1. Introduction
Different fish species play an important role in human nutrition as an excellent source of proteins and unsaturated fatty acids, especially long-chain omega-3 fatty acids, and vitamin D. However, raw fish and many mildly processed fish products are highly perishable due to neutral pH and high water activity, and the oxidative process that starts almost immediately when fish are caught [
1]. Smoking and salting enhance the taste of many fish products, and at the same time, they are traditional ways to increase their safety. Food preservatives provide longer shelf life, but consumers tend to prefer clean label ingredients and are driving the food industry to search for “green” food preservation technologies [
2]. This has pushed both food researchers and food industry to seek alternative processing methods, such as high pressure processing (HPP), for various food items, including hot and cold smoked rainbow trout (
Oncorhynchus mykiss).
HPP research started sometime in the 1990s and since then, experiments with different kinds of food have been investigated, and later also utilized, in industrial applications. The essential advantage of HPP is based on the isostatic principle that pressure is uniformly distributed throughout the product independently of its size or geometry. Moreover, HPP preserves nutritional properties better than processes involving heating. Compared with non-treated samples, HPP extends the shelf-life and tackles challenges regarding microbial quality. As with any food processing method, HPP also has its challenges: it requires pre-packaging and product-specific optimal conditions. One factor that needs to be considered is the high initial investment of the equipment [
3,
4,
5].
De Oliveira et al. [
1] reviewed the effects of high pressure on the quality of pressurised fish meat. High pressure changes the quaternary, tertiary and secondary structures of proteins, leading to dissociation, unfolding, denaturation, aggregation, precipitation, and gelatinization in varying intensities. Already pressures 150–200 MPa may be able to dissociate oligomeric proteins, potentially leading to protein aggregation or precipitation, and beyond those pressures, unfolding of proteins starts to take place [
6]. Depending on compression rate and rearrangements in the secondary structures of proteins, pressures above 300–700 MPa can cause non-reversible denaturation [
6]. Denaturation of fish proteins by HPP opens the structure of the protein, enabling better action of hydrolytic enzymes and thus, potentially contributes to the digestibility and bioavailability of proteins [
7]. However, along with denaturation, the hydrophobic components of proteins are revealed which, together with other interactions, can lead to protein aggregation. Excessive protein aggregation limits the activity of peptidase enzymes and thus, reduces protein digestibility [
7]. Therefore, HPP may have both positive and negative impacts on protein digestibility and bioavailability. Zhang et al. [
8] found that the digestibility of cod (
Gadus morhua) was improved when the samples were processed at 200 MPa for 20 min. HPP treatment can also cause protein oxidation in fish [
1]. Oxidation of proteins due to pressure can produce free radicals that cause various oxidizing reactions. As a result, the digestibility of food proteins may be reduced. Data on the effects of high-pressure treatment on the digestibility of fish proteins, especially hot or cold smoked rainbow trout, is limited. The compaction of muscle fibres leads to changes in hardness by decreasing sarcomere length, and the liquid release. The fluid loss retained by capillary forces alters the water holding capacity (WHC). Protein denaturation alters protein solubility and may lead to exposure of acidic or alkaline amino acid residues and hydrogen bonds, which can alter the pH, and furthermore, cause changes in WHC [
1].
HPP can also induce changes in the sensory quality of fish; for example, protein denaturation affects fish muscle texture and colour [
9]. In a study by Yagiz et al. [
10] rainbow trout samples were subjected to 150, 300, 450 and 600 MPa pressure for 15 min in room temperature. Two latter pressures induced higher hardness and cohesiveness of fish meat. Similar results were obtained by Yagiz et al. [
11], when they studied salmon (
Salmo salar) samples processed at 150 and 300 MPa for 15 min at 4 °C. Pressure increased hardness, gumminess, and chewiness, and decreased adhesiveness when compared to the control. No significant differences in hardness, adhesiveness, gumminess, and chewiness were observed for samples subjected to 150 MPa and the control. This was further demonstrated in Gudbjornsdottir et al. [
12] when testing cutting strength with salmon. They found no difference in toughness between the control and the pressurized samples (400–900 MPa for 20 s), but on the other hand, samples subjected to 900 MPa for 10, 30 or 60 s were tougher than the samples processed at 400 MPa. While the previous literature indicates that at least 400–600 MPa might be needed to inactivate spoiling and pathogenic microorganisms in fish products, pressures higher than 300 MPa may risk sensory quality; yet, the pressure thresholds might be affected by the fish species in question [
13]. All these findings reflect the importance of testing the appropriate processing parameters case by case, as the amount of pressure, holding time and temperature all influence the food item itself and thus, the consumer acceptance. Rainbow trout is among the most important farmed fish species [
14]; yet, only a limited number of papers have been published on the effects of HPP parameters on rainbow trout quality.
