1. Introduction
Reinforced concrete, a common and widely used civil engineering material, is often employed in complex and harsh environments. Examples include coastal pump stations, hydroelectric power plant buildings, and cross-sea bridges. These structures frequently operate under the coupled effects of stray currents and chloride environments. When the chloride ion concentration in these service environments exceeds the critical threshold [
1], weak areas of the passive film in concrete structures are destroyed, forming localized pitting. As corrosion progresses, the relatively intact passive film surrounding these pits is also eroded, exposing a larger area of the substrate and accelerating metal phase corrosion.
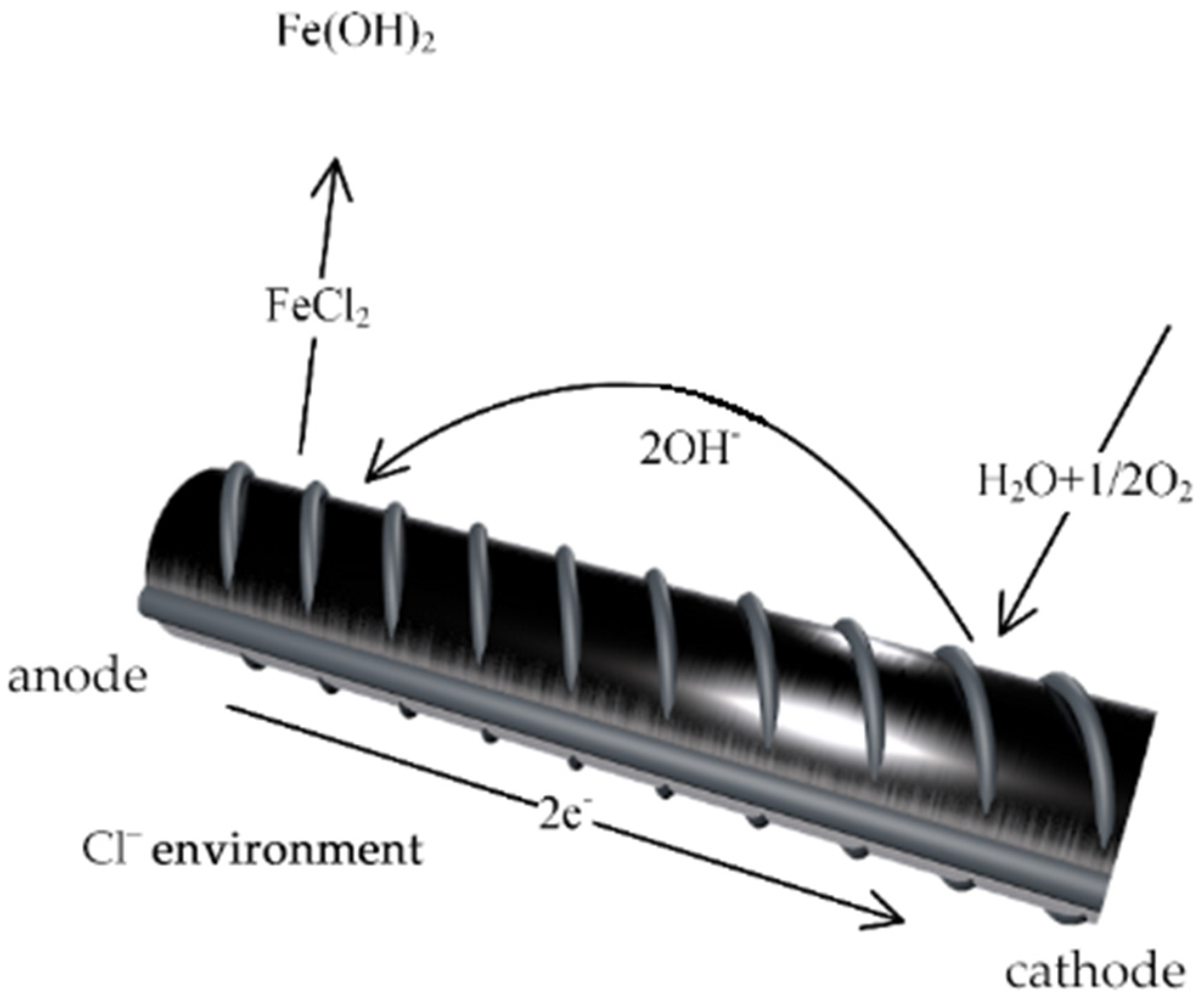
Figure 1 illustrates the chemical reactions of chloride ion attack on the metal phase in concrete materials [
2]. Additionally, chloride ions in reinforced concrete interact with one of concrete’s hydration products—monosulfate (AFm). This interaction promotes the formation of corrosion by-products, namely Friedel salts [
3]. This chemical reaction signifies ongoing corrosion activity. The accumulation of these salts leads to concrete volume expansion, thereby increasing the risk of damage to concrete structures.
