Tau Protein Accumulation Trajectory-Based Brain Age Prediction in the Alzheimer’s Disease Continuum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Image Acquisition and Processing
2.3. Brain Age Estimation
2.4. Statistical Analyses
3. Results
3.1. Subject Characteristics
3.2. Pathologic Tau Accumulation in AD Continuum
3.3. Brain Age Prediction and Its Gap
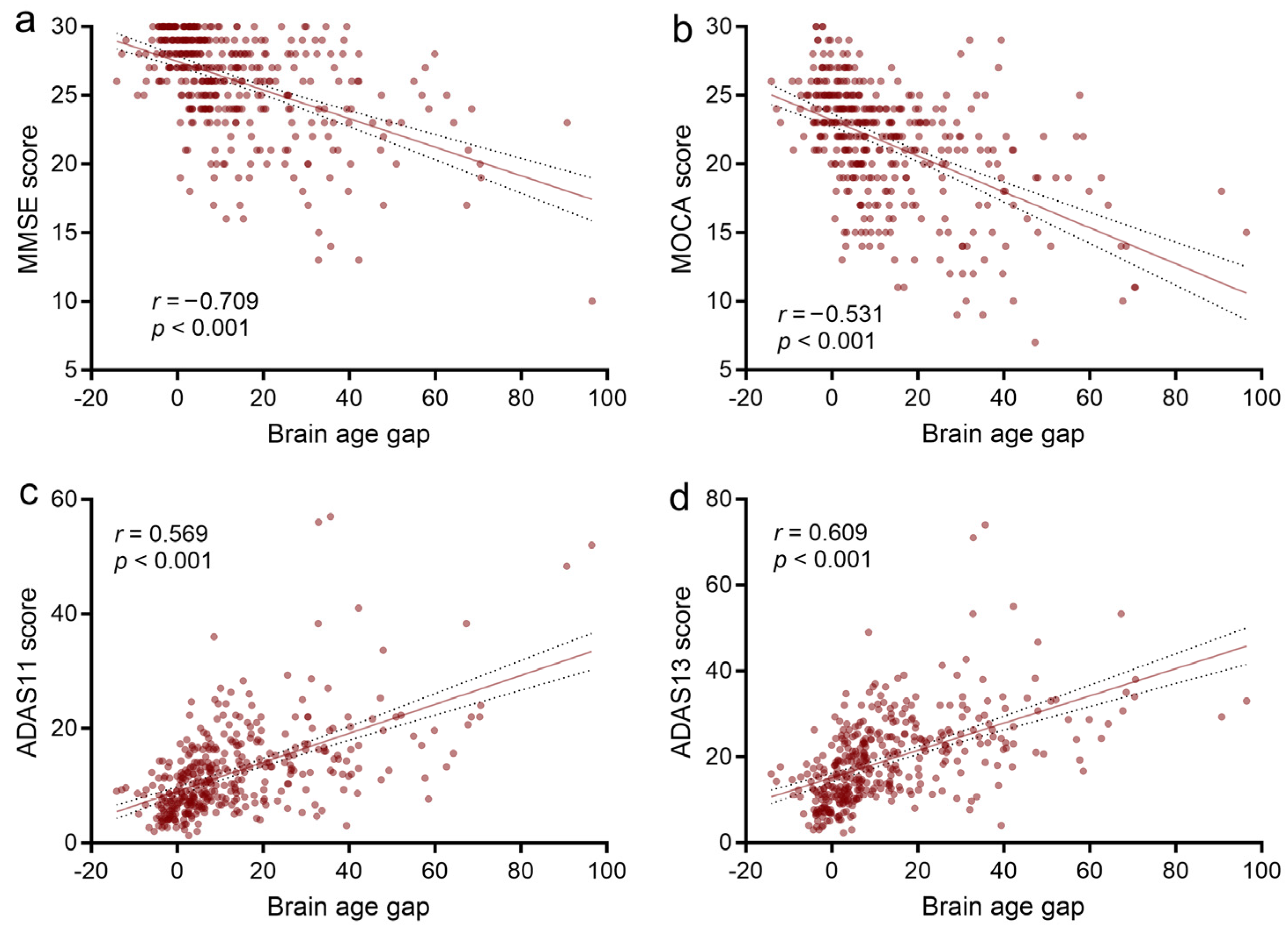
3.4. Associations between Brain Age Gap and Neuropsychological Assessments and the AD Biomarker
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 2023, 19, 1598–1695. [CrossRef]
- Jack, C.R.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Haeberlein, S.B.; Holtzman, D.M.; Jagust, W.; Jessen, F.; Karlawish, J.; et al. NIA-AA Research Framework: Toward a Biological Definition of Alzheimer’s Disease. Alzheimers Dement. 2018, 14, 535–562. [Google Scholar] [CrossRef]
- Jack, C.R.; Holtzman, D.M. Biomarker Modeling of Alzheimer’s Disease. Neuron 2013, 80, 1347–1358. [Google Scholar] [CrossRef]
- Jansen, W.J.; Ossenkoppele, R.; Knol, D.L.; Tijms, B.M.; Scheltens, P.; Verhey, F.R.; Visser, P.J.; Aalten, P.; Aarsland, D.; Alcolea, D.; et al. Prevalence of Cerebral Amyloid Pathology in Persons without Dementia: A Meta-Analysis. JAMA 2015, 313, 1924–1938. [Google Scholar] [CrossRef]
- Murphy, M.P.; LeVine, H. Alzheimer’s Disease and the Amyloid-Beta Peptide. J. Alzheimers Dis. 2010, 19, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Thal, D.R.; Ronisz, A.; Tousseyn, T.; Rijal Upadhaya, A.; Balakrishnan, K.; Vandenberghe, R.; Vandenbulcke, M.; von Arnim, C.A.F.; Otto, M.; Beach, T.G.; et al. Different Aspects of Alzheimer’s Disease-Related Amyloid β-Peptide Pathology and Their Relationship to Amyloid Positron Emission Tomography Imaging and Dementia. Acta Neuropathol. Commun. 2019, 7, 178. [Google Scholar] [CrossRef]
- Vogel, J.W.; Young, A.L.; Oxtoby, N.P.; Smith, R.; Ossenkoppele, R.; Strandberg, O.T.; La Joie, R.; Aksman, L.M.; Grothe, M.J.; Iturria-Medina, Y.; et al. Four Distinct Trajectories of Tau Deposition Identified in Alzheimer’s Disease. Nat. Med. 2021, 27, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Strikwerda-Brown, C.; Hobbs, D.A.; Gonneaud, J.; St-Onge, F.; Binette, A.P.; Ozlen, H.; Provost, K.; Soucy, J.-P.; Buckley, R.F.; Benzinger, T.L.S.; et al. Association of Elevated Amyloid and Tau Positron Emission Tomography Signal with Near-Term Development of Alzheimer Disease Symptoms in Older Adults without Cognitive Impairment. JAMA Neurol. 2022, 79, 975–985. [Google Scholar] [CrossRef] [PubMed]
