From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions
Abstract
:1. Introduction
- -
- By upgrading biogas from the AD of biodegradable organic matter;
- -
- Via the thermochemical conversion of biomass (gasification), followed by syngas methanation;
- -
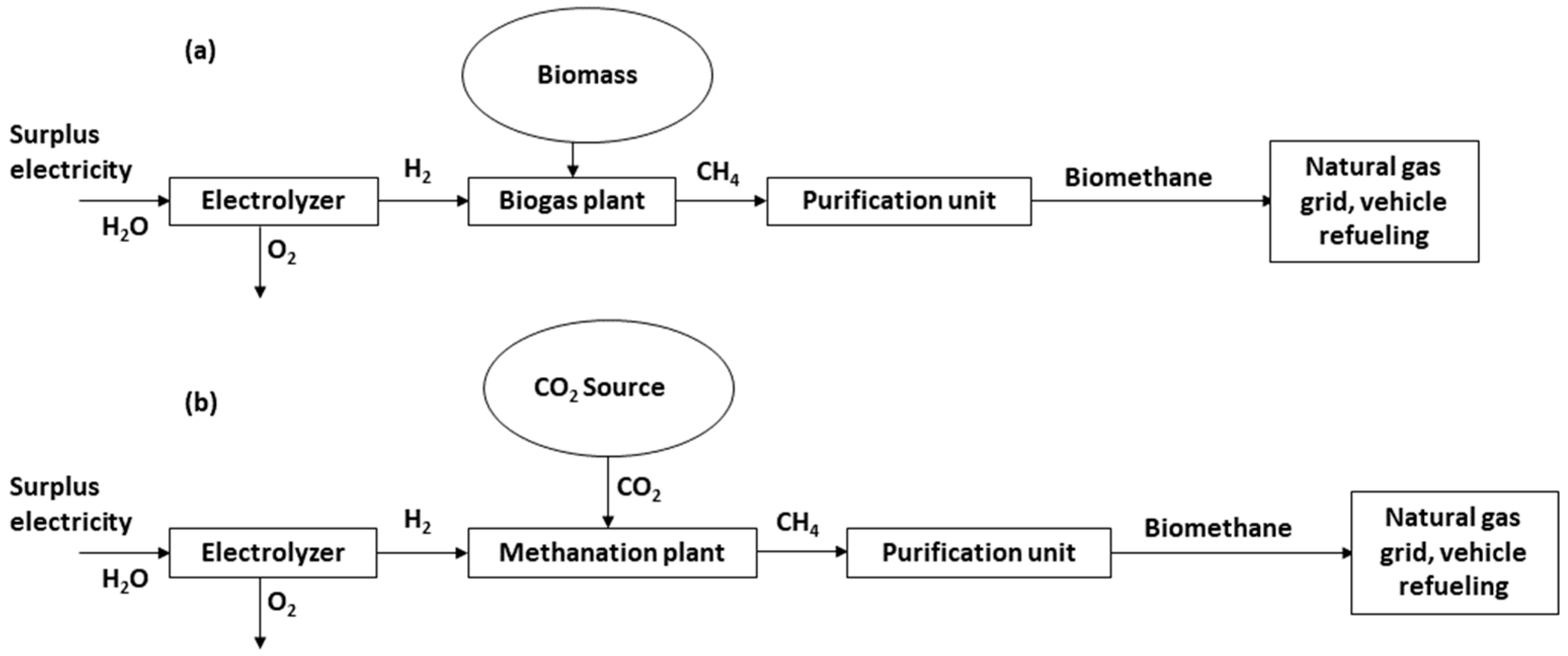
- Via the methanation of hydrogen, produced through water electrolysis, and a biogenic CO2 source stream, also called “Power-to-Gas” or, more specifically, “Power-to-Methane” (PtM).
2. Biogas Cleaning and Upgrading
3. Developed Biogas Upgrading Technologies
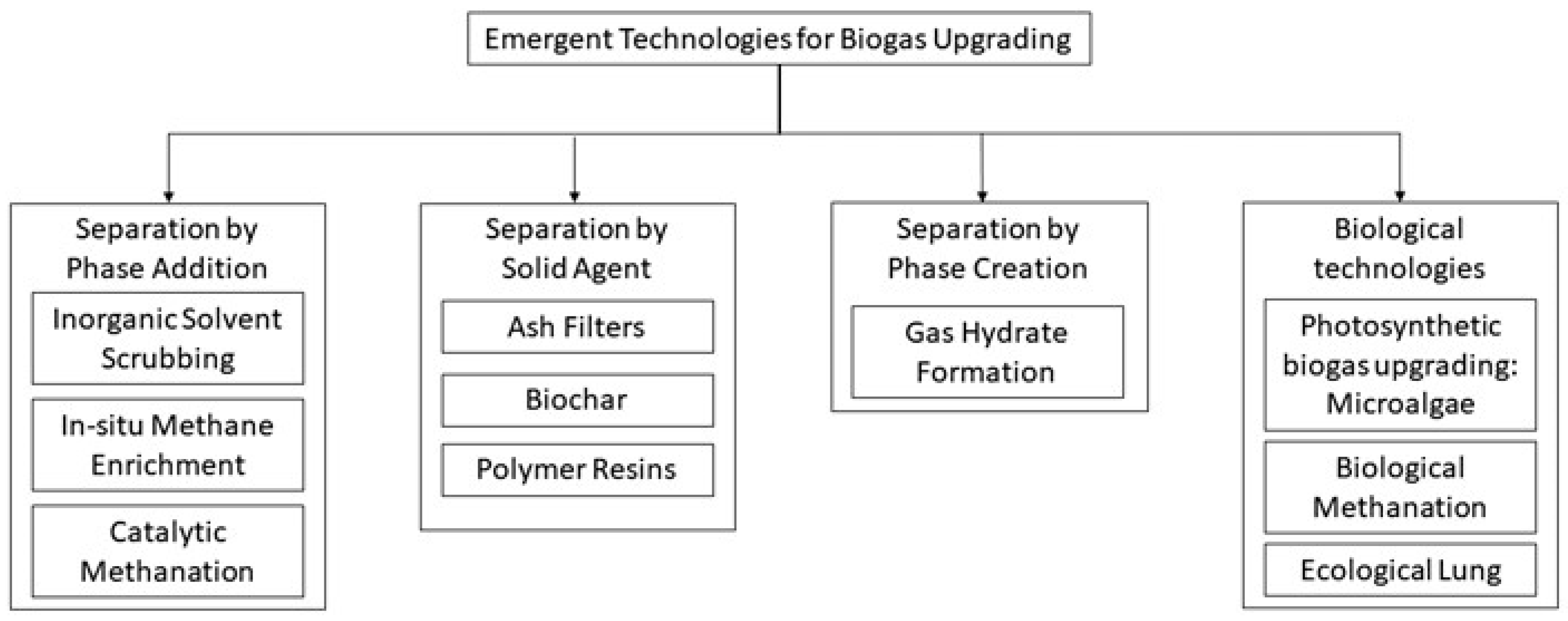
4. Emerging Biogas Upgrading Technologies
4.1. Separation via Phase Addition
4.1.1. Inorganic Solvent Scrubbing
| Solvent and Concentration | Apparatus | Operating Conditions | Biogas Composition (% Vol) | Product Purity (Highest CH4, Lowest CO2) | Loading Capacity (kg CO2/kg Solvent Solution) | Comments | Case/ Reference |
|---|---|---|---|---|---|---|---|
| 1.5 M NaOH | Glass cylinder (H = 54 cm; D = 5 cm) | Ambient T | 65.79% CH4 34.2% CO2 | 96.23% CH4 3.76% CO2 | 0.29 | Saturation after 12 min | 1/[86] (lab-scale tests) |
| 1.5 M KOH | 92.1% CH4 7.89% CO2 | 0.18 | |||||
| 0.5 M Ca(OH)2 | 85.99% CH4 14% CO2 | 0.36 | |||||
| NH4OH (10% aqueous sol.) | 97.44% CH4 2.55% CO2 | 0.27 | |||||
| 12% vol NaOH | Packed column (H = 2 m; D = 0.1 m) Packing: plastic bioballs | p = 0.1 MPa T = 298 K Liquid flow: 72 kg/h Gas flow: 6 kg/h | 59.8% CH4 40.1% CO2 | 85.9% CH4 10.8% CO2 3.3% H2O | - | NH4 scrubbing can be improved by adding a WS process afterwards to reduce NH3 concentration | 2/[85] (lab-scale tests) |
| 5% vol NH3 | 76.0% CH4 8.9% CO2 3.4% H2O 7.2% NH3 | ||||||
| 10% NaOH | Packed column (H = 1.8 m; D = 0.3 m) Packing: polyethylene balls | p = 0.1 MPa T = 303 K | 60.59% CH4 36.03% CO2 | 93.62% CH4 2.05% CO2 | - | Saturation after 6 h and cleaning of the system for corrosion after 3 h | 3/[72] (pilot-scale tests) |
| 0.1 M NaOH | Glass cylinder (H = 1 m; D = 0.07 m) Packing: plastic bioballs | p = 0.1 MPa T = 303 K | 53.1% CH4 46.8% CO2 2150 ppm H2S | 95.5% CH4 3.2% CO2 0 ppm H2S | 0.22 | Saturation after 100 min | 4/[89] (lab-scale tests) |
| 0.1 M Ca(OH)2 | 95.0% CH4 4.0% CO2 0 ppm H2S | 0.18 | Saturation after 50 min | ||||
| Pig manure: 1 g NH4+/L | Lab-scale stirrer bath | p = 0.1 Mpa T = 313 K | 70% CH4 30% CO2 | - | 0.0031 | Saturation after 15 min; fertilizer rich in N and C | 5/[90] (lab-scale tests) |
| Effluent from a digester treating pig manure: 1.1 g NH4+/L | 87.5% CH4 12.5% CO2 (theoretical) | 0.0045 | |||||
| 53% mass concentration KOH | Column filled with laboratory packing (H = 0.99 m; D = 0.08 m) | p = 0.1 Mpa T (KOH) = 313–318 K T (gas) = 288–293 K | 60% CH4 40% CO2 (landfill gas) | 97% CH4 2% CO2 | - | - | 6/[83] (pilot-scale tests) |
| 0.2 M Ca(OH)2 | - | Biogas flow: 5 L/min Solution flow: 30 L/min | 51% CH4 39.36% CO2 0.21% O2 2940.61 ppm H2S | 89.3% CH4 | - | - | 7/[91] (lab-scale tests) |
4.1.2. In Situ Methane Enrichment
4.1.3. Catalytic Methanation
4.2. Separation via a Solid Agent
4.2.1. Ash Filters
4.2.2. Biochar
4.2.3. Polymer Resins
4.3. Separation via Phase Creation
Gas Hydrate Formation
4.4. Biological Technologies
4.4.1. Photosynthetic Biogas Upgrading: Microalgae
4.4.2. Biological Methanation
4.4.3. Ecological Lung
5. Discussion and Future Perspectives
5.1. Process Complexity
5.2. Environmental Challenges
5.3. Economic Challenges
5.4. Future Research Prospects
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Communication from the Commission to the European Parliament, the European Council, the Council, the European Economic and Social Committee, and the Committee of the Regions. A Green Deal Industrial Plan for the Net-Zero Age. Available online: https://commission.europa.eu/strategy-and-policy/priorities-2019-2024/european-green-deal/green-deal-industrial-plan_en (accessed on 20 December 2023).
