Abstract
In recent years, pediatric dentistry has evolved considerably in terms of both the techniques and materials used. A clear, odorless liquid called silver diamine fluoride (SDF) is recommended for molars affected by hypoplasia and dentinal hypersensitivity. In adults and children who are at a high risk of developing caries, it is also helpful to prevent the progression of carious lesions. Teeth too compromised to be reconstructed that are not accompanied by discomfort or infection can be treated with SDF. The current review aims to provide an overview of the most recent research over the last 10 years on the potential of SDF, focusing on the clinical evidence of professionally applied fluoride therapy to prevent and arrest caries in children’s primary molars. Research was conducted according to the guidelines of the PRISMA statement. “silver diamine fluoride”, “primary molar”, and “primary molars” were the search terms utilized on the databases Scopus, Web of Science, and PubMed, with the Boolean operator “AND” and “OR”. Of these 195 studies, 13 primary studies were included in this review. The quality of the included papers was assessed using the reputable Cochrane risk of bias assessment for randomized trials. The studies reviewed consistently support the effectiveness of SDF in stopping the progression of existing caries in deciduous molars; on the contrary, there is insufficient evidence to draw conclusions about SDF being effective for the prevention of carious lesions in deciduous molars.
1. Introduction
Dental caries remain a significant global health concern, particularly among pediatric populations [1,2,3,4,5,6,7,8,9,10,11,12,13].
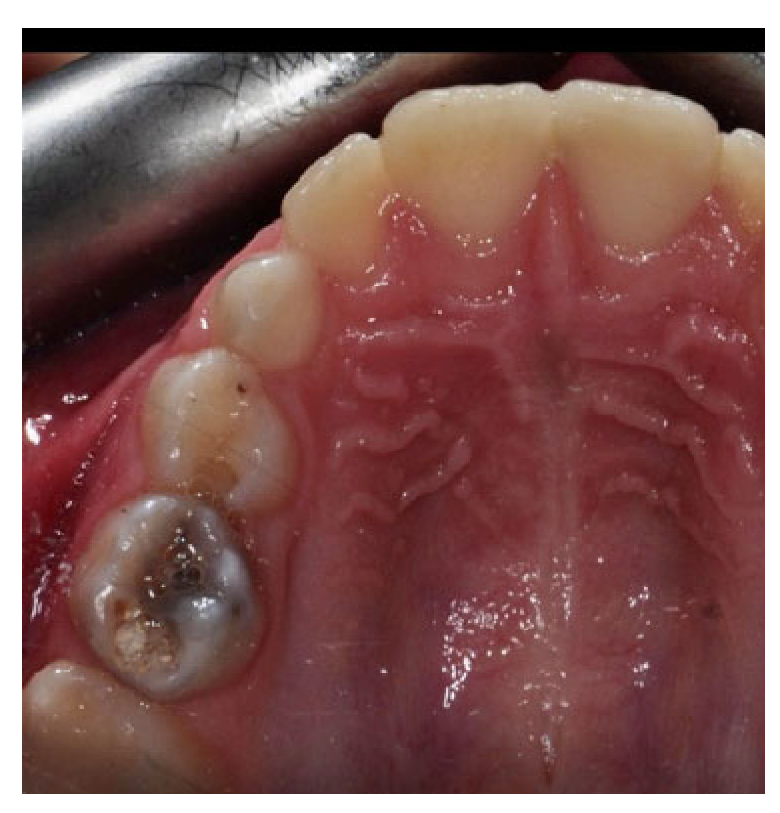
Early childhood caries (ECCs) stand out as one of the most prevalent chronic conditions in childhood. This phenomenon significantly impacts children’s health, necessitating careful attention from parents and healthcare practitioners (Figure 1) [14,15,16,17,18,19,20,21,22].
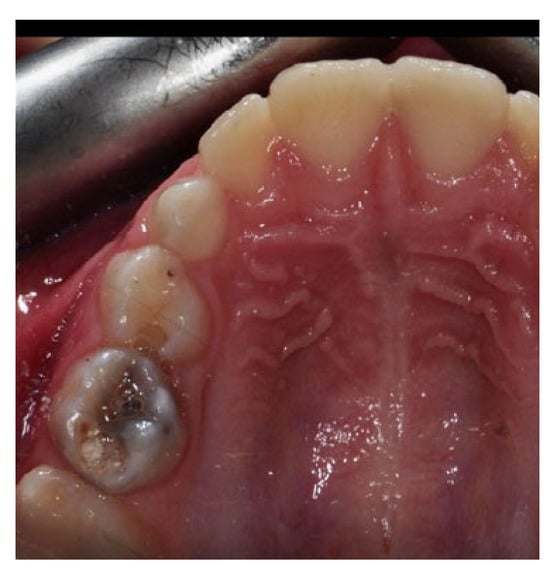
Figure 1.
Decayed second deciduous molar.
In the traditional approach, addressing a cavitated carious lesion typically requires the use of a dental bur attached to a high-speed handpiece for the removal of carious dentin. The audible noise and tactile sensations associated with the dental bur in the oral cavity can evoke fear in the child, resulting in uncooperative behavior and making dental procedures more challenging to carry out [12,23,24,25,26,27,28,29,30,31]. The challenges presented by these behavioral issues significantly impede the successful implementation of restorative treatments for dental caries in young children [32,33,34,35,36,37,38,39].
If ECC persists without intervention, it can impact various facets of a child’s well-being, including oral health–related quality of life, body weight, growth, school attendance, and academic performance [40,41,42,43,44,45,46,47,48,49,50,51].
The endeavor to devise effective, minimally invasive strategies for caries prevention and management has fueled a surge of interest in alternative interventions. Among these, silver diamine fluoride (SDF) has emerged as a noteworthy candidate, demonstrating considerable potential in impeding the progression of carious lesions in the challenging domain of molar teeth in children [52,53,54].
SDF has demonstrated promising outcomes in hindering the progression of caries, especially in molar teeth among children [55,56,57,58,59].
It is a solution that contains a diamine-silver ion and fluoride ion, serving as both an antibacterial and a remineralizing agent. By decreasing biofilm counts and impeding collagen degradation, this product promotes the remineralization of enamel and dentin, effectively halting the progression of caries [60,61,62,63,64].
Silver diamine fluoride (SDF) exerts its preventive action against dental caries through a multifaceted mechanism. Upon its application, SDF dissociates into silver, fluoride, and ammonium ions. The silver ions exhibit antimicrobial properties by disrupting bacterial cell membranes and inhibiting bacterial enzyme activity, thereby reducing the population of cariogenic bacteria such as Streptococcus mutans. Additionally, fluoride ions promote remineralization by enhancing the formation of fluorapatite, which strengthens the tooth structure and renders it more resistant to acid dissolution. Furthermore, SDF facilitates the formation of a protective layer of silver phosphate and calcium fluoride on the tooth’s surface, acting as a physical barrier against acid attack and inhibiting demineralization. This comprehensive approach makes SDF an effective agent in preventing dental caries, particularly in primary molars where traditional interventions may be challenging [65,66,67]. Inizio moduloFine modulo.
SDF finds application in a variety of dental procedures; it is utilized for the halting of early childhood caries, the management of root caries, and the prevention of pit and fissure caries [61]. Additionally, it plays a role in impeding secondary caries, promoting the remineralization of hypomineralized teeth, and treating infected root canals [68,69,70]. Furthermore, SDF is employed to desensitize hypersensitive teeth [71]. Some practitioners have explored the potential of using SDF in the management of dental erosion, while others have suggested its use as a diagnostic dye to identify carious tissues, aiding in the guidance of caries excavation [71,72,73,74,75]. In the realm of research, the combination of laser irradiation with SDF therapy has been studied for its efficacy in preventing tooth fracture post-endodontic treatment [76,77,78,79,80,81,82,83,84,85,86,87,88,89]. Moreover, researchers have investigated its ability to prevent the development of root caries by enhancing fluoride uptake in dentine [90,91].
SDF stands out as an alternative treatment for managing dental caries in situations where other approaches are not feasible. It represents a minimally invasive, cost-effective, and straightforward method capable of alleviating fear and anxiety, especially in young children. Furthermore, it holds potential for application in community settings [60,92,93,94,95,96,97].
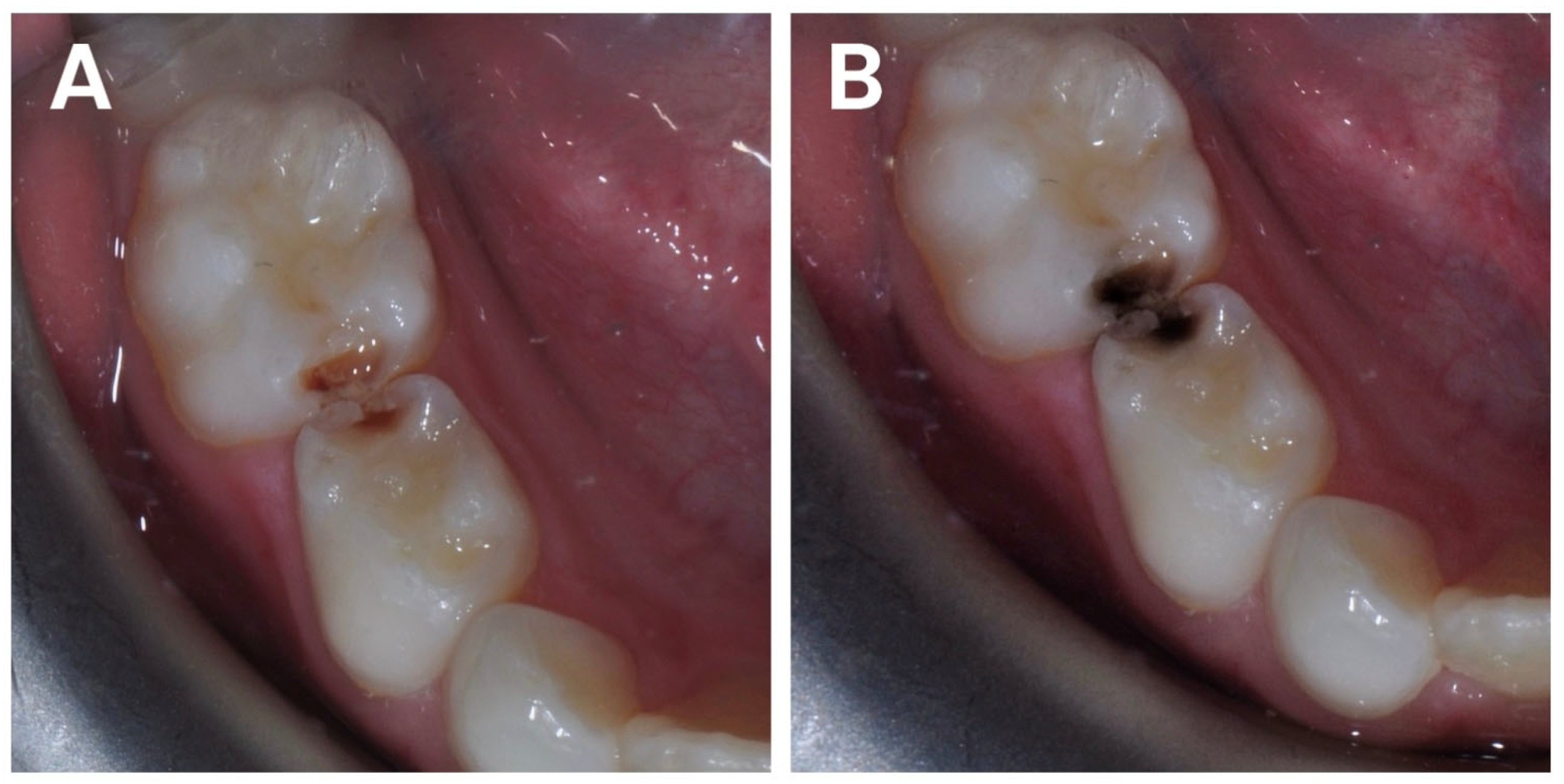
Its ease of application, low cost, and the potential to address caries in non-cooperative pediatric patients have positioned SDF as a compelling intervention (Figure 2) [59,98,99,100].
