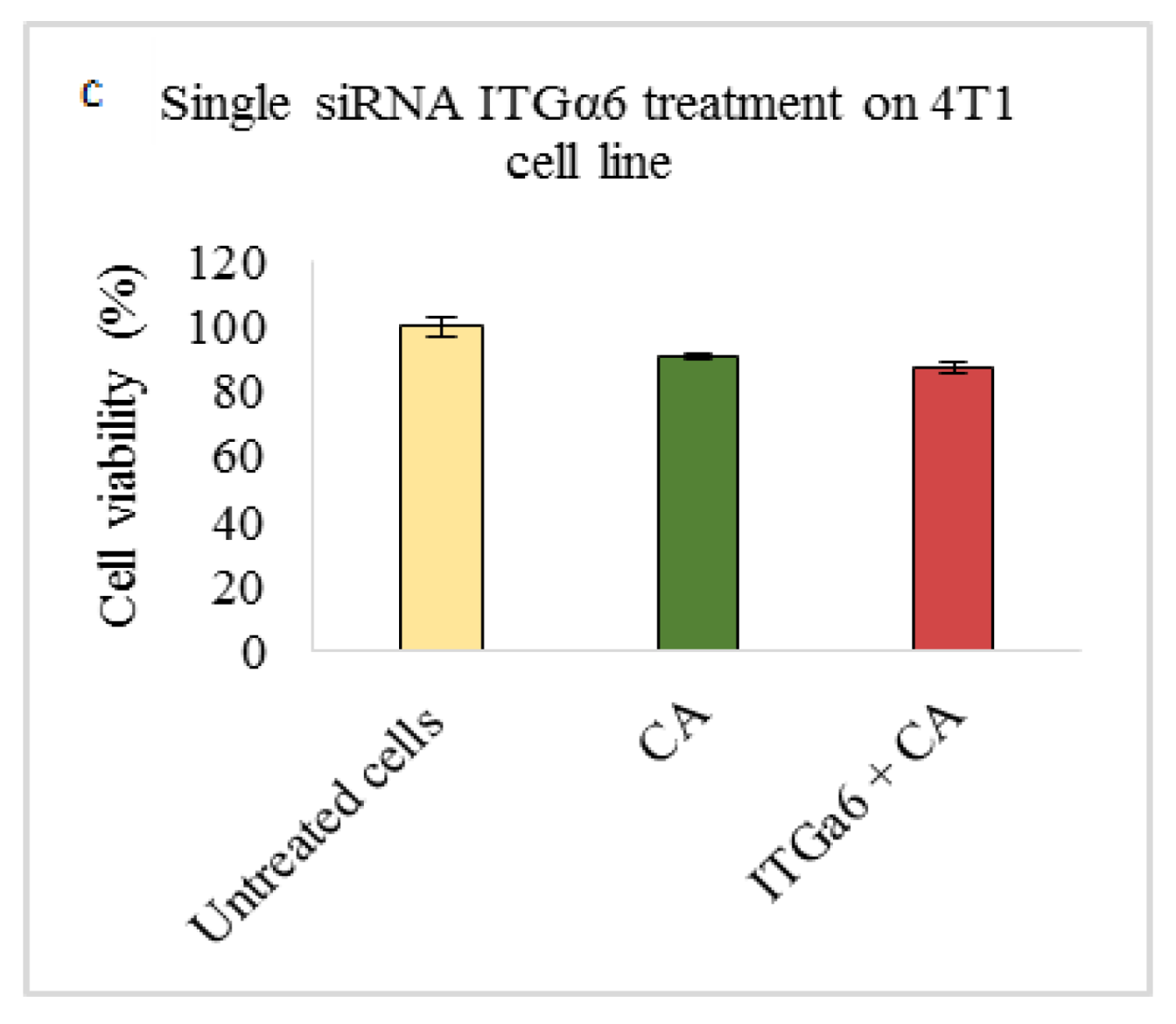
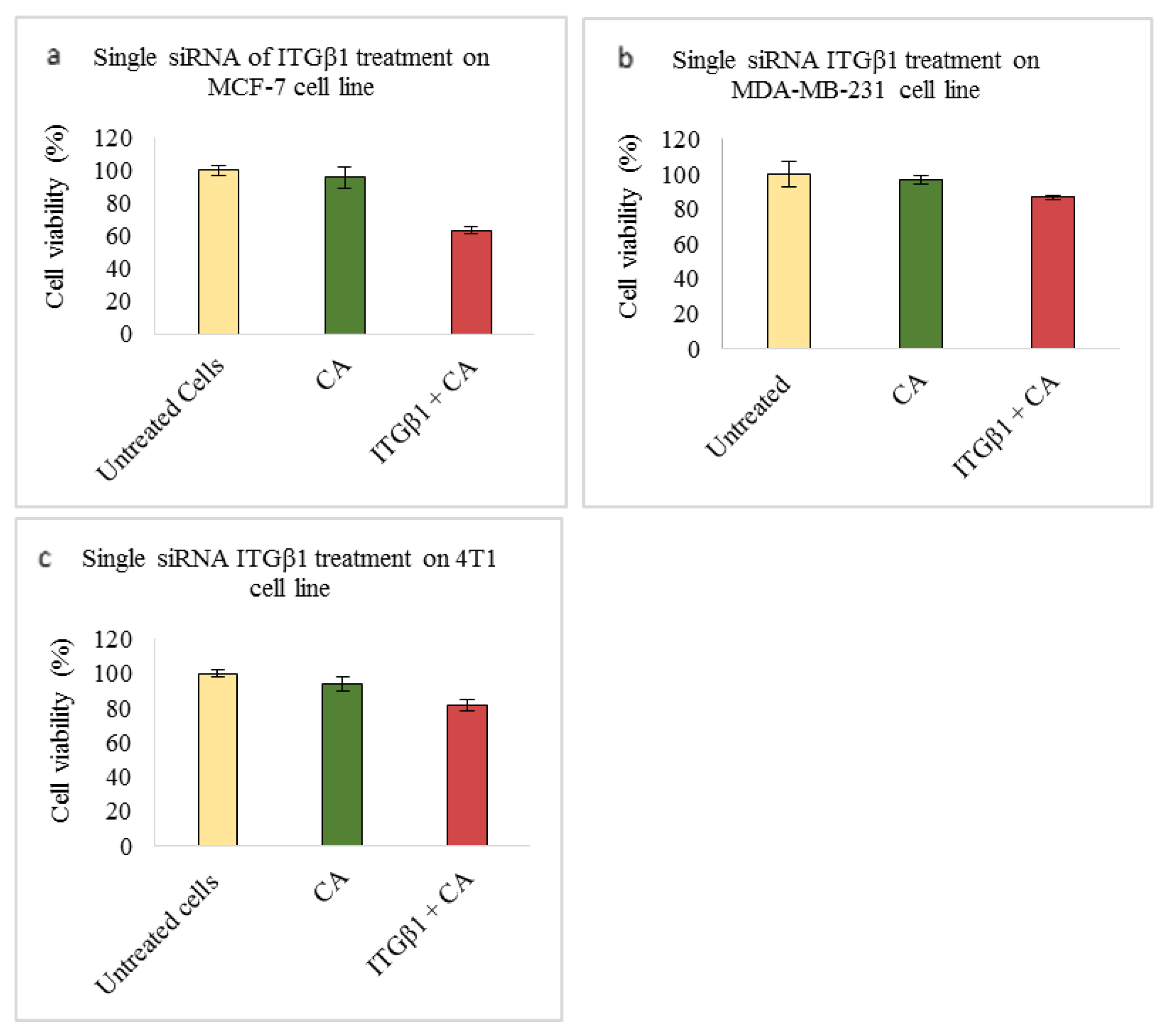
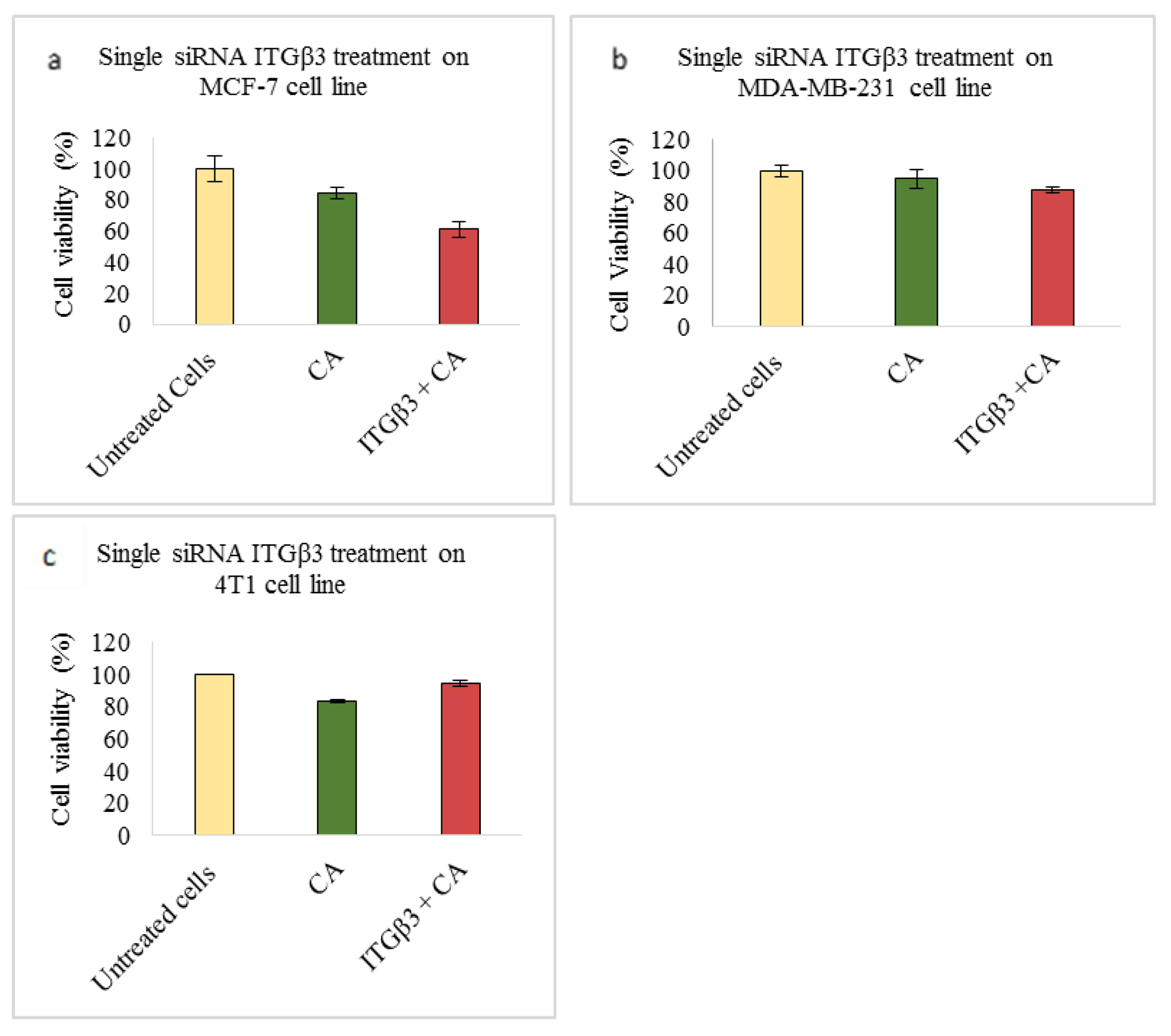
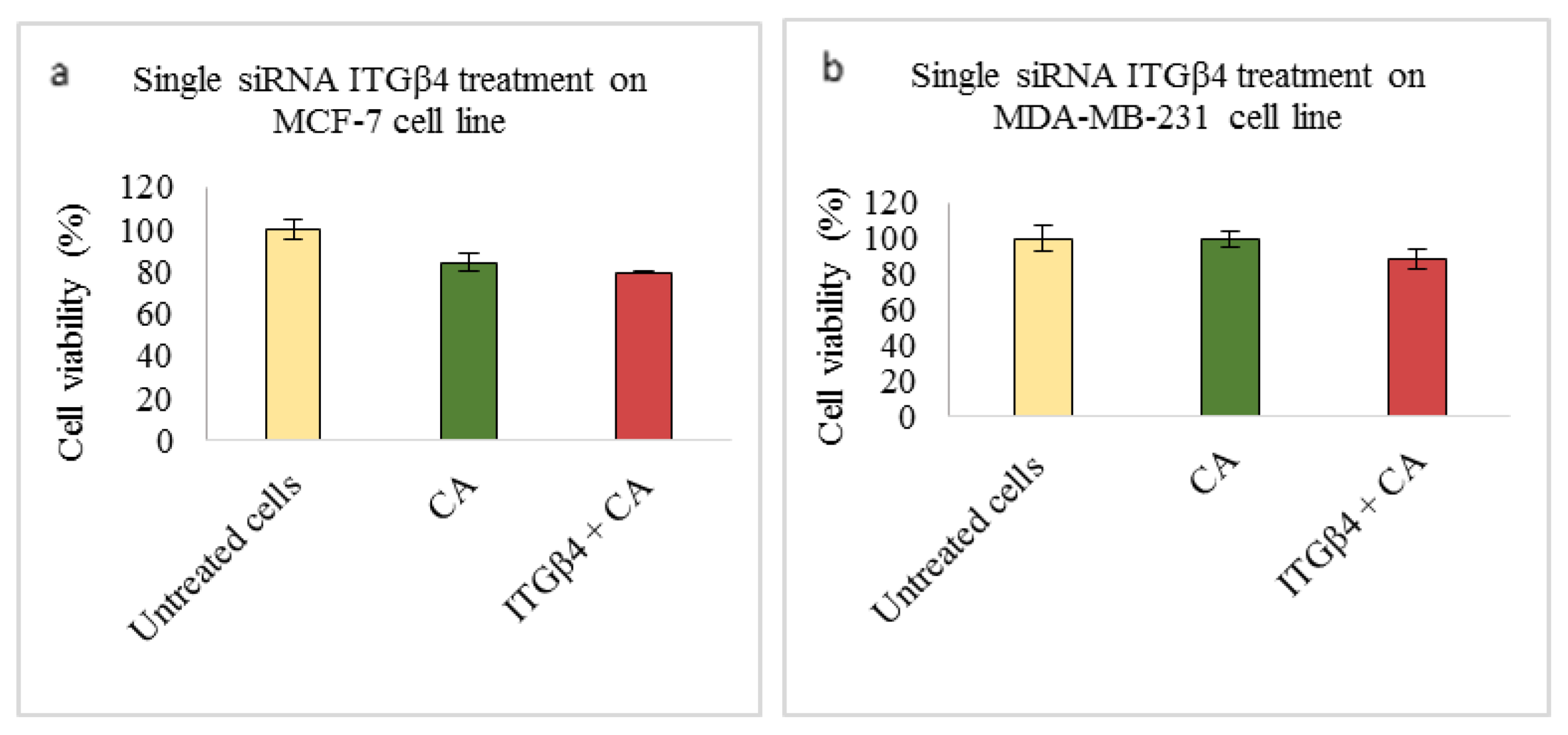
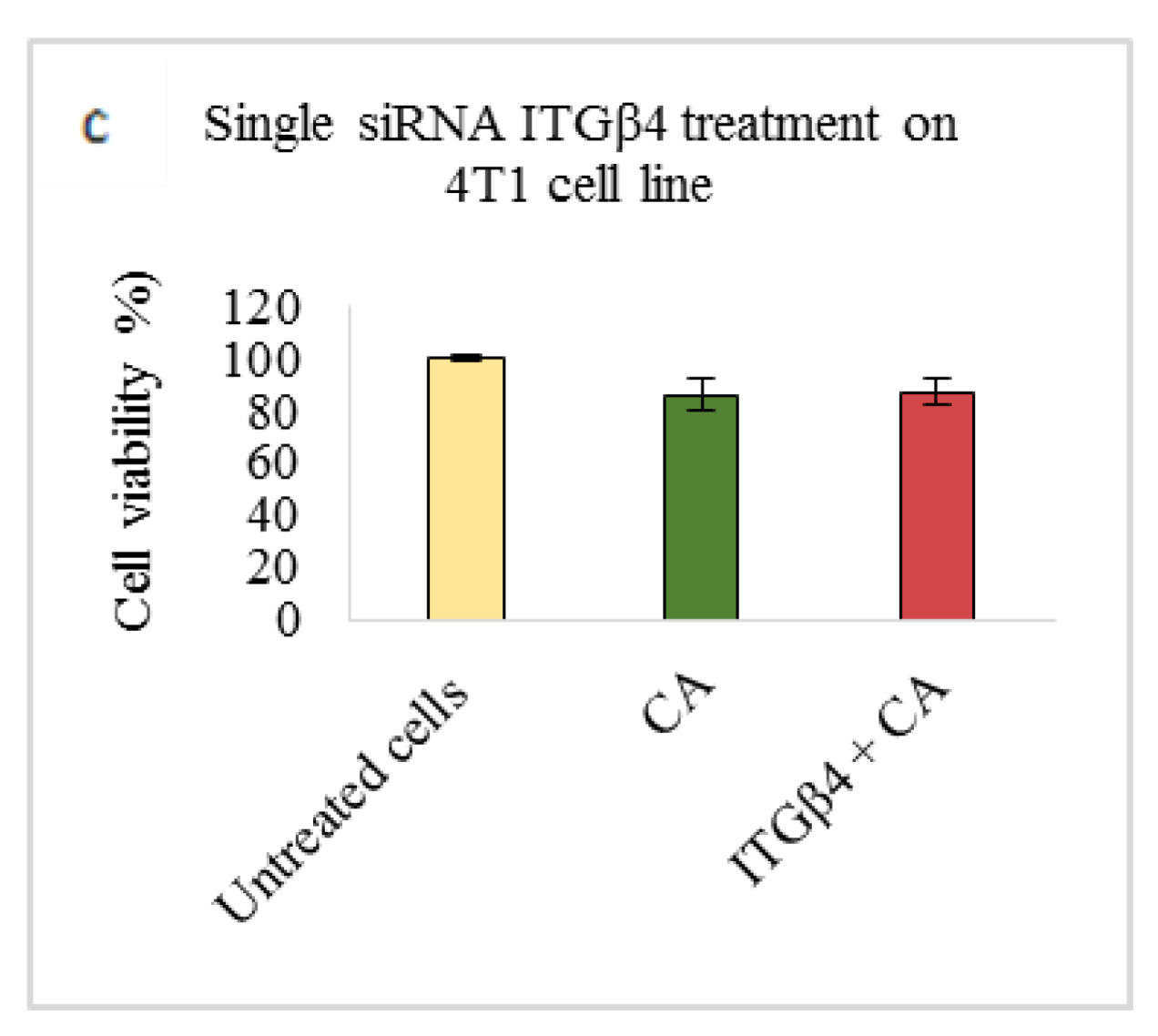
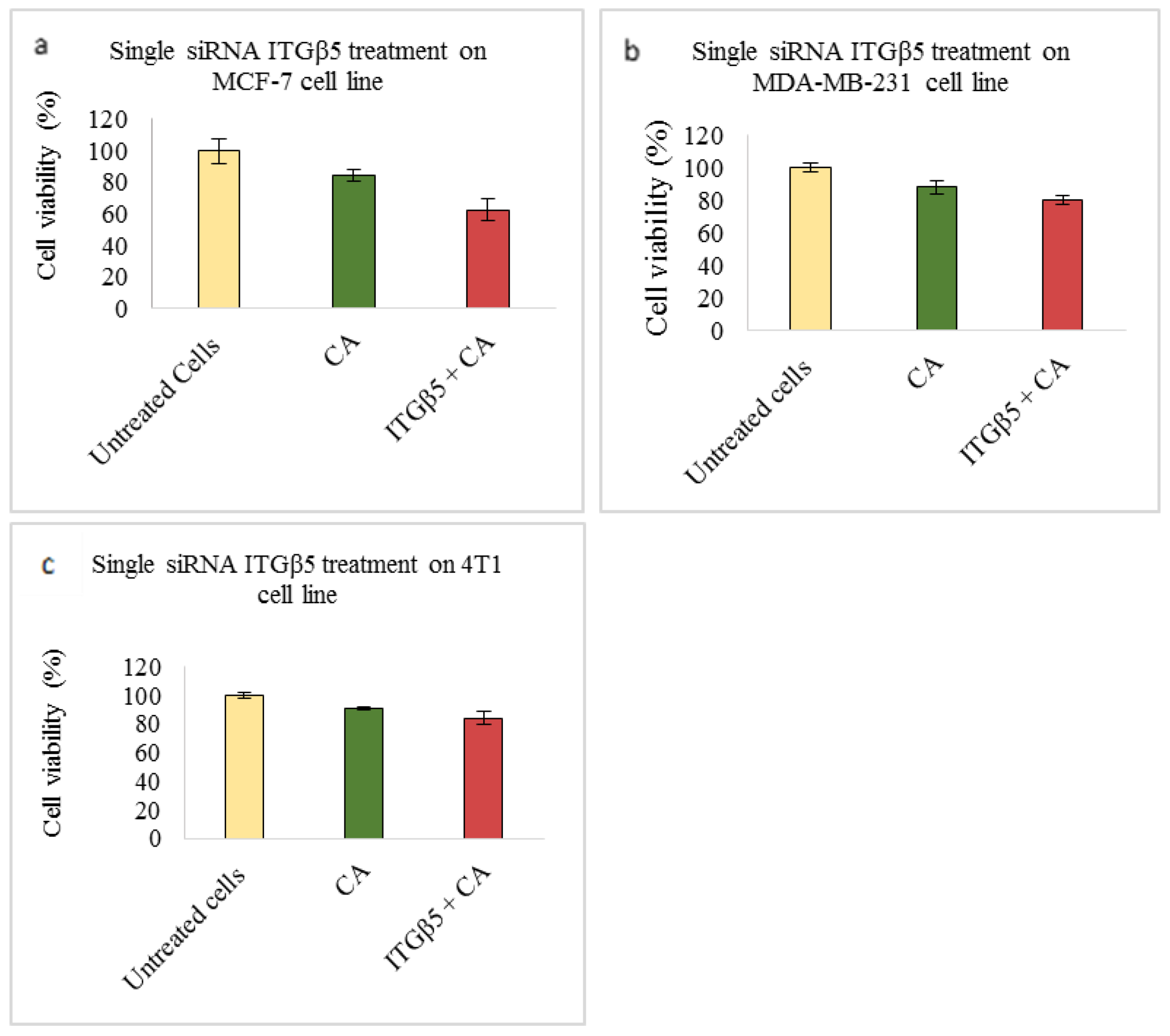
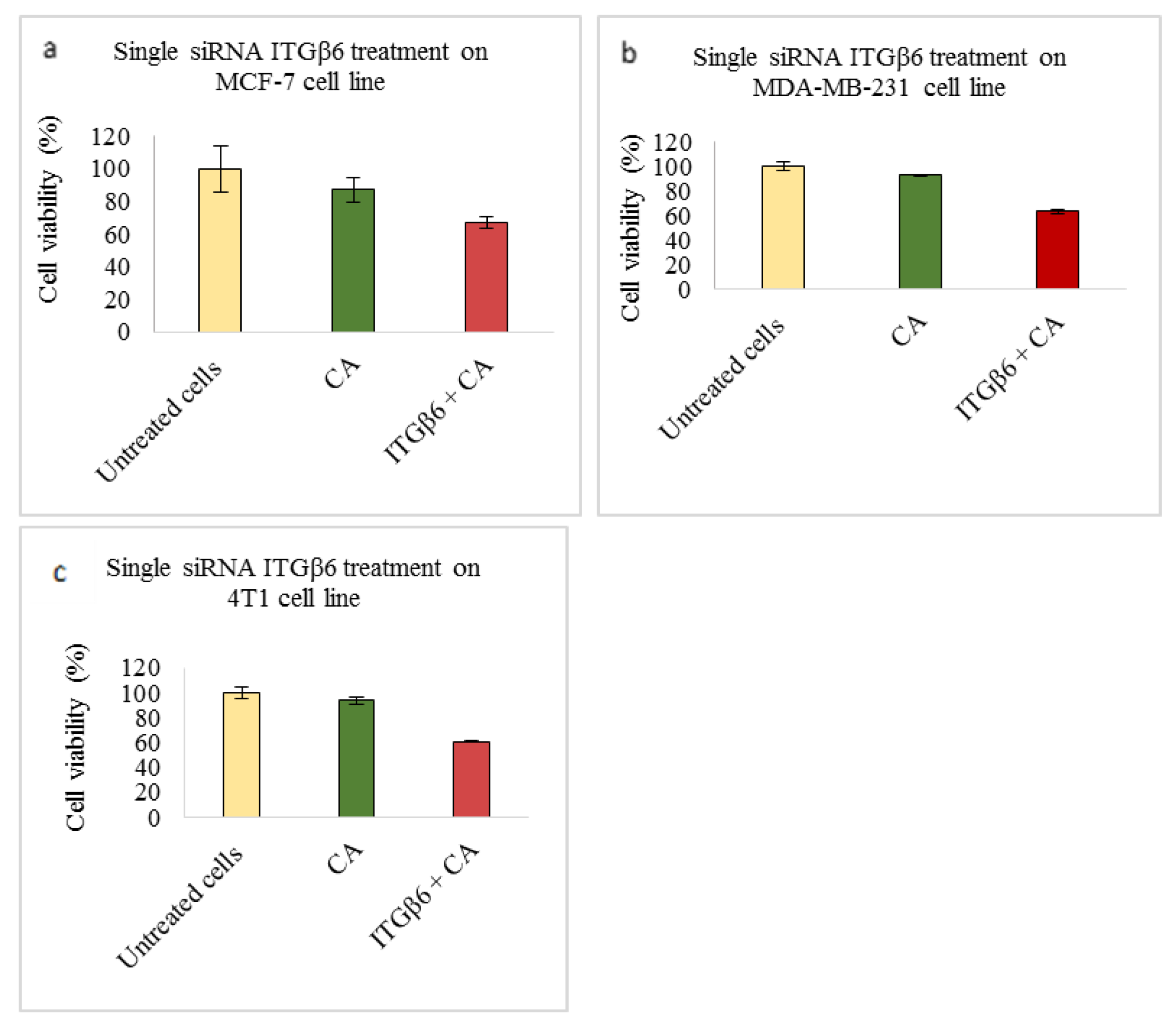
Based on MTT results we found that delivery of siRNA ITGαv loaded with CA showed less cell viability in MDA-MB-231 and 4T1 cells (Figure 1b,c respectively) while in MCF-7 cells no such visible reduction was observed (Figure 1a). However, siRNA ITGα6-CA complex reduced cell viability only in MCF-7 cells (Figure 2a), remaining unaffected in MDA-MB-231 and 4T1 cells (Figure 2b,c respectively). Upon treatment with ITGβ1, siRNAs complexed with CA, cell viability decreased in MCF-7, MDA-MB-231 and 4T1 cells (Figure 3a–c respectively). siRNA ITGβ3-CA complex Figure 4a) reduced cell viability only in MCF-7 cells, remaining unaffected in MDA-MB-231 and 4T1 cells (Figure 4b,c respectively). Treatment of ITGβ4 siRNA-CA delivery did not reduction in cell viability in MCF-7 and 4T1 cells (Figure 5a, c respectively). However, in MDA-MB-231, cell viability was reduced (Figure 5b). Similar to ITGβ1, treatment with, ITGβ5, and ITGβ6 siRNAs complexed with CA also reduced the cell viability in all the three cell lines (Figure 6a–c and Figure 7a–c respectively). Table 2 shows the actual cytotoxicity caused in MCF-7; Table 3 and Table 4 show the actual cytotoxicity caused in MDA-MB-231 and 4T1 cells respectively after delivery of siRNA-CA complex.
