Effect of the Lateral Bone Augmentation Procedure in Correcting Peri-Implant Bone Dehiscence Defects: A 7-Years Retrospective Study
Abstract
:1. Introduction
- When the GBR procedure is used for implant dehiscence treatment, it does not influence the marginal bone loss around the implant in 7 years of observation;
- There are no gender-related differences in marginal bone loss around implants;
- There is no difference in marginal bone loss around implants between age groups.
2. Materials and Methods
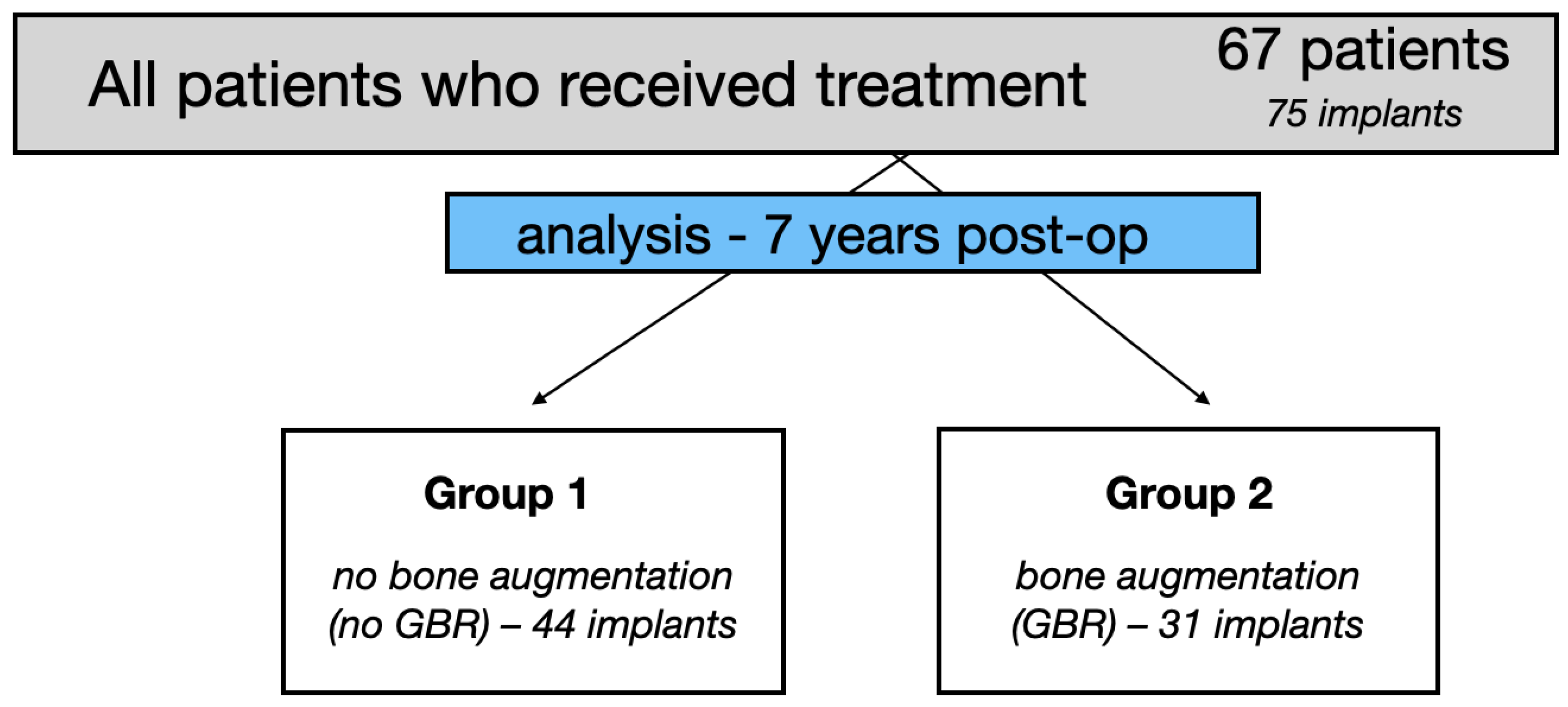
2.1. Study Design
2.2. Clinical Data, Groups of Patients, Surgery, Implant Loading
2.3. Clinical Outcomes
2.4. Marginal Bone Loss
2.5. Statistical Analysis
3. Results
3.1. Implant Survival Rate
3.2. Peri-Implantitis
3.3. Marginal Bone Loss
3.4. Soft Tissue
- The first research hypothesis was accepted. No statistically significant (p < 0.05) differences in terms of marginal bone loss were found between the groups with and without guided bone regeneration;
- The second research hypothesis was accepted. No gender-related differences were found in marginal bone loss;
- The third hypothesis was rejected. A statistically significant difference in marginal bone loss around the implant neck was found between the age group 25–40 and 60–72.
