1. Introduction
The management of acute and chronic wounds has a significant financial, clinical, and social impact on healthcare and affects the living conditions of over 2.5% of the population. Understanding the wound’s etiology, chronicity, healing mechanism, and biology is necessary for providing ideal wound care [
1]. Despite the numerous treatments already established for both chronic and acute wounds, their management continues to constitute an urgent and burdensome issue in modern times, necessitating the development of novel therapies.
Injuries have always been difficult to treat and continue to put a tremendous strain on the medical system. Over 8 million Americans suffered from wounds in 2014, with an estimated USD 30 billion in medical expenses. High-risk comorbidities rise in tandem with an aging and obese population, with the wound closure product market expected to reach USD 21.4 billion in 2022 with a compound annual growth rate of 4.15% from 2023 to 2030 [
2].
The goal of commercially accessible biomaterials for wound healing is usually symptom treatment (infection, pressure reduction, scarring, moisture balance, fluid exudation, etc.). On the other hand, sophisticated biomaterials for wound healing are being created to offer biophysical cues inspired by extracellular matrix (ECM) and immune response modulation for sufficient inflammation resolution. These treatments may combine the use of pharmaceutical and biological product therapies, and they are usually designed as injectable or biomaterial-based delivery systems. Basic research has demonstrated how biophysical signals can be included into biomaterials to regulate the activity of cells. Systems of delivery based on biomaterials, such as hydrogels, can offer stimuli-responsive and sustained-release characteristics. These ideas could further encourage patient adherence to novel treatments and get around the drawbacks and hazards of systemic delivery [
2,
3].
Numerous studies have suggested that fish oil supplements may lessen the severity of a number of skin conditions, including melanogenesis, dermatitis, allergies, photoaging, and skin cancer. The correlation between fish oil and skin protection and homeostasis has garnered significant attention, particularly with the omega-3 polyunsaturated fatty acids (PUFAs), namely docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA). The two main ways that polyunsaturated fatty acids (PUFAs) reduce cutaneous inflammation are by competing with inflammatory arachidonic acid and by preventing the synthesis of proinflammatory eicosanoid molecules [
4].
PUFAs in fish oil, on the other hand, may function as regulators to change the synthesis and activity of cytokines for promoting wound healing. Proinflammatory cytokines are mostly responsible for the initiation and amplification of the cellular and molecular processes that occur during the inflammatory phase of wound healing. PUFAs have the ability to control the production and function of cytokines. It has been demonstrated that these fatty acids are essential for the formation of cell membranes and anabolic processes that occur during skin tissue regeneration. The local inflammatory response at wound sites may be modulated or enhanced by omega-3 and omega-6 polyunsaturated fatty acids, which could hasten the healing process [
5]. Using the most recent research, this study attempts to provide a bibliographic survey on eicosapentaenoic acid and its therapeutic properties.
Methodology: The academic literature related to this specific bioactive compound and its potential wound healing effect were studied. The literature search was based on scientific databases, including Scopus, PubMed, Science Direct, and Google Scholar, mainly from 2017 to 2024. A total of 66 articles, including research papers and book chapters, were downloaded, of which 32 more recent articles on closer relevance to the topic were selected for writing the review. The keywords used were eicosapentaenoic acid, wound healing, skin, diabetes, and in vivo.
We included pre-clinical and clinical studies and we avoided including articles that were more focused on the biochemical mechanism of action than the results of the administration of EPA.
We think it is worth searching the literature during the last seven years (2017–2024), since we found enough articles relevant to the clinical application of ERA, since the support of wound healing and many dermatological problems by supplements is a trend of the recent years.
2. Skin and Its Healing Mechanisms
The skin, which covers 1.85 m
2 of surface area on average, is the biggest organ in the human body. It is the natural barrier against pathogenic microbes, extreme temperature, mechanical damage, toxic agents, and UV radiation [
6]. According to their impact and underlying causes, skin wounds are divided into acute and chronic categories [
7]. In contrast to chronic wounds, which are challenging to heal, acute wounds typically heal in 4–12 weeks with the recovery of the functional and anatomical integrity of the skin. Chronic wounds take longer to heal, a fact that increases the risk of infection and hinders the full and effective restoration of skin integrity. Adults usually exhibit scarring rather than total tissue regeneration as a result of persistent or deep wounds.
