Hepatic Oxidative Stress and Cell Death Influenced by Dietary Lipid Levels in a Fresh Teleost
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Diets
2.2. Management and Feeding Practices for the Fish
2.3. Sample Collection
2.4. Biochemical Analysis
2.5. Quantitative Real-Time Reverse-Transcriptase Polymerase Chain Reaction (qRT-PCR)
2.6. Protein Extraction and WB Analysis
2.7. Fe2+ Content Assay
2.8. Liver Histology
2.9. Statistical Analysis
3. Results
3.1. Fish Growth Performance
3.2. Fish Proximate Composition
3.3. Plasma and Liver Parameters
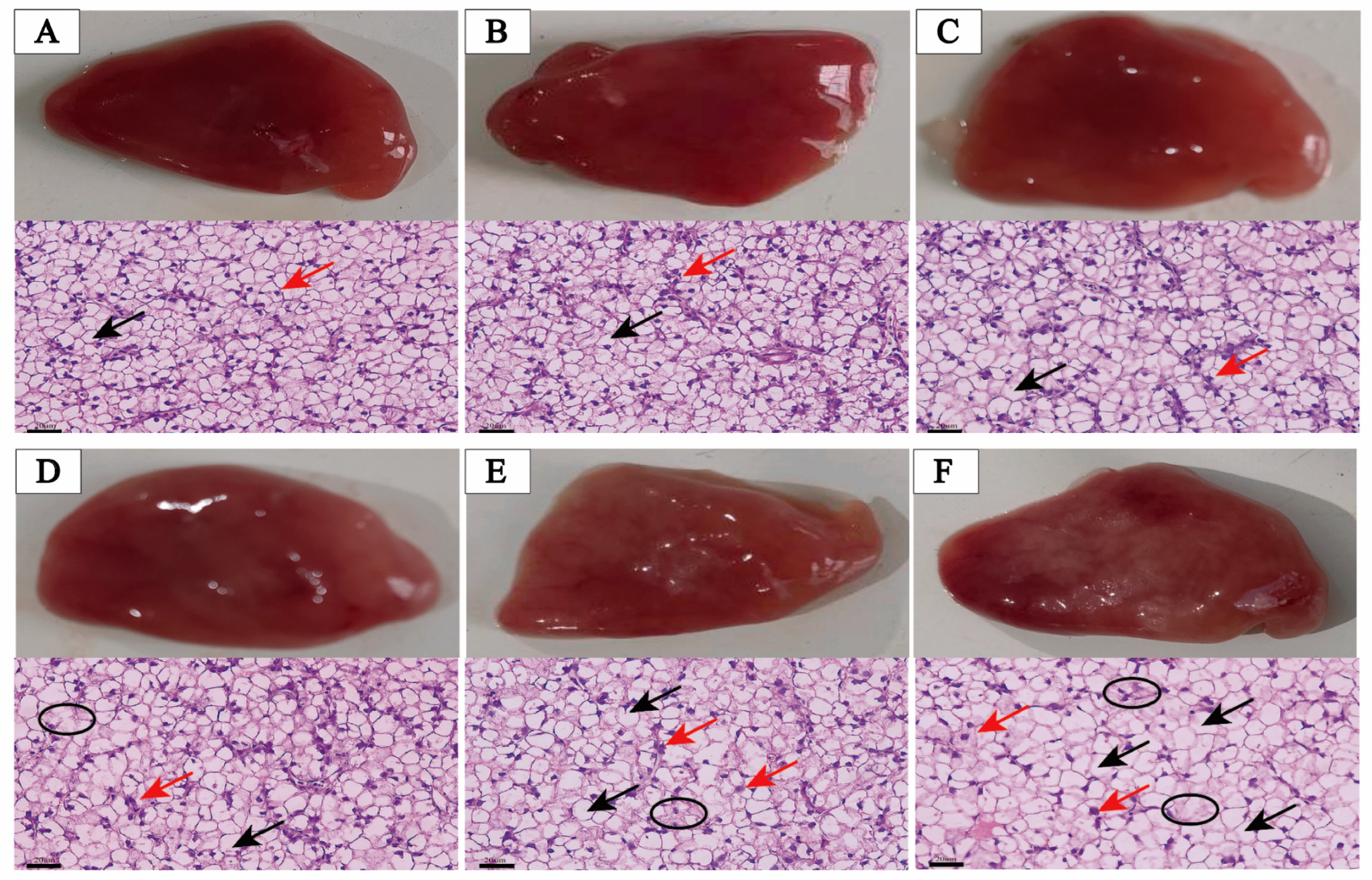
3.4. Liver Examination and Histology
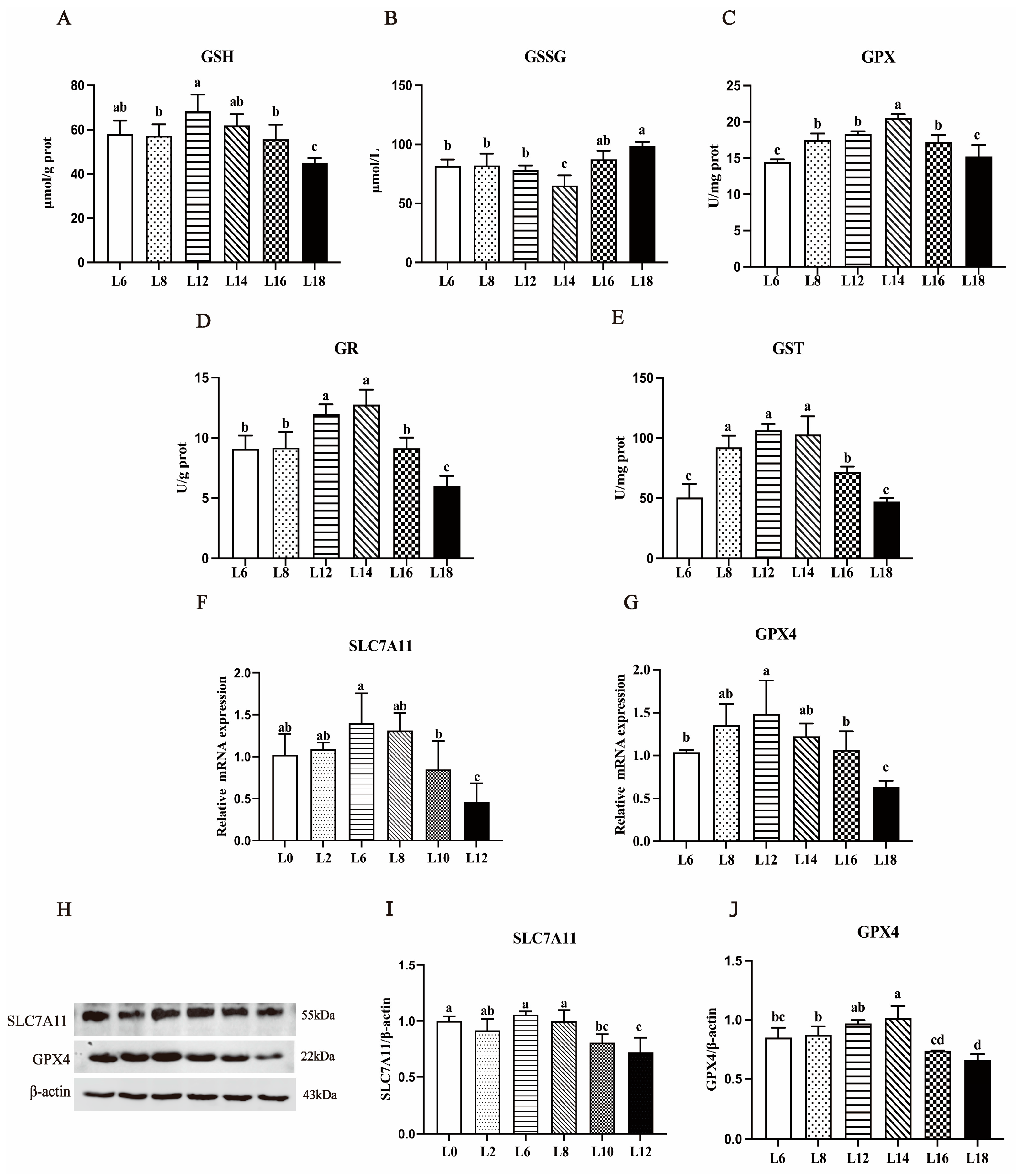
3.5. Liver Antioxidant Indicators
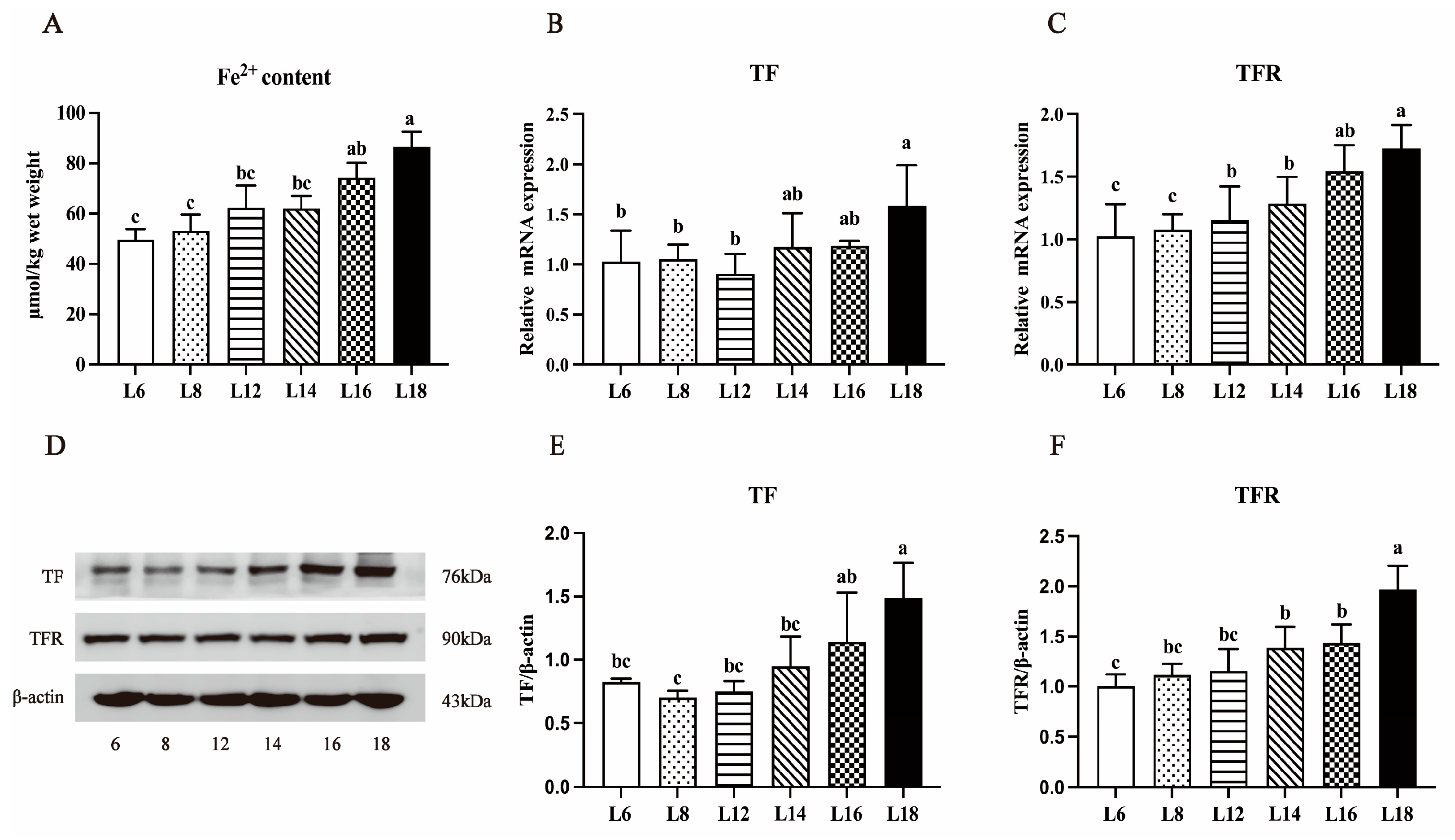
3.6. Liver Fe2+ Concentration and the Expressions of TF and TFR
3.7. ACSL4/LPCAT3/LOX12 Axis
4. Discussion
4.1. Effects of Lipid Levels on Growth Performance, Feed Utilization, and Body Composition of Largemouth Bass
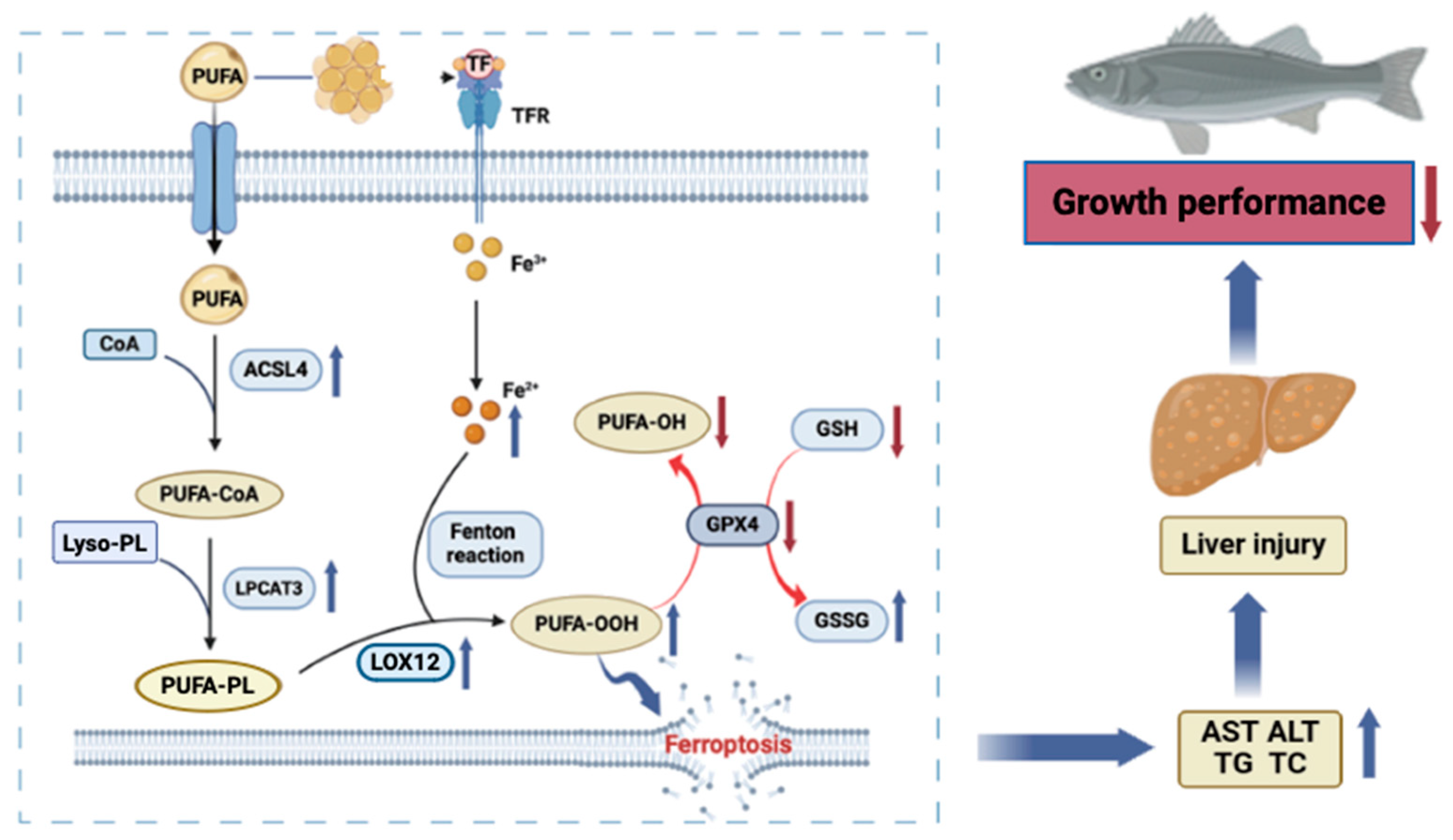
4.2. High Lipid Level Diet Can Up-Regulate the ACSL4/LPCAT3/LOX12 Axis and Promote Hepatic Oxidative Stress and Ferroptosis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Q.; Wang, C.; Sun, Y.; Chen, Y.; Chen, S.; Han, T.; Wang, J. Excessive Substitution of Fish Meal with Fermented Soybean Meal Induces Oxidative Stress by Impairing Glutathione Metabolism in Largemouth Bass (Micropterus salmoides). Antioxidants 2023, 12, 2096. [Google Scholar] [CrossRef]
- Cai, W.; Fu, L.; Liu, H.; Yi, J.; Yang, F.; Hua, L.; He, L.; Han, D.; Zhu, X.; Yang, Y.; et al. Dietary yeast glycoprotein supplementation improves the growth performance, intestinal health and disease resistance of largemouth bass (Micropterus salmoides) fed low-fishmeal diets. Front. Immunol. 2023, 14, 1164087. [Google Scholar] [CrossRef] [PubMed]
- Peng, D.; Peng, B.; Li, J.; Zhang, Y.; Luo, H.; Xiao, Q.; Tang, S.; Liang, X.-F. Effects of three feed attractants on the growth, biochemical indicators, lipid metabolism and appetite of Chinese perch (Siniperca chuatsi). Aquac. Rep. 2022, 23, 101075. [Google Scholar] [CrossRef]
- Xiao, W.; Zou, Z.; Li, D.; Zhu, J.; Yue, Y.; Yang, H. Effect of dietary phenylalanine level on growth performance, body composition, and biochemical parameters in plasma of juvenile hybrid tilapia, Oreochromis niloticus × Oreochromis aureus. J. World Aquac. Soc. 2020, 51, 437–451. [Google Scholar] [CrossRef]
- Huang, D.; Wu, Y.; Lin, Y.; Chen, J.; Karrow, N.; Ren, X.; Wang, Y. Dietary Protein and Lipid Requirements for Juvenile Largemouth Bass, Micropterus salmoides. J. World Aquac. Soc. 2017, 48, 782–790. [Google Scholar] [CrossRef]
- Li, X.; Zheng, S.; Ma, X.; Cheng, K.; Wu, G. Effects of dietary protein and lipid levels on the growth performance, feed utilization, and liver histology of largemouth bass (Micropterus salmoides). Amino Acids 2020, 52, 1043–1061. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, H.; Guo, B.; Li, X.; Leng, X. Dietary effects of protein and lipid levels on growth performance and flesh quality of large-size largemouth bass (Micropterus salmoides). Aquac. Rep. 2023, 33, 101852. [Google Scholar] [CrossRef]
- Chen, N.S.; Xiao, W.W.; Liang, Q.L.; Zhou, H.Y.; Ma, X.L.; Zhao, M. Effects of dietary lipid to protein ratios on growth performance, body composition and non-specific immunity of largemouth bass (Micropterus salmoides). J. Fish. China 2012, 36, 1270–1280. [Google Scholar] [CrossRef]
