1. Introduction
Diesel engines have been widely used on various occasions because of their excellent thermal efficiency and reliability. However, nitrogen oxide (NO
x) emissions from diesel engines have an adverse effect on human beings and the environment [
1], and regulations for diesel engine NO
x emission have been put forward by governing bodies all over the world [
2]. According to Euro VI legislation, NO
x are restricted under 0.40 g/kW·h (steady-state testing) and 0.46 g/kW·h (transient-state testing), respectively [
3]. Even so, the super ultra-low NO
x (ULN) emission standards for medium-duty vehicles, established by the California Air Resources Board, requires that the total emissions of nonmethane organic gases and NO
x are no more than 0.02 g/mile under the Heavy-Duty Federal Test Procedure (HD-FTP) [
4]. This requires further alteration and improvement in NO
x emission control technologies.
Current legislation limitations already strictly require combining aftertreatment technologies. As the mainstream exhaust aftertreatment (EAT) technology route, the combination of a diesel oxidation catalyst (DOC) + a catalytic diesel particulate filter (CDPF) + selective catalytic reduction (SCR) + an ammonia slip catalyst (ASC) presents excellent performance on emissions control at the cost of limited decrease in thermal efficiency [
5]. Even though the current method has already achieved above 95% NO
x conversion, the EAT system scheme still requires further adjustment and optimization in schemes and strategies for ULN emission regulation. Zavala et al. [
4] proposed different aftertreatment configurations for ULN emission control, with light-off selective catalyst reduction (LO-SCR) applied close to the engine to take advantage of the higher exhaust temperatures.
SCR is the critical device in the EAT De-NO
x progress. The Cu-zeolite catalyst is a common choice to meet current regulations. Metkar et al. [
6] studied the effects of the Fe-ZSM-5 and Cu-CHA catalysts on the NO
x reduction performance of NH
3, gave the kinetic parameter values for Cu-CHA catalysts, and established a global kinetic model, which can better predict the conversion rate of NO
x and NH
3. Mohan et al. [
7] reviewed the performance of NH
3-SCR using Cu-based catalysts in reducing NO
x at low temperatures and compared the ability of several Cu-based catalysts to reduce NO
x (standard SCR reaction) in different temperature ranges. Among them, Cu/SSZ is currently the most suitable catalyst for low-temperature reduction of NO
x, and the NO
x removal rate can reach 100% when the temperature is between 150 and 350 °C. When the temperature is lower than 150 °C, the NO
x reduction activity of Cu/SSZ drops sharply. Lei et al. [
8] studied the effect of exhaust temperature on the efficiency of selective catalytic reduction (SCR) denitrification. Lei summarizes that the change in SCR denitrification efficiency generally includes three states based on exhaust gas temperature. In state I, the SCR carrier temperature is high and urea is injected, resulting in high NO
x conversion efficiency (>90%); state III is a cold-start condition with the lowest NO
x conversion efficiency (<50%). State II is a transitional stage involving ammonia storage. The NO
x conversion efficiency is linearly related to exhaust temperature and increases with increasing temperature; the slope of the NO
x conversion efficiency decreases with increasing exhaust temperature. Gholami et al. [
9] reviewed different NO
x emission-reduction technologies and introduced the chemical reaction mechanism of NH
3-SCR (standard SCR) under aerobic and anaerobic conditions. Xie et al. [
10] conducted experimental research on the NO
x conversion efficiency and NH
3 leakage of three aftertreatment schemes by introducing SCR size strategies. Xie used postprocessing systems with different SCR lengths of the same SCR diameter and found that the nitrogen oxide emissions of the three SCR systems were similar, but the average NH
3 leakage varied greatly. As the SCR length decreased, the average NH
3 leakage increased. Ciardelli et al. [
11] proposed a novel catalytic mechanism for the fast SCR reaction of NH
3, NO, and NO
2 at low temperatures and supported a theoretical basis for fast SCR-related research. Yang et al. [
12] studied the effect of fast SCR reaction on commercial SCR catalysts and found that fast SCR can apparently promote catalytic activity and decrease catalyst consumption and replacement frequency.