- Communication from the Commission to the European Parliament, the European Council, the Council, the European Economic and Social Committee, and the Committee of the Regions. The European Green Deal. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=COM%3A2019%3A640%3AFIN (accessed on 18 November 2023).
- European Commission. EU Energy in Figures—Statistical Pocketbook 2023; Publications Office of the European Union: Luxembourg, 2023. [Google Scholar]
- Communication from the Commission to the European Parliament and the Council. European Energy Security Strategy. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52014DC0330 (accessed on 20 September 2023).
- European Environment Agency. A Framework Strategy for a Resilient Energy Union with a Forward-Looking Climate Change Policy. Available online: https://www.eea.europa.eu/policy-documents/com-2015-80-final (accessed on 20 September 2023).
- European Commission. Green Paper on the Security of Energy Supply. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=LEGISSUM:l27037 (accessed on 20 September 2023).
- IEA (International Energy Agency). Russia’s War on Ukraine. Available online: https://www.iea.org/topics/russias-war-on-ukraine (accessed on 14 February 2024).
- European Biogas Association. EBA Statistical Report 2022. Available online: https://www.europeanbiogas.eu/SR-2022/EBA (accessed on 12 December 2023).
- Communication from the Commission to the European Parliament, the European Council, the Council, the European Economic and Social Committee, and the Committee of the Regions. REPowerEU Plan. Available online: https://commission.europa.eu/strategy-and-policy/priorities-2019-2024/european-green-deal/repowereu-affordable-secure-and-sustainable-energy-europe_es (accessed on 20 December 2023).
- European Commission. Climate Strategies and Targets 2018. Available online: https://ec.europa.eu/clima/policies/strategies_en (accessed on 26 September 2023).
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions: Energy 2020—A Strategy for Competitive, Sustainable and Secure Energy. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=LEGISSUM:en0024 (accessed on 20 December 2023).
- El País Economía. Available online: https://elpais.com/economia/2018/05/04/actualidad/1525444638_932682.html (accessed on 26 May 2023).
- Perez, A. La Trampa Global Del Gas. Un Puente al Desastre, 1st ed.; Rosa Luxemburg Stiftung; ODG: Brussels, Belgium, 2017. [Google Scholar]
- Kollamthodi, S.; Norris, J.; Dun, C.; Brannigan, C.; Twisse, F.; Biedka, M.; Bates, J. The Role of Natural Gas and Biomethane in the Transport Sector; Ricardo Energy & Environment: Harwell Oxford, UK, 2016. [Google Scholar]
- European Commission. Methane Emissions. Available online: https://energy.ec.europa.eu/topics/oil-gas-and-coal/methane-emissions_en (accessed on 13 February 2024).
- Financial Times. Available online: https://www.ft.com/content/2cce7954-7587-11e8-b326-75a27d27ea5f (accessed on 15 June 2023).
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions on an EU Strategy to Reduce Methane Emissions. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A52020DC0663 (accessed on 26 November 2023).
- Appels, L.; Baeyens, J.; Degrève, J.; Dewil, R. Principles and Potential of the Anaerobic Digestion of Waste-Activated Sludge. Prog. Energy Combust. Sci. 2008, 34, 755–781. [Google Scholar] [CrossRef]
- Martín-Hernández, E.; Guerras, L.S.; Martín, M. Optimal Technology Selection for the Biogas Upgrading to Biomethane. J. Clean. Prod. 2020, 267, 122032. [Google Scholar] [CrossRef]
- Nguyen, L.N.; Kumar, J.; Vu, M.T.; Mohammed, J.A.H.; Pathak, N.; Commault, A.S.; Sutherland, D.; Zdarta, J.; Tyagi, V.K.; Nghiem, L.D. Biomethane Production from Anaerobic Co-Digestion at Wastewater Treatment Plants: A Critical Review on Development and Innovations in Biogas Upgrading Techniques. Sci. Total Environ. 2021, 765, 142753. [Google Scholar] [CrossRef] [PubMed]
- Duic, N.; Cajner, F.; Puksec, T. Factsheets on Current Biogas/Biomethane Handling Practices; University of Zagreb: Zagreb, Croatia, 2016. [Google Scholar]
- IRENA (International Renewable Energy Agency). Biogas for Road Vehicles: Technology Brief. Available online: https://www.irena.org/publications/2017/Mar/Biogas-for-road-vehicles-Technology-brief (accessed on 15 November 2023).
- European Commission. Mandate to CEN for Standards for Biomethane for Use in Transport and Injection in Natural Gas Pipelines 2010. Available online: https://law.resource.org/pub/eu/mandates/m475.pdf (accessed on 20 September 2023).
- European Commission. Assistance to Assessing Options Improving Market Conditions for Bio-Methane and Gas Market Rules. Available online: https://op.europa.eu/en/publication-detail/-/publication/d24343db-5ee8-11ec-9c6c-01aa75ed71a1/language-en (accessed on 28 December 2023).
- European Biogas Association (EBA). Six National Biomethane Registries Are Developing the Foundation for Cross-Border Biomethane Trade in Europe. Available online: https://www.europeanbiogas.eu/six-national-biomethane-registries-developing-foundation-cross-border-biomethane-trade-europe/ (accessed on 26 October 2023).
- Álvarez Rodríguez, J. La Co-Digestión Anaerobia Como Un Sistema de Producción Energética. In Biocombustibles Retos y Oportunidades; Lema Rodicio, J.M., Bello Bugallo, P.M., Eds.; USC: Santiago de Compostela, Spain, 2009; ISBN 978-84-613-46-721. [Google Scholar]
- Achinas, S.; Achinas, V.; Euverink, G.J.W. A Technological Overview of Biogas Production from Biowaste. Engineering 2017, 3, 299–307. [Google Scholar] [CrossRef]
- Ariunbaatar, J.; Panico, A.; Esposito, G.; Pirozzi, F.; Lens, P.N.L. Pretreatment Methods to Enhance Anaerobic Digestion of Organic Solid Waste. Appl. Energy 2014, 123, 143–156. [Google Scholar] [CrossRef]
- van Lier, J.B.; Mahmoud, N.; Zeeman, G. Anaerobic Wastewater Treatment; IWA Publishing: London, UK, 2008. [Google Scholar]
- Angelidaki, I.; Xie, L.; Luo, G.; Zhang, Y.; Oechsner, H.; Lemmer, A.; Munoz, R.; Kougias, P.G. Biogas Upgrading: Current and Emerging Technologies. In Biofuels: Alternative Feedstocks and Conversion Processes for the Production of Liquid and Gaseous Biofuels; Elsevier: Amsterdam, The Netherlands, 2019; pp. 817–843. [Google Scholar]
- Sikarwar, V.S.; Zhao, M.; Fennell, P.S.; Shah, N.; Anthony, E.J. Progress in Biofuel Production from Gasification. Prog. Energy Combust. Sci. 2017, 61, 189–248. [Google Scholar] [CrossRef]
- Rajendran, K.; Drielak, E.; Sudarshan Varma, V.; Muthusamy, S.; Kumar, G. Updates on the Pretreatment of Lignocellulosic Feedstocks for Bioenergy Production—A Review. Biomass Convers. Biorefin 2018, 8, 471–483. [Google Scholar] [CrossRef]
- Molino, A.; Chianese, S.; Musmarra, D. Biomass Gasification Technology: The State of the Art Overview. J. Energy Chem. 2016, 25, 10–25. [Google Scholar] [CrossRef]
- Woolcock, P.J.; Brown, R.C. A Review of Cleaning Technologies for Biomass-Derived Syngas. Biomass Bioenergy 2013, 52, 54–84. [Google Scholar] [CrossRef]
- Le, T.A.; Kim, M.S.; Lee, S.H.; Kim, T.W.; Park, E.D. CO and CO2 Methanation over Supported Ni Catalysts. Catal. Today 2017, 293–294, 89–96. [Google Scholar] [CrossRef]
- Götz, M.; Lefebvre, J.; Mörs, F.; McDaniel Koch, A.; Graf, F.; Bajohr, S.; Reimert, R.; Kolb, T. Renewable Power-to-Gas: A Technological and Economic Review. Renew. Energy 2016, 85, 1371–1390. [Google Scholar] [CrossRef]
- Mazza, A.; Bompard, E.; Chicco, G. Applications of Power to Gas Technologies in Emerging Electrical Systems. Renew. Sustain. Energy Rev. 2018, 92, 794–806. [Google Scholar] [CrossRef]
- Ghaib, K.; Ben-Fares, F.-Z. Power-to-Methane: A State-of-the-Art Review. Renew. Sustain. Energy Rev. 2018, 81, 433–446. [Google Scholar] [CrossRef]
- EBA. Statistical Report of the European Biogas Association 2023. Available online: https://www.europeanbiogas.eu/eba-statistical-report-2023/ (accessed on 12 February 2024).