Additionally, stray currents in service environments refer to electrical currents that do not flow along designed or specified circuits. These currents induce electrochemical corrosion [
4], affecting metals such as the reinforcing steel in civil engineering structures. When stray currents flow through reinforcing steel in concrete, areas with intact passive layers act as cathodes in the “corrosion cell”, undergoing hydrogen evolution corrosion and generating hydrogen gas. This gas, unable to quickly escape from the concrete, creates hydrostatic pressure in the corrosion area. Consequently, the metal phase separates from the surrounding concrete, significantly reducing the concrete’s grip and protective capacity on the metal. Conversely, areas where the metal has been depassivated expose the metal substrate, acting as anodes in the “corrosion cell”. The metal substrate oxidizes, producing substantial corrosion products that often increase the volume of the metal phase. For instance, the volume expansion caused by various oxide products from steel corrosion increases local stress in the concrete, damaging the protective layer around the reinforcing steel. Furthermore, the presence of stray currents accelerates chloride ion transport in concrete. The coupled effect of these two environmental factors poses a serious threat to the durability and safety of concrete structures [
5]. Therefore, studying the corrosive effects of stray currents and chlorides on reinforced concrete structures is of significant importance.
In practical engineering, corrosion inhibitors are widely applied as an economical and effective anti-corrosion measure. Their primary function is to suppress the bipolar reactions of metal electrochemical corrosion. Specifically, the inhibiting components react on the metal surface to form an adsorption film. This film elevates the corrosion potential or increases charge transfer resistance [
6], thereby mitigating corrosion effects under stray current and chloride ion attack.
Currently, environmentally friendly corrosion inhibitors are a research focus in this field. Corrosion inhibitors based on plant extracts [
7,
8] have significant potential for application. Academic reports have identified several plants with notable corrosion inhibition effects. These include perilla [
9], Spartina alterniflora [
10], orange peel [
11], green tea [
12] and so on. Ref. [
9] introduced a plant-based corrosion inhibitor extracted from perilla seeds. They employed HPLC-MS, FT-IR, XPS, and chemical computation methods. The results indicated the presence of two effective components in perilla seeds: luteolin and apigenin. The adsorption behavior conformed to Langmuir adsorption theory. The adsorption free energy was −22.7 kJ/mol. A higher absolute value of adsorption energy correlates with better corrosion inhibition effects. Pradipta et al. compared the corrosion inhibition effects of green tea extract and standard commercial Ca(NO
2)
2 inhibitor [
13] on reinforced concrete in mortar. At equivalent concentrations, both showed similar steel corrosion inhibition efficacy. In electrochemical tests, changes in the anodic and cathodic slopes of steel, within a corrosion cell, proved green tea extract to be a composite inhibitor. It formed a protective layer on the steel surface, enhancing its polarization resistance.
Xanthium sibiricum is a novel, environmentally friendly plant-based corrosion inhibitor that has received limited academic attention.
Xanthium sibiricum, as the fruit of a common wild plant, is mainly distributed in China and some other Asian countries and regions, making it widely available and easy to obtain. Its surface is covered with stiff, hooked bristles, and the color is generally brown. It is relatively hard in texture, and due to the presence of the bristles, the surface has an irregular texture. From a chemical perspective, it contains various alkaloids and flavonoids, which have antioxidant properties [
14]. The 1,5-dicaffeoylquinic acid it contains is considered a potential corrosion inhibitor component.