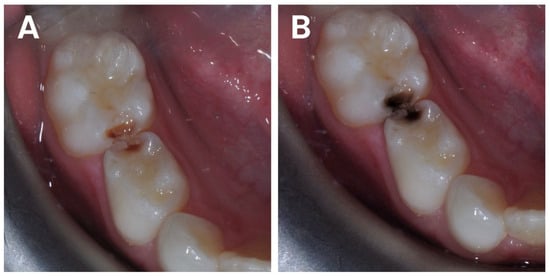
Figure 2.
Before (A) and after (B) the application of SDF around the embrasures of carious lesions with the use of a microbrush.
By synthesizing the existing literature and analyzing clinical trials, observational studies, and other relevant research, this review aims to provide evidence-based insights into the effectiveness of SDF applications in arresting carious lesions, preventing the progression of caries, and its potential adverse effects, thereby informing clinical practice and guiding future research in the field of dentistry.
2. Materials and Methods
2.1. Protocol and Registration
This systematic review was conducted by the standards of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 statement [101]. The review protocol was registered at PROSPERO under the unique number ID: CRD 505141.
2.2. Search Processing
“Silver diamine fluoride”, “primary molar”, and “primary molars” were the search terms utilized on the databases (Scopus, Web of Science, and Pubmed) to select the papers under evaluation, with the Boolean operator “AND” and “OR”. The research was conducted in December 2023 and was restricted to just items released in English over the previous ten years (December 2013–December 2023) (Table 1).

Table 1.
Database search indicators.
2.3. Eligibility Criteria
The reviewers, who worked in pairs, chose works that satisfied the following criteria for inclusion: (1) human subject-only research; (2) clinical studies (3); research concerning children treated with SDF on deciduous molars.
Exclusion criteria were (1) in vitro studies (2), animal studies, (3) systematic reviews, narrative reviews, and meta-analyses (4) case reports. Duplicate studies were removed manually.
The review was conducted using PICO criteria:
- −
- Population: children, both male and female, who had a primary molar treated with SDF;
- −
- Intervention: SDF on deciduous molar;
- −
- Comparison: untreated deciduous molars with SDF;
- −
- Outcome: effectiveness of SDF in arresting caries or preventing its development on deciduous molars.
2.4. Data Processing
Disagreements among reviewers on the selection of the articles were discussed and resolved.
2.5. Article Identification Procedure
The appropriateness evaluation was performed independently by two reviewers, F.I. and G.L. An additional manual search was conducted to increase the number of articles available for full-text analysis. English-language articles that met the inclusion criteria were taken into consideration, and duplicates and items that did not qualify were marked with the reason they were not included.
2.6. Study Evaluation
The screening process allowed for the exclusion of any publications that did not fit the themes examined. It was carried out by G.L. and L.R. independently by reading the article titles and abstracts selected in the earlier identification step.
After being found to meet the predefined inclusion criteria, the full text of the publications was read.
2.7. Quality Assessment
The quality of the included papers was assessed by two reviewers, RS and LR, using the reputable Cochrane risk of bias assessment for randomized trials (RoB 2) [76]. This test assesses six potential areas of bias as follows: random sequence generation, allocation concealment, blinding participants and personnel, blinding outcome assessment, incomplete outcome data, and selective reporting. In the event of a disagreement, a third reviewer (FI) was consulted until a consensus was obtained. Because all articles have a study group and a control group, we were able to use the Rob 2 tool to assess the risk of bias among the identified studies.
3. Results
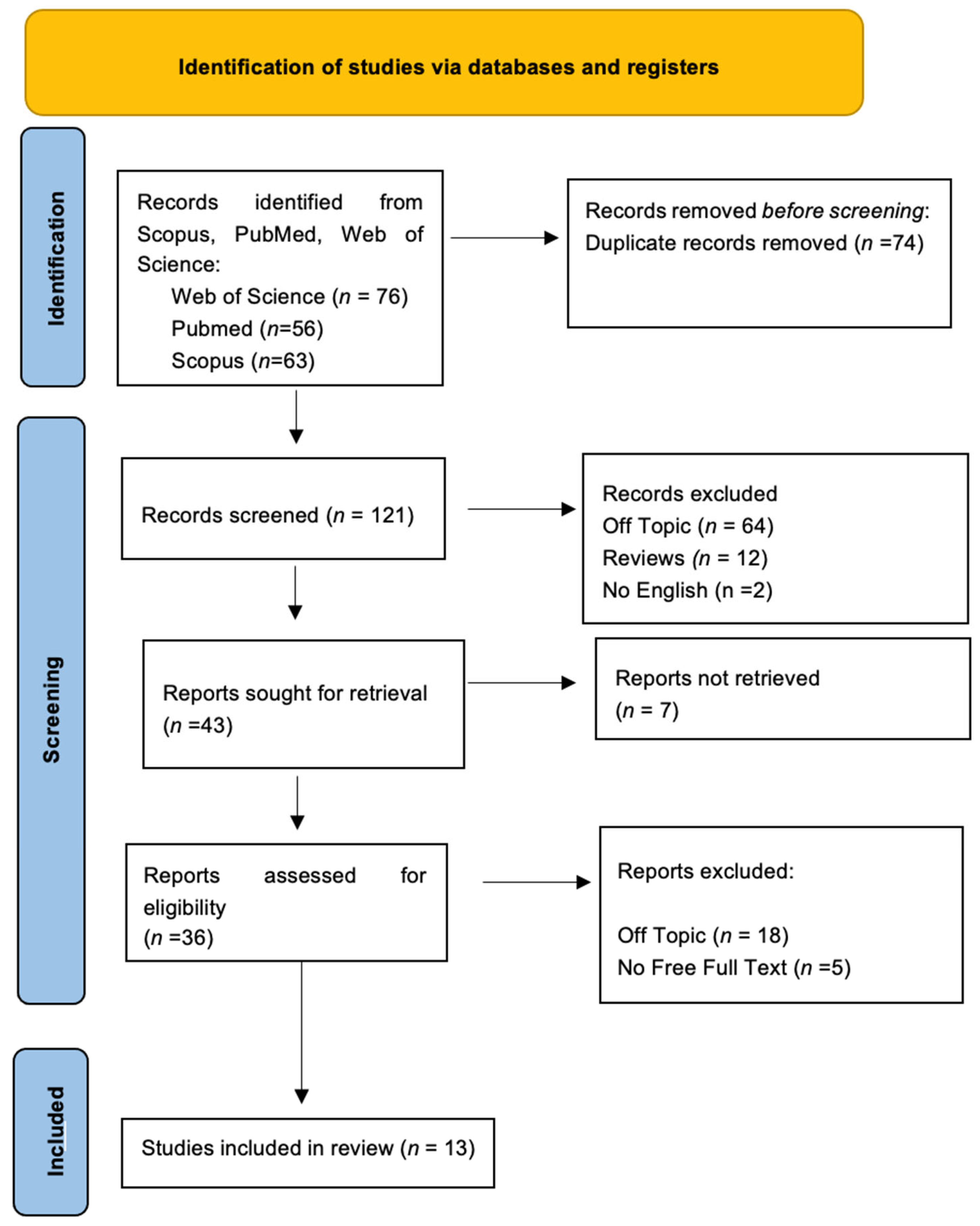
Keyword searches of the Web of Science (76), Scopus (63), and Pubmed (56) databases yielded a total of 195 articles. The subsequent elimination of duplicates (74) resulted in the inclusion of 121 articles. Of these 121 studies, 78 were excluded because they deviated from the previously defined inclusion criteria, leading to 43 records being selected. Subsequently, 7 records not retrieved were excluded, and then 23 reports were excluded because they did not meet the criteria (18 were off-topic, and 5 were reviews). After their eligibility was confirmed, 13 primary studies were selected for qualitative analysis. The selection process and the summary of selected records are shown in Figure 3 and Table 2, respectively.
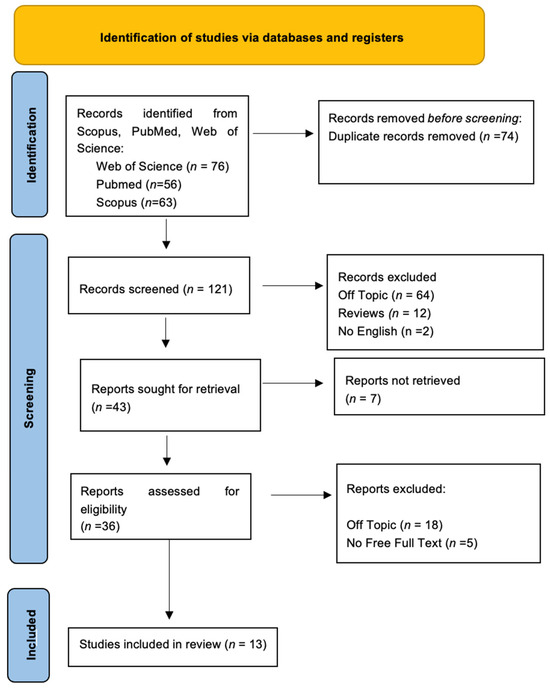
Figure 3.