The aim of this study was to investigate how HPP with different treatment intensities in regards to pressure level affects protein solubility, protein digestibility, and WHC of cold smoked and hot smoked rainbow trout. In addition, we wanted to examine how these treatments alter the consumer perceived sensory properties and whether the protein-related variables contribute to the observed changes in sensory quality.
2. Materials and Methods
2.1. Fish Samples
For the in vitro digestions and WHC analyses, cold smoked rainbow trout (CSRT) fillets and hot smoked rainbow trout (HSRT) fillets were obtained from a local supermarket in Kuopio, Finland. Fish were cut into ca. 50 g pieces and packed in polyamide/polyethylene bags. Bags were vacuumed-sealed with a domestic vacuum machine (Chef; OBH Nordica, Stockholm, Sweden). Samples were stored at 5 °C for 16 h before high pressure processing (HPP) and transported on ice to the processing site.
For the consumer evaluation, packed CSRT fillets (ca. 150 g) and HSRT fillets (ca. 700 g) were provided by Escamar Seafood Ltd. (Kuopio, Finland) two days prior to the first assessment day. Samples were stored at 5 °C until HPP, taking place on the day before the first assessment day.
2.2. High Pressure Processing
HPP was conducted at Toripiha Ltd. (Suonenjoki, Finland) with a hydrostatic pressure equipment (ThyssenKrupp UHDE 350-60, Hagen, Germany). Samples were treated with 0, 200, 400, or 600 MPa for 3 min at 4 °C. The pressurisation rate was approximately 1.7 MPa/s with immediate decompression time after pressurisation. After HPP, samples were stored at 5 °C.
2.3. In Vitro Digestion
HPP-processed CSRT and HSRT samples (200, 400, and 600 MPa), and unprocessed (0 MPa) control samples were digested by INFOGEST method [
15], with some modifications. Each fish sample was mixed 1:1 (
w/v) with artificial saliva (15.1 mM KCl, 3.7 mM KH
2PO
4, 13.6 mM NaHCO
3, 0.15 mM MgCl
2(H
2O)
6, 1.1 mM HCl, 1.5 mM CaCl
2(H
2O)
2). Samples were homogenized in mortar with the saliva solution for 2 min. The fish-saliva homogenates were mixed 1:1 with 1% pepsin (Sigma-Aldrich, P7125, 400 U/mg; 0.1 M HCl in 47.4 mM NaCl; pH 3.0) and incubated for 2 h at 37 °C (Certomat HK, BBI Biotech, Germany), with shaking (150 rpm; Orbital Shaker SO1, Stuart Scientific, UK). The pepsin digesta were mixed 1:1 with pancreatin-bile mixture (0.2% pancreatin (8×); 0.82% ox-bile (Millipore, 70168); 0.2 M NaHCO
3 in 38.4 mM NaCl; pH 7.0), and the samples were incubated for 2 h at 37 °C, with shaking (200 rpm). To inactivate the digestive enzymes, the intestinal digesta were heated for 15 min at 95 °C. Each sample type was treated three times in separate digestions.
2.4. Level of Proteolysis
The level of proteolysis (LP) was determined based on the increase in non-protein nitrogen (NPN) after in vitro digestion. NPN was extracted from fish samples and from intestinal digesta as follows: each fish sample was thoroughly homogenized with a domestic stick blender in ultrapure water (1:7 w/v). From these homogenates, pH was measured with pH meter instrument (WTW InoLab, Xylem Inc., Washington, DC, USA). Fish homogenates and intestinal digesta samples were extracted with 60% acetone (1:3 v/v) for 10 min at room temperature with vigorous shaking. Extracts were centrifuged (3026× g, 20 min, 20 °C; Centrifuge BR 4i, Jouan SA, France) and supernatants were collected. The remaining pellets were extracted with 15 % trichloroacetic acid (TCA; original sample size (g) to TCA ratio 1:2 w/v) for 10 min at 5 °C. Extracts were centrifuged (3026× g, 20 min, 20 °C), and supernatants were collected. Acetone and TCA extracts were combined and filtered with 40 µm filter paper.
