Dexborneol Amplifies Pregabalin’s Analgesic Effect in Mouse Models of Peripheral Nerve Injury and Incisional Pain
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Mouse Model of Spared Nerve Injury and Incisional Pain
2.3. Behavioral Tests in Mice
2.4. Pharmacological Treatments
2.5. The Calculation Method for Kim Jong-Kyun’s q Value
2.6. Cells
2.7. Treatment of BV2 Cells
2.8. Real-Time Quantitative PCR Analysis
2.9. Detection of Oxidative Stress Levels
2.10. Pharmacokinetic Experiments with Pregabalin and Dexborneol
2.11. HPLC-MS/MS
2.12. UPLC-HRMS Analysis for Lipidomic Studies
2.13. Perfusion and Cryo-Sectioning
2.14. Immunofluorescence Staining
2.15. Chemicals
3. Statistics
4. Results
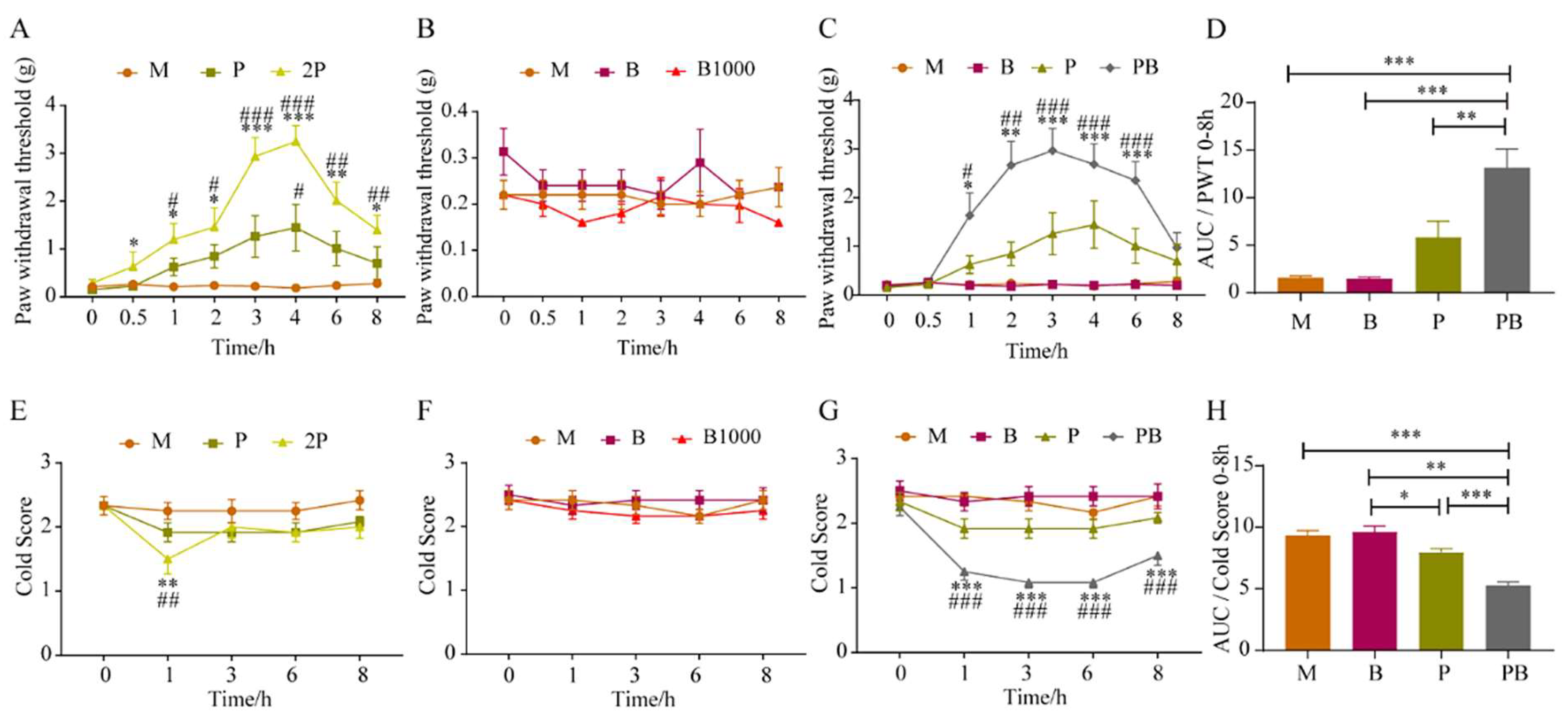
4.1. Dexborneol Augments Pregabalin’s Analgesic Effect in a Mouse Model of Peripheral Nerve Injury
4.2. Dexborneol Amplifies Pregabalin’s Analgesic Effectiveness in a Mouse Model of Incisional Pain
4.3. Dexborneol Can Promote Pregabalin Entry into the Central Nervous System to Exert Analgesic Effect
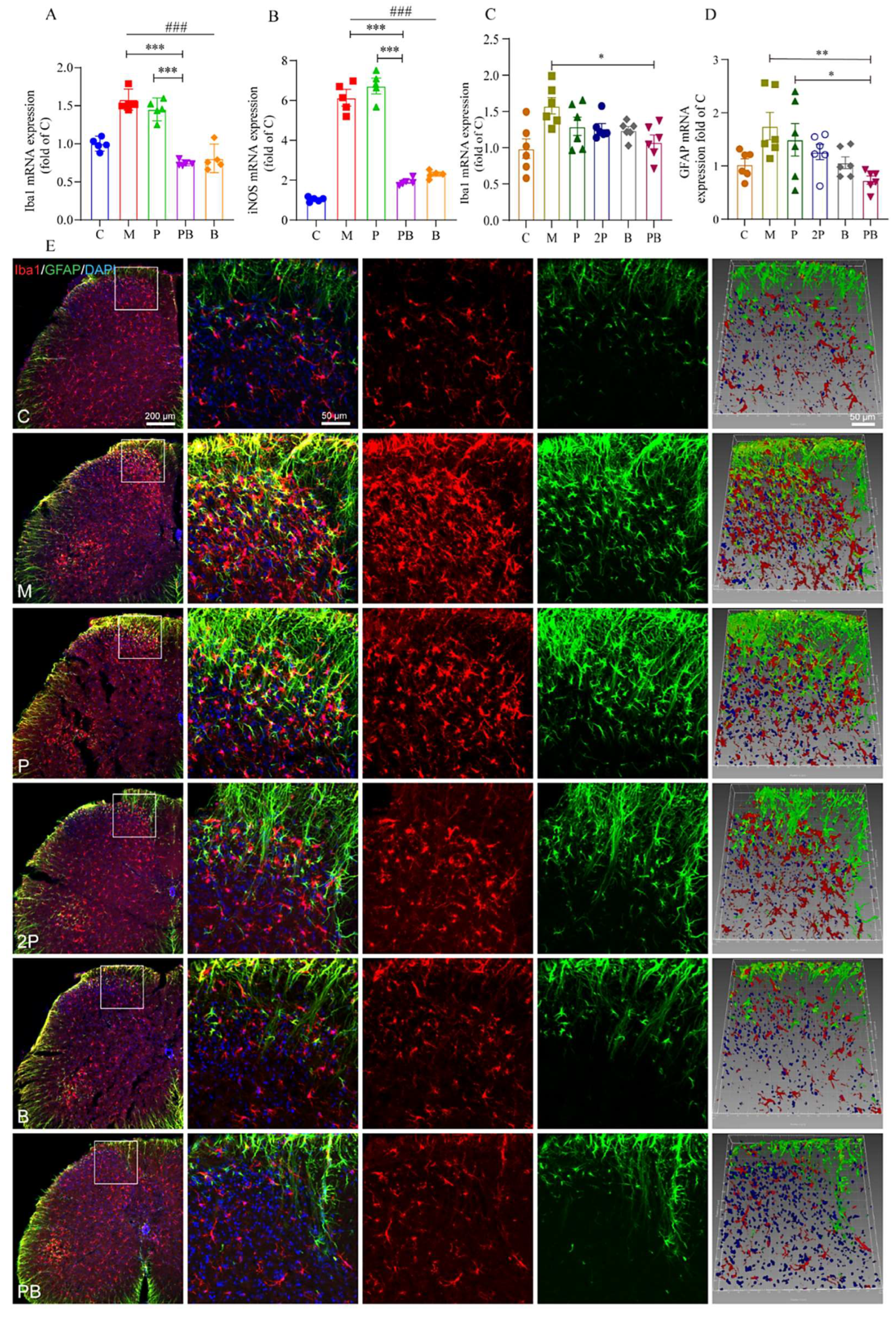
4.4. Pregabalin-Dexborneol Suppresses Glial Cell Activation in a Mouse Model of Peripheral Nerve Injury
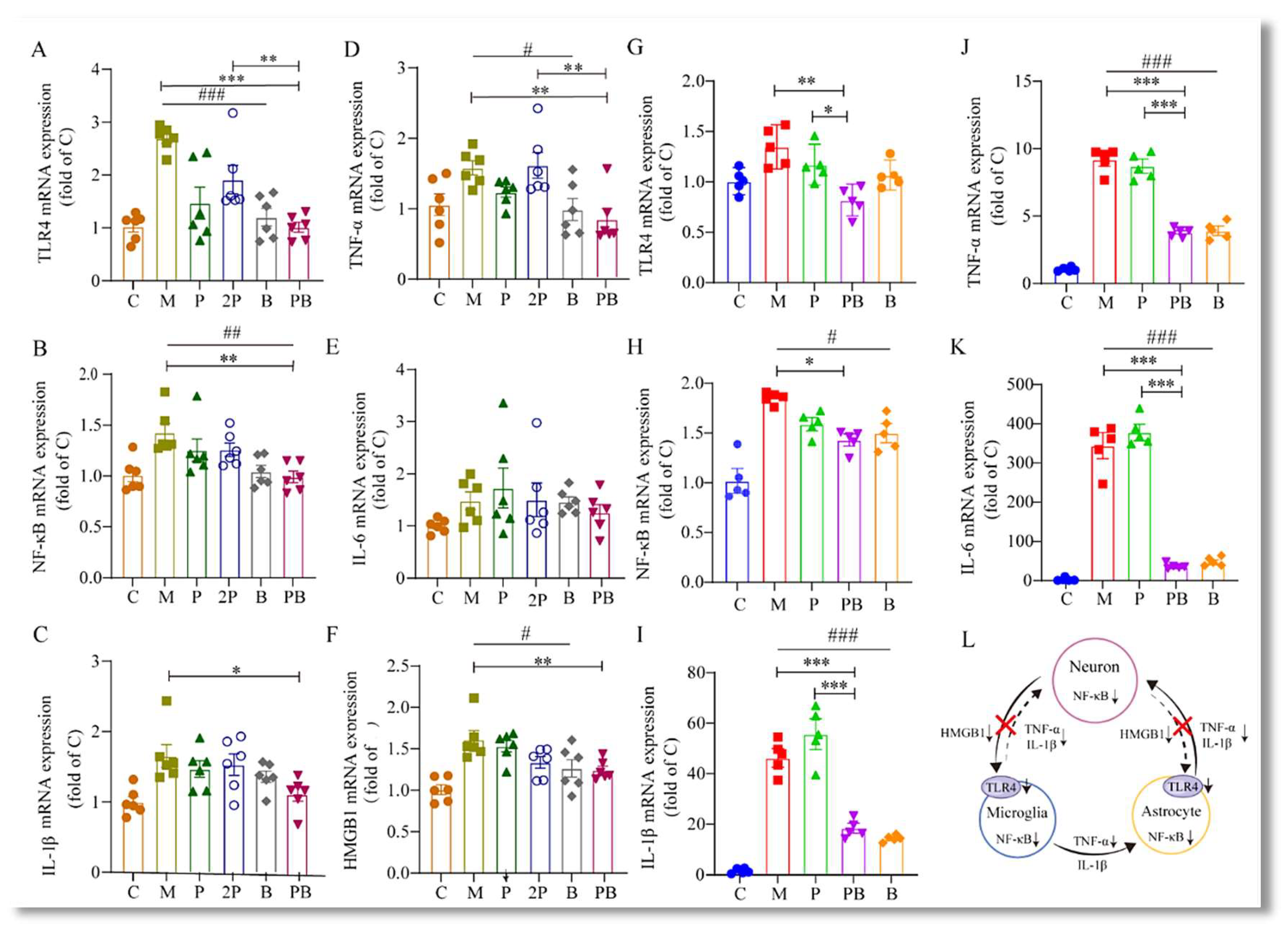
4.5. Pregabalin-Dexborneol Suppresses Detrimental SNI-Induced Neuroimmune Crosstalk and Neuroinflammation via HMGB1/TLR4/NF-κB Inhibition
4.6. Pregabalin-Dexborneol Activates Nrf2 to Regulate Oxidative Stress Levels and Disrupts a Self-Amplifying Oxidative Cycle in a Mouse Model of Peripheral Nerve Injury
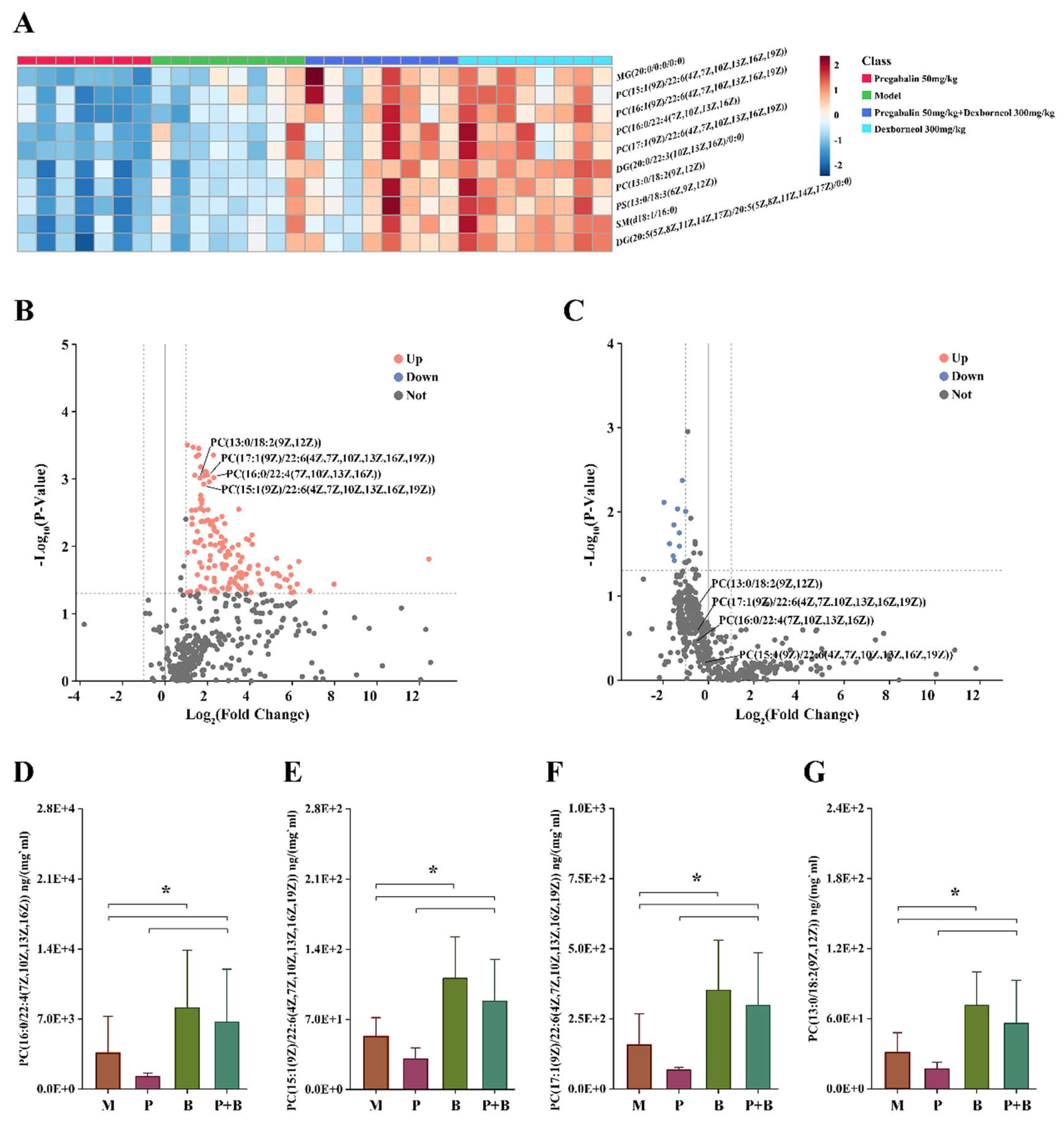
4.7. Dexborneol Exerts Analgesic Modulatory Effects through Lipid Mediators in a Mouse Model of Peripheral Nerve Injury
5. Discussion
5.1. Dexborneol Enhances Pregabalin’s Ability to Enter the CNS
5.2. Pregabalin Modulates Neuronal Signal Transmission
5.3. Dexborneol Alters the CNS Microenvironment
5.4. Pregabalin-Dexborneol Breaks HMGB1-Driven Inflammatory Cycle
5.5. Nrf2 Activation by Pregabalin-Dexborneol Disrupts a Self-Amplifying Oxidative Cycle
5.6. Lipid Metabolism as Key Upstream Mechanism Driving Pregabalin-Dexborneol’s Neuroprotection
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CDC | Centers for Disease Control and Prevention |
| GABA | γ-aminobutyric acid |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| ATP | adenosine triphosphate |
| MDA | malondialdehyde |
| LMs | lipid mediators |
| C | control group |
| P | pregabalin group |
| PB | combination of pregabalin and dexborneol group |
| B | dexborneol group |
| iNOS | inducible nitric oxide synthase |
| GFAP | glial fibrillary acidic protein |
| PC | phosphatidylcholine |
| DHA | docosahexaenoic acid |
| LAT1 | L-type Amino Acid Transporter 1 |
| NMDA | N-Methyl-D-aspartic acid |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| DMS | N,N-dimethylsphingosine |