The literature search of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram and database search indicators.

Table 2.
Descriptive summary of item selection.
3.1. Quality Assessment and Risk of Bias
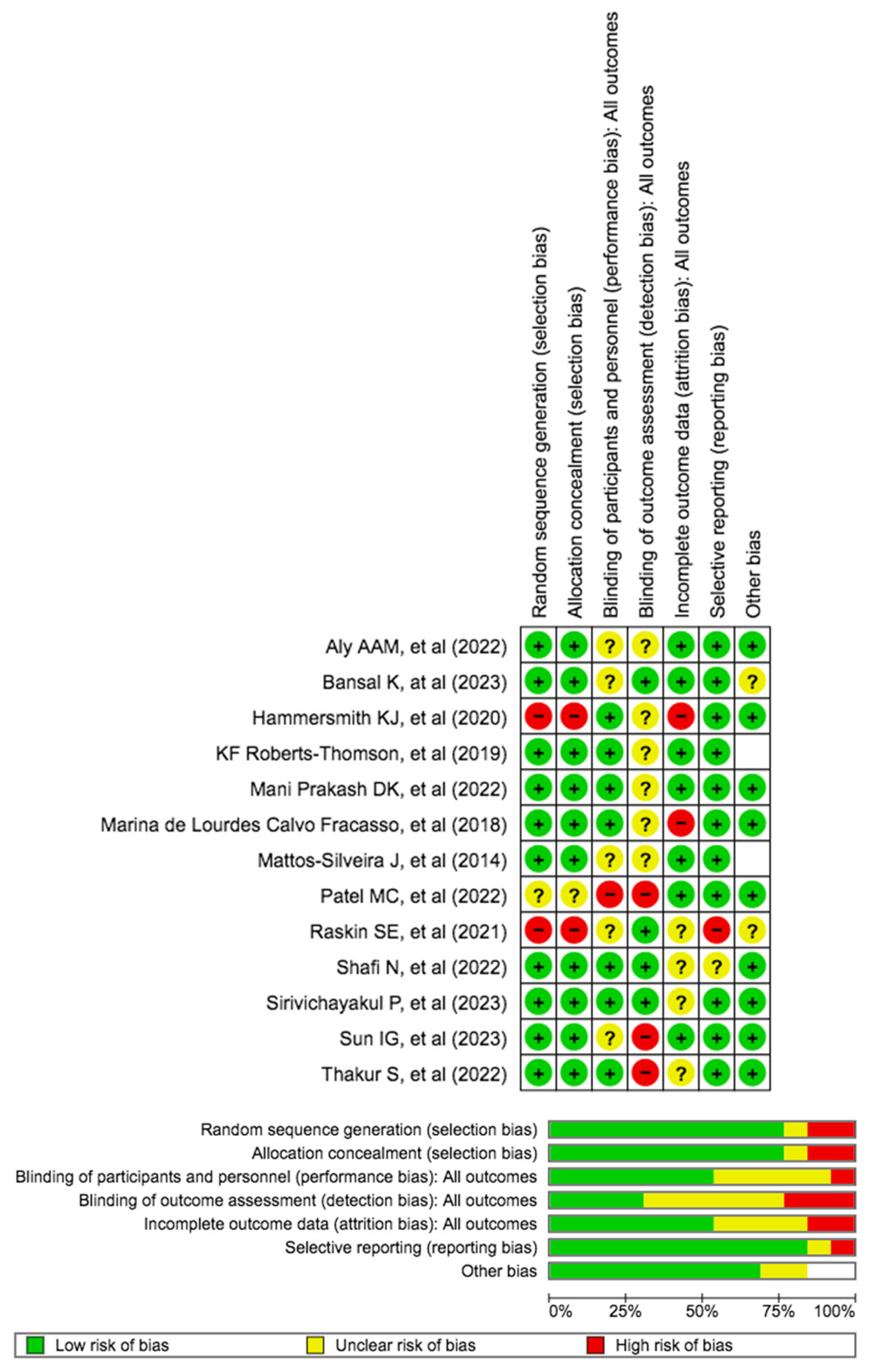
The risk of bias was calculated using RoB 2 and is displayed in Figure 4. Concerning randomization, a low risk of bias was guaranteed by all trials. Regarding allocation concealment, 75% of articles had a low risk of bias. The results of the self-reported study revealed that 80% had a minimal risk of bias. The objective results, blinding of participants and staff, and blinding of the outcome evaluation showed little chance of bias at 50% and 30%, respectively. A 55% low probability of bias was indicated by the papers’ incomplete outcome data (objective measures). Regarding selective reporting, there was little chance of bias in every study.
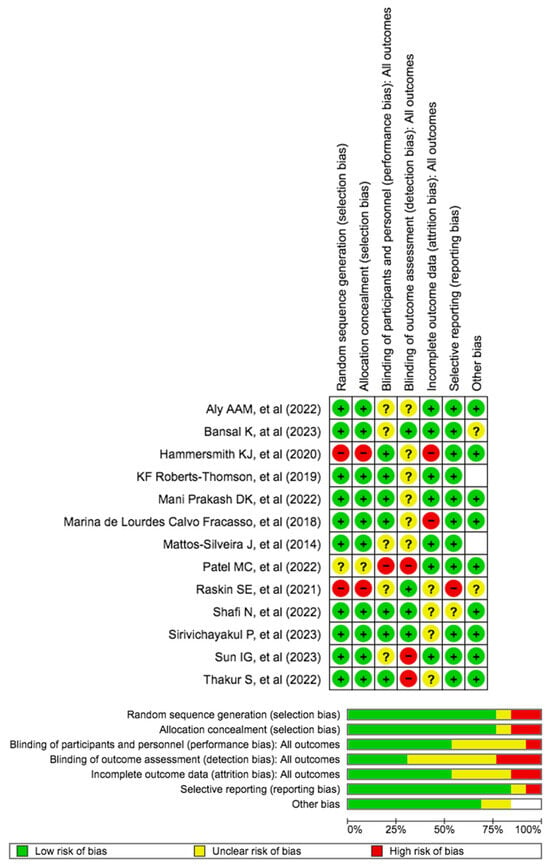
Figure 4.
Risk of bias domains of the included studies.
3.2. Relevant Findings of the Studies
There was a total of 13 studies analyzed, including 8 randomized clinical trials, 2 prospective clinical studies, 1 naturalistic study, and 1 retrospective study. The average age of participants across the studies reviewed varied slightly. Studies included children ranging from 4 to 8 years old, with some focusing on preschoolers and others including children up to 9 years old. This indicates that the research on the effectiveness of silver diamine fluoride (SDF) in preventing and treating caries in primary molars spans a broad age range typical of primary dentition.
Regarding the techniques studied, a variety of interventions were investigated in the reviewed literature. These included comparisons between SDF and traditional methods like the atraumatic restorative technique (ART) or conventional restoration, as well as evaluations of SDF against other materials such as glass ionomer cement (SMART), sodium fluoride varnish, and resin sealants. Additionally, some studies assessed the effectiveness of SDF, either alone or in combination with other treatments like sedative filling or light cure calcium hydroxide. This diversity in studied techniques reflects the ongoing exploration of different approaches to managing caries in primary molars and highlights the interest in finding effective, minimally invasive interventions for young children.
4. Discussion
Since the late 1960s, SDF has been utilized for many years worldwide, with concentrations ranging from 10% to 38% [53,98,102,103,104,105,106,107,108,109,110,111,112].
The Food and Drug Administration approved it in 2014 as a desensitizing agent, and since then, its use and research interest have increased both in the US and, more recently, in Europe [113,114,115,116].
While its utilization has expanded globally, a comprehensive examination of the existing body of literature is imperative to distill the evidence surrounding its efficacy, safety, non-invasive nature, child-friendly quality in dental care practices, and the nuances of its application in the specific context of molar caries prevention and arrest in children.
This systematic literature review aims to comprehensively assess and synthesize existing research on the utilization of SDF as an intervention for both preventing and arresting caries in the molars of pediatric patients [117].
The synthesis of evidence from a series of studies over the last ten years contributed significantly to our understanding of the effectiveness and implications of SDF in preventing and arresting pediatric dental caries. This discussion will be structured around three major themes, each delving into various facets of the research, encompassing its comparative efficacy, clinical performance, application protocols, and broader population-level impact. All studies showed low bias in both the placement and possible systematic errors in evaluating the effect of SDF on caries prevention [118].
4.1. Comparative Efficacy of SDF in Various Treatment Modalities
The systematic review identified several studies assessing the comparative efficacy of SDF in different treatment modalities [119]. For instance, Roberts-Thomson et al. in 2019 were pivotal in assessing the efficacy of SDF in pediatric dental caries prevention and treatment in remote Aboriginal communities. The randomized groups received an atraumatic restorative technique (ART) or SDF. The study found that SDF-treated teeth showed more retreatment needs, particularly in 5-year-olds, leading to a higher prevalence ratio (PR: 1.77). However, when retreatment was considered routine, the PR for negative outcomes was lower for SDF (PR: 0.18). This highlighted the staining issues associated with SDF but emphasized its effectiveness and simplicity compared to ART. Despite its contributions, the study faced challenges in recruitment, posing potential limitations on its generalizability [77].
Similarly, Bansal et al. demonstrated comparable success rates of silver diamine fluoride-modified atraumatic restorative technique (SMART) with conventional methods, emphasizing its potential as a child-friendly alternative. They compared the use of the SDF-modified atraumatic restorative technique (SMART) with the conventional drill and fill method in treating deep asymptomatic caries in primary molars. The study, involving 226 children aged 4–8, showed that SMART had comparable success rates with the conventional method. However, SMART demonstrated better treatment acceptance (79% vs. 56%), making it a potential alternative for uncooperative children. This underscores the significance of child-friendly approaches in pediatric dentistry, aligning with the paradigm shift towards minimally invasive techniques [78].
However, Sirivichayakul et al. challenged the presumed superiority of SDF over traditional fluoride varnishes in preventing caries in high-risk preschool children, suggesting contextual effectiveness. This study involved engaging in an 18-month randomized clinical trial comparing the efficacy of two topical fluoride applications and a placebo in preventing approximal caries in the primary teeth of preschool children. Participants were assigned to three groups: placebo control, 5% sodium fluoride (NaF) varnish, and 38% SDF. Surprisingly, the study found no significant differences in the development of approximal caries among the groups, challenging the presumed superiority of SDF over traditional fluoride varnishes. This suggests that, in certain contexts, traditional fluoride varnishes may be as effective as SDF in preventing caries in high-risk preschool children [80].