- Ossenkoppele, R.; Pichet Binette, A.; Groot, C.; Smith, R.; Strandberg, O.; Palmqvist, S.; Stomrud, E.; Tideman, P.; Ohlsson, T.; Jögi, J.; et al. Amyloid and Tau PET-Positive Cognitively Unimpaired Individuals Are at High Risk for Future Cognitive Decline. Nat. Med. 2022, 28, 2381–2387. [Google Scholar] [CrossRef]
- Biel, D.; Brendel, M.; Rubinski, A.; Buerger, K.; Janowitz, D.; Dichgans, M.; Franzmeier, N. Alzheimer’s Disease Neuroimaging Initiative (ADNI) Tau-PET and in Vivo Braak-Staging as Prognostic Markers of Future Cognitive Decline in Cognitively Normal to Demented Individuals. Alzheimers Res. Ther. 2021, 13, 137. [Google Scholar] [CrossRef]
- Pontecorvo, M.J.; Devous, M.D.; Kennedy, I.; Navitsky, M.; Lu, M.; Galante, N.; Salloway, S.; Doraiswamy, P.M.; Southekal, S.; Arora, A.K.; et al. A Multicentre Longitudinal Study of Flortaucipir (18F) in Normal Ageing, Mild Cognitive Impairment and Alzheimer’s Disease Dementia. Brain 2019, 142, 1723–1735. [Google Scholar] [CrossRef] [PubMed]
- Whittington, A.; Gunn, R.N. Alzheimer’s Disease Neuroimaging Initiative TauIQ: A Canonical Image Based Algorithm to Quantify Tau PET Scans. J. Nucl. Med. 2021, 62, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Whittington, A.; Gunn, R.N. Amyloid Load: A More Sensitive Biomarker for Amyloid Imaging. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2019, 60, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.E.; Cropley, V.; Maier, A.B.; Lautenschlager, N.T.; Breakspear, M.; Zalesky, A. Heterogeneous Aging across Multiple Organ Systems and Prediction of Chronic Disease and Mortality. Nat. Med. 2023, 29, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Beheshti, I.; Ganaie, M.A.; Paliwal, V.; Rastogi, A.; Razzak, I.; Tanveer, M. Predicting Brain Age Using Machine Learning Algorithms: A Comprehensive Evaluation. IEEE J. Biomed. Health Inform. 2022, 26, 1432–1440. [Google Scholar] [CrossRef] [PubMed]
