Abstract
Mass vaccination against COVID-19 is necessary to control the pandemic. COVID-19 vaccines are now recommended during pregnancy to prevent the disease. A systematic review of the literature in the electronic databases PubMed and EMBASE was performed and we aimed to investigate the attitude of documents towards COVID-19 vaccination and the prognostic factors of vaccination hesitation. A meta-analysis was also conducted to estimate the overall percentage of pregnant women who were willing to be vaccinated or had been vaccinated against COVID-19. A total of 18 studies were included in the review and meta-analysis. The acceptance rate of vaccination against COVID-19 among pregnant women ranged from 17.6% to 84.5%. The pooled proportion of acceptance of vaccination against COVID-19 in pregnant women was 0.53 (95% CI: 0.44–0.61). Predictors of acceptance of COVID-19 vaccination were older age, White race, occupational status, higher level of education, comorbidities, third trimester of pregnancy, influenza vaccination, knowledge about COVID-19, and confidence that vaccines for COVID-19 are safe and effective. The prevalence of COVID-19 vaccination in pregnant women is low. Targeted information campaigns are needed to increase vaccine education in this population.
1. Introduction
Pregnancy is considered a state of relative immunosuppression with a decrease in cellular immunity and possible susceptibility to infections. Also, there are changes in hormone levels, such as human chorionic gonadotropin (hCG), progesterone, and cortisol. In addition, the increase in uterus size causes the diaphragm to increase by 4 cm, widening the transverse diameter of the chest by 2 cm and affecting lung capacity. Furthermore, the immaturity of the immune system of fetuses and newborns makes them more vulnerable to infections. Therefore, pregnant women and neonates could be considered high-risk groups for infection during the current pandemic [1].
Although COVID-19 infection increases the risk of severe morbidity and mortality in pregnancy, pregnant women were not included in the initial vaccine trials which aimed to explore the efficacy and safety of vaccines against this novel virus. This has resulted in a lack of data on the safety of vaccination in pregnant women. The Centers for Disease Control and Prevention (CDC) collected reports through the National Notifiable Diseases Surveillance System (NNDSS) between 22 January and 3 October 2020, and a total of 1,300,938 women of reproductive age tested positive for SARS-CoV-2 [2]. Data on pregnancy status were available for 461,825 (35.5%) women, of whom 6.6% were pregnant. Among 461,825 women of reproductive age (15–44 years), 409,462 (88.7%) were symptomatic. Among all symptomatic women, 23,434 (5.7%) were pregnant. After adjustment for age, ethnicity/nationality, and underlying medical conditions, pregnant women were significantly more likely to be admitted to an Intensive Care Unit (ICU) [adjusted risk ratio (aRR) = 3.0; 95% confidence interval (CI) = 2.6–3.4], to need invasive ventilation (aRR = 2.9; 95% CI = 2.2–3.8), to receive extracorporeal membrane oxygenation (ECMO) (aRR = 2.4; 95% CI = 1.5–4.0) and to die (aRR = 1.7; 95% CI = 1.2–2.4), compared to non-pregnant women [2].
Unvaccinated pregnant women with a symptomatic COVID-19 infection are at high risk for preterm delivery, admission to an Intensive Care Unit (ICU), and invasive ventilation [3]. Also, unvaccinated women with symptomatic COVID-19 infection during pregnancy are at higher risk of death than non-pregnant women with a symptomatic COVID-19 infection [2]. In addition, unvaccinated pregnant women have a higher risk of hospitalization for COVID-19 than vaccinated pregnant women [4]. Anti-SARS-CoV-2 immunoglobulins confer immunity to neonates, and vaccines do not cause adverse effects associated with them [5,6].
Engjom et al. [7] studied the characteristics of 214 pregnant women with COVID-19 in five Nordic countries from 1 March to 30 June 2020. Out of a total of 214, 56 had to be hospitalized due to COVID-19 disease. Artymuk et al. [8] assessed the incidence of COVID-19 in pregnant women in the Far Eastern Federal District and the Siberian Federal District over 10 months. During the first year of the SARS-CoV-2 pandemic, 8485 cases of COVID-19 were reported among pregnant women in the Far Eastern Federal District and the Siberian Federal District, representing 5.9% of registered pregnant women and 1.71% of the total affected population. The morbidity rate in pregnant women was 3.02 times higher than in the general population: 5933.2 versus 1960.8 per 100,000 of the population. Pregnant women seem to be associated with greater susceptibility to transmission, presenting more severe forms of the disease or high risk for pregnancy complications [9]. COVID-19 is a risk factor for increased maternal and perinatal morbidity, possibly due to higher preterm birth rates in infected mothers [1].
Thus, several organizations worldwide now recommend vaccination against COVID-19 in pregnancy and women trying to get pregnant, or who may become pregnant in the future, to prevent severe maternal morbidity and adverse birth outcomes. The safety and efficacy of COVID-19 vaccines during pregnancy is a particular concern affecting the decision to vaccinate this vulnerable group [10]. Globally, significant reluctance was observed among pregnant women to accept vaccination against COVID-19. This is likely due to the limited evidence on the safety of vaccines in pregnancy at the start of the pandemic and the conflicting and changing advice given to pregnant women as the pandemic progresses [11]. Vaccination against COVID-19 appears to be highly effective in pregnancy for preventing COVID-19, without increasing the risk of adverse pregnancy outcomes. The risks of COVID-19 outweigh the rare risks of vaccination in pregnancy, and pregnant women should be encouraged to continue vaccination, even in the first trimester. However, the lack of high-quality (i.e., low risk of bias) studies uniformly reporting clinically relevant outcomes and other vaccine types used in low- to middle-income countries is problematic [10]. Regarding the vaccination strategy against COVID-19, the World Health Organization (WHO) recommends vaccination for pregnant women only when the benefits of vaccination outweigh the potential risks, but current recommendations for pregnant women to receive vaccinations against COVID-19 differ from country to country [12].