The nitrogen contents of fish samples (Ntot), fish NPN extracts (NPNfish) and digesta NPN extracts (NPNdigesta) were measured with the Kjeldahl method [
16]. LP was calculated as follows: (NPNdigesta − NPNfish)/Ntot × 100.
2.5. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis
Soluble muscle protein profiles and digestion patterns of fish proteins were observed with Sodium Dodecyl Sulphate Polyacrylamide Gel Electrophoresis (SDS-PAGE). All samples were analysed in triplicates.
Soluble myofibrillar proteins and sarcoplasmic proteins were extracted with NaCl and water, respectively. Twelve ml aliquots of each fish meat homogenate (1:7 w/v in ultrapure H2O) were first centrifuged (3026× g, 15 min, 4 °C) and the supernatants collected. The remaining fish pellets were extracted with 2 × 10 mL 0.5 M NaCl and 1 × 10 mL H2O, respectively. Between the extraction steps, the samples were centrifuged (3026× g, 15 min, 4/20 °C) and the supernatants were collected and combined. Final soluble protein (SP) samples were filtered with 40 µm filter paper. Protein contents in the SP extracts were determined with BCA protein assay kit (Pierce Biotechnology, Rockford, IL, USA). The amount of soluble proteins was calculated as mg per g of fish muscle sample.
Mini-PROTEAN® 3 (Bio-Rad, Hercules, CA, USA) equipment was used to run the gels (resolving gel and stacking gel containing 12% and 4% polyacrylamide, respectively). Protein contents in the intestinal digesta were determined with BCA protein assay kit, and the amount of protein that was loaded on the gels was normalized to 7.5 µg for each SP and digesta sample. Samples were mixed 1:1 with Laemmli loading buffer (62.5 mM Tris base, 25% (w/v) glycerol, 2% (w/v) SDS, 5% (v/v) β-mercaptoethanol, 0.01% (w/v) bromophenol blue) and heated at 95 °C for 4 min before loading on the gel with a protein molecular marker (PageRuler Plus Prestained Protein Ladder, Thermo Scientific, Waltham, Massachusetts, US). Gels were run with 150 V and upon completion, they were fixed with methanol-acetic acid solution (50% and 10%, respectively) and stained with Coomassie Brilliant Blue R-250 (Bio-Rad).
2.6. Water Holding Capacity
WHC was tested according to Teixeira et al. [
17]. The samples were treated in triplicates.Approximately 2 g (±0.04 g) of each fish sample was weighted (Ws) and wrapped in two layers of filter paper; the filter papers were also weighted beforehand (Wi). Samples were centrifuged in 50 mL test tubes for 10 min (3000×
g, 20 °C). After that, the filter papers were weighted again (Wf). Water content (H; (fresh weight − dry weight)/fresh weight) of the samples was determined by drying ca. 1 g (±0.08 g) of each sample at 50 °C for 48 h. WHC was calculated as follows: WHC = Ws × H − (Wf − Wi)/Ws × H × 100
2.7. Consumer Sensory Evaluation
The consumer sensory evaluation was carried out for CSRT and WSRT samples. The evaluations were performed in the sensory evaluation laboratory of the University of Eastern Finland [
18]. Sensory evaluation sessions were designed, and data collected with EyeQuestion software (Elst, The Netherlands, version 5.0.8.3).
Sensory evaluation was conducted following the ethical principles of the University of Eastern Finland. All participants provided their informed consent for inclusion. The non-trained consumers were recruited from the Kuopio area (Finland) by distributing a research call in different social media platforms. The exclusion criteria were pregnancy, breastfeeding, daily smoking, and fish allergy. All participants had to consume fish at least once every two weeks. A total of 53 consumers (37 females, 16 males) aged 18–65 years participated in the study (
Appendix A).