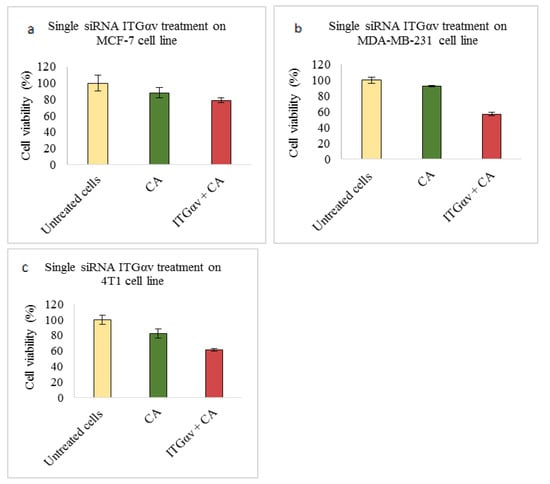
Figure 1.
NP-loaded siRNA effect on cell viability against ITGαv single gene after 48 h of MTT assay. CA-siRNA complexes were prepared at 1 mL final volume consisting of 1 nM siRNAs + 4 mM CaCl2 into bicarbonate buffered DMEM (pH 7.4), followed by incubation for 30 min. The prepared complex was transfected for 48 h and absorbance was taken at 595 nm with 630 nm as reference wavelength. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
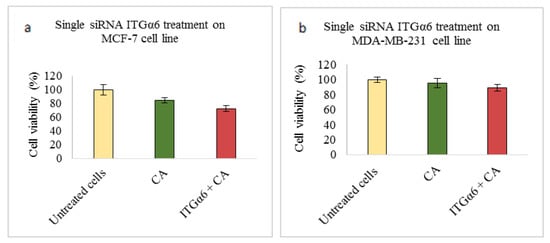
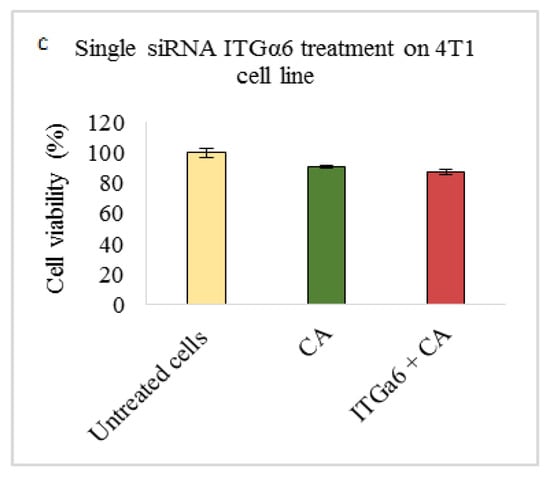
Figure 2.
NP-loaded siRNA effect on cell viability against ITGα6 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
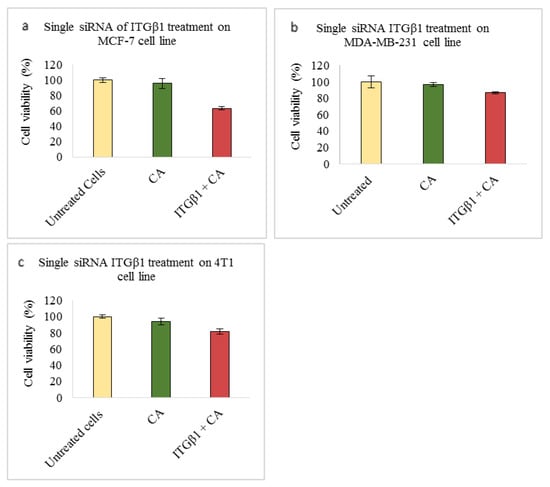
Figure 3.
NP-loaded siRNA effect on cell viability against ITGβ1 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
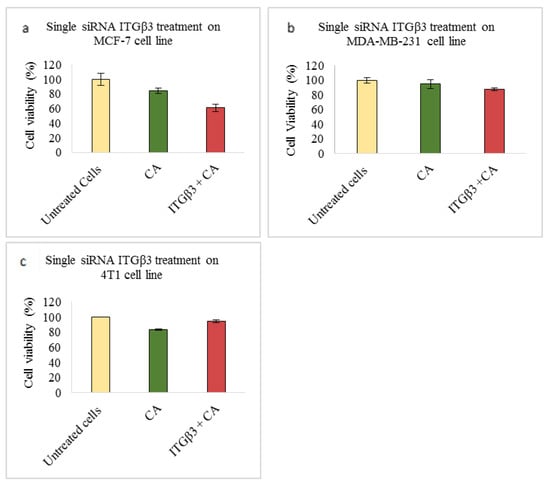
Figure 4.
NP-loaded siRNA effect on cell viability against ITGβ3 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
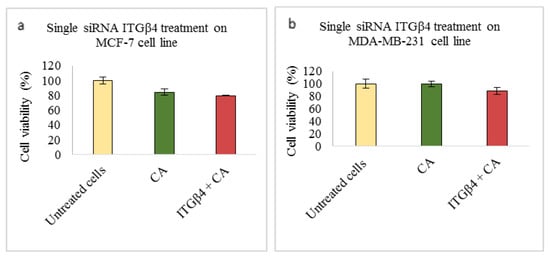
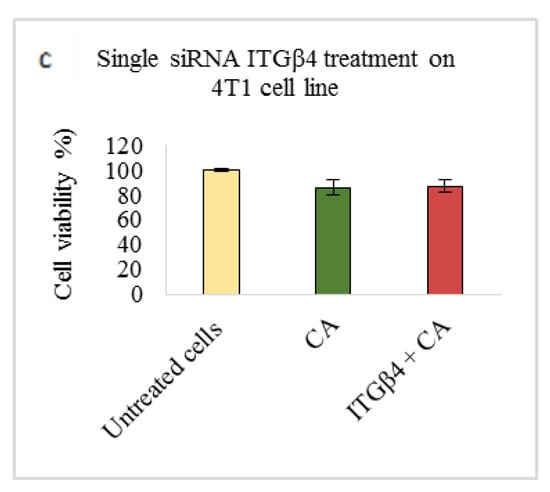
Figure 5.
NP-loaded siRNA effect on cell viability against ITGβ4 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
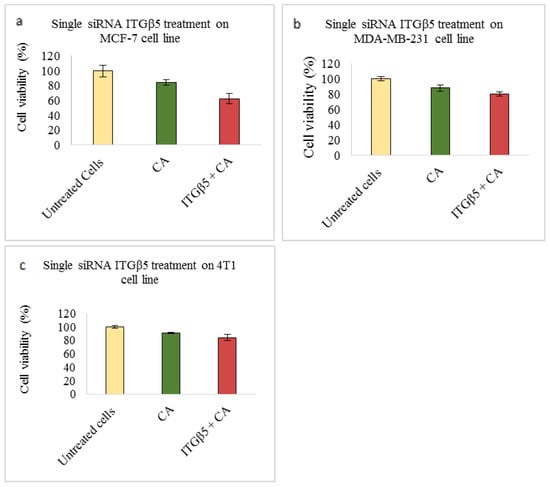
Figure 6.
NP-loaded siRNA effect on cell viability against ITGβ5 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.
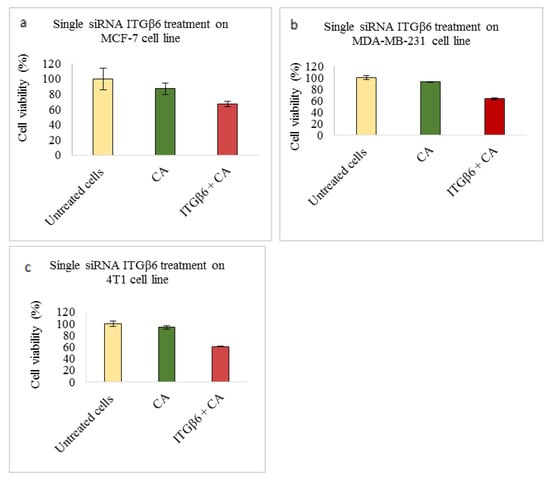
Figure 7.
NP-loaded siRNA effect on cell viability against ITGβ6 single gene after 48 h of MTT assay. Data are shown as mean ± S.D for (a) MCF-7 cells, (b) MDA-MB-231 cells, and (c) 4T1 cells.

Table 2.
Enhancement of cytotoxicity of single integrin siRNA CA transfections in MCF-7 cells.

Table 3.
Enhancement of cytotoxicity of single integrin siRNA transfections in MDA-MB-231 cells.

Table 4.
Cytotoxicity enhancement of single integrin siRNA transfections in 4T1 cells.
4.2. Integrins Link with PI3-Kinase/AKT and MAPK Signalling Pathways in Breast Cancer
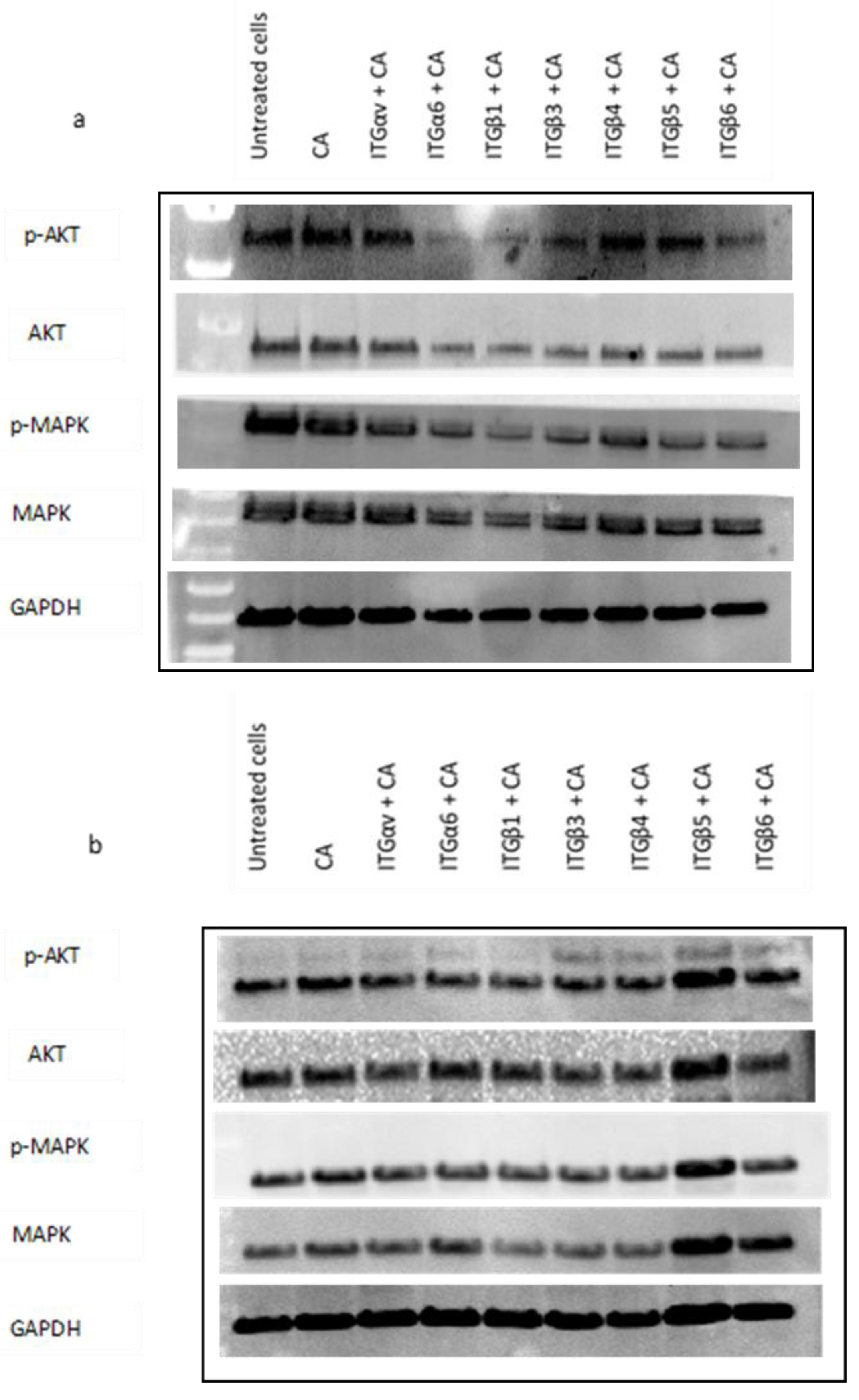
To study whether delivery of single integrin siRNAs loaded with CA-NP in cell lines can regulate the expression of total and phosphorylated AKT and MAPK, we performed Western blot assay. In MCF-7 cells, from Figure 8a we can see the expression of phosphorylated AKT (p-AKT), and total MAPK was significantly reduced in α6, β1, β3, β4, β5, and β6 integrin siRNA treatments delivered using CA complex compared to CA control. The expression of total AKT and phosphorylated MAPK (p-MAPK) was reduced in all the seven treatments.
