Impact of Lactic Acid Bacteria on Immunoreactivity of Oat Beers
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Brewing
2.3. Determination of Protein Immunoreactivity
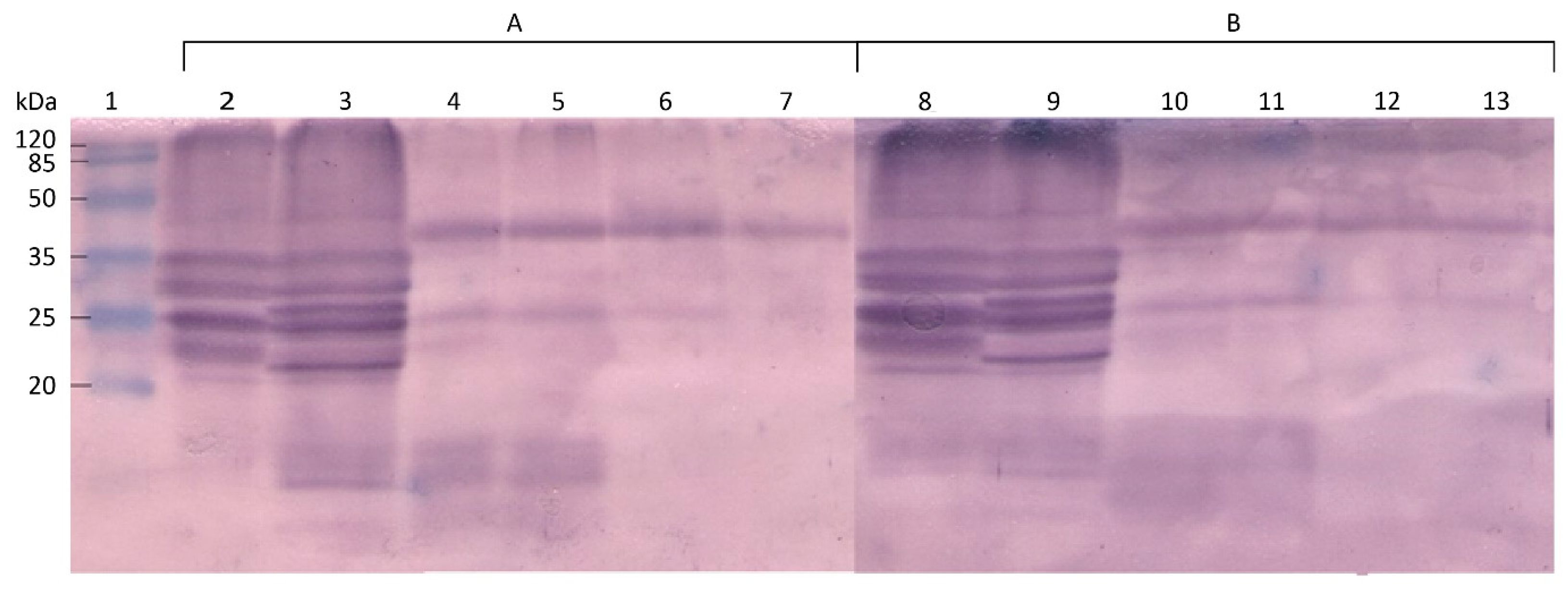
2.4. Identification of the Protein Fractions Involved in the Immunoenzymatic Reaction
2.5. Statistical Analysis
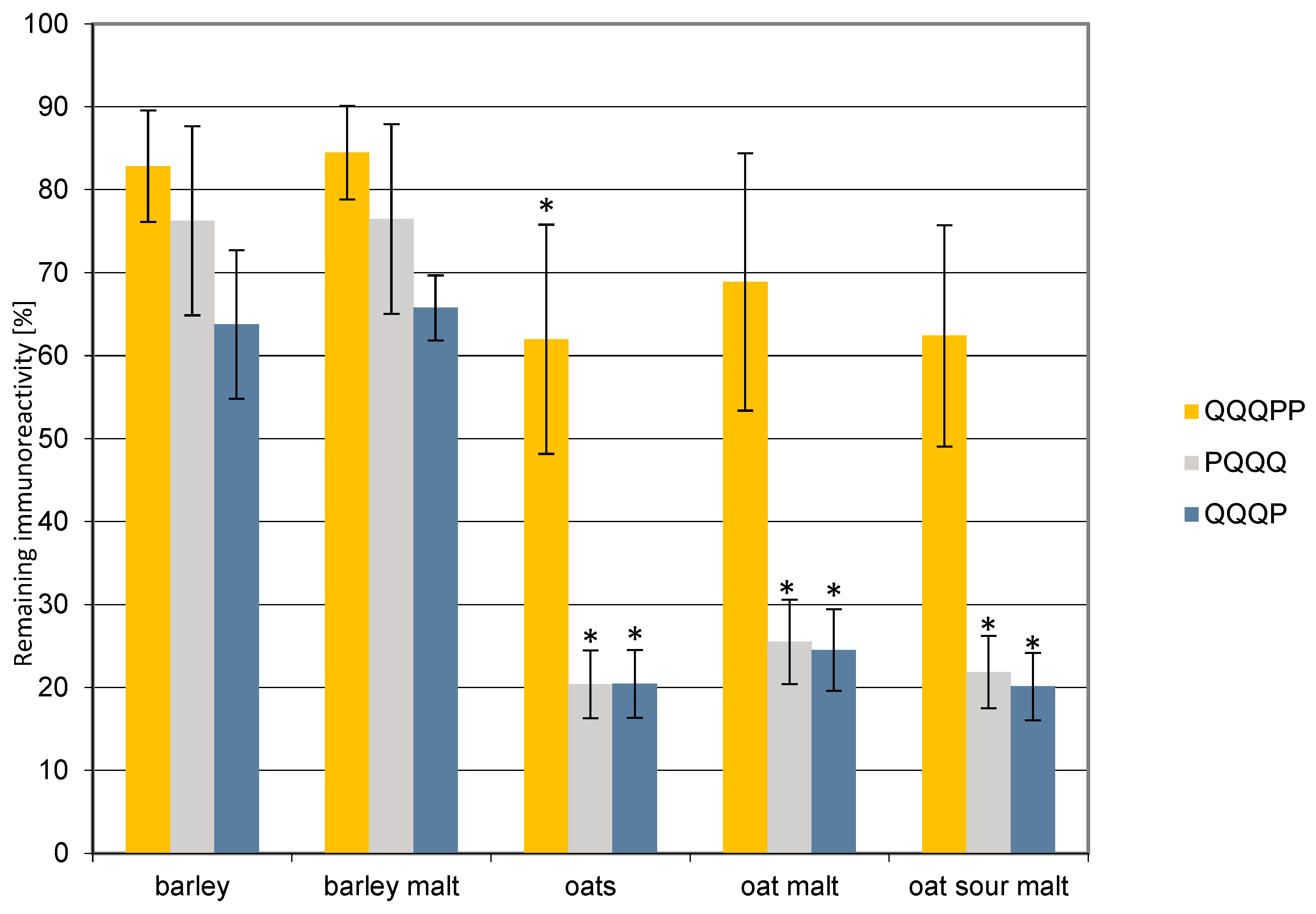
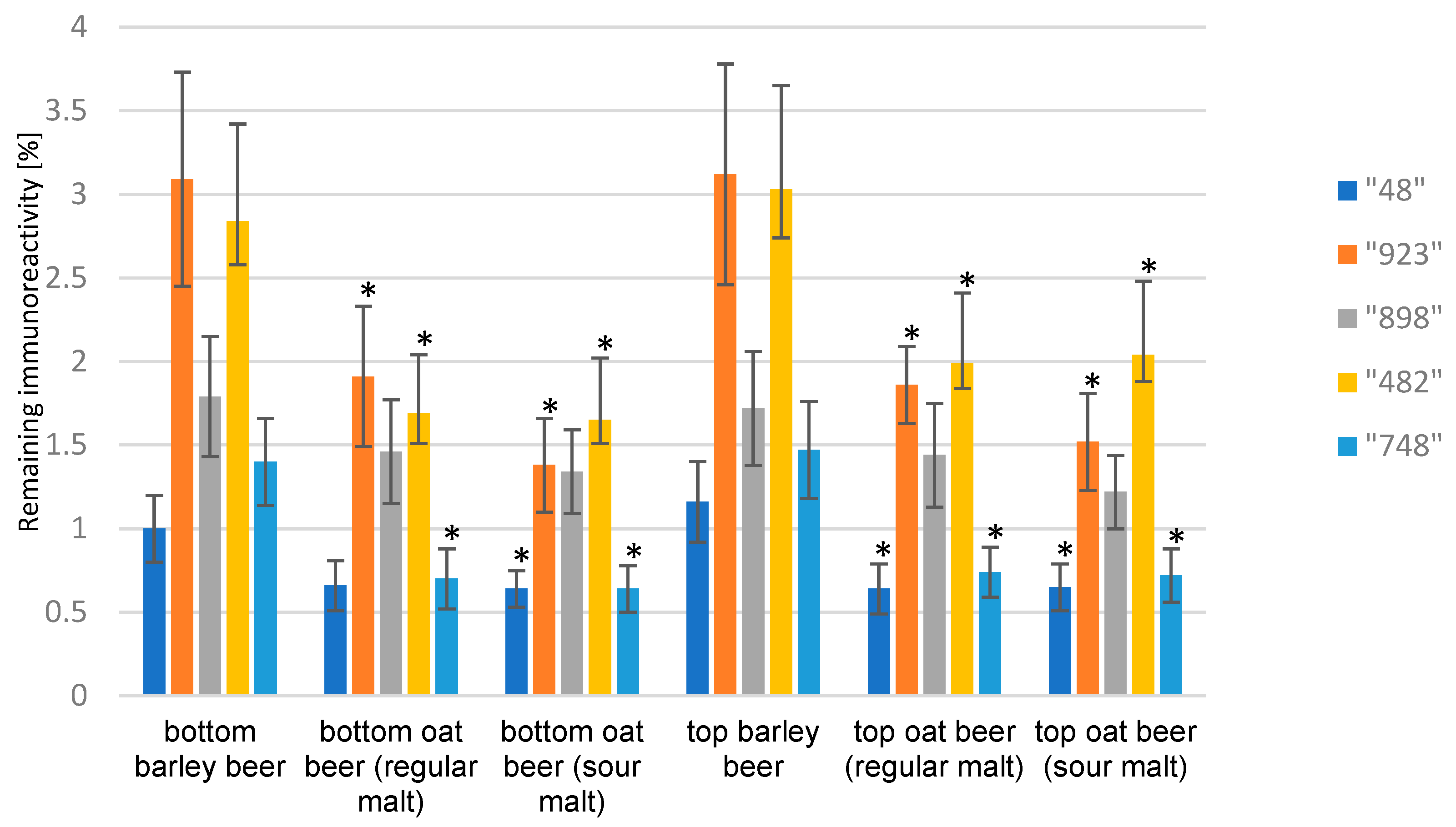
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lohi, S.; Mustalahti, K.; Kaukinen, K.; Lauril, K.; Colli, P.; Lohi, O.; Bravi, E.; Gasparin, M.; Reunanen, A.; Mäki, M. Increasing prevalence of celiac disease over time. Aliment. Pharmacol. Ther. 2007, 26, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- Bascunan, K.A.; Vespa, M.C.; Araya, M. Celiac disease: Understanding the gluten-free diet. Eur. J. Nutr. 2017, 56, 449–459. [Google Scholar] [PubMed]
- Ortiz, C.; Valenzuela, R.; Lucero, A.Y. Celiac disease, non celiac gluten sensitivity and wheat allergy: Comparison of 3 different diseases triggered by the same food. Rev. Child. Pediatr. 2017, 88, 417–423. [Google Scholar]
- Vici, G.; Belli, L.; Biondi, M.; Polzonetti, V. Gluten free diet and nutrient deficiencies: A review. Clin. Nutr. ESPEN 2016, 35, 1236–1241. [Google Scholar]
- Gobbetti, M.; Pontonio, E.; Filannino, P.; Rizello, C.; De Angelis, M.; Di Cagno, R. How to improve the gluten-free diet: The state of the art. From a food science perspective. Int. Food Res. 2018, 10, 22–29. [Google Scholar]
- Melini, V.; Melini, F. Gluten-free diet: Gaps and needs for a healthier diet. Nutrients 2019, 11, 170. [Google Scholar] [CrossRef]
- Kosová, K.; Leišová-Svobodová, L.; Dvořáček, V. Oats as a safe alternative to Triticeae cereals for people suffering from celiac disease? A review. Plant Foods Hum. Nutr. 2020, 75, 131–141. [Google Scholar] [CrossRef]
- Valido, E.; Stoyanov, J.; Bertolo, A.; Hertig-Godeschalk, A.; Zeh, R.M.; Flueck, J.L.; Minder, B.; Stojic, S.; Metzger, B.; Bussler, W.; et al. Systematic review of the effects of oat iIntake on gastrointestinal health. J. Nutr. 2021, 151, 3075–3090. [Google Scholar]