However, Cu-based catalysts may generate N
2O and have little capability to clarify it. Yao et al. [
13] suggested reducing the amount of Cu in Cu-zeolite SCR to reduce N
2O generation without increasing NO
x emission. Kim et al. [
14] examined the Fe-zeolite catalyst and achieved significant suppression of N
2O formation. Zhang and Yang analyzed the formation pathways of N
2O in a combined zeolite-supported SCR with both Fe and Cu catalysts. The later research of Sharp et al. also selected this method for better N
2O reduction [
15].
The DOC is applied to oxidize CO, HC, and other combustible emissions of diesel engines [
16]. A DOC coupled with DPF or CDPF has been widely applied for particulate matter clarification [
17]. The oxidization process within DOC also affects NO, which can benefit the passive regeneration in downstream CDPF [
18] and SCR. As the EAT thermal management largely focused on the DOC, the exhaust temperature and constituents of NO
x, mainly NO and NO
x, will also affect the NH
3–NO
x reactions of downstream SCR [
19]. Nova et al. [
20] suggested that, compared to other NO
2/NO
x ratios, the efficiency of the SCR reaction can be promoted to the greatest extent when the NO
2/NO
x ratio reaches 50%. Based on the inherent characteristics of high NO emission (more than 90%) of diesel engines, a feasible solution to improve De-NO
x efficiency and decrease urea water solution consumption is increasing the SCR inlet NO/NO
2 ratio to 1:1 [
21]. Li et al. [
22] proposed that the optimal ratio of NO
2 to NO
x in SCR may be 1:2, at which the nitrogen removal efficiency and the amount of N
2 generated are maximized, with relatively less N
2O produced. By increasing the ratio of NO
2 to NO
x, the NO content at the outlet gradually decreases, while the N
2 content first increases and then decreases and the N
2O content increases.
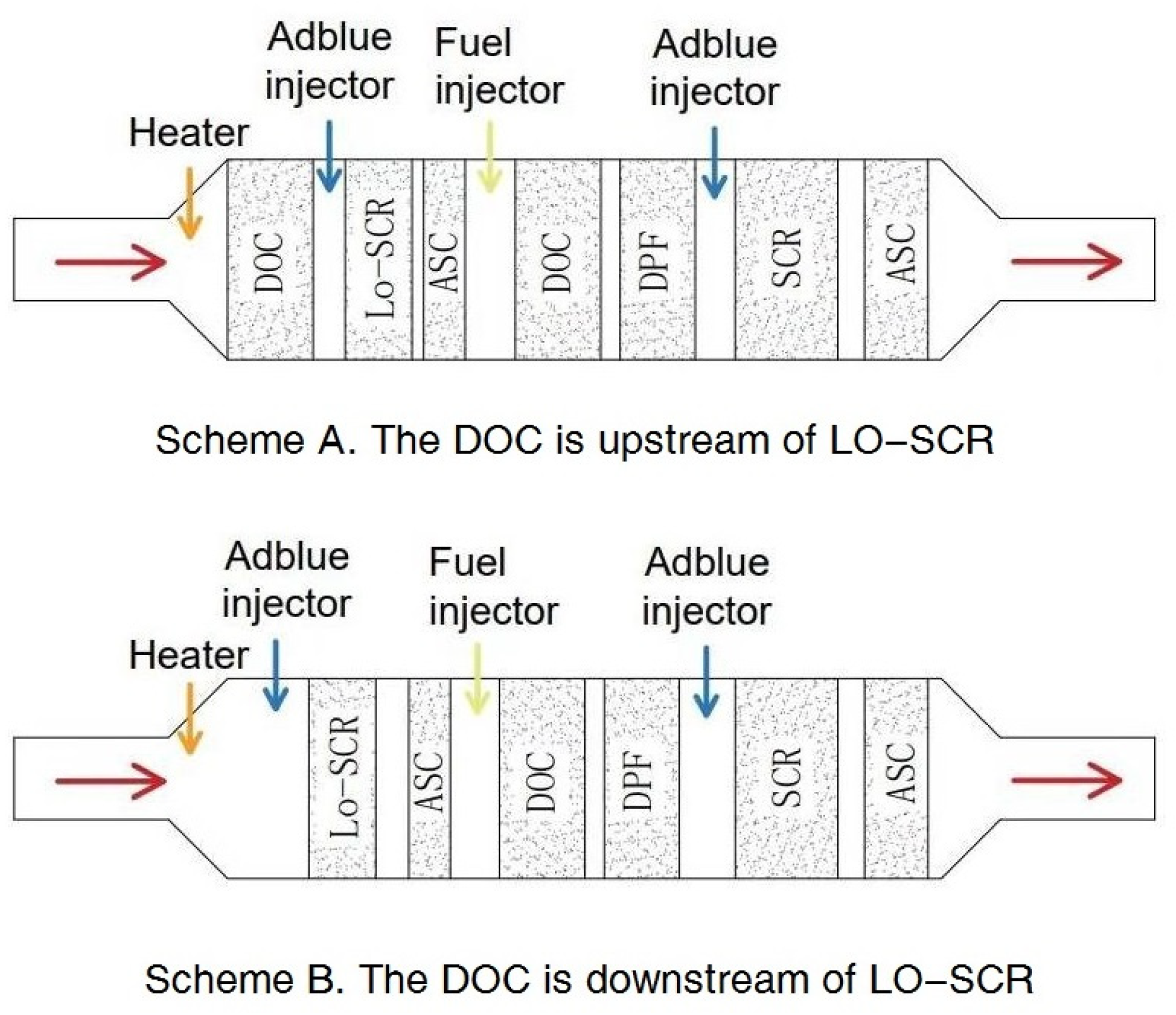
The relative location of the DOC, whether upstream (scheme A in
Figure 1) or downstream (scheme B in
Figure 1) of LO-SCR, also requires consideration. A DOC deployed upstream provides the possibility to partially oxidize NO into NO
2 and increase the proportion of fast reactions. However, at lower exhaust temperatures, a higher NO
2 concentration will increase N
2O generation, [
23], while, at higher exhaust temperatures, possible NO
