Abstract
Background: This systematic review and meta-analysis summarizes the evidence for the association between endometriosis and adverse pregnancy outcome, including gestational hypertension, pre-eclampsia, low birth weight, and small for gestational age, preterm birth, placenta previa, placental abruption, cesarean section, stillbirth, postpartum hemorrhage, spontaneous hemoperitoneum in pregnancy, and spontaneous bowel perforation in pregnancy. Methods: We performed the literature review in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA), by searches in PubMed and EMBASE, until 1 November 2020 (PROSPERO ID CRD42020213999). We included peer-reviewed observational cohort studies and case-control studies and scored them according to the Newcastle–Ottawa Scale, to assess the risk of bias and confounding. Results: 39 studies were included. Women with endometriosis had an increased risk of gestational hypertension, pre-eclampsia, preterm birth, placenta previa, placental abruption, cesarean section, and stillbirth, compared to women without endometriosis. These results remained unchanged in sub-analyses, including studies on spontaneous pregnancies only. Spontaneous hemoperitoneum in pregnancy and bowel perforation seemed to be associated with endometriosis; however, the studies were few and did not meet the inclusion criteria. Conclusions: The literature shows that endometriosis is associated with an increased risk of gestational hypertension, pre-eclampsia, preterm birth, placenta previa, placental abruption, cesarean section, and stillbirth.
1. Introduction
Endometriosis affects about 10% of women of reproductive age [1]. It is a chronic gynecological disease whereby estrogen-dependent endometrial fragments are located on organs other than the uterus, with the development of inflammation, adhesions, and pain [1], with the ovaries and the posterior cul-de-sac (pouch of Douglas) most often affected [2]. Several pathogenic mechanisms are possible, but the most supported is retrograde bleeding through the fallopian tubes, due to dysperistaltic uterine contractions [3]. Peritoneal levels of inflammatory mediators, such as cytokines, chemokines, and prostaglandins, increase, leading to a state of chronic inflammation in women with endometriosis [4].
Many pathogenic consequences of endometriosis have been suggested to play a role in the decreased fertility seen in these women, e.g., structural changes in the junctional zone, chronic inflammation, mechanical defects, and ovarian dysfunction [5,6]. Furthermore, abnormal remodeling of the spiral arteries in the junctional zone may cause defective deep placentation, thus increasing the risk of adverse pregnancy outcome [5].
Due to infertility, many women with endometriosis use assisted reproductive technology (ART) to conceive [7], which may increase the risk of adverse pregnancy outcome per se [8].
In recent years, many studies have investigated the association between endometriosis and pregnancy complications; however, the results have been ambiguous [9,10,11,12]. This systematic review and meta-analysis set out to evaluate the association between endometriosis and adverse pregnancy outcomes, including gestational hypertension, pre-eclampsia, low birth weight, small for gestational age (SGA), preterm birth, antepartum hemorrhage, placenta previa, placental abruption, cesarean section, stillbirth, and postpartum hemorrhage (PPH). We also reviewed studies on endometriosis related to spontaneous hemoperitoneum in pregnancy (SHiP) and spontaneous bowel perforation in pregnancy.
2. Materials and Methods
We performed the study in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [13] (Supplementary Materials S1). The protocol was published to PROSPERO in November 2020. ID for protocol: CRD42020213999.
2.1. Information Sources
We systematically searched PubMed and EMBASE from its inception until 1 November 2020, for all studies on endometriosis and adverse pregnancy outcome. The reference lists of the included studies were screened for additional literature. Prior to submission of the review (10 January 2021), an additional search was performed to ensure that no newer studies had been published since the latest literature search.
A priori, we decided to focus on the following outcomes: gestational hypertension, pre-eclampsia, low birth weight, SGA, preterm birth, placenta previa, placental abruption, cesarean section, stillbirth, PPH, SHiP, and spontaneous bowel perforation in pregnancy.
We used “MeSH” (PubMed) and “Emtree” (EMBASE) terms as well as free text words. The following MeSH-terms were used: endometriosis, adenomyosis, pre-eclampsia, pregnancy-induced hypertension, infant low birth weight, small for gestational age, premature birth, gestational age, placenta previa, abruptio placentae, hemoperitoneum, intestinal perforation, cesarean section, stillbirth, and postpartum hemorrhage. Please see the exact search strings in Supplementary Materials S2.
2.2. Study Selection and Data Extraction
Studies had to investigate the association between endometriosis and at least one of the predefined outcomes.
We included cohort studies and case-control studies. Thus, case reports, case series, commentaries, letters, editorials, and conference abstracts were excluded. No restrictions by language or time period were applied. However, studies in other languages than English were excluded during the full-text assessment.
Data extracted, by use of a structured extraction sheet, included information on bibliography and study design, characteristics of participants, information on confounders and intermediate factors, how endometriosis was diagnosed, how the reference group was recruited, definitions of outcomes, number and proportions per group with the specific outcome, and effect estimates, including crude or adjusted odds ratios (cOR or aOR), crude or adjusted relative risks (cRR or aRR), and 95% confidence intervals (CIs). Data extraction was performed by Kjerstine Breintoft (KB) and Regitze Pinnerup (RP) and in case of no consensus, a third author was consulted (Linn Håkonsen Arendt (LHA)). For cohort studies not providing risk estimates, we extracted information on numbers of outcomes among exposed and non-exposed.
2.3. Screening of Studies
Duplicates were removed, using EndNote (X9, Clarivate Analytics, Philadelphia, PA, U.S.) and Covidence (Veritas Health Innovation Ltd, Melbourne, Australia). Studies were screened by title and abstract by Axel Forman (AF) and KB. Any discrepancies were resolved by discussion and if in doubt a third author was consulted (LHA). Relevant studies were reviewed in full text by KB and RP, and any disagreements were resolved by discussion and by consulting a third author (LHA).
2.4. Assessment of Bias
The included studies were assessed by KB and RP, using the Newcastle–Ottawa quality assessment Scale (NOS). Consensus was reached by discussion in case of disagreement. In case of no consensus, a third author was consulted (LHA). NOS evaluates the studies based on the selection and comparability of the groups. Furthermore, the ascertainment of exposure and outcomes is assessed. A priori, based on directed Acyclic graphs (DAGs) and the existing literature, we selected the following relevant potential confounding factors: maternal age, smoking, body mass index (BMI), and socioeconomic status. These factors have been shown to be associated with endometriosis [14,15,16] and increase the risk of several adverse pregnancy outcomes [17,18,19,20]. Thus, we considered these the most important potential confounders. Adjustment for at least two of these resulted in one point, and adjustment for all resulted in two points. Studies could receive a score between zero and nine based on criteria defined a priori (Supplementary Materials S3). Adjustment, stratification, or sampling for ART did not result in a higher score. However, ART is discussed.
2.5. Meta-Analyses
Most studies estimated the association by providing odds ratios or relative risks with 95% CIs. We used aORs or aRRs if available. EpiBasic (V4.4, Svend Juul and Morten Frydenberg, Aarhus, Denmark) was used to calculate odds ratios based on available data if odds ratios or relative risks were not provided.
We used Review Manager [21] to conduct the meta-analyses. We used a random-effect inverse-variance weighted model providing a combined OR with 95% CI. Our main meta-analysis only included studies with a NOS score ≥7, as indicated in Table 1. To evaluate whether the results of the main analyses changed by including all studies regardless of study quality, a secondary analysis for each outcome was also conducted. Furthermore, to avoid the influence of ART on the associations, sub-analyses were performed including only studies investigating spontaneous conceived pregnancies in both the exposed and non-exposed groups.

Table 1.
Characteristics of the 39 included cohort studies investigating the association between endometriosis and adverse pregnancy outcome.
Furthermore, we created funnel plots, using Software for Statistics and Data Science (STATA, 16, STATACorp LLC, Texas, TX, USA) to visualize the likelihood of publication bias if the number of studies was more than ten, in accordance with the Cochrane Handbook of Systematic Reviews [22]. The cohort studies without adverse outcomes in women with or without endometriosis were excluded from the funnel plots because odds ratios could not be calculated.
2.6. Heterogeneity Assessment
We assessed the heterogeneity between studies by considering the study characteristics including study design, setting, population, and definition of pregnancy outcome. Furthermore, in the meta-analyses, we used the I2 statistics to assess the statistical heterogeneity, as recommended by the Cochrane Handbook for Systematic Reviews threshold recommendations [23]. An I2 value of 0% to 40% suggests that the heterogeneity may not be important, 30% to 60% suggests moderate heterogeneity, 50% to 90% suggests substantial heterogeneity, and 75% to 100% suggests significant heterogeneity [23].