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balshi, T.; Wolfinger, G.; Stein, B.; Balshi, S. A Long-term Retrospective Analysis of Survival Rates of Implants in the Mandible. Int. J. Oral Maxillofac. Implant. 2015, 30, 1348–1354. [Google Scholar] [CrossRef] [PubMed]
- Chappuis, V.; Buser, R.; Brägger, U.; Bornstein, M.M.; Salvi, G.E.; Buser, D. Long-Term Outcomes of Dental Implants with a Titanium Plasma-Sprayed Surface: A 20-Year Prospective Case Series Study in Partially Edentulous Patients. Clin. Implant Dent. Relat. Res. 2013, 15, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Elgali, I.; Omar, O.; Dahlin, C.; Thomsen, P. Guided bone regeneration: Materials and biological mechanisms revisited. Eur. J. Oral Sci. 2017, 125, 315–337. [Google Scholar] [CrossRef] [PubMed]
- Aghaloo, T.; Misch, C.; Lin, G.-H.; Iacono, V.; Wang, H.-L. Bone Augmentation of the Edentulous Maxilla for Implant Placement: A Systematic Review. Int. J. Oral Maxillofac. Implant. 2017, 31, s19–s30. [Google Scholar] [CrossRef] [PubMed]
- Retzepi, M.; Donos, N. Guided Bone Regeneration: Biological principle and therapeutic applications. Clin. Oral Implant. Res. 2010, 21, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Soldatos, N.K.; Stylianou, P.; Koidou, P.; Angelov, N.; Yukna, R.; Romanos, G.E. Limitations and options using resorbable versus nonresorbable membranes for successful guided bone regeneration. Quintessence Int. 2017, 48, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Wickramasinghe, M.L.; Dias, G.J.; Premadasa, K.M.G.P. A novel classification of bone graft materials. J. Biomed. Mater. Res. Part B Appl. Biomater. 2022, 110, 1724–1749. [Google Scholar] [CrossRef]
- Kao, S.T.; Scott, D.D. A Review of Bone Substitutes. Oral Maxillofac. Surg. Clin. N. Am. 2007, 19, 513–521. [Google Scholar] [CrossRef]
- Sbricoli, L.; Guazzo, R.; Annunziata, M.; Gobbato, L.; Bressan, E.; Nastri, L. Selection of collagen membranes for bone regeneration: A literature review. Materials 2020, 13, 30786. [Google Scholar] [CrossRef]
- Ren, Y.; Fan, L.; Alkildani, S.; Liu, L.; Emmert, S.; Najman, S.; Rimashevskiy, D.; Schnettler, R.; Jung, O.; Xiong, X.; et al. Barrier Membranes for Guided Bone Regeneration (GBR): A Focus on Recent Advances in Collagen Membranes. Int. J. Mol. Sci. 2022, 23, 14987. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Zhou, Z.; Lai, H.; Xu, P.; Liao, L.; Wei, J. Biodegradable polymer membranes applied in guided bone/tissue regeneration: A review. Polymers 2016, 8, 115. [Google Scholar] [CrossRef]
- Testori, T.; Weinstein, T.; Scutellà, F.; Wang, H.L.; Zucchelli, G. Implant placement in the esthetic area: Criteria for positioning single and multiple implants. Periodontology 2000 2018, 77, 176–196. [Google Scholar] [CrossRef]
- Chiapasco, M.; Zaniboni, M. Clinical outcomes of GBR procedures to correct peri-implant dehiscences and fenestrations: A systematic review. Clin. Oral Implant. Res. 2009, 20, 113–123. [Google Scholar] [CrossRef]
- Puzio, M.; Hadzik, J.; Błaszczyszyn, A.; Gedrange, T.; Dominiak, M. Soft tissue augmentation around dental implants with connective tissue graft (CTG) and xenogenic collagen matrix (XCM). 1-year randomized control trail. Ann. Anat. 2020, 230, 151484. [Google Scholar] [CrossRef]