While the study by Roberts-Thomson et al. highlighted the effectiveness of SDF in remote Aboriginal communities, it also brought attention to the need for routine retreatment and staining issues associated with SDF. Bansal et al.’s findings regarding SMART underscored its comparable success rates with conventional methods, along with its superior treatment acceptance, indicating its potential as a child-friendly alternative. Additionally, Sirivichayakul et al.’s study challenged the presumed superiority of SDF over traditional fluoride varnishes in preventing caries in high-risk preschool children, suggesting that traditional varnishes may be equally effective in certain contexts.
4.2. Clinical Performance and Long-Term Outcomes of SDF
The studies reviewed also focused on the clinical performance and long-term outcomes of SDF interventions. For example, Mohamed Ali et al. found comparable clinical performance between SMART and ART, with SMART demonstrating cost-effectiveness and shorter treatment times. Additionally, Guofang Sun et al. addressed the lack of consensus in post-SDF instructions, contributing evidence for effective SDF treatment protocols. Calvo Fracasso et al. aimed to assess the clinical performance of preventive materials, including a resin-modified glass ionomer cement, resin sealant, and SDF, on primary molars over 36 months. The study, involving 32 children, found no significant difference in material retention between the groups. The resin sealant exhibited higher loss (44%) than glass ionomer cement (35%). Incipient caries incidence was highest in the SDF group (20%), but differences were not significant. Overall, the study concluded that no significant differences in material retention and caries were found, emphasizing the importance of clinical judgment in selecting appropriate preventive materials for high-risk pediatric patients [81].
Mohamed Ali et al. assessed the clinical performance and cost-effectiveness of SMART and ART in primary molars over 12 months [120]. The study involved 67 children with active caries randomly assigned to SMART or ART. Both SMART and ART techniques showed comparable clinical performance, with mean survival times of 11.8 and 11.6 months, respectively. SMART had a significantly lower mean treatment time (7.8 min.) and total cost per restoration, suggesting that, despite similar clinical outcomes, SMART is less time-consuming and more cost-effective than ART in treating single-surface occlusal lesions in primary molars [82].
The 12-month randomized controlled clinical trial by Guofang Sun et al. aimed to compare post-treatment instructions for SDF therapy in arresting dentine caries in kindergarten children. The study involved at least 254 children randomized into two groups, receiving 38% SDF topically. Group A rinsed immediately, while Group B refrained from rinsing, eating, and drinking for 30 min. The primary outcome was the proportion of arrested caries lesions at the 12-month examination. Secondary outcomes included parental satisfaction and adverse effects [121]. This trial addressed the lack of consensus in post-SDF instructions and contributed evidence for effective SDF treatment protocols [83].
Mattos-Silveira et al.’s study aimed to assess the effectiveness and cost-effectiveness of SDF in preventing and arresting initial caries lesions on approximal surfaces in pediatric patients. The randomized clinical trial included three groups: SDF, resin infiltration, and a control group with exclusive flossing. The primary outcome was caries progression, evaluated through clinical and radiographic examinations. Secondary outcomes included patient discomfort, satisfaction, and cost analysis. The hypothesis suggested SDF as the most cost-effective option. The trial, with ethical approval, involved children and adolescents, with interventions and placebos standardized. This study addressed the challenge of controlling caries lesions on approximal surfaces, emphasizing the importance of early intervention [84].
4.3. Optimizing SDF Application Protocols and Treatment Outcomes
Thakur et al. explored the optimal duration of SDF application in pediatric dental caries prevention. The study, involving different application durations (30, 60, and 120 s), found no significant difference in overall caries arrest at 6 months [122]. This challenges the notion that longer application times necessarily lead to better outcomes, providing insights into the efficiency of SDF application in minimally cooperative children [88]. Shafi et al. investigated the potential of 1:10-diluted SDF as an option for indirect pulp capping in primary molars, comparing it with light-cured calcium hydroxide. The study found no statistically significant difference in success rates between the two groups, suggesting that diluted SDF can indeed be a viable option. This aligns with the broader trend of exploring alternative applications of SDF in pediatric dentistry [89].
In a clinical interventional study by Patel et al. involving 60 primary molar teeth in children aged 4–8 years, a comparative analysis was conducted between SMART and conventional treatments. Teeth were assessed at the baseline, 3, 6, and 12 months. SMART involved SDF application followed by glass ionomer cement restoration and stainless-steel crown placement. The results showed that both SMART and conventional methods effectively managed deep caries lesions, highlighting the potential of SDF in minimally invasive dentistry [86].
In the double-blinded, randomized controlled trial of Prakash et al., 34 children (age 6–9 years) with 68 cavitated carious primary molars were divided into two groups: 38% SDF and 5% NaF varnish. The study aimed to evaluate the caries-arresting potential at 6- and 12-month intervals. The results indicated statistically significant differences in favor of SDF at both intervals (82% vs. 45% at 6 months, 77.4% vs. 41.9% at 12 months) [123]. This study suggests SDF’s superior efficacy in arresting cavitated carious lesions compared to NaF varnish, emphasizing its potential as a non-invasive treatment for disadvantaged communities [87].
Patel et al.’s clinical interventional study and Prakash et al.’s randomized controlled trial both demonstrate the efficacy of SDF-based treatments in managing deep caries lesions, with SDF showing superior caries-arresting potential compared to conventional methods and fluoride varnish, respectively. These findings underscore the potential of SDF as a non-invasive and effective treatment option, particularly for disadvantaged communities.
The study of Hammersmith et al. aimed to assess how often interproximal caries arrest occurred in primary dentition after SDF and fluoride varnish applications. A retrospective review of the dental records of 131 pediatric patients who received SDF treatment with woven floss was performed. In total, 84% of 185 interproximal carious lesions had radiographic evidence of non-progression during a 12-month follow-up. Regarding the type of insurance or the type of teeth, no discernible changes were detected. According to the study, SDF applied with woven floss is linked to low-risk children’s interproximal caries arrest. Limitations include the retrospective design and the need for future placebo-controlled studies [79].
Collectively, these studies contribute to the growing body of evidence supporting the efficacy and versatility of SDF in pediatric dental care, paving the way for its wider adoption and application in clinical practice.
4.4. Population-Level Impact and Public Health Considerations
The study by Raskin et al. analyzed data from a large cohort, assessing the outcomes of SDF across demographics. These findings supported the efficacy of SDF for non-cavitated lesions and early decay, emphasizing its potential impact on the population level [124]. Notably, a single yearly SDF application was found to effectively arrest caries, with minor restorations common upon SDF failure [90]. This highlights the significance of SDF in public health strategies for managing dental caries at a broader scale [27,120].
4.5. Limitations
The studies on silver diamine fluoride (SDF) in pediatric dental care, while informative, have the following notable limitations:
- Heterogeneous designs: Variability in methodologies and participant characteristics hampers direct comparisons, impacting generalizability.
- Staining and acceptance concerns: aesthetic issues related to SDF staining raise acceptance challenges, introducing subjective biases in evaluation.
- Recruitment challenges and sample size variability: recruitment difficulties and varying sample sizes across studies affect representativeness and statistical reliability.
- Short to medium-term follow-up: many studies have relatively short follow-up periods (a week), limiting insights into the long-term (months) effectiveness and side effects of SDF.
- Retrospective nature: studies with retrospective designs, like Hammersmith et al., face challenges related to incomplete data and potential bias.
- Applicability to high-risk populations: generalizing SDF’s effectiveness to diverse pediatric populations with varying risk profiles remains a challenge.
- Inconsistent outcome measures: diverse outcome measures across studies complicate the synthesis of evidence and hinder standardization.
- Limited socioeconomic and cultural diversity: some studies lack diverse socioeconomic and cultural representation, impacting the broad applicability of findings.
4.6. Strengths of the Review
The clinical efficacy of SDF is an evolving field of research, and limitations in the scientific literature may vary over time. There are still many variations in protocols as follows: a lack of standardization of treatment protocols, such as how to apply the product, duration, and frequency of sessions, makes it difficult to compare the results between different studies. Our research refers to the last 10 years and is, therefore, up-to-date with the latest scientific studies.
4.7. Implications for Future Research
Experts could define a core set of outcomes that might help clinical trialists understand the advantages and disadvantages of nonrestorative treatment for managing dental caries. As a result, this would assist those who create systematic reviews in defining and compiling results. Furthermore, we could not find any published RCTs despite our interest in the impact of SDF on non-cavitated carious lesions on approximate surfaces. Finally, future researchers should strive to improve the overall caliber of their studies by submitting a more thorough report of their procedures and lowering the possibility of bias using appropriate randomization and allocation concealment techniques.
5. Conclusions
Considering various studies, the multifaceted efficacy of SDF in the prevention and treatment of pediatric dental caries is highlighted. While acknowledging staining issues and recruitment challenges, SDF emerges as a valuable asset in minimizing treatment time and costs, especially in non-cavitated lesions. The comparison with traditional methods and exploration of optimal application durations provides insights for tailored interventions. These findings, supported by large-scale analyses, substantiate the population-level impact of SDF, advocating for its strategic inclusion in public health initiatives. Overall, this synthesis informs evidence-based approaches, paving the way for refined protocols and the comprehensive management of pediatric dental caries.