- Zhou, Y.L.; Guo, J.L.; Tang, R.J.; Ma, H.J.; Chen, Y.J.; Lin, S.M. High dietary lipid level alters the growth, hepatic metabolism enzyme, and anti-oxidative capacity in juvenile largemouth bass Micropterus salmoides. Fish Physiol. Biochem. 2020, 46, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.L.; Zhou, Y.L.; Zhao, H.; Chen, W.Y.; Chen, Y.J.; Lin, S.M. Effect of dietary lipid level on growth, lipid metabolism and oxidative status of largemouth bass, Micropterus salmoides. Aquaculture 2019, 506, 394–400. [Google Scholar] [CrossRef]
- Borges, P.; Valente, L.M.P.; Veron, V.; Dias, K.; Panserat, S.; Medale, F. High Dietary Lipid Level Is Associated with Persistent Hyperglycaemia and Downregulation of Muscle Akt-mTOR Pathway in Senegalese Sole (Solea senegalensis). PLoS ONE 2014, 9, e102196. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.; Yan, J.; Mai, K.; Ai, Q. Dietary lipid concentration affects liver mitochondrial DNA copy number, gene expression and DNA methylation in large yellow croaker (Larimichthys crocea). Comp. Biochem. Physiol. Part B Mol. Integr. Physiol. 2016, 193, 25–32. [Google Scholar] [CrossRef]
- Wang, J.; Liu, T.; Zheng, P.; Xu, H.; Su, H.; Tao, H.; Yang, Y. Effect of dietary lipid levels on growth performance, body composition, and feed utilization of juvenile spotted knifejaw Oplegnathus punctatus. Aquac. Rep. 2021, 21, 100797. [Google Scholar] [CrossRef]
- Cho, C.Y.; Kaushik, S.J. Nutritional energetics in fish: Energy and protein utilization in rainbow trout (Salmo gairdneri). World Rev. Nutr. Diet. 1990, 61, 132–172. [Google Scholar] [PubMed]
- Kaushik, S.J.; Médale, F. Energy requirements, utilization and dietary supply to salmonids. Aquaculture 1994, 124, 81–97. [Google Scholar] [CrossRef]
- Kim, H.; Park, M.; Shin, J.H.; Kwon, O. Ethanolic Extract of Acanthopanax koreanum Nakai Alleviates Alcoholic Liver Damage Combined with a High-Fat Diet in C57BL/6J Mice. Molecules 2016, 21, 681. [Google Scholar] [CrossRef]
- Qi, J.; Kim, J.W.; Zhou, Z.; Lim, C.-W.; Kim, B. Ferroptosis Affects the Progression of Nonalcoholic Steatohepatitis via the Modulation of Lipid Peroxidation–Mediated Cell Death in Mice. Am. J. Pathol. 2020, 190, 68–81. [Google Scholar] [CrossRef]
- Tsurusaki, S.; Tsuchiya, Y.; Koumura, T.; Nakasone, M.; Sakamoto, T.; Matsuoka, M.; Imai, H.; Yuet-Yin Kok, C.; Okochi, H.; Nakano, H.; et al. Hepatic ferroptosis plays an important role as the trigger for initiating inflammation in nonalcoholic steatohepatitis. Cell Death Dis. 2019, 10, 449. [Google Scholar] [CrossRef]
- Zhang, X.J.; Zhou, L.; Lu, W.J.; Du, W.X.; Mi, X.Y.; Li, Z.; Li, X.Y.; Wang, Z.W.; Wang, Y.; Duan, M.; et al. Comparative transcriptomic analysis reveals an association of gibel carp fatty liver with ferroptosis pathway. BMC Genom. 2021, 22, 328. [Google Scholar] [CrossRef]
- Chen, X.; Li, J.; Kang, R.; Klionsky, D.J.; Tang, D. Ferroptosis: Machinery and regulation. Autophagy 2021, 17, 2054–2081. [Google Scholar] [CrossRef]
- Li, Y.; Feng, D.; Wang, Z.; Zhao, Y.; Sun, R.; Tian, D.; Liu, D.; Zhang, F.; Ning, S.; Yao, J.; et al. Ischemia-induced ACSL4 activation contributes to ferroptosis-mediated tissue injury in intestinal ischemia/reperfusion. Cell Death Differ. 2019, 26, 2284–2299. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Yang, W.S.; Stockwell, B.R. Ferroptosis: Death by Lipid Peroxidation. Trends Cell Biol. 2016, 26, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- YunFei, Z.; ChaoMing, S.; Yuelang, Z.; YongJun, C.; ShiMei, L.; RenJun, T. Optimum dietary fiber level could improve growth, plasma biochemical indexes and liver function of largemouth bass, Micropterus salmoides. Aquaculture 2020, 518, 734661. [Google Scholar]