- Hadzik, J.; Błaszczyszyn, A.; Gedrange, T.; Dominiak, M. Soft-Tissue Augmentation around Dental Implants with a Connective Tissue Graft (CTG) and Xenogeneic Collagen Matrix (CMX)—5-Year Follow-Up. J. Clin. Med. 2023, 12, 924. [Google Scholar] [CrossRef]
- Puzio, M.; Błaszczyszyn, A.; Hadzik, J.; Dominiak, M. Ultrasound assessment of soft tissue augmentation around implants in the aesthetic zone using a connective tissue graft and xenogeneic collagen matrix—1-year randomised follow-up. Ann. Anat. 2018, 217, 129–141. [Google Scholar] [CrossRef]
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Figuero, E.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S286–S291. [Google Scholar] [CrossRef]
- Cha, H.S.; Kim, J.W.; Hwang, J.H.; Ahn, K.M. Frequency of bone graft in implant surgery. Maxillofac. Plast. Reconstr. Surg. 2016, 38, 1–4. [Google Scholar] [CrossRef]
- Zhang, S.; Li, X.; Qi, Y.; Ma, X.; Qiao, S.; Cai, H.X.; Zhao, B.C.; Jiang, H.B.; Lee, E.S. Comparison of Autogenous Tooth Materials and Other Bone Grafts. Tissue Eng. Regen. Med. 2021, 18, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Dominiak, M.; Dominiak, S.; Targonska, S.; Gedrange, T. Three-Dimensional Bone Block Planning for Mandibular Sagittal Bone Defect Reconstruction. J. Healthc. Eng. 2020, 2020, 8829288. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Yang, R.; Cooper, P.R.; Khurshid, Z.; Shavandi, A.; Ratnayake, J. Bone grafts and substitutes in dentistry: A review of current trends and developments. Molecules 2021, 26, 3007. [Google Scholar] [CrossRef] [PubMed]
- Elnayef, B.; Porta, C.; del Amo, F.; Mordini, L.; Gargallo-Albiol, J.; Hernández-Alfaro, F. The Fate of Lateral Ridge Augmentation: A Systematic Review and Meta-Analysis. Int. J. Oral Maxillofac. Implant. 2018, 33, 622–635. [Google Scholar] [CrossRef] [PubMed]
- Barbu, H.M.; Iancu, S.A.; Rapani, A.; Stacchi, C. Guided bone regeneration with concentrated growth factor enriched bone graft matrix (Sticky bone) vs. bone-shell technique in horizontal ridge augmentation: A retrospective study. J. Clin. Med. 2021, 10, 3953. [Google Scholar] [CrossRef] [PubMed]
- Jain, G.; Blaauw, D.; Chang, S. A Comparative Study of Two Bone Graft Substitutes–InterOss® Collagen and OCS-B Collagen®. J. Funct. Biomater. 2022, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Canellas, J.V.d.S.; Soares, B.N.; Ritto, F.G.; Vettore, M.V.; Vidigal Júnior, G.M.; Fischer, R.G.; Medeiros, P.J.D.A. What grafting materials produce greater alveolar ridge preservation after tooth extraction? A systematic review and network meta-analysis. J. Cranio-Maxillofac. Surg. 2021, 49, 1064–1071. [Google Scholar] [CrossRef]
- Özkan, Y.; Akoǧlu, B.; Kulak-Özkan, Y. Maxillary sinus floor augmentation using bovine bone grafts with simultaneous implant placement: A 5-year prospective follow-up study. Implant Dent. 2011, 20, 455–459. [Google Scholar] [CrossRef]