Author Contributions
Conceptualization, L.R., G.L. and R.S.; methodology, F.I.; software, A.D.I. and A.M.I.; validation, G.D., A.M. and A.P.; formal analysis, L.R. and R.S.; investigation, G.L.; resources, R.S.; data curation, F.I. and G.D.; writing—original draft preparation, A.M.I. and A.P.; writing—review and editing, L.R.; visualization, G.L.; supervision, R.S.; project administration, F.I.; funding acquisition, L.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data generated or analyzed during this study are included in the published article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| AgF | Stannous fluoride |
| ART | Atraumatic restorative technique |
| ECC | Early childhood caries |
| NaF | Sodium-fluoride |
| SDF | Silver Diamine Fluoride |
| SMART | Silver-modified atraumatic restorative technique |
References
- Campanella, V.; Gallusi, G.; Nardi, R.; Mea, A.; Di Taranto, V.; Montemurro, E.; Marzo, G.; Libonati, A. Dentinal Substrate Variability and Bonding Effectiveness: Sem Investigation. J. Biol. Regul. Homeost. Agents 2020, 34, 49–54. [Google Scholar]
- Marchetti, E.; Tecco, S.; Caterini, E.; Casalena, F.; Quinzi, V.; Mattei, A.; Marzo, G. Alcohol-Free Essential Oils Containing Mouthrinse Efficacy on Three-Day Supragingival Plaque Regrowth: A Randomized Crossover Clinical Trial. Trials 2017, 18, 154. [Google Scholar] [CrossRef]
- Mummolo, S.; Tieri, M.; Tecco, S.; Mattei, A.; Albani, F.; Giuca, M.R.; Marzo, G. Clinical Evaluation of Salivary Indices and Levels of Streptococcus Mutans and Lactobacillus in Patients Treated with Occlus-o-Guide. Eur. J. Paediatr. Dent. Off. J. Eur. Acad. Paediatr. Dent. 2014, 15, 367–370. [Google Scholar]
- Rosa, M.; Quinzi, V.; Marzo, G. Paediatric Orthodontics Part 1: Anterior Open Bite in the Mixed Dentition. Eur. J. Paediatr. Dent. 2019, 20, 80–82. [Google Scholar] [CrossRef]
- Crincoli, V.; Anelli, M.G.; Quercia, E.; Piancino, M.G.; Di Comite, M. Temporomandibular Disorders and Oral Features in Early Rheumatoid Arthritis Patients: An Observational Study. Int. J. Med. Sci. 2019, 16, 253–263. [Google Scholar] [CrossRef]
- Inchingolo, A.D.; Malcangi, G.; Inchingolo, A.M.; Piras, F.; Settanni, V.; Garofoli, G.; Palmieri, G.; Ceci, S.; Patano, A.; De Leonardis, N.; et al. Benefits and Implications of Resveratrol Supplementation on Microbiota Modulations: A Systematic Review of the Literature. Int. J. Mol. Sci. 2022, 23, 4027. [Google Scholar] [CrossRef]
- Di Stasio, D.; Lauritano, D.; Romano, A.; Salerno, C.; Minervini, G.; Minervini, G.; Gentile, E.; Serpico, R.; Lucchese, A. In Vivo Characterization of Oral Pemphigus Vulgaris by Optical Coherence Tomography. J. Biol. Regul. Homeost. Agents 2015, 29, 39–41. [Google Scholar]
- Inchingolo, A.M.; Inchingolo, A.D.; Latini, G.; Garofoli, G.; Sardano, R.; De Leonardis, N.; Dongiovanni, L.; Minetti, E.; Palermo, A.; Dipalma, G.; et al. Caries Prevention and Treatment in Early Childhood: Comparing Strategies. A Systematic Review. Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 11082–11092. [Google Scholar] [CrossRef]
- Contaldo, M.; Della Vella, F.; Raimondo, E.; Minervini, G.; Buljubasic, M.; Ogodescu, A.; Sinescu, C.; Serpico, R. Early Childhood Oral Health Impact Scale (ECOHIS): Literature Review and Italian Validation. Int. J. Dent. Hyg. 2020, 18, 396–402. [Google Scholar] [CrossRef]
- Di Spirito, F.; Amato, A.; Di Palo, M.P.; Ferraro, G.A.; Baroni, A.; Serpico, R.; Contaldo, M. COVID-19 Related Information on Pediatric Dental Care Including the Use of Teledentistry: A Narrative Review. Children 2022, 9, 1942. [Google Scholar] [CrossRef]
- Malcangi, G.; Inchingolo, A.D.; Inchingolo, A.M.; Santacroce, L.; Marinelli, G.; Mancini, A.; Vimercati, L.; Maggiore, M.E.; D’oria, M.T.; Hazballa, D.; et al. COVID-19 Infection in Children, Infants and Pregnant Subjects: An Overview of Recent Insights and Therapies. Microorganisms 2021, 9, 1964. [Google Scholar] [CrossRef]
- Patano, A.; Cirulli, N.; Beretta, M.; Plantamura, P.; Inchingolo, A.D.; Inchingolo, A.M.; Bordea, I.R.; Malcangi, G.; Marinelli, G.; Scarano, A.; et al. Education Technology in Orthodontics and Paediatric Dentistry during the COVID-19 Pandemic: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 6056. [Google Scholar] [CrossRef]
- Dipalma, G.; Inchingolo, A.D.; Inchingolo, F.; Charitos, I.A.; Di Cosola, M.; Cazzolla, A.P. Focus on the Cariogenic Process: Microbial and Biochemical Interactions with Teeth and Oral Environment. J. Biol. Regul. Homeost. Agents 2021, 35, 429–440. [Google Scholar] [CrossRef]
- Inchingolo, F.; Tatullo, M.; Abenavoli, F.M.; Marrelli, M.; Inchingolo, A.D.; Palladino, A.; Inchingolo, A.M.; Dipalma, G. Oral Piercing and Oral Diseases: A Short Time Retrospective Study. Int. J. Med. Sci. 2011, 8, 649–652. [Google Scholar] [CrossRef]
- Minervini, G.; Franco, R.; Marrapodi, M.M.; Di Blasio, M.; Ronsivalle, V.; Cicciù, M. Children Oral Health and Parents Education Status: A Cross Sectional Study. BMC Oral Health 2023, 23, 787. [Google Scholar] [CrossRef]
- Minervini, G.; Franco, R.; Marrapodi, M.M.; Fiorillo, L.; Cervino, G.; Cicciù, M. The Association between Parent Education Level, Oral Health, and Oral-Related Sleep Disturbance. An Observational Crosssectional Study. Eur. J. Paediatr. Dent. 2023, 24, 218–223. [Google Scholar] [CrossRef]
- Rossi, F.; Tortora, C.; Paoletta, M.; Marrapodi, M.M.; Argenziano, M.; Di Paola, A.; Pota, E.; Di Pinto, D.; Di Martino, M.; Iolascon, G. Osteoporosis in Childhood Cancer Survivors: Physiopathology, Prevention, Therapy and Future Perspectives. Cancers 2022, 14, 4349. [Google Scholar] [CrossRef]
- Rossi, F.; Tortora, C.; Di Martino, M.; Di Paola, A.; Di Pinto, D.; Marrapodi, M.M.; Argenziano, M.; Pota, E. Alteration of Osteoclast Activity in Childhood Cancer Survivors: Role of Iron and of CB2/TRPV1 Receptors. PLoS ONE 2022, 17, e0271730. [Google Scholar] [CrossRef]
- Geduk, N.; Ozdemir, M.; Erbas Unverdi, G.; Ballikaya, E.; Cehreli, Z.C. Clinical and Radiographic Performance of Preformed Zirconia Crowns and Stainless-Steel Crowns in Permanent First Molars: 18-Month Results of a Prospective, Randomized Trial. BMC Oral Health 2023, 23, 828. [Google Scholar] [CrossRef]
- Benjamin, R.M. Oral Health: The Silent Epidemic. Public Health Rep. 2010, 125, 158–159. [Google Scholar] [CrossRef]
- Inchingolo, F.; Tatullo, M.; Abenavoli, F.M.; Marrelli, M.; Inchingolo, A.D.; Corelli, R.; Mingrone, R.; Inchingolo, A.M.; Dipalma, G. Simple Technique for Augmentation of the Facial Soft Tissue. Sci. World J. 2012, 2012, 262989. [Google Scholar] [CrossRef][Green Version]
- Tecco, S.; Crincoli, V.; Di Bisceglie, B.; Saccucci, M.; Macrĺ, M.; Polimeni, A.; Festa, F. Signs and Symptoms of Temporomandibular Joint Disorders in Caucasian Children and Adolescents. Cranio J. Craniomandib. Pract. 2011, 29, 71–79. [Google Scholar] [CrossRef]
- Minervini, G.; Franco, R.; Marrapodi, M.M.; Almeida, L.E.; Ronsivalle, V.; Cicciù, M. Prevalence of Temporomandibular Disorders (TMD) in Obesity Patients: A Systematic Review and Meta-Analysis. J. Oral Rehabil. 2023, 50, 1544–1553. [Google Scholar] [CrossRef]
- Contaldo, M.; Luzzi, V.; Ierardo, G.; Raimondo, E.; Boccellino, M.; Ferati, K.; Bexheti-Ferati, A.; Inchingolo, F.; Di Domenico, M.; Serpico, R.; et al. Bisphosphonate-Related Osteonecrosis of the Jaws and Dental Surgery Procedures in Children and Young People with Osteogenesis Imperfecta: A Systematic Review. J. Stomatol. Oral Maxillofac. Surg. 2020, 121, 556–562. [Google Scholar] [CrossRef]
- Inchingolo, A.D.; Patano, A.; Coloccia, G.; Ceci, S.; Inchingolo, A.M.; Marinelli, G.; Malcangi, G.; Montenegro, V.; Laudadio, C.; Di Pede, C.; et al. The Efficacy of a New AMCOP® Elastodontic Protocol for Orthodontic Interceptive Treatment: A Case Series and Literature Overview. Int. J. Environ. Res. Public Health 2022, 19, 988. [Google Scholar] [CrossRef]
- Inchingolo, F.; Tatullo, M.; Abenavoli, F.M.; Marrelli, M.; Inchingolo, A.D.; Villabruna, B.; Inchingolo, A.M.; Dipalma, G. Severe Anisocoria after Oral Surgery under General Anesthesia. Int. J. Med. Sci. 2010, 7, 314–318. [Google Scholar] [CrossRef]