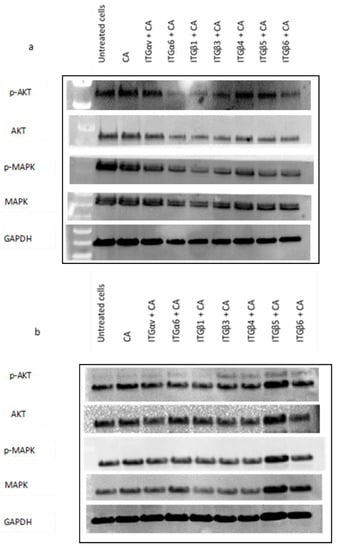
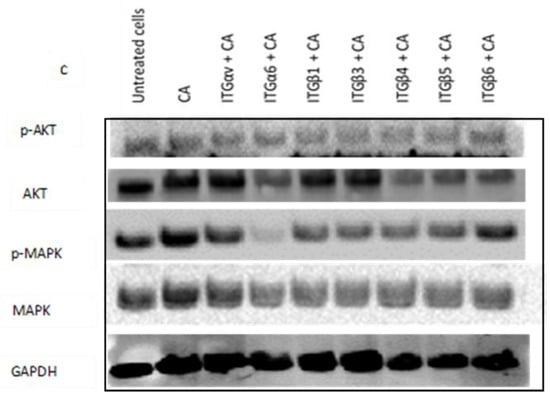
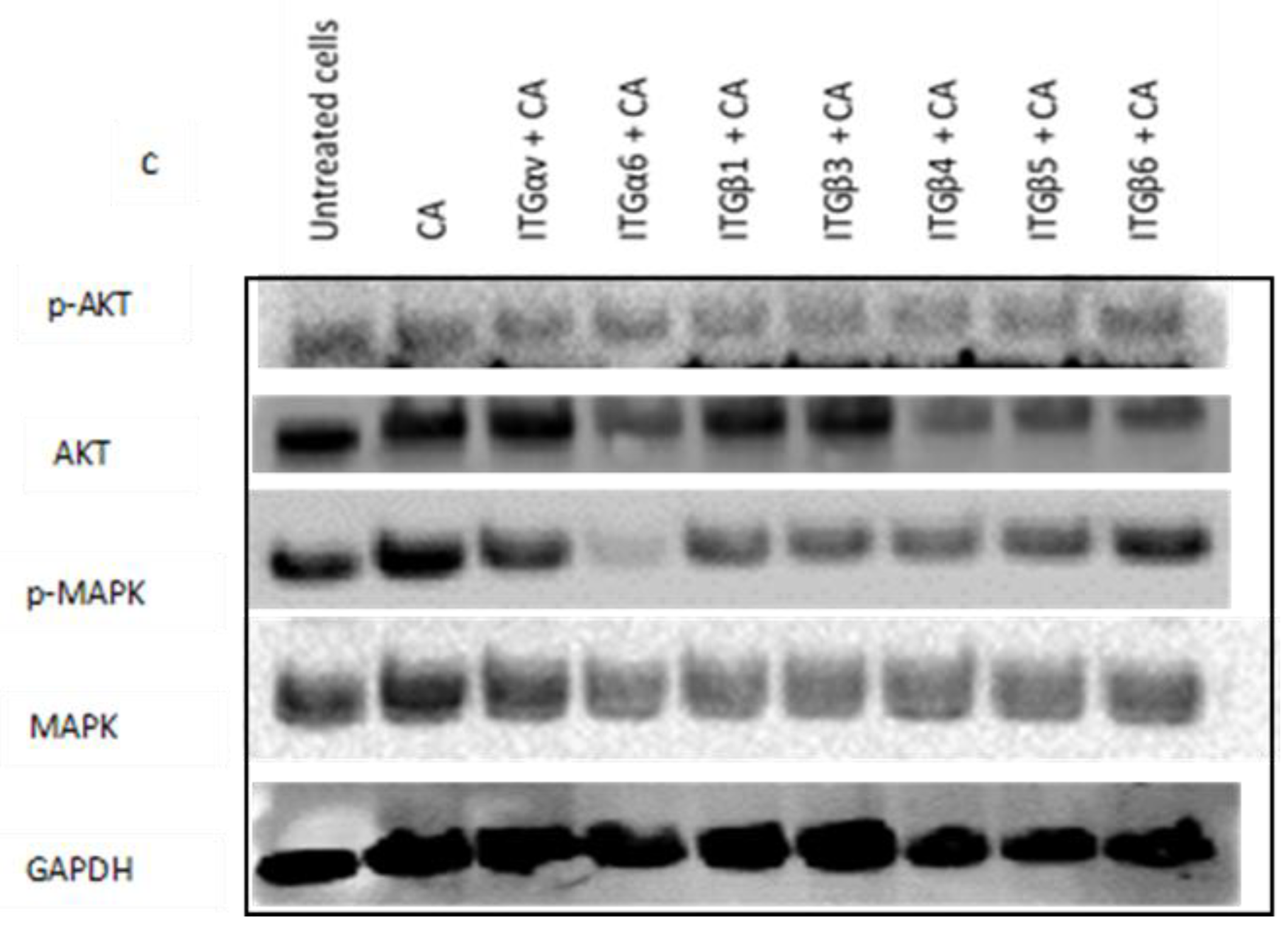
Figure 8.
Impact of CA loaded single integrin siRNA intracellular delivery in (a) MCF-7, (b) MDA-MB-231, and (c) 4T1 cells. Cells were incubated with CA loaded single integrin (ITGαv, ITGα6, ITGβ1, ITGβ3, ITGβ4, ITGβ5, and ITGβ6) siRNAs for 48 h. Post incubation cells were lysed for Western blot assay. Then, 9 µg of proteins were loaded uniformly on SDS-PAGE, and transferred onto nitrocellulose membrane for detection of p-AKT, total AKT, p-MAPK, total MAPK, and housekeeping gene GAPDH expression.
In MDA-MB-231 cells, the siRNA delivery with CA nanoparticles reduced the intensity of p-AKT and p-MAPK in ITGαv, ITGα6, ITGβ1, ITGβ3, ITGβ4, and ITGβ6 siRNAs-CA treatments compared to CA control. The intensity of total AKT was reduced with integrin ITGαv, ITGβ1, ITGβ3, ITGβ4, and ITGβ6 siRNAs-CA treatment. On the other hand, the intensity of total MAPK was reduced with ITGαv, ITGβ1, ITGβ3, and ITGβ4 siRNA-CA treatments (Figure 8b).
Western blot analysis showed reduced expression of p-AKT in 4T1 cells upon all the siRNA-CA treatments except siRNA ITGβ6 + CA treatment, while as total AKT expression was reduced in all the groups excluding siRNA ITGαv + CA treatment group. On the other hand, p-MAPK and total MAPK reduction was observed in all the treatment groups (Figure 8c).
4.3. In Vivo Delivery of Single siRNA Integrins Loaded with CA Nanoparticles
4.3.1. Body Weight of Mice
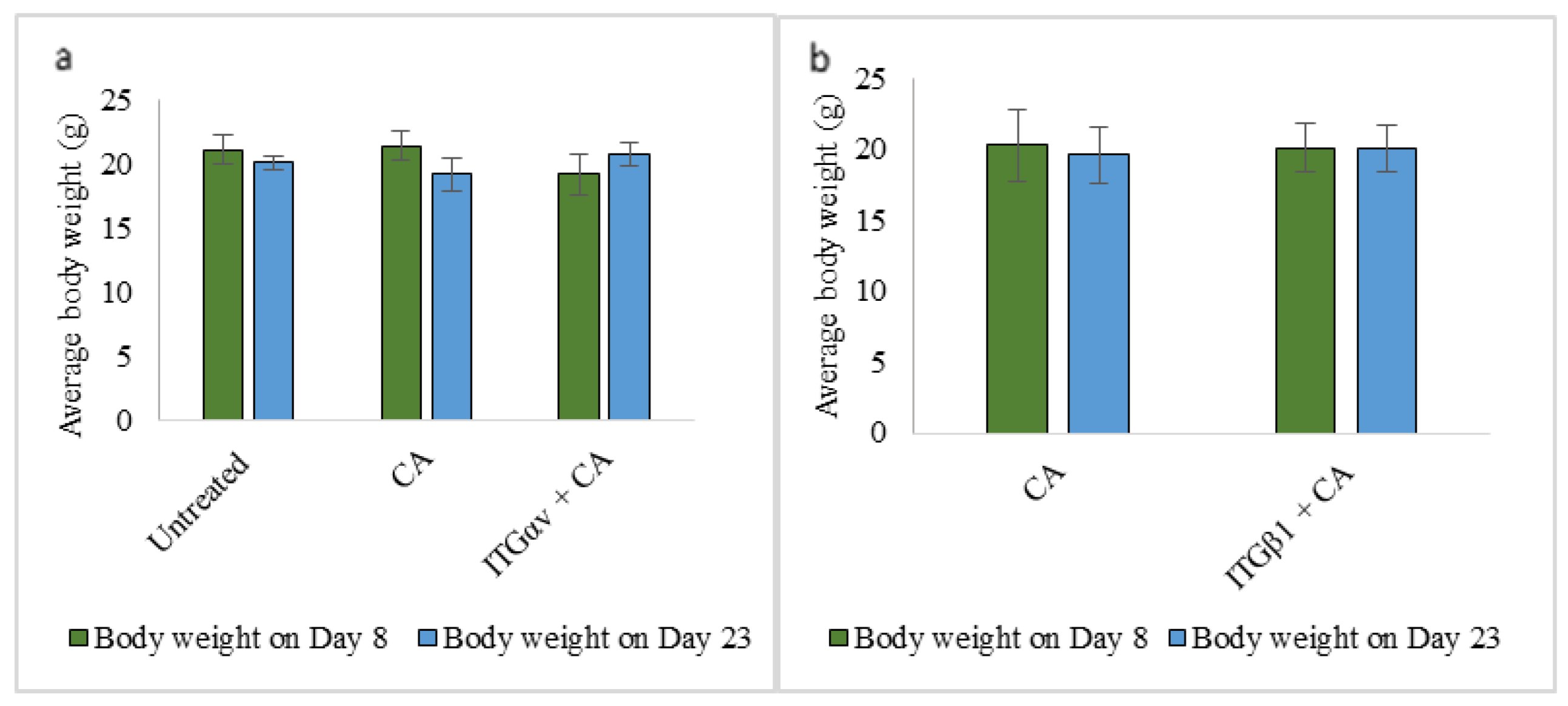
Average body weight in both treatment and control groups remained unchanged. For data simplicity, only two treatment group body weights have been shown (Figure 9a,b).
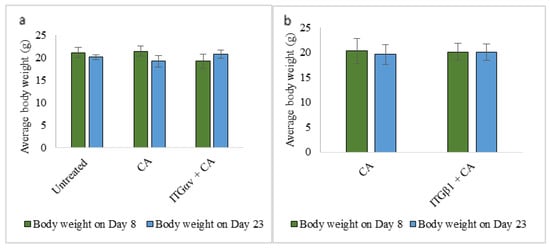
Figure 9.
Average body weight of mice on Day 8 and Day 23 of tumor regression for untreated, CA treated and (a) ITGαv + CA (b) ITGβ1+ CA treatment. Data are shown as average body weight of 5 mice ± S.D.
4.3.2. Tumor Regression Analysis
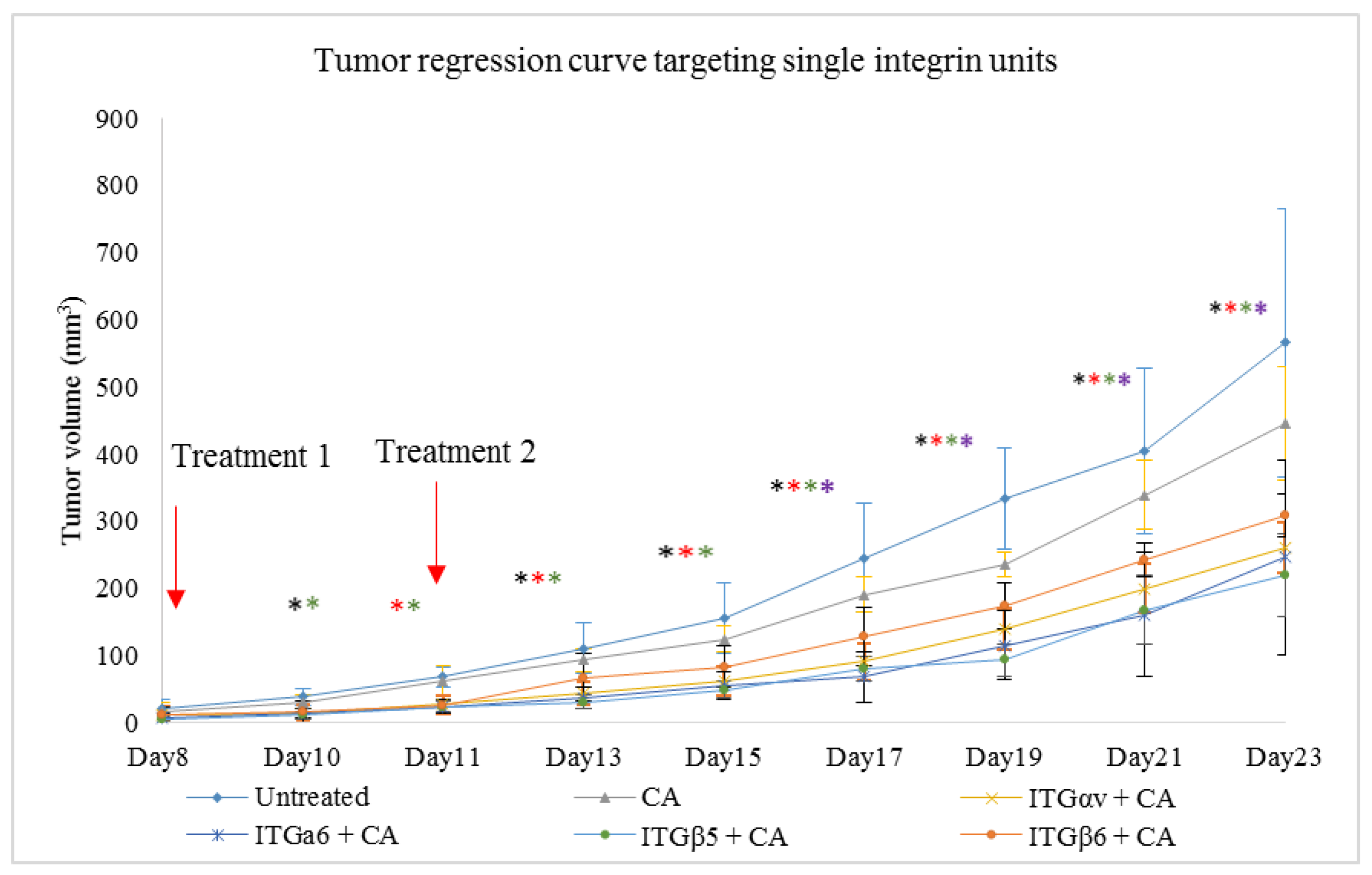
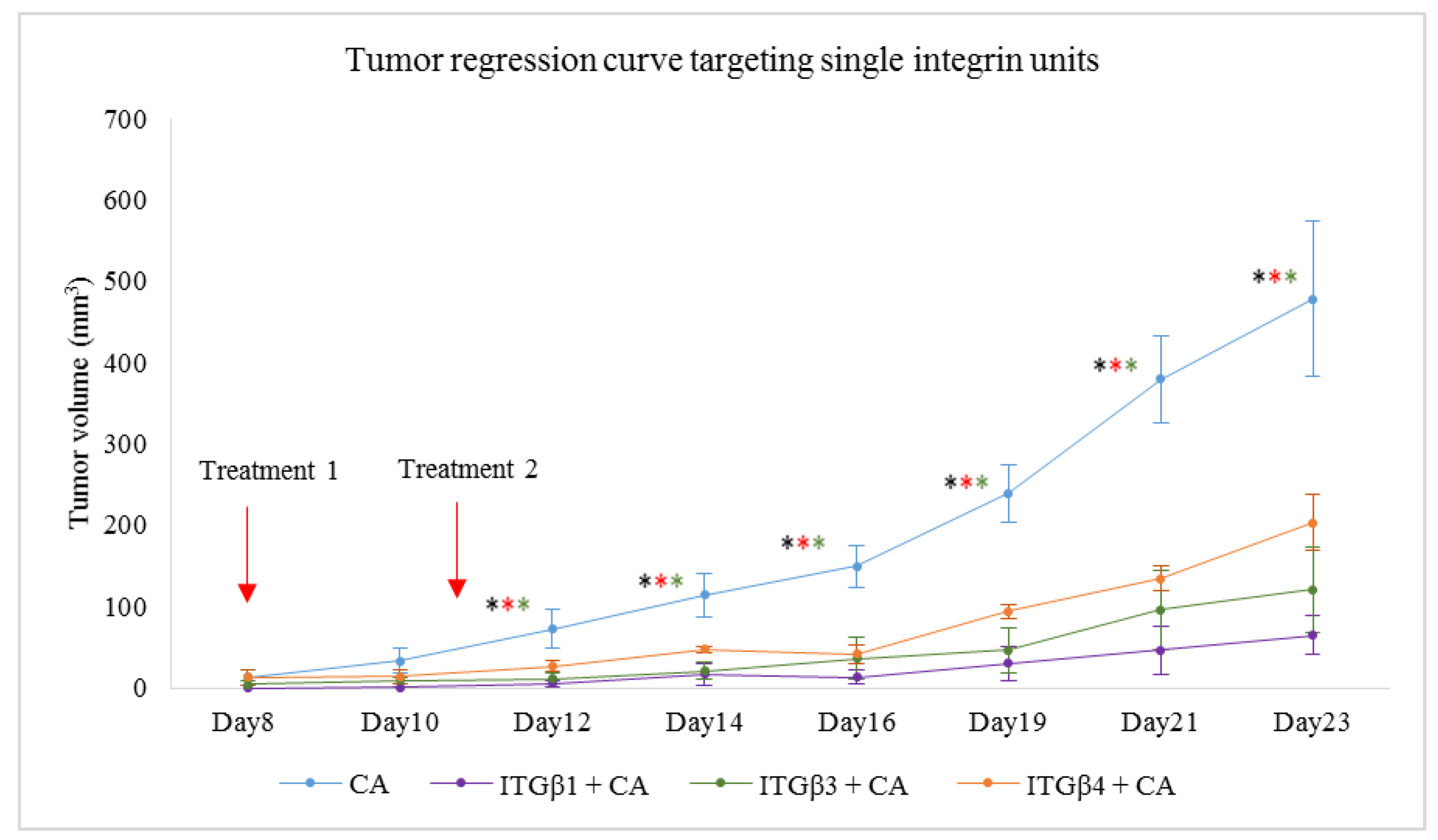
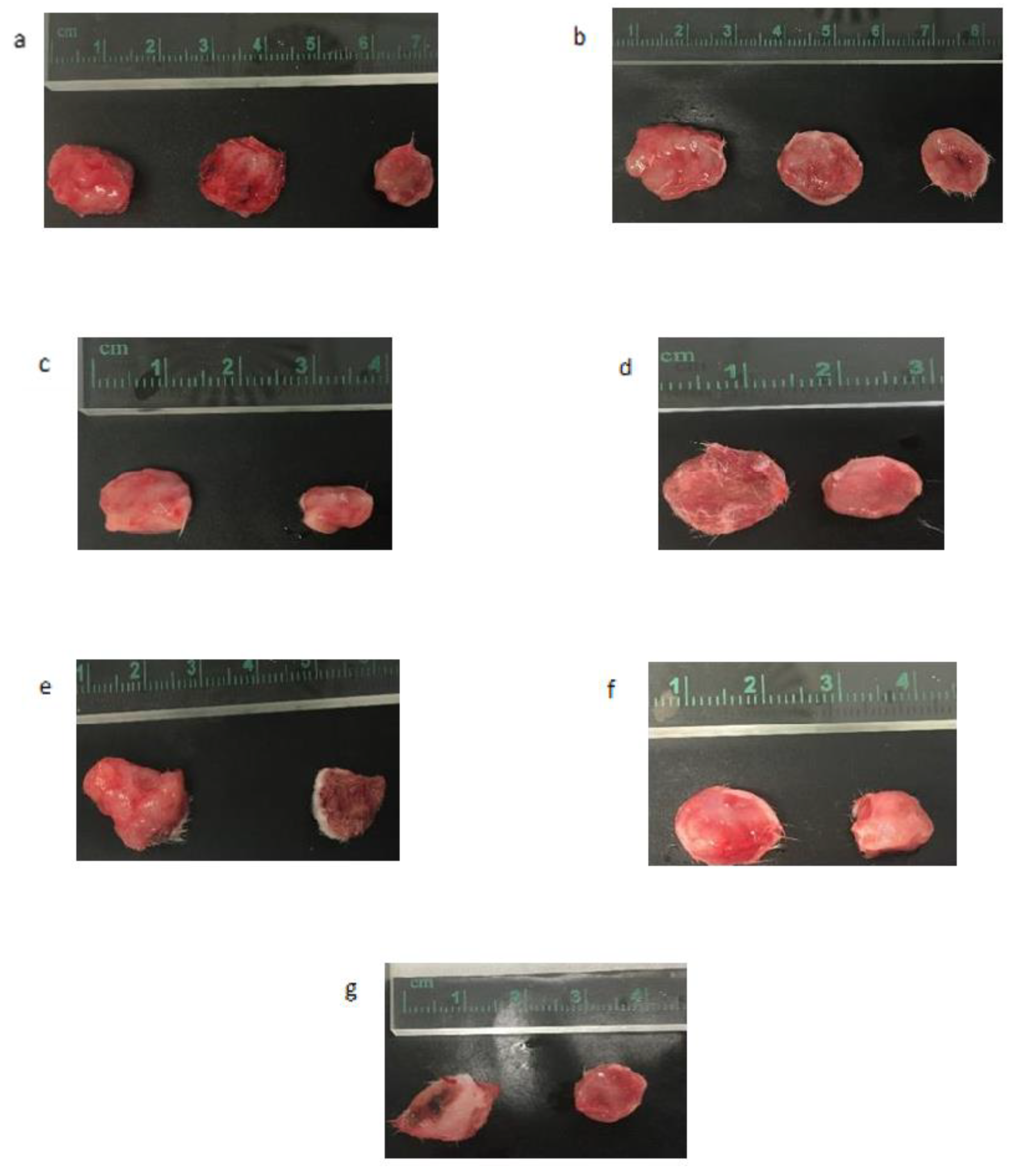
Delivery of single integrin siRNAs using CA-NPs caused reduction in tumor volumes compared to both untreated and CA-NP treated groups. Compared to in vitro study where 1 nM of the siRNA was used to transfect the cells in each well of a 24-well plate, 50-fold more siRNA was administered intravenously per dose in the form of 100 ul of particle suspension. From Figure 10, we can see that, compared to untreated and CA controls, the tumor volume was lesser in the treatment groups. The significant decrease in tumor volume with p < 0.05 compared to CA-NP treated group was observed on Day 11, Day 13, Day 15, Day 17, Day 19, Day 21, and Day 23 in siRNA ITGαv + CA and ITGβ5 +CA treatments. For siRNA ITGα6 +CA, the tumor regression was significant on Day 11, Day 13, Day 15, Day 17, Day 19, and Day 21 of measurements. For siRNA ITGβ6 + CA treated group, while the trend for tumor regression was reduced, the significant difference with p < 0.05 was observed on Day 17, Day 19, Day 21, and Day 23. Table 5 shows the fold decrease in the tumor volume compared to CA-NP treated groups at different time points. Figure 11 shows reduced tumor volumes for ITGβ1, ITGβ3 and ITGβ4 siRNA treatments loaded with CA. As there was no significant difference between untreated and CA treated groups, the untreated group was not repeated for further experiments. Treatment of siRNA ITGβ1 + CA, siRNA ITGβ3 + CA, and siRNA ITGβ4 + CA showed significant reduction in tumor (p < 0.05) at all the time points compared to CA. Table 6 shows the fold decrease in the tumor volume compared to CA groups on different days. Excised tumors at the end of the study of siRNA + CA treated and CA treated groups are shown in Figure 12, with tumor of treatment groups smaller compared to CA control.