References
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.; Ringkamp, M.; Sluka, K.; et al. The Revised International Association for the Study of Pain Definition of Pain: Concepts, Challenges, and Compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Zhen, Y.; Zhang, T.; Yang, X.; Feng, Z.; Qiu, F.; Xin, G.; Liu, J.; Nie, F.; Jin, X.; Liu, Y. A Survey of Chronic Pain in China. Libyan J. Med. 2020, 15, 1730550. [Google Scholar] [CrossRef]
- Ji, R.-R.; Nackley, A.; Huh, Y.; Terrando, N.; Maixner, W. Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. Anesthesiology 2018, 129, 343–366. [Google Scholar] [CrossRef]
- Teixeira-Santos, L.; Albino-Teixeira, A.; Pinho, D. Neuroinflammation, Oxidative Stress and Their Interplay in Neuropathic Pain: Focus on Specialized Pro-Resolving Mediators and Nadph Oxidase Inhibitors as Potential Therapeutic Strategies—Sciencedirect. Pharmacol. Res. 2020, 162, 105280. [Google Scholar] [CrossRef]
- Hendrix, J.; Nijs, J.; Ickmans, K.; Godderis, L.; Ghosh, M.; Polli, A. The Interplay between Oxidative Stress, Exercise, and Pain in Health and Disease: Potential Role of Autonomic Regulation and Epigenetic Mechanisms. Antioxidants 2020, 9, 1166. [Google Scholar] [CrossRef]
- Finnerup, N.B.; Kuner, R.; Jensen, T.S. Neuropathic Pain: From Mechanisms to Treatment. Physiol. Rev. 2020, 101, 259–301. [Google Scholar] [CrossRef]
- Carley, M.E.; Chaparro, L.E.; Choinière, M.; Kehlet, H.; Moore, R.A.; Kerkhof, E.V.D.; Gilron, I. Pharmacotherapy for the Prevention of Chronic Pain after Surgery in Adults: An Updated Systematic Review and Meta-analysis. Anesthesiology 2021, 135, 304–325. [Google Scholar] [CrossRef]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpää, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for Neuropathic Pain in Adults: A Systematic Review and Meta-Analysis. Lancet Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef]
- Bannister, K.; Sachau, J.; Baron, R.; Dickenson, A.H. Neuropathic Pain: Mechanism-Based Therapeutics. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 257–274. [Google Scholar] [CrossRef]
- Wiffen, P.J.; Derry, S.; Bell, R.F.; Rice, A.S.; Tölle, T.R.; Phillips, T.; Moore, R.A. Gabapentin for Chronic Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2017, 6, CD007938. [Google Scholar] [CrossRef] [PubMed]
- Macintyre, P.E.; Quinlan, J.; Levy, N.; Lobo, D.N. Current Issues in the Use of Opioids for the Management of Postoperative Pain: A Review. JAMA Surg. 2022, 157, 158–166. [Google Scholar] [CrossRef]
- Chowdhury, D.; Mundra, A.; Datta, D.; Duggal, A.; Krishnan, A.; Koul, A. Efficacy and Tolerability of Combination Treatment of Topiramate and Greater Occipital Nerve Block versus Topiramate Monotherapy for the Preventive Treatment of Chronic Migraine: A Randomized Controlled Trial. Cephalalgia 2022, 42, 859–871. [Google Scholar] [CrossRef]