- Thompson, T. Oats and the gluten-free diet. J. Am. Diet. Assoc. 2003, 103, 376–379. [Google Scholar]
- Klose, C.; Arendt, E.K. Proteins in oats; their synthesis and changes during germination: A review. Crit. Rev. Food Sci. Nutr. 2012, 52, 629–639. [Google Scholar]
- Hüttner, E.K.; Arendt, E.K. Recent advances in gluten-free baking and the current status of oats. Trends Food Sci. Technol. 2010, 21, 303–312. [Google Scholar]
- Pawłowska, P.; Diowksz, A.; Kordialik-Bogacka, E. State-of-the-art on incorporation of oats into a gluten-free diet. Food Rev. Int. 2012, 28, 330–342. [Google Scholar]
- Hoffmanova, I.; Sanchez, D.; Szczepankova, A.; Tlaskalova-Hogenova, H. The pros and cons of using oat in a gluten-free diet for celiac patients. Nutrients 2019, 11, 2345. [Google Scholar] [CrossRef]
- Sternaa, V.; Zuteb, S.; Brunavaa, L. Oat grain composition and its nutrition benefice. Agric. Agric. Sci. Procedia 2016, 8, 252–256. [Google Scholar] [CrossRef]
- Anttila, H.; Sontag-Strohm, T.; Salovaara, H. Viscosity of beta-glucan in oat products. Agric. Food Sci. 2004, 13, 80–87. [Google Scholar]
- Angelov, A.; Gotcheva, V.; Kuncheva, R.; Hristozova, T. Development of a new oat-based probiotic drink. Int. J. Food Microbiol. 2006, 112, 75–80. [Google Scholar]
- Størsrud, S.; Malmheden Yman, I.; Lenner, R.A. Gluten contamination in oat products and products naturally free from gluten. Eur. Food Res. Technol. 2003, 217, 481–485. [Google Scholar] [CrossRef]
- Hernando, A.; Mujico, J.R.; Mena, M.C.; Lombardia, M.; Méndez, E. Measurement of wheat gluten and barley hordeins in contaminated oats from Europe, the United States and Canada by sandwich R5 ELISA. Eur. J. Gastroenterol. Hepatol. 2008, 20, 545–554. [Google Scholar]
- Wieser, H.; Segura, V.; Ruiz-Carnicer, A.; Sousa, C.; Comino, I. Food safety and cross-contamination of gluten-free products: A narrative review. Nutrients 2021, 13, 2244. [Google Scholar] [CrossRef]