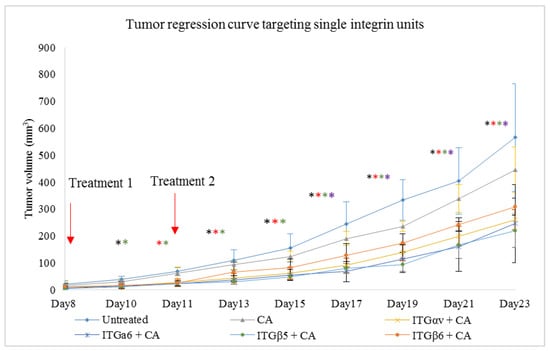
Figure 10.
CA bound siRNA treatments against integrin genes in 4T1 tumor induced mice. Subcutaneous injection of 4T1 cells was done in the mice mammary pad. Once tumors were palpable, mice were treated intravenously via tail vein with 100 μL of CA-siRNA(s) generated in 4 μL of 1 M CaCl2 and individual siRNA against ITGαv, ITGα6, ITGβ5, and ITGβ6 genes. Two treatment doses were given, one on Day 8 and the second on Day 11. In total, 5 mice per group were used and data were represented as mean ± SD. Tumor outgrowth of mice treated with CA-siRNA against ITGαv, ITGα6, ITGβ5, and ITGβ6 genes values are significant for * representing p < 0.05 for ITGαv + CA, * representing p < 0.05 for ITGα6 + CA, * representing p < 0.05 for ITGβ5 + CA, * representing p < 0.05 for ITGβ6 + CA.

Table 5.
CA loaded siRNA ITGαv, ITGα6, ITGβ5, and ITGβ6 treated group (in folds) with tumor reduction in comparison to CA-NP treatment.
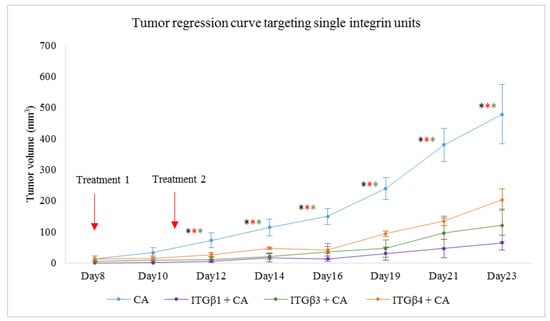
Figure 11.
Tumor induced mice were treated intravenously with 100 μL of CA-siRNA(s) formed in 4 μL of 1 M CaCl2 and individual siRNA against ITGβ1, ITGβ3, and ITGβ4 genes. Two treatment doses were given, on Day 8 and on Day 11. A total of 5 mice per group were used and data were represented as mean ± SD. Tumor outgrowth of mice treated with CA-siRNA against ITGαv, ITGα6, ITGβ5, and ITGβ6 genes values are significant for * representing p < 0.05 for ITGβ1 + CA, * representing p < 0.05 for ITGβ3 + CA, * representing p < 0.05 for ITGβ4 + CA.

Table 6.
CA loaded siRNA ITGβ1, ITGβ3, and ITGβ4 treated group (in folds) with tumor reduction in comparison to CA-NP treatment.
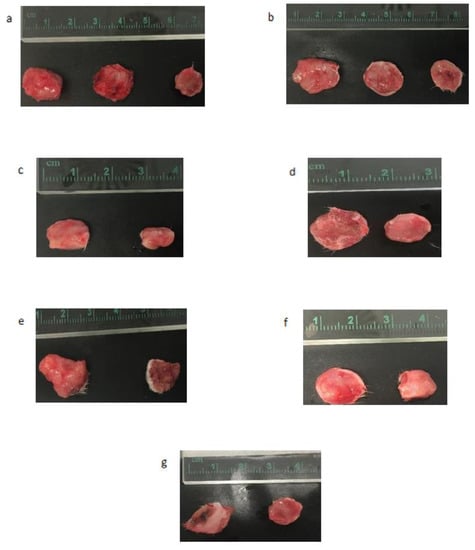
Figure 12.
Excised tumors at the end of the treatment for various single siRNAs loaded with CA nanoparticles, targeting various integrin genes in comparison to CA control. (a) Untreated, CA, and ITGαv + CA. (b) Untreated, CA, and ITGα6 + CA. (c) CA and Ca + ITGβ1. (d) CA and Ca + ITGβ3. (e) CA and Ca + ITGβ4. (f) CA and Ca + ITGβ5. (g) CA and Ca + ITGβ6.
5. Discussion
Integrins maintain cellular architecture, survival, proliferation, migration, and overall tissue integrity by binding to ECM, cytoskeleton, and connecting via various cell adaptor proteins and ligands [16,18]. Integrin αv, α6, β1, β3, β4, β5, and β6 subunits are the essential targets responsible for tumor proliferation, progression, and growth. High level of integrins or high activity of the signaling components associated with them synergize with oncogenes, growth factors, and other cell adhesion proteins, leading to the activation of signaling pathways and affecting both upstream and downstream molecules [6,21,28,31,44,45,46,47,48]. Therefore, they serve an essential targeting molecule to control tumor development. Dysregulation in the integrin expressions which participate in breast cancer development using siRNA-CA nano-formulation may lead to reduced association of the integrins with other cell adhesion molecules and proteins, thus hampering the cancer proliferation and survival.