Prior to the evaluation, samples were cut in pieces (15 g) and stored at 6 °C in transparent plastic plates covered with a transparent lid. Samples were taken at room temperature 10 min prior to evaluation and the lid removed. All the samples were coded with random three-digit-numbers and presented to the panellists in a randomized order. The HPP treated (200, 400 or 600 MPa) and untreated control samples were evaluated in separate sets for CSRT and HSRT. The participants were asked to rinse their mouth with filtered tap water and eat a piece of gluten-free crispbread prior to each sample.
The Rate-All-That-Apply (RATA) method [
19] was applied to profile the sensory properties of rainbow trout samples treated with HPP. In addition, participants were asked to evaluate the appeal of the samples prior to tasting using a 9-point scale (1 = not appealing at all, 9 = very appealing) and overall liking after tasting the samples (1 = I do not like it at all, 9 = I like it very much). The list of RATA terms was chosen based on the literature [
20,
21,
22] and modified based on the consensus of the research team. Altogether, 18 RATA terms were grouped into three categories most likely to be affected by the HPP treatment: texture (11 terms), appearance (four terms), and overall impression (three terms). The order of the RATA terms in each category was randomized. Consumers were asked to choose the RATA terms they perceived applicable to describe the samples and then rate their intensity with a 5-point scale (1 = slightly applicable, 5 = very applicable). In the end, participants were able to provide voluntary written comments regarding the evaluated samples. The sensory evaluation protocol was validated with a trained in-house panel (n = 3) prior to consumer sensory evaluation.
2.8. Statistical Analyses
Statistical analyses were conducted using IBM SPSS Statistics software version 26 (IBP Corp., Armonk, NY, USA). SP, LP, and WHC results were tested with one-way analysis of variance (ANOVA). Due to the small sample size (n = 3 per each HPP treatment), Dunnett’s T3 post-hoc test was used for pair-wise comparisons. Significance level was set to 0.05. Since appeal, overall liking and RATA data did not follow a normal distribution (Shapiro–Wilk test), the Friedman and Wilcoxon tests were applied to analyse statistically significant differences between samples (
p < 0.05). The Bonferroni correction was used with pair-wise comparisons. If a participant had not chosen a RATA term, it was rated as zero (0 = not applicable for the sample) to form a scale of 0–6 [
23].
Principal Component Analysis (PCA) was conducted on CSRT samples to observe potential correlations between sensory attributes and physical variables (namely SP and WHC) showing statistically significant differences according to the intensity of HPP treatment. Pressure levels were included in the analysis as a binary variable (1 = pressure level applied, 0 = pressure level not applied). To obtain an equal number of observations (n = 53) for sensory and physical variables, SP and WHC results were propagated within each sample type in random order using Excel (Microsoft Corp., Redmond, WA, US). Components were extracted based on eigenvalue threshold 1. PCA was not conducted on HSRT samples in order to avoid overinterpretation of the results due to the lack of significant differences in SP and WHC between the pressure treatments.
4. Discussion
For HSRT samples, LP was found to be highest in HSRT-200, showing LP of 75%, which was significantly higher than LP of 68% in HSRT-600. Previous studies have reported similar levels of in vitro proteolysis in fish muscle: depending on the pre-treatment and/or cooking methods, pancreatic digestibility of fish proteins have been reported to vary between ca. 56% and 80% [
8,
24]. HPP treatment of 200 MPa at 25 °C for 20 min has been found to increase pancreatic digestibility of cod proteins in comparison to fresh samples [
8]. Although there are considerable differences in the study designs and raw materials, it could be speculated that 200 MPa might be a safe and even beneficial pressure level regarding fish protein digestibility. Based on the present results, it can also be suggested that physical protein-related variables did not affect LP. While CSRT samples showed significant differences in SP and WHC, but no significant differences in LP, HSRT samples did not vary in SP or WHC, but demonstrated small, yet significant changes in LP according to applied HPP treatment.
Indeed, WHC in CSRT samples was significantly reduced when 400 and 600 MPa pressure levels were used. Teixeira et al. [
17] reported reduced WHC of sea bass fillets when the samples were treated at 250 or 400 MPa for 0–30 min instead of 100 MPa (6 °C). In comparison to control samples, 100 MPa did not have a significant effect on WHC. Reduced WHC in HPP-treated muscle samples was probably due to protein denaturation and degradation [
25]. This is supported by the fact that protein solubility was reduced in CSRT samples, but not in HSRT samples, suggesting protein denaturation in CSRT as a result of HPP. Pressures higher than 200 MPa have been observed to reduce solubility of sarcoplasmic proteins in beef longissimus muscle [
26]. Furthermore, actin, a myofibrillar protein, has been suggested to be susceptible to denaturation by HPP in high-fat fish [
9], such as rainbow trout.