- Shen, Y.; Li, X.; Bao, Y.; Zhu, T.; Wu, Z.; Yang, B.; Jiao, L.; Zhou, Q.; Jin, M. Differential regulatory effects of optimal or excessive dietary lipid levels on growth, lipid metabolism and physiological response in black seabream (Acanthopagrus schlegelii). Aquaculture 2022, 560, 738532. [Google Scholar] [CrossRef]
- Xu, J.H.; Qin, J.; Yan, B.L.; Zhu, M.; Luo, G. Effects of dietary lipid levels on growth performance, feed utilization and fatty acid composition of juvenile Japanese seabass (Lateolabrax japonicus) reared in seawater. Aquac. Int. 2011, 19, 79–89. [Google Scholar] [CrossRef]
- Fei, S.; Chen, Z.; Duan, Y.; Liu, H.; Jin, J.; Yang, Y.; Han, D.; Zhu, X.; Xie, S. Growth, reproduction, fatty acid profiles and offspring performance of broodstock yellow catfish Pelteobagrus fulvidraco fed diets with different lipid levels. Aquaculture 2024, 580, 740273. [Google Scholar] [CrossRef]
- Bertucci, J.I.; Blanco, A.M.; Navarro, J.C.; Unniappan, S.; Canosa, L.F. Dietary protein:lipid ratio modulates somatic growth and expression of genes involved in somatic growth, lipid metabolism and food intake in Pejerrey fry (Odontesthes bonariensis). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2022, 270, 111231. [Google Scholar] [CrossRef]
- Calo, J.; Conde-Sieira, M.; Comesana, S.; Soengas, J.L.; Blanco, A.M. Fatty acids of different nature differentially modulate feed intake in rainbow trout. Aquaculture 2023, 563, 738961. [Google Scholar] [CrossRef]
- Ruyter, B.; Moya-Falcón, C.; Rosenlund, G.; Vegusdal, A. Fat content and morphology of liver and intestine of Atlantic salmon (Salmo salar): Effects of temperature and dietary soybean oil. Aquaculture 2006, 252, 441–452. [Google Scholar] [CrossRef]
- Abdel-Ghany, H.M.; Salem, M.E.-S.; Ezzat, A.A.; Essa, M.A.; Helal, A.M.; Ismail, R.F.; El-Sayed, A.-F.M. Effects of different levels of dietary lipids on growth performance, liver histology and cold tolerance of Nile tilapia (Oreochromis niloticus). J. Therm. Biol. 2021, 96, 102833. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, W.D.; Wu, P.; Liu, Y.; Jin, X.W.; Jiang, J.; Tang, J.Y.; Feng, L.; Zhou, X.Q. Dietary selenium promoted muscle growth (myofiber hypertrophy rather than hyperplasia) by reducing endoplasmic reticulum stress in juvenile grass carp (Ctenopharyngodon idella). Aquaculture 2023, 575, 739796. [Google Scholar] [CrossRef]
- Wang, J.-T.; Liu, Y.-J.; Tian, L.-X.; Mai, K.-S.; Du, Z.-Y.; Wang, Y.; Yang, H.-J. Effect of dietary lipid level on growth performance, lipid deposition, hepatic lipogenesis in juvenile cobia (Rachycentron canadum). Aquaculture 2005, 249, 439–447. [Google Scholar] [CrossRef]
- Ghanawi, J.; Roy, L.; Davis, D.A.; Saoud, I.P. Effects of dietary lipid levels on growth performance of marbled spinefoot rabbitfish Siganus rivulatus. Aquaculture 2011, 310, 395–400. [Google Scholar] [CrossRef]
- Vergara, J.M.; López, C.G.; Robaina, L.; Caballero, M.J.; Montero, D.; Izquierdo, M.S.; Aksnes, A. Growth, feed utilization and body lipid content of gilthead seabream (Sparus aurata) fed increasing lipid levels and fish meals of different quality. Aquaculture 1999, 179, 35–44. [Google Scholar] [CrossRef]