- Gao, T.; Hao, J.; Wiesenfeld-Hallin, Z.; Wang, D.-Q.; Xu, X.-J. Analgesic Effect of Sinomenine in Rodents after Inflammation and Nerve Injury. Eur. J. Pharmacol. 2013, 721, 5–11. [Google Scholar] [CrossRef]
- Sun, H.; Li, X.; Chen, M.; Zhong, M.; Li, Y.; Wang, K.; Du, Y.; Zhen, X.; Gao, R.; Wu, Y.; et al. Multi-Omics-Guided Discovery of Omicsynins Produced by Streptomyces sp. 1647: Pseudo-Tetrapeptides Active Against Influenza A Viruses and Coronavirus HCoV-229E. Engineering 2021, 16, 176–186. [Google Scholar] [CrossRef]
- Hong, B.; Jiang, J.-D. Treating Chronic Diseases by Regulating the Gut Microbiota. Engineering 2022, 18, 17–20. [Google Scholar] [CrossRef]
- Gao, T.; Li, T.; Jiang, W.; Fan, W.; Xu, X.-J.; Zhao, X.; Yin, Z.; Guo, H.; Wang, L.; Gao, J.; et al. Antinociceptive Effects of Sinomenine Combined with Ligustrazine or Paracetamol in Animal Models of Incisional and Inflammatory Pain. Front. Physiol. 2021, 11, 523769. [Google Scholar] [CrossRef]
- Gao, T.; Shi, T.; Wiesenfeld-Hallin, Z.; Li, T.; Jiang, J.-D.; Xu, X.-J. Sinomenine Facilitates the Efficacy of Gabapentin or Ligustrazine Hydrochloride in Animal Models of Neuropathic Pain. Eur. J. Pharmacol. 2019, 854, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Briones-Valdivieso, C.; Briones, F.; Orellana-Urzúa, S.; Chichiarelli, S.; Saso, L.; Rodrigo, R. Novel Multi-Antioxidant Approach for Ischemic Stroke Therapy Targeting the Role of Oxidative Stress. Biomedicines 2024, 12, 501. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Y.; Shi, J.; Jiang, W.; Huang, W.; Chen, K.; Wang, X.; Zhang, G.; Li, Y.; Cao, C.; et al. Edaravone Dexborneol Alleviates Ischemic Injury and Neuroinflammation by Modulating Microglial and Astrocyte Polarization While Inhibiting Leukocyte Infiltration. Int. Immunopharmacol. 2024, 130, 111700. [Google Scholar] [CrossRef]
- Liu, S.; Long, Y.; Yu, S.; Zhang, D.; Yang, Q.; Ci, Z.; Cui, M.; Zhang, Y.; Wan, J.; Li, D. Borneol in Cardio-Cerebrovascular Diseases: Pharmacological Actions, Mechanisms, and Therapeutics. Pharmacol. Res. 2021, 169, 105627. [Google Scholar] [CrossRef] [PubMed]
- Decosterd, I.; Woolf, C.J. Spared Nerve Injury: An Animal Model of Persistent Peripheral Neuropathic Pain. Pain 2000, 87, 149–158. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration. Guidance for Industry Nonclinical Safety Evaluation of Drug Combinations. 2005. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents (accessed on 15 March 2006).
- LI, T.; Zhao, X.-l.; Gao, T.-l.; Jiao, Y.; Gao, W.-y.; Liu, Y.; Zhang, M.-y.; Wang, Z.-g.; Wang, D.-q. Microdialysis Sampling and Hplc-Ms/Ms Quantification of Sinomenine, Ligustrazine, Gabapentin, Paracetamol, Pregabalin and Amitriptyline in Rat Blood and Brain Extracellular Fluid. Acta Pharm. Sin. 2020, 55, 2198–2206. [Google Scholar]