3. Results
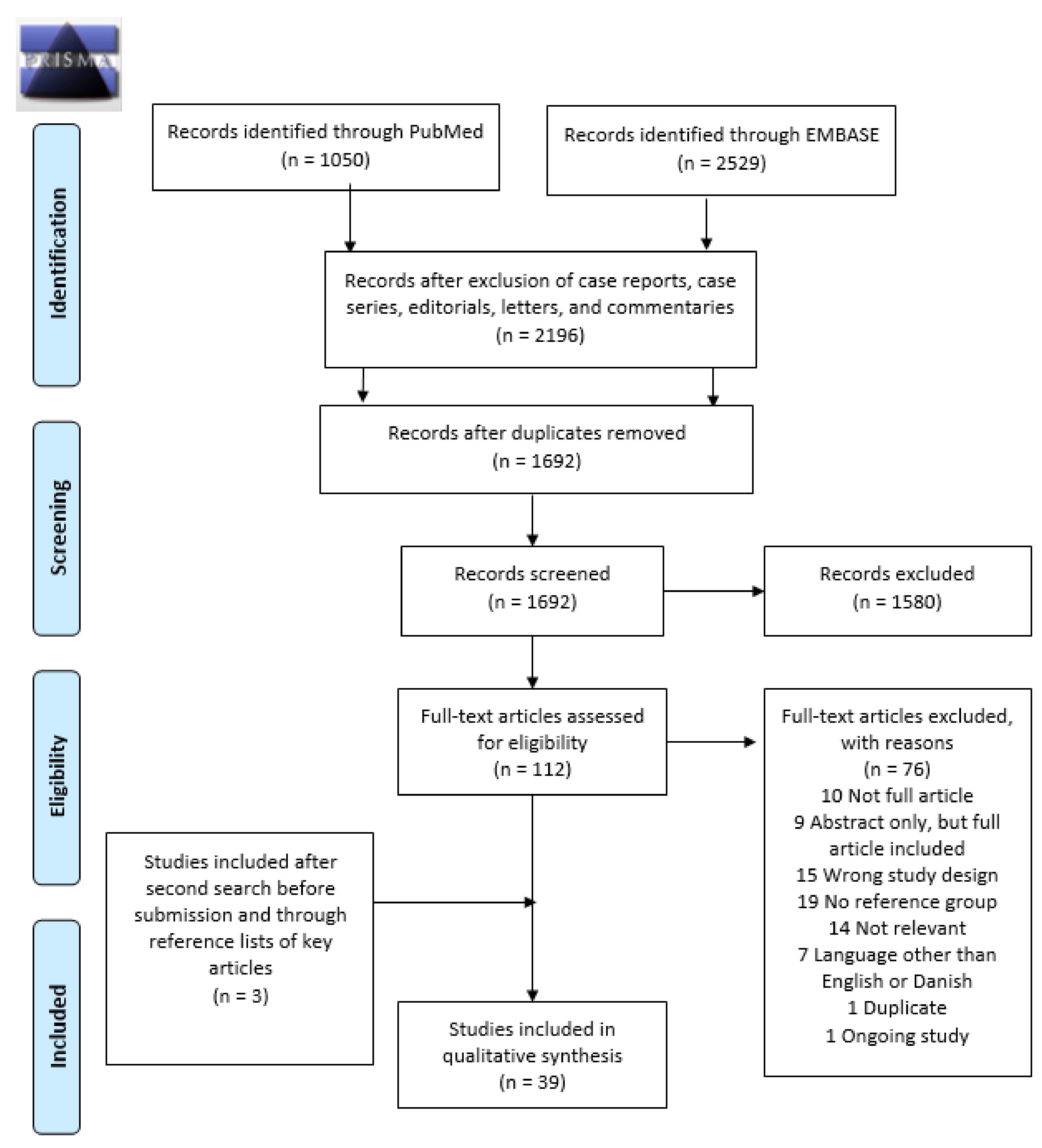
A total of 1692 records were identified by the initial search. After screening titles and abstracts, 112 papers were reviewed in full text. This revealed 36 relevant papers. By scrutiny of their references, one additional paper was added. Prior to submission, an additional literature search revealed two relevant papers. A total of 39 cohort studies and no case-control studies were included in the systematic review. Figure 1 illustrates the PRISMA flowchart of study selection for the systematic review. Furthermore, characteristics and main results of the included studies are presented in Table 1 and Table 2.
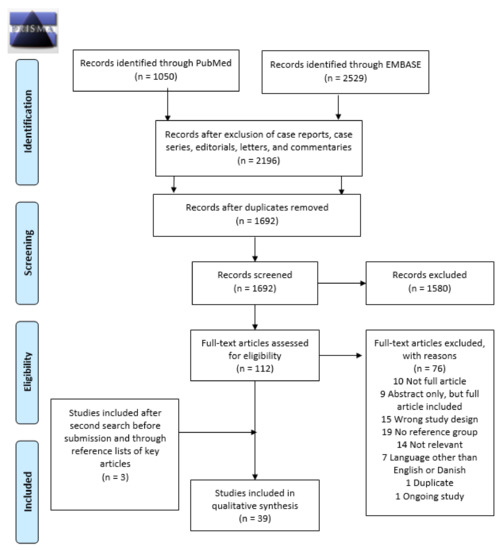
Figure 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flowchart identifying the inclusion of studies. Note: n, number.

Table 2.
Main results of the 39 included cohort studies investigating the association between endometriosis and adverse pregnancy outcome.
For the outcomes SHiP and spontaneous bowel perforation in pregnancy, only one cohort study was available. The rest of the existing literature on these outcomes comprised of reviews, case reports, or case series. Thus, it was not possible to systematically review these outcomes. However, because of their severity, the existing literature was summarized and discussed.
Fifteen studies received a NOS score ≥7 and were thus included in the main meta-analyses. The most frequent reasons for studies receiving a NOS score <7, and thus not being included in the main meta-analysis, were lack of adjustment for confounders, small sample size, comparison of pregnancies conceived by ART to spontaneous pregnancies, and inability to verify information on exposure and outcomes in medical records. The pooled ORs from the main meta-analyses were compared to the pooled ORs of the secondary analyses including all studies regardless of study quality. Six studies were eligible for the sub-analyses investigating only spontaneous pregnancies.
3.1. Heterogeneity Assessment
A priori, we considered the heterogeneity of the studies including sample size, country, timing, population, and data sources (Table 1). All studies were cohort studies.
Most studies originated from Italy, USA, Japan, or Scandinavia. Furthermore, the studies were conducted in varying time periods, from the 1970s, but with the majority published within the last five years (2015–2020). Data were mostly derived from medical records or registers. However, many of the studies also gathered information from questionnaires or did not state how information was collected.
In the main meta-analyses, the I2 statistic was used to quantify the statistical heterogeneity and this revealed a high heterogeneity between studies for most outcomes, ranging from 0% to 96%. Gestational hypertension revealed an I2 of 0%; however, only four studies were included in the meta-analysis for this outcome [23].
3.2. Hypertensive Disorders of Pregnancy
Hypertensive disorders in pregnancy are associated with an increased risk of maternal and fetal morbidity and affect around 10% of all pregnant women worldwide [63]. A severe manifestation of this condition is pre-eclampsia which is characterized by hypertension, proteinuria, and maternal organ dysfunction after 20 weeks of gestation [64].
3.2.1. Hypertensive Disorder in Pregnancy Overall
A total of 11 cohort studies investigated hypertensive disorders in pregnancy overall [25,30,34,38,44,45,49,51,53,59,60]. Five were eligible for the main meta-analysis [30,34,38,51,53], without statistically significant difference between exposed and non-exposed to endometriosis (Figure 2). The main meta-analysis showed a pooled OR of 1.20 (95% CI: 0.92–1.55). Heterogeneity was high (I2 = 91%). The secondary analysis, including all studies regardless of study quality, did not change the direction of the estimated association between endometriosis and hypertensive disorders in pregnancy overall based on the high-quality studies (Supplementary Figure S1). Sub-analysis including only spontaneous pregnancies was not conducted as only one study was eligible [45].

Figure 2.
Forest plot for endometriosis and hypertensive disorders in pregnancy overall including studies with NOS ≥7. Chen et al., (2018) [30] and Farland et al., (2019) [34] used adjusted relative risks; Hadfield et al., (2009) [38], Pan et al., (2017) [51], and Saraswat et al., (2017) [53] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S2).
3.2.2. Gestational Hypertension
We identified 14 cohort studies on the association between endometriosis and gestational hypertension [24,27,28,29,31,33,46,47,48,50,52,55,57,61], four of which were included in the main-analysis [24,27,28,57] (Figure 3). Endometriosis showed to increase the risk of gestational hypertension with a pooled OR of 1.14 (95% CI: 1.00–1.31). Heterogeneity was low (I2 = 0%). The secondary analysis including all studies showed a pooled OR of 1.00 (95% CI: 0.79–1.27) (Supplementary Figure S3a). The sub-analyses including only spontaneous pregnancies showed similar results as the main meta-analysis, but with more uncertainty, as only three studies were included (Supplementary Figure S3b) [48,52,57].

Figure 3.
Forest plot for endometriosis and gestational hypertension, including studies with NOS ≥7. Aris et al., (2014) [24] and Benaglia et al., (2016) [27] used crude odds ratios; Berlac et al., (2017) [28] and Stern et al. (2015) [57] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S4).