- Han, L.K.M.; Dinga, R.; Hahn, T.; Ching, C.R.K.; Eyler, L.T.; Aftanas, L.; Aghajani, M.; Aleman, A.; Baune, B.T.; Berger, K.; et al. Brain Aging in Major Depressive Disorder: Results from the ENIGMA Major Depressive Disorder Working Group. Mol. Psychiatry 2021, 26, 5124–5139. [Google Scholar] [CrossRef] [PubMed]
- Constantinides, C.; Han, L.K.M.; Alloza, C.; Antonucci, L.A.; Arango, C.; Ayesa-Arriola, R.; Banaj, N.; Bertolino, A.; Borgwardt, S.; Bruggemann, J.; et al. Brain Ageing in Schizophrenia: Evidence from 26 International Cohorts via the ENIGMA Schizophrenia Consortium. Mol. Psychiatry 2023, 28, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- Beheshti, I.; Nugent, S.; Potvin, O.; Duchesne, S. Disappearing Metabolic Youthfulness in the Cognitively Impaired Female Brain. Neurobiol. Aging 2021, 101, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Franke, K.; Ziegler, G.; Klöppel, S.; Gaser, C. Estimating the Age of Healthy Subjects from T1-Weighted MRI Scans Using Kernel Methods: Exploring the Influence of Various Parameters. NeuroImage 2010, 50, 883–892. [Google Scholar] [CrossRef]
- Beheshti, I.; Mishra, S.; Sone, D.; Khanna, P.; Matsuda, H. T1-Weighted MRI-Driven Brain Age Estimation in Alzheimer’s Disease and Parkinson’s Disease. Aging Dis. 2020, 11, 618–628. [Google Scholar] [CrossRef]
- Sone, D.; Beheshti, I.; Maikusa, N.; Ota, M.; Kimura, Y.; Sato, N.; Koepp, M.; Matsuda, H. Neuroimaging-Based Brain-Age Prediction in Diverse Forms of Epilepsy: A Signature of Psychosis and Beyond. Mol. Psychiatry 2021, 26, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Nenadić, I.; Dietzek, M.; Langbein, K.; Sauer, H.; Gaser, C. BrainAGE Score Indicates Accelerated Brain Aging in Schizophrenia, but Not Bipolar Disorder. Psychiatry Res. Neuroimaging 2017, 266, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.S.; Vlassenko, A.G.; Blazey, T.M.; Su, Y.; Couture, L.E.; Durbin, T.J.; Bateman, R.J.; Benzinger, T.L.-S.; Morris, J.C.; Raichle, M.E. Loss of Brain Aerobic Glycolysis in Normal Human Aging. Cell Metab. 2017, 26, 353–360.e3. [Google Scholar] [CrossRef] [PubMed]