- Redexis Foundation. Available online: https://fundacion.redexis.es/assets/docs/Redexis-BIOMETANO_23062022.pdf (accessed on 16 February 2024).
- The European Network of Transmission System Operators for Gas (ENTSOG). Ten Years Network Development Plan. Scenario Report. Annex II: Methodology. Available online: https://www.entsog.eu/tyndp#entsog-ten-year-network-development-plan-2018 (accessed on 20 December 2023).
- AEBIOM. A Biogas Road Map for Europe. Brussels: 2009. Available online: https://www.big-east.eu/downloads/Brochure_BiogasRoadmap_WEB%5B1%5D.pdf (accessed on 3 September 2023).
- Kampman, B.; Leguijt, C.; Scholten, T.; Tallat-Kelpsaite, J.; Brückmann, R.; Maroulis, G.; Lesschen, J.P.; Meesters, K.; Sikirica, N.; Elbersen, B. Optimal Use of Biogas from Waste Streams: An Assessment of the Potential of Biogas from Digestion in the EU Beyond 2020; European Commission: Brussels, Belgium, 2017. [Google Scholar]
- EBA. How Gas Can Help to Achievethe Paris Agreement Target in an Affordable Way. Available online: https://www.europeanbiogas.eu/how-gas-can-help-to-achievethe-paris-agreement-target-in-an-affordable-way/ (accessed on 20 September 2023).
- Svensson, M. Biomethane for Transport Applications. In The Biogas Handbook; Elsevier: Amsterdam, The Netherlands, 2013; pp. 428–443. [Google Scholar]
- Statistical Report of the European Biogas Association 2018. Available online: https://www.europeanbiogas.eu/eba-statistical-report-2018/ (accessed on 3 September 2023).
- Green Gas Grids. Proposal for a European Biomethane Roadmap. Available online: https://www.europeanbiogas.eu/green-gas-grids-project-proposal-european-biomethane-roadmap/ (accessed on 22 October 2023).
- International Energy Agency. Natural Gas Information; International Energy Agency: Paris, France, 2018. [Google Scholar]
- Petersson, A.; Wellinger, A. Biogas Upgrading Technologies—Developments and Innovations; IEA Bioenergy: Paris, France, 2010. [Google Scholar]
- Ryckebosch, E.; Drouillon, M.; Vervaeren, H. Techniques for Transformation of Biogas to Biomethane. Biomass Bioenergy 2011, 35, 1633–1645. [Google Scholar] [CrossRef]
- Awe, O.W.; Zhao, Y.; Nzihou, A.; Minh, D.P.; Lyczko, N. A Review of Biogas Utilisation, Purification and Upgrading Technologies. Waste Biomass Valoriz. 2017, 8, 267–283. [Google Scholar] [CrossRef]
- Muñoz, R.; Meier, L.; Diaz, I.; Jeison, D. A Review on the State-of-the-Art of Physical/Chemical and Biological Technologies for Biogas Upgrading. Rev. Environ. Sci. Biotechnol. 2015, 14, 727–759. [Google Scholar] [CrossRef]
- Gaj, K.; Cichuta, K. Combined Biological Method for Simultaneous Removal of Hydrogen Sulphide and Volatile Methylsiloxanes from Biogas. Energies 2022, 16, 100. [Google Scholar] [CrossRef]
- Miltner, M.; Makaruk, A.; Harasek, M. Review on Available Biogas Upgrading Technologies and Innovations towards Advanced Solutions. J. Clean. Prod. 2017, 161, 1329–1337. [Google Scholar] [CrossRef]
- Linders, M.; Latsuzbaia, R.; Van Heck, R.; Goetheer, E. A Novel Electrochemical Process for H2S Removal from Biogas. In Proceedings of the 5th International Conference on Renewable Energy Gas Technology, Tolouse, France, 3–4 May 2018. [Google Scholar]
- Atelge, M.R.; Senol, H.; Djaafri, M.; Hansu, T.A.; Krisa, D.; Atabani, A.; Eskicioglu, C.; Muratçobanoğlu, H.; Unalan, S.; Kalloum, S.; et al. A Critical Overview of the State-of-the-Art Methods for Biogas Purification and Utilization Processes. Sustainability 2021, 13, 11515. [Google Scholar] [CrossRef]
- Gaj, K. Adsorptive Biogas Purification from Siloxanes—A Critical Review. Energies 2020, 13, 2605. [Google Scholar] [CrossRef]
- Singh, R.; Shukla, A. A Review on Methods of Flue Gas Cleaning from Combustion of Biomass. Renew. Sustain. Energy Rev. 2014, 29, 854–864. [Google Scholar] [CrossRef]
- Petersson, A. Biogas Cleaning. In The Biogas Handbook; Elsevier: Amsterdam, The Netherlands, 2013; pp. 329–341. [Google Scholar]
- Seader, J.D.; Henley, E.J. Separation Process Principles, 2nd ed.; Wiley: Hoboken, NJ, USA, 2006; ISBN 978-0471464808. [Google Scholar]
- Ullah Khan, I.; Hafiz Dzarfan Othman, M.; Hashim, H.; Matsuura, T.; Ismail, A.F.; Rezaei-DashtArzhandi, M.; Wan Azelee, I. Biogas as a Renewable Energy Fuel—A Review of Biogas Upgrading, Utilisation and Storage. Energy Convers. Manag. 2017, 150, 277–294. [Google Scholar] [CrossRef]
- Chen, X.Y.; Vinh-Thang, H.; Ramirez, A.A.; Rodrigue, D.; Kaliaguine, S. Membrane Gas Separation Technologies for Biogas Upgrading. RSC Adv. 2015, 5, 24399–24448. [Google Scholar] [CrossRef]
- Aile, N. Biogas to Biomethane Technology Review; Vienna University of Technology: Vienna, Austria, 2012. [Google Scholar]
- Kadam, R.; Panwar, N.L. Recent Advancement in Biogas Enrichment and Its Applications. Renew. Sustain. Energy Rev. 2017, 73, 892–903. [Google Scholar] [CrossRef]
- Bauer, F.; Hulteberg, C.P.; Persson, T.; Tamm, D. Biogas Upgrading—Review of Commercial Technologies; Lund University: Lund, Sweden, 2013. [Google Scholar]
- Sun, Q.; Li, H.; Yan, J.; Liu, L.; Yu, Z.; Yu, X. Selection of Appropriate Biogas Upgrading Technology—A Review of Biogas Cleaning, Upgrading and Utilisation. Renew. Sustain. Energy Rev. 2015, 51, 521–532. [Google Scholar] [CrossRef]
- Zhou, K.; Chaemchuen, S.; Verpoort, F. Alternative Materials in Technologies for Biogas Upgrading via CO2 Capture. Renew. Sustain. Energy Rev. 2017, 79, 1414–1441. [Google Scholar] [CrossRef]
- Abdeen, F.R.H.; Mel, M.; Jami, M.S.; Ihsan, S.I.; Ismail, A.F. A Review of Chemical Absorption of Carbon Dioxide for Biogas Upgrading. Chin. J. Chem. Eng. 2016, 24, 693–702. [Google Scholar] [CrossRef]
- Beil, M.; Beyrich, W. Biogas Upgrading to Biomethane. In The Biogas Handbook; Elsevier: Amsterdam, The Netherlands, 2013; pp. 342–377. [Google Scholar]
- Scholz, M.; Frank, B.; Stockmeier, F.; Falß, S.; Wessling, M. Techno-Economic Analysis of Hybrid Processes for Biogas Upgrading. Ind. Eng. Chem. Res. 2013, 52, 16929–16938. [Google Scholar] [CrossRef]
- Yang, L.; Ge, X. Biogas and Syngas Upgrading. Adv. Bioenergy 2016, 1, 125–188. [Google Scholar]
- Zhao, Q.; Leonhardt, E.; MacConnell, C.; Frear, C.; Chen, S. Purification Technologies for Biogas Generated by Anaerobic Digestion. CSANR Research Report 2010-001. In Climate Friendly Farming; Compressed Biomethane; Washington State University: Wenatchee, WA, USA, 2010; Chapter 9; pp. 1–24. [Google Scholar]
- Esposito, E.; Dellamuzia, L.; Moretti, U.; Fuoco, A.; Giorno, L.; Jansen, J.C. Simultaneous Production of Biomethane and Food Grade CO2 from Biogas: An Industrial Case Study. Energy Environ. Sci. 2019, 12, 281–289. [Google Scholar] [CrossRef]
- Baena-Moreno, F.M.; Rodríguez-Galán, M.; Vega, F.; Vilches, L.F.; Navarrete, B.; Zhang, Z. Biogas Upgrading by Cryogenic Techniques. Environ. Chem. Lett. 2019, 17, 1251–1261. [Google Scholar] [CrossRef]
- Angelidaki, I.; Treu, L.; Tsapekos, P.; Luo, G.; Campanaro, S.; Wenzel, H.; Kougias, P.G. Biogas Upgrading and Utilization: Current Status and Perspectives. Biotechnol. Adv. 2018, 36, 452–466. [Google Scholar] [CrossRef] [PubMed]
- Niesner, J.; Jecha, D.; Stehlik, P. Biogas Upgrading Technologies: State of Art Review in European Region. Chem. Eng. Trans. 2013, 35, 517–522. [Google Scholar] [CrossRef]
- Sahota, S.; Shah, G.; Ghosh, P.; Kapoor, R.; Sengupta, S.; Singh, P.; Vijay, V.; Sahay, A.; Vijay, V.K.; Thakur, I.S. Review of Trends in Biogas Upgradation Technologies and Future Perspectives. Bioresour. Technol. Rep. 2018, 1, 79–88. [Google Scholar] [CrossRef]
- Bailón Allegue, L.; Hinge, J. Biogas and Bio-Syngas Upgrading; Danish Technological Institute: Aarhus, Denmark, 2012. [Google Scholar]
- Blom, H.; Mccann, M.; Westman, J. Small-Scale Upgrading and Refinement of Biogas; Poeyry SwedPower AB: Stockholm, Sweden, 2012. [Google Scholar]
- International Energy Agency. Biogas Upgrading Plants List. Available online: https://www.iea.org/reports/outlook-for-biogas-and-biomethane-prospects-for-organic-growth/an-introduction-to-biogas-and-biomethane (accessed on 22 September 2023).