- Jiang, Y.D.; Wang, J.T.; Han, T.; Li, X.Y.; Hu, S.X. Effect of dietary lipid level on growth performance, feed utilization and body composition by juvenile red spotted grouper (Epinephelus akaara). Aquac. Int. 2015, 23, 99–110. [Google Scholar] [CrossRef]
- Song, L.P.; An, L.; Zhu, Y.A.; Li, X.; Wang, A.Y. Effects of Dietary Lipids on Growth and Feed Utilization of Jade Perch. J. World Aquac. Soc. 2009, 40, 266–273. [Google Scholar] [CrossRef]
- Borges, P.; Casal, S.; Conceição, L.; Dias, J.; Oliveira, B.; Valente, L.M.P. Dietary lipid level affects growth performance and nutrient utilisation of Senegalese sole (Solea senegalensis) juveniles. Br. J. Nutr. 2009, 102, 1007–1014. [Google Scholar] [CrossRef]
- Lu, K.L.; Xu, W.N.; Liu, W.B.; Wang, L.N.; Zhang, C.N.; Li, X.F. Association of mitochondrial dysfunction with oxidative stress and immune suppression in Blunt Snout Bream Megalobrama amblycephala fed a high-fat diet. J. Aquat. Anim. Health 2014, 26, 100–112. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Hou, C.; Gao, Y.; Wang, Y. Physiological and molecular changes in large yellow croaker (Pseudosciaena crocea R.) with high-fat diet-induced fatty liver disease. Aquac. Res. 2015, 46, 272–282. [Google Scholar] [CrossRef]
- Jia, H.; Liu, N.; Zhang, Y.; Wang, C.; Yang, Y.; Wu, Z. 3-Acetyldeoxynivalenol induces cell death through endoplasmic reticulum stress in mouse liver. Environ. Pollut. 2021, 286, 117238. [Google Scholar] [CrossRef] [PubMed]
- Astuti, S.D.; Febriastri, A.R.; Mukti, A.T.; Yaqubi, A.K.; Susilo, Y.; Syahrom, A. Photobiomodulation effect of infra-red laser on the level of gonad maturity in the Simese Catfish (Pangasianodon hypophthalmus). Heliyon 2023, 9, e23253. [Google Scholar] [CrossRef] [PubMed]
- Abulikemu, A.; Zhao, X.; Xu, H.; Li, Y.; Ma, R.; Yao, Q.; Wang, J.; Sun, Z.; Li, Y.; Guo, C. Silica nanoparticles aggravated the metabolic associated fatty liver disease through disturbed amino acid and lipid metabolisms-mediated oxidative stress. Redox Biol. 2023, 59, 102569. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, W.; Yang, Z.; Kong, Q.; Liu, P.; Liao, H.; Cui, Z.; Tang, H. Dietary Lactobacillus plantarum can alleviate high starch diet-induced liver lipid deposition, tissue damage and oxidative stress in largemouth bass (Micropterus salmoides). Aquac. Rep. 2024, 35, 101955. [Google Scholar] [CrossRef]
- Zhang, J.K.; Zhou, X.L.; Wang, X.Q.; Zhang, J.X.; Yang, M.L.; Liu, Y.P.; Cao, J.X.; Cheng, G.G. Que Zui tea ameliorates hepatic lipid accumulation and oxidative stress in high fat diet induced nonalcoholic fatty liver disease. Food Res. Int. 2022, 156, 111196. [Google Scholar] [CrossRef]
- Fernández-Sánchez, A.; Madrigal-Santillán, E.; Bautista, M.; Esquivel-Soto, J.; Morales-González, A.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sánchez-Rivera, G.; Valadez-Vega, C.; Morales-González, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Vasudevan, D.M. Alcohol-induced oxidative stress. Life Sci. 2007, 81, 177–187. [Google Scholar] [CrossRef]
- Xu, Y.; Li, Y.; Li, J.; Chen, W. Ethyl carbamate triggers ferroptosis in liver through inhibiting GSH synthesis and suppressing Nrf2 activation. Redox Biol. 2022, 53, 102349. [Google Scholar] [CrossRef]