- Knopman, D.S.; Jack, C.R.; Wiste, H.J.; Lundt, E.S.; Weigand, S.D.; Vemuri, P.; Lowe, V.J.; Kantarci, K.; Gunter, J.L.; Senjem, M.L.; et al. 18F-Fluorodeoxyglucose Positron Emission Tomography, Aging, and Apolipoprotein E Genotype in Cognitively Normal Persons. Neurobiol. Aging 2014, 35, 2096–2106. [Google Scholar] [CrossRef]
- Bonte, S.; Vandemaele, P.; Verleden, S.; Audenaert, K.; Deblaere, K.; Goethals, I.; Van Holen, R. Healthy Brain Ageing Assessed with 18F-FDG PET and Age-Dependent Recovery Factors after Partial Volume Effect Correction. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Thal, D.R.; Ghebremedhin, E.; Del Tredici, K. Stages of the Pathologic Process in Alzheimer Disease: Age Categories from 1 to 100 Years. J. Neuropathol. Exp. Neurol. 2011, 70, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Burkett, B.J.; Min, H.-K.; Senjem, M.L.; Lundt, E.S.; Botha, H.; Graff-Radford, J.; Barnard, L.R.; Gunter, J.L.; Schwarz, C.G.; et al. Deep Learning-Based Brain Age Prediction in Normal Aging and Dementia. Nat. Aging 2022, 2, 412–424. [Google Scholar] [CrossRef] [PubMed]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The Diagnosis of Dementia Due to Alzheimer’s Disease: Recommendations from the National Institute on Aging-Alzheimer’s Association Workgroups on Diagnostic Guidelines for Alzheimer’s Disease. Alzheimers Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R.; Wiste, H.J.; Weigand, S.D.; Therneau, T.M.; Lowe, V.J.; Knopman, D.S.; Gunter, J.L.; Senjem, M.L.; Jones, D.T.; Kantarci, K.; et al. Defining Imaging Biomarker Cut Points for Brain Aging and Alzheimer’s Disease. Alzheimers Dement. 2017, 13, 205–216. [Google Scholar] [CrossRef]
- MacMahon, S.; Peto, R.; Cutler, J.; Collins, R.; Sorlie, P.; Neaton, J.; Abbott, R.; Godwin, J.; Dyer, A.; Stamler, J. Blood Pressure, Stroke, and Coronary Heart Disease. Part 1, Prolonged Differences in Blood Pressure: Prospective Observational Studies Corrected for the Regression Dilution Bias. Lancet 1990, 335, 765–774. [Google Scholar] [CrossRef]