- Yang, L.; Ge, X.; Wan, C.; Yu, F.; Li, Y. Progress and Perspectives in Converting Biogas to Transportation Fuels. Renew. Sustain. Energy Rev. 2014, 40, 1133–1152. [Google Scholar] [CrossRef]
- Huang, H.; Chang, S.-G.; Dorchak, T. Method to Regenerate Ammonia for the Capture of Carbon Dioxide. Energy Fuels 2002, 16, 904–910. [Google Scholar] [CrossRef]
- Lombardia, L.; Corti, A.; Carnevale, E.; Baciocchi, R.; Zingaretti, D. Carbon Dioxide Removal and Capture for Landfill Gas Up-Grading. Energy Procedia 2011, 4, 465–472. [Google Scholar] [CrossRef]
- McLeod, A.; Jefferson, B.; McAdam, E.J. Biogas Upgrading by Chemical Absorption Using Ammonia Rich Absorbents Derived from Wastewater. Water Res. 2014, 67, 175–186. [Google Scholar] [CrossRef]
- Abdeen, F.R.H.; Mel, M.; Jami, M.S.; Ihsan, S.I.; Ismail, A.F. Improvement of Biogas Upgrading Process using Chemical Absorption at Ambient Conditions. J. Teknol. 2017, 80, 1. [Google Scholar] [CrossRef]
- Ahmad, F.; Khan, S.; Gupta, N. Application of Alkaline Chemicals for Biogas Methane Enrichment. Ecol. Environ. Conserv. 2016, 22, 983–992. [Google Scholar]
- Siriwardane, R.V.; Robinson, C.; Shen, M.; Simonyi, T. Novel Regenerable Sodium-Based Sorbents for CO2 Capture at Warm Gas Temperatures. Energy Fuels 2007, 21, 2088–2097. [Google Scholar] [CrossRef]
- Yeh, J.T.; Resnik, K.P.; Rygle, K.; Pennline, H.W. Semi-Batch Absorption and Regeneration Studies for CO2 Capture by Aqueous Ammonia. Fuel Process. Technol. 2005, 86, 1533–1546. [Google Scholar] [CrossRef]
- Tippayawong, N.; Thanompongchart, P. Biogas Quality Upgrade by Simultaneous Removal of CO2 and H2S in a Packed Column Reactor. Energy 2010, 35, 4531–4535. [Google Scholar] [CrossRef]
- Unidad Mixta de Movilidad Sostenible. El Gas Natural y El Biometano Como Combustible Para Movilidad Marítima y Agraria. Available online: https://energylab.es/listado/unidad-mixta-movilidad-sostenible/ (accessed on 20 May 2023).
- Srichat, A.; Suntivarakorn, R.; Kamwilaisak, K. A Development of Biogas Purification System Using Calcium Hydroxide and Amine Solution. Energy Procedia 2017, 138, 441–445. [Google Scholar] [CrossRef]
- Hsueh, H.T.; Chien, T.W.; Ko, T.H.; Huang, T.T.; Chu, H. The Regeneration of Ammonia Solution by Calcium Hydroxide for Carbon Dioxide Absorption from the Flue Gas. Glob. Environ. Eng. 2015, 2, 23–28. [Google Scholar] [CrossRef]
- Baciocchi, R.; Carnevale, E.; Costa, G.; Lombardi, L.; Olivieri, T.; Paradisi, A.; Zanchi, L.; Zingaretti, D. Pilot-Scale Investigation of an Innovative Process for Biogas Upgrading with CO2 Capture and Storage. Energy Procedia 2013, 37, 6026–6034. [Google Scholar] [CrossRef]
- Baciocchi, R.; Carnevale, E.; Corti, A.; Costa, G.; Lombardi, L.; Olivieri, T.; Zanchi, L.; Zingaretti, D. Innovative Process for Biogas Upgrading with CO2 Storage: Results from Pilot Plant Operation. Biomass Bioenergy 2013, 53, 128–137. [Google Scholar] [CrossRef]
- Baciocchi, R.; Carnevale, E.; Costa, G.; Gavasci, R.; Lombardi, L.; Olivieri, T.; Zanchi, L.; Zingaretti, D. Performance of a Biogas Upgrading Process Based on Alkali Absorption with Regeneration Using Air Pollution Control Residues. Waste Manag. 2013, 33, 2694–2705. [Google Scholar] [CrossRef] [PubMed]
- Astrup, T.F. Management of APC Residues from W-t-E Plants. An Overview of Management Options and Treatment Methods; ISWA General Secretariat: Rotterdam, The Netherlands, 2008. [Google Scholar]
- UK Essays. Air Pollution Control Residues. Available online: https://www.ukessays.com/essays/engineering/air-pollution-control-residues.php?cref=1 (accessed on 26 May 2023).
- Yellezuome, D.; Zhu, X.; Liu, X.; Liu, X.; Liu, R.; Wang, Z.; Li, Y.; Sun, C.; Hemida Abd-Alla, M.; Rasmey, A.-H.M. Integration of Two-Stage Anaerobic Digestion Process with in Situ Biogas Upgrading. Bioresour. Technol. 2023, 369, 128475. [Google Scholar] [CrossRef]
- Boontawee, S.; Koonaphapdeelert, S. In-Situ Biomethane Enrichment by Recirculation of Biogas Channel Digester Effluent Using Gas Stripping Column. Energy Procedia 2016, 89, 78–84. [Google Scholar] [CrossRef]
- Hansson, M.; Laurell, J.; Nordberg, Å.; Rasmuson, Å.C.; Liu, J.; Nistor, M.F.; Strömberg, S.; Costa, J. In-Situ Methane Enrichment of Raw Biogas in the Anaerobic Digestion Process; Svenskt Gastekniskt Center AB: Malmö, Sweden, 2013. [Google Scholar]
- Andersson, J.; Olsson, H.; Ascue, J.; Rogstrand, G.; Edström, M.; Nordberg, Å. In-Situ Methane Enrichment during Anaerobic Digestion of Manure—Pilot Scale Tests; Report 50; Swedish Institute of Agricultural and Environmental Engineering: Uppsala, Sweden, 2014. [Google Scholar]
- Lecker, B.; Illi, L.; Lemmer, A.; Oechsner, H. Biological Hydrogen Methanation—A Review. Bioresour. Technol. 2017, 245, 1220–1228. [Google Scholar] [CrossRef]
- Memon, M.; Jiang, Y.; Hassan, M.; Ajmal, M.; Wang, H.; Liu, Y. Heterogeneous Catalysts for Carbon Dioxide Methanation: A View on Catalytic Performance. Catalysts 2023, 13, 1514. [Google Scholar] [CrossRef]
- Tan, C.H.; Nomanbhay, S.; Shamsuddin, A.H.; Park, Y.-K.; Hernández-Cocoletzi, H.; Show, P.L. Current Developments in Catalytic Methanation of Carbon Dioxide—A Review. Front. Energy Res. 2022, 9, 795423. [Google Scholar] [CrossRef]
- Rönsch, S.; Schneider, J.; Matthischke, S.; Schlüter, M.; Götz, M.; Lefebvre, J.; Prabhakaran, P.; Bajohr, S. Review on Methanation—From Fundamentals to Current Projects. Fuel 2016, 166, 276–296. [Google Scholar] [CrossRef]
- Khan, M.U.; Lee, J.T.E.; Bashir, M.A.; Dissanayake, P.D.; Ok, Y.S.; Tong, Y.W.; Shariati, M.A.; Wu, S.; Ahring, B.K. Current Status of Biogas Upgrading for Direct Biomethane Use: A Review. Renew. Sustain. Energy Rev. 2021, 149, 111343. [Google Scholar] [CrossRef]
- Dannesboe, C.; Hansen, J.B.; Johannsen, I. Catalytic Methanation of CO2 in Biogas: Experimental Results from a Reactor at Full Scale. React. Chem. Eng. 2020, 5, 183–189. [Google Scholar] [CrossRef]
- Balzarotti, R.; Drago Ferrante, G.; Italiano, C.; Laganà, M.; Francis, L.F.; Vita, A.; Cristiani, C.; Pino, L. RhNi/CeO2 Catalytic Activation of Alumina Open Cell Foams by Dip-Spin Coating for the CO2 Methanation of Biogas. Surf. Coat. Technol. 2022, 441, 128563. [Google Scholar] [CrossRef]
- Guilera, J.; Filipe, M.; Montesó, A.; Mallol, I.; Andreu, T. Carbon Footprint of Synthetic Natural Gas through Biogas Catalytic Methanation. J. Clean. Prod. 2021, 287, 125020. [Google Scholar] [CrossRef]
- Tommasi, M.; Degerli, S.N.; Ramis, G.; Rossetti, I. Advancements in CO2 Methanation: A Comprehensive Review of Catalysis, Reactor Design and Process Optimization. Chem. Eng. Res. Des. 2024, 201, 457–482. [Google Scholar] [CrossRef]