The epidermis, dermis, and subcutaneous adipose tissue, which run from the outer to the inner skin layer, must cooperate to perform particular sequences to foster healing when the skin is wounded. There are four stages of healing, which include hemostasis (I); inflammation (II); proliferation (III); and granulation tissue creation, re-epithelialization, and (IV) remodeling, which take place in a certain order or overlap.
Trauma causes the wounded blood vessels to constrict first, followed by the activation of platelets to form a clot. The clot seals the wound temporarily, preventing the spread of germs, and serves as a scaffold for arriving inflammatory cells [
8]. Damaged epithelial and endothelial cells, along with the clot, generate several chemotactic substances that draw in inflammatory cells to commence the following phase of healing. After hemostasis, the inflammatory phase follows, where immune cells travel to the site of the wound as a reaction to damage signaling factors to ward off microbial infection [
7].
At the location of the wound, there is an inflow of various inflammatory cells, that consequently produce a range of mediators and cytokines that facilitate angiogenesis and re-epithelialization. Hemostasis, chemotaxis, and enhanced vascular permeability are the characteristics of the inflammatory phase, which limit additional damage, seal the wound, cleanse cellular debris and microorganisms, and accelerate cell migration [
9]. The clot thrombus is instantly inhabited by neutrophils as the first line of resistance against pathogens. Within 48–96 h of injury, monocytes are drawn in to change into tissue-activated macrophages in the damaged area. To fight self- and foreign antigens, the adaptive immune system, which consists of Langerhans cells, dermal dendritic cells, and T cells, is also activated [
8].
Acute phase signals are first presented to skin cells, which are then recognized by toll-like receptors that start and maintain inflammation. To make it to the injury site, leukocytes, most notably neutrophils, migrate along a gradient of chemokines that keeps growing. Additionally, neutrophils release a variety of pro-inflammatory cytokines which boost the inflammatory response [
6]. Apoptosis, or necrosis, is followed by macrophage engulfment, or efferocytosis, to start the process of clearing out neutrophils. Neutrophils must be eliminated quickly since their removal initiates the process of inflammation resolution [
8]. The adaptive immune system includes activated regulatory T cells. Regulatory T cells, in addition to leukocytes, have the ability to reduce tissue inflammation by lowering the production of interferon-γ as well as the accumulation of proinflammatory macrophages. Macrophages are one of the significant factors in the progression from inflammation phase to proliferation. Pathogen-specific and damage-related molecular patterns activate macrophages. This causes their differentiation into the M1 subset, which is linked to phagocytic activity, scavenging, and also the synthesis of pro-inflammatory mediators, in the early stages of wound repair. Later, M1 changes into the M2 subset, displaying reparative characteristics. M2 macrophages play a role in fibroblast proliferation, the creation of the extracellular matrix (ECM), the synthesis of anti-inflammatory mediators, and angiogenic procedures. M2 macrophages operate as a sort of cleanup crew by phagocytosing neutrophils, bacteria, and cellular debris, eliminating additional damage to the wound site late in the healing process [
6]. The inflammatory phase, which typically lasts from one to four days following injury, clears the wound of debris, and performs significant phagocytosis to prepare it for regeneration. Various cells, including fibroblasts, keratinocytes, and endothelial cells, are recruited by growth factors and cytokines released by immune system cells throughout the inflammatory phase to kick off the proliferation phase [
7]. This stage’s duration might be several weeks [
9]. Fibroblasts, the ECM creation for granulation tissue formation, the proliferation and migration of endothelial cells (angiogenesis), and the repair of the epidermal barrier by means of wound re-epithelialization all participate in the process of skin tissue remodeling.