- Ma, X.; Bi, Q.; Kong, Y.; Xu, H.; Liang, M.; Mai, K.; Zhang, Y. Dietary lipid levels affected antioxidative status, inflammation response, apoptosis and microbial community in the intestine of juvenile turbot (Scophthalmus maximus L.). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2022, 264, 111118. [Google Scholar] [CrossRef]
- Xu, H.; Han, T.; Li, X.; Wang, J.; Zheng, P.; Yin, F.; Wang, C. Effects of dietary lipid levels on survival, growth performance, and antioxidant ability of the early juvenile Scylla paramamosain. Aquaculture 2020, 528, 735559. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, J.; Gao, Q.; Shan, X.; Wang, J.; Lv, Z. Study on the attenuated effect of Ginkgolide B on ferroptosis in high fat diet induced nonalcoholic fatty liver disease. Toxicology 2020, 445, 152599. [Google Scholar] [CrossRef]
- Xu, S.; Xiaojing, L.; Xinyue, S.; Wei, C.; Honggui, L.; Shiwen, X. Pig lung fibrosis is active in the subacute CdCl2 exposure model and exerts cumulative toxicity through the M1/M2 imbalance. Ecotoxicol. Environ. Saf. 2021, 225, 112757. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, W.; Gladstone, S.; Ng, W.-K.; Zhang, J.; Shao, Q. Effects of isoenergetic diets with varying protein and lipid levels on the growth, feed utilization, metabolic enzymes activities, antioxidative status and serum biochemical parameters of black sea bream (Acanthopagrus schlegelii). Aquaculture 2019, 513, 734397. [Google Scholar] [CrossRef]
- Li, X.; Wang, T.X.; Huang, X.; Li, Y.; Sun, T.; Zang, S.; Guan, K.L.; Xiong, Y.; Liu, J.; Yuan, H.X. Targeting ferroptosis alleviates methionine-choline deficient (MCD)-diet induced NASH by suppressing liver lipotoxicity. Liver Int. 2020, 40, 1378–1394. [Google Scholar] [CrossRef]
- Wei, S.; Qiu, T.; Wang, N.; Yao, X.; Jiang, L.; Jia, X.; Tao, Y.; Zhang, J.; Zhu, Y.; Yang, G.; et al. Ferroptosis mediated by the interaction between Mfn2 and IREα promotes arsenic-induced nonalcoholic steatohepatitis. Environ. Res. 2020, 188, 109824. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.J.; Zhang, G.F.; Zheng, J.Y.; Sun, J.H.; Ding, S.B. Targeting Mitochondrial ROS-Mediated Ferroptosis by Quercetin Alleviates High-Fat Diet-Induced Hepatic Lipotoxicity. Front. Pharmacol. 2022, 13, 876550. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Cho, S.S.; Ki, S.H. Emerging roles of ferroptosis in liver pathophysiology. Pharmacal Res. 2020, 43, 985–996. [Google Scholar] [CrossRef]
- Buzzetti, E.; Petta, S.; Manuguerra, R.; Luong, T.V.; Cabibi, D.; Corradini, E.; Craxì, A.; Pinzani, M.; Tsochatzis, E.; Pietrangelo, A. Evaluating the association of serum ferritin and hepatic iron with disease severity in non-alcoholic fatty liver disease. Liver Int. 2019, 39, 1325–1334. [Google Scholar] [CrossRef]
- Crawford, D.H.G.; Ross, D.G.F.; Jaskowski, L.A.; Burke, L.J.; Britton, L.J.; Musgrave, N.; Briskey, D.; Rishi, G.; Bridle, K.R.; Subramaniam, V.N. Iron depletion attenuates steatosis in a mouse model of non-alcoholic fatty liver disease: Role of iron-dependent pathways. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2021, 1867, 166142. [Google Scholar] [CrossRef]
- Dongiovanni, P.; Lanti, C.; Gatti, S.; Rametta, R.; Recalcati, S.; Maggioni, M.; Fracanzani, A.L.; Riso, P.; Cairo, G.; Fargion, S.; et al. High fat diet subverts hepatocellular iron uptake determining dysmetabolic iron overload. PLoS ONE 2015, 10, e0116855. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Kang, R.; Tang, D. Signaling pathways and defense mechanisms of ferroptosis. FEBS J. 2022, 289, 7038–7050. [Google Scholar] [CrossRef]