In our integrin siRNA transfection experiments MCF-7 cells showed no visible decrease in cell viability when they were transfected with CA loaded with ITGαv siRNA (Figure 1a), while a significant reduction in viability of ~57% was observed in MDA-MB-231 (Figure 1b), causing about 35% cytotoxicity (Table 3) and viability of ~61% in 4T1 cells (Figure 1c) which caused about 21% cytotoxicity (Table 4). Research shows MDA-MB-231 cells contain high expression of ITGαv causing different signaling [25]. Targeting this sub-unit in MDA-MB-231 cells might have reduced the viability by mainly targeting AKT and MAPK pathways, as from Western blot analysis we could see reduced band intensities after siRNA-CA transfection (Figure 8b). On the other hand, in MCF-7 cells phosphorylated AKT and total MAPK showed no decrease in band intensity while total AKT and phosphorylated MAPK showed significant reduction compared to CA (Figure 8a). Breast cancer is a heterogeneous disease, and each cell line represents a different model, with different characteristics and association with different protein molecules and signaling pathways. The expression of integrin αv can vary among different metastatic and non-metastatic cells [25]. The upregulation of certain pathways in the cells upon delivery of αv integrin might be due to cross talks with other pathways. One such pathway which could interfere is TGF-β pathway. TGF-β signaling mutually influence each other and its cross-talks with other pathways result in successful induction of cancer progression [48].
Upregulated in invasive and distant metastasis, studies show ITGαv associates with various β sub-units and contributes towards breast cancer metastasis [25,30]. When 4T1 cells were induced in the in vivo model of female Balb/c mice for tumor, after first dose of treatment of CA complexed with ITGαv siRNA, reduced tumor volume trend was observed compared to CA treated and untreated groups (Figure 10) with highest fold decrease of 2.3 on Day 11 (Table 5). The tumor volume remained smaller compared to the control groups (Figure 12a). Statistical analysis performed between CA nanoparticle treated and siRNA-CA treated groups showed significant difference with p < 0.05 on selected time points (Figure 10). The CA loaded delivery of the integrin siRNA molecule increases its delivery efficiency to the target site. This is achieved by its ability to cause instability in the endosome, and release prior to its loss due to endosomal degradation, thus causing a sufficient decrease in the tumor volume [39]. The body weight remaining the same for both control and the treatment group indicates that there is no effect on the overall physiology of animals. One of the essential roles of ITGαv is during angiogenesis and progression in breast cancer. Besides frequently being overexpressed in neoplastic cell surfaces and involved in invasion of cancer cells and metastatic dissemination, ITGαv via its ligand association interacts with various cell proteases such as MMP2 which helps in enhancing the activity of tumor cells to degrade ECM and eventually migrate within the environment [49]. Several pathways are responsible for the migration of tumor cells. As we could see, transfection of this integrin siRNA caused reduction in protein expressions of phosphorylated AKT and MAPK as well as total MAPK (Figure 8c). This indicates that these pathways have a critical role in modulation of ITGαv expression. However, apart from AKT and MAPK, the TGF-β pathway also plays an essential role in integrin signaling and these pathways have been found to be co-related to each other. In 4T1 cells, research shows that TGF-β can induce EMT, cell migration, and phosphorylation of mothers against decapentaplegic homolog (Smad)-2 protein and transcriptional responses are dependent on the PI3K-AKT pathway [50]. Further, elevated endogenous levels of various MAPK proteins by this sub-unit have also been found responsible in increasing the tumor invasiveness [51]. Therefore, inhibiting these pathways with the help of antagonist such as ITGαv siRNA could modulate the morphogenic, transcriptional, and migratory activities of activated signaling pathways such as TGF-β, AKT, and MAPK in 4T1 models (in vitro and in vivo). While αv integrin can form heterodimers with other β integrin units as well as connect with additional cell cytoskeletal proteins [52], disrupting its function could possibly decrease the association with these molecules, which could further reduce the invasion of the tumor cells. Therefore, its knockdown could be vital in reducing the metastatic dissemination of the cancer in breasts and eventually reducing the cell invasion.
Integrin α6 (ITGα6), widely upregulated in tumors, participates in migration and invasion in cancer cells and as compared to normal cells, has a high level of protein and mRNA expressions [26,53], causing poor prognosis and survival. Transfection of CA-ITGα6 siRNA slightly decreased cell viability (~72%) in MCF-7 cells (Figure 2a) with cytotoxicity of ~11% (Table 2). ITGα6 siRNA transfection also showed the decrease expression of phosphorylated and total AKT and MAPK proteins in MCF-7 cells (Figure 8a). A study performed by Cariati et al. (2008) showed ITGα6 was necessary for MCF-7 tumorigenicity and overexpression of α6 has been found to provide growth and survival of tumor cells. Further, within this cell line, the sub population of MSS-derived cells attribute the stemness, which is stem cell characteristics and ITGα6 is one of its markers for identification. Therefore, by reducing the expression of this integrin via these proteins the invasive and migration properties of these cells could be reduced [54]. On the other hand, in MDA-MB-231 and 4T1 cells (Figure 2b,c respectively) no visible decrease in the viability was observed. As the level of α6 integrin is very high in TNBC and in metastatic breast cancers, the transfection at smaller dose may not be sufficient to carry out the reduction in cell viability in the respective MDA-MB-231 and 4T1 cell lines. Additionally, ITGα6is found in two transcript variants [26,53,54]. Hence, with one of the variants more expressed than the other, it may be possible that this pre-designed siRNA is not sufficient to reduce the activity of both the variants in the cells, leading to unaffected cell cytotoxicity. Reduction in levels of phosphorylated and total AKT and MAPK in MDA-MB-231 and in 4T1 cells (Figure 8b,c respectively) suggest MAPK and AKT pathways are critical in overregulated α6. They play an essential role in causing chemoresistance in breast cancer via α6 [26], and by controlling their expression by down regulating molecules such as ITGα6 they could prove beneficial for chemotherapeutics. However, at the same time targeting these pathways may not be enough to reduce the cell viability in certain high proliferative and metastatic in vitro cells. Cross talks might affect more downstream molecules of AKT and MAPK pathway, leading to increased viability, and involvement of other pathways and molecules such as growth factors, TGF-β, hedgehog, cell adhesion, and p63 might play critical role [55,56]. Studies show upon knockdown of ITGα6, reduction in the cell migration, invasion, and pulmonary metastasis can be observed. This can be seen in our in vivo model as well, where clear reduction in the tumor volume was observed post second dose of siRNA-CA treatment (Day 11 onwards) with maximum fold reduction of 2.65 on Day 17 (Table 5) and small tumors were observed at the end of the treatment (Figure 12b). As we can see from Figure 10, the trend for outgrowth of tumor over the time for ITGα6 siRNA complexed with CA nanoparticle was smaller compared to CA nanoparticle and untreated group. Statistical analysis performed between nanoparticle treated and siRNA-CA nanoparticle treated groups showed significant difference (*) with p < 0.05 on selected time points. Research shows apart from targeting main cell proliferation pathways such as PI3K/AKT or ERK/MAPK one of the ways the reduction of ITGα6 expression could take place is also by inhibiting cells to enter S phase. This is done by up-regulating p27, a cyclin protein, which results in down-regulation of cyclin E/CDK2 complexes [57].
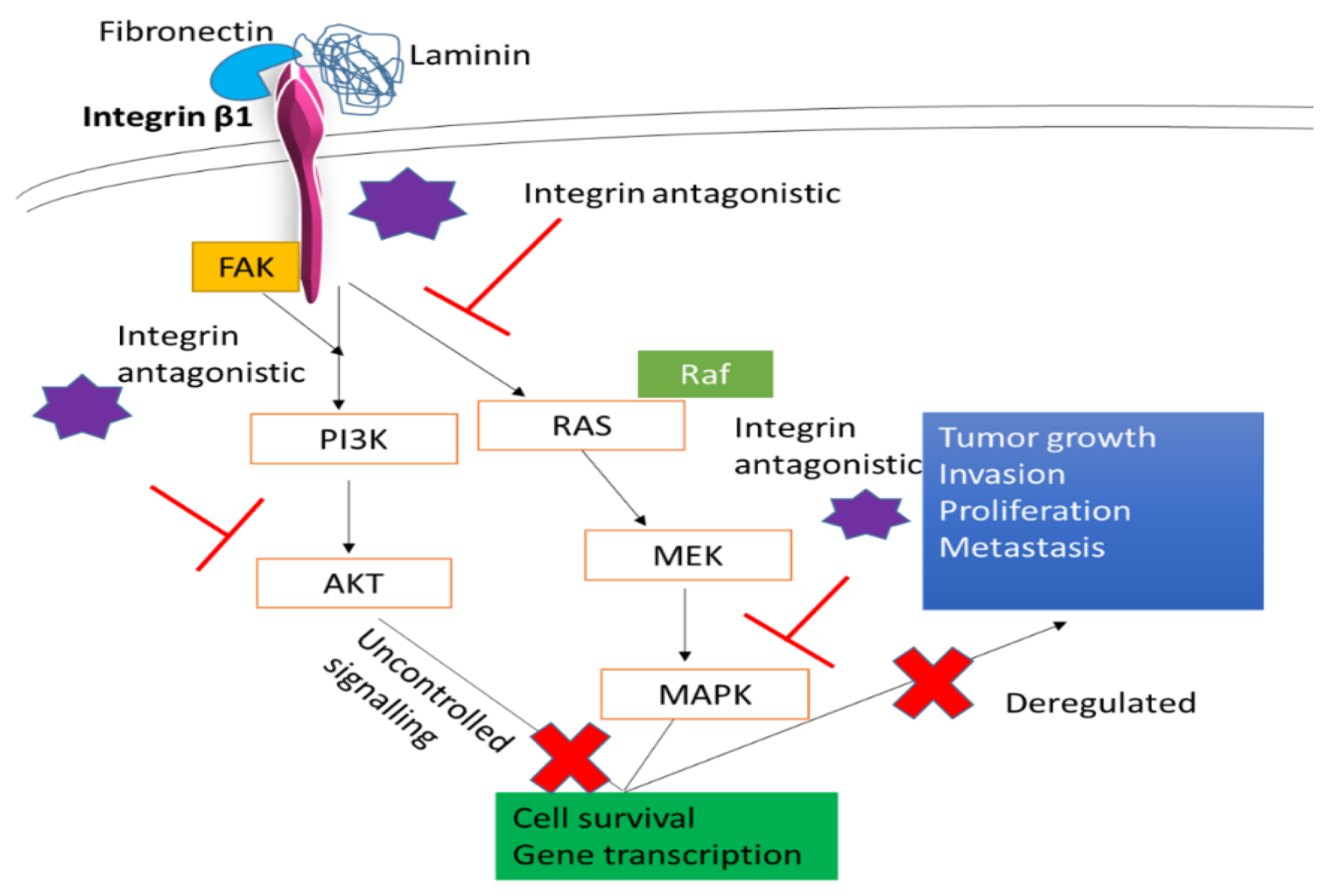
Integrin β1 (ITGβ1), the largest subgroup of integrin family, plays a vital role in breast cancer. Responsible for tumor proliferation, causing the switch of tumor cells from the dormant stage to metastatic stage, and providing resistance to tumor cells against adjuvant therapy and ionization radiation [58], targeting this component of integrin could be a promising strategy in reducing the tumor metastasis and increasing the efficiency in therapy. Based on our MTT results significant reduction in the cell viability after 48 h of transfection with this siRNA in all three cell lines was seen. While MCF-7 showed cell viability ~63% (Figure 3a) and cytotoxicity of ~32% (Table 2), MDA-MB-231 showed cell viability of ~86% (Figure 3b) and cytotoxicity ~10% (Table 3) and 4T1 showed cell viability of ~81% and cytotoxicity of ~12.5% (Table 4). The reduced expression of phosphorylated and total MAPK and AKT levels was observed in MCF-7, MDA-MB-231, and 4T1 cells (Figure 8a–c respectively). In animal model, tumor regression analysis also showed decrease in the tumor volume upon delivery of this integrin siRNA sub-unit along with CA nanoparticles (Figure 11). The significant difference was observed post first dose of treatment till the mice were sacrificed with highest fold decrease of 6.3 in tumor volume (Table 6) and smaller tumor compared to CA group (Figure 12c). From these results the essential role of this integrin subunit in various types and stages of breast cancer and its significant correlation with AKT and MAPK pathways can be inferred. Previous studies have shown knockdown of β1 in MDA-MB-231 cell line decreased the expression levels of β1-associated major α subunits and decreased the phosphorylation levels of FAK, thereby disrupting the focal adhesion and inhibiting mobility [59]. ITGβ1 is involved in bidirectional signaling where the domain interacts with cytoskeleton as well as the intracellular proteins in the cell. This helps ITGβ1 to contribute towards several tumor generating steps such as initiation, its survival, progression, reversion, and eventually metastasis. Various experimental studies, in vitro and in vivo, have shown, by targeting this sub-unit, its downstream signaling mediator is able to prevent the transition of the dormant tumor cells to function into metastatic cells and attenuate cell proliferation [58,60]. Further, by inhibiting its ability to cease apoptosis caused by various drugs such as paclitaxel could aid in reducing the chemoresistance via deactivation of AKT and MAPK pathways and effector molecules such as B-cell lymphoma-2 (Bcl-2) [45]. In Figure 13 we have elucidated how this integrin sub-unit could involve in signaling pathways, and introduction of an antagonistic could help in hampering the pathways at various sites.
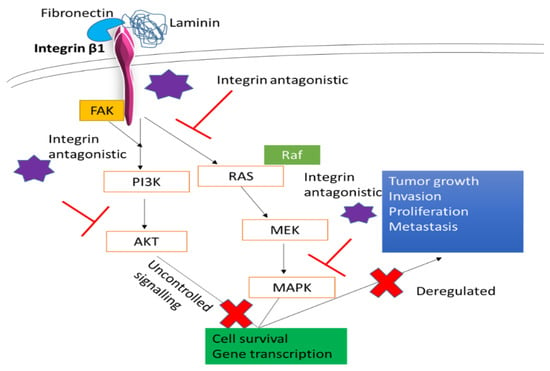
Figure 13.
A schematic representation of targeting ITGβ1 in the cells, and possible ways of hampering essential tumorigenic pathways.