The ECM components created by fibroblasts’ proliferation and formation of granulation tissue serve as a substrate for the development of new blood vessels [
7]. Macrophages, fibroblasts, blood vessels, and a loose extracellular matrix consisting of type I collagen, glycoprotein, fibronectin, and hyaluronic acid compose the granulation tissue. Re-epithelialization caused by keratinocyte differentiation and proliferation covers the wound with overlying epidermal tissue. Following this stage is the remodeling or maturation phase, during which the granulation tissue is replaced by type I collagen and various skin components, i.e., sweat glands, sebaceous glands, and hair follicles [
7]. To provide the skin with better tensile strength, during this phase, which starts around the third week and can last up to 12 months [
9], fibroblasts drop into myofibroblasts and type III collagen transforms into type I collagen [
7]. The tensile strength reaches its peak after 11 to 14 weeks. Only around 80% of the original tensile strength remains in the resulting scar, which means it will never regain its full original tensile strength [
9]. In the case of acute wounds, wound healing occurs when all of these stages have been completed. Chronic wounds, however, do not go through all of these stages, and the healing process is impeded [
10].
2.1. Sources of Eicosapentaenoic Acid, Structure, and Physico-Chemical Properties
The polyunsaturated fatty acid (PUFA) class of omega-3 fatty acids includes eicosapentaenoic acid (EPA) (
Figure 1). Humans can consume EPA through their diets by eating fatty fish, different kinds of edible algae, or by supplementation with fish oil or algal oil. Cod and plaice oil are the fish oils with the greatest concentrations of EPA (
Table 1). Alpha linolenic acid (ALA), which is absorbed by the body, is partially converted into EPA. Humans require a sufficient amount of ALA because it is a necessary fatty acid. However, the efficiency of converting ALA to EPA is significantly less than the absorption of EPA from food intake [
11].