2 surplus caused by an increased generation rate in the DOC will further hinder the NH
3–NO
x reactions in SCR. Another problem for the upstream DOC scheme is its effect on exhaust temperature; at cold start conditions, the cold DOC upstream will delay the catalyst temperature increase of LO-SCR [
4].
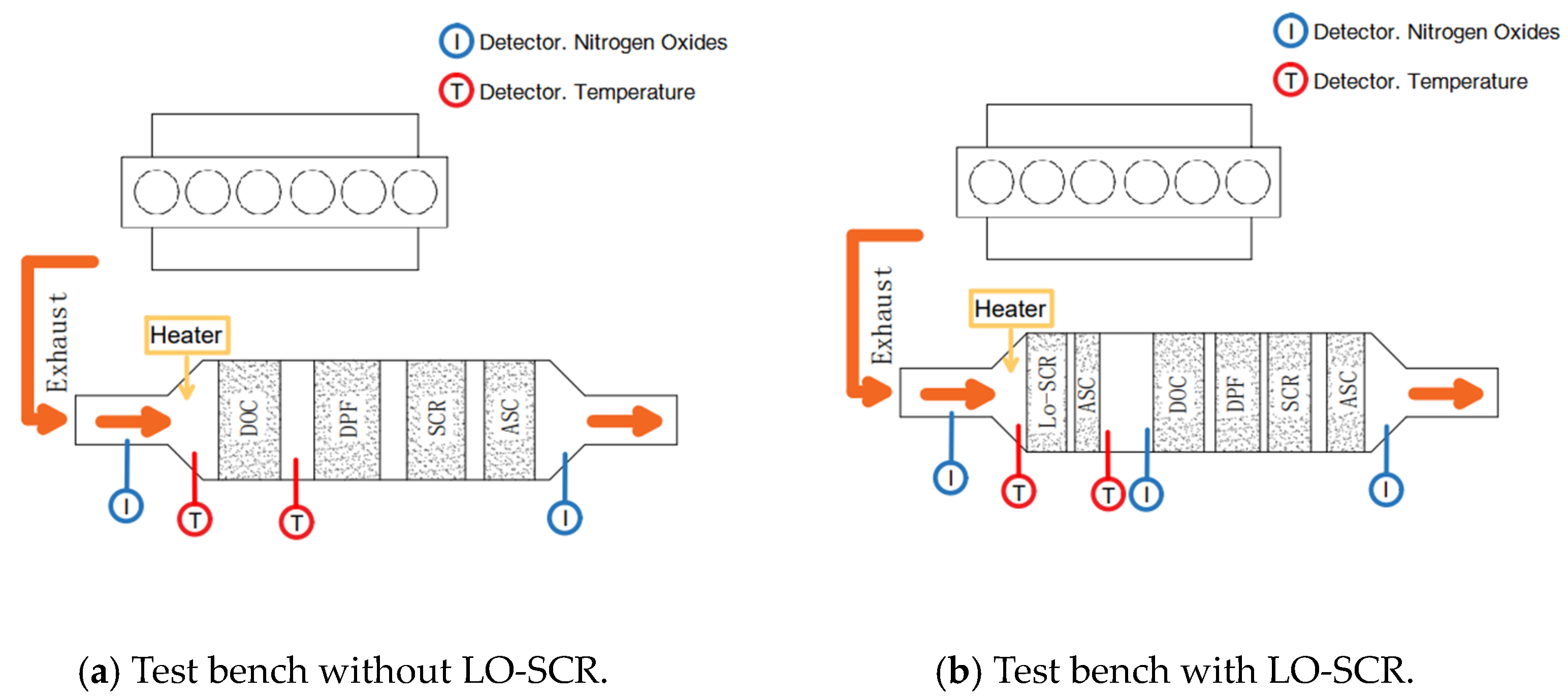
Therefore, to achieve ULN emission control at mid to low load, the EAT thermal management strategy requires detailed analysis. This work focused on the NH3–NOx reactions in LO-SCR and related thermal management strategy optimization. Two major aspects were evaluated in this work: the effect of thermal management strategy on exhaust parameters before SCR and the effect of thermal management strategy on LO-SCR performance at ULN emission control. The former was analyzed through a diesel engine EAT bench test, focused on exhaust temperature increase with an electric heater and NO oxidization in a DOC. The latter was evaluated through simulation, mainly for the comparison of different EAT system schemes, and the final Lo-SCR performance with the optimized thermal management strategy. This paper can be a reference for ULN emission control at mid- to low-load working conditions.