- Colonna, M.; Butovsky, O. Microglia Function in the Central Nervous System During Health and Neurodegeneration. Annu. Rev. Immunol. 2017, 35, 441–468. [Google Scholar] [CrossRef]
- Filomeni, G.; De Zio, D.; Cecconi, F. Oxidative stress and Autophagy: The Clash between Damage and Metabolic Needs. Cell Death Differ. 2015, 22, 377–388. [Google Scholar] [CrossRef]
- Morgan, M.J.; Liu, Z.-g. Crosstalk of Reactive Oxygen Species and Nf-Κb Signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef]
- Jin, M.; Wang, H.; Liu, H.; Xia, Y.; Ruan, S.; Huang, Y.; Qiu, J.; Du, S.; Xu, L. Oxidative Stress Response and Proteomic Analysis Reveal the Mechanisms of Toxicity of Imidazolium-Based Ionic Liquids against Arabidopsis thaliana. Environ. Pollut. 2020, 260, 114013. [Google Scholar] [CrossRef]
- Li, M.; Wang, Y.; Banerjee, R.; Marinelli, F.; Silberberg, S.; Faraldo-Gomez, J.D.; Hattori, M.; Swartz, K.J. Molecular Mechanisms of Human P2x3 Receptor Channel Activation and Modulation by Divalent Cation Bound Atp. eLife 2019, 8, e47060. [Google Scholar] [CrossRef]
- Qiao, W.-L.; Li, Q.; Hao, J.-W.; Wei, S.; Li, X.-M.; Liu, T.-T.; Qiu, C.-Y.; Hu, W.-P. Enhancement of P2x3 Receptor-Mediated Currents by Lysophosphatidic Acid in Rat Primary Sensory Neurons. Front. Pharmacol. 2022, 13, 928647. [Google Scholar] [CrossRef]
- Xie, M.; Dunford, N.T. Lipid Composition and Emulsifying Properties of Canola Lecithin from Enzymatic Degumming. Food Chem. 2017, 218, 159–164. [Google Scholar] [CrossRef]
- Bianchetti, G.; Clementi, M.E.; Sampaolese, B.; Serantoni, C.; Abeltino, A.; De Spirito, M.; Sasson, S.; Maulucci, G. Metabolic Imaging and Molecular Biology Reveal the Interplay between Lipid Metabolism and DHA-Induced Modulation of Redox Homeostasis in RPE Cells. Antioxidants 2023, 12, 339. [Google Scholar] [CrossRef]
- Li, G.; Li, Y.; Xiao, B.; Cui, D.; Lin, Y.; Zeng, J.; Li, J.; Cao, M.-J.; Liu, J. Antioxidant Activity of Docosahexaenoic Acid (Dha) and Its Regulatory Roles in Mitochondria. J. Agric. Food Chem. 2021, 69, 1647–1655. [Google Scholar] [CrossRef]
- Shahine, Y.; El-Aal, S.A.A.; Reda, A.M.; Sheta, E.; Atia, N.M.; Abdallah, O.Y.; Ibrahim, S.S.A. Diosmin Nanocrystal Gel Alleviates Imiquimod-Induced Psoriasis in Rats Via Modulating Tlr7, 8/Nf-Κb/Micro Rna-31, Akt/Mtor/P70s6k Milieu, and Tregs/Th17 Balance. Inflammopharmacology 2023, 31, 1341–1359. [Google Scholar] [CrossRef]
- Zhu, W.; Li, Y.; Zhao, D.; Li, H.; Zhang, W.; Xu, J.; Hou, J.; Feng, X.; Wang, H. Dihydroartemisinin Suppresses Glycolysis of Lncap Cells by Inhibiting Pi3k/Akt Pathway and Downregulating Hif-1α Expression. Life Sci. 2019, 233, 116730. [Google Scholar] [CrossRef]