Therefore, this review aimed to examine pregnant women’s attitudes towards vaccination against COVID-19 and to study predictors of vaccination hesitancy.
2. Materials and Methods
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Figure 1: Flow Chart).
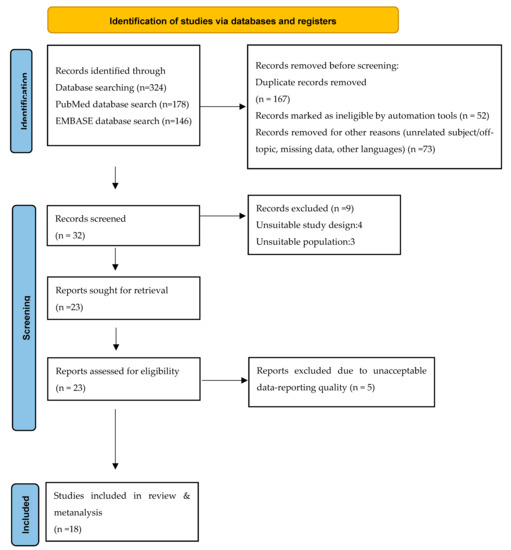
Figure 1.
PRISMA flow diagram. Visualization of the process involving identification of records from databases, screening of records, assessing reports for eligibility, inclusion of eligible studies, and exclusion of non-eligible reports. The number of records or reports in each step of the process is shown in brackets.
To search for studies related to the purpose of the review, the method followed was based on the PICOST procedure, where P represents population, I intervention, C comparison, O outcome, S the type of studies (Study Design), and T the year of publication (Timeframe). In more detail, the selection/exclusion criteria of the publications for inclusion in this review were as follows:
- Population: Studies involved pregnant women or women who had recently given birth.
- Intervention: Investigating pregnant women’s attitudes towards vaccination against COVID-19 through a questionnaire. Studies investigating pregnant women’s attitudes towards vaccines for diseases other than COVID-19 were rejected.
- Comparison: Studies compared demographic, social, and occupational characteristics of pregnant women with their attitudes toward vaccination against COVID-19.
- Outcome: The studied outcomes of the publications/studies were the rate of acceptance of the vaccine against COVID-19 and the factors related to hesitancy or refusal of vaccination.
- Study Design: The studies were primary quantitative studies. Secondary studies (reviews, meta-analyses), case studies, and qualitative studies were not included in the review.
- Timeframe: The studies must have been published by 31 December 2021 and written in English or Greek. Studies published in a language other than English and Greek were not included. Furthermore, studies in which the full text could not be located were also excluded.
A systematic review of the international literature was performed in the electronic databases PubMed/MEDLINE and EMBASE during the period of July–September 2022. The keywords used to search for studies were: (pregnancy) AND (COVID-19) AND (vaccination) AND (attitude OR hesitancy OR decline OR acceptance OR concern).
A literature search was conducted in the MEDLINE (PubMed) and EMBASE databases following the predefined search strategy (Figure 1). The electronic search yielded 178 abstracts from PubMed and 146 abstracts from EMBASE. Both sets of records were downloaded from each database to the bibliographic software package EndNote X7 (Clarivate Analytics, Philadelphia, PA, USA) and merged into one core database to remove duplicate records and facilitate retrieval of relevant articles. All potentially relevant reports identified after searching other nonelectronic sources were entered into EndNote manually. After the elimination of duplicates, 157 studies were identified. Additionally, seventy-three new studies were removed for missing data, being off topic, and unknown languages (Figure 1).
The titles and abstracts of all identified studies were examined by two reviewers independently (A.S. and V.K.) according to predefined inclusion and exclusion criteria. Review authors were not blinded to the names of the authors, institutions, journal of publication, or results of the studies. All records identified by the searches were primarily checked on the basis of the title and abstract. Records that were obviously irrelevant were excluded, and the full text of all remaining records was obtained. If the relevant information for meeting the inclusion criteria was not available from the abstract and/or title, we obtained the full text of the report. In this way, 32 studies were selected for full-text reading and were assessed independently by the same two reviewers. Articles that did not meet all inclusion criteria after the full-text assessment (N = 9) were excluded from further examination. Figure 1 depicts and summarizes the complete study selection process.
The publications resulting from the literature review were screened as to their title, and those whose title was not compatible with the purpose of the systematic review were rejected. The abstracts of the remaining studies were then read, and those that did not meet the criteria for inclusion in the review were discarded. The remaining studies were searched as full texts and those that did not provide the necessary information regarding the topic and purpose of the review were rejected.