- Inchingolo, F.; Inchingolo, A.M.; Malcangi, G.; De Leonardis, N.; Sardano, R.; Pezzolla, C.; de Ruvo, E.; Di Venere, D.; Palermo, A.; Inchingolo, A.D.; et al. The Benefits of Probiotics on Oral Health: Systematic Review of the Literature. Pharmaceuticals 2023, 16, 1313. [Google Scholar] [CrossRef]
- Mungara, J.; Injeti, M.; Joseph, E.; Elangovan, A.; Sakthivel, R.; Selvaraju, G. Child’s Dental Fear: Cause Related Factors and the Influence of Audiovisual Modeling. J. Indian Soc. Pedod. Prev. Dent. 2013, 31, 215–220. [Google Scholar] [CrossRef]
- Marrelli, M.; Tatullo, M.; Dipalma, G.; Inchingolo, F. Oral Infection by Staphylococcus Aureus in Patients Affected by White Sponge Nevus: A Description of Two Cases Occurred in the Same Family. Int. J. Med. Sci. 2012, 9, 47–50. [Google Scholar] [CrossRef]
- Marinelli, G.; Inchingolo, A.D.; Inchingolo, A.M.; Malcangi, G.; Limongelli, L.; Montenegro, V.; Coloccia, G.; Laudadio, C.; Patano, A.; Inchingolo, F.; et al. White Spot Lesions in Orthodontics: Prevention and Treatment. A Descriptive Review. J. Biol. Regul. Homeost. Agents 2021, 35, 227–240. [Google Scholar] [CrossRef]
- Patano, A.; Malcangi, G.; Sardano, R.; Mastrodonato, A.; Garofoli, G.; Mancini, A.; Inchingolo, A.D.; Di Venere, D.; Inchingolo, F.; Dipalma, G.; et al. White Spots: Prevention in Orthodontics-Systematic Review of the Literature. Int. J. Environ. Res. Public Health 2023, 20, 5608. [Google Scholar] [CrossRef]
- Inchingolo, A.D.; Ceci, S.; Patano, A.; Inchingolo, A.M.; Montenegro, V.; Di Pede, C.; Malcangi, G.; Marinelli, G.; Coloccia, G.; Garibaldi, M.; et al. Elastodontic Therapy of Hyperdivergent Class II Patients Using AMCOP® Devices: A Retrospective Study. Appl. Sci. 2022, 12, 3259. [Google Scholar] [CrossRef]
- Lanteri, V.; Cossellu, G.; Farronato, M.; Ugolini, A.; Leonardi, R.; Rusconi, F.; De Luca, S.; Biagi, R.; Maspero, C. Assessment of the Stability of the Palatal Rugae in a 3D-3D Superimposition Technique Following Slow Maxillary Expansion (SME). Sci. Rep. 2020, 10, 2676. [Google Scholar] [CrossRef]
- Maspero, C.; Fama, A.; Cavagnetto, D.; Abate, A.; Farronato, M. Treatment of Dental Dilacerations. J. Biol. Regul. Homeost. Agents 2019, 33, 1623–1628. [Google Scholar]
- Paolantoni, G.; Marenzi, G.; Blasi, A.; Mignogna, J.; Sammartino, G. Findings of a Four-Year Randomized Controlled Clinical Trial Comparing Two-Piece and One-Piece Zirconia Abutments Supporting Single Prosthetic Restorations in Maxillary Anterior Region. BioMed Res. Int. 2016, 2016, 8767845. [Google Scholar] [CrossRef]
- Minervini, G.; Marrapodi, M.M.; Cicciù, M. Online Bruxism-related Information: Can People Understand What They Read? A Cross-Sectional Study. J. Oral Rehabil. 2023, 50, 1211–1216. [Google Scholar] [CrossRef]
- Crystal, Y.O.; Niederman, R. Silver Diamine Fluoride Treatment Considerations in Children’s Caries Management. Pediatr. Dent. 2016, 38, 466–471. [Google Scholar]
- Malcangi, G.; Patano, A.; Morolla, R.; De Santis, M.; Piras, F.; Settanni, V.; Mancini, A.; Di Venere, D.; Inchingolo, F.; Inchingolo, A.D.; et al. Analysis of Dental Enamel Remineralization: A Systematic Review of Technique Comparisons. Bioengineering 2023, 10, 472. [Google Scholar] [CrossRef]
- Sharma, A.; Tyagi, R. Behavior Assessment of Children in Dental Settings: A Retrospective Study. Int. J. Clin. Pediatr. Dent. 2011, 4, 35–39. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4999635/ (accessed on 23 January 2024).
- Sheiham, A. Dental Caries Affects Body Weight, Growth and Quality of Life in Pre-School Children. Br. Dent. J. 2006, 201, 625–626. [Google Scholar] [CrossRef]
- Jackson, S.L.; Vann, W.F., Jr.; Kotch, J.B.; Pahel, B.T.; Lee, J.Y. Impact of Poor Oral Health on Children’s School Attendance and Performance. Am. J. Public Health 2011, 101, 1900–1906. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3222359/ (accessed on 20 January 2024). [CrossRef]
- De Grauwe, A.; Aps, J.K.; Martens, L.C. Early Childhood Caries (ECC): What’s in a Name? Eur. J. Paediatr. Dent. 2004, 5, 62–70. [Google Scholar]
- Feldens, C.A.; Giugliani, E.R.J.; Duncan, B.B.; de Drachler, M.L.; Vítolo, M.R. Long-Term Effectiveness of a Nutritional Program in Reducing Early Childhood Caries: A Randomized Trial. Community Dent. Oral Epidemiol. 2010, 38, 324–332. [Google Scholar] [CrossRef]
- Inchingolo, A.D.; Malcangi, G.; Semjonova, A.; Inchingolo, A.M.; Patano, A.; Coloccia, G.; Ceci, S.; Marinelli, G.; Di Pede, C.; Ciocia, A.M.; et al. Oralbiotica/Oralbiotics: The Impact of Oral Microbiota on Dental Health and Demineralization: A Systematic Review of the Literature. Children 2022, 9, 1014. [Google Scholar] [CrossRef]
- Finlayson, T.L.; Siefert, K.; Ismail, A.I.; Sohn, W. Psychosocial Factors and Early Childhood Caries among Low-Income African-American Children in Detroit. Community Dent. Oral Epidemiol. 2007, 35, 439–448. [Google Scholar] [CrossRef]
- Nunn, M.E.; Braunstein, N.S.; Krall Kaye, E.A.; Dietrich, T.; Garcia, R.I.; Henshaw, M.M. Healthy Eating Index Is a Predictor of Early Childhood Caries. J. Dent. Res. 2009, 88, 361–366. [Google Scholar] [CrossRef]
- Berkowitz, R.J. Causes, Treatment and Prevention of Early Childhood Caries: A Microbiologic Perspective. J. Can. Dent. Assoc. 2003, 69, 304–307. Available online: https://pubmed.ncbi.nlm.nih.gov/12734024/ (accessed on 20 January 2024).
- Palmer, C.A.; Kent, R., Jr.; Loo, C.Y.; Hughes, C.V.; Stutius, E.; Pradhan, N.; Dahlan, M.; Kanasi, E.; Arevalo Vasquez, S.S.; Tanner, A.C. Diet and Caries-Associated Bacteria in Severe Early Childhood Caries. Dent. Res. 2010, 89, 1224–1229. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2954266/ (accessed on 20 January 2024). [CrossRef]
- Crescente, G.; Minervini, G.; Spagnuolo, C.; Moccia, S. Cannabis Bioactive Compound-Based Formulations: New Perspectives for the Management of Orofacial Pain. Molecules 2023, 28, 106. [Google Scholar] [CrossRef]
- Minervini, G.; Del Mondo, D.; Russo, D.; Cervino, G.; D’Amico, C.; Fiorillo, L. Stem Cells in Temporomandibular Joint Engineering: State of Art and Future Persectives. J. Craniofac. Surg. 2022, 33, 2181–2187. [Google Scholar] [CrossRef]
- Minervini, G.; Lucchese, A.; Perillo, L.; Serpico, R.; Minervini, G. Unilateral Superior Condylar Neck Fracture with Dislocation in a Child Treated with an Acrylic Splint in the Upper Arch for Functional Repositioning of the Mandible. Cranio J. Craniomandib. Pract. 2017, 35, 337–341. [Google Scholar] [CrossRef]
- Ballini, A.; Cantore, S.; Signorini, L.; Saini, R.; Scacco, S.; Gnoni, A.; Inchingolo, A.D.; De Vito, D.; Santacroce, L.; Inchingolo, F.; et al. Efficacy of Sea Salt-Based Mouthwash and Xylitol in Improving Oral Hygiene among Adolescent Population: A Pilot Study. Int. J. Environ. Res. Public Health 2021, 18, 44. [Google Scholar] [CrossRef]
- Zaffarano, L.; Salerno, C.; Campus, G.; Cirio, S.; Balian, A.; Karanxha, L.; Cagetti, M.G. Silver Diamine Fluoride (SDF) Efficacy in Arresting Cavitated Caries Lesions in Primary Molars: A Systematic Review and Metanalysis. Int. J. Environ. Res. Public Health 2022, 19, 2917. [Google Scholar] [CrossRef]
- Shah, S.; Bhaskar, V.; Venkatraghavan, K.; Choudhary, P.; Trivedi, K. Silver Diamine Fluoride: A Review and Current Applications. J. Adv. Oral Res. 2014, 5, 15–35. Available online: https://www.academia.edu/89754115/Silver_Diamine_Fluoride_A_Review_and_Current_Applications (accessed on 21 January 2024). [CrossRef]
- Zheng, F.M.; Yan, I.G.; Duangthip, D.; Gao, S.S.; Lo, E.C.M.; Chu, C.H. Silver Diamine Fluoride Therapy for Dental Care. Jpn. Dent. Sci. Rev. 2022, 58, 249–257. [Google Scholar] [CrossRef]
- Daga, S.; Shetty, V.; Hegde, A. Silver Diamine Fluoride in Arresting Dentinal Caries in School Children. Indian J. Public Health Res. Dev. 2020, 11, 530–535. [Google Scholar] [CrossRef]
- Llodra, J.C.; Rodriguez, A.; Ferrer, B.; Menardia, V.; Ramos, T.; Morato, M. Efficacy of Silver Diamine Fluoride for Caries Reduction in Primary Teeth and First Permanent Molars of Schoolchildren: 36-Month Clinical Trial. J. Dent. Res. 2005, 84, 721–724. [Google Scholar] [CrossRef]
- Gao, S.S.; Chen, K.J.; Duangthip, D.; Wong, M.C.M.; Lo, E.C.M.; Chu, C.H. Arresting Early Childhood Caries Using Silver and Fluoride Products—A Randomised Trial. J. Dent. 2020, 103, 103522. [Google Scholar] [CrossRef]