While research findings on biomass corrosion inhibitors are abundant, most studies remain focused on evaluating corrosion inhibition efficiency and investigating the mechanisms of action, with less emphasis on the practical engineering applications of biomass corrosion inhibitors. There is also limited consideration of the impact of corrosion inhibitors on the durability of concrete structures when they are incorporated. On the other hand, current research on the corrosion of reinforced concrete primarily concentrates on single ion or multi-ion exposure environments, with less attention given to studies of corrosion resistance and the exploration of chloride ion transport mechanisms under the combined effects of stray currents and high chloride salt environments. Therefore, this study first employed the natural potential method [
15], corrosion area [
16] and steel weight loss method [
17], and linear polarization method [
18] to verify the inhibitory effect of
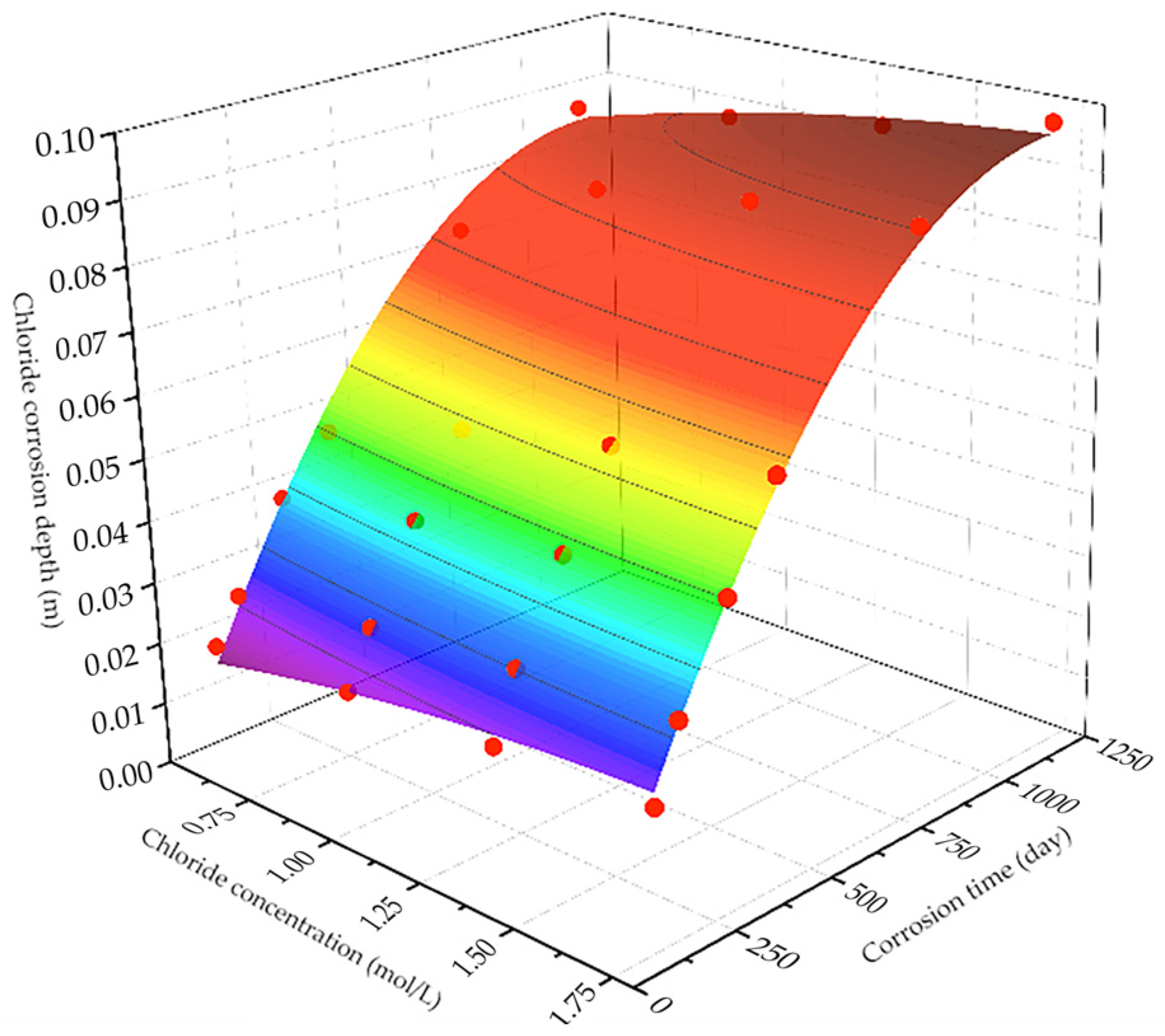
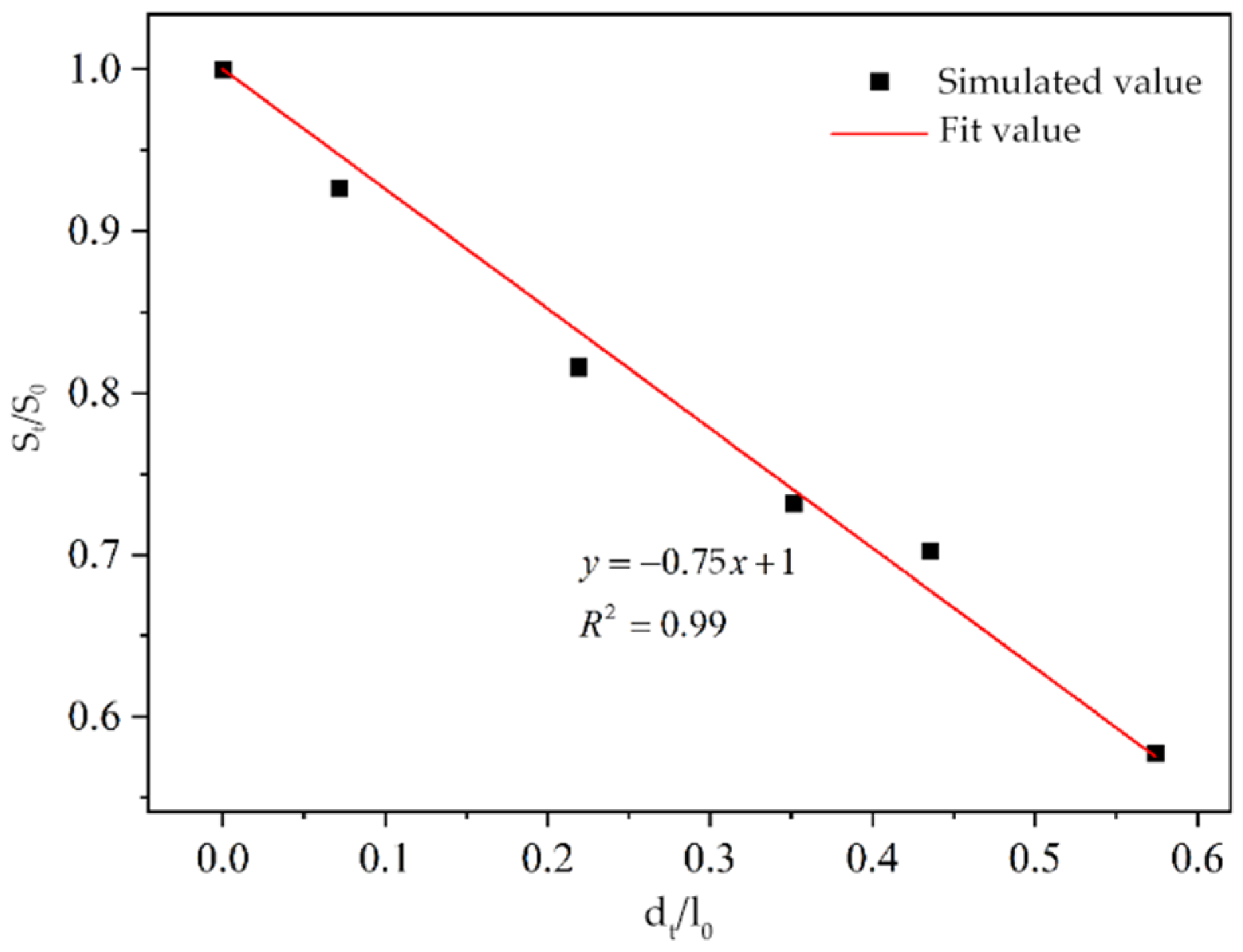