The data selected to be extracted are the following:
- General characteristics of the study (name of 1st author, year of publication, country of conduct, type of study)
- Sample characteristics (sample size, average age of pregnant women, gestational week)
- Outcome (vaccination acceptance/refusal rate)
- Significant findings (finding, or non-statistically significant association of vaccination acceptance/hesitancy with demographic, social, and occupational characteristics)
Statistical Analysis
The effect size used was the acceptance rate of vaccination against COVID-19 (Proportion) in pregnant women. The meta-analysis was performed using the metaprop command and the results are presented in the form of a table and plot of findings of the individual studies and overall analysis (forest plot). Study heterogeneity was checked with the I2 test for all studies that provided results. When this was statistically significant (>50%), a random effect model was applied using the DerSimonian and Laird (D+L) method, while a fixed effect model was used otherwise. Publication bias refers to the non-representative publication of research reports, which is not due to the quality of the respective study. The present study did not check for publication bias, either by drawing a funnel plot or with the statistical criterion of Egger’s test, as according to studies on proportional data these tests are not sufficiently adjusted. The assumption that positive results are published more often does not necessarily apply to proportional studies, as there is no clear definition nor consensus on what constitutes a positive result in a proportional meta-analysis. Therefore, it is not recommended in these proportional meta-analyses to conduct publication bias checks [13]. Furthermore, no meta-analysis was performed on factors influencing the vaccination acceptance rate among pregnant women, as the data were highly heterogeneous. The analysis was performed with the statistical package STATA 13. STATA 13 is a statistical software package created in 1985 by StataCorp for data analysis, data management, and graphics. It is a general-purpose statistical software package used in research, especially in the fields of biomedicine and epidemiology.
3. Results
From the search through international literature, 324 studies were found. Out of these, 292 were rejected after reading the title, nine after reading the abstract, and five after reading the full text. Ultimately, 18 studies were included in the present review (Figure 1: Flow Chart).
In Table 1 the characteristics of the included studies are demonstrated (author, year, research type, methodology, and main findings). Table 2 shows the acceptance rate of vaccination against COVID-19 among pregnant women according to the studies included in the review and related factors of hesitancy (i.e., maternal age, gestational age, educational level, marital status, race/ethnicity, and presence of comorbidities). As shown in the table, the acceptance rate of vaccination against COVID-19 among pregnant women ranged from 17.6% [14,15] to 84.5% [15].

Table 1.
Characteristics of studies included in review & metanalysis.

Table 2.
Acceptance rate of vaccination against COVID-19 among pregnant women and related factors of hesitancy.
Of the six studies that investigated whether age is a predictor of COVID-19 vaccination acceptance among pregnant women, only two found age to be a predictor [15,18]. More specifically, in the study by Ghamri et al. [18], it was found that for every year of increase in the age of a pregnant woman, the probability of accepting vaccination increases by 2% [OR: 1.02; 95% CI: 1.018–1.036). Moreover, in Levy’s et al. study, pregnant women aged 18–24 years were found to be 0.35 times less likely to be vaccinated against COVID-19 than women aged 31–35 years [20].
Out of the nine studies that investigated whether education level is a predictor of acceptance of COVID-19 vaccination among pregnant women, seven studies showed that education level is a predictor of acceptance [16,18,20,24,28,30,31]. More specifically, in the study by Battarbee et al. [16], it was found that pregnant women who had a graduate school degree were 2.4 times more likely (OR: 2.4; 95% CI: 1.3–4.7) to accept being vaccinated against COVID-19 compared to pregnant women who were high school graduates. In a study by Ghamri et al. [18], pregnant women with primary education were 2.8 times more likely (OR: 2.853; 95% CI: 1.207–6.745) to accept vaccination against COVID-19 than illiterate pregnant women. In a study by Levy et al. [20], female high school graduates were 0.14 times less likely (OR: 0.14; 95% CI: 0.07–0.25) to accept being vaccinated against COVID-19 in relation to pregnant women who had a Bachelor’s degree. In a study by Riad et al. [24], pregnant women with Master’s degree and a Ph.D. were 5.99 times more likely (OR: 5.99; 95% CI: 1.12–32.16) to accept getting a COVID-19 vaccine dose in relation to pregnant women who had received only basic education. Also, in a study by Skjefte et al. [28], in comparison with pregnant women who were high school graduates, pregnant women with lower than secondary education were 0.76 times less likely to be vaccinated (OR: 0.76; 95% CI: 0.58–0.99), while pregnant women who held university degrees (OR: 1.25; 95% CI: 1.03–1.53) and had completed postgraduate studies (either MSc or Ph.D. (OR: 1.26; 95% CI: 1.10–1.44) were 1.25 and 1.26 times more likely to get a COVID 19 vaccine, respectively.
Only the study by Riad et al. [24] showed that pregnant women who had a partner were 5.43 times more likely to accept being vaccinated against COVID-19 than those who were single.
Six out of the ten studies showed that occupational status is a predictor of acceptance of COVID-19 vaccination among pregnant women [16,18,20,26,28,30]. Employed pregnant women were found to be more likely to accept being vaccinated against COVID-19 than pregnant women who were unemployed/in charge of housekeeping [16,18,26,32]. Also, full-time employed women were more likely to get vaccinated against COVID-19 than part-time employed pregnant women [20,30]. Finally, pregnant physicians [28] and health professionals [26] were more likely to be vaccinated against COVID-19.
Three out of the five studies showed that ethnicity is a predictor of COVID-19 vaccination acceptance among pregnant individuals [16,20,29]. Relative to pregnant white women, those who were black [16,20,29] and Hispanic [16,20,29] were less likely to be vaccinated against COVID-19.
Only two [18,28] out of seven studies showed that the presence of underlying diseases in pregnant women increases with statistical significance the likelihood of accepting vaccination against COVID-19 among this population.
Two studies found that pregnant women in their third trimester were more likely to accept vaccination against COVID-19 than pregnant women in early pregnancy [24,31].
Table 3 shows the results of the studies included in the review regarding influenza vaccination as a factor of COVID-19 vaccination acceptance in pregnancy. Six studies [16,18,20,28,29,30] stated that pregnant women who had been vaccinated against influenza either during their pregnancy or in the previous year were more likely to accept the vaccination against COVID-19 than pregnant women who had never received an influenza vaccination.