Integrin β3 (ITGβ3) a critical sub-unit which plays role in tumor invasion, neoangiogenesis, and inflammation, makes it one of the promising cancer targets. Tumor cells which show high expression of ITGβ3 exhibit enhancement in their proliferation and metastasis, especially to bones [61,62]. The role of this integrin in breast cancer has been studied in various experiments, in vitro as well as in vivo, suggesting its knockdown or deletion can play significant role in decreasing the metastasis. In our MTT assay, delivery of β3 siRNA loaded CA reduced the cell viability to ~60% in MCF-7 (Figure 4a), with cytotoxicity of ~14% (Table 2), while in MDA-MB-231 and 4T1 cell lines no reduction in cell viability was seen (Figure 4b,c respectively). However, in vivo model showed contrary results compared to cell line. Significant tumor reduction was observed from Day 10 (Figure 11), with highest of 4.9-fold decrease on Day 12 compared to CA control (Table 6) and reduced tumor size at the end of tumor excision (Figure 12d). Protein expression study showed the transfection of CA loaded β3 integrin siRNA caused reduction of total and phosphorylated MAPK and AKT proteins in MCF-7, MDA-MB-23, and 4T1 cells (Figure 8a–c respectively). One reason for high cell viability despite reduced AKT and MAPK pathways may be due to cross talks with signaling molecules such as that of signal transducer and activator of transcription (STAT)-6/STAT1. These cross talks could have resulted in controlling the balance between anti-tumor and pro-tumor immune cells [61] in the in vitro model. In several biological contexts, the locus of ITGB3 is regulated epigenetically, which in turn is responsible for regulating its function and expression. This regulation is seen to control tumor senescence as well. For example, increased levels of endogenous β3 via transcriptional regulation of polycomb protein chromobox 7 (CBX7) is observed during oncogene-induced senescence (OIS) with concomitant increase in β3 mRNA expression [62]. Therefore, by hampering expression of this integrin along with other transcriptional proteins via an efficient vehicle may provide improved efficacy in tumor regulation.
Integrin β4 (ITGβ4) overexpression, chemoresistance to drugs such as tamoxifien, and its related aggressiveness has been found especially in basal-like breast cancers. Cell migration and invasion takes place with the aid of actin binding proteins, which help in stabilization of actin protrusions [31]. Targeting this subtype of integrin has shown reduction in tumors in aggressive cancers such as TNBC. Based on our results, ITGβ4 siRNA-CA delivery did not caused visible reduction in cell viability in MCF-7 and 4T1 cells (Figure 5a,c respectively), while in MDA-MB-231, cell viability of ~88% (Figure 5b), with cytotoxicity of ~11% (Table 3) was observed. Presence of integrin β4 has been observed in TNBC patients. Inter and intra tumoral heterogeneity and presence of cancer stem like cells (CSC) being critical feature of this breast cancer [63]. Therefore, reducing its expression may help in reduction of these stem-like cells and result in lesser tumor heterogeneity. ITGβ4 binds directly to laminin protein which in turn activates PI3K/AKT pathways and promotes proliferation, invasion, and survival of cells [64]. Protein expression study by Western blot technique showed total and phosphorylated AKT and MAPK expression was reduced upon β4 integrin siRNA transfection, in all the three cell lines (Figure 8a–c). Reduction in the expression of this integrin reportedly reduces the lung metastasis in aggressive breast cancers [64]. Reduction in protein expression without decrease in cell viability indicates that, in cells such as MCF-7 and 4T1, other pathways are actively involved in causing the upregulation of this gene. For example, ERα signaling plays an essential role in upregulating ITGβ4 signaling. Research done by Yi Ho et al. (2016) showed estrogen enhances the viability of breast cancer cells by activating ERα-∆Np63-integrin β4 signaling. Additionally, cross talk with various growth factors, and activation of Ras homolog family member A (RhoA)/Rho-associated protein kinase 2 (ROCK-2) signaling cascades could enhance the cell motility and viability in vitro [64]. Even though no reduction in cell viability was observed in 4T1 cells, the tumor regression model showed significant decrease (p < 0.05) in the tumor volume compared to CA control (Figure 11) and smaller tumor at the end of the treatment (Figure 12e). This indicates targeting this integrin and reduced signaling of vital cell proliferation pathways such as AKT and MAPK pathways play vital role in regulating β4 expression and its tumor induced modelling. While various cross talks are involved between the signaling molecules and several upstream and downstream molecules are targeted, one of the ways by which integrins cooperate with AKT and ERK is by suppressing cyclin-dependent kinase inhibitor p21/cyclin dependent kinase inhibitor p27 jointly, which causes shift in the balance of forkhead box O3 (Foxo3a), an essential transcriptional activator for apoptosis, from the nucleus to cytosol [65]. Similarly, it plays role in hampering of pathways involved with cell cycle, thereby causing a significant reduction in tumors.
Integrin β5 (ITGβ5), when highly expressed, participates in tumor growth and angiogenesis. After delivering ITGβ5 siRNA along with CA nanoparticles and incubating the cells for 48 h, MTT assay revealed a decrease in cell viability of ~63% in MCF-7 (Figure 6a) with ~21% cytotoxicity (Table 2), ~84% in MDA-MB-231 (Figure 6b) with slight cytotoxicity of ~8% (Table 3), and cell viability of ~84% in 4T1 cells (Figure 6c) with cytotoxicity of ~7% (Table 4). Protein expression was reduced for both phosphorylated and total AKT and MAPK in MCF-7 and 4T1 cells (Figure 8a,c respectively), while in MDA-MB-231 no knockdown was seen (Figure 8b). Variation in the protein expressions may be due to different phenotypic and genotypic characteristics of cells, and while PI3K-ERK serves as one of the pathways which contribute to signaling via this integrin, several other pathways such as Src-FAK, MEK may be involved in regulating the cell viability in MDA-MB-231 cells [28]. At the end of the study, tumor regression showed significant (p < 0.05) reduction in tumor volume compared to CA control (Figure 11), with smaller tumor in comparison with CA control (Figure 12f), indicating successful delivery of target antagonist via CA delivery vehicle. Recent studies have shown integrin β5 contributes to TGF-β-induced EMT and resistance to radiotherapy and chemotherapy in various stages of breast cancer [28]. Therefore, by sequestering this molecule, besides targeting AKT and MAPK oncogenic proliferation pathways, vital EMT pathways could also be affected. Hence, this integrin sub-unit might serve as an efficient therapeutic target in the breast cancers which are resistant to most of the therapies.
Integrin β6 (ITGβ6) gene shows its expression in several carcinoma cell lines, including breast cancer [66,67]. All three cell lines showed reduced cell viability of ~67% (Figure 7a), ~63% (Figure 7b), and ~60% (Figure 7c). High cytotoxicity of ~16% in MCF-7 (Table 2), ~29% in MDA-MB-231 (Table 3) and ~33% in 4T1 cells (Table 4), upon transfection with β6 siRNA loaded CA nanoparticles was observed. At protein level, expression of phosphorylated and total AKT and MAPK was reduced in MCF-7 (Figure 8a). In MDA-MB-231 cells, total MAPK showed no reduction (Figure 8b) compared to CA treated control, while in 4T1 cell line undetectable reduction was seen in p-AKT (Figure 8c). In vivo model showed reduction in tumor volume with significant difference (p < 0.05) at certain time points post treatment doses (Figure 11) and smaller tumor post excision compared to CA control (Figure 12g). With only 50-fold more siRNA (roughly equivalent to 3.325 μg/kg in a 20 g mouse) compared to that used in each well of a 24 well-plate, the anti-tumor effect was remarkably high, which could be due to the efficient tumor targeting ability of siRNA-loaded CA nanoparticles and, finally, intracellular release of the therapeutic siRNA following endocytosis and pH-responsive dissolution of the particles. The expression of p-AKT may have contributed towards significant reduction only at certain days. Mechanism of this integrin is mainly regulated at transcription level. Studies report various transcription factors such as ETS Proto-Oncogene 1 (Ets-1), Signal transducer, and activator of transcription 3 (STAT3) (involved in basal epithelial cells), and Smad3 mediate the initiation of ITGβ6 expression. While the exact mechanism underlying integrin β6 regulation is unknown, it serves as a prognostic indicator in various invasive breast cancers, especially HER2+ subtype [67,68].
The contrasting in vitro and in vivo results could be due to varied microenvironments in both systems and different sensitivity to siRNAs in cells compared to animal model. While in vivo cells are biologically embedded in the ECM, which in itself is a complex three-dimensional gel, providing mechanical support, directing cellular behavior, and mimicking the physiology and pathology of human systems, in vitro support system provides a two-dimensional environment which not only provides a less suitable complex system for cell-substrate interaction but also results in certain limitations such as dissimilarities in cellular adhesion, migration, and cytoskeletal organization [69]. Further, slight, or undetectable change in the expression of proteins in certain studies could indicate that the proteins were already expressed before the knockdown by siRNA treatment. In addition, integrin signaling is complex and, besides MAPK and AKT, there are several pathways and molecules, which play distinct roles in each type of integrin regulation. Hence, in treatments where knockdown in these pathways is observed, without cell viability being affected, it may be the other pathways which enhance the proliferation.
6. Conclusions
Using CA as a delivery vehicle, the siRNA/gene degradation can be reduced and delivery efficiency to the target site could be enhanced. Delivery of single integrins (ITGαv, ITGα6, ITGβ1, ITGβ3, ITGβ4, ITGβ5, and ITGβ6) siRNAs loaded with CA-NP in vitro and in vivo showed promising effect in causing the reduction in cell number and in reducing the tumor volume significantly. Further, the siRNA-CA transfection was able to reduce band intensities for phosphorylated and total AKT and MAPK in MCF-7, MDA-MB-231, and 4T1 cell lines. Thus, targeting dysregulated integrin genes, regulating their expression, and abrogating their dysregulated pathways via delivery of antagonistic molecules such as siRNAs with the aid of suitable inorganic CA nanocarrier could provide a potential source of developing improvised therapeutics for breast cancer with promising effect in reducing the proliferation, invasion, and metastasis of breast cancer.
Author Contributions
M.A. carried out experiments and prepared the first draft and E.H.C. designed and supervised the project. All authors have read and agreed to the published version of the manuscript.
Funding
This project was financially supported by a grant from Ministry of Higher Education, Malaysia (MoHE) (FRGS/1/2018/STG05/MUSM/02/3).
Institutional Review Board Statement
All the in vivo work was done under Monash University Animal Ethics Committee approval (MARP/2016/126).
Informed Consent Statement
Not applicable.
Data Availability Statement
All animal work was done according to the procedure and guideline approved by Monash University Animal Ethics Committee (MARP/2016/126).
Conflicts of Interest
The authors declare no conflict of interest.
References
- WHO. Breast Cancer. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer (accessed on 25 October 2021).
- National Cancer Institute. Cancer Stat Facts: Female Breast Cancer. Available online: https://seer.cancer.gov/statfacts/html/breast.html (accessed on 25 October 2021).