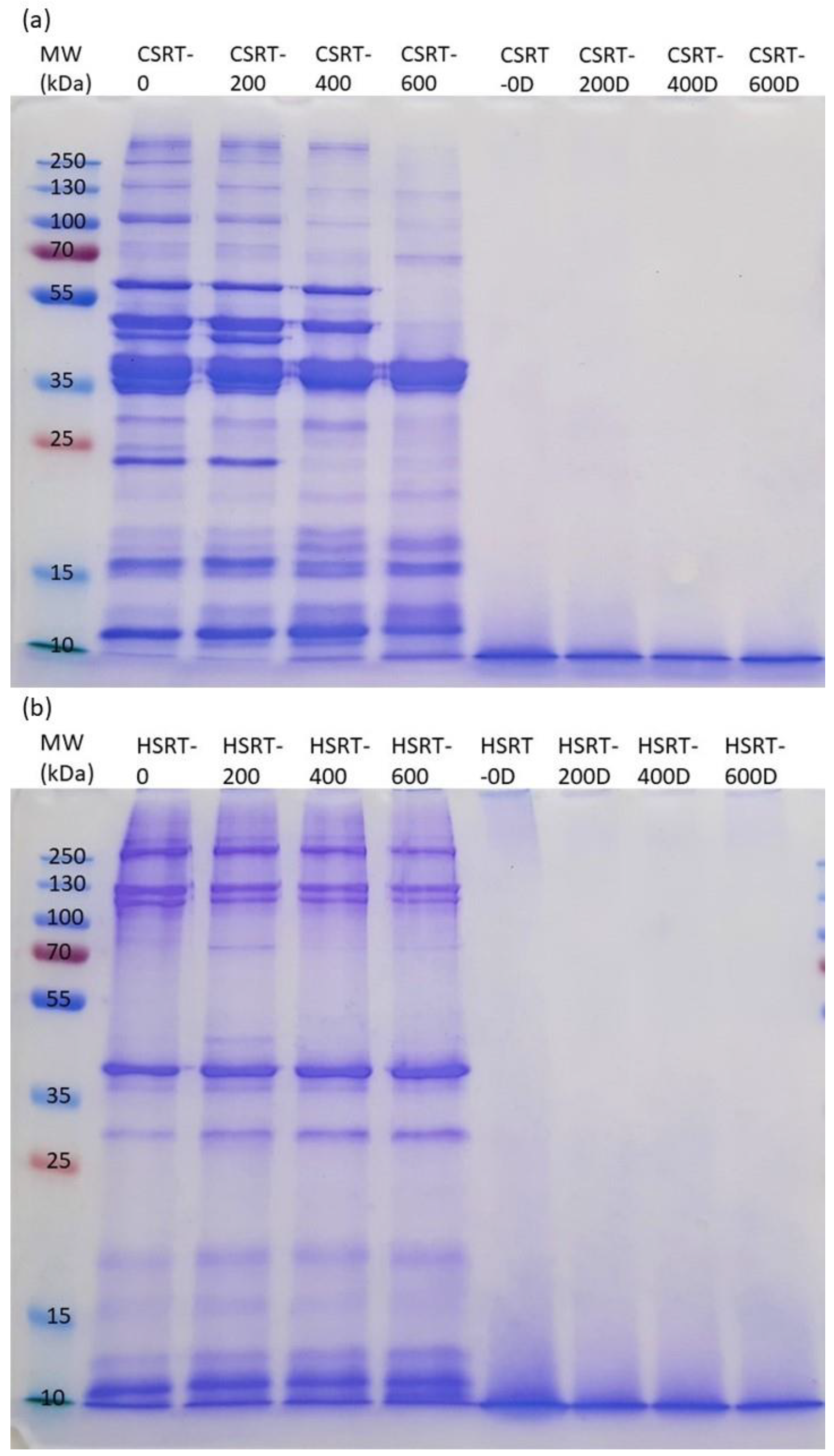
Protein bands in SDS-PAGE indicate that sarcoplasmic proteins [
27] in HSRT were already largely insolubilized at the smoking phase. Instead, in CSRT-0, many sarcoplasmic and myofibrillar proteins [
28] are still soluble, while especially sarcoplasmic proteins, but also some myofibrillar proteins are insolubilized by HPP at 400 and 600 MPa. Furthermore, high pressures may potentially break down muscle proteins [
26]. For CSRT, this could explain the increase in band intensity in SDS-PAGE at the lower end of the molecular weight spectrum with higher HPP treatment intensity. Cioca et al. [
29] studied rainbow trout samples with variable pressures (100, 200, 400 or 600 MPa) either for 3 or 6 min and found that the use of lower pressure levels 100 or 200 MPa did not have a major influence on myosin or actin, but instead, higher pressures 400 or 600 MPa did. These results are well in line with ours, although in their study, the ambient temperature during HPP treatment was approximately 35–47 °C, which remarkably differs from our study, where the processing temperature was 4 °C.