Table 3.
Influenza vaccination as a factor of COVID-19 vaccination acceptance among pregnant women.
Table 4 shows the results of the studies included in the review regarding the existence of a positive COVID-19 test as a factor of vaccination acceptance against COVID-19 among pregnant women, their knowledge regarding COVID-19, and their confidence that vaccines are safe and effective. One study found that pregnant women who had tested positive for COVID-19 were more likely to be vaccinated against COVID-19 than those who never had a positive COVID-19 test [18].

Table 4.
Positive COVID-19 test, knowledge of COVID-19, and confidence that vaccines are safe and effective as factors in acceptance of vaccination against COVID-19 among pregnant women.
Likewise, two studies found that a higher level of knowledge about COVID-19 or the vaccine was a predictor of acceptance of vaccination against COVID-19 among pregnant women [29,30]. Finally, the perception that the COVID-19 vaccine is safe [23,28] and effective [16,28] was found to be a predictor of vaccination acceptance against COVID-19 in the participating pregnant population.
Quantitative Synthesis of Studies–Meta-Analysis
The 18 studies included in the review were used to investigate the pooled acceptance proportion of vaccination against COVID-19 in pregnant women. Since a statistically significant heterogeneity among the studies was detected (I2 = 99.29%, p < 0.001), an analysis was performed using the random effect model. This analysis revealed that the pooled proportion of vaccination uptake against COVID-19 in pregnant women was 0.53 (95% CI: 0.44–0.61). (Figure 2).
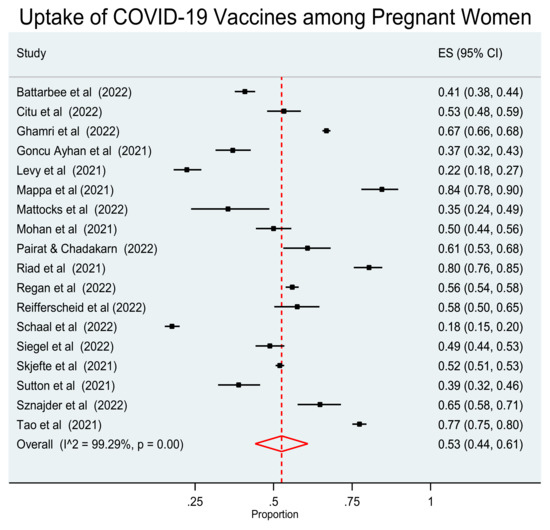
Figure 2.
Forest plot of studies reporting vaccination acceptance against COVID-19 in pregnant women. Vertical ticks and horizontal lines show the mean effect and 95% confidences interval for each study. The red diamond at the bottom shows the cumulative effect with 95% confidence intervals.
4. Discussion
The present systematic review and meta-analysis included 18 studies that met certain inclusion and exclusion criteria and aimed to estimate the acceptance of COVID-19 vaccination among pregnant women. Additionally, we intended to investigate factors associated with vaccination acceptance, as well as reasons for vaccination hesitancy.
The acceptance rate of vaccination against COVID-19 among pregnant women ranged from 17.6% to 84.5%. The pooled proportion of vaccination uptake against COVID-19 in the pregnant population was 0.53 (95% CI: 0.44–0.61). The results of our study are similar to the results of two earlier meta-analyses [33,34] and found that the global prevalence of pregnant women receiving the vaccine for COVID-19 was approximately 49–54%. However, in a meta-analysis investigating the pooled proportion of women who had been vaccinated against COVID-19, it was 29% [35].
Acceptance of COVID-19 vaccination was associated with several factors, including older age, ethnicity, occupational status, higher level of education, presence of comorbidities, advanced gestation, influenza vaccination, knowledge about COVID-19, and confidence that COVID-19 vaccines are safe and effective. More specifically, older age was found to be associated with higher acceptance of COVID vaccines [18,20]. This finding is plausible because pregnancy at advanced maternal age is known to be a risk factor for adverse outcomes, such as higher rates of NICU admission for the neonate, preterm birth, miscarriage, preeclampsia, low birth weight, worse Apgar scores, and cesarean deliveries. In addition, older age is associated with higher mortality due to COVID-19. It is probable that older pregnant women face COVID-19 with added fear, resulting in their higher acceptance of COVID-19 vaccination [33,34,35].
Moreover, in our review, the rate of COVID-19 vaccination acceptance was highest among White and Asian pregnant women and lowest among those of Black and Hispanic origin [16,20,29]. Latins and Black or African American race are associated with refusal of vaccination against COVID-19 during pregnancy [16,20,29]. Similar racial and ethnic disparities have been reported for the uptake of other recommended vaccinations during pregnancy, such as tetanus, influenza, and pertussis, with black and Hispanic women having the lowest vaccination coverage [33,34,35].
The safety and effectiveness of COVID-19 vaccines during pregnancy is a particular concern affecting vaccination uptake by this vulnerable population [10]. Regarding the vaccination strategy against COVID-19, the World Health Organization (WHO) recommends vaccination for pregnant women only when the benefits of vaccination outweigh the potential risks, but current recommendations for pregnant women vaccinations against COVID-19 differ from country to country. For example, the USA, the UK, and other European countries encourage pregnant women to get vaccinated against COVID-19, but China does not [36]. For ethical and other reasons, evidence from randomized controlled trials (RCTs) on the safety and efficacy of COVID-19 vaccines for the pregnant population is scarce. Therefore, the real-world study (RWS) on the safety and efficacy of COVID-19 vaccines for pregnant women may provide additional evidence [36].