- Løvik, A.; Skodje, G.; Bratlie, J.; Brottveit, M.; Lundin, K.E.A. Diet adherence and gluten exposure in coeliac disease and self-reported non-coeliac gluten sensitivity. Clin. Nutr. 2017, 36, 275–280. [Google Scholar]
- Mazzola, A.M.; Zammarchi, I.; Valerii, M.C.; Spisni, E.; Saracino, I.M.; Lanzarotto, F.; Ricci, C. Gluten-Free Diet and Other Celiac Disease Therapies: Current Understanding and Emerging Strategies. Nutrients 2024, 16, 1006. [Google Scholar] [CrossRef] [PubMed]
- Ellis, H.J.; Ciclitira, P.J. Should coeliac sufferers be allowed their oats? Eur. J. Gastroenterol. Hepatol. 2008, 20, 492–493. [Google Scholar]
- Hassani, A.; Procopio, S.; Becker, T. Influence of malting and lactic acid fermentation on functional bioactive components in cereal-based raw materials: A review paper. Int. J. Food Sci. Technol. 2016, 51, 14–22. [Google Scholar]
- Klose, C.; Mauch, A.; Wunderlich, S.; Thiele, F.; Zarnkow, M.; Jacob, F.; Arendt, E.K. Brewing with 100% oat malt. J. Inst. Brew. 2011, 117, 411–421. [Google Scholar]
- Kordialik-Bogacka, E.; Bogdan, P.; Diowksz, A. Malted and unmalted oats in brewing. J. Inst. Brew. 2014, 120, 390–398. [Google Scholar]
- Zdaniewicz, M.; Pater, A.; Knapik, A.; Duliński, R. The effect of different oat (Avena sativa L.) malt contents in a top-fermented beer recipe on the brewing process performance and product quality. J. Cereal Sci. 2021, 101, 103301. [Google Scholar]
- Kumar, J.; Kumar, M.; Pandey, R.; Chauhan, N.S. Physiopathology and management of gluten-Induced celiac disease. J. Food Sci. 2017, 82, 270–277. [Google Scholar]
- Wieser, H.; Koehler, P. The Biochemical Basis of Celiac Disease. Cereal Chem. 2008, 85, 1–13. [Google Scholar]
- Cornell, H.J.; Stelmasiak, T. The Significance of Key Amino Acid Sequences in the Digestibility and Toxicity of Gliadin Peptides in Celiac Disease. IJCD 2016, 4, 113–120. [Google Scholar]
- Vriz, R.; Moreno, F.J.; Koning, F.; Fernandez, A. Ranking of immunodominant epitopes in celiac disease: Identification of reliable parameters for the safety assessment of innovative food proteins. Food Chem. Toxicol. 2021, 157, 112584. [Google Scholar]