- Kusnere, Z.; Spalvins, K.; Bataitis, M. Wood Ash Filter Material Characterization as a Carrier Material for Ex-Situ Biomethanation of Biogas in Biotrickling Filter Reactors. Environ. Clim. Technol. 2023, 27, 92–102. [Google Scholar] [CrossRef]
- Andersson, J. Uppgradering Av Biogas Med Aska Från Trädbränslen; Swedish University of Agricultural Sciences: Uppsala, Sweden, 2013; p. 48. [Google Scholar]
- Mostbauer, P.; Lombardi, L.; Olivieri, T.; Lenz, S. Pilot Scale Evaluation of the BABIU Process—Upgrading of Landfill Gas or Biogas with the Use of MSWI Bottom Ash. Waste Manag. 2014, 34, 125–133. [Google Scholar] [CrossRef]
- Han, B.; Yu, X.; Ou, J. Self-Sensing Concrete in Smart Structures, 1st ed.; Butterworth-Heinemann: Oxford, UK, 2014; ISBN 9780128005170. [Google Scholar]
- Gopinath, S.; Mehra, A. Carbon Dioxide Sequestration Using Steel Slag—Modeling and Experimental Investigation. In Carbon Dioxide Sequestration in Cementitious Construction Materials; Pacheco-Torgal, F., Shi, C., Palomo Sanchez, A., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 65–80. [Google Scholar]
- Andersson, J.; Nordberg, Å. Biogas Upgrading Using Ash from Combustion of Wood Fuels: Laboratory Experiments. Energy Environ. Res. 2017, 7, 38. [Google Scholar] [CrossRef]
- Fernández-Delgado Juárez, M.; Mostbauer, P.; Knapp, A.; Müller, W.; Tertsch, S.; Bockreis, A.; Insam, H. Biogas Purification with Biomass Ash. Waste Manag. 2018, 71, 224–232. [Google Scholar] [CrossRef]
- Sarperi, L.; Surbrenat, A.; Kerihuel, A.; Chazarenc, F. The Use of an Industrial By-Product as a Sorbent to Remove CO2 and H2S from Biogas. J. Environ. Chem. Eng. 2014, 2, 1207–1213. [Google Scholar] [CrossRef]
- Shen, Y.; Linville, J.L.; Ignacio-de Leon, P.A.A.; Schoene, R.P.; Urgun-Demirtas, M. Towards a Sustainable Paradigm of Waste-to-Energy Process: Enhanced Anaerobic Digestion of Sludge with Woody Biochar. J. Clean. Prod. 2016, 135, 1054–1064. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, H.; He, S.; Zhao, Q.; Wei, L. A Review of Biochar in Anaerobic Digestion to Improve Biogas Production: Performances, Mechanisms and Economic Assessments. Bioresour. Technol. 2021, 341, 125797. [Google Scholar] [CrossRef] [PubMed]
- Younas, M.; Sohail, M.; Leong, L.K.; Bashir, M.J.; Sumathi, S. Feasibility of CO2 Adsorption by Solid Adsorbents: A Review on Low-Temperature Systems. Int. J. Environ. Sci. Technol. 2016, 13, 1839–1860. [Google Scholar] [CrossRef]
- Lee, J.; Kim, K.-H.; Kwon, E.E. Biochar as a Catalyst. Renew. Sustain. Energy Rev. 2017, 77, 70–79. [Google Scholar] [CrossRef]
- Shen, Y.; Linville, J.L.; Urgun-Demirtas, M.; Schoene, R.P.; Snyder, S.W. Producing Pipeline-Quality Biomethane via Anaerobic Digestion of Sludge Amended with Corn Stover Biochar with In-Situ CO2 Removal. Appl. Energy 2015, 158, 300–309. [Google Scholar] [CrossRef]
- Sahota, S.; Vijay, V.K.; Subbarao, P.M.V.; Chandra, R.; Ghosh, P.; Shah, G.; Kapoor, R.; Vijay, V.; Koutu, V.; Thakur, I.S. Characterization of Leaf Waste Based Biochar for Cost Effective Hydrogen Sulphide Removal from Biogas. Bioresour. Technol. 2018, 250, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Linville, J.L.; Shen, Y.; Ignacio-de Leon, P.A.; Schoene, R.P.; Urgun-Demirtas, M. In-Situ Biogas Upgrading during Anaerobic Digestion of Food Waste Amended with Walnut Shell Biochar at Bench Scale. Waste Manag. Res. J. A Sustain. Circ. Econ. 2017, 35, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Raab, K.; Lamprecht, M.; Brechtel, K.; Scheffknecht, G. Innovative CO2 Separation of Biogas by Polymer Resins: Operation of a Continuous Lab-scale Plant. Eng. Life Sci. 2012, 12, 327–335. [Google Scholar] [CrossRef]
- Safai, D.; Schmid, M.-O.; Scheffknecht, G. Biogas Upgrading with Polymer Resins in a Technical Scale Plant—Scale-up and First Operating Points. In Proceedings of the 5th International Conference on Renewable Energy Gas Technology, Tolouse, France, 3–4 May 2018. [Google Scholar]
- Meng, Y.; Jiang, J.; Gao, Y.; Aihemaiti, A.; Ju, T.; Xu, Y.; Liu, N. Biogas Upgrading to Methane: Application of a Regenerable Polyethyleneimine-Impregnated Polymeric Resin (NKA-9) via CO2 Sorption. Chem. Eng. J. 2019, 361, 294–303. [Google Scholar] [CrossRef]
- Arca, S.; Poletti, L.; Poletti, R.; D’Alessandro, E. Upgrading of Biogas Technology through the Application of Gas Hydrates. In Proceedings of the 7th International Conference on Gas Hydrates (ICGH 2011), Edinburgh, UK, 17–21 July 2011; Curran Associates: New York, NY, USA, 2011. [Google Scholar]
- Castellani, B.; Rossi, F.; Filipponi, M.; Nicolini, A. Hydrate-Based Removal of Carbon Dioxide and Hydrogen Sulphide from Biogas Mixtures: Experimental Investigation and Energy Evaluations. Biomass Bioenergy 2014, 70, 330–338. [Google Scholar] [CrossRef]
- Di Profio, P.; Canale, V.; D’Alessandro, N.; Germani, R.; Di Crescenzo, A.; Fontana, A. Separation of CO2 and CH4 from Biogas by Formation of Clathrate Hydrates: Importance of the Driving Force and Kinetic Promoters. ACS Sustain. Chem. Eng. 2017, 5, 1990–1997. [Google Scholar] [CrossRef]
- Sinehbaghizadeh, S.; Saptoro, A.; Amjad-Iranagh, S.; Mohammadi, A.H. Understanding the Influences of Different Associated Gas Impurities and the Kinetic Modelling of Biogas Hydrate Formation at the Molecular Scale. Energy 2023, 282, 128893. [Google Scholar] [CrossRef]
- EBI ChEBI Database. Available online: https://www.ebi.ac.uk/chebi/searchId.do?chebiId=CHEBI:132140 (accessed on 30 December 2023).
- Moghaddam, E.A.; Larsolle, A.; Tidåker, P.; Nordberg, Å. Gas Hydrates as a Means for Biogas and Biomethane Distribution. Front. Energy Res. 2021, 9, 568879. [Google Scholar] [CrossRef]
- Castellani, B.; Morini, E.; Bonamente, E.; Rossi, F. Experimental Investigation and Energy Considerations on Hydrate-Based Biogas Upgrading with CO2 Valorization. Biomass Bioenergy 2017, 105, 364–372. [Google Scholar] [CrossRef]
- Passos, F.; Mota, C.; Donoso-Bravo, A.; Astals, S.; Jeison, D.; Muñoz, R. Biofuels from Microalgae: Biomethane. In Energy from Microalgae; Springer: Cham, Switzerland, 2018; pp. 247–270. [Google Scholar]
- Accettola, F.; Guebitz, G.M.; Schoeftner, R. Siloxane Removal from Biogas by Biofiltration: Biodegradation Studies. Clean. Technol. Environ. Policy 2008, 10, 211–218. [Google Scholar] [CrossRef]
- Meier, L.; Pérez, R.; Azócar, L.; Rivas, M.; Jeison, D. Photosynthetic CO2 Uptake by Microalgae: An Attractive Tool for Biogas Upgrading. Biomass Bioenergy 2015, 73, 102–109. [Google Scholar] [CrossRef]
- Meier, L.; Barros, P.; Torres, A.; Vilchez, C.; Jeison, D. Photosynthetic Biogas Upgrading Using Microalgae: Effect of Light/Dark Photoperiod. Renew. Energy 2017, 106, 17–23. [Google Scholar] [CrossRef]
- Noticias Jurídicas. Available online: https://noticias.juridicas.com/base_datos/Admin/res211212-iet.html (accessed on 24 June 2023).