In the recent systematic review and meta-analysis by Prasad et al. [10], which included 23 studies with 117,552 pregnant women vaccinated against COVID-19, it was found that the efficacy of mRNA vaccination against RT-PCR confirmed SARS-CoV-2 seven days after the second dose was 89.5% (95% CI = 69.0–96.4%, 18,828 vaccinated pregnant women). The risk of stillbirth was significantly lower in vaccinated pregnant women by 15% (pooled OR = 0.85; 95% CI = 0.73–0.99; 66,067 vaccinated vs. 424,624 unvaccinated). There was no evidence of a higher risk of adverse outcomes, including miscarriage, preterm birth, placental abruption, pulmonary embolism, postpartum hemorrhage, maternal death, ICU admission, lower birth weight, or NICU admission (p > 0.05 for all). Therefore, vaccination with COVID-19 mRNA in pregnancy appears to be safe and associated with a reduction in stillbirth.
Ma et al. [36] explored the safety and efficacy of vaccines against COVID-19 in pregnant women through the RWS systematic review and meta-analysis. The researchers included a total of six studies. An analysis of the studies found that vaccination prevented the pregnant population from SARS-CoV-2 infection (OR = 0.50; 95% CI = 0.35–0.79) and hospitalization related to COVID-19 (OR = 0.50; 95% CI = 0.31–0.82). mRNA vaccines could reduce the risk of infection in pregnant women (OR = 0.13; 95% CI = 0.03–0.57). Also, no adverse effects of vaccination against COVID-19 were found in pregnant, fetal, or neonatal outcomes.
A question of particular interest is whether the risk of miscarriage increases after vaccination against COVID-19 in early pregnancy. This is particularly important, as up to 40% of pregnancies are unintended and may remain unrecognized until the 4th–8th week of gestation, and thus inadvertent vaccination in early pregnancy is likely to be common [37]. The mRNA vaccine elicits both antibody and cellular immune responses. Given the importance of T-cell suppression in placental development and fetal well-being, concern has been raised that the vaccine may increase the risk of miscarriage [38]. Social media is full of reports that have fueled this concern, and many pregnant women have cited this fear as their main reason for hesitating to vaccinate. Data from the meta-analysis by Prasad et al. [10] do not support such concerns.
Also, vaccination against COVID-19 is associated with a lower incidence of stillbirth. COVID-19 in pregnancy is associated with an increased risk of stillbirth, particularly during the period of Delta variant dominance [10]. A population-based study in Scotland found that after SARS-CoV-2 infection in unvaccinated pregnant women, the perinatal mortality rate was 22.6 per 1000 births, whereas, in contrast, no vaccinated pregnant women with primary infection suffered a perinatal death [39].
In Brazil, there has been a report of maternal death after vaccination with Astra Zeneca, and other cases are also reported in the published literature. With the Oxford/Astra Zeneca virus vaccine, there is a rare risk of vaccine-induced immune thrombotic thrombocytopenia [40]. This has prompted some countries (UK, Canada, and USA) to exclude this vaccine in people under 40 years of age. These very rare complications should not discourage the scientific community, health workers, and policymakers from disseminating information about the clear benefits of vaccination against COVID-19 in pregnancy to both mother and newborn [10].
Similarly, cases of myocarditis have been reported after vaccination with mRNA, estimated to occur at two cases per million women and 10 cases per million men aged 18–40 years. Such reactions are typically mild and rapidly self-limiting and occur more often in association with COVID-19 infection. However, no cases of myocarditis have been reported after vaccination in pregnant women [10].
Globally, significant reluctance was observed among pregnant women to accept vaccination against COVID-19. This is likely due to the limited evidence on the safety of vaccines in pregnancy at the start of the pandemic and the conflicting and changing advice given to pregnant women as the pandemic progresses [11]. Vaccination against COVID-19 appears to be highly effective in pregnancy for preventing infection, without increasing the risk of adverse pregnancy outcomes. The risks of COVID-19 outweigh the rare risks of vaccination in pregnancy, and pregnant women should be encouraged to continue vaccination, even in their first trimester. However, the lack of high-quality (i.e., low risk of bias) studies uniformly reporting clinically relevant outcomes, as well as reporting other vaccine types used in low- to middle-income countries, is rather challenging.
Confidence in COVID-19 vaccines and fewer concerns about the safety and side effects of COVID-19 vaccines are predictors of acceptance of COVID-19 vaccination [16,28]. Similar factors, such as confidence in the safety and effectiveness of COVID-19 vaccines, confidence in the information received about vaccination against COVID-19, confidence in childhood vaccines, and influenza vaccination in the previous year, are associated with a higher rate of intention of pregnant women to receive a COVID-19 vaccine [16,18,20,28,29,30]. In general, high levels of information and knowledge about COVID-19 vaccines reduce fear and have a significant effect on a pregnant woman’s decision to get vaccinated against COVID-19 [33,34,35]. A recent systematic review and meta-analysis found that vaccination for COVID-19 protects pregnant women from SARS-CoV-2 infection and COVID-19-related hospitalization and has no adverse effects on pregnant women, fetuses, or neonates. Since COVID-19 vaccines have been shown to be safe and effective in pregnant women, policymakers should use this information to improve confidence in COVID-19 vaccines and reduce hesitancy [33,34,35].
The present review and meta-analysis is characterized by some limitations. Initially, publications were searched only in two databases, and studies published only in English and Greek were included. Another limitation of the review is the heterogeneity of the studies in terms of their design and the study population. Finally, the quality of the publications was not thoroughly assessed.