CSRT underwent more significant changes in texture and appearance according to HPP treatments in comparison to HSRT. For both samples, juiciness, dryness, and moistness were significantly affected by HPP. Appearance was significantly altered only in CSRT. Hardness was significantly increased in CSRT-600 in comparison to CSRT-0 and CSRT-200 samples, which is in line with the study of Yagiz et al. [
10] who showed that HPP at 450 or 600 MPa significantly increased hardness in rainbow trout in comparison to samples treated at 150 or 300 MPa. Christensen et al. [
30] studied salmon, cod and mackerel for the sensory quality parameters including texture and colour, with an HPP treatment of 200 and 500 MPa for 2 min at 8–9 °C. The main findings were that hardness and lightness increased at 500 MPa and redness decreased. Same was true for CSRT-400 and CSRT-600 samples in our study. On the other hand, Mengden et al. [
31] used a trained ten-person panel to evaluate mild smoked rainbow trout and fresh European catfish fillets treated with HPP at 200, 400 or 600 MPa for 1 or 5 min at room temperature and found only minor effects on the overall appearance, colour and texture of rainbow trout fillets. Instead, catfish tended to become paler and thus, a treatment with 600 MPa for 5 min for rainbow trout fillets was recommended. The challenge in comparing different studies investigating the effects of HPP on sensory properties of fish products is the variability in treatment parameters and fish raw materials.
Interestingly, CSRT-200 was rated with the highest overall liking score among all CSRT samples in our study. The same sample was also rated as most juicy and freshest. Although overall liking was more strongly loaded together with appeal and pleasantness in the PCA, some positive correlation was also seen between overall liking and the PC with positive loadings of juiciness and freshness. Meanwhile, a small negative correlation was seen between overall liking and the PC with chewiness, hardness, dryness, and firmness. In the present study, WHC and SP were positively associated with intense red colour and negatively associated with light colour of CSRT samples. This was highlighted in samples treated at 600 MPa. Protein denaturation is related with colour changes of fish muscle, and it has been shown to contribute to fish surface lightness [
32,
33]. In HPP-treated cod and salmon samples, HPP was found to reduce redness and to increase lightness, and especially denaturation of myosin was suggested to be a major factor [
9]. Pressure of 300 MPa was suggested as a threshold for both protein denaturation and colour changes, as the treatment at 150 MPa did not cause significant changes in these variables in comparison to untreated control samples. In our analyses as well, samples treated with 0 or 200 MPa showed some positive correlation with intense red colour, WHC, and SP while being negatively associated with light colour. Against expectations, WHC did not correlate with juiciness, dryness, or moistness in our study. However, although WHC in HSRT samples did not vary at a statistically significant level, the trends in WHC of HSRT were in parallel with juiciness and moistness while showing an opposite trend with dryness. PCA on CSRT samples potentially indicated the drop in WHC when increasing the pressure from 0 to 200 MPa and the small recovery of WHC when increasing the pressure from 400 to 600 MPa.
HPP may also cause shifts in fish muscle pH [
34], and consequently, changes in taste and consumer liking [
35]. In our study, the pH levels varied between 6.21 and6.27 in CSRT and between 6.36 and6.46 in HSRT, without any significant differences according to treatment pressure levels (
Appendix B). This finding is in accordance with a previous study by Rode and Hovda [
36].