- Urban, I.A.; Nagursky, H.; Lozada, J.L.; Nagy, K. Horizontal Ridge Augmentation with a Collagen Membrane and a Combination of Particulated Autogenous Bone and Anorganic Bovine Bone–Derived Mineral: A Prospective Case Series in 25 Patients. Int. J. Periodontics Restor. Dent. 2013, 33, 299–307. [Google Scholar] [CrossRef]
- Arnal, H.M.; Angioni, C.D.; Gaultier, F.; Urbinelli, R.; Urban, I.A. Horizontal guided bone regeneration on knife-edge ridges: A retrospective case–control pilot study comparing two surgical techniques. Clin. Implant Dent. Relat. Res. 2022, 24, 211–221. [Google Scholar] [CrossRef]
- Selahi, D.; Spiegel, M.; Hadzik, J.; Pitułaj, A.; Michalak, F.; Kubasiewicz-ross, P.; Dominiak, M. The Appliance of A-PRF and CGF in the Treatment of Impacted Mandibular Third Molar Extraction Sockets—Narrative Review. Appl. Sci. 2023, 13, 165. [Google Scholar] [CrossRef]
- Pietruszka, P.; Chruścicka, I.; Duś-Ilnicka, I.; Paradowska-Stolarz, A. PRP and PRF—Subgroups and Divisions When Used in Dentistry. J. Pers. Med. 2021, 11, 944. [Google Scholar] [CrossRef]
- Aludden, H.C.; Mordenfeld, A.; Hallman, M.; Dahlin, C.; Jensen, T. Lateral ridge augmentation with Bio-Oss alone or Bio-Oss mixed with particulate autogenous bone graft: A systematic review. Int. J. Oral Maxillofac. Surg. 2017, 46, 1030–1038. [Google Scholar] [CrossRef]
- Sanz-Sánchez, I.; Carrillo de Albornoz, A.; Figuero, E.; Schwarz, F.; Jung, R.; Sanz, M.; Thoma, D. Effects of lateral bone augmentation procedures on peri-implant health or disease: A systematic review and meta-analysis. Clin. Oral Implant. Res. 2018, 29, 18–31. [Google Scholar] [CrossRef]
- Zitzmann, N.U.; Schärer, P.; Marinello, C.P. Long-term results of implants treated with guided bone regeneration: A 5-year prospective study. Int. J. Oral Maxillofac. Implant. 2001, 16, 355–366. [Google Scholar]
- Severi, M.; Simonelli, A.; Farina, R.; Tu, Y.K.; Lan, C.H.; Shih, M.C.; Trombelli, L. Effect of lateral bone augmentation procedures in correcting peri-implant bone dehiscence and fenestration defects: A systematic review and network meta-analysis. Clin. Implant Dent. Relat. Res. 2022, 24, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Avila-Ortiz, G.; Chambrone, L.; Vignoletti, F. Effect of alveolar ridge preservation interventions following tooth extraction: A systematic review and meta-analysis. J. Clin. Periodontol. 2019, 46, 195–223. [Google Scholar] [CrossRef] [PubMed]
- Majzoub, J.; Ravida, A.; Starch-Jensen, T.; Tattan, M.; Suárez-López del Amo, F. The Influence of Different Grafting Materials on Alveolar Ridge Preservation: A Systematic Review. J. Oral Maxillofac. Res. 2019, 10, 10306. [Google Scholar] [CrossRef] [PubMed]
- Kubasiewicz-Ross, P.; Fleischer, M.; Pitułaj, A.; Hadzik, J.; Nawrot-Hadzik, I.; Bortkiewicz, O.; Dominiak, M.; Jurczyszyn, K. Evaluation of the three methods of bacterial decontamination on implants with three different surfaces. Adv. Clin. Exp. Med. 2020, 29, 177–182. [Google Scholar] [CrossRef]
- Diaz, P.; Gonzalo, E.; Villagra, L.J.G.; Miegimolle, B.; Suarez, M.J. What is the prevalence of peri-implantitis? A systematic review and meta-analysis. BMC Oral Health 2022, 22, 1–13. [Google Scholar] [CrossRef]