3.2.3. Pre-Eclampsia
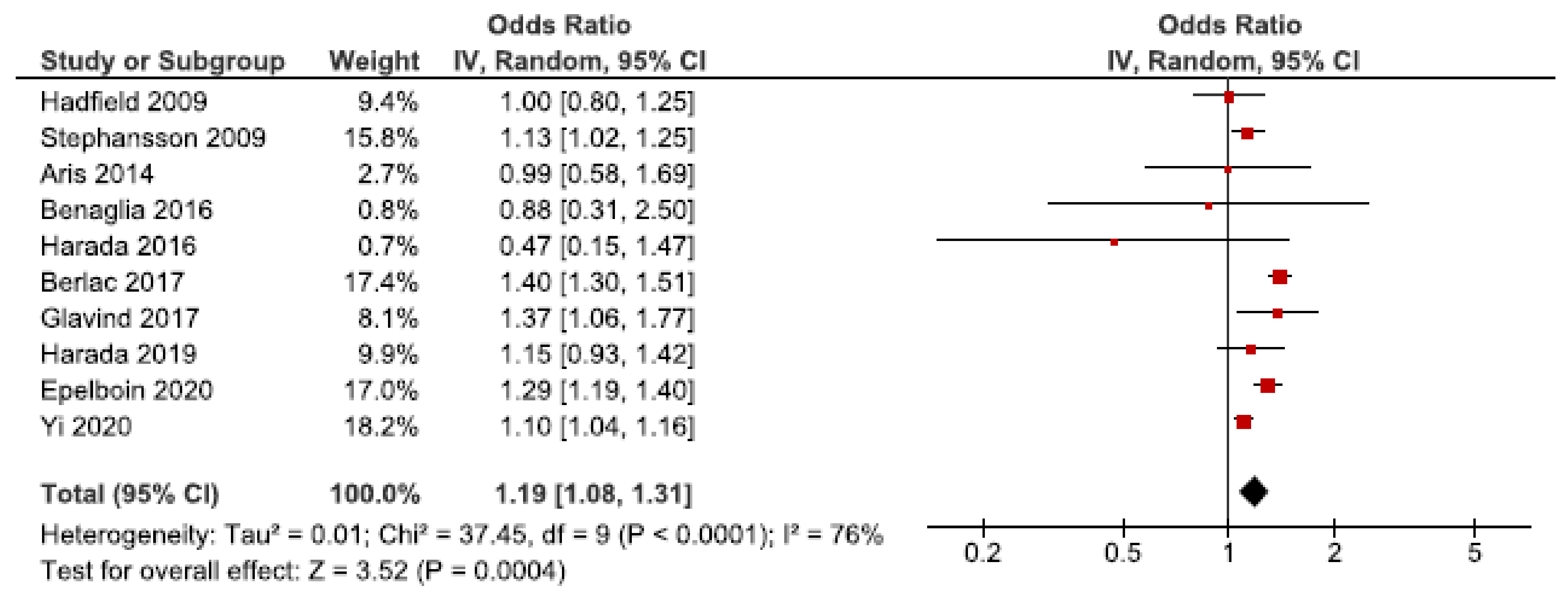
We identified 21 cohort studies regarding endometriosis and pre-eclampsia [24,26,27,28,29,31,32,37,38,39,40,41,42,43,50,52,54,55,56,61,62]. Ten of these were included in the main meta-analysis [24,27,28,32,37,38,39,40,56,62] (Figure 4). Women with endometriosis showed to be at increased risk of pre-eclampsia (OR: 1.19, 95% CI: 1.08–1.31). Heterogeneity was high (I2 = 76%). Neither the secondary analysis including all studies regardless of quality nor the sub-analysis including only spontaneous pregnancies [32,52,54] changed the direction of the association between endometriosis and pre-eclampsia (Supplementary Figure S5).
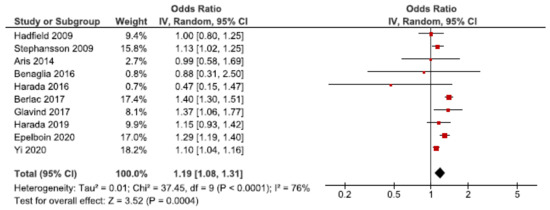
Figure 4.
Forest plots for endometriosis and pre-eclampsia including studies with NOS ≥7. Aris et al., (2014) [24], Benaglia et al., (2016) [27], Hadfield et al., (2009) [38], and Harada et al., (2019) [40] used crude odds ratios; Berlac et al., (2017) [28], Epelboin et al., (2020) [32], Glavind et al., (2017) [37], Harada et al., (2016) [39], Stephansson et al., (2009) [56], and Yi et al., (2020) [62] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S6).
3.3. Low Birth Weight
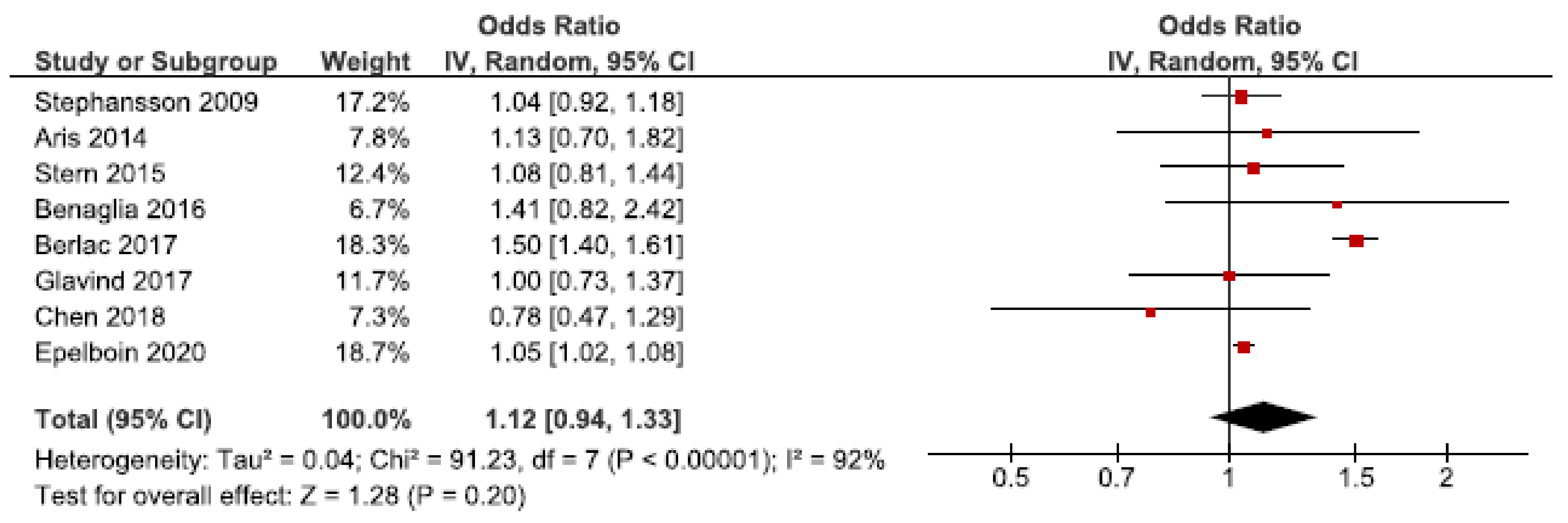
Low birth weight may occur as a result of preterm birth or SGA [65]. We identified 14 cohort studies on endometriosis and low birth weight [24,26,27,30,34,42,43,46,49,52,53,57,58,62]. Seven were eligible for the main meta-analysis [24,27,30,34,53,57,62], without reaching statistically significance (Figure 5). We found a pooled OR of 1.22 (95% CI: 0.99–1.49). Heterogeneity was high I2 = 90%. Neither the secondary analysis including all studies nor the sub-analysis including only spontaneous pregnancies [52,57] changed the direction of the main results (Supplementary Figure S7).
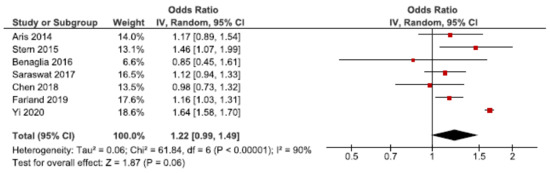
Figure 5.
Forest plot for endometriosis and low birth weight, including studies with NOS ≥7. Chen et al., 2018 [30] and Farland et al., 2019 [34] used adjusted relative risks; Aris et al., (2014) [24] and Benaglia et al., (2016) [27] used crude odds ratios; Saraswat et al., (2017) [53], Stern et al., (2015) [57], and Yi et al., (2020) [62] used adjusted odds ratios.
The funnel plot did not indicate publication bias (Supplementary Figure S8).
3.4. Small for Gestational Age
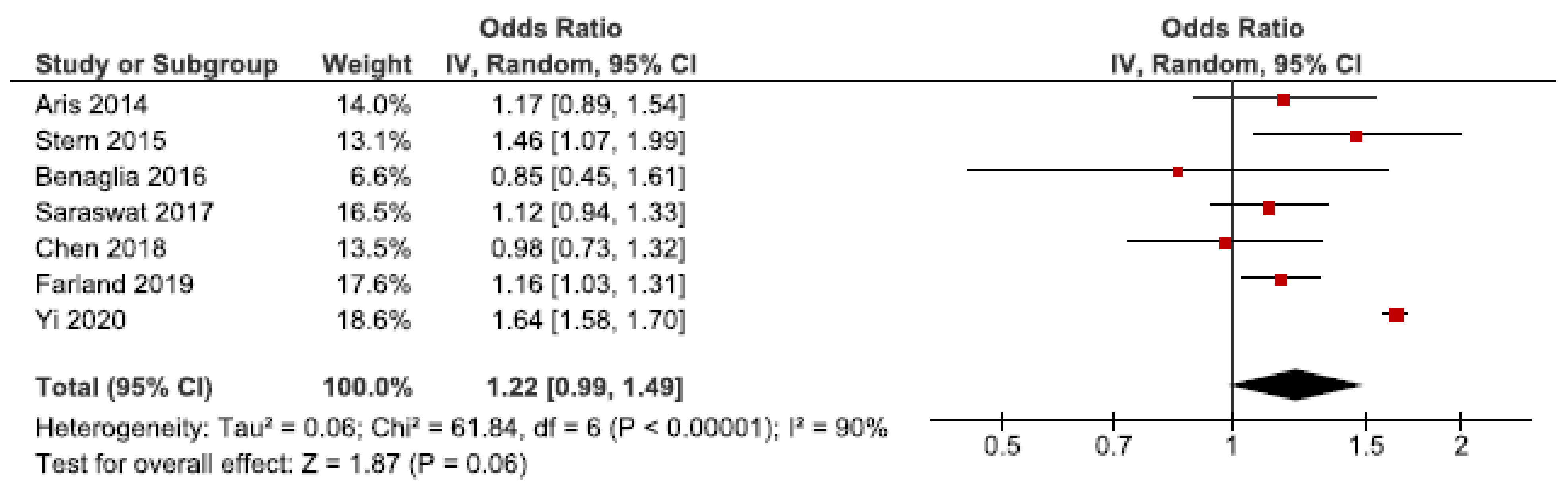
Neonatal mortality and morbidity is increased in SGA infants [66]. We identified 22 cohort studies regarding the association between endometriosis and SGA [24,26,27,28,30,31,32,33,35,36,37,42,43,45,46,47,48,49,50,54,56,57]. Eight of these were included in the main meta-analysis [24,27,28,30,32,37,56,57], without reaching statistically significance (Figure 6). The pooled OR for the association between endometriosis and SGA was 1.12 (92% CI: 0.94–1.33). Heterogeneity was high (I2 = 92%). The secondary analysis, including all studies regardless of study quality, showed a pooled OR of 1.18 (1.02–1.36) (Supplementary Figure S9a). Five studies were eligible for the sub-analysis including only spontaneous pregnancies [32,45,48,54,57]. This showed an attenuated association with an OR of 1.05 (1.02–1.08) (Supplementary Figure S9b). Thus, the risk of SGA was only increased when including all studies regardless of study quality or only including spontaneous pregnancies.