The membranes of cells contain EPA. EPA forms eicosanoids during an inflammatory reaction by being metabolized by the cyclooxygenase and lipoxygenase enzymes. Because EPA is used to metabolize docosahexaenoic acid (DHA) as its precursor, which requires additional metabolic energy to produce, it is important to ensure that there is an adequate amount of EPA in the diet. Eicosanoids produced from EPA are generally strong inducing agents of vasoconstriction, inflammation, and blood coagulation [
12].
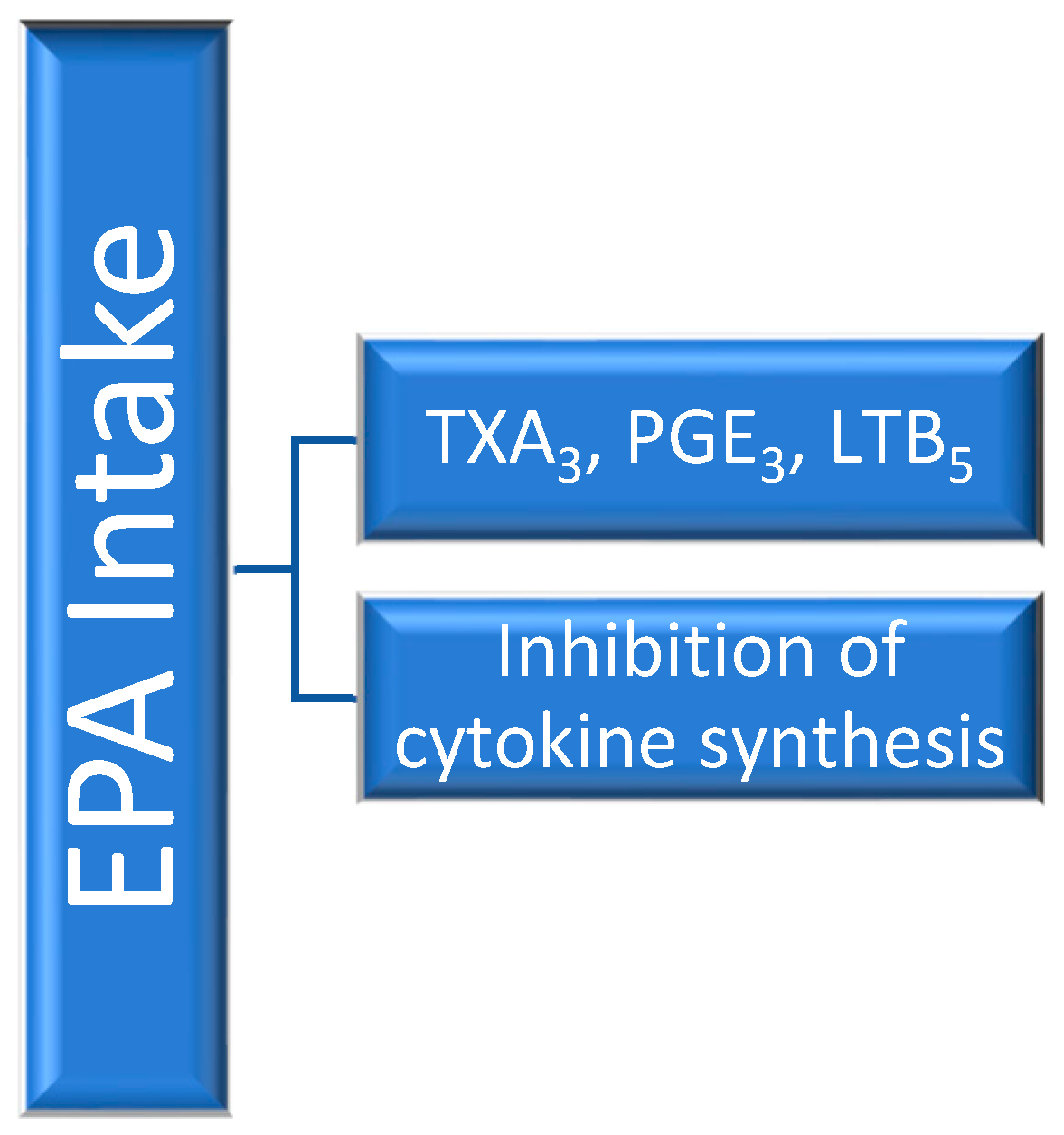
Thromboxane (TX)A3 derived from EPA is weakly proaggregatory when compared with the arachidonic acid (AA)-derived TXA2. Also, the influence of pro-inflammatory PGE2 and the leukotriene (LT)B4, which is a chemoattractant and activator of neutrophils, is affected by the presence of EPA. Although PGE3 has a similar action as PGE2, it is produced with low efficiency. LTB5 has little inflammatory activity when compared with LTB4.
Of particular interest are findings on the highly inducible COX-2 which is involved in the overproduction of prostaglandins in inflammatory sites. EPA greatly decreased the interleukin-(IL-)1-induced COX-2 expression in endothelial cells. Although it is well documented that ω-3 fatty acids suppress cytokine production such as tumor necrosis factor-(TNF-)α and IL-1, the underlying mechanisms still remain unclear. It appears that interfering with the synthesis of TXA
2 and PGE
2 also affects cytokine production (
Figure 2) [
13].
2.2. Recent Studies on Skin
EPA has a healing effect, as proven by Eugenia Sofrona and her team. In that study, lipophilic and hydrophilic extracts of the marine isopod
Ceratothoa oestroides were produced, and their ability to promote wound healing after being applied topically to wounds induced on SKH-hr1 hairless mice was assessed. Measurements of skin biophysical parameters such as TEWL, hydration, elasticity, and skin thickness and histological examination were primarily used to evaluate wound healing daily. Using an EPA-rich fraction to treat wounds resulted in the most activity, according to the findings of the preliminary evaluation of the various extracts. Mice’s wounds entirely healed after being treated locally with the bioactive fraction, and the skin completely returned to normal without any signs of inflammation [
14].