Previous studies have stated COVID-19 vaccine intention among pregnant women; our study is among the first to compare vaccination behavior in the light of vaccine acceptance and hesitancy attitudes toward vaccination in this specific population.
5. Conclusions
Pregnant individuals are at increased risk of severe COVID-19 infection. Although vaccination is suggested, COVID-19 vaccination rates are lower among pregnant women compared to the non-pregnant population.
The present analysis shows that approximately one in two pregnant women is willing to be vaccinated against COVID-19. Numerous factors, including advanced maternal age, White race, employment, educational level, underlying diseases, influenza vaccination, knowledge about COVID-19 infection, and confidence that vaccines for COVID-19 are safe and effective, are associated with COVID-19 vaccination acceptance.
COVID-19 vaccine acceptance during pregnancy is not widespread and public health intervention will be vital to continue to increase vaccine coverage. Efforts should be made in order to encourage uptake and facilitate access to vaccination. These actions may contribute to higher uptake in pregnant populations of lower socioeconomic status and all those associated with lower vaccine acceptance.
The safety of the COVID-19 vaccine is a principal concern for pregnant people when deciding to receive the vaccine. As pregnant and lactating women were not included in the COVID anti-viral and vaccine trials, it is imperative that safety information is communicated to this high-risk population as it occurs, in order to advance empirical evidence.
Information delivery methods about the benefits of vaccination during pregnancy should be improved to increase the likelihood that someone will act on it, as the elevated risk of COVID-19 disease in pregnancy is either poorly understood or underrated.
Author Contributions
Conceptualization, A.S.; methodology, V.E.K. and A.S.; software, V.E.K.; validation, V.E.K., C.T. and A.S.; formal analysis, A.S.; investigation, V.E.K.; resources, C.N.; data curation, V.E.K. and C.T.; writing—original draft preparation, V.E.K. and A.S.; writing—review and editing, A.S.; visualization, A.S.; supervision, A.S.; funding acquisition, A.L. All authors have read and agreed to the published version of the manuscript.
Funding
The APC was funded by the Special Account for Research Grants, University of West Attica, Athens, Greece.
Institutional Review Board Statement
Not Applicable. It wasn’t required to seek institutional ethics approval before commencing this systematic review as publicly accessible documents are used as evidence. All datasets on which the conclusions of this paper rely are deposited in openly available repositories available to readers.
Informed Consent Statement
Data from previously published studies in which informed consent was obtained by primary investigators were retrieved and analyzed in this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Michailidou, D.; Stavridou, A.; Panagouli, E.D.; Sergentanis, T.N.; Psaltopoulou, T.; Bacopoulou, F.; Baltag, V.; Greydanus, D.E.; Mastorakos, G.; Chrousos, G.P.; et al. The impact of COVID-19 during pregnancy on maternal and neonatal outcomes: A systematic review. EMBnet. J. 2021, 26, e969. [Google Scholar] [CrossRef] [PubMed]
- Zambrano, L.D.; Ellington, S.; Strid, P.; Galang, R.R.; Oduyebo, T.; Tong, V.T.; Woodworth, K.R.; Nahabedian, J.F., III; Azziz-Baumgartner, E.; Gilboa, S.M.; et al. Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status-United States. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 1641–1647. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; von Dadelszen, P.; Kalafat, E.; Sebghati, M.; Ladhani, S.; Ugwumadu, A.; Draycott, T.; O’Brien, P.; Magee, L. Change in obstetric attendance and activities during the COVID-19 pandemic. Lancet Infect. Dis. 2021, 21, e115. [Google Scholar] [CrossRef] [PubMed]
- Iacobucci, G. Covid-19 and pregnancy: Vaccine hesitancy and how to overcome it. BMJ 2021, 375, n2862. [Google Scholar] [CrossRef] [PubMed]
- Falsaperla, R.; Leone, G.; Familiari, M.; Ruggieri, M. COVID-19 vaccination in pregnant and lactating women: A systematic review. Expert Rev. Vaccines 2021, 20, 1619–1628. [Google Scholar] [CrossRef]
- Garg, I.; Shekhar, R.; Sheikh, A.B.; Pal, S. COVID-19 Vaccine in Pregnant and Lactating Women: A Review of Existing Evidence and Practice Guidelines. Infect. Dis. Rep. 2021, 13, 685–699. [Google Scholar] [CrossRef]
- Engjom, H.; Aabakke, A.J.M.; Klungsøyr, K.; Svanvik, T.; Äyräs, O.; Jonasdottir, E.; Thurn, L.; Jones, E.; Pettersson, K.; Nyfløt, L.T.; et al. COVID-19 in pregnancy—Characteristics and outcomes of pregnant women admitted to hospital because of SARS-CoV-2 infection in the Nordic countries. Acta Obstet. Gynecol. Scand. 2021, 100, 1611–1619. [Google Scholar] [CrossRef]
- Artymuk, N.V.; Belokrinitskaya, T.E.; Filippov, O.S.; Frolova, N.I.; Surina, M.N. Perinatal outcomes in pregnant women with COVID-19 in Siberia and the Russian Far East. J. Matern.-Fetal Neonatal Med. 2021, 35, 5427–5430. [Google Scholar] [CrossRef]
- Ayala-Ramírez, P.; González, M.; Escudero, C.; Quintero-Arciniegas, L.; Giachini, F.R.; Alves de Freitas, R.; Damiano, A.E.; García-Robles, R. Severe Acute Respiratory Syndrome Coronavirus 2 Infection in Pregnancy. A Non-systematic Review of Clinical Presentation, Potential Effects of Physiological Adaptations in Pregnancy, and Placental Vascular Alterations. Front. Physiol. 2022, 13, 384. [Google Scholar] [CrossRef]
- Prasad, S.; Kalafat, E.; Blakeway, H.; Townsend, R.; O’Brien, P.; Morris, E.; Draycott, T.; Thangaratinam, S.; Le Doare, K.; Ladhani, S.; et al. Systematic review and meta-analysis of the effectiveness and perinatal outcomes of COVID-19 vaccination in pregnancy. Nat. Commun. 2022, 13, 2414. [Google Scholar] [CrossRef]
- RCOG. COVID-19 Vaccines, Pregnancy and Breastfeeding FAQs. Royal College of Obstetricians & Gynaecologists. 2021. Available online: https://www.rcog.org.uk/guidance/coronavirus-covid-19-pregnancy-and-women-s-health/vaccination/covid-19-vaccines-pregnancy-and-breastfeeding-faqs/ (accessed on 5 March 2022).
