Mathematical Modeling for the Growth of Salmonella spp. and Staphylococcus aureus in Cake at Fluctuating Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Analysis of Water Activity (aw) and Proximate Composition
2.2. Inocula Preparation
2.3. Comparisons of Salmonella spp. and S. aureus Growth in Various Cake Samples
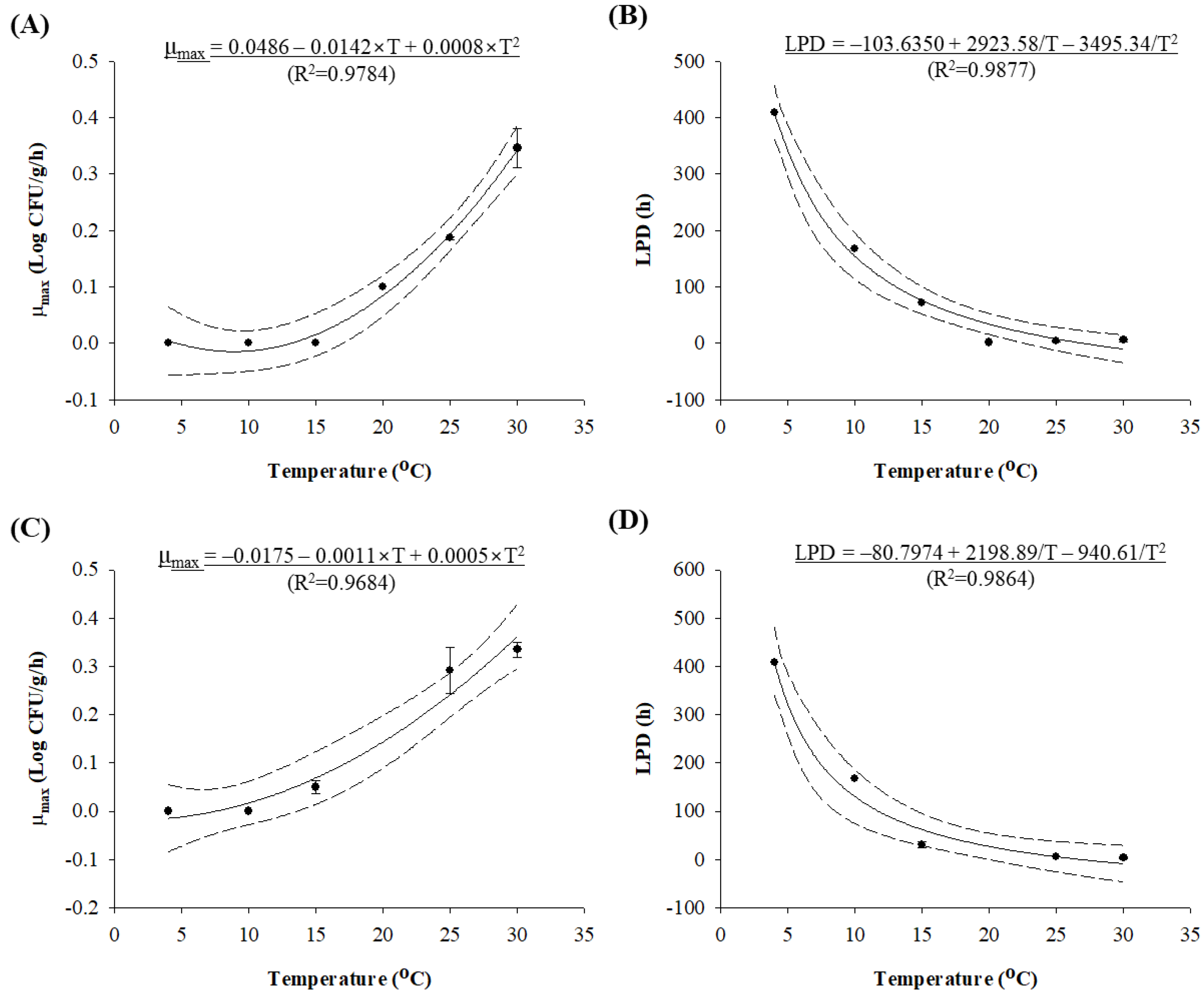
2.4. Development of Predictive Models for Isothermal Temperature
2.5. Development of the Dynamic Model
2.6. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Canadian Food Inspection Agency. Bacterial Pathogens in Pre-Prepared, Refrigerated Baked Desserts. 1 April 2016 to 31 March 2018. Food Microbiology-Targeted Surveys-Final Report. Available online: https://tinyurl.com/6h8cs3u (accessed on 2 March 2021).