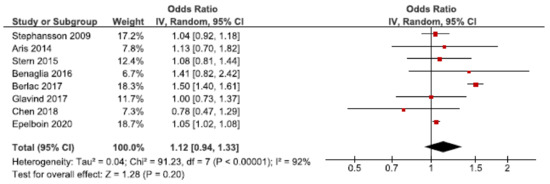
Figure 6.
Forest plot for endometriosis and SGA, including studies with NOS ≥7. Chen et al., 2018 [30] used adjusted relative risk; Aris et al., 2014 [24] and Benaglia et al., 2016 [27] used crude odds ratios; Berlac et al., 2017 [28], Epelboin et al., 2020 [32], Glavind et al., 2017 [37], Stephansson et al., 2009 [56], and Stern et al., 2015 [57] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S10).
3.5. Gestational Age at Birth and Preterm Birth
Gestational age is defined as the estimated time from the first day of the last menstrual period until birth, and preterm birth is defined as birth before 37 completed weeks of gestation [67]. The estimation was usually carried out by early pregnancy ultrasound scanning.
3.5.1. Gestational Age at Birth
A total of 13 cohorts were found on endometriosis and gestational age at birth. Seven studies found women with endometriosis to have a shorter duration of pregnancy compared to women without endometriosis [31,39,40,41,43,55,60]. Two of these received a NOS score ≥7 [39,40]. Six studies found no difference in gestational age between women with and without endometriosis [25,42,44,49,54,61]. None of these received a NOS score ≥7. Ten of these studies also investigated preterm birth. No meta-analysis or funnel plot was performed for gestational age.
3.5.2. Preterm Birth
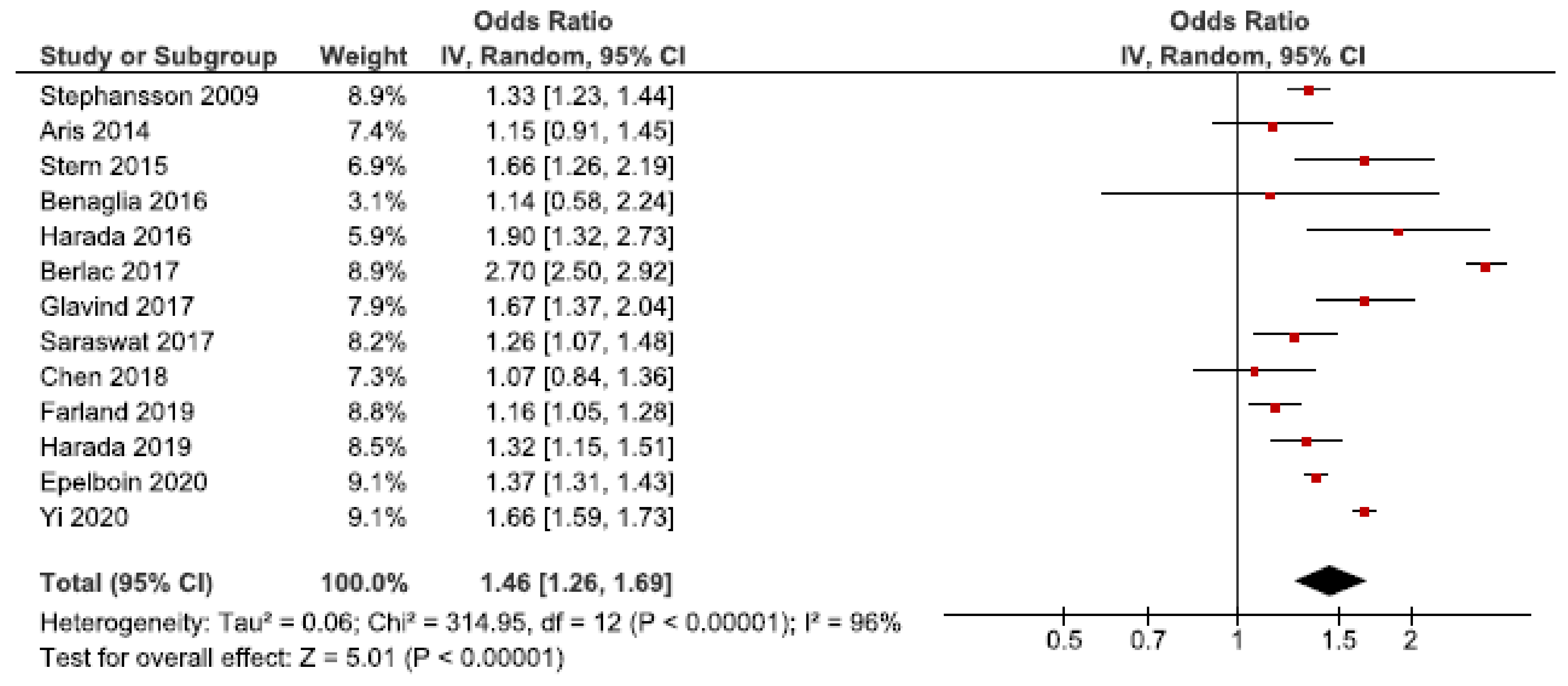
We identified 33 cohort studies on the association between endometriosis and preterm birth [24,25,26,27,28,30,31,32,33,34,35,36,37,39,40,41,42,43,44,45,46,47,48,49,50,52,53,56,57,58,59,61,62]. Thirteen of these were included in the main meta-analysis [24,27,28,30,32,34,37,39,40,53,56,57,62] (Figure 7). The pooled estimate showed an increased risk of preterm birth in women with endometriosis with an OR of 1.46 (95% CI: 1.26–1.69). Heterogeneity was high (I2 = 96%). Neither the secondary analysis including all studies regardless of quality nor the sub-analysis including only spontaneous pregnancies changed the direction of the results [32,45,48,52,57] (Supplementary Figure S11).
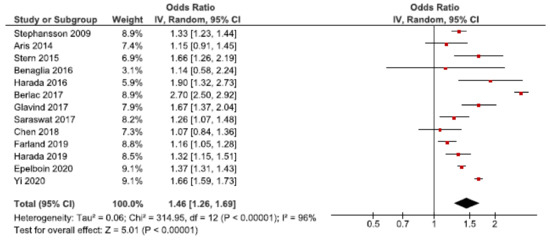
Figure 7.
Forest plot for endometriosis and preterm birth including studies with NOS ≥7. Chen et al., 2018 [30] and Farland et al., 2019 [34] used adjusted relative risks; Aris et al., 2014 [24] and Harada et al., 2016 [39] used crude odds ratios; Benaglia et al., 2016 [27], Berlac et al., 2017 [28], Epelboin et al., 2020 [32], Glavind et al., 2017 [37], Harada et al., 2019 [40], Saraswat et al., 2017 [53], Stephansson et al., 2009 [56], Stern et al., 2015 [57], and Yi et al., 2020 [62] used adjusted odds ratios.
The funnel plot indicated no publication bias (Supplementary Figure S12).
3.6. Antepartum Hemorrhage
Antepartum hemorrhage, including placenta previa and placental abruption, is one of the leading causes of maternal mortality worldwide [68].
The two large register-based studies by Stephansson et al. [56] and Berlac et al. [28] investigated all types of antepartum hemorrhage as one outcome. They both found endometriosis to be associated with antepartum hemorrhage.
3.6.1. Placenta Previa
Twenty-four cohort studies investigated the association between endometriosis and placenta previa [25,26,27,28,30,32,33,36,39,40,41,42,43,44,45,46,47,49,50,52,53,60,61,62]. Eight studies were eligible for the main meta-analysis [27,28,30,32,39,40,53,62] (Figure 8). It showed an increased risk of placenta previa in women with endometriosis with a pooled OR of 2.99 (95% CI: 2.54–3.53). Heterogeneity was high (I2 = 86%). Neither the secondary analysis including all studies regardless of quality nor the sub-analysis only including spontaneous pregnancies [32,45,52] changed the direction of the results (Supplementary Figure S13).
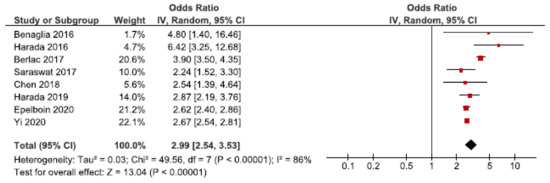
Figure 8.