- World Health Organization (WHO). Update on WHO Interim Recommendations on COVID-19 Vaccination of Pregnant and Lactating Women. 2021. Available online: https://www.who.int/publications/m/item/update-on-who-interim-recommendations-on-covid-19-vaccination-of-pregnant-and-lactating-women (accessed on 24 August 2022).
- Barker, P.; Hartley, D.; Beck, A.F.; Oliver, G.; Sampath, B.; Roderick, T.; Miff, S. Rethinking Herd Immunity: Managing the Covid-19 Pandemic in a Dynamic Biological and Behavioral Environment. NEJM Catalyst. 2021, 10, 5. [Google Scholar]
- Schaal, N.K.; Zöllkau, J.; Hepp, P.; Fehm, T.; Hagenbeck, C. Pregnant and breastfeeding women’s attitudes and fears regarding the COVID-19 vaccination. Arch. Gynecol. Obstet. 2022, 306, 365–372. [Google Scholar] [CrossRef]
- Mappa, I.; Luviso, M.; Distefano, F.A.; Carbone, L.; Maruotti, G.M.; Rizzo, G. Women perception of SARS-CoV-2 vaccination during pregnancy and subsequent maternal anxiety: A prospective observational study. J. Matern.-Fetal Neonatal Med. 2021, 35, 6302–6305. [Google Scholar] [CrossRef]
- Battarbee, A.N.; Stockwell, M.S.; Varner, M.; Newes-Adeyi, G.; Daugherty, M.; Gyamfi-Bannerman, C.; Tita, A.T.; Vorwaller, K.; Vargas, C.; Subramaniam, A.; et al. Attitudes Toward COVID-19 Illness and COVID-19 Vaccination among Pregnant Women: A Cross-Sectional Multicenter Study during August-December 2020. Am. J. Perinatol. 2020, 39, 75–83. [Google Scholar] [CrossRef]
- Citu, C.; Chiriac, V.D.; Citu, I.M.; Gorun, O.M.; Burlea, B.; Bratosin, F.; Popescu, D.E.; Ratiu, A.; Buca, O.; Gorun, F. Appraisal of COVID-19 Vaccination Acceptance in the Romanian Pregnant Population. Vaccines 2022, 10, 952. [Google Scholar] [CrossRef]
- Ghamri, R.A.; Othman, S.S.; Alhiniah, M.H.; Alelyani, R.H.; Badawi, A.M.; Alshahrani, A.A. Acceptance of COVID-19 Vaccine and Associated Factors Among Pregnant Women in Saudi Arabia. Patient Prefer. Adherence 2022, 16, 861–873. [Google Scholar] [CrossRef]
- Ayhan Goncu, S.; Oluklu, D.; Atalay, A.; Menekse Beser, D.; Tanacan, A.; Moraloglu Tekin, O.; Sahin, D. COVID-19 vaccine acceptance in pregnant women. Int. J. Gynaecol. Obstet. 2021, 154, 291–296. [Google Scholar] [CrossRef]
- Levy, A.T.; Singh, S.; Riley, L.E.; Prabhu, M. Acceptance of COVID-19 vaccination in pregnancy: A survey study. Am. J. Obstet. Gynecol. Mfm 2021, 3, 100399. [Google Scholar] [CrossRef]
- Mattocks, K.M.; Kroll-Desrosiers, A.; Moore Simas, T.A.; Bastian, L.A.; Marteeny, V.; Walker, L.; Sheahan, K.; Elwy, A.R. Examining Pregnant Veterans’ Acceptance and Beliefs Regarding the COVID-19 Vaccine. J. Gen. Intern. Med. 2022, 37 (Suppl. S3), 671–678. [Google Scholar] [CrossRef]
- Mohan, S.; Reagu, S.; Lindow, S.; Alabdulla, M. COVID-19 vaccine hesitancy in perinatal women: A cross-sectional survey. J. Perinat. Med. 2021, 49, 678–685. [Google Scholar] [CrossRef]
- Pairat, K.; Phaloprakarn, C. Acceptance of COVID-19 vaccination during pregnancy among Thai pregnant women and their spouses: A prospective survey. Reprod. Health 2022, 19, 74. [Google Scholar] [CrossRef] [PubMed]
- Riad, A.; Jouzová, A.; Üstün, B.; Lagová, E.; Hruban, L.; Janků, P.; Pokorná, A.; Klugarová, J.; Koščík, M.; Klugar, M. COVID-19 Vaccine Acceptance of Pregnant and Lactating Women (PLW) in Czechia: An Analytical Cross-Sectional Study. Int. J. Environ. Res. Public Health 2021, 18, 13373. [Google Scholar] [CrossRef] [PubMed]
- Regan, A.K.; Kaur, R.; Nosek, M.; Swathi, P.A.; Gu, N.Y. COVID-19 vaccine acceptance and coverage among pregnant persons in the United States. Prev. Med. Rep. 2022, 29, 101977. [Google Scholar] [CrossRef]