- Cristofori, F.; Francavilla, R.; Capobianco, D.; Dargenio, V.N.; Filardo, S.; Mastromarino, P. Bacterial-Based Strategies to Hydrolyze Gluten Peptides and Protect Intestinal Mucosa. Front. Immunol. 2020, 11, 567801. [Google Scholar]
- Aldred, P.; Kanauchi, M.; Bamforth, C.W. An investigation into proteolysis in mashing. J. Inst. Brew. 2021, 127, 21–26. [Google Scholar]
- Ledley, A.J.; Elias, R.J.; Cockburn, D.W. Evaluating the Role of Mashing in the Amino Acid Profiles of Worts Produced from Gluten-Free Malts. Beverages 2023, 9, 10. [Google Scholar] [CrossRef]
- Allred, L.K.; Lesko, K.; McKiernan, D.; Kupper, C.; Guandalini, S. The Celiac Patient Antibody Response to Conventional and Gluten-Removed Beer. J. AOAC Int. 2017, 100, 485–491. [Google Scholar]
- Watson, H.G.; Decloedt, A.I.; Hemeryck, L.Y.; Van Landschoot, A.; Prenni, J. Peptidomics of an industrial gluten-free barley malt beer and its non-gluten-free counterpart: Characterisation and immunogenicity. Food Chem. 2021, 355, 129597. [Google Scholar]
- Gerez, C.L.; Dallagnol, A.; Rollán, G.; De Valdez, G.F. A combination of two lactic acid bacteria improves the hydrolysis of gliadin during wheat dough fermentation. Food Microbiol. 2012, 32, 427–430. [Google Scholar]
- Brzozowski, B.; Stasiewicz, K.; Ostolski, M.; Adamczak, M. Reducing immunoreactivity of gliadins and coeliac-toxic peptides using peptidases from L. acidophilus 5e2 and A. niger. Catalysts 2020, 10, 923. [Google Scholar] [CrossRef]
- Kõiv, V.; Tenson, T. Gluten-degrading bacteria: Availability and applications. Appl. Microbiol. Biotechnol. 2021, 105, 3045–3059. [Google Scholar]
- Lowe, D.P.; Arendt, E.K. The use and effects of lactic acid bacteria in malting and brewing with their relationships to antifungal activity, mycotoixins and gushing: A review. J. Inst. Brew. 2004, 110, 163–180. [Google Scholar]
- Lowe, D.P.; Arendt, E.K.; Soriano, A.M.; Ulmer, H.M. The influence of lactic acid bacteria on the quality of malt. J. Inst. Brew. 2005, 111, 42–50. [Google Scholar]