- Ladd-Wilson, S.G.; Morey, K.; Koske, S.E.; Burkhalter, B.; Bottichio, L.; Brandenburg, J.; Fontana, J.; Tenney, K.; Kutumbaka, K.K.; Samadpour, M.; et al. Notes from the Field: Multistate Outbreak of Salmonella Agbeni Associated with Consumption of Raw Cake Mix—Five States, 2018. MMWR. Morb. Mortal. Wkly. Rep. 2019, 68, 751–752. [Google Scholar] [CrossRef] [PubMed]
- Eun, Y.; Jeong, H.; Kim, S.; Park, W.; Ahn, B.; Kim, D.; Kim, E.; Park, E.; Hwang, I.; Son, H.; et al. A large outbreak of Salmonella enterica serovar Thompson infections associated with chocolate cake in Busan, Korea. Epidemiol. Health 2019, 41, e2019002. [Google Scholar] [CrossRef] [PubMed]
- Solhan, S.; Chan, P.P.; Kurupatham, L.; Foong, B.H.; Ooi, P.L.; James, L.; Phua, L.; Tan, A.L.; Koh, D.; Goh, K.T. An outbreak of Salmonella enterica serotype Enteritidis traced to cream cakes. West. Pac. Surveil. Response J. 2011, 2, 23–30. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Outbreak of Salmonella Infections. Available online: https://tinyurl.com/y5ba37b8 (accessed on 2 March 2021).
- NSW Food Authority. Microbiological Quality of High Risk Bakery Products. A Survey to Determine the Microbiological Quality of Bakery Products Sold in NSW. Available online: https://tinyurl.com/5d2u8tru (accessed on 2 March 2021).
- Ercoli, L.; Gallina, S.; Nia, Y.; Auvray, F.; Primavilla, S.; Guidi, F.; Pierucci, B.; Graziotti, C.; Decastelli, L.; Scuota, S. Investi-gation of a Staphylococcal Food Poisoning Outbreak from a Chantilly Cream Dessert, in Umbria (Italy). Foodborne Pathg. Dis. 2017, 14, 407–413. [Google Scholar] [CrossRef] [PubMed]
- FSN (Food Safety News). Staph Found in Illinois Bakery Tied to Outbreak. Available online: https://tinyurl.com/rjw9a4mw (accessed on 2 March 2021).
- Lee, H.; Yoon, Y. Etiological Agents Implicated in Foodborne Illness World Wide. Food Sci. Anim. Resour. 2021, 41, 1–7. [Google Scholar] [CrossRef] [PubMed]
- EFSA (European Food Safety Authority). Salmonella the Most Common Cause of Foodborne Outbreaks in the European Union. Available online: https://tinyurl.com/34ujs4u3 (accessed on 2 March 2020).
- CDC (Center for Disease Control and Prevention). Salmonella and Food. Available online: https://tinyurl.com/36cct7yt (accessed on 2 March 2020).
- CDC (Center for Disease Control and Prevention). Staphylococcal (Staph) Food Poisoning. Available online: https://tinyurl.com/5fraxfxw (accessed on 2 March 2020).