- Bashyam, V.M.; Erus, G.; Doshi, J.; Habes, M.; Nasrallah, I.M.; Truelove-Hill, M.; Srinivasan, D.; Mamourian, L.; Pomponio, R.; Fan, Y.; et al. MRI Signatures of Brain Age and Disease over the Lifespan Based on a Deep Brain Network and 14,468 Individuals Worldwide. Brain 2020, 143, 2312–2324. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Gong, W.; Beckmann, C.F.; Vedaldi, A.; Smith, S.M. Accurate Brain Age Prediction with Lightweight Deep Neural Networks. Med. Image Anal. 2021, 68, 101871. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, B.A.; Bjornsdottir, G.; Thorgeirsson, T.E.; Ellingsen, L.M.; Walters, G.B.; Gudbjartsson, D.F.; Stefansson, H.; Stefansson, K.; Ulfarsson, M.O. Brain Age Prediction Using Deep Learning Uncovers Associated Sequence Variants. Nat. Commun. 2019, 10, 5409. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.H.; Franke, K. Predicting Age Using Neuroimaging: Innovative Brain Ageing Biomarkers. Trends Neurosci. 2017, 40, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Benvenutto, A.; Giusiano, B.; Koric, L.; Gueriot, C.; Didic, M.; Felician, O.; Guye, M.; Guedj, E.; Ceccaldi, M. Imaging Biomarkers of Neurodegeneration in Alzheimer’s Disease: Distinct Contributions of Cortical MRI Atrophy and FDG-PET Hypometabolism. J. Alzheimers Dis. 2018, 65, 1147–1157. [Google Scholar] [CrossRef] [PubMed]
- Ossenkoppele, R.; Smith, R.; Ohlsson, T.; Strandberg, O.; Mattsson, N.; Insel, P.S.; Palmqvist, S.; Hansson, O. Associations between Tau, Aβ, and Cortical Thickness with Cognition in Alzheimer Disease. Neurology 2019, 92, e601–e612. [Google Scholar] [CrossRef] [PubMed]
- Wuestefeld, A.; Pichet Binette, A.; Berron, D.; Spotorno, N.; van Westen, D.; Stomrud, E.; Mattsson-Carlgren, N.; Strandberg, O.; Smith, R.; Palmqvist, S.; et al. Age-Related and Amyloid-Beta-Independent Tau Deposition and Its Downstream Effects. Brain 2023, 146, 3192–3205. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Brown, J.A.; Kim, H.R.; Joie, R.L.; Cho, H.; Lyoo, C.H.; Rabinovici, G.D.; Seong, J.-K.; Seeley, W.W. Regional Aβ-Tau Interactions Promote Onset and Acceleration of Alzheimer’s Disease Tau Spreading. Neuron 2022, 110, 1932–1943.e5. [Google Scholar] [CrossRef]
- Schoonhoven, D.N.; Coomans, E.M.; Millán, A.P.; van Nifterick, A.M.; Visser, D.; Ossenkoppele, R.; Tuncel, H.; van der Flier, W.M.; Golla, S.S.V.; Scheltens, P.; et al. Tau Protein Spreads through Functionally Connected Neurons in Alzheimer’s Disease: A Combined MEG/PET Study. Brain 2023, 146, 4040–4054. [Google Scholar] [CrossRef]