- Derry, S.; Bell, R.F.; Straube, S.; Wiffen, P.J.; Aldington, D.; Moore, R.A. Pregabalin for neuropathic pain in adults. Cochrane Database Syst. Rev. 2019, 2019, CD007076. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Huang, H.; Zhao, H.; Liu, R.; Sun, Z.; Liu, Y.; Chen, N.; Zhang, Z. Edaravone Dexborneol Protects against Cerebral Ischemia/Reperfusion-Induced Blood-Brain Barrier Damage by Inhibiting Ferroptosis Via Activation of Nrf-2/Ho-1/Gpx4 Signaling. Free. Radic. Biol. Med. 2024, 217, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zhang, K.; Shi, W.; Tang, Z. Research Progress on the Regulation and Mechanism of Borneol on the Blood-Brain Barrier in Pathological States: A Narrative Review Focused on Ischemic Stroke and Cerebral Glioma. Transl. Cancer Res. 2023, 12, 3198–3209. [Google Scholar] [CrossRef]
- Chen, Q.; Cai, Y.; Zhu, X.; Wang, J.; Gao, F.; Yang, M.; Mao, L.; Zhang, Z.; Sun, B. Edaravone Dexborneol Treatment Attenuates Neuronal Apoptosis and Improves Neurological Function by Suppressing 4-Hne-Associated Oxidative Stress After Subarachnoid Hemorrhage. Front. Pharmacol. 2022, 13, 848529. [Google Scholar] [CrossRef] [PubMed]
- Tampio, J.; Markowicz-Piasecka, M.; Montaser, A.; Rysä, J.; Kauppinen, A.; Huttunen, K.M. L-type Amino Acid Transporter 1 Utilizing Ferulic Acid Derivatives Show Increased Drug Delivery in the Mouse Pancreas Along with Decreased Lipid Peroxidation and Prostaglandin Production. Mol. Pharm. 2022, 19, 3806–3819. [Google Scholar] [CrossRef]
- Bushnell, M.C.; Čeko, M.; Low, L.A. Cognitive and Emotional Control of Pain and Its Disruption in Chronic Pain. Nat. Rev. Neurosci. 2013, 14, 502–511. [Google Scholar] [CrossRef]
- Bauer, C.S.; Nieto-Rostro, M.; Rahman, W.; Tran-Van-Minh, A.; Ferron, L.; Douglas, L.; Kadurin, I.; Sri Ranjan, Y.; Fernandez-Alacid, L.; Millar, N.S. 327 the A2d Ligand Pregabalin Inhibits the Increased Trafficking of the Calcium Channel Subunit A2d-1 to Presynaptic Terminals in Neuropathic Pain. Eur. J. Pain Suppl. 2010, 1, 94. [Google Scholar] [CrossRef]
- Deng, M.; Chen, S.-R.; Pan, H.-L. Presynaptic Nmda Receptors Control Nociceptive Transmission at the Spinal Cord Level in Neuropathic Pain. Cell. Mol. Life Sci. 2019, 76, 1889–1899. [Google Scholar] [CrossRef]
- Sills, G.J.; Rogawski, M.A. Mechanisms of Action of Currently Used Antiseizure Drugs. Neuropharmacology 2020, 168, 107966. [Google Scholar] [CrossRef]
- Yang, Q.; Zhou, J. Neuroinflammation in the Central Nervous System: Symphony of Glial Cells. Glia 2018, 67, 1017–1035. [Google Scholar] [CrossRef]
- Liu, Y.; Shen, X.; Zhang, Y.; Zheng, X.; Cepeda, C.; Wang, Y.; Duan, S.; Tong, X. Interactions of Glial Cells with Neuronal Synapses, from Astrocytes to Microglia and Oligodendrocyte Lineage Cells. Glia 2023, 71, 1383–1401. [Google Scholar] [CrossRef]