- Jia, M.; Zhang, H.; Qin, Q.; Hou, Y.; Zhang, X.; Chen, D.; Zhang, H.; Chen, Y. Ferroptosis as a new therapeutic opportunity for nonviral liver disease. Eur. J. Pharmacol. 2021, 908, 174319. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Ou, Z.; Xie, M.; Kang, R.; Fan, Y.; Niu, X.; Wang, H.; Cao, L.; Tang, D. HSPB1 as a novel regulator of ferroptotic cancer cell death. Oncogene 2015, 34, 5617–5625. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yan, Y.; Han, Z.; Zheng, Y.; Wang, X.; Zhang, M.; Li, H.; Xu, J.; Chen, X.; Ding, Z.; et al. Comparative effects of dietary soybean oil and fish oil on the growth performance, fatty acid composition and lipid metabolic signaling of grass carp, Ctenopharyngodon idella. Aquac. Rep. 2022, 22, 101002. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, Z.; Liang, Z.; Xie, Y.; Su, J.; Luo, Q.; Zhu, J.; Liu, Q.; Han, T.; Wang, A. Effects of dietary fish oil replacement by soybean oil and l-carnitine supplementation on growth performance, fatty acid composition, lipid metabolism and liver health of juvenile largemouth bass, Micropterus salmoides. Aquaculture 2020, 516, 734596. [Google Scholar] [CrossRef]
- Kagan, V.E.; Mao, G.; Qu, F.; Angeli, J.P.F.; Doll, S.; Croix, C.S.; Dar, H.H.; Liu, B.; Tyurin, V.A.; Ritov, V.B.; et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 2017, 13, 81–90. [Google Scholar] [CrossRef]
- Doll, S.; Freitas, F.P.; Shah, R.; Aldrovandi, M.; da Silva, M.C.; Ingold, I.; Goya Grocin, A.; da Silva, T.N.X.; Panzilius, E.; Scheel, C.H.; et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature 2019, 575, 693–698. [Google Scholar] [CrossRef]
- Hansen, J.Ø.; Berge, G.M.; Hillestad, M.; Krogdahl, Å.; Galloway, T.F.; Holm, H.; Holm, J.; Ruyter, B. Apparent digestion and apparent retention of lipid and fatty acids in Atlantic cod (Gadus morhua) fed increasing dietary lipid levels. Aquaculture 2008, 284, 159–166. [Google Scholar] [CrossRef]
- Dixon, S.J.; Winter, G.E.; Musavi, L.S.; Lee, E.D.; Snijder, B.; Rebsamen, M.; Superti-Furga, G.; Stockwell, B.R. Human Haploid Cell Genetics Reveals Roles for Lipid Metabolism Genes in Nonapoptotic Cell Death. ACS Chem. Biol. 2015, 10, 1604–1609. [Google Scholar] [CrossRef]
- Brown, C.W.; Amante, J.J.; Goel, H.L.; Mercurio, A.M. The α6β4 integrin promotes resistance to ferroptosis. J. Cell Biol. 2017, 216, 4287–4297. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Minikes, A.M.; Gao, M.; Bian, H.; Li, Y.; Stockwell, B.R.; Chen, Z.-N.; Jiang, X. Intercellular interaction dictates cancer cell ferroptosis via NF2-YAP signalling. Nature 2019, 572, 402. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Kim, K.J.; Gaschler, M.M.; Patel, M.; Shchepinov, M.S.; Stockwell, B.R. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc. Natl. Acad. Sci. USA 2016, 113, E4966–E4975. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Maher, P.; Schubert, D. A role for 12-lipoxygenase in nerve cell death caused by glutathione depletion. Neuron 1997, 19, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Lei, G.; Zhang, Y.; Koppula, P.; Liu, X.; Zhang, J.; Lin, S.H.; Ajani, J.A.; Xiao, Q.; Liao, Z.; Wang, H.; et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020, 30, 146–162. [Google Scholar] [CrossRef]
- Yang, M.; Lu, Z.; Li, F.; Shi, F.; Zhan, F.; Zhao, L.; Li, Y.; Li, J.; Lin, L.; Qin, Z. Escherichia coli induced ferroptosis in red blood cells of grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol. 2021, 112, 159–167. [Google Scholar] [CrossRef]