- Millar, P.R.; Gordon, B.A.; Luckett, P.H.; Benzinger, T.L.; Cruchaga, C.; Fagan, A.M.; Hassenstab, J.J.; Perrin, R.J.; Schindler, S.E.; Allegri, R.F.; et al. Multimodal Brain Age Estimates Relate to Alzheimer Disease Biomarkers and Cognition in Early Stages: A Cross-Sectional Observational Study. eLife 2023, 12, e81869. [Google Scholar] [CrossRef]
- Biel, D.; Luan, Y.; Brendel, M.; Hager, P.; Dewenter, A.; Moscoso, A.; Otero Svaldi, D.; Higgins, I.A.; Pontecorvo, M.; Römer, S.; et al. Combining Tau-PET and fMRI Meta-Analyses for Patient-Centered Prediction of Cognitive Decline in Alzheimer’s Disease. Alzheimers Res. Ther. 2022, 14, 166. [Google Scholar] [CrossRef] [PubMed]
- Saint-Aubert, L.; Lemoine, L.; Chiotis, K.; Leuzy, A.; Rodriguez-Vieitez, E.; Nordberg, A. Tau PET Imaging: Present and Future Directions. Mol. Neurodegener. 2017, 12, 19. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.; Zhang, F.; Niu, X.; Heywood, A.; Stocks, J.; Feng, G.; Popuri, K.; Beg, M.F.; Wang, L. Investigating the Temporal Pattern of Neuroimaging-Based Brain Age Estimation as a Biomarker for Alzheimer’s Disease Related Neurodegeneration. Neuroimage 2022, 263, 119621. [Google Scholar] [CrossRef]
- Bejanin, A.; Schonhaut, D.R.; La Joie, R.; Kramer, J.H.; Baker, S.L.; Sosa, N.; Ayakta, N.; Cantwell, A.; Janabi, M.; Lauriola, M.; et al. Tau Pathology and Neurodegeneration Contribute to Cognitive Impairment in Alzheimer’s Disease. Brain 2017, 140, 3286–3300. [Google Scholar] [CrossRef] [PubMed]
- Ossenkoppele, R.; van der Kant, R.; Hansson, O. Tau Biomarkers in Alzheimer’s Disease: Towards Implementation in Clinical Practice and Trials. Lancet Neurol. 2022, 21, 726–734. [Google Scholar] [CrossRef]
- Eavani, H.; Habes, M.; Satterthwaite, T.D.; An, Y.; Hsieh, M.-K.; Honnorat, N.; Erus, G.; Doshi, J.; Ferrucci, L.; Beason-Held, L.L.; et al. Heterogeneity of Structural and Functional Imaging Patterns of Advanced Brain Aging Revealed via Machine Learning Methods. Neurobiol. Aging 2018, 71, 41–50. [Google Scholar] [CrossRef]
- Liu, W.; Dong, Q.; Sun, S.; Shen, J.; Qian, K.; Hu, B. Risk Prediction of Alzheimer’s Disease Conversion in Mild Cognitive Impaired Population Based on Brain Age Estimation. IEEE Trans. Neural Syst. Rehabil. Eng. 2023, 31, 2468–2476. [Google Scholar] [CrossRef]