Forest plot for endometriosis and placenta previa including studies with NOS ≥7. Chen et al., 2018 [30] used adjusted relative risk; Benaglia et al., 2016 [27], Berlac et al., 2017 [28], Epelboin et al., 2020 [32], Harada et al., 2016 [39], Harada et al., 2019 [40], Saraswat et al., 2017 [53], and Yi et al., 2020 [62] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S14).
3.6.2. Placental Abruption
Twenty cohort studies investigated placental abruption in endometriosis patients [26,27,28,30,32,33,39,40,42,43,44,45,46,49,50,52,53,59,61,62]. Eight studies were included in the main meta-analysis [27,28,30,32,39,40,53,62] (Figure 9). We found an increased risk of placental abruption in women with endometriosis with a pooled OR of 1.40 (95% CI: 1.12–1.76). Heterogeneity was high (I2 = 82%). Neither the secondary analysis including all studies or the sub-analysis only including spontaneous pregnancies changed the direction of the results (Supplementary Figure S15). However, in the sub-analysis the CIs were broad as two studies were included [32,45].
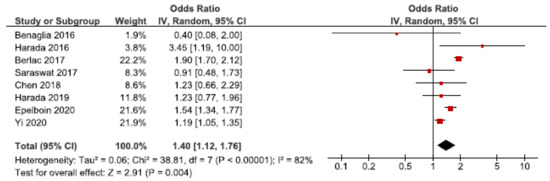
Figure 9.
Forest plot for endometriosis and placental abruption including studies with NOS ≥7. Chen et al., 2018 [30] used adjusted relative risk; Benaglia et al., 2016 [27] and Harada et al., 2019 [40] used crude odds ratios; Berlac et al., 2017 [28], Epelboin et al., 2020 [32], Harada et al., 2016 [39], Saraswat et al., 2017 [53], and Yi et al., 2020 [62] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S16).
3.7. Cesarean Section
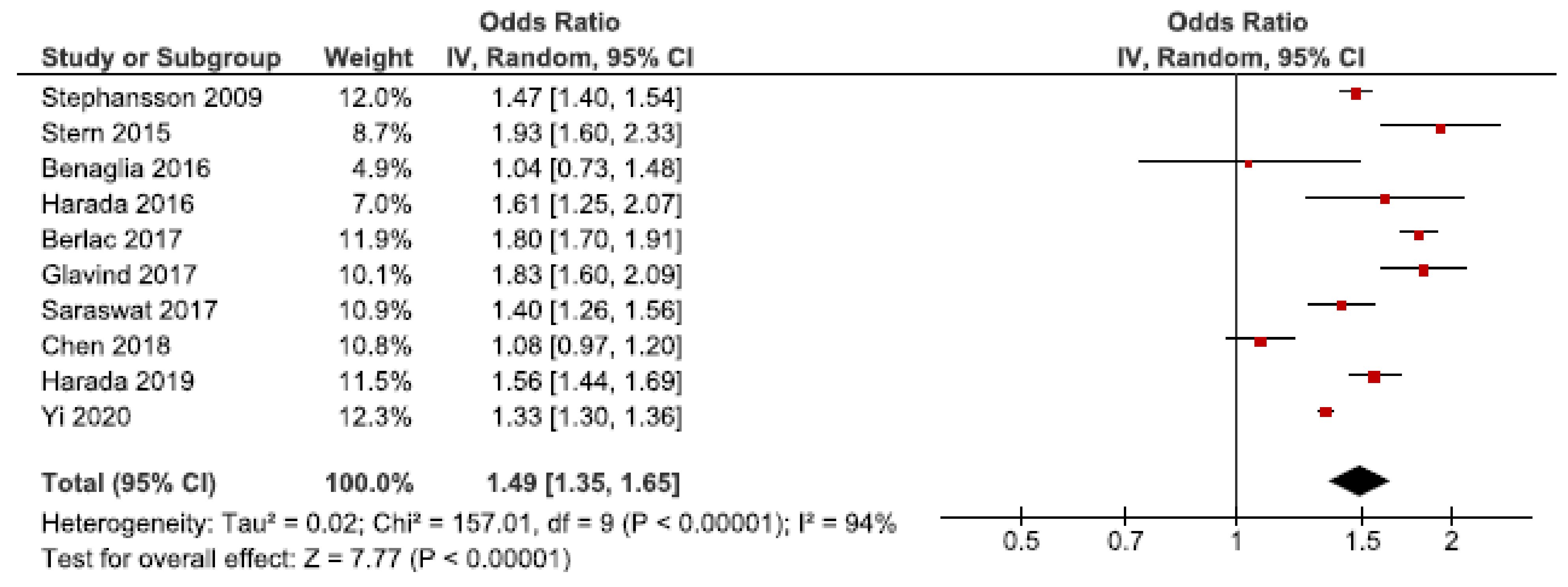
Cesarean section is indicated based on maternal (e.g., pre-eclampsia) or fetal complications (e.g., fetal distress). However, cesarean sections may cause severe maternal complications compared to vaginal deliveries [69]. We identified 28 cohort studies investigating the risk of cesarean section in women with endometriosis [25,26,27,28,30,31,33,37,39,40,41,42,44,45,46,47,48,49,50,52,53,55,56,57,59,60,61,62]. Ten of these were included in the main meta-analysis [27,28,30,37,39,40,53,56,57,62] (Figure 10), which showed an increased risk of cesarean section with an OR of 1.49 (95% CI: 1.35–1.65). Heterogeneity was high (I2 = 94%). Neither the secondary analysis including all studies regardless of study quality not the sub-analysis including only spontaneous pregnancies [45,48,52,57] changed the direction of the association between endometriosis and cesarean section (Supplementary Figure S17).
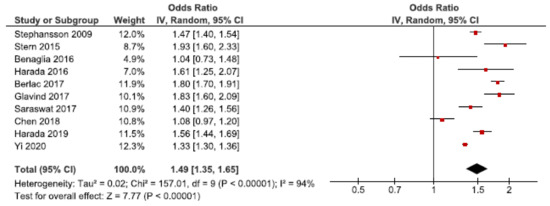
Figure 10.
Forest plot for endometriosis and cesarean section including studies with NOS ≥7. Chen et al., 2018 [30] used adjusted relative risk; Benaglia et al., 2016 [27], Harada et al., 2016 [39], and Harada et al., 2019 [40] used crude odds ratios; Berlac et al., 2017 [28], Glavind et al., 2017 [37], Saraswat et al., 2017 [53], Stephansson et al., 2009 [56], Stern et al., 2015 [57], and Yi et al., 2020 [62] used adjusted odds ratios.
The funnel plot was rather symmetrical (Supplementary Figure S18).
3.8. Stillbirth
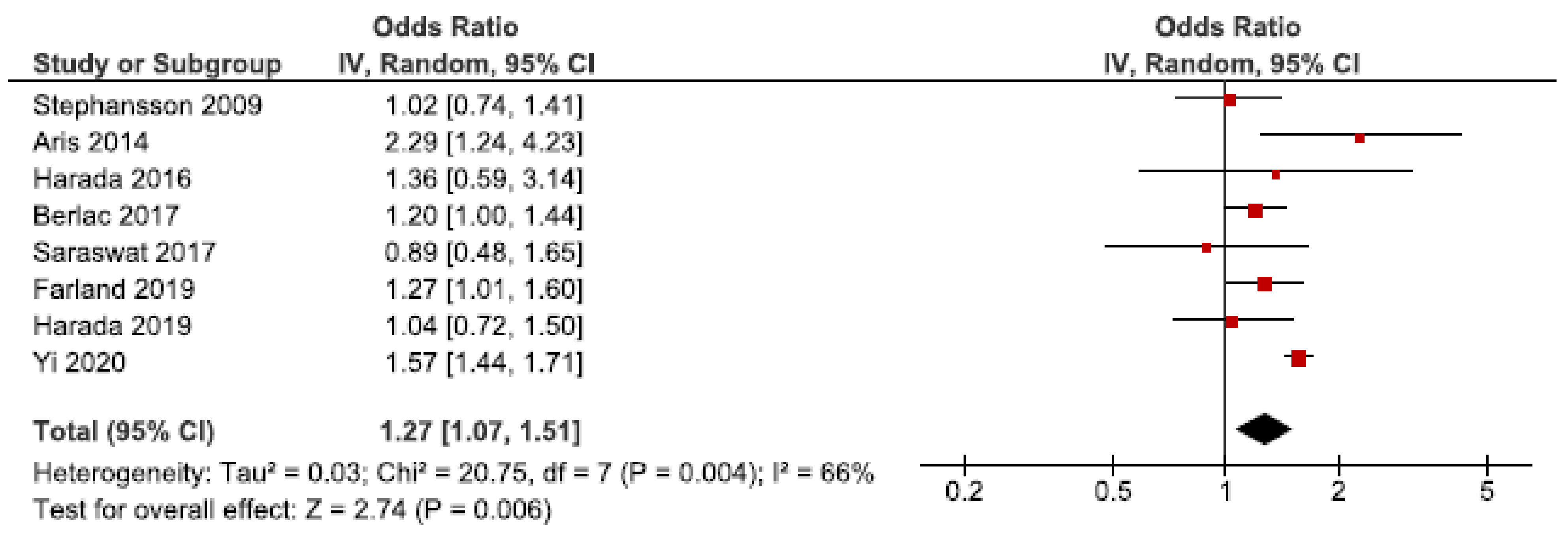
In 2009, 2.64 million stillbirths were recorded across 42 countries [70]. We identified nine cohort studies regarding stillbirth [24,28,34,39,40,52,53,56,62]. Eight of these were eligible for the main meta-analysis [24,28,34,39,40,53,56,62], which showed an association between endometriosis and stillbirth (OR: 1.27, 95% CI: 1.07–1.51) (Figure 11). Heterogeneity was rather high (I2 = 66%). The secondary analysis including all studies did not change the direction of the results (Supplementary Figure S19). The sub-analysis, including only spontaneous pregnancies, was not conducted as only one study was eligible [52].