- Fiore, N.T.; Debs, S.R.; Hayes, J.P.; Duffy, S.S.; Moalem-Taylor, G. Pain-Resolving Immune Mechanisms in Neuropathic Pain. Nat. Rev. Neurol. 2023, 19, 199–220. [Google Scholar] [CrossRef]
- Inoue, K.; Tsuda, M. Microglia in Neuropathic Pain: Cellular and Molecular Mechanisms and Therapeutic Potential. Nat. Rev. Neurosci. 2018, 19, 138–152. [Google Scholar] [CrossRef]
- Takeda, I.; Yoshihara, K.; Cheung, D.L.; Kobayashi, T.; Agetsuma, M.; Tsuda, M.; Eto, K.; Koizumi, S.; Wake, H.; Moorhouse, A.J.; et al. Controlled Activation of Cortical Astrocytes Modulates Neuropathic Pain-like Behaviour. Nat. Commun. 2022, 13, 4100. [Google Scholar] [CrossRef]
- Zhang, Z.; Jiang, J.; He, Y.; Cai, J.; Xie, J.; Wu, M.; Xing, M.; Zhang, Z.; Chang, H.; Yu, P.; et al. Pregabalin Mitigates Microglial Activation and Neuronal Injury By Inhibiting Hmgb1 Signaling Pathway in Radiation-Induced Brain Injury. J. Neuroinflammation 2022, 19, 231. [Google Scholar] [CrossRef]
- Khan, A.; Shal, B.; Khan, A.U.; Baig, M.W.; Ul Haq, I.; Khan, S. Withametelin, a Steroidal Lactone, Isolated from Datura Innoxa Attenuates Stz-Induced Diabetic Neuropathic Pain in Rats through Inhibition of Nf-Kb/Mapk Signaling. Food Chem. Toxicol. 2023, 175, 113742. [Google Scholar] [CrossRef]
- Tomić, M.; Pecikoza, U.; Micov, A.; Vučković, S.; Stepanović-Petrović, R. Antiepileptic Drugs as Analgesics/Adjuvants in Inflammatory Pain: Current Preclinical Evidence. Pharmacol. Ther. 2018, 192, 42–64. [Google Scholar] [CrossRef]
- Wang, C.; Gu, H.-Q.; Dong, Q.; Xu, A.; Wang, N.; Yang, Y.; Wang, F.; Wang, Y. Rationale and Design of Treatment of Acute Ischaemic Stroke with Edaravone Dexborneol II (Taste-2): A Multicentre Randomised Controlled Trial. Stroke Vasc. Neurol. 2024. [Google Scholar] [CrossRef]
- Li, L.; He, G.; Shi, M.; Zhu, J.; Cheng, Y.; Chen, Y.; Chen, J.; Xue, Q. Edaravone Dexborneol Ameliorates Cognitive Impairment by Regulating the Nf-Κb Pathway through Ahr and Promoting Microglial Polarization Towards the M2 Phenotype in Mice with Bilateral Carotid Artery Stenosis (Bcas). Eur. J. Pharmacol. 2023, 957, 176036. [Google Scholar] [CrossRef]
- Zhang, D.; Li, X.; Jing, B.; Chen, Z.; Shi, H.; Zheng, Y.; Chang, S.; Sun, J.; Zhao, G. α-Asarone Attenuates Chronic Sciatica by Inhibiting Peripheral Sensitization and Promoting Neural Repair. Phytotherapy Res. 2023, 37, 151–162. [Google Scholar] [CrossRef]
- Mo, Y.; Chen, K. The Role of Hmgb1 in Spinal Cord Injury. Front. Immunol. 2023, 13, 1094925. [Google Scholar] [CrossRef] [PubMed]
- Shi, T.; van Soest, D.M.; Polderman, P.E.; Burgering, B.M.; Dansen, T.B. DNA Damage and Oxidant Stress Activate p53 through Differential Upstream Signaling Pathways. Free. Radic. Biol. Med. 2021, 172, 298–311. [Google Scholar] [CrossRef]