Recently, Vitsos A. et al., studied the healing efficacy of composite dressings containing Pycnogenol™ and/or
Ceratothoa oestroides extracts, with the goal of improving their efficiency and stability while applying a low-temperature vacuum technique. Surgical wounds were inflicted on SKH-hr2 hairless mice. The assessment of wound healing was mainly conducted by clinical and histological examination, with an Antera 3D camera and biophysical measurements. Dressings remained constant and did not affect the
C. oestroides extract’s medicinal qualities. As a standard product, all interventions were contrasted with the
C. oestroides ointment. By the fifteenth day, the majority of the wounds treated with the reference formulation and the
C. oestrodes dressing had closed, with histological scores of 6.5 and 7, respectively. Considering the formulations that contained
C. oestroides olive oil extract, the results showed that, in comparison to the control, the extract improved the healing process [
15].
Karoud et al., reported that the usually wasted hake heads from
M. merluccius might be used as a unique raw material for the production of oil. This fish oil is a good source of n-6 PUFAs, including arachidonic acid, and n-3 PUFAs, especially EPA and DHA. This study presented data on the anti-inflammatory and antioxidant activities of hake head oil (HHO). Using a round seal, a uniform wound area measuring 1 cm in diameter was created on the mouse’s previously cleaned and shaved back. Three mouse groups, each with six mice, were given the appropriate treatment: Group I received sterile saline treatment and was assigned as the negative control. Group III received a topical application of 300 mg/kg of hake head oil (HHO), and Group II received a topical application of 0.13 mg/mm
2 of CICAFLORA
® (comparison medication), designated as the reference group. Every two days, HHO and the reference medication, CICAFLORA
®, were administered until the wound had healed completely. When compared to the negative control, the HHO- and CICAFLORA
®-treated groups showed appreciable evolutions in wound contraction rates (
p < 0.05). The HHO-treated group showed the strongest healing contraction rate during the cicatrization process as compared to the CICAFLORA
®-treated group. In fact, on the sixth day, they observed a reduction in the wound area for HHO and CICAFLORA
® of 55.66% and 67.89%, respectively. In actuality, the HHO-treated group showed 100% wound healing at the conclusion of the experiment, compared to only 80% for the CICAFLORA
®-treated group. The abundance of n-3 and n-6 polyunsaturated fats in this oil, along with its antioxidant and anti-inflammatory qualities, may be contributing factors to HHO’s potential to heal wounds [
16].
Ontoria-Oviedo et al., examined the impact on wound healing of a marine lipid concentrate developed from anchovy and sardine oil that is high in SPMs (specialized lipid mediators), EPA, and precursor substance DHA (docosahexaenoic acid), demonstrating the healing power of EPA. The marine lipid concentrates enhanced wound closure in vitro, decreased the number of proinflammatory macrophages (M1), and demonstrated biocompatibility with keratinocytes and fibroblasts. In this study, daily administration of EPA-rich marine oil to open wounds in db/db mice accelerated the healing process by reducing inflammation, promoting neoangiogenesis, and polarizing M1/M2 macrophages. This process improved wound closure [
17].
In a recent study, the effect of lipid derivatives from EPA and DHA on burn wound healing was investigated. In Yorkshire pigs, after partial and full thickness burn wounds were created, punch biopsies were collected at various time points during treatment and analyzed for measurements of approximately 45 EPA and DHA derivatives. The results showed nine lipid mediators of EPA and DHA to be elevated on the seventh day of treatment in full-thickness burns, thus suggesting the healing effect of these fatty acids on wounds [
18].
2.3. Oral Consumption
As EPA belongs to the omega-3 fatty acids, which partake in many functions of the body, and has recognized beneficial properties, it is reasonable to see why it is a component of many food supplements. EPA is available in fish oil capsules, along with DHA [docosahexaenoic acid]. The majority of commercially available dietary supplements are made from fish oil and are often offered as triglycerides, ethyl ester, or phospholipid EPA.