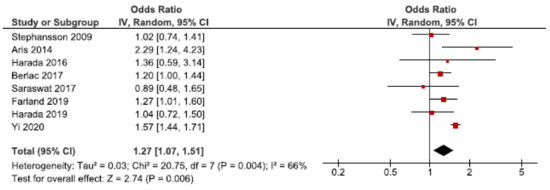
Figure 11.
Forest plot for endometriosis and stillbirth including studies with NOS ≥7. Farland et al., 2019 [34] used adjusted relative risk; Aris et al., 2014 [24], Harada et al., 2016 [39], and Harada et al., 2019 [40] used crude odds ratios; Berlac et al., 2017 [28], Saraswat et al., 2017 [53], Stephansson et al., 2009 [56], and Yi et al., 2020 [62] used adjusted odds ratios.
No funnel plot was made since only nine studies investigated stillbirth in women with endometriosis.
3.9. Postpartum Hemorrhage
Hemorrhage is the leading cause of maternal death worldwide, and PPH accounts for two-thirds of these [71]. We identified 15 cohort studies investigating the association between endometriosis and PPH [28,30,31,37,41,44,47,49,50,52,53,55,59,60,62]. Five of these were included in the main meta-analysis [28,30,37,53,62], without reaching statistically significance (Figure 12). We found a pooled OR of 1.05 (95% CI: 0.93–1.19). Heterogeneity was high (I2 = 84%). The secondary analysis, including all studies regardless of quality, did not change the direction of the association between endometriosis and PPH (Supplementary Figure S20). The sub-analysis, including only spontaneous pregnancies, was not conducted as only one study was eligible [52].
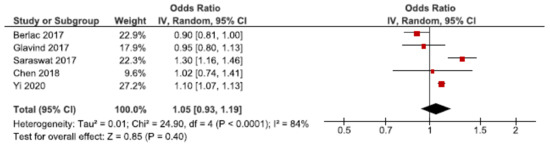
Figure 12.
Forest plot for endometriosis and postpartum hemorrhage, including studies with NOS ≥7. Chen et al., 2018 [30] used adjusted relative risk; Berlac et al., 2017 [28], Glavind et al., 2017 [37], Saraswat et al., 2017 [53], and Yi et al., 2020 [62] used adjusted odds ratios.
The funnel plot did not indicate publication bias (Supplementary Figure S21).
3.10. Spontaneous Hemoperitoneum in Pregnancy
SHiP is a rare pregnancy complication associated with fetal and maternal mortality [10]. Exacoustos et al. conducted the only cohort study on SHiP in women with endometriosis [33]. They found the odds to be 24 times as high in women with endometriosis, compared to women without the disease (OR: 24.6, 95% CI: 1.15–528) [33], but this conclusion was based on two cases of SHiP in women with endometriosis and no cases in women without endometriosis [33]. Still, recent reviews support an increased but poorly defined risk of SHiP in women with endometriosis [10,72,73]. The review by Lier et al. found that 33 of 59 cases of SHiP occurred in women with endometriosis [73]. In the initial search for this review, another five case reports, including eight cases of SHiP associated with endometriosis, were identified [74,75,76,77,78]. Taken together, the available evidence indicates that endometriosis may be associated with increased risk of SHiP.
3.11. Spontaneous Bowel Perforation in Pregnancy
Spontaneous bowel perforation in pregnancy is an abdominal emergency [10]. Setúbal et al. reported on three cases of bowel perforation due to endometriosis, at their own center, as well as 12 cases through a search of the literature until 2013 [79]. A further three cases of spontaneous bowel perforation during pregnancy in endometriosis patients were reported in 2014 [80], 2016 [81], and 2018 [82]. All cases had the diagnosis proven either prior to pregnancy or through biopsies taken during surgery. Thus, available data indicate that spontaneous bowel perforation may occur with increased risk in patients with bowel endometriosis.
4. Discussion
In this systematic review and meta-analysis, we addressed the association between endometriosis and adverse pregnancy outcomes. Overall, existing evidence pointed towards an association between endometriosis and gestational hypertension, pre-eclampsia, preterm birth, placenta previa, placental abruption, cesarean section, and stillbirth. However, the results on low birth weight, SGA, and PPH showed no statistical significance, and future studies on these outcomes are encouraged. The literature on SHiP and bowel perforation in pregnancy was sparse but indicated that endometriosis seemed to increase the risk of these rare, but severe complications.
4.1. Methodological Considerations
When evaluating the results from this systematic review and meta-analyses, several methodological aspects of the included studies need to be addressed. Only cohort studies were included, and thus only methodological considerations regarding cohort studies were considered. We performed quality assessment of the studies, focusing on the risk of selection bias, information bias, and confounding. Studies with a NOS score ≥7 were included in the main meta-analysis and compared to the results from the secondary meta-analysis including all studies regardless of study quality and the results from the sub-analyses including only spontaneous pregnancies. Results from the meta-analyses including all studies regardless of study quality and the sub-analysis including only spontaneous pregnancies were similar to those of the meta-analyses only including studies with a NOS score ≥7. However, we consider the main meta-analysis with the high-quality studies most valid.
A limitation of this systematic review is the high heterogeneity between the studies, which may explain, e.g., why hypertensive disorders in pregnancy, overall, had no statistically significant association with endometriosis, whereas gestational hypertension and pre-eclampsia did. This could well be due to the heterogeneous study populations of women with endometriosis, with various locations and types and severity of endometriosis. This review did not consider these aspects.
Selection bias is present if the association between endometriosis and adverse pregnancy outcomes differs between those included in the studies and the background population. Most studies did not report their participation rate, and among those that did, a rather small participation for women with endometriosis was reported [33,50]. A low participation rate may increase the risk of selection bias which could explain the different results found throughout the studies. Selection bias can be present if the frequency of adverse pregnancy outcome was higher in women with endometriosis who participated compared to women with endometriosis who did not participate in these studies, it could lead to bias away from the null.
Adjustment for potential confounders varied throughout the studies, and unadjusted or residual confounding may hamper interpretation of results. The covariates most often adjusted for were maternal age, parity, BMI, and smoking status. When scoring the original studies included in this review, we defined essential confounders as maternal age, socio-economic status, BMI, and smoking. We decided not to include parity in the NOS score because parity may be an intermediate factor between endometriosis and adverse pregnancy outcome. Adjusting for an intermediate factor may lead to bias towards the null and underestimate the association [83]. For pre-eclampsia, preterm birth, and stillbirth, the studies that adjusted for potential confounders tended to find a stronger association, as compared to the studies that did not adjust for any confounders. For placenta previa and cesarean section, most studies found an association regardless of adjustment for confounders. Furthermore, studies that adjusted for confounders tended to find a smaller association between endometriosis and adverse pregnancy outcome after adjustment
Women with endometriosis more often need fertility treatment, and ART may also affect pregnancy outcome. Thus, ART may act as an intermediate factor in the association between endometriosis and adverse pregnancy outcome [7,8].
Adjusting for ART may result in bias towards the null, thus leading to an underestimation of the association between endometriosis and adverse pregnancy outcomes [83]. The studies included handled ART in different ways, some by selection only within ART patients, while others adjusted, stratified, or excluded ART patients. Four studies included in this review adjusted for ART [28,30,31,49]. Berlac et al., showed a decreasing association when adjusting for ART between endometriosis and all outcomes apart from PPH [28]. The same pattern was seen in the studies by Chen et al., [30] and Miura et al., [49]. Conti et al., claimed that multivariate analysis including ART failed to change their results [31].
Two studies stratified their results by ART [37,56]. Glavind et al., found similar results in women with and without ART [37]. Stephansson et al., only stratified preterm birth by ART and found that the association attenuated in the ART group [56]. Furthermore, 14 of the included 37 studies neither stratified nor adjusted their results by ART.
The inclusion of women who conceived by ART or spontaneous pregnancies in only the exposed or non-exposed group may lead to bias. An association found when comparing ART pregnancies and spontaneous pregnancies might be due to ART and not endometriosis. Stern et al. [57] and Epelboin et al. [32] excluded ART pregnancies in their non-exposed group and divided their exposed group in ART and non-ART pregnancies. Exacoustos et al. only excluded ART pregnancies in their non-exposed group [33]. Lastly, Kuivasaari-Pirinen et al. [43] and Fernando et al. [35] compared ART pregnancies in endometriosis patients to spontaneous pregnancies in women without endometriosis.
Epelboin et al. compared women with endometriosis spontaneously or by ART. They found ART to act as an independent risk factor for placenta previa, preterm birth and SGA [32]. However, data are limited, and more studies on this aspect are encouraged.
The classification of exposure varied across the studies and may be important to consider. Miura et al. included women diagnosed with endometriosis through symptoms [49] and Harada et al. from 2016 and 2019 gathered information on endometriosis from questionnaires [39,40]. This may have led to women without a verified diagnosis of endometriosis being included in the exposed group and thus non-differentiated misclassification and bias towards the null. The three studies all received a lower NOS score (Supplementary Materials S3).