- Reifferscheid, L.; Marfo, E.; Assi, A.; Dubé, E.; MacDonald, N.E.; Meyer, S.B.; Bettinger, J.A.; Driedger, S.M.; Robinson, J.; Sadarangani, M.; et al. COVID-19 vaccine uptake and intention during pregnancy in Canada. Can. J. Public Health 2022, 113, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.R.; Lumbreras-Marquez, M.I.; James, K.; McBay, B.R.; Gray, K.J.; Schantz-Dunn, J.; Diouf, K.; Goldfarb, I.T. Perceptions and Attitudes toward COVID-19 Vaccination among Pregnant and Postpartum Individuals. Am. J. Perinatol. 2022, 29, 1489–1495. [Google Scholar] [CrossRef]
- Skjefte, M.; Ngirbabul, M.; Akeju, O.; Escudero, D.; Hernandez-Diaz, S.; Wyszynski, D.F.; Wu, J.W. COVID-19 vaccine acceptance among pregnant women and mothers of young children: Results of a survey in 16 countries. Eur. J. Epidemiol. 2021, 36, 197–211. [Google Scholar] [CrossRef]
- Sutton, D.; D’Alton, M.; Zhang, Y.; Kahe, K.; Cepin, A.; Goffman, D.; Staniczenko, A.; Yates, H.; Burgansky, A.; Coletta, J.; et al. COVID-19 vaccine acceptance among pregnant, breastfeeding, and nonpregnant reproductive-aged women. Am. J. Obstet. Gynecol. MFM 2021, 3, 100403. [Google Scholar] [CrossRef]
- Sznajder, K.K.; Kjerulff, K.H.; Wang, M.; Hwang, W.; Ramirez, S.I.; Gandhi, C.K. COVID-19 vaccine acceptance and associated factors among pregnant women in Pennsylvania 2020. Prev. Med. Rep. 2022, 26, 101713. [Google Scholar] [CrossRef]
- Tao, L.; Wang, R.; Han, N.; Liu, J.; Yuan, C.; Deng, L.; Han, C.; Sun, F.; Liu, M.; Liu, J. Acceptance of a COVID-19 vaccine and associated factors among pregnant women in China: A multi-center cross-sectional study based on health belief model. Hum. Vaccines Immunother. 2021, 17, 2378–2388. [Google Scholar] [CrossRef]
- Niemi, M.E.K.; Karjalainen, J.; Liao, R.G.; Neale, B.M.; Daly, M.; Ganna, A.; Pathak, G.A.; Andrews, S.J.; Kanai, M.; Veerapen, K.; et al. Mapping the human genetic architecture of COVID-19. Nature 2021, 600, 472–477. [Google Scholar] [CrossRef]
- Carbone, L.; Di Girolamo, R.; Mappa, I.; Saccone, G.; Raffone, A.; Di Mascio, D.; De Vivo, V.; D’Antonio, F.; Guida, M.; Rizzo, G.; et al. Worldwide beliefs among pregnant women on SARS-CoV-2 vaccine: A systematic review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 268, 144–164. [Google Scholar] [CrossRef]
- Nikpour, M.; Sepidarkish, M.; Omidvar, S.; Firouzbakht, M. Global prevalence of acceptance of COVID-19 vaccines and associated factors in pregnant women: A systematic review and meta-analysis. Expert Rev. Vaccines 2022, 21, 843–851. [Google Scholar] [CrossRef]
- Galanis, P.; Vraka, I.; Siskou, O.; Konstantakopoulou, O.; Katsiroumpa, A.; Kaitelidou, D. Uptake of COVID-19 Vaccines among Pregnant Women: A Systematic Review and Meta-Analysis. Vaccines 2022, 10, 766. [Google Scholar] [CrossRef]
- Ma, Y.; Deng, J.; Liu, Q.; Du, M.; Liu, M.; Liu, J. Effectiveness and Safety of COVID-19 Vaccine among Pregnant Women in Real-World Studies: A Systematic Review and Meta-Analysis. Vaccines 2022, 10, 246. [Google Scholar] [CrossRef]
- Singh, S.; Sedgh, G.; Hussain, R. Unintended pregnancy: Worldwide levels, trends, and outcomes. Stud. Fam. Plann. 2010, 41, 241–250. [Google Scholar] [CrossRef]
- Saito, S.; Nakashima, A.; Shima, T.; Ito, M. Th1/Th2/Th17 and regulatory T-cell paradigm in pregnancy. Am. J. Reprod. Immunol. 2010, 63, 601–610. [Google Scholar] [CrossRef]
- Stock, S.J.; Carruthers, J.; Calvert, C.; Denny, C.; Donaghy, J.; Goulding, A.; Hopcroft, L.E.M.; Hopkins, L.; McLaughlin, T.; Pan, J.; et al. SARS-CoV-2 infection and COVID-19 vaccination rates in pregnant women in Scotland. Nat. Med. 2022, 28, 504–512. [Google Scholar] [CrossRef]
- Wolf, M.E.; Luz, B.; Niehaus, L.; Bhogal, P.; Bäzner, H.; Henkes, H. Thrombocytopenia and Intracranial Venous Sinus Thrombosis after “COVID-19 Vaccine AstraZeneca” Exposure. J. Clin. Med. 2021, 10, 1599. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).