McDaniel J. C. and his team, taking advantage of the healing effect of EPA, compared the efficacy of EPA + DHA fish oil capsule therapy with a placebo in reducing polymorphonuclear leukocyte activation in chronic venous leg ulcers. In this study, 40 patients with chronic venous leg ulcers were randomly assigned to either placebo [Control Group] or EPA + DHA supplementation. Polymorphonuclear leukocyte (CD15) and activated polymorphonuclear leukocyte (CD66b) markers and protease levels were measured on days 0, 28, and 56 of treatment. The collective findings concluded that oral EPA + DHA treatment can modulate the activity of polymorphonuclear leukocytes and facilitate the healing of chronic venous leg ulcers [
19].
The same team of researchers also investigated a novel oral therapy containing eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), two bioactive components of fish oil, to target and reduce the elevated amounts of activated polymorphonuclear leukocytes in the site of the wound that keep chronic venous leg ulcers “enslaved” in a chronic inflammatory state. In this clinical research, 248 adults over 55 years of age with chronic venous leg ulcers were divided into two groups and randomly assigned to be given either EPA + DHA (1.87 g/day EPA + 1.0 g/day DHA) or placebo daily for 12 weeks. Then, during the treatment, they examined factors such as reduction in the wound area, active polymorphonuclears in the blood, lipid metabolites, etc., in the patients. The results provided new evidence on the efficacy of EPA + DHA treatment, which may facilitate healing and thus be a novel adjunctive therapy for chronic venous leg ulcers in the aging population [
20].
Soleimani et al. examined the benefits of flaxseed oil supplementation with additional omega-3 fatty acids on wound healing and metabolic status in patients with DFU focused on healing such ulcers. Patients for the study were divided at random into two groups and given either 1000 mg of flaxseed supplements containing omega-3 fatty acids [EPA and DHA] or a placebo twice daily for 12 weeks. When compared to the placebo group, the supplement group exhibited a substantial decrease in dimensional extent of the ulcer. Significant decreases in insulin resistance and blood insulin levels were seen. Overall, measurements of diabetic foot ulcer size and indicators of the metabolism of insulin benefited from flaxseed supplementation with omega-3 fatty acids [
21].
Previous research has indicated that the decreased production of growth factors such as platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), keratinocyte growth factor (KGF), and excess protease activity may be associated with potential etiologies for impaired wound healing in diabetes. A crucial part of diabetic wound healing is played by mast cells. Mast cell degranulation can be influenced by omega-3 fatty acids in a dose-dependent manner. Babaei et al. investigated the association between the presence and degranulation of mast cells in the diabetic wound region and omega-3 fatty acids. After shaving their backs, the rats had an extensive wound (20 mm × 20 mm) that measured and reached the level of the panniculus carnosus muscle. Omegaven (Fluka/Sigma-Aldrich
®, St. Louis, MO, USA) was administered intraperitoneally to the experimental group (
n = 35) twice a day at a dose of 1 mL per rat (100 mL of Omegaven contains 2.82 g of EPA and 3.09 g of DHA). Before the trial began, administration was initiated intraperitoneally at a dose of 1 mL once daily. This was continued until the conclusion of each analysis (biomechanical test and histologic examination). When comparing the wound area of experimental and control groups on day 7 and the quantity of grade three mast cells on days 3 and 5, it was shown to be significantly reduced by omega-3 fatty acids. It was also found that the wound strength significantly increased in the experimental group on day 15 [
22].