Measurement error and misclassification of the outcomes may be present but is unlikely to depend on endometriosis status. The definitions of outcomes varied throughout the studies. Notably, regarding stillbirth, the studies which found an association all defined stillbirth as fetal loss occurring after 20 completed weeks of gestation, while in the studies that found no association included gestational ages ranged from 22 to 28 weeks and above. Furthermore, PPH was defined differently throughout the studies. However, these differences were present in both the studies that found an association and those that did not; thus, they should not alter the results.
Additionally, not all studies stated how the outcome information was obtained [31,36,42,44,45,47,48]. Furthermore, three studies gathered information on outcomes by questionnaires and telephone interviews [25,27,34]. Self-reporting of outcomes may cause differential misclassification, as women with endometriosis might over-report adverse outcomes compared to women without endometriosis, which could then lead to an overestimation of the studied association.
The external validity of several of the included studies may be limited as they were based on selected populations. Eight studies recruited their exposed group among women with previous endometriosis surgery [25,31,33,36,44,45,47,50]. Though this procedure ensures a verified diagnosis, these women no longer represent the general population of women with endometriosis. Another important aspect to consider is that several studies focused only on women who conceived spontaneously [45,48,52,54] or following ART [26,27,29,36,41,46,58]. We conducted sub-analyses only including spontaneous pregnancies, and, overall, these provided similar results. However, women with endometriosis who conceived spontaneously will most likely have milder forms of endometriosis compared to women with endometriosis who conceive by ART. If the severity of endometriosis affects the risk of adverse pregnancy outcome this might bias the results towards the null.
4.2. Potential Mechanisms
Various theories exist regarding both the increased risk of infertility and the increased risk of adverse pregnancy outcome in women with endometriosis.
First, the thickening of the junctional zone, especially seen in women with advanced stages of endometriosis [84], may lead to abnormal remodeling of the spiral arteries and thus defective deep placentation [5,85]. The increased risk of placental dysfunction in women with endometriosis may lead to both adverse fetal outcomes (e.g., stillbirth) and adverse maternal outcomes (e.g., pre-eclampsia, placenta previa, placental abruption, and PPH) [5].
Furthermore, the inflammatory response caused by extrauterine endometrial cells may lead to increased levels of prostaglandins, cytokines, and macrophage activity, and by that to preterm labor contractions [86]. Additionally, the progesterone resistance and the increased estrogen levels seen in women with endometriosis may also contribute to an abnormal inflammatory response [87]. Furthermore, an increase in the activity of proteases and the breakdown of the extracellular matrix may lead to preterm pre-labor rupture of membranes (PPROM), and thus spontaneous preterm birth [86,88]. Although only sparsely studied, PPROM has previously been shown to be more frequent in women with endometriosis [31,39]. However, other studies failed to support this [27,30,39]. This could indicate that spontaneous preterm births, and not induced preterm births due to maternal or fetal complications, may be the main issue in endometriosis patients. This systematic review did not distinguish between spontaneous and induced preterm birth which may limit the results. Future studies on these aspects are encouraged.
Adenomyosis is a condition, associated with endometriosis, in which the endometrium invades the myometrium. Adenomyosis has not been included in this review but could play a pathogenetic role since Kunz et al. found that 90% of women suffering from pelvic endometriosis also had a diagnosis of adenomyosis [89]. The thickened junctional zone is diagnostic for adenomyosis and can lead to a disrupted placental bed and thereby obstetrical complications [85,89]. A recent review showed adenomyosis to have an even stronger association with preterm birth and SGA than endometriosis [90].
The rare outcome SHiP is a potentially life-threatening condition during pregnancy. Lier et al. found that bleeding originated from endometriotic implants, ruptured utero-ovarian vessels, hemorrhagic nodules of decidualized tissue, or a combination of these [91]
Spontaneous bowel perforations during pregnancy are mostly located in deep infiltrating bowel endometriosis in the sigmoid and rectum [92], and decidualization of these lesions represents a possible, yet unproven pathogenetic mechanism [79,93].
4.3. Clinical Aspects
The majority of original studies has rather consistently shown that women with endometriosis face a higher risk of preterm birth, irrespective of use of ART. However, future studies are needed to investigate preterm birth in more detail and to distinguish between medically indicated preterm birth and spontaneous preterm birth. Moreover, studies should investigate the effect of screening and potential interventions.
Furthermore, the risk of placenta previa was substantially increased, and we encourage heightened awareness for this complication during pregnancy in women with endometriosis.
The risk of cesarean section was also increased in women with endometriosis; however, this systematic review was limited by not differentiating between elective and acute cesarean section or investigating the underlying cause of cesarean section. Future studies are needed to determine whether the increased risk of cesarean sections is driven by acute or elective cesarean sections and whether they are carried out for fetal or maternal reasons in women with endometriosis.
The low incidence of SHiP and spontaneous bowel perforation in pregnancy implies that large-scale collaboration is needed to further define the risk profile and proper management of these serious pregnancy complications. With our present knowledge, an increased awareness in pregnant women with endometriosis seems reasonable when abdominal emergencies are encountered.
5. Conclusions
This systematic review with meta-analyses supports the notion that endometriosis is associated with an increased risk of several adverse pregnancy outcomes, including gestational hypertension, pre-eclampsia, preterm birth, placenta previa, placental abruption, cesarean section, and stillbirth. Furthermore, the two severe complications, SHiP and spontaneous bowel perforation in pregnancy, may also be associated with endometriosis, but large observational studies are needed to explore this further.
Supplementary Materials
The following are available online at https://www.mdpi.com/2077-0383/10/4/667/s1, Supplementary Material S1: PRISMA checklist, Supplementary Material S2: Search strings, Supplementary Material S3: Explanation of Newcastle–Ottawa quality assessment scale, Figure S1: Forest plot for endometriosis and hypertensive disorders in pregnancy overall including all studies regardless of study quality, Figure S2: Funnel plot for endometriosis and hypertensive disorders in pregnancy overall, Figure S3: Forest plot for endometriosis and gestational hypertension including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S4: Funnel plot for endometriosis and gestational hypertension, Figure S5: Forest plot for endometriosis and pre-eclampsia including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S6: Funnel plot for endometriosis and pre-eclampsia, Figure S7: Forest plot for endometriosis and low birth weight including all studies regardless of study (a) and including only studies with spontaneous pregnancies (b), Figure S8: Funnel plot for endometriosis and low birth weight, Figure S9: Forest plot for endometriosis and small for gestational age including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S10: Funnel plot for endometriosis and small for gestational age, Figure S11: Forest plot for endometriosis and preterm birth including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S12: Funnel plot for endometriosis and preterm birth, Figure S13: Forest plot for endometriosis and placenta previa including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S14: Funnel plot for endometriosis and placenta previa, Figure S15: Forest plot for endometriosis and placental abruption including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S16: Funnel plot for endometriosis and placental abruption, Figure S17: Forest plot for endometriosis and cesarean section including all studies regardless of study quality (a) and including only studies with spontaneous pregnancies (b), Figure S18: Funnel plot for endometriosis and cesarean section, Figure S19: Forest plot endometriosis and stillbirth including for all studies regardless of study quality, Figure S20: Forest plot for endometriosis and postpartum hemorrhage including all studies regardless of study quality, Figure S21: Funnel plot for endometriosis and postpartum hemorrhage.