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Riet, K.V. T Modeling of the Bacterial Growth Curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, H.; Lee, J.-Y.; Skandamis, P.; Park, B.-Y.; Oh, M.-H.; Yoon, Y. Mathematical Models To Predict Kinetic Behavior and Growth Probabilities of Listeria monocytogenes on Pork Skin at Constant and Dynamic Temperatures. J. Food Prot. 2013, 76, 1868–1872. [Google Scholar] [CrossRef] [PubMed]
- Dalgaard, P.; Mejlholm, O. Modeling Growth of Listeria and Lactic Acid Bacteria in Food Environments. Methods Mol. Biol. 2018, 247–264. [Google Scholar] [CrossRef]
- Ross, T.; McMeekin, T.A. Modeling Microbial Growth Within Food Safety Risk Assessments. Risk Anal. 2003, 23, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Kapetanakou, A.E.; Makariti, I.P.; Nazou, E.N.; Manios, S.G.; Karavasilis, K.; Skandamis, P.N. Modelling the effect of osmotic adaptation and temperature on the non–thermal inactivation of Salmonella spp. on brioche-type products. Int. J. Food Microbiol. 2019, 296, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Panagou, E.Z.; Nychas, G.J. Dynamic modeling of Listeria monocytogenes growth in pasteurized vanilla cream after postpro-cessing contamination. J. Food Prot. 2008, 71, 456–457. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of AOAC International, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Baranyi, J.; Roberts, T.A. A dynamic approach to predicting bacterial growth in food. Int. J. Food Microbiol. 1994, 23, 277–294. [Google Scholar] [CrossRef]
- Baranyi, J.; Ross, T.; McMeekin, T.; Roberts, T. Effects of parameterization on the performance of empirical models used in ‘predictive microbiology’. Food Microbiol. 1996, 13, 83–91. [Google Scholar] [CrossRef]
- Lathrop, A.A.; Taylor, T.; Schnepf, J. Survival of Salmonella during Baking of Peanut Butter Cookies. J. Food Prot. 2014, 77, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Ross, T. Indices for performance evaluation of predictive models in food microbiology. J. Appl. Bacteriol. 1996, 81, 501–508. [Google Scholar] [PubMed]
- Ministry of Food and Drug Safety (MFDS). Food code. Available online: http://www.foodsafetykorea.go.kr/foodcode/01_01.jsp (accessed on 18 January 2021).
- Korea Meteorological Administration. Climate Statistics Analysis. Available online: https://tinyurl.com/4pnukj5n (accessed on 2 March 2021).

| Type of Cake | aw | Proximate Composition (%) | Bacterial Density (Log CFU/g) 1 | |||||
|---|---|---|---|---|---|---|---|---|
| Moisture | Fat | Protein | Carbohydrate | Salmonella spp. | S. aureus | |||