| Ingredients (g kg−1) 1 | Dietary Lipid Levels (%) | |||||
|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 14 | 16 | 18 | |
| Soybean oil | 0 | 20 | 60 | 80 | 100 | 120 |
| Microcrystalline cellulose | 127 | 107 | 67 | 47 | 27 | 7 |
| Fishmeal | 430 | 430 | 430 | 430 | 430 | 430 |
| Chicken powder | 70 | 70 | 70 | 70 | 70 | 70 |
| Pork powder | 90 | 90 | 90 | 90 | 90 | 90 |
| Gluten | 70 | 70 | 70 | 70 | 70 | 70 |
| Fermented soybean meal | 50 | 50 | 50 | 50 | 50 | 50 |
| Wheat meal | 50 | 50 | 50 | 50 | 50 | 50 |
| Cassava starch | 50 | 50 | 50 | 50 | 50 | 50 |
| Lysine | 2 | 2 | 2 | 2 | 2 | 2 |
| DL-Methionine | 2 | 2 | 2 | 2 | 2 | 2 |
| CaH2PO4 | 10 | 10 | 10 | 10 | 10 | 10 |
| Phospholipid oil | 20 | 20 | 20 | 20 | 20 | 20 |
| Choline chloride | 4 | 4 | 4 | 4 | 4 | 4 |
| Vitamin premix 2 | 15 | 15 | 15 | 15 | 15 | 15 |
| Mineral premix 3 | 10 | 10 | 10 | 10 | 10 | 10 |
| Nutrients content (%) 4 | ||||||
| Crude protein | 47.15 | 47.22 | 47.25 | 47.24 | 47.64 | 47.50 |
| Crude lipid | 6.45 | 8.58 | 12.46 | 14.56 | 16.37 | 18.24 |
| Ash | 16.49 | 16.37 | 16.43 | 16.90 | 16.24 | 16.55 |
| Moisture | 7.46 | 7.31 | 7.39 | 7.55 | 7.46 | 7.36 |
| Gross energy (MJ kg−1) | 16.05 | 16.89 | 18.41 | 19.23 | 20.02 | 20.72 |
| Name 1 | Sequences | Tm (°C) 2 | Accession Number | E (%) 3 |
|---|---|---|---|---|
| ACSL4-QF | GATCTGCACTCACCCCGACA | 61.4 | XM_038699899 | 92.3 |
| ACSL4-QR | GCTCTGGACTCAAATGCACCT | |||
| LPCAT3-QF | CAGCCCTTCTGGTATCGTTG | 63.3 | XM_038711111 | 99.3 |
| LPCAT3-QR | ATACACCCTCCGCTATAACCC | |||
| LOX12-QF | ATGGTGCATACCTGACACCTC | 61.4 | XM_038731340 | 92.6 |
| LOX12-QR | TCCCTCACTTGGCCTTTCTTG | |||
| TF-QF TF-QR | GGGCAACAATCCCCAAACT TCATCCACCAGACACTGAAAGG | 61.4 | XM_038718037 | 94.4 |
| TFR-QF TFR-QR | CTTCCTGTCGCCCTATGAGTC GTCTGCCTTAGGGTTGTTGGT | 64.5 | XM_038718573 | 100.4 |
| GPX4-QF | GTTTACGCATCCTTGCCTTCC | 59.0 | XM_038716292 | 97.7 |
| GPX4-QR | GCTCTTTCAGCCACTTCCACAA | |||
| SLC7A11-QF SLC7A11-QR | GGGGCTACAGATCACACGAG ACACTACAGCCCCTTTGACC | 57 | XM_038699722 | 95.8 |
| β-actin-QF | CCCCATCCACCATGAAGA | 55.7 | XM_038695351 | 93.6 |
| β-actin-QR | CCTGCTTGCTGATCCACAT | |||
| 18S-QF | TGAATACCGCAGCTAGGAATAATG | 59.0 | MH_018569.1 | 99.5 |
| 18S-QR | CCTCCGACTTTCGTTCTTGATT |
| Items | Dietary Lipid Levels | |||||
|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 14 | 16 | 18 | |
| IBW 2 | 14.39 ± 0.13 | 14.31 ± 0.07 | 14.38 ± 0.03 | 14.37 ± 0.02 | 14.31 ± 0.16 | 14.40 ± 0.07 |
| FBW | 82.86 ± 1.00 bc | 86.28 ± 3.80 b | 92.93 ± 3.05 a | 91.33 ± 0.42 a | 81.16 ± 1.11 bc | 79.24 ± 2.07 c |
| SR | 98.33 ± 1.70 | 98.67 ± 1.25 | 99.67 ± 0.47 | 99.33 ± 0.47 | 99.33 ± 0.47 | 99.00 ± 1.41 |
| WG | 475.91 ± 10.55 cd | 502.87 ± 27.71 bc | 546.10 ± 19.73 a | 535.48 ± 3.50 ab | 467.28 ± 3.42 cd | 450.45 ± 14.78 d |
| SGR | 2.27 ± 0.02 cd | 2.33 ± 0.06 bc | 2.42 ± 0.04 a | 2.40 ± 0.01 ab | 2.25 ± 0.01 d | 2.21 ± 0.03 d |
| FI | 61.54 ± 2.13 bc | 63.76 ± 5.18 ab | 67.55 ± 2.21 a | 67.20 ± 1.31 a | 59.53 ± 0.42 bc | 58.55 ± 2.17 c |
| FE | 111.31.24 ± 1.85 b | 112.98 ± 1.93 ab | 116.27 ± 2.71 a | 114.53 ± 1.49 ab | 112.30 ± 1.83 ab | 110.78 ± 2.98 b |
| FCR | 89.85 ± 1.51 a | 88.53 ± 1.50 ab | 86.04 ± 2.02 b | 87.32 ± 1.14 ab | 89.06 ± 1.44 ab | 90.31 ± 2.46 a |
| PER | 2.40 ± 0.04 | 2.37 ± 0.01 | 2.33 ± 0.03 | 2.34 ± 0.04 | 2.36 ± 0.04 | 2.36 ± 0.02 |