- Chu, C.H.; Mei, L.; Seneviratne, C.J.; Lo, E.C.M. Effects of Silver Diamine Fluoride on Dentine Carious Lesions Induced by Streptococcus Mutans and Actinomyces Naeslundii Biofilms. Int. J. Paediatr. Dent. 2012, 22, 2–10. [Google Scholar] [CrossRef]
- Zhi, Q.H.; Lo, E.C.M.; Lin, H.C. Randomized Clinical Trial on Effectiveness of Silver Diamine Fluoride and Glass Ionomer in Arresting Dentine Caries in Preschool Children. J. Dent. 2012, 40, 962–967. [Google Scholar] [CrossRef]
- Holan, G.; Rahme, M.A.; Ram, D. Parents’ Attitude toward Their Children’s Appearance in the Case of Esthetic Defects of the Anterior Primary Teeth. J. Clin. Pediatr. Dent. 2009, 34, 141–145. [Google Scholar] [CrossRef]
- Holan, G.; Fuks, A.; Ketlz, N. Success Rate of Formocresol Pulpotomy in Primary Molars Restored with SSC vs. Amalgam. Pediatr. Dent. 2001, 24, 212–216. [Google Scholar]
- Contreras, V.; Toro, M.J.; Elías-Boneta, A.R.; Encarnación-Burgos, A. Effectiveness of Silver Diamine Fluoride in Caries Prevention and Arrest: A Systematic Literature Review. Gen. Dent. 2017, 65, 22–29. [Google Scholar]
- Nishino, M.; Yoshida, S.; Sobue, S.; Kato, J.; Nishida, M. Effect of Topically Applied Ammoniacal Silver Fluoride on Dental Caries in Children. J. Osaka Univ. Dent. Sch. 1969, 9, 149–155. [Google Scholar]
- Lo, E.C.; Chu, C.H.; Lin, H.C. A Community-Based Caries Control Program for Pre-School Children Using Topical Fluorides: 18-Month Results. J. Dent. Res. 2001, 80, 2071–2074. [Google Scholar] [CrossRef]
- Inchingolo, F.; Tatullo, M.; Pacifici, A.; Gargari, M.; Inchingolo, A.D.; Inchingolo, A.M.; Dipalma, G.; Marrelli, M.; Abenavoli, F.M.; Pacifici, L. Use of Dermal-Fat Grafts in the Post-Oncological Reconstructive Surgery of Atrophies in the Zygomatic Region: Clinical Evaluations in the Patients Undergone to Previous Radiation Therapy. Head Face Med. 2012, 8, 33. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3527323/ (accessed on 21 January 2024). [CrossRef]
- Bijella, M.F.T.B.; Bijella, V.T.; Silva, M.S.M.B.D.; Lopes, E.S. Avaliação clínica da aplicação de diamino-fluoreto de prata a 12. Rev. Paul. Odontol. 1991, 13, 28–35. [Google Scholar]
- Mauro, S.; García Robles, E.; Cinque, C.; Squassi, A.F.; Bordoni, N.E. Eficiencia de tres fluoruros concentrados para la estabilización de caries de esmalte. Bol. Asoc. Argent. Odontol. Niños 2004, 33, 4–11. [Google Scholar]
- Mei, M.L.; Lo, E.C.-M.; Chu, C.-H. Clinical Use of Silver Diamine Fluoride in Dental Treatment. Compend. Contin. Educ. Dent. 2016, 37, 93–98. [Google Scholar]
- Boccellino, M.; Di Stasio, D.; Dipalma, G.; Cantore, S.; Ambrosio, P.; Coppola, M.; Quagliuolo, L.; Scarano, A.; Malcangi, G.; Borsani, E.; et al. Steroids and Growth Factors in Oral Squamous Cell Carcinoma: Useful Source of Dental-Derived Stem Cells to Develop a Steroidogenic Model in New Clinical Strategies. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 8730–8740. [Google Scholar] [CrossRef]
- Vimercati, L.; De Maria, L.; Quarato, M.; Caputi, A.; Gesualdo, L.; Migliore, G.; Cavone, D.; Sponselli, S.; Pipoli, A.; Inchingolo, F.; et al. Association between Long COVID and Overweight/Obesity. J. Clin. Med. 2021, 10, 4143. [Google Scholar] [CrossRef]
- Inchingolo, F.; Tatullo, M.; Abenavoli, F.M.; Marrelli, M.; Inchingolo, A.D.; Gentile, M.; Inchingolo, A.M.; Dipalma, G. Non-Syndromic Multiple Supernumerary Teeth in a Family Unit with a Normal Karyotype: Case Report. Int. J. Med. Sci. 2010, 7, 378–384. [Google Scholar] [CrossRef]
- Inchingolo, F.; Ballini, A.; Cagiano, R.; Inchingolo, A.D.; Serafini, M.; De Benedittis, M.; Cortelazzi, R.; Tatullo, M.; Marrelli, M.; Inchingolo, A.M.; et al. Immediately Loaded Dental Implants Bioactivated with Platelet-Rich Plasma (PRP) Placed in Maxillary and Mandibular Region. Clin. Ter. 2015, 166, e146–e152. [Google Scholar] [CrossRef]
- Franco, R.; Gianfreda, F.; Miranda, M.; Barlattani, A.; Bollero, P. The Hemostatic Properties of Chitosan in Oral Surgery. Biomed. Biotechnol. Res. J. 2020, 4, 186. [Google Scholar] [CrossRef]
- Bollero, P.; Di Renzo, L.; Franco, R.; Rampello, T.; Pujia, A.; Merra, G.; De Lorenzo, A.; Docimo, R. Effects of New Probiotic Mouthwash in Patients with Diabetes Mellitus and Cardiovascular Diseases. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 5827–5836. [Google Scholar] [CrossRef]
- Arrigoni, R.; Ballini, A.; Santacroce, L.; Cantore, S.; Inchingolo, A.; Inchingolo, F.; Di Domenico, M.; Quagliuolo, L.; Boccellino, M. Another Look at Dietary Polyphenols: Challenges in Cancer Preventionand Treatment. Curr. Med. Chem. 2022, 29, 1061–1082. [Google Scholar] [CrossRef]
- Santacroce, L.; Di Cosola, M.; Bottalico, L.; Topi, S.; Charitos, I.A.; Ballini, A.; Inchingolo, F.; Cazzolla, A.P.; Dipalma, G. Focus on HPV Infection and the Molecular Mechanisms of Oral Carcinogenesis. Viruses 2021, 13, 559. [Google Scholar] [CrossRef]
- Inchingolo, A.D.; Inchingolo, A.M.; Bordea, I.R.; Xhajanka, E.; Romeo, D.M.; Romeo, M.; Zappone, C.M.F.; Malcangi, G.; Scarano, A.; Lorusso, F.; et al. The Effectiveness of Osseodensification Drilling Protocol for Implant Site Osteotomy: A Systematic Review of the Literature and Meta-Analysis. Materials 2021, 14, 1147. [Google Scholar] [CrossRef]
- Crincoli, V.; Scivetti, M.; Di Bisceglie, M.B.; Pilolli, G.P.; Favia, G. Unusual Case of Adverse Reaction in the Use of Sodium Hypochlorite during Endodontic Treatment: A Case Report. Quintessence Int. Berl. Ger. 2008, 39, e70–e73. [Google Scholar]
- Di Domenico, M.; Pinto, F.; Quagliuolo, L.; Contaldo, M.; Settembre, G.; Romano, A.; Coppola, M.; Ferati, K.; Bexheti-Ferati, A.; Sciarra, A.; et al. The Role of Oxidative Stress and Hormones in Controlling Obesity. Front. Endocrinol. 2019, 10, 540. [Google Scholar] [CrossRef]
- Pisano, M.; Romano, A.; Di Palo, M.P.; Baroni, A.; Serpico, R.; Contaldo, M. Oral Candidiasis in Adult and Pediatric Patients with COVID-19. Biomedicines 2023, 11, 846. [Google Scholar] [CrossRef]
- Favia, G.; Tempesta, A.; Limongelli, L.; Crincoli, V.; Piattelli, A.; Maiorano, E. Metastatic Breast Cancer in Medication-Related Osteonecrosis Around Mandibular Implants. Am. J. Case Rep. 2015, 16, 621–626. [Google Scholar] [CrossRef]
- Camerota, L.; Ritelli, M.; Wischmeijer, A.; Majore, S.; Cinquina, V.; Fortugno, P.; Monetta, R.; Gigante, L.; Marfan Syndrome Study Group Tor Vergata University Hospital; Sangiuolo, F.C.; et al. Genotypic Categorization of Loeys-Dietz Syndrome Based on 24 Novel Families and Literature Data. Genes 2019, 10, 764. [Google Scholar] [CrossRef]
- Sammartino, G.; Gasparro, R.; Marenzi, G.; Trosino, O.; Mariniello, M.; Riccitiello, F. Extraction of Mandibular Third Molars: Proposal of a New Scale of Difficulty. Br. J. Oral Maxillofac. Surg. 2017, 55, 952–957. [Google Scholar] [CrossRef]
- Saccomanno, S.; Berretin-Felix, G.; Paskay, L.C.; Manenti, R.J.; Quinzi, V. Myofunctional Therapy Part 4: Prevention and Treatment of Dentofacial and Oronasal Disorders. Eur. J. Paediatr. Dent. 2021, 22, 332–334. [Google Scholar] [CrossRef]
- Görürgöz, C.; Yangıncı, Y.; Akçam, M.O.; Orhan, K. Is It Possible to Reveal a Typical Swallowing Pattern for Specific Skeletal Malocclusion Types Using M-Mode Sonographic Imaging of Tongue Movements? J. Orofac. Orthop. 2023, 84, 392–404. [Google Scholar] [CrossRef]
- Van der Plas, P.P.J.M.; Streppel, M.; Pullens, B.; Koudstaal, M.J.; Mathijssen, I.M.J.; van Heesch, G.G.M.; Wolvius, E.B.; Joosten, K.F.M. Feeding and Swallowing Outcomes Following Mandibular Distraction Osteogenesis: An Analysis of 22 Non-Isolated Paediatric Cases. Int. J. Oral Maxillofac. Surg. 2022, 51, 892–899. [Google Scholar] [CrossRef]
- Grechi, T.H.; Itikawa, C.E.; Gallarreta, F.W.M.; Anselmo-Lima, W.T.; Valera, F.C.P.; Trawitzki, L.V.V. Effect of Rapid Maxillary Expansion on Masticatory and Swallowing Functions in Children with Posterior Crossbite. Braz. J. Otorhinolaryngol. 2023, 89, 101304. [Google Scholar] [CrossRef]
- Zhang, C.; Kimura, Y.; Matsumoto, K. The Effects of Pulsed Nd:YAG Laser Irradiation with Fluoride on Root Surface. J. Clin. Laser Med. Surg. 1996, 14, 399–403. [Google Scholar] [CrossRef]
- Miranda, M.; Martinez, L.S.; Franco, R.; Forte, V.; Barlattani, A.; Bollero, P. Differences between Warfarin and New Oral Anticoagulants in Dental Clinical Practice. Oral Implantol. 2016, 9, 151–156. [Google Scholar]