4. Discussion
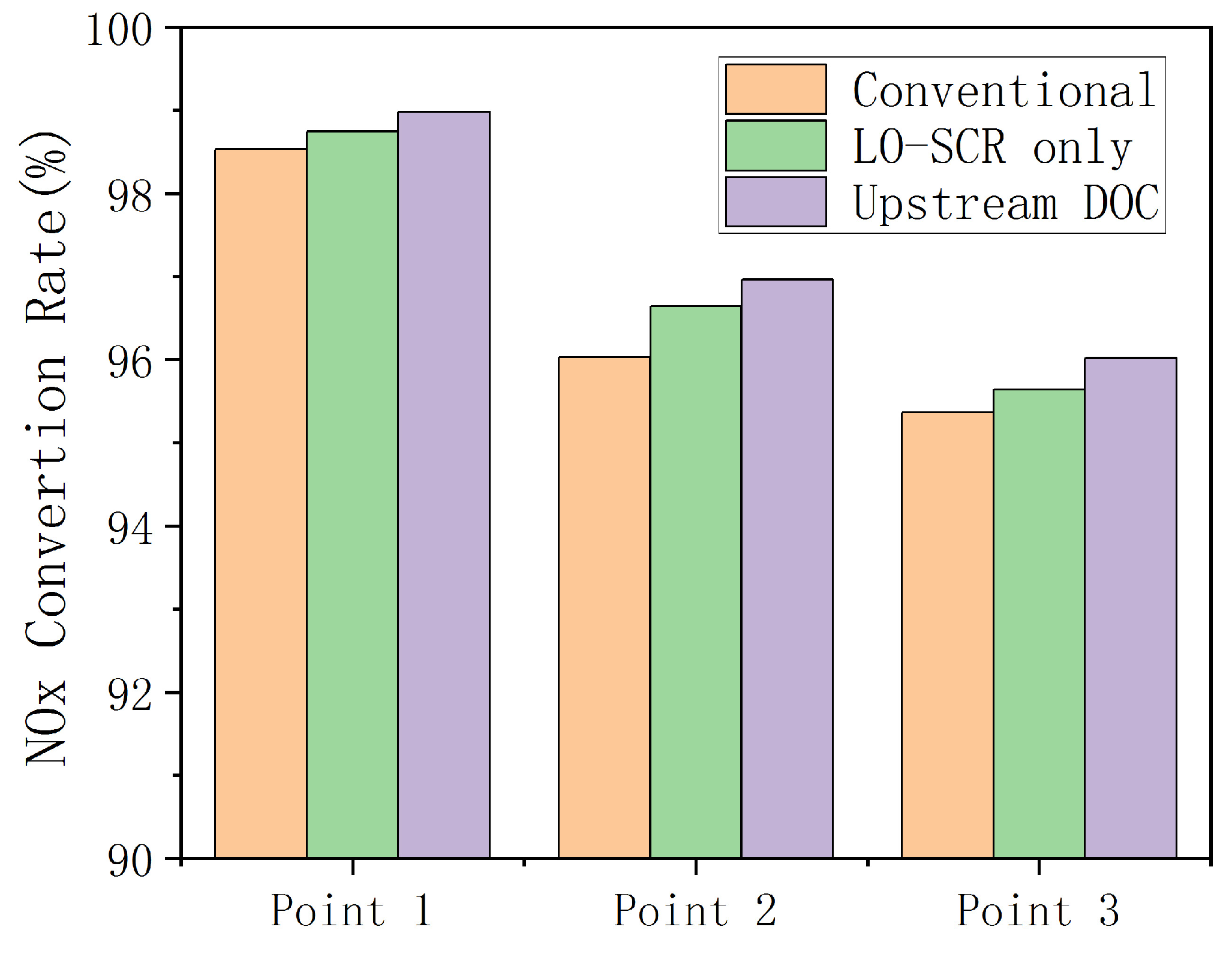
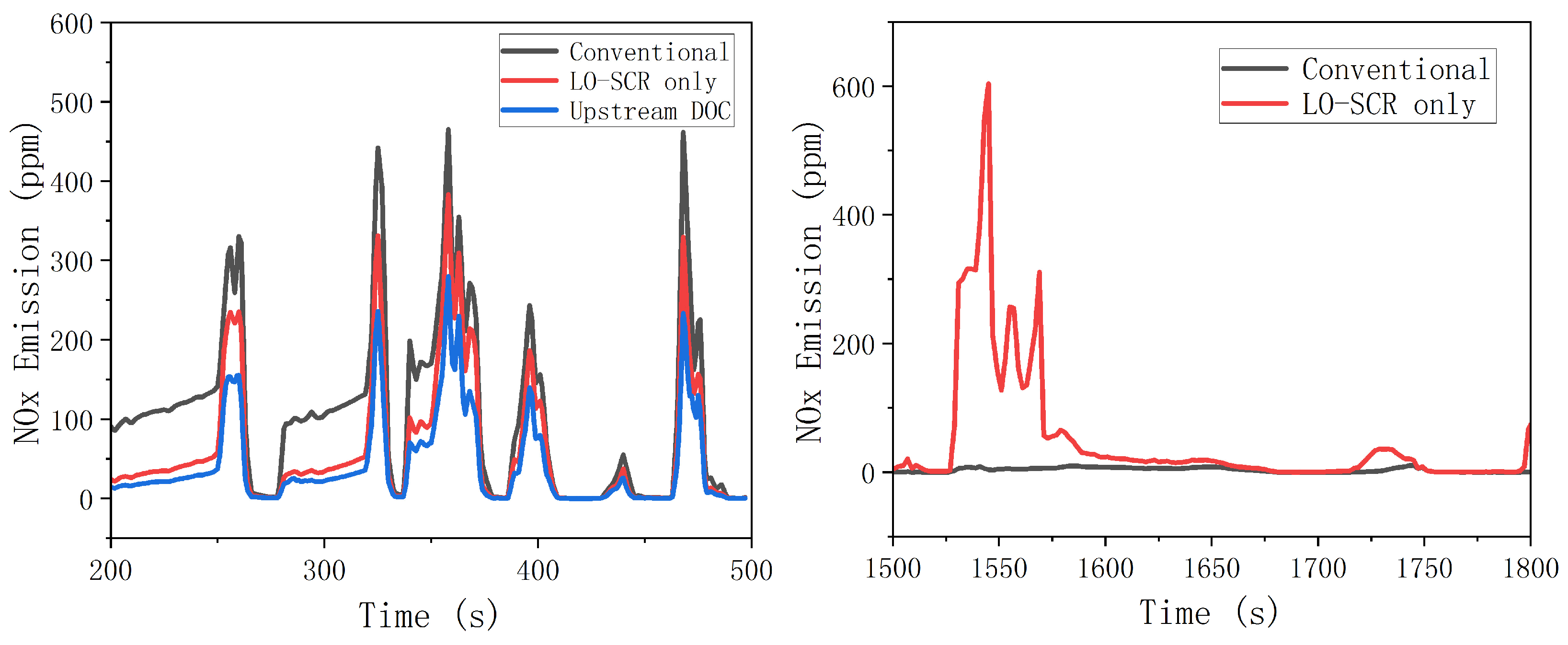
To effectively reduce NO
x emission at mid- to low-load working points, an electric heater was applied at SCR upstream. During the entire WHTC bench test, the exhaust was heated before entering the EAT system to improve the activity of the SCR catalyst at a lower exhaust temperature. The simulation result is shown in
Figure 13. Increased temperature has improved the activity of cold-start SCR catalyst, and the effect of thermal management on NO
x emission is significant. During 200~500 s of WHTC test, after electrical heating, the average NO
x emission of the LO-SCR system is 61.5 ppm, a reduction of 45% compared to the value without electrical heating before. The effect of upstream DOC is also moderately positive, as the NO
x emission decreased by 63%, reaching 41.9 ppm. However, the effect of DOC will inevitably hinder the temperature increase of LO-SCR during the early stage of cold start. With higher exhaust temperature, the DOC before LO-SCR may generate NO
2 in a larger proportion, which will increase the possibility of N
2O generation. The moderate positive effect is insufficient to compromise these two concerns.
From 1500 to 1800 s, there was a significant difference in nitrogen oxide emissions between the EAT system with LO-SCR and the conventional arrangement system of 1500~1600 s. The increase in average exhaust temperature caused by thermal management increases the reaction rate between ammonia and oxygen, resulting in the increase in the ammonia chemical loss. If the urea input is not adjusted after thermal management, the ammonia storage is insufficient at 1500 s, which, in turn, leads to significant NOx emission. The problem can be largely avoided by proper urea input adjustment.