| CF | 2.20 ± 0.04 | 2.17 ± 0.03 | 2.19 ± 0.06 | 2.18 ± 0.03 | 2.20 ± 0.03 | 2.14 ± 0.06 |
| VSI | 7.92 ± 0.13 d | 8.37 ± 0.12 c | 8.74 ± 0.17 bc | 8.91 ± 0.20 b | 9.42 ± 0.09 a | 9.74 ± 0.29 a |
| HSI | 2.82 ± 0.04 b | 2.68 ± 0.07 b | 2.99 ± 0.05 b | 2.93 ± 0.42 b | 3.45 ± 0.13 a | 3.64 ± 0.22 a |
| Items | Dietary Lipid Levels | |||||
|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 14 | 16 | 18 | |
| Moisture | 70.76 ± 0.52 | 70.61 ± 0.24 | 70.69 ± 0.19 | 70.26 ± 0.37 | 70.92 ± 0.46 | 70.93 ± 0.24 |
| Protein | 17.28 ± 0.37 d | 18.41 ± 0.15 b | 19.42 ± 0.33 a | 18.93 ± 0.66 a | 18.00 ± 0.48 bc | 17.85 ± 0.13 c |
| Lipid | 6.88 ± 0.41 d | 7.56 ± 0.57 c | 8.17 ± 0.20 b | 8.65 ± 0.29 ab | 8.57 ± 0.48 ab | 8.94 ± 0.19 a |
| Ash | 4.19 ± 0.12 a | 4.00 ± 0.04 b | 3.67 ± 0.06 cd | 3.72 ± 0.10 c | 3.59 ± 0.11 d | 3.57 ± 0.05 d |
| Items | Dietary Lipid Levels | |||||
|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 14 | 16 | 18 | |
| Plasma 2 | ||||||
| ALT | 10.32 ±2.57 b | 12.14 ± 3.97 b | 14.19 ± 2.68 b | 14.20 ± 1.52 b | 20.11 ± 3.45 a | 21.34 ± 0.51 a |
| AST | 33.53 ± 1.88 c | 37.14 ± 2.26 bc | 37.58 ± 1.74 bc | 38.87 ± 1.10 b | 41.51 ± 3.44 ab | 43.97 ± 2.49 a |
| Liver | ||||||
| TG | 0.157 ± 0.012 b | 0.158 ± 0.008 b | 0.191 ± 0.005 b | 0.192 ± 0.032 b | 0.256 ± 0.030 a | 0.292 ± 0.013 a |
| TC | 0.020 ± 0.006 d | 0.028 ± 0.008 cd | 0.040 ± 0.014 bc | 0.054 ± 0.010 ab | 0.055 ± 0.008 ab | 0.070 ± 0.006 a |
| Items | Dietary Lipid Levels | |||||
|---|---|---|---|---|---|---|
| 6 | 8 | 12 | 14 | 16 | 18 | |
| MDA 2 | 0.95 ± 0.02 b | 0.75 ± 0.08 c | 0.76 ± 0.09 c | 0.86 ± 0.06 bc | 1.27 ± 0.09 a | 1.28 ± 0.14 a |
| PC | 7.50 ± 1.20 ab | 6.44 ± 0.70 b | 4.90 ± 0.86 c | 4.75 ± 0.51 c | 7.55 ± 0.47 ab | 8.23 ± 0.81 a |
| T-SOD | 160.99 ± 11.87 abc | 166.64 ± 5.93 ab | 165.96 ± 5.83 ab | 172.76 ± 6.26 a | 154.29 ± 11.03 bc | 147.04 ± 7.27 c |
| CAT | 9.85 ± 0.33 c | 11.33 ± 0.21 b | 12.35 ± 0.43 a | 12.07 ± 0.43 a | 11.25 ± 0.28 b | 10.86 ± 0.11 b |
| ASA | 433.08 ± 13.78 d | 441.95 ± 12.53 d | 728.31 ± 19.85 b | 824.39 ± 73.38 a | 730.97 ± 15.50 b | 627.67 ± 11.02 c |
| AHR | 122.04 ± 4.50 d | 130.49 ± 5.34 cd | 141.91 ± 2.52 b | 167.96 ± 6.45 a | 134.15 ± 4.82 bc | 93.58 ± 1.93 e |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, L.; Zhang, Y.; Cao, Q.; Shan, H.; Zong, J.; Feng, L.; Jiang, W.; Wu, P.; Zhao, J.; Liu, H.; et al. Hepatic Oxidative Stress and Cell Death Influenced by Dietary Lipid Levels in a Fresh Teleost. Antioxidants 2024, 13, 808. https://doi.org/10.3390/antiox13070808
He L, Zhang Y, Cao Q, Shan H, Zong J, Feng L, Jiang W, Wu P, Zhao J, Liu H, et al. Hepatic Oxidative Stress and Cell Death Influenced by Dietary Lipid Levels in a Fresh Teleost. Antioxidants. 2024; 13(7):808. https://doi.org/10.3390/antiox13070808
Chicago/Turabian StyleHe, Lingjie, Yupeng Zhang, Quanquan Cao, Hongying Shan, Jiali Zong, Lin Feng, Weidan Jiang, Pei Wu, Juan Zhao, Haifeng Liu, and et al. 2024. "Hepatic Oxidative Stress and Cell Death Influenced by Dietary Lipid Levels in a Fresh Teleost" Antioxidants 13, no. 7: 808. https://doi.org/10.3390/antiox13070808
APA StyleHe, L., Zhang, Y., Cao, Q., Shan, H., Zong, J., Feng, L., Jiang, W., Wu, P., Zhao, J., Liu, H., & Jiang, J. (2024). Hepatic Oxidative Stress and Cell Death Influenced by Dietary Lipid Levels in a Fresh Teleost. Antioxidants, 13(7), 808. https://doi.org/10.3390/antiox13070808