- Laitila, A.; Sweins, H.; Vilpola, A.; Kotaviita, E.; Olkku, J.; Home, S.; Haikara, A. Lactobacillus plantarum and Pediococcus pentosaceus starter cultures as a tool for microflora management in malting for enhancement of malt processability. J. Agric. Food Chem. 2006, 54, 3840–3851. [Google Scholar] [CrossRef] [PubMed]
- Peyer, L.C.; De Kruijf, M.; O’Mahony, J.; De Colli, L.; Danaher, M.; Zarnkow, M.; Jacob, F.; Arendt, E.K. Lactobacillus brevis R2Δ as starter culture to improve biological and technological qualities of barley malt. Eur. Food Res. Technol. 2017, 243, 1363–1374. [Google Scholar] [CrossRef]
- Di Cagno, R.; De Angelis, M.; Lavermicocca, P.; De Vincenzi, M.; Giovannini, C.; Faccia, M.; Gobbetti, M. Proteolysis of sourdough lactic acid bacteria: Effects on wheat flour protein fractions and gliadin peptides involved in human cereal intolerance. Appl. Environ. Microbiol. 2002, 68, 623–633. [Google Scholar] [CrossRef]
- De Angelis, M.; Coda, R.; Silano, M.; Minervini, F.; Rizzello, C.G.; Di Cagno, R.; Vicentini, O.; De Vincenzi, M.; Gobbetti, M. Fermentation by selected sourdough lactic acid bacteria to decrease coeliac intolerance to rye flour. J. Cereal Sci. 2006, 43, 301–314. [Google Scholar] [CrossRef]
- Ogilviea, O.; Roberts, S.; Sutton, K.; Domigana, L.; Larsen, N.; Gerrarda, J.; Demaraisa, N. Proteomic modelling of gluten digestion from a physiologically relevant food system: A focus on the digestion of immunogenic peptides from wheat implicated in celiac disease. Food Chem. 2020, 333, 127466. [Google Scholar] [CrossRef] [PubMed]
- Bradauskiene, V.; Vaiciulyte-Funk, L.; Shah, B.R.; Cernauskas, D.; Tita, M.A. Recent advances in biotechnological methods for wheat gluten immunotoxicity abolishment—A review. Pol. J. Food Nutr. Sci. 2021, 71, 5–20. [Google Scholar] [CrossRef]
- Berti, C.; Trovato, C.; Bardella, M.T.; Forlani, F. IgA anti-gliadin antibody immunoreactivity to food proteins. Food Agric. Immunol. 2003, 15, 217–223. [Google Scholar] [CrossRef]