- Selvi, R. Breast Diseases: Imaging and Clinical Management; Springer: Pune, India, 2014. [Google Scholar]
- Irvin, W.J., Jr.; Carey, L.A. What is triple-negative breast cancer? Eur. J. Cancer 2008, 44, 2799–2805. [Google Scholar] [CrossRef]
- Anders, C.; Carey, L.A. Understanding and treating triple-negative breast cancer. Oncology 2008, 22, 1233–1239. [Google Scholar] [PubMed]
- Yilmaz, M.; Christofori, G. EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev. 2009, 28, 15–33. [Google Scholar] [CrossRef] [Green Version]
- Jin, X.; Mu, P. Targeting breast cancer metastasis. Breast Cancer Basic Clin. Res. 2015, 9 (Suppl. 1), 23–34. [Google Scholar] [CrossRef] [Green Version]
- Parekh, A.; Weaver, A.M. Regulation of cancer invasiveness by the physical extracellular matrix environment. Cell Adhes. Migr. 2009, 3, 288–292. [Google Scholar] [CrossRef] [Green Version]
- Bendas, G.; Borsig, L. Cancer cell adhesion and metastasis: Selectins, integrins, and the inhibitory potential of heparins. Int. J. Cell Biol. 2012, 676731. [Google Scholar] [CrossRef]
- Golias, C.H.; Charalabopoulos, A.; Peschos, D.; Maritsi, D.; Charalabopoulos, K.; Batistatou, A. Adhesion molecules in cancer invasion and metastasis. Hippokratia 2005, 9, 106–114. [Google Scholar]
- Okegawa, T.; Pong, R.C.; Li, Y.; Hsieh, J.T. The role of cell adhesion molecule in cancer progression and its application in cancer therapy. Acta Biochim. Pol. -Engl. Ed. 2004, 51, 445–458. [Google Scholar] [CrossRef] [Green Version]
- Glukhova, M.A.; Streuli, C.H. How integrins control breast biology. Curr. Opin. Cell Biol. 2013, 25, 633–641. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Kåresen, R.; Axcrona, U.; Noren, T.; Sauer, T. Expression pattern of adhesion molecules (E-cadherin, α-, β-, γ-catenin and claudin-7), their influence on survival in primary breast carcinoma, and their corresponding axillary lymph node metastasis. Apmis 2007, 115, 52–65. [Google Scholar] [CrossRef]
- Hsu, K.S.; Kao, H.Y. Alpha-actinin 4 and tumorigenesis of breast cancer. Vitam. Horm. 2013, 93, 323–351. [Google Scholar] [PubMed] [Green Version]
- Berx, G.; Van, R.F. The E-cadherin/catenin complex: An important gatekeeper in breast cancer tumorigenesis and malignant progression. Breast Cancer Res. 2001, 3, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Hood, J.D.; Cheresh, D.A. Role of integrins in cell invasion and migration. Nat. Rev. Cancer 2002, 2, 91–100. [Google Scholar] [CrossRef]
- Sosnoski, D.; Emanuel, B.S.; Hawkins, A.L.; Van Tuinen, P.; Ledbetter, D.H.; Nussbaum, R.L.; Kaos, F.T.; Schwartz, E.; Phillips, D.; Bennett, J.S. Chromosomal localization of the genes for the vitronectin and fibronectin receptors alpha subunits and for platelet glycoproteins IIb and IIIa. J. Clin. Investig. 1988, 81, 1993–1998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berman, A.; Kozlova, N. Integrins: Structure and functions. Membr. Cell Biol. 2000, 13, 207–244. [Google Scholar] [PubMed]
- Qin, J.; Vinogradova, O.; Plow, E.F. Integrin bidirectional signaling: A molecular view. PLoS Biol. 2004, 2, e169. [Google Scholar] [CrossRef] [Green Version]
- Springer, T.A.; Wang, J.-H. The three-dimensional structure of integrins and their ligands, and conformational regulation of cell adhesion. Adv. Protein Chem. 2004, 68, 29–63. [Google Scholar]
- White, D.E.; Muller, W.J. Multifaceted roles of integrins in breast cancer metastasis. J. Mammary Gland. Biol. Neoplasia 2007, 12, 135–142. [Google Scholar] [CrossRef]
- Weis, S.M.; Cheresh, D.A. αV integrins in angiogenesis and cancer. Cold Spring Harb. Perspect. Med. 2011, 1, a006478. [Google Scholar] [CrossRef] [Green Version]
- Donahue, J.P.; Sugg, N.; Hawiger, J. The integrin αv gene: Identification and characterization of the promoter region. Biochim. Biophys. Acta (BBA) 1994, 1219, 228–232. [Google Scholar] [CrossRef]
- Desgrosellier, J.S.; Barnes, L.A.; Shields, D.J.; Huang, M.; Lau, S.K.; Prévost, N.; Tarin, D.; Shattil, S.J.; Cheresh, D.A. An integrin α v β 3–c-Src oncogenic unit promotes anchorage-independence and tumor progression. Nat. Med. 2009, 15, 1163–1169. [Google Scholar] [CrossRef] [Green Version]
- Taherian, A.; Li, X.; Liu, Y.; Haas, T.A. Differences in integrin expression and signaling within human breast cancer cells. BMC Cancer 2011, 11, 293. [Google Scholar] [CrossRef] [Green Version]
- Hu, T.; Zhou, R.; Zhao, Y.; Wu, G. Integrin alpha6/Akt/Erk signaling is essential for human breast cancer resistance to radiotherapy. Sci. Rep. 2016, 6, 33376. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.; Mercurio, A.M. Contributions of the α6 Integrins to Breast Carcinoma Survival and Progression. Mol. Cells 2004, 17, 203–209. [Google Scholar] [PubMed]
- Bianchi-Smiraglia, A.; Paesante, S.; Bakin, A.V. Integrin beta5 contributes to the tumorigenic potential of breast cancer cells through the Src-FAK and MEK-ERK signaling pathways. Oncogene 2013, 32, 3049–3058. [Google Scholar] [CrossRef] [Green Version]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Felding-Habermann, B.; O’Toole, T.E.; Smith, J.W.; Fransvea, E.; Ruggeri, Z.M.; Ginsberg, M.H.; Hughes, P.E.; Pampori, N.; Shattil, S.J.; Saven, A.; et al. Integrin activation controls metastasis in human breast cancer. Proc. Natl. Acad. Sci. USA 2001, 98, 1853–1858. [Google Scholar] [CrossRef] [Green Version]
- Nisticò, P.; Di Modugno, F.; Spada, S.; Bissell, M.J. β1 and β4 integrins: From breast development to clinical practice. Breast Cancer Res. 2014, 16, 459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schittenhelm, J.; Klein, A.; Tatagiba, M.S.; Meyermann, R.; Fend, F.; Goodman, S.L.; Sipos, B. Comparing the expression of integrins αvβ3, αvβ5, αvβ6, αvβ8, fibronectin and fibrinogen in human brain metastases and their corresponding primary tumors. Int. J. Clin. Exp. Pathol. 2013, 6, 2719–2732. [Google Scholar]
- dos Santos, P.B.; Zanetti, J.S.; Ribeiro-Silva, A.; Beltrão, E.I. Beta 1 integrin predicts survival in breast cancer: A clinicopathological and immunohistochemical study. Diagn. Pathol. 2012, 7, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Behlke, M.A. Progress towards in vivo use of siRNAs. Mol. Ther. 2006, 13, 644–670. [Google Scholar] [CrossRef]
- Cao, Q.; Cai, W.; Li, T.; Yang, Y.; Chen, K.; Xing, L.; Chen, X. Combination of integrin siRNA and irradiation for breast cancer therapy. Biochem. Biophys. Res. Commun. 2006, 351, 726–732. [Google Scholar] [CrossRef]
- Xu, C.-F.; Wang, J. Delivery systems for siRNA drug development in cancer therapy. Asian J. Pharm. Sci. 2015, 10, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, E. pH-sensitive nano-crystals of carbonate apatite for smart and cell-specific transgene delivery. Expert Opin. Drug Deliv. 2007, 4, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, E.; Akaike, T. High performance DNA nano-carriers of carbonate apatite: Multiple factors in regulation of particle synthesis and transfection efficiency. Int. J. Nanomed. 2007, 2, 101–106. [Google Scholar] [CrossRef]
- Chowdhury, E.H. pH-Sensitive Nanocrystals of Carbonate Apatite-a Powerful and Versatile Tool for Efficient Delivery of Genetic Materials to Mammalian Cells. In Advances in Biomaterials Science and Biomedical Applications; InTechOpen: London, UK, 2013. [Google Scholar]
- Hossain, S.; Stanislaus, A.; Chua, M.J.; Tada, S.; Tagawa, Y.I.; Chowdhury, E.H.; Akaike, T. Carbonate apatite-facilitated intracellularly delivered siRNA for efficient knockdown of functional genes. J. Control. Release 2010, 147, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Mozar, F.S.; Chowdhury, E.H. Surface-modification of carbonate apatite nanoparticles enhances delivery and cytotoxicity of gemcitabine and anastrozole in breast cancer cells. Pharmaceutics 2017, 9, 21. [Google Scholar] [CrossRef] [Green Version]
- Tiash, S.; Othman, I.; Rosli, R.; Hoque Chowdhury, E. Methotrexate- and cyclophosphamide-embedded pure and strontiumsubstituted carbonate apatite nanoparticles for augmentation of chemotherapeutic activities in breast cancer cells. Curr. Drug Deliv. 2014, 11, 214–222. [Google Scholar] [CrossRef]
- Kunnath, A.P.; Tiash, S.; Fatemian, T.; Morshed, M.; Mohamed, S.M.; Chowdhury, E.H. Intracellular delivery of ERBB2 siRNA and p53 gene synergistically inhibits the growth of established tumour in an immunocompetent mouse. J. Cancer Sci. Ther. 2014, 6, 99–104. [Google Scholar] [CrossRef]
- Shaw, L.M. Integrin function in breast carcinoma progression. J. Mammary Gland. Biol. Neoplasia 1999, 4, 367–376. [Google Scholar] [CrossRef]
- Aoudjit, F.; Vuori, K. Integrin signaling in cancer cell survival and chemoresistance. Chemother. Res. Pract. 2012, 2012, 283181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eliceiri, B.P.; Cheresh, D.A. Role of alpha v integrins during angiogenesis. Cancer J. 2000, 6, S245-9. [Google Scholar]
- Koistinen, P.; Heino, J. Integrins in Cancer Cell Invasion; Landes Bioscience: Austin, TX, USA, 2013. [Google Scholar]
- Mamuya, F.A.; Duncan, M.K. aV integrins and TGF-β-induced EMT: A circle of regulation. J. Cell. Mol. Med. 2012, 16, 445–455. [Google Scholar] [CrossRef] [PubMed]
- Nejjari, M.; Hafdi, Z.; Gouysse, G.; Fiorentino, M.; Béatrix, O.; Dumortier, J.; Pourreyron, C.; Barozzi, C.; D’errico, A.; Grigioni, W.F.; et al. Expression, regulation, and function of αV integrins in hepatocellular carcinoma: An in vivo and in vitro study. Hepatology 2002, 36, 418–426. [Google Scholar] [CrossRef]
- Bakin, A.V.; Tomlinson, A.K.; Bhowmick, N.A.; Moses, H.L.; Arteaga, C.L. Phosphatidylinositol 3-kinase function is required for transforming growth factor beta-mediated epithelial to mesenchymal transition and cell migration. J. Biol. Chem. 2000, 275, 36803–36810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Tomlinson, A.K.; Bhowmick, N.A.; Moses, H.L.; Arteaga, C.L. αv integrin, p38 mitogen-activated protein kinase, and urokinase plasminogen activator are functionally linked in invasive breast cancer cells. J. Biol. Chem. 2001, 276, 47901–47905. [Google Scholar] [CrossRef] [Green Version]
- Weber, G.F.; Bjerke, M.A.; DeSimone, D.W. Integrins and cadherins join forces to form adhesive networks. J. Cell Sci. 2011, 124, 1183–1193. [Google Scholar] [CrossRef] [Green Version]
- Ms, L.; Shauntell, N. The Role of Integrin Alpha 6 on Tumor Metastasis. 2016. Available online: https://digitalcommons.georgiasouthern.edu/honors-theses/209/ (accessed on 25 July 2021).
- Cariati, M.; Naderi, A.; Brown, J.P.; Smalley, M.J.; Pinder, S.E.; Caldas, C.; Purushotham, A.D. Alpha-6 integrin is necessary for the tumourigenicity of a stem cell-like subpopulation within the MCF7 breast cancer cell line. Int. J. Cancer 2008, 122, 298–304. [Google Scholar] [CrossRef]
- Shimizu, H.; Koyama, N.; Asada, M.; Yoshimatsu, K. Aberrant expression of integrin and erbB subunits in breast cancer cell lines. Int. J. Oncol. 2002, 21, 1073–1079. [Google Scholar] [CrossRef]
- Hu, M.; Yao, J.; Carroll, D.K.; Weremowicz, S.; Chen, H.; Carrasco, D.; Richardson, A.; Violette, S.; Nikolskaya, T.; Nikolsky, Y.; et al. Regulation of in situ to invasive breast carcinoma transition. Cancer Cell 2008, 13, 394–406. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Shenouda, S.; Baranwal, S.; Rathinam, R.; Jain, P.; Bao, L.; Hazari, S.; Dash, S.; Alahari, S.K. Integrin subunits alpha5 and alpha6 regulate cell cycle by modulating the chk1 and Rb/E2F pathways to affect breast cancer metastasis. Mol. Cancer 2011, 10, 84. [Google Scholar] [CrossRef] [Green Version]
- Barkan, D.; Green, J.E.; Chambers, A.F. Extracellular matrix: A gatekeeper in the transition from dormancy to metastatic growth. Eur. J. Cancer 2010, 46, 1181–1188. [Google Scholar] [CrossRef] [Green Version]
- Hou, S.; Isaji, T.; Hang, Q.; Im, S.; Fukuda, T.; Gu, J. Distinct effects of β1 integrin on cell proliferation and cellular signaling in MDA-MB-231 breast cancer cells. Sci. Rep. 2016, 6, 18430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zawistowski, J.S.; Nakamura, K.; Parker, J.S.; Granger, D.A.; Golitz, B.T.; Johnson, G.L. miR-9-3p targets integrin beta 1 to sensitize claudin-low breast cancer cells to MEK inhibition. Mol. Cell. Biol. 2013, 33, 2260–2274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, X.; Nakamura, K.; Parker, J.S.; Granger, D.A.; Golitz, B.T.; Johnson, G.L. Antagonizing integrin β3 increases immunosuppression in cancer. Cancer Res. 2016, 76, 3484–3495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rapisarda, V.; Rapisarda, V.; Borghesan, M.; Miguela, V.; Encheva, V.; Snijders, A.P.; Lujambio, A.; O’Loghlen, A. Integrin Beta 3 Regulates Cellular Senescence by Activating the TGF-β Pathway. Cell Rep. 2017, 18, 2480–2493. [Google Scholar] [CrossRef] [Green Version]
- Bierie, B.; Pierce, S.E.; Kroeger, C.; Stover, D.G.; Pattabiraman, D.R.; Thiru, P.; Donaher, J.L.; Reinhardt, F.; Chaffer, C.L.; Keckesova, Z.; et al. Integrin-β4 identifies cancer stem cell-enriched populations of partially mesenchymal carcinoma cells. Proc. Natl. Acad. Sci. USA 2017, 114, E2337–E2346. [Google Scholar] [CrossRef] [Green Version]
- Ho, J.-Y.; Pierce, S.E.; Kroeger, C.; Stover, D.G.; Pattabiraman, D.R.; Thiru, P.; Donaher, J.L.; Reinhardt, F.; Chaffer, C.L.; Keckesova, Z.; et al. Estrogen enhances the cell viability and motility of breast cancer cells through the ERα-ΔNp63-integrin β4 signaling pathway. PLoS ONE 2016, 11, e0148301. [Google Scholar] [CrossRef]
- Moreno-Layseca, P.; Streuli, C.H. Signalling pathways linking integrins with cell cycle progression. Matrix Biol. 2014, 34, 144–153. [Google Scholar] [CrossRef]
- Breuss, J.M.; Gallo, J.; DeLisser, H.M.; Klimanskaya, I.V.; Folkesson, H.G.; Pittet, J.F.; Nishimura, S.L.; Aldape, K.; Landers, D.V.; Carpenter, W. Expression of the beta 6 integrin subunit in development, neoplasia and tissue repair suggests a role in epithelial remodeling. J. Cell Sci. 1995, 108, 2241–2251. [Google Scholar] [CrossRef]
- Xu, M.; Chen, X.; Yin, H.; Yin, L.; Liu, F.; Fu, Y.; Yao, J.; Deng, X. Cloning and characterization of the human integrin β6 gene promoter. PLoS ONE 2015, 10, e0121439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desai, K.; Nair, M.G.; Prabhu, J.S.; Vinod, A.; Korlimarla, A.; Rajarajan, S.; Aiyappa, R.; Kaluve, R.S.; Alexander, A.; Hari, P.S.; et al. High expression of integrin β6 in association with the Rho–Rac pathway identifies a poor prognostic subgroup within HER 2 amplified breast cancers. Cancer Med. 2016, 5, 2000–2011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavo, M.; Fato, M.; Peñuela, L.; Beltrame, F.; Raiteri, R.; Scaglione, S. Microenvironment complexity and matrix stiffness regulate breast cancer cell activity in a 3D in vitro model. Sci. Rep. 2016, 6, 35367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).