- Cheng, J.; Liu, Z.; Guan, H.; Wu, Z.; Zhu, H.; Jiang, J.; Wen, W.; Tao, D.; Liu, T. Brain Age Estimation From MRI Using Cascade Networks With Ranking Loss. IEEE Trans. Med. Imaging 2021, 40, 3400–3412. [Google Scholar] [CrossRef]
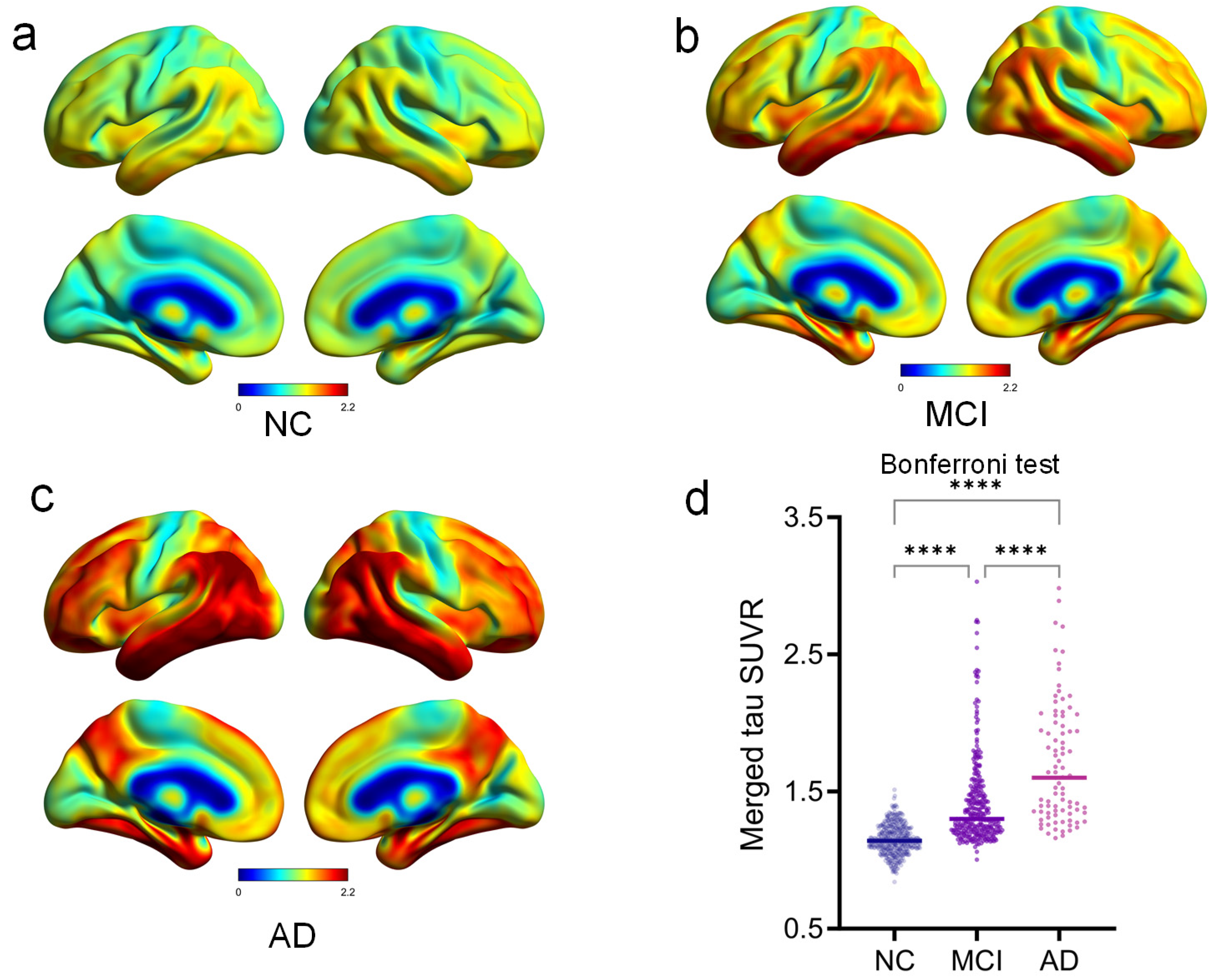



| NC | MCI | AD | p | Post Hoc p | |||
|---|---|---|---|---|---|---|---|
| p1 | p2 | p3 | |||||
| Number | 418 | 306 | 86 | - | - | - | - |
| Sex (M/F) | 259/159 | 170/136 | 54/32 | 0.203 | 0.083 | 0.885 | 0.232 |
| Age (years) | 72.7 ± 7.6 | 74.6 ± 7.3 | 76.9 ± 7.9 | <0.001 | 0.0014 | <0.001 | 0.341 |
| Education (years) | 16.7 ± 2.2 | 16.4 ± 2.5 | 15.4 ± 2.5 | <0.001 | 0.231 | <0.001 | 0.003 |
| APOE4 carriers (%) | 32.3 | 53.9 | 61.6 | - | - | - | - |
| ADAS11 | 5.2 ± 2.5 | 9.7 ± 4.4 | 20.6 ± 8.6 | <0.001 | <0.001 | <0.001 | <0.001 |
| ADAS13 | 8.2 ± 3.9 | 15.8 ± 6.8 | 31.5 ± 10.1 | <0.001 | <0.001 | <0.001 | <0.001 |
| MMSE | 29.2 ± 1.0 | 27.3 ± 2.3 | 21.5 ± 4.2 | <0.001 | <0.001 | <0.001 | <0.001 |
| MOCA | 26.2 ± 2.6 | 22.9 ± 3.2 | 16.3 ± 4.4 | <0.001 | <0.001 | <0.001 | <0.001 |
| Merged tau SUVR | 1.14 ± 0.12 | 1.42 ± 0.45 | 1.71 ± 0.63 | <0.001 | <0.001 | <0.001 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Wei, M.; Wang, L.; Song, J.; Rominger, A.; Shi, K.; Jiang, J., on behalf of the Alzheimer’s Disease Neuroimaging Initiative. Tau Protein Accumulation Trajectory-Based Brain Age Prediction in the Alzheimer’s Disease Continuum. Brain Sci. 2024, 14, 575. https://doi.org/10.3390/brainsci14060575
Wang M, Wei M, Wang L, Song J, Rominger A, Shi K, Jiang J on behalf of the Alzheimer’s Disease Neuroimaging Initiative. Tau Protein Accumulation Trajectory-Based Brain Age Prediction in the Alzheimer’s Disease Continuum. Brain Sciences. 2024; 14(6):575. https://doi.org/10.3390/brainsci14060575
Chicago/Turabian StyleWang, Min, Min Wei, Luyao Wang, Jun Song, Axel Rominger, Kuangyu Shi, and Jiehui Jiang on behalf of the Alzheimer’s Disease Neuroimaging Initiative. 2024. "Tau Protein Accumulation Trajectory-Based Brain Age Prediction in the Alzheimer’s Disease Continuum" Brain Sciences 14, no. 6: 575. https://doi.org/10.3390/brainsci14060575