- Derks, J.; Schaller, D.; Håkansson, J.; Wennström, J.L.; Tomasi, C.; Berglundh, T. Effectiveness of Implant Therapy Analyzed in a Swedish Population: Prevalence of Peri-implantitis. J. Dent. Res. 2016, 95, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Wada, M.; Mameno, T.; Otsuki, M.; Kani, M.; Tsujioka, Y.; Ikebe, K. Prevalence and risk indicators for peri-implant diseases: A literature review. Jpn. Dent. Sci. Rev. 2021, 57, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Skorik, Y.A.; Puppi, D.; Wiessner, A.; Wassmann, T.; Wiessner, J.M.; Schubert, A.; Wiechens, B.; Hampe, T.; Bürgers, R. In Vivo Biofilm Formation on Novel PEEK, Titanium, and Zirconia Implant Abutment Materials. Int. J. Mol. Sci. 2023, 24, 1779. [Google Scholar] [CrossRef]
- Hadzik, J.; Kubasiewicz-Ross, P.; Gębarowski, T.; Waloszczyk, N.; Maciej, A.; Stolarczyk, A.; Gedrange, T.; Dominiak, M.; Szajna, E.; Simka, W. An Experimental Anodized Titanium Surface for Transgingival Dental Implant Elements—Preliminary Report. J. Funct. Biomater. 2023, 14, 34. [Google Scholar] [CrossRef]
- Hadzik, J.; Kubasiewicz-Ross, P.; Simka, W.; Gębarowski, T.; Barg, E.; Cieśla-Niechwiadowicz, A.; Szajna, A.T.; Szajna, E.; Gedrange, T.; Kozakiewicz, M.; et al. Fractal Dimension and Texture Analysis in the Assessment of Experimental Laser-Induced Periodic Surface Structures (LIPSS) Dental Implant Surface-In Vitro Study Preliminary Report. Materials 2022, 15, 82713. [Google Scholar] [CrossRef] [PubMed]
- Kordbacheh Changi, K.; Finkelstein, J.; Papapanou, P.N. Peri-implantitis prevalence, incidence rate, and risk factors: A study of electronic health records at a U.S. dental school. Clin. Oral Implant. Res. 2019, 30, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Linkevicius, T.; Apse, P.; Grybauskas, S.; Puisys, A. The influence of soft tissue thickness on crestal bone changes around implants: A 1-year prospective controlled clinical trial. Int. J. Oral Maxillofac. Implant. 2009, 24, 712–719. [Google Scholar]
- Lissek, M.; Boeker, M.; Happe, A. How thick is the oral mucosa around implants after augmentation with different materials: A systematic review of the effectiveness of substitute matrices in comparison to connective tissue grafts. Int. J. Mol. Sci. 2020, 21, 5043. [Google Scholar] [CrossRef] [PubMed]
- Hadzik, J.; Kubasiewicz-Ross, P.; Nawrot-Hadzik, I.; Gedrange, T.; Pitułaj, A.; Dominiak, M. Short (6 mm) and regular dental implants in the posterior maxilla–7-years follow-up study. J. Clin. Med. 2021, 10, 940. [Google Scholar] [CrossRef]
- Gianfilippo, R.D.; Valente, N.A.; Toti, P.; Wang, H.L.; Barone, A. Influence of implant mucosal thickness on early bone loss: A systematic review with meta-analysis. J. Periodontal Implant Sci. 2020, 50, 209–225. [Google Scholar] [CrossRef]
- Hadzik, J.; Botzenhart, U.; Krawiec, M.; Gedrange, T.; Heinemann, F.; Vegh, A.; Dominiak, M. Comparative evaluation of the effectiveness of the implantation in the lateral part of the mandible between short tissue level (TE) and bone level (BL) implant systems. Ann. Anat. 2017, 213, 78–82. [Google Scholar] [CrossRef]