- Tanabe, S.; Arai, S.; Yanagihara, Y.; Mita, H.; Takahashi, K.; Watanabe, M. A major wheat alergen has a Gln-Gln-Gln-Pro-Pro motif identified as an IgE-binding epitope. Biochem. Biophys. Res. Commun. 1996, 219, 290–293. [Google Scholar] [CrossRef]
- Słowianek, M.; Mańkowska, D.; Leszczyńska, J. Reaction of spice proteins with serum antibodies from celiac patients and rabbit antibodies raised to specific glutamine/proline-containing peptides. Food Agric. Immunol. 2016, 28, 134–148. [Google Scholar] [CrossRef]
- Watanabe, M.; Tanabe, S.; Suzuki, T.; Ikezawa, Z.; Arai, S. Primary structure of an allergenic peptide occuring in the chymotryptic hydrolysate of gluten. Biosci. Biotechnol. Biochem. 1995, 59, 1596–1995. [Google Scholar] [CrossRef]
- Fritz, R.D.; Chen, Y. Oat safety for celiac disease patients: Theoretical analysis correlates adverse symptoms in clinical studies to contaminated study oats. Nutr. Res. 2018, 60, 54–67. [Google Scholar]
- Dostálek, P.; Hochel, I.; Méndez, E.; Hernando, A.; Gabrovská, D. Immunochemical determination of gluten in malts and beers. Food Addit. Contam. 2006, 23, 1074–1078. [Google Scholar]
- Müntz, K.; Belozersky, M.A.; Dunaevsky, Y.E.; Schlereth, A.; Tiedemann, J. Stored proteinases and the initiation of storage protein mobilization in seed during germination and seedling growth. J. Exp. Bot. 2001, 52, 1741–1752. [Google Scholar] [PubMed]
- Klose, C.; Schlehl, B.D.; Arendt, E.K. Fundamental study of protein changes taking place during malting of oats. J. Cereal Sci. 2009, 49, 83–91. [Google Scholar]
- Lewis, M.J. Celiac disease, beer and brewing. Tech. Q. MBAA Commun. 2005, 42, 45–48. [Google Scholar]
- Steiner, E.; Gastl, M.; Becker, T. Protein changes during malting and brewing with focus on haze and foam formation: A review. Eur. Food Res. Technol. 2011, 232, 191–204. [Google Scholar]
- Tsuji, H.; Kimoto, M.; Natori, Y. Allergen in major crops. Nutr. Res. 2001, 21, 925–934. [Google Scholar]
- Mikola, M.; Mikkonen, M. Occurrence and stabilities of oat trypsin and chymotrypsin inhibitors. J. Cereal Sci. 1999, 30, 227–235. [Google Scholar]