- Yin, C.; Liu, B.; Wang, P.; Li, X.; Li, Y.; Zheng, X.; Tai, Y.; Wang, C.; Liu, B. Eucalyptol Alleviates Inflammation and Pain Responses in a Mouse Model of Gout Arthritis. Br. J. Pharmacol. 2019, 177, 2042–2057. [Google Scholar] [CrossRef]
- Xia, L.-P.; Luo, H.; Ma, Q.; Xie, Y.-K.; Li, W.; Hu, H.; Xu, Z.-Z. GPR151 in Nociceptors Modulates Neuropathic Pain via Regulating P2x3 Function and Microglial Activation. Brain 2021, 144, 3405–3420. [Google Scholar] [CrossRef] [PubMed]
- Giniatullin, R.; Nistri, A. Role of Atp in Migraine Mechanisms: Focus on P2x3 receptors. J. Headache Pain 2023, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Hybertson, B.M.; Gao, B.; Bose, S.K.; McCord, J.M. Oxidative Stress in Health and Disease: The Therapeutic Potential of Nrf2 Activation. Mol. Asp. Med. 2011, 32, 234–246. [Google Scholar] [CrossRef]
- Li, J.; Tian, M.; Hua, T.; Wang, H.; Yang, M.; Li, W.; Zhang, X.; Yuan, H. Combination of Autophagy and Nfe2l2/Nrf2 Activation as a Treatment Approach for Neuropathic Pain. Autophagy 2021, 17, 4062–4082. [Google Scholar] [CrossRef]
- Wu, Z.; Bagarolo, G.I.; Thoröe-Boveleth, S.; Jankowski, J. “Lipidomics”: Mass Spectrometric and Chemometric Analyses of Lipids. Adv. Drug Deliv. Rev. 2020, 159, 294–307. [Google Scholar] [CrossRef] [PubMed]
- Patti, G.J.; Yanes, O.; Shriver, L.P.; Courade, J.-P.; Tautenhahn, R.; Manchester, M.; Siuzdak, G. Metabolomics Implicates Altered Sphingolipids in Chronic Pain of Neuropathic Origin. Nat. Chem. Biol. 2012, 8, 232–234. [Google Scholar] [CrossRef]
- Li, J.; Satyshur, K.A.; Guo, L.W.; Ruoho, A.E. Sphingoid Bases Regulate the Sigma-1 Receptor—Sphingosine and N, N’-Dimethylsphingosine Are Endogenous Agonists. Int. J. Mol. Sci. 2023, 24, 3103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xia, Y.; Cao, L.; Ji, B.; Ge, Z.; Zheng, Q.; Qi, Z.; Ding, S. Pc 18: 1/18: 1 Mediates the Anti-Inflammatory Effects of Exercise and Remodels Tumor Microenvironment of Hepatocellular Carcinoma. Life Sci. 2024, 336, 122335. [Google Scholar] [CrossRef]





| Models and Pains | Pregabalin 50 mg/kg | Dexborneol 300 mg/kg | Pregabalin 50 mg/kg + Dexborneol 300 mg/kg | Jong-Kyun’s q Value |
|---|---|---|---|---|
| mechanical pain of SNI | 13.95 | −0.44 | 37.91 | 2.79 |
| cold pain of SNI | 14.73 | −3.12 | 43.30 | 3.59 |
| mechanical pain of INCI | 6.32 | −0.24 | 21.54 | 3.54 |
| cold pain of INCI | 14.41 | 6.80 | 26.69 | 1.32 |
| Plasma | Brain | ||||
|---|---|---|---|---|---|
| Parameter | Units | Pregabalin 50 mg/kg | Pregabalin 50 mg/kg + Dexborneol 300 mg/kg | Pregabalin 50 mg/kg | Pregabalin 50 mg/kg + Dexborneol 300 mg/kg |
| AUC (0–t) | μg/L·h | 111,623.20 ± 7951.00 | 131,453.10 ± 14,515.35 | 16,913.72 ± 1675.77 | 21,313.56 ± 3665.82 |
| AUC (0–∞) | μg/L·h | 111,921.78 ± 7824.53 | 131,821.18 ± 14,443.95 | 17,763.44 ± 1819.73 | 22,617.36 ± 4462.52 |