| Sponge | A | 0.86 ± 0.00 B | 27.7 ± 0.0 F | 16.6 ± 0.4 DE | 8.6 ± 0.1 A | 45.9 ± 0.6 B | 1.5 ± 0.0 C | <LOD2 D |
| B | 0.82 ± 0.02 C | 20.4 ± 0.1 G | 31.0 ± 0.6 B | 6.6 ± 0.0 C | 41.3 ± 0.6 C | 2.9 ± 0.1 A | 5.8 ± 0.4 B | |
| C | 0.80 ± 0.02 C | 27.7 ± 0.0 F | 17.2 ± 0.1 D | 7.2 ± 0.0 B | 47.1 ± 0.1 A | 3.1 ± 0.1 A | 5.5 ± 0.4 B | |
| Mousse | D | 0.92 ± 0.01 A | 43.9 ± 0.1 B | 14.1 ± 0.4 F | 6.3 ± 0.0 D | 35.0 ± 0.2 E | 3.4 ± 0.9 A | 7.7 ± 0.0 A |
| E | 0.90 ± 0.01 A | 40.1 ± 0.0 D | 14.2 ± 0.2 F | 5.9 ± 0.0 E | 39.1 ± 0.2 D | 2.6 ± 0.2 AB | 7.5 ± 0.1 A | |
| F | 0.90 ± 0.01 A | 45.1 ± 0.0 A | 10.0 ± 0.1 G | 3.9 ± 0.1 G | 40.6 ± 0.1 C | 2.6 ± 0.2 AB | 7.8 ± 0.1 A | |
| Cheese | G | 0.90 ± 0.00 A | 32.1 ± 0.1 E | 23.8 ± 0.6 C | 8.6 ± 0.1 A | 34.7 ± 0.8 E | 2.6 ± 0.2 AB | 7.6 ± 0.1 A |
| Brownie | H | 0.70 ± 0.01 D | 12.4 ± 0.1 H | 34.1 ± 0.1 A | 5.7 ± 0.1 F | 46.4 ± 0.3 AB | 1.9 ± 0.1 BC | 3.5 ± 0.0 C |
| Tiramisu | I | 0.91 ± 0.01 A | 43.3 ± 0.3 C | 16.0 ± 0.5 E | 7.4 ± 0.1 B | 32.3 ± 0.3 F | 3.0 ± 0.7 A | 7.9 ± 0.1 A |
| Bacteria | Storage Temperature (°C) | LPD1 (h) | μmax 2 (Log CFU/g/h) | N03 (Log CFU/g) | Nmax4 (Log CFU/g) | R2 |
|---|---|---|---|---|---|---|
| Salmonella spp. | 20 | 1.6 ± 0.0 A | 0.10 ± 0.00 B | 1.9 ± 0.1 | 7.0 ± 0.0 | 0.990–0.993 |
| 25 | 4.0 ± 1.9 A | 0.19 ± 0.00 B | 3.3 ± 0.5 | 7.5 ± 0.0 | 0.992–0.994 | |
| 30 | 5.2 ± 3.8 A | 0.35 ± 0.05 A | 3.8 ± 0.0 | 7.1 ± 0.3 | 0.955–0.972 | |
| S. aureus | 15 | 30.1 ± 8.5 a | 0.05 ± 0.02 b | 3.7 ± 0.0 | 7.2 ± 0.1 | 0.971–0.996 |
| 25 | 6.3 ± 0.2 b | 0.29 ± 0.07 a | 4.0 ± 0.3 | 8.1 ± 0.4 | 0.985–0.989 | |
| 30 | 4.2 ± 2.5 b | 0.33 ± 0.02 a | 3.7 ± 0.7 | 7.6 ± 0.3 | 0.942–0.965 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Park, J.H.; Park, Y.K.; Kim, H.J. Mathematical Modeling for the Growth of Salmonella spp. and Staphylococcus aureus in Cake at Fluctuating Temperatures. Appl. Sci. 2021, 11, 2475. https://doi.org/10.3390/app11062475
Lee H, Park JH, Park YK, Kim HJ. Mathematical Modeling for the Growth of Salmonella spp. and Staphylococcus aureus in Cake at Fluctuating Temperatures. Applied Sciences. 2021; 11(6):2475. https://doi.org/10.3390/app11062475
Chicago/Turabian StyleLee, Heeyoung, Jin Hwa Park, Yu Kyoung Park, and Hyun Jung Kim. 2021. "Mathematical Modeling for the Growth of Salmonella spp. and Staphylococcus aureus in Cake at Fluctuating Temperatures" Applied Sciences 11, no. 6: 2475. https://doi.org/10.3390/app11062475
APA StyleLee, H., Park, J. H., Park, Y. K., & Kim, H. J. (2021). Mathematical Modeling for the Growth of Salmonella spp. and Staphylococcus aureus in Cake at Fluctuating Temperatures. Applied Sciences, 11(6), 2475. https://doi.org/10.3390/app11062475