- Hejgaard, J.; Hauge, S. Serpins of oat (Avena sativa) grain with distinct reactive centres and inhibitory specificity. Physiol. Plant 2002, 116, 153–163. [Google Scholar]
- Maruyama, N.; Ichsie, K.; Katsube, T.; Kishimoto, T.; Kawase, S.; Matsumura, Y.; Takeuchi, Y.; Sawada, T.; Utsumi, S. Identification of major wheat allergens by means of Escherichia coli expression system. Eur. J. Biochem. 1998, 225, 739–745. [Google Scholar]
- Leszczyńska, J.; Łącka, A.; Pytasz, U.; Szemraj, J.; Lukamowicz, J.; Lewiński, A. The effect of proteolysis on the gliadin immunogenicity. Pol. J. Food Nutr. Sci. 2002, 52, 145–148. [Google Scholar]
- Szabo, E.; Hajos, G.; Matuz, J. Identification of major allergens of cereal proteins by electrophoretic methods. Pol. J. Food Nutr. Sci. 2002, 52, 131–134. [Google Scholar]
- Waga, J. Structure and allergenicity of wheat gluten proteins—A review. Pol. J. Food Nutr. Sci. 2004, 54, 327–338. [Google Scholar]
- Mills, E.N.C.; Madsen, C.; Shewry, P.R.; Wichers, H.J. Food allergens of plant origin—Their molecular and evolutionary relationship. Trends Food Sci. Technol. 2003, 14, 145–156. [Google Scholar] [CrossRef]
- Kushimoto, H.; Aoki, T. Masked type I wheat allergy—Relation to exercise-induced anaphylaxis. Arch. Dermatol. 1985, 121, 355–360. [Google Scholar] [CrossRef]
- Simonato, B.; Lazzari, F.D.; Pasini, G.; Polato, F.; Giannattasio, M.; Gemignani, C.; Peruffo, A.D.B.; Santucci, B.; Plebani, M.; Curioni, A. IgE binding to soluble and insoluble wheat flour proteins in atopic and non-atopic patients suffering from gastrointestinal symptoms after wheat ingestion. Clin. Exp. Allergy 2001, 31, 1771–1778. [Google Scholar] [CrossRef]
- Scherf, K.A.; Catassi, C.; Chirdo, F.; Ciclitira, P.J.; Feighery, C.; Gianfrani, C.; Koning, F.; Lundin, K.E.A.; Schuppan, D.; Smulders, M.J.M.; et al. Recent Progress and Recommendations on Celiac Disease From the Working Group on Prolamin Analysis and Toxicity. Front. Nutr. 2020, 7, 29. [Google Scholar] [CrossRef]