| MRT (0–t) | h | 1.80 ± 0.22 | 1.96 ± 0.22 | 2.88 ± 0.24 | 3.09 ± 0.19 |
| MRT (0–∞) | h | 1.82 ± 0.22 | 1.99 ± 0.21 | 3.24 ± 0.32 | 3.49 ± 0.64 |
| t1/2z | h | 0.84 ± 0.10 | 0.86 ± 0.12 | 1.60 ± 0.27 | 1.49 ± 0.60 |
| Tmax | h | 0.58 ± 0.20 | 0.83 ± 0.61 | 1.17 ± 0.41 | 2.17 ± 0.41 |
| Vz/F | L/kg | 0.54 ± 0.10 | 0.48 ± 0.10 | 6.48 ± 0.94 | 4.74 ± 1.45 |
| CLz/F | L/h/kg | 0.45 ± 0.03 | 0.38 ± 0.04 | 2.84 ± 0.27 | 2.28 ± 0.45 |
| Cmax | μg/L | 56,254.25 ± 8451.01 | 60,860.60 ± 13,515.34 | 4288.62 ± 450.78 | 5581.82 ± 1368.57 |
| Compounds | Molecular Formula | ANOVA | ANOVA | FC |
|---|---|---|---|---|
| (Inter Groups) | (PB vs. P) | (PB vs. P) | ||
| PC(16:0/22:4(7Z,10Z,13Z,16Z)) | C46H84NO8P | 2.45 × 10−4 | 3.34 × 10−3 | 4.94 |
| PC(15:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C45H76NO8P | 5.28 × 10−5 | 4.17 × 10−4 | 3.54 |
| PC(17:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C47H80NO8P | 1.07 × 10−4 | 1.99 × 10−3 | 4.02 |
| PC(13:0/18:2(9Z,12Z)) | C39H74NO8P | 9.63 × 10−6 | 6.03 × 10−4 | 3.15 |
| PC(16:1(9Z)/22:6(4Z,7Z,10Z,13Z,16Z,19Z)) | C46H78NO8P | 7.94 × 10−1 | 9.85 × 10−1 | 1.45 |
| SM(d18:1/16:0) | C39H79N2O6P | 5.22 × 10−6 | 7.15 × 10−4 | 3.63 |
| DG(20:5(5Z,8Z,11Z,14Z,17Z)/20:5(5Z,8Z,11Z,14Z,17Z)/0:0) | C43H64O5 | 5.37 × 10−3 | 6.79 × 10−2 | 38.54 |
| DG(20:0/22:3(10Z,13Z,16Z)/0:0) | C45H82O5 | 1.64 × 10−3 | 5.54 × 10−2 | 8.29 |
| PS(13:0/18:3(6Z,9Z,12Z)) | C37H66NO10P | 4.96 × 10−3 | 8.15 × 10−2 | 2.96 |
| MG(20:0/0:0/0:0) | C23H46O4 | 4.28 × 10−5 | 2.66 × 10−4 | 4.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Z.; Guo, Y.-D.; Tang, M.-Z.; Zhou, P.; Su, Y.-X.; Shen, H.-R.; Li, T.; Jiang, W.; Han, Y.-X.; Tie, C.; et al. Dexborneol Amplifies Pregabalin’s Analgesic Effect in Mouse Models of Peripheral Nerve Injury and Incisional Pain. Antioxidants 2024, 13, 803. https://doi.org/10.3390/antiox13070803
Shen Z, Guo Y-D, Tang M-Z, Zhou P, Su Y-X, Shen H-R, Li T, Jiang W, Han Y-X, Tie C, et al. Dexborneol Amplifies Pregabalin’s Analgesic Effect in Mouse Models of Peripheral Nerve Injury and Incisional Pain. Antioxidants. 2024; 13(7):803. https://doi.org/10.3390/antiox13070803
Chicago/Turabian StyleShen, Zhen, Yun-Dan Guo, Ming-Ze Tang, Ping Zhou, Yu-Xin Su, Hao-Ran Shen, Tao Li, Wei Jiang, Yan-Xing Han, Cai Tie, and et al. 2024. "Dexborneol Amplifies Pregabalin’s Analgesic Effect in Mouse Models of Peripheral Nerve Injury and Incisional Pain" Antioxidants 13, no. 7: 803. https://doi.org/10.3390/antiox13070803
APA StyleShen, Z., Guo, Y.-D., Tang, M.-Z., Zhou, P., Su, Y.-X., Shen, H.-R., Li, T., Jiang, W., Han, Y.-X., Tie, C., Cui, J.-J., Gao, T.-L., & Jiang, J.-D. (2024). Dexborneol Amplifies Pregabalin’s Analgesic Effect in Mouse Models of Peripheral Nerve Injury and Incisional Pain. Antioxidants, 13(7), 803. https://doi.org/10.3390/antiox13070803