- Bose, A.; O’Shea, R.; Lin, R.; Murphy, J.D. A Perspective on Novel Cascading Algal Biomethane Biorefinery Systems. Bioresour. Technol. 2020, 304, 123027. [Google Scholar] [CrossRef]
- Xu, B.; Liu, J.; Zhao, C.; Sun, S.; Xu, J.; Zhao, Y. Induction of Vitamin B12 to Purify Biogas Slurry and Upgrade Biogas Using Co-culture of Microalgae and Fungi. Water Environ. Res. 2021, 93, 1254–1262. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, B.; Zhao, C.; Liu, J.; Zhao, Y.; Sun, S.; Wei, J. Simultaneous Biogas Upgrading and Biogas Slurry Treatment by Different Microalgae-Based Technologies under Various Strigolactone Analog (GR24) Concentrations. Bioresour. Technol. 2022, 351, 127033. [Google Scholar] [CrossRef]
- Rusmanis, D.; O’Shea, R.; Wall, D.M.; Murphy, J.D. Biological Hydrogen Methanation Systems—An Overview of Design and Efficiency. Bioengineered 2019, 10, 604–634. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.-K.; Zhao, L.; Zheng, X.-C.; Sun, Z.-F.; Wang, Z.-H.; Chen, C.; Xing, D.-F.; Yang, S.-S.; Ren, N.-Q. Recovery of Methane and Acetate during Ex-Situ Biogas Upgrading via Novel Dual-Membrane Aerated Biofilm Reactor. Bioresour. Technol. 2023, 382, 129181. [Google Scholar] [CrossRef] [PubMed]
- Mattiasson, B. Ekologisk Lunga för Biogasuppgradering; Svenska Biogasföreningen: Lund, Sweden, 2005. [Google Scholar]
- Persson, M.; Jonsson, O.; Wellinger, A. Task 37—Biogas Upgrading to Vehicle Fuel Standards and Grid Injection. IEA Bioenergy 2006, 37, 20–21. [Google Scholar]
- Alper, E.; Lohse, M.; Deckwer, W.-D. On the Mechanism of Enzyme-Catalysed Gas—Liquid Reactions: Absorption of CO2, into Buffer Solutions Containing Carbonic Anhydrase. Chem. Eng. Sci. 1980, 35, 2147–2156. [Google Scholar] [CrossRef]
- Zhang, Y.-T.; Zhang, L.; Chen, H.-L.; Zhang, H.-M. Selective Separation of Low Concentration CO2 Using Hydrogel Immobilized CA Enzyme Based Hollow Fiber Membrane Reactors. Chem. Eng. Sci. 2010, 65, 3199–3207. [Google Scholar] [CrossRef]
- Fosbøl, P.L.; Gaspar, J.; Jacobsen, B.; Glibstrup, J.; Gladis, A.; Diaz, K.M.; Thomsen, K.; Woodley, J.M.; von Solms, N. Design and Simulation of Rate-Based CO2 Capture Processes Using Carbonic Anhydrase (CA) Applied to Biogas. Energy Procedia 2017, 114, 1434–1443. [Google Scholar] [CrossRef]
- Fradette, L.; Lefebvre, S.; Carley, J. Demonstration Results of Enzyme-Accelerated CO2 Capture. Energy Procedia 2017, 114, 1100–1109. [Google Scholar] [CrossRef]
- Mignogna, D.; Ceci, P.; Cafaro, C.; Corazzi, G.; Avino, P. Production of Biogas and Biomethane as Renewable Energy Sources: A Review. Appl. Sci. 2023, 13, 10219. [Google Scholar] [CrossRef]
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and Committee of the Regions Powering a Climate-Neutral Economy: An EU Strategy for Energy System Integration. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A52020DC0299 (accessed on 28 December 2023).
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and Committee of the Regions A Farm to Fork Strategy for a Fair, Healthy and Environmentally Friendly Food System. Available online: https://food.ec.europa.eu/horizontal-topics/farm-fork-strategy_en (accessed on 28 December 2023).
- Tomczak, W.; Gryta, M.; Grubecki, I.; Miłek, J. Biogas Production in AnMBRs via Treatment of Municipal and Domestic Wastewater: Opportunities and Fouling Mitigation Strategies. Appl. Sci. 2023, 13, 6466. [Google Scholar] [CrossRef]
- Wang, X.-H.; Xu, X.-J.; Cai, J.; Zheng, H.-X.; Chen, Y.-X.; Pang, W.-X.; Yu, Y.; Sun, C.-Y.; Chen, G.-J. CO2 Concentration in Aqueous Solution from Gas–Liquid Equilibrium System to Gas–Liquid–Hydrate Coexistence System. Gas. Sci. Eng. 2023, 115, 205024. [Google Scholar] [CrossRef]
- Bajón Fernández, Y.; Soares, A.; Villa, R.; Vale, P.; Cartmell, E. Carbon Capture and Biogas Enhancement by Carbon Dioxide Enrichment of Anaerobic Digesters Treating Sewage Sludge or Food Waste. Bioresour. Technol. 2014, 159, 1–7. [Google Scholar] [CrossRef]
- Wang, P.; Peng, H.; Adhikari, S.; Higgins, B.; Roy, P.; Dai, W.; Shi, X. Enhancement of Biogas Production from Wastewater Sludge via Anaerobic Digestion Assisted with Biochar Amendment. Bioresour. Technol. 2020, 309, 123368. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Deng, Y.F.; Wang, F.; Davaritouchaee, M.; Yao, Y.Q. A Review on Biochar-Mediated Anaerobic Digestion with Enhanced Methane Recovery. Renew. Sustain. Energy Rev. 2019, 115, 109373. [Google Scholar] [CrossRef]
- He, P.; Zhang, H.; Duan, H.; Shao, L.; Lü, F. Continuity of Biochar-Associated Biofilm in Anaerobic Digestion. Chem. Eng. J. 2020, 390, 124605. [Google Scholar] [CrossRef]
- Pan, J.; Ma, J.; Liu, X.; Zhai, L.; Ouyang, X.; Liu, H. Effects of Different Types of Biochar on the Anaerobic Digestion of Chicken Manure. Bioresour. Technol. 2019, 275, 258–265. [Google Scholar] [CrossRef]
- Maroušek, J.; Strunecký, O.; Stehel, V. Biochar Farming: Defining Economically Perspective Applications. Clean. Technol. Environ. Policy 2019, 21, 1389–1395. [Google Scholar] [CrossRef]
- Romero-Güiza, M.S.; Vila, J.; Mata-Alvarez, J.; Chimenos, J.M.; Astals, S. The Role of Additives on Anaerobic Digestion: A Review. Renew. Sustain. Energy Rev. 2016, 58, 1486–1499. [Google Scholar] [CrossRef]
- Sailer, G.; Eichermüller, J.; Poetsch, J.; Paczkowski, S.; Pelz, S.; Oechsner, H.; Müller, J. Optimizing Anaerobic Digestion of Organic Fraction of Municipal Solid Waste (OFMSW) by Using Biomass Ashes as Additives. Waste Manag. 2020, 109, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Ajay, C.M.; Mohan, S.; Dinesha, P.; Rosen, M.A. Review of Impact of Nanoparticle Additives on Anaerobic Digestion and Methane Generation. Fuel 2020, 277, 118234. [Google Scholar] [CrossRef]
- Yun, S.; Zhang, C.; Wang, Y.; Zhu, J.; Huang, X.; Du, T.; Li, X.; Wei, Y. Synergistic Effects of Fe Salts and Composite Additives on Anaerobic Digestion of Dairy Manure. Int. Biodeterior. Biodegrad. 2019, 136, 82–90. [Google Scholar] [CrossRef]