- Saleh, M.H.A.; Ravidà, A.; Suárez-López del Amo, F.; Lin, G.H.; Asa’ad, F.; Wang, H.L. The effect of implant-abutment junction position on crestal bone loss: A systematic review and meta-analysis. Clin. Implant Dent. Relat. Res. 2018, 20, 617–633. [Google Scholar] [CrossRef]
- Makowiecki, A.; Hadzik, J.; Błaszczyszyn, A.; Gedrange, T.; Dominiak, M. An evaluation of superhydrophilic surfaces of dental implants—A systematic review and meta-analysis. BMC Oral Health 2019, 19, 79. [Google Scholar] [CrossRef] [PubMed]
- Valles, C.; Rodríguez-Ciurana, X.; Clementini, M.; Baglivo, M.; Paniagua, B.; Nart, J. Influence of subcrestal implant placement compared with equicrestal position on the peri-implant hard and soft tissues around platform-switched implants: A systematic review and meta-analysis. Clin. Oral Investig. 2018, 22, 555–570. [Google Scholar] [CrossRef] [PubMed]




| G1 | G2 | G1, Excluded Peri-Implantitis | G2, Excluded Peri-Implantitis | |
|---|---|---|---|---|
| mean MBL (in mm) | 0.63 | 0.98 | 0.60 | 0.72 |
| SD | 0.6 | 1.0 | 0.57 | 0.65 |
| minimum | 0 | 0 | 0 | 0 |
| maximum | 2.12 | 3.97 | 2.17 | 2.60 |
| Male | Female | Male Peri-Implantitis Excluded | Female Peri-Implantitis Excluded | |
|---|---|---|---|---|
| mean MBL (in mm) | 0.85 | 0.73 | 0.60 | 0.67 |
| SD | 0.93 | 0.75 | 0.56 | 0.64 |
| minimum | 0 | 0 | 0 | 0 |
| maximum | 3.97 | 3.5 | 1.95 | 2.58 |
| All Implants | 25–40 Years Old | 41–60 Years Old | 60–72 Years Old |
| mean MBL (in mm) | 0.60 | 0.67 | 1.13 |
| SD | 0.68 | 0.80 | 0.88 |
| minimum | 0 | 0 | 0 |
| maximum | 2.60 | 3.97 | 3.50 |
| Excluded peri-implantitis | 25–40 years old | 41–60 years old | 60–72 years old |
| mean MBL (in mm) | 0.51 | 0.57 | 0.95 |
| SD | 0.54 | 0.57 | 0.67 |
| minimum | 0 | 0 | 0 |
| maximum | 1.95 | 2.17 | 2.58 |
| G1 | G2 | G1, Excluded Peri-Implantitis | G2, Excluded Peri-Implantitis | |
|---|---|---|---|---|
| mean TKT (in mm) | 1.95 | 1.74 | 1.95 | 1.73 |
| SD | 0.72 | 0.65 | 0.72 | 0.67 |
| minimum | 0.72 | 0.45 | 0.72 | 0.45 |
| maximum | 3.45 | 2.9 | 3.45 | 2.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadzik, J.; Błaszczyszyn, A.; Gedrange, T.; Dominiak, M. Effect of the Lateral Bone Augmentation Procedure in Correcting Peri-Implant Bone Dehiscence Defects: A 7-Years Retrospective Study. Appl. Sci. 2023, 13, 2324. https://doi.org/10.3390/app13042324
Hadzik J, Błaszczyszyn A, Gedrange T, Dominiak M. Effect of the Lateral Bone Augmentation Procedure in Correcting Peri-Implant Bone Dehiscence Defects: A 7-Years Retrospective Study. Applied Sciences. 2023; 13(4):2324. https://doi.org/10.3390/app13042324
Chicago/Turabian StyleHadzik, Jakub, Artur Błaszczyszyn, Tomasz Gedrange, and Marzena Dominiak. 2023. "Effect of the Lateral Bone Augmentation Procedure in Correcting Peri-Implant Bone Dehiscence Defects: A 7-Years Retrospective Study" Applied Sciences 13, no. 4: 2324. https://doi.org/10.3390/app13042324
APA StyleHadzik, J., Błaszczyszyn, A., Gedrange, T., & Dominiak, M. (2023). Effect of the Lateral Bone Augmentation Procedure in Correcting Peri-Implant Bone Dehiscence Defects: A 7-Years Retrospective Study. Applied Sciences, 13(4), 2324. https://doi.org/10.3390/app13042324