Notwithstanding the generally acknowledged health advantages of omega-3 fatty acids, particular research has indicated unfavorable outcomes of these substances in the process of wound healing. Burger et al. (2019) examined how oral eicosapentaenoic acid (EPA)-rich oil delivery affects mice’s ability to repair wounds. Following four weeks of supplementation with 2 g/kg of body weight with EPA-rich oil, mice’s serum concentrations of docosahexaenoic acid (DHA; 22:6ω-3) (33%) and EPA (20:5ω-3) were higher than those of control mice. The mice who were fed EPA also had skin that contained omega-3 fatty acids. Compared to control mice, mice that received EPA-rich oil showed a delay in the healing process at the 3- and 7-day marks after wounding; however, this delay did not affect the overall time needed for wound closure. Following EPA-rich oil supplementation, collagen reorganization—which affects the integrity of the wound tissue—was compromised. These effects were linked to increased amounts of interleukin-10 (IL-10) in tissue during the early phases of wound healing, as well as twice as many M2 macrophages compared to control rats. Wound closure and collagen organization returned to normal in the absence of IL-10 (IL-10
−/− mice), indicating that the harmful effects of EPA-rich oil supplementation were caused by the overproduction of IL-10. To sum up, the quality of wound healing was compromised by oral administration of EPA-rich oil, but the wound closure time remained unaffected. This is probably because the anti-inflammatory cytokine IL-10 was elevated [
23].
Candreva et al. (2019) employed two distinct methods. Two types of mice were used: (1) FAT-1 transgenic mice that were able to produce ω-3 FA naturally; and (2) wild-type (WT) mice that were given DHA-enriched fish oil orally. Compared to WT mice, FAT-1 mice exhibited elevated levels of ω-3 FA, primarily docosahexaenoic acid (DHA), in both their systemic (serum) and local (skin tissue) samples. Three days following the formation of the wound, the cutaneous wound tissue of FAT-1 mice exhibited decreased IL-10 levels, increased myeloperoxidase (MPO) activity, and higher levels of CXCL-1 and CXCL-2. The presence of inflammatory cells, edema, and an elevated TNF-α concentration all contributed to the maintenance of inflammation. Isolated from FAT-1 mice, neutrophils and macrophages also produced lower levels of IL-10 and higher levels of TNF-α. On the final day of analysis, which was 14 days after the wound, the mice’s wounds were six times larger than those of the WT group due to a delayed wound closure. This was linked to structural features in the restored tissue as well as improper collagen fiber orientation. In a similar vein, the DHA group saw a late inflammatory phase delay. On the third day following skin injury, this group exhibited elevated TNF-α content and CD45+F4/80+ cells along with reduced concentrations of ω-6 FA metabolites and increased concentrations of key metabolites generated from ω-3, such as 18-HEPE. In conclusion, both the FAT-1 and DHA groups’ increased DHA content retarded the resolution of inflammation and decreased the quality of the skin tissue that healed [
24].
Burger et al. [
25] showed that EPA supplementation has a negative effect on diabetic mice’s ability to heal wounds due to decreased collagen gene expression, poor collagen organization, and a resulting decrease in the resistance of the repaired tissue, mechanistically, via activating PPAR-γ (peroxisome proliferator-activated receptors) [
19]. EPA supplementation increased neutrophils’ production of IL-10. Simultaneously, EPA and IL-10 decreased collagen expression and fibroblasts’ synthesis of TGF-β. The discovery that PPAR-γ blockage in vivo prevented the EPA-induced decrease in collagen organization could be the most significant. In more detail, EPA-rich oil reduced the ω-6/ω-3 ratio by increasing the incorporation of ω-3 and decreasing the ω-6 fatty acids, according to the gas chromatography study of serum and skin. Ten days after wounding, EPA induced neutrophils in the wound to produce more IL-10, decreased collagen deposition, and eventually postponed wound closure and compromised the quality of the recovered tissue. This effect was dependent on PPAR-γ. In vitro fibroblasts produced less collagen when exposed to EPA and IL-10. In vivo, topical PPAR-γ-blockade in diabetic mice mitigated the detrimental effects of EPA on collagen organization and wound closure. In diabetic animals given the PPAR-γ blocker topically, neutrophils produced less IL-10. These findings demonstrated how oral EPA-rich oil supplementation hinders the healing of skin wounds in diabetics by influencing both inflammatory and non-inflammatory cells (
Figure 3) [
25].