- Barua, S.; Zakaria, B.S.; Lin, L.; Dhar, B.R. Magnetite Doped Granular Activated Carbon as an Additive for High-Performance Anaerobic Digestion. Mater. Sci. Energy Technol. 2019, 2, 377–384. [Google Scholar] [CrossRef]
- Ding, M.; Jiang, H.-L. One-Step Assembly of a Hierarchically Porous Phenolic Resin-Type Polymer with High Stability for CO2 Capture and Conversion. Chem. Commun. 2016, 52, 12294–12297. [Google Scholar] [CrossRef] [PubMed]
- Kida, M.; Goda, H.; Sakagami, H.; Minami, H. CO2 Capture from CH4–CO2 Mixture by Gas–Solid Contact with Tetrahydrofuran Clathrate Hydrate. Chem. Phys. 2020, 538, 110863. [Google Scholar] [CrossRef]
- Fu, Y.; Jiang, Y.-B.; Dunphy, D.; Xiong, H.; Coker, E.; Chou, S.S.; Zhang, H.; Vanegas, J.M.; Croissant, J.G.; Cecchi, J.L.; et al. Ultra-Thin Enzymatic Liquid Membrane for CO2 Separation and Capture. Nat. Commun. 2018, 9, 990. [Google Scholar] [CrossRef]
- Karlsson, M.; Nygren, P. Enzymatic Upgrading of Biogas; Energiforsk: Stockholm, Sweden, 2015. [Google Scholar]
- Fougerit, V. Development of an Innovative Process for Biogas Purification through the Implementation of Membrane Contactors; Université Paris Saclay: Paris, France, 2017. [Google Scholar]
- González, J.M.; Fisher, S.Z. Carbonic Anhydrases in Industrial Applications. Subcell Biochem. 2014, 75, 405–426. [Google Scholar]
- Thema, M.; Bauer, F.; Sterner, M. Power-to-Gas: Electrolysis and Methanation Status Review. Renew. Sustain. Energy Rev. 2019, 112, 775–787. [Google Scholar] [CrossRef]
- de Meyer, F.; Jouenne, S. Industrial Carbon Capture by Absorption: Recent Advances and Path Forward. Curr. Opin. Chem. Eng. 2022, 38, 100868. [Google Scholar] [CrossRef]
- Ahmed, S.F.; Mofijur, M.; Tarannum, K.; Chowdhury, A.T.; Rafa, N.; Nuzhat, S.; Kumar, P.S.; Vo, D.-V.N.; Lichtfouse, E.; Mahlia, T.M.I. Biogas Upgrading, Economy and Utilization: A Review. Environ. Chem. Lett. 2021, 19, 4137–4164. [Google Scholar] [CrossRef]
- Andlar, M.; Belskaya, H.; Morzak, G.; Ivančić Šantek, M.; Rezić, T.; Petravić Tominac, V.; Šantek, B. Biogas Production Systems and Upgrading Technologies: A Review. Food Technol. Biotechnol. 2021, 59, 387–412. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.E.; Steiman, M.W.; St. Angelo, S.K. Biogas Digestate as a Renewable Fertilizer: Effects of Digestate Application on Crop Growth and Nutrient Composition. Renew. Agric. Food Syst. 2021, 36, 173–181. [Google Scholar] [CrossRef]
- Samoraj, M.; Mironiuk, M.; Izydorczyk, G.; Witek-Krowiak, A.; Szopa, D.; Moustakas, K.; Chojnacka, K. The Challenges and Perspectives for Anaerobic Digestion of Animal Waste and Fertilizer Application of the Digestate. Chemosphere 2022, 295, 133799. [Google Scholar] [CrossRef]
- Bidart, C.; Wichert, M.; Kolb, G.; Held, M. Biogas Catalytic Methanation for Biomethane Production as Fuel in Freight Transport—A Carbon Footprint Assessment. Renew. Sustain. Energy Rev. 2022, 168, 112802. [Google Scholar] [CrossRef]
- Kim, H.; Choi, H.; Lee, C. The Potential Use of Human Urine as a Solvent for Biogas Upgrading. J. Water Process Eng. 2020, 36, 101343. [Google Scholar] [CrossRef]
- Gutiérrez-Martín, F.; Rodríguez-Antón, L.M.; Legrand, M. Renewable Power-to-Gas by Direct Catalytic Methanation of Biogas. Renew. Energy 2020, 162, 948–959. [Google Scholar] [CrossRef]
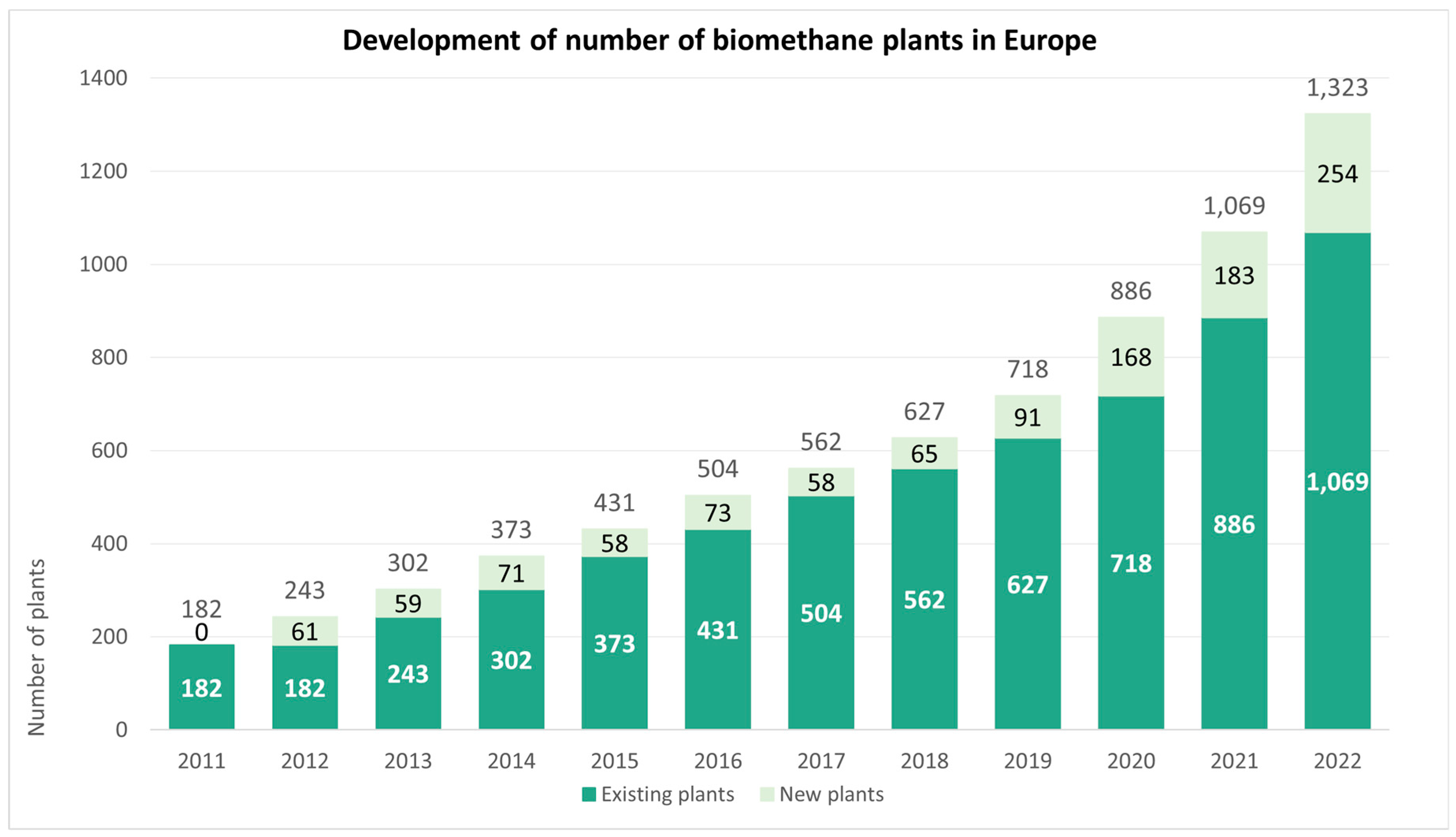
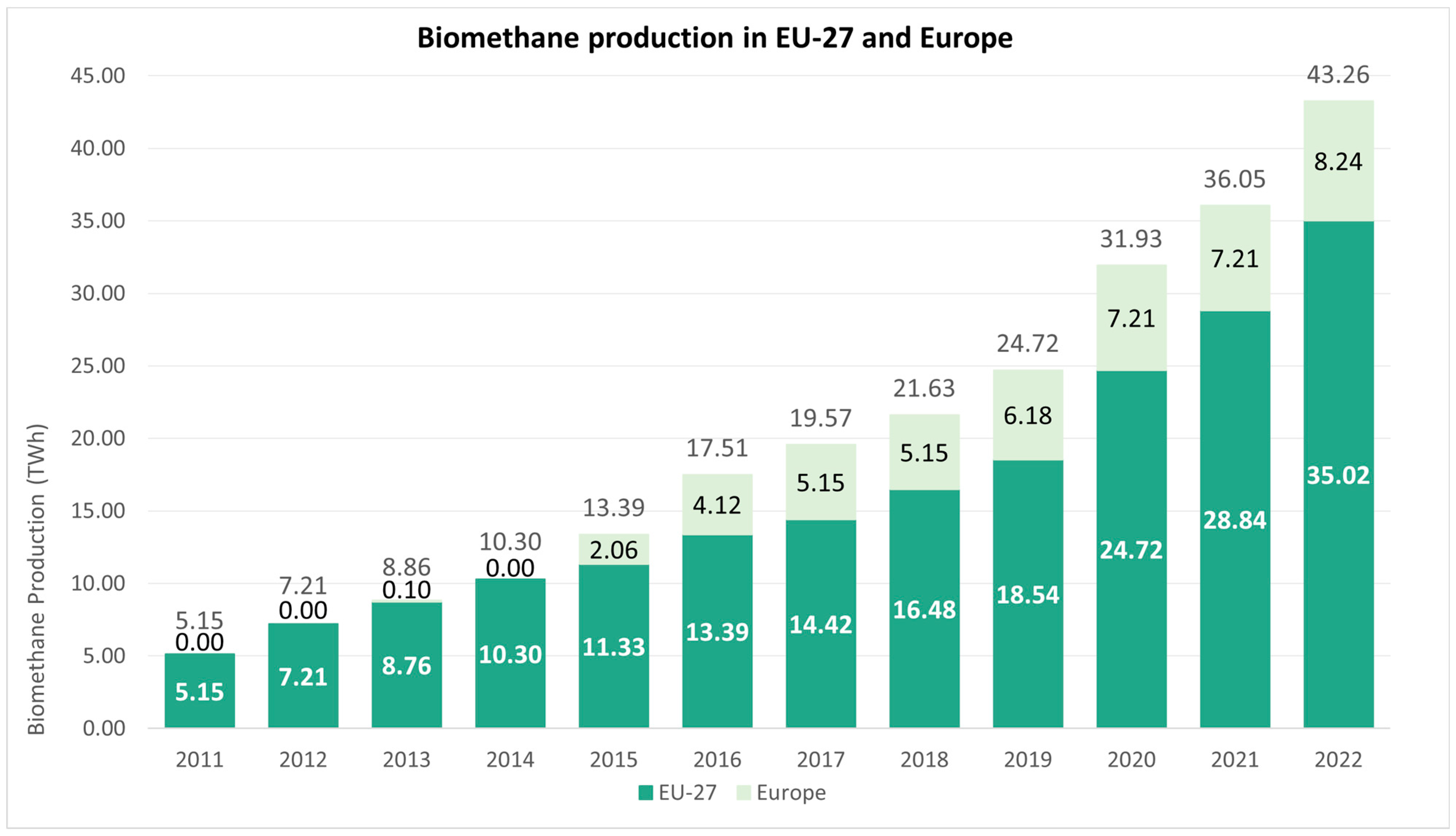
| Biomethane Production Pathway | Production in 2020 (TWh/y) | Production in 2030 (TWh/y) | Production in 2050 (TWh/y) | Total Existing Potential (TWh/y) |
|---|---|---|---|---|
| Biogas upgrading | 24.7–79.4 1 [41] 510.67 1,2,3 [42] 221.11–278.89 1 [43] | 148.3–369.4 1 [41] 334.94–467.6 (1) [43] | 1088.9 1 [44] | 865.6 1,3 [42] 500 1,4 [45] |
| Gasification with methanation | - | 885 1,5 [46] | - | - |
| PtM | - | 5.9–13.9 1 [41] | 266.7 1 [44] | - |
| Biogas upgrading and gasification with methanation | 88.61 1 [47] | 200–222.2 1 [47] | - | 1521.4–2467.8 1 [47] |
| Characteristics | Adsorption | Absorption | MS | CS | ||
|---|---|---|---|---|---|---|
| PSA | WS | OPS | AS | |||
| Compounds that must be pre-removed | H2S and H2O [51,61,63,69] | H2S (if the concentration is high) [61,63,65] | H2S (recommended) [61,63] | H2S (if the concentration is high) [61,63] O2 [68] | H2S and H2O [61,63] | H2S and H2O [52,61] |
| Technology separates N2 and O2 | Yes [51,54,66,69] | No [65,66] | Yes [61] No [65,66] | No [65,66] | Partly separates O2 but not N2 [65] | Possible if LBM is obtained [51,55] |
| Methane content in upgraded gas (%vol CH4) | 95–99 [54,61,63] >96 [69] 98 [52,76] 96–98 [77] | >97 [52,61] 95–99 [63] 95–98 [77] | >97 [61,65] 96–98.5 [51] 98 [52] 95–99 [63] 93–98 [77] | 99 [61] 97–99 [66] >99 [52,63] >98 [77] | 91 [61] 92 [78] 98 [51,65] 99 [54] 95–99 [63] 90–99 [77] | 98 [61] 97–99 [54,66] >97 [52] 99 [77] |
| Water demand (dm3/Nm3 biogas) [65] | 0 | 0.4–4 | 0 | 0.03 | 0 | 0 |
| Heat demand (kWh/Nm3 biogas) | 0 [65,69,78] | 0 [65,69,78] | <0.2 [78] 0.10–0.15 [69] | 0.55 [65] 0.50–0.75 [78] 0.4–0.8 [69] | 0 [65,69,78] | 0 [69,78] |
| Electricity consumption (kWh/Nm3 biogas) | 0.15–0.30 [65] 0.23–0.30 [78] 0.16–0.35 [69] 0.16–0.43 [77] | 0.20–0.30 [65,69] 0.3 [78] <0.25 [78] 0.2–0.5 [77] | 0.20–0.29 [65] 0.2–0.3 [78] 0.23–0.33 [69] 0.10–0.33 [77] | 0.12–0.14 [65] 0.10–0.15 [78] 0.06–0.17 [69] 0.05–0.18 [77] | 0.20–0.30 [65] 0.18–0.21 [78] 0.18–0.35 [69,77] | 0.20–0.28 [78] 0.72 [78] 1.05 [78] 0.18–0.25 [69,77] 0.4–1.0 [52] |
| Other consumables [65] | Lubricant oil for compressors | Anti-foaming agents (in some cases) and lubricant oil for compressors | Organic solvent addition once a year and lubricant oil for compressors | Anti-foaming agents, make-up of amine, and lubricant oil for compressors | Membrane replacement (5–10 years) and lubricant oil for compressors | Lubricant oil for compressors |