Xanthium sibiricum on steel in chloride-containing concrete. Subsequently, a two-dimensional multiphase ion transport model distinguishing coarse aggregates and cement mortar was constructed to simulate chloride ion transport in concrete under a stray current and chloride environment. Based on the simulation results, predictive formulas for the residual compressive strength of reinforced concrete were derived for various voltages and chloride ion concentrations after a specific corrosion period. Finally, field tests were conducted on an actual project operating in similar environments. These tests determined the optimal dosage of the
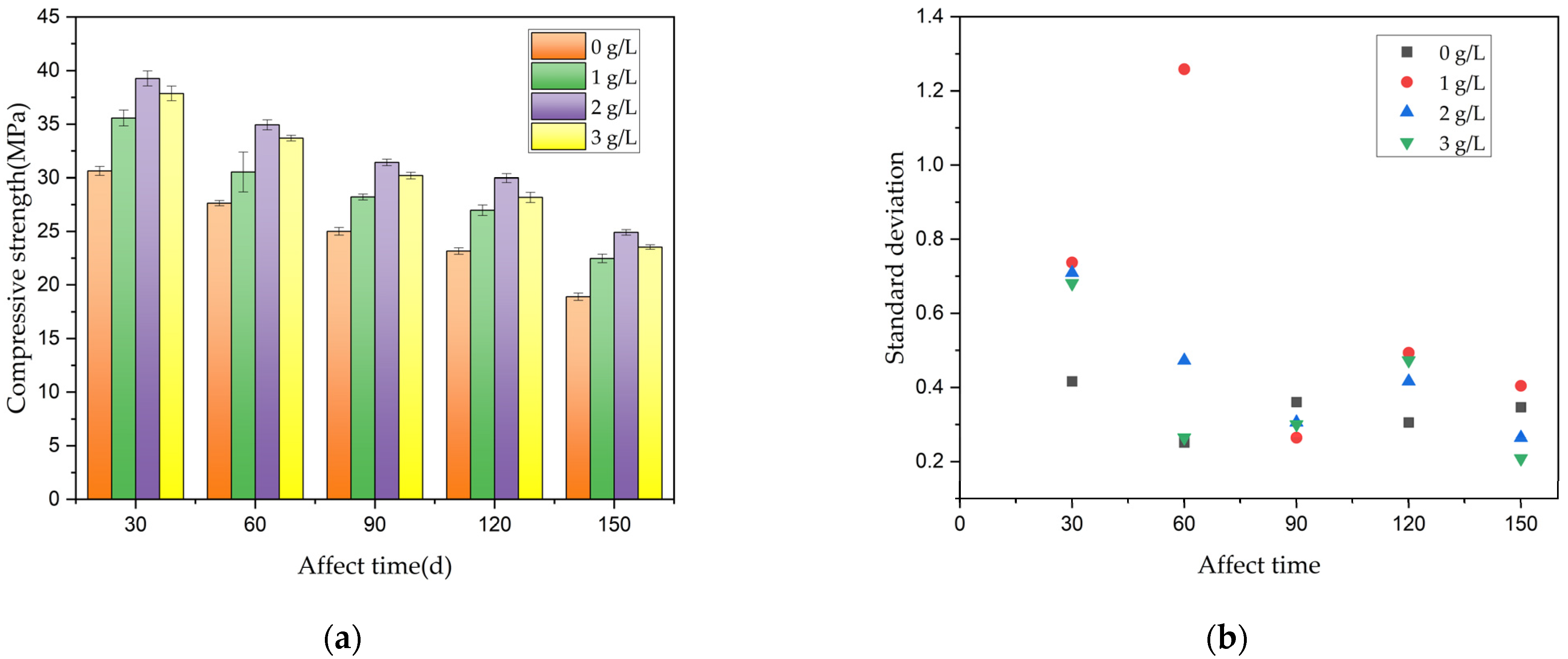
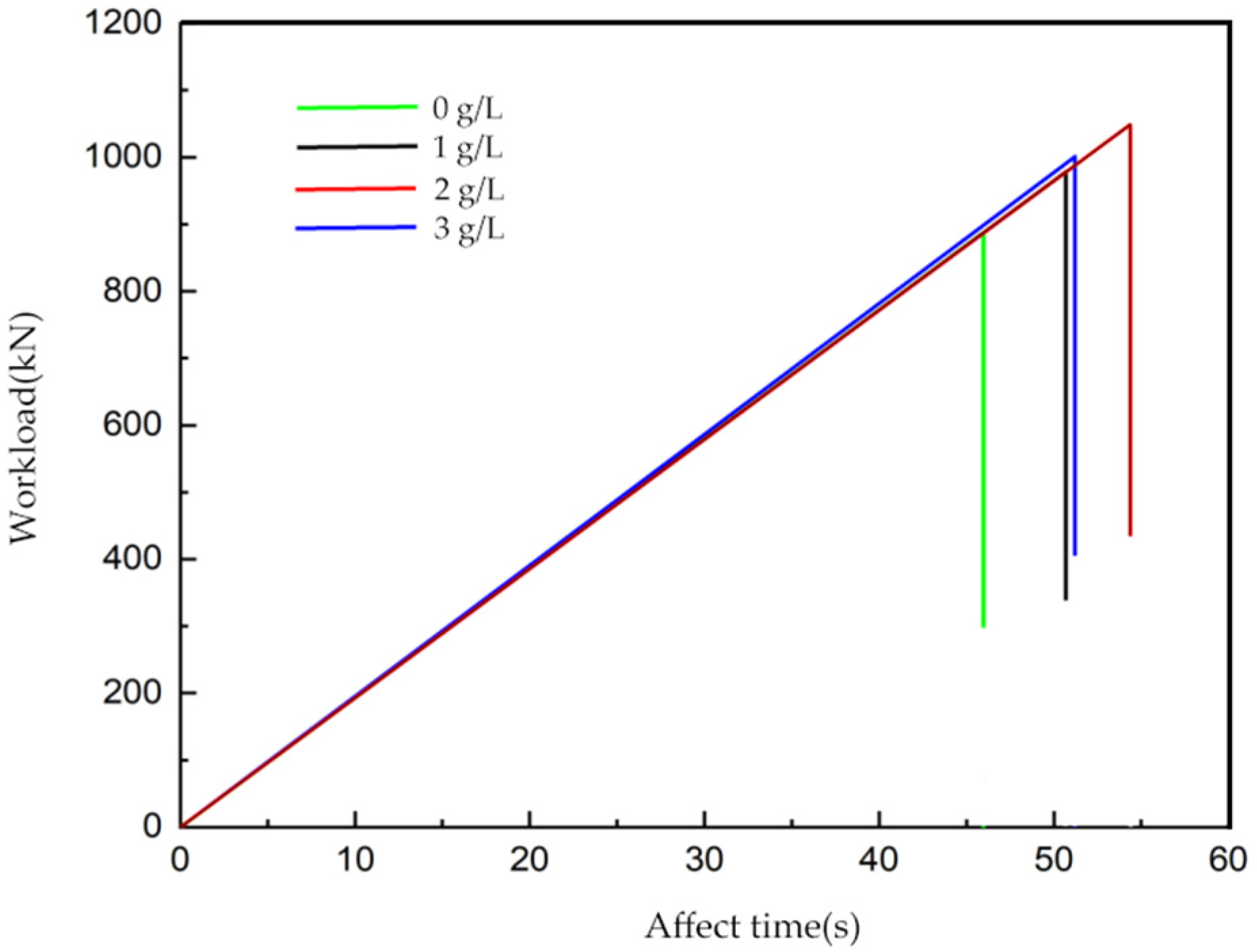
Xanthium sibiricum corrosion inhibitor for practical applications. Furthermore, in connection with the above formulas, a prediction formula was developed. This formula estimates the remaining compressive strength of reinforced concrete after a specific corrosion period, taking into account various dosages of corrosion inhibitors.