3. Discussion
EPA is widely known for the management or prevention of various heart-related diseases, such as blocked heart arteries [coronary artery disease] and heart attacks. It also reduces triglyceride levels in those with very elevated levels [
26]. It finds application along with the appropriate medicines, in the treatment of schizophrenia, personality disorders, Alzheimer’s disease, depression, and attention deficit hyperactivity disorder (ADHD), among other mental illnesses [
27]. In addition, it is used for other diseases such as psoriasis, asthma, cystic fibrosis, and diabetes as an adjuvant nutritional supplement in treatment. EPA helps in various kinds of cancer, mostly assisting in body weight maintenance and lessening the negative effects of chemotherapy in cancer patients [
28]. When used by women, it lessens the risk of delayed fetal development, high blood pressure during high-risk pregnancy, and menopause symptoms [
29]. In fish oil preparations, EPA and DHA are frequently combined for several disorders, such as avoiding heart disease and lowering irregular heartbeats. The idea that encapsulated EPA and DHA supplements improve cardiovascular outcomes in people with known cardiovascular disease or at risk of developing it has been tested over the course of the last 20 years in a number of clinical trials. Nevertheless, using EPA and DHA combination capsules to lower cardiovascular risk is not advised based on the information that is currently available. According to certain research, using EPA on its own may reduce the risk of cardiovascular disease [
30].
According to our review study, EPA is used as a supplement after surgery to lessen infections, accelerate wound healing, and speed up recovery [
31]. It has been demonstrated that fish oil supplements accelerate the rates at which wounds epithelialize and that omega-3 fatty acids change the inflammatory composition of wounds. It has been demonstrated that EPA and DHA modulate the rate at which wound healing proceeds. The physiology of the skin is changed at the cellular and molecular levels by these distinctive omega-3 fatty acids and their metabolites [
30]. On the other hand, there is some controversial data in the literature regarding the oral supplantation of omega-3-fatty acids [
30]. It has been observed that oral consumption of omega-3 fatty acids, particularly the DHA and EPA combination, significantly slow down the healing of wounds and disrupt the structure of collagen through several mechanisms [
5]. Omega-3 fatty acids work through a variety of mechanisms, such as modifications to the structure of membranes, the synthesis of lipid mediators, and the activation of intracellular receptors that modify the expression of genes. Omega-3 fatty acids stimulate the peroxisome proliferator-activated receptors (PPARs) and therefore could have negative impact on the healing of diabetic ulcers [
23,
25]. Characteristic genes of mast cells in the skin tissues of patients with type 2 diabetes mellitus (T2DM) were described at a single-cell level. These genes and enriched signaling pathways provide a theoretical basis and data support for further research on dermatopathy in patients with diabetes mellitus [
32]. Although the topical application of omega-3 fatty acids could improve wound healing, further research is needed regarding the influence of oral supplementation, particularly on diabetic ulcers.
A number of additional cutting-edge technologies are hitting the market. Among these are products such as Woundchek (DEN180014) (Systagenix) for detecting elevated protease activity as a proxy for impaired wounds; Cellutome (KCI) and Recell (BP170122) (Avita) for epidermal harvest and suspension systems; SofPulse (K070541) (Endonovo) for targeted pulsed electromagnetic therapy; TWO2 (WoundSource) for topical wound oxygen therapy; and UltraMIST (K1407828) (WoundSource) for ultrasound therapy. Numerous growth factors, including platelet-derived growth factor (PDGF), fibroblast growth factor, transforming growth factor-β, and epidermal growth factor, have been identified in the intricate environment of wound healing. Growth factors such as PDGF supplementation [Regranex (BLA103691) (Smith + Nephew)] have been developed as adjuncts treating persistent wounds, such as diabetic neuropathic ulcers, as a result of ongoing technological advancement [
2].
However, there are still a lot of obstacles to overcome in the treatment of both acute and chronic wounds, even with the creation of numerous new products. Fundamental knowledge of the pathophysiological mechanisms behind injury and the enhanced identification and comprehension of unmet clinical requirements are necessary. These discoveries will ultimately result in personalized medicine for the treatment of chronic wounds and will enable scientists to determine the optimal application for products with single molecular targets.