| Methane losses (%) | <2 [63,65] 2–4 [66] 10–12 [66] 1.5–10.0 [69] <3 [77] | <2 [52,54,61,77] 3–5 [66] 0.5–2 [69] 2 [63] 1 [65] | 1–4 [65] <2 [51] <1 [52] <4 [63,66,77] | <0.1 [52,65,69] 0.04 [63] <0.5 [77] 4 [51] | <0.5 [54,65] 1–15 [69] <1 [52] 0.5–20 [63] <5 [77] | <1 [51,61] <2 [52,65] 0.1–2.0 [69] <0.1 [77] |
| Parameters | PSA | WS | OPS | AS | MS | CS | |
|---|---|---|---|---|---|---|---|
| Specific CAPEX for different flows (€/(Nm3/h)) | 1000 Nm3/h biogas | 1750 [77] 1782 [61] 2000 [65,66] | 1000 [77] 1620 [61] 1700 [65] 1800 [66] | 1000 [77] 1944 [61] 2000 [65] 2200 [66] | 2000 [77] 2106 [61] 2500 [65] 2400 [66] | 1782 [61] 2000 [65,77] 1300–2400 [66] | - |
| 500 Nm3/h biogas | 2592 [61] 2800 [65] 2900 [66] 3700 [62] | 2187 [61] 2700 [65] 2500 [66] 3500 [62] | 3078 [61] 3300 [65] 3500 [62] | 2916 [61] 3300 [66] 3500 [62] | 2750 [65] 3500–3700 [62] | 600 Nm3/h: 2300 [66] | |
| 250 Nm3/h biogas | 4455 [61] 5400 [62,66] | 4000 [65] 5500 [62] | 3890 [61] 4500 [65] 4400 [66] 5000 [62,66] | 4455 [61] 5000 [62] | 3000–3500 [65] 4700–4900 [62,66] | - | |
| 100 Nm3/h biogas | 3000 [79] | 5000 [66,79] | - | - | 5000 [79] 6000 [52,66] | - | |
| Maintenance costs (% of CAPEX) [65] | - | 2–3 | 2–3 | 3 | 3–4 | - | |
| Maintenance costs for 1000 m3/h biogas (m€/y) [61] | 56 | 15 | 39 | 59 | 25 | - | |
| Availability (%) | 94 [61,78] 94–96 [52] | 96 [61,78] 95–96 [65] | 96 [61,78] 96–98 [65] | 91 [61,78] 96 [65] | 98 [61,78] 95–98 [65] | - | |
| Number of plants in Europe (2019) [80] | 79 | 175 1 (WS + PSA) | 17 | 103 | 173 1 (MS + PSA) | 10 (MS + CS) | |
| Substrate Type | Desorption Compound | Biogas Composition (%vol CH4) | CH4 in Upgraded Gas (%vol) | CH4 Losses (%) | Comments | Case/Reference |
|---|---|---|---|---|---|---|
| Sludges | Air | - | 87 | 8 | - | 1/[49] (best performance from different experiments) |
| Sewage sludge | Air | 58 | 69 | 1.3 | Low sludge flow rate and high gas flow rate lead to higher CH4 concentration and lower CH4 losses | 2/[100] (computer simulations) |
| Nitrogen-rich substrate | Air | 69 | 66 | - | Bad performance due to ammonia inhibition and pH increase | 3/[100] (lab-based platform) |
| Vacuum (40 kPa) | 63 | - | ||||
| Substrate mixture with less N than 3/ | Air | 55 | 64 | - | Addition of organic acids to air stripping may achieve better results | 4/[100] (lab-based platform) |
| Vacuum (40 kPa) | 60 | 62 | - | |||
| Vacuum (40 kPa) with organic acids | 49 | 61 | - | |||
| Manure | - | 60 | 81 | 1.8 | H2S reduction from 1100–1500 ppm to 150–300 ppm and reduction of nitrogen by 11–21% | 5/[101] (pilot plant) |
| Chicken manure | Air | 55 | 60–70 | 3.7–10.3 | Up to 60% H2S removal | 6/[99] (lab-based platform) |
| Ash Filter Type | Biogas Composition (%Vol) | Upgraded Gas Composition (%vol) | Loading Capacity (kg CO2/t Ash) | Comments | Case/Reference |
|---|---|---|---|---|---|
| BA from MSWI | 58% CH4 95 ppm H2S | 99% CH4 1.4 ppm H2S | 13.9 | This process is called BABIU (Bottom Ash for Biogas Upgrading) | 1/[113] (pilot plant) |
| 54.7% CH4 | 95% CH4 | 23 (less BA than in the previous result) | |||
| Ash from a biomass incineration plant | Gas rich in CO2 | - | 15.4 | Saturation time * of 300 min | 2/[90] (lab-scale tests) |
| Mussel shell ash | - | 1.4 | Saturation time * of 30 min | 3/[90] (lab-scale tests) | |
| Wood chip ash | 65% CH4 35% CO2 | 98% CH4 | 0.14 (dry ash) | Two serial reactors utilize the ash’s total CO2 capacity; the pH of the ash (approx. 13) was reduced by 2–3 units | 4/[116] (lab-scale tests) |
| Wood pellet ash | 99% CH4 | 0.2 (dry ash) | |||
| Mixture of fly ash (10%) and BA (90%) from a biomass incineration plant | 65% CH4 35% CO2 100–600 ppm H2S | Almost 100% CH4 and 0 ppm H2S | 35–135 (wet ash) 0.56–1.25 kg H2S/t wet ash | Pure CH4 was achieved for a few hours, while H2S removal was longer-lasting | 5/[117] (pilot plant) |
| Basic Oxygen Furnace (BOF) slags | 68.4% CH4 31.2% CO2 250 ppm H2S | 95% CH4 5% CO2 <2 ppm H2S | 48 | Saturation time * of 42 min and 300–350 kg of BOF to produce bio-compressed NG to fill up one vehicle | 6/[118] (lab-scale tests) |
| Biochar Type | Biogas Composition (%Vol CH4) | Case/(Reference) |
|---|---|---|
| Corn stover | 90 <5 ppb H2S | 1/[123] |
| Pine | 92.3 (mesophilic T) 79 (thermophilic T) | 2/[119] |
| White oak | 89.8 (mesophilic T) 78.5 (thermophilic T) | 3/[119] |
| Walnut shell | 77.5–98.1 | 4/[125] |
| Method | Operating Conditions | Promoter | Biogas Composition (%vol) | CH4 in Upgraded Gas (%Vol) | CO2 in Upgraded Gas (%Vol) | Case/ (Reference) | |
|---|---|---|---|---|---|---|---|
| T (K) | p (MPa) | ||||||
| CO2 hydrates | 278.15 | 6 | SDS 300 ppm | 60% CH4 40% CO2 | 70 | 30 | 1/[130] (up-scaled apparatus) |
| CO2 hydrates | 275.1 | 4.5 | 55% CH4, 37.5% CO2, 5% N2, 2% O2, 0.5% H2 | 70.1 | 22.4 | 2/[135] (up-scaled apparatus) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Francisco López, A.; Lago Rodríguez, T.; Faraji Abdolmaleki, S.; Galera Martínez, M.; Bello Bugallo, P.M. From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions. Appl. Sci. 2024, 14, 2342. https://doi.org/10.3390/app14062342
Francisco López A, Lago Rodríguez T, Faraji Abdolmaleki S, Galera Martínez M, Bello Bugallo PM. From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions. Applied Sciences. 2024; 14(6):2342. https://doi.org/10.3390/app14062342
Chicago/Turabian StyleFrancisco López, Ahinara, Telma Lago Rodríguez, Shoeib Faraji Abdolmaleki, Marta Galera Martínez, and Pastora M. Bello Bugallo. 2024. "From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions" Applied Sciences 14, no. 6: 2342. https://doi.org/10.3390/app14062342
APA StyleFrancisco López, A., Lago Rodríguez, T., Faraji Abdolmaleki, S., Galera Martínez, M., & Bello Bugallo, P. M. (2024). From Biogas to Biomethane: An In-Depth Review of Upgrading Technologies That Enhance Sustainability and Reduce Greenhouse Gas Emissions. Applied Sciences, 14(6), 2342. https://doi.org/10.3390/app14062342