- DiCagno, R.; De Angelis, M.; Auricchio, S.; Greco, L.; Clarke, C.; De Vincenzi, M.; Giovanni, C.; D’Archivio, M.; Landolfo, F.; Parrilli, G.; et al. Sourdough bread made from wheat and non-toxic flours and started with selected Lactobacilli is tolerated in celiac patients. Appl. Environ. Microbiol. 2004, 70, 1088–1096. [Google Scholar] [CrossRef]
- Arentz-Hansen, H.; Fleckenstein Molberg, Ø.; Scott, H.; Koning, F.; Jung, G.; Roepstorff, P.; Lundin, K.E.A.; Sollid, L.M. The molecular basis for oat intolerance in patients with celiac disease. PLoS Med. 2004, 1, 84–92. [Google Scholar] [CrossRef]
- Guennouni, M.; Admou, B.; El Khoudri, N.; Bourrhouat, A.; Zogaam, L.G.; Elmoumou, L.; Hilali, A. Gluten contamination in labelled gluten-free, naturally gluten-free and meals in food services in low-, middle- and high-income countries: A systematic review and meta-analysis. Br. J. Nutr. 2022, 127, 1528–1542. [Google Scholar] [CrossRef]
- Voisine, J.; Abadie, V. Interplay Between Gluten, HLA, Innate and Adaptive Immunity Orchestrates the Development of Coeliac Disease. Front. Immunol. 2021, 12, 674313. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diowksz, A.; Pawłowska, P.; Kordialik-Bogacka, E.; Leszczyńska, J. Impact of Lactic Acid Bacteria on Immunoreactivity of Oat Beers. Appl. Sci. 2025, 15, 3887. https://doi.org/10.3390/app15073887
Diowksz A, Pawłowska P, Kordialik-Bogacka E, Leszczyńska J. Impact of Lactic Acid Bacteria on Immunoreactivity of Oat Beers. Applied Sciences. 2025; 15(7):3887. https://doi.org/10.3390/app15073887
Chicago/Turabian StyleDiowksz, Anna, Paulina Pawłowska, Edyta Kordialik-Bogacka, and Joanna Leszczyńska. 2025. "Impact of Lactic Acid Bacteria on Immunoreactivity of Oat Beers" Applied Sciences 15, no. 7: 3887. https://doi.org/10.3390/app15073887
APA StyleDiowksz, A., Pawłowska, P., Kordialik-Bogacka, E., & Leszczyńska, J. (2025). Impact of Lactic Acid Bacteria on Immunoreactivity of Oat Beers. Applied Sciences, 15(7), 3887. https://doi.org/10.3390/app15073887