- Horst, J.A.; Ellenikiotis, H.; Milgrom, P.L. UCSF Protocol for Caries Arrest Using Silver Diamine Fluoride: Rationale, Indications and Consent. J. Calif. Dent. Assoc. 2016, 44, 16–28. [Google Scholar] [CrossRef]
- Oliveira, B.H.; Rajendra, A.; Veitz-Keenan, A.; Niederman, R. The Effect of Silver Diamine Fluoride in Preventing Caries in the Primary Dentition: A Systematic Review and Meta-Analysis. Caries Res. 2019, 53, 24–32. [Google Scholar] [CrossRef]
- Patel, M.; McTigue, D.J.; Thikkurissy, S.; Fields, H.W. Parental Attitudes Toward Advanced Behavior Guidance Techniques Used in Pediatric Dentistry. Pediatr. Dent. 2016, 38, 30–36. [Google Scholar]
- Food and Drug Administration, HHS. Food Labeling: Health Claims; Dietary Noncariogenic Carbohydrate Sweeteners and Dental Caries. Final Rule. Fed. Regist. 2006, 71, 15559–15564. [Google Scholar]
- Yamaga, R.; Nishino, M.; Yoshida, S.; Yokomizo, I. Diammine Silver Fluoride and Its Clinical Application. J. Osaka Univ. Dent. Sch. 1972, 12, 1–20. [Google Scholar]
- Ballikaya, E.; Ünverdi, G.E.; Cehreli, Z.C. Management of Initial Carious Lesions of Hypomineralized Molars (MIH) with Silver Diamine Fluoride or Silver-Modified Atraumatic Restorative Treatment (SMART): 1-Year Results of a Prospective, Randomized Clinical Trial. Clin. Oral Investig. 2022, 26, 2197–2205. [Google Scholar] [CrossRef]
- Mulder, R.; Potgieter, N.; Noordien, N. Penetration of SDF and AgF from the Infected Dentine towards the Unaffected Tooth Structure. Front. Oral Health 2023, 4, 1298211. [Google Scholar] [CrossRef]
- Sayed, M.; Nikaido, T.; Abdou, A.; Burrow, M.F.; Tagami, J. Potential Use of Silver Diammine Fluoride in Detection of Carious Dentin. Dent. Mater. J. 2021, 40, 820–826. [Google Scholar] [CrossRef]
- Ainoosah, S.E.; Levon, J.; Eckert, G.J.; Hara, A.T.; Lippert, F. Effect of Silver Diamine Fluoride on the Prevention of Erosive Tooth Wear in Vitro. J. Dent. 2020, 103, 100015. [Google Scholar] [CrossRef]
- Campanella, V.; Syed, J.; Santacroce, L.; Saini, R.; Ballini, A.; Inchingolo, F. Oral Probiotics Influence Oral and Respiratory Tract Infections in Pediatric Population: A Randomized Double-Blinded Placebo-Controlled Pilot Study. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 8034–8041. [Google Scholar] [CrossRef]
- Fanali, S.; Tumedei, M.; Pignatelli, P.; Inchingolo, F.; Pennacchietti, P.; Pace, G.; Piattelli, A. Implant Primary Stability with an Osteocondensation Drilling Protocol in Different Density Polyurethane Blocks. Comput. Methods Biomech. Biomed. Engin. 2021, 24, 14–20. [Google Scholar] [CrossRef]
- Mei, M.L.; Ito, L.; Zhang, C.F.; Lo, E.C.M.; Chu, C.H. Effect of Laser Irradiation on the Fluoride Uptake of Silver Diamine Fluoride Treated Dentine. Lasers Med. Sci. 2015, 30, 985–991. [Google Scholar] [CrossRef]
- Crystal, Y.O.; Janal, M.N.; Hamilton, D.S.; Niederman, R. Parental Perceptions and Acceptance of Silver Diamine Fluoride Staining. J. Am. Dent. Assoc. 2017, 148, 510–518.e4. [Google Scholar] [CrossRef]
- Yee, R.; Holmgren, C.; Mulder, J.; Lama, D.; Walker, D.; van Palenstein Helderman, W. Efficacy of Silver Diamine Fluoride for Arresting Caries Treatment. J. Dent. Res. 2009, 88, 644–647. [Google Scholar] [CrossRef]
- Peng, J.J.-Y.; Botelho, M.G.; Matinlinna, J.P. Silver Compounds Used in Dentistry for Caries Management: A Review. J. Dent. 2012, 40, 531–541. [Google Scholar] [CrossRef]
- Rosenblatt, A.; Stamford, T.C.M.; Niederman, R. Silver Diamine Fluoride: A Caries “Silver-Fluoride Bullet”. J. Dent. Res. 2009, 88, 116–125. [Google Scholar] [CrossRef]
- Mei, M.-L.; Chu, C.-H.; Low, K.-H.; Che, C.-M.; Lo, E.-C.-M. Caries Arresting Effect of Silver Diamine Fluoride on Dentine Carious Lesion with S. Mutans and L. Acidophilus Dual-Species Cariogenic Biofilm. Med. Oral Patol. Oral Cirugia Bucal 2013, 18, e824–e831. [Google Scholar] [CrossRef]
- Sharma, G.; Puranik, M.P.; Sowmya, K.R. Approaches to Arresting Dental Caries: An Update. J. Clin. Diagn. Res. 2015, 9, ZE08–ZE11. [Google Scholar] [CrossRef]
- Mei, M.L.; Lo, E.C.M.; Chu, C.H. Arresting Dentine Caries with Silver Diamine Fluoride: What’s Behind It? J. Dent. Res. 2018, 97, 751–758. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A Revised Tool for Assessing Risk of Bias in Randomised Trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Roberts-Thomson, K.F.; Ha, D.H.; Wooley, S.; Meihubers, S.; Do, L.G. Community Trial of Silver Fluoride Treatment for Deciduous Dentition Caries in Remote Indigenous Communities. Aust. Dent. J. 2019, 64, 175–180. [Google Scholar] [CrossRef]
- Bansal, K.; Shamoo, A.; Mani, K.; Verma, A.; Mathur, V.P.; Tewari, N. Silver Diamine Fluoride Modified Atraumatic Restorative Treatment Compared to Conventional Restorative Technique on Carious Primary Molars-A Randomized Controlled Trial. J. Dent. 2023, 138, 104698. [Google Scholar] [CrossRef]
- Hammersmith, K.J.; DePalo, J.R.; Casamassimo, P.S.; MacLean, J.K.; Peng, J. Silver Diamine Fluoride and Fluoride Varnish May Halt Interproximal Caries Progression in the Primary Dentition. J. Clin. Pediatr. Dent. 2020, 44, 79–83. [Google Scholar] [CrossRef]
- Sirivichayakul, P.; Jirarattanasopha, V.; Phonghanyudh, A.; Tunlayadechanont, P.; Khumsub, P.; Duangthip, D. The Effectiveness of Topical Fluoride Agents on Preventing Development of Approximal Caries in Primary Teeth: A Randomized Clinical Trial. BMC Oral Health 2023, 23, 349. [Google Scholar] [CrossRef]
- De Fracasso, M.L.C.; Venante, H.S.; Santin, G.C.; Salles, C.L.F.; Provenzano, M.G.A.; Maciel, S.M. Performance of Preventive Methods Applied to the Occlusal Surface of Primary Teeth: A Randomized Clinical Study. Pesqui. Bras. Odontopediatria Clin. Integr. 2018, 18, 3675. [Google Scholar] [CrossRef]
- Aly, A.A.M.; Aziz, A.M.A.; Elghazawy, R.K.; El Fadl, R.K.A. Survival Analysis and Cost Effectiveness of Silver Modified Atraumatic Restorative Treatment (SMART) and ART Occlusal Restorations in Primary Molars: A Randomized Controlled Trial. J. Dent. 2023, 128, 104379. [Google Scholar] [CrossRef]
- Sun, I.G.; Duangthip, D.; Lo, E.C.M.; Chu, C.H. The Caries-Arrest Effectiveness of Silver Diamine Fluoride Treatment with Different Post-Treatment Instructions in Preschool Children: A Study Protocol for a Randomized Controlled Trial. Dent. J. 2023, 11, 145. [Google Scholar] [CrossRef]
- Mattos-Silveira, J.; Floriano, I.; Ferreira, F.R.; Viganó, M.E.F.; Frizzo, M.A.; Reyes, A.; Novaes, T.F.; Moriyama, C.M.; Raggio, D.P.; Imparato, J.C.P.; et al. New Proposal of Silver Diamine Fluoride Use in Arresting Approximal Caries: Study Protocol for a Randomized Controlled Trial. Trials 2014, 15, 448. [Google Scholar] [CrossRef]
- Raskin, S.E.; Tranby, E.P.; Ludwig, S.; Okunev, I.; Frantsve-Hawley, J.; Boynes, S. Survival of Silver Diamine Fluoride among Patients Treated in Community Dental Clinics: A Naturalistic Study. BMC Oral Health 2021, 21, 35. [Google Scholar] [CrossRef]
- Patel, M.C.; Makwani, D.A.; Bhatt, R.K.; Raj, V.; Patel, C.; Patel, F. Evaluation of Silver-Modified Atraumatic Restorative Technique versus Conventional Pulp Therapy in Asymptomatic Deep Carious Lesion of Primary Molars—A Comparative Prospective Clinical Study. J. Indian Soc. Pedod. Prev. Dent. 2022, 40, 383–390. [Google Scholar]
- Prakash, D.K.M.; Vinay, C.; Uloopi, K.S.; RojaRamya, K.S.; Penmatsa, C.; Chandana, N. Evaluation of Caries Arresting Potential of Silver Diamine Fluoride and Sodium Fluoride Varnish in Primary Molars: A Randomized Controlled Trial. J. Indian Soc. Pedod. Prev. Dent. 2022, 40, 377. [Google Scholar]
- Thakur, S.; Sojan, M.; Singhal, P.; Chauhan, D. A Comparative Study to Evaluate the Effectiveness of Silver Diamine Fluoride at Different Time Durations of Application in Treating Carious Primary Teeth: A Randomized Trial. Int. J. Clin. Pediatr. Dent. 2022, 15, S147–S150. [Google Scholar] [CrossRef]
- Shafi, N.; Kaur, H.; Choudhary, R.; Yeluri, R. Dilute Silver Diamine Fluoride (1:10) Versus Light Cure Calcium Hydroxide as Indirect Pulp Capping Agents in Primary Molars—A Randomized Clinical Trial. J. Clin. Pediatr. Dent. 2022, 46, 273–279. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).