Author Contributions
Conceptualization, K.B., A.F., N.U., D.R., T.B.H. and L.H.A.; methodology, A.F., T.B.H., N.U., D.R., R.P., L.H.A. and K.B.; software, not relevant; validation, L.H.A., R.P. and K.B.; formal analysis, L.H.A., R.P. and K.B.; investigation, K.B.; resources, not relevant; data curation, A.F., K.B., L.H.A. and R.P.; writing—original draft preparation, K.B.; writing—review and editing, L.H.A., A.F., T.B.H., N.U., D.R., R.P. and K.B.; visualization, K.B., A.F., N.U., T.B.H., D.R., R.P. and L.H.A.; supervision, L.H.A., A.F. and T.B.H.; project administration, L.H.A., A.F. and K.B.; funding acquisition, no funding was provided for this article. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors thank librarian, Anne Vils Møller, from Health Sciences, Aarhus University for her contribution in the systematic literature search.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Giudice, L.C.; Kao, L.C. Endometriosis. Lancet 2004, 364, 1789–1799. [Google Scholar] [CrossRef]
- Gruppo Italiano per lo Studio dell’Endometriosi. Prevalence and anatomical distribution of endometriosis in women with selected gynaecological conditions: Results from a multicentric Italian study. Hum. Reprod. 1994, 9, 1158–1162. [Google Scholar] [CrossRef]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef]
- Symons, L.K.; Miller, J.E.; Kay, V.R.; Marks, R.M.; Liblik, K.; Koti, M.; Tayade, C. The Immunopathophysiology of Endometriosis. Trends Mol. Med. 2018, 24, 748–762. [Google Scholar] [CrossRef] [PubMed]
- Brosens, I.; Brosens, J.J.; Fusi, L.; Al-Sabbagh, M.; Kuroda, K.; Benagiano, G. Risks of adverse pregnancy outcome in endometriosis. Fertil. Steril. 2012, 98, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Goldberg, J.M.; Aziz, N.; Goldberg, E.; Krajcir, N.; Agarwal, A. Pathogenic mechanisms in endometriosis-associated infertility. Fertil. Steril. 2008, 90, 247–257. [Google Scholar] [CrossRef]
- De Ziegler, D.; Borghese, B.; Chapron, C. Endometriosis and infertility: Pathophysiology and management. Lancet 2010, 376, 730–738. [Google Scholar] [CrossRef]
- Qin, J.; Liu, X.; Sheng, X.; Wang, H.; Gao, S. Assisted reproductive technology and the risk of pregnancy-related complications and adverse pregnancy outcomes in singleton pregnancies: A meta-analysis of cohort studies. Fertil. Steril. 2016, 105, 73–85.e6. [Google Scholar] [CrossRef]
- Zullo, F.; Spagnolo, E.; Saccone, G.; Acunzo, M.; Xodo, S.; Ceccaroni, M.; Berghella, V. Endometriosis and obstetrics complications: A systematic review and meta-analysis. Fertil. Steril. 2017, 108, 667–672.e5. [Google Scholar] [CrossRef]
- Glavind, M.T.; Møllgaard, M.V.; Iversen, M.L.; Arendt, L.H.; Forman, A. Obstetrical outcome in women with endometriosis including spontaneous hemoperitoneum and bowel perforation: A systematic review. Best Pr. Res. Clin. Obstet. Gynaecol. 2018, 51, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Maggiore, U.L.R.; Ferrero, S.; Mangili, G.; Bergamini, A.; Inversetti, A.; Giorgione, V.; Viganò, P.; Candiani, M. A systematic review on endometriosis during pregnancy: Diagnosis, misdiagnosis, complications and outcomes. Hum. Reprod. Updat. 2016, 22, 70–103. [Google Scholar] [CrossRef] [PubMed]
- Lalani, S.; Choudhry, A.J.; Firth, B.; Bacal, V.; Walker, M.; Wen, S.W.; Singh, S.; Amath, A.; Hodge, M.; Chen, I. Endometriosis and adverse maternal, fetal and neonatal outcomes, a systematic review and meta-analysis. Hum. Reprod. 2018, 33, 1854–1865. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; for the PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Facchin, F.; Buggio, L.; Ottolini, F.; Barbara, G.; Saita, E.; Vercellini, P. Preliminary insights on the relation between endometriosis, pelvic pain, and employment. Gynecol. Obstet. Investig. 2018, 84, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Parazzini, F.; Esposito, G.; Tozzi, L.; Noli, S.; Bianchi, S. Epidemiology of endometriosis and its comorbidities. Eur. J. Obstetrics Gynecol. Reprod. Biol. 2017, 209, 3–7. [Google Scholar] [CrossRef]
- Tang, Y.; Zhao, M.; Lin, L.; Gao, Y.; Chen, G.Q.; Chen, S.; Chen, Q. Is body mass index associated with the incidence of endometriosis and the severity of dysmenorrhoea: A case–control study in China? BMJ Open 2020, 10, e037095. [Google Scholar] [CrossRef]
- Frederiksen, L.E.; Ernst, A.; Brix, N.; Lauridsen, L.L.B.; Roos, L.; Ramlau-Hansen, C.H.; Ekelund, C.K. Risk of Adverse Pregnancy Outcomes at Advanced Maternal Age. Obstet. Gynecol. 2018, 131, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Marchi, J.; Berg, M.V.D.; Dencker, A.; Olander, E.K.; Begley, C. Risks associated with obesity in pregnancy, for the mother and baby: A systematic review of reviews. Obes. Rev. 2015, 16, 621–638. [Google Scholar] [CrossRef]
- Meyer, M.B.; Jonas, B.S.; A Tonascia, J. Perinatal events associated with maternal smoking during pregnancy. Am. J. Epidemiol. 1976, 103, 464–476. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Lee, S.M.; Bae, S.-H.; Kim, H.J.; Lim, N.G.; Yoon, S.-J.; Lee, J.Y.; Jo, M.-W. Socioeconomic status can affect pregnancy outcomes and complications, even with a universal healthcare system. Int. J. Equity Health 2018, 17, 2. [Google Scholar] [CrossRef]
- Review Manager (RevMan), Version 5.4; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2014; Available online: https://training.cochrane.org/online-learning/core-software-cochrane-reviews/revman (accessed on 18 January 2021).
- Page, M.J.; Higgins, J.P.T.; Sterne, J.A.C. Assessing risk of bias due to missing results in a synthesis. In Cochrane Handbook for Systematic Reviews of Interventions; Version 6.1; Higgins, J.P.T., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: Spokane, WA, USA, 2020; Chapter 13. [Google Scholar]
- Deeks, J.J.; Higgins, J.P.T.; Altman, D.G. Analysing data and undertaking meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; Version 6.1; Higgins, J.P.T., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: Spokane, WA, USA, 2020; Chapter 10. [Google Scholar]
- Aris, A. A 12-year cohort study on adverse pregnancy outcomes in Eastern Townships of Canada: Impact of endometriosis. Gynecol. Endocrinol. 2013, 30, 34–37. [Google Scholar] [CrossRef]
- Baggio, S.; Pomini, P.; Zecchin, A.; Garzon, S.; Bonin, C.; Santi, L.; Festi, A.; Franchi, M.P. Delivery and pregnancy outcome in women with bowel resection for deep endometriosis: A retrospective cohort study. Gynecol. Surg. 2015, 12, 279–285. [Google Scholar] [CrossRef]
- Benaglia, L.; Bermejo, A.; Somigliana, E.; Scarduelli, C.; Ragni, G.; Fedele, L.; Garcia-Velasco, J.A. Pregnancy outcome in women with endometriomas achieving pregnancy through IVF. Hum. Reprod. 2012, 27, 1663–1667. [Google Scholar] [CrossRef]
- Benaglia, L.; Candotti, G.; Papaleo, E.; Pagliardini, L.; Leonardi, M.; Reschini, M.; Quaranta, L.; Munaretto, M.; Viganò, P.; Candiani, M.; et al. Pregnancy outcome in women with endometriosis achieving pregnancy with IVF. Hum. Reprod. 2016, 31, 2730–2736. [Google Scholar] [CrossRef] [PubMed]
- Berlac, J.F.; Hartwell, D.; Skovlund, C.W.; Langhoff-Roos, J.; Lidegaard, Ø. Endometriosis increases the risk of obstetrical and neonatal complications. Acta Obstet. Gynecol. Scand. 2017, 96, 751–760. [Google Scholar] [CrossRef]
- Brosens, I.A.; De Sutter, P.; Hamerlynck, T.; Imeraj, L.; Yao, Z.; Cloke, B.; Brosens, J.J.; Dhont, M. Endometriosis is associated with a decreased risk of pre-eclampsia. Hum. Reprod. 2007, 22, 1725–1729. [Google Scholar] [CrossRef]
- Chen, I.; Lalani, S.; Xie, R.-H.; Shen, M.; Singh, S.S.; Wen, S.-W. Association between surgically diagnosed endometriosis and adverse pregnancy outcomes. Fertil. Steril. 2018, 109, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Conti, N.; Cevenini, G.; Vannuccini, S.; Orlandini, C.; Valensise, H.; Gervasi, M.T.; Ghezzi, F.; Di Tommaso, M.; Severi, F.M.; Petraglia, F. Women with endometriosis at first pregnancy have an increased risk of adverse obstetric outcome. J. Matern. Fetal Neonatal Med. 2014, 28, 1795–1798. [Google Scholar] [CrossRef]
- Epelboin, S.; Labrosse, J.; Fauque, P.; Levy, R.; Gervoise-Boyer, M.-J.; Devaux, A.; Bergère, M.; De Vienne, C.; Jonveaux, P.; De Mouzon, J.; et al. Endometriosis and assisted reproductive techniques independently related to mother–child morbidities: A French longitudinal national study. Reprod. Biomed. Online 2020. [Google Scholar] [CrossRef]
- Exacoustos, C.; Lauriola, I.; Lazzeri, L.; De Felice, G.; Zupi, E. Complications during pregnancy and delivery in women with untreated rectovaginal deep infiltrating endometriosis. Fertil. Steril. 2016, 106, 1129–1135.e1. [Google Scholar] [CrossRef] [PubMed]
- Farland, L.V.; Prescott, J.; Sasamoto, N.; Tobias, D.K.; Gaskins, A.J.; Stuart, J.J.; Carusi, D.A.; Chavarro, J.E.; Horne, A.W.; Rich-Edwards, J.W.; et al. Endometriosis and Risk of Adverse Pregnancy Outcomes. Obstet. Gynecol. 2019, 134, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Fernando, S.; Breheny, S.; Jaques, A.M.; Halliday, J.L.; Baker, G.; Healy, D. Preterm birth, ovarian endometriomata, and assisted reproduction technologies. Fertil. Steril. 2009, 91, 325–330. [Google Scholar] [CrossRef]
- Fujii, T.; Wada-Hiraike, O.; Nagamatsu, T.; Harada, M.; Hirata, T.; Koga, K.; Fuji, T.; Osuga, Y. Assisted reproductive technology pregnancy complications are significantly associated with endometriosis severity before conception: A retrospective cohort study. Reprod. Biol. Endocrinol. 2016, 14. [Google Scholar] [CrossRef]
- Glavind, M.T.; Forman, A.; Arendt, L.H.; Nielsen, K.; Henriksen, T.B. Endometriosis and pregnancy complications: A Danish cohort study. Fertil. Steril. 2017, 107, 160–166. [Google Scholar] [CrossRef]
- Hadfield, R.M.; Lain, S.J.; Raynes-Greenow, C.H.; Morris, J.M.; Roberts, C.L. Is there an association between endometriosis and the risk of pre-eclampsia? A population based study. Hum. Reprod. 2009, 24, 2348–2352. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Taniguchi, F.; Onishi, K.; Kurozawa, Y.; Hayashi, K.; Harada, T. Japan Environment and Children’s Study Group, Obstetrical complications in women with endometriosis: A cohort study in Japan. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Taniguchi, F.; Amano, H.; Kurozawa, Y.; Ideno, Y.; Hayashi, K.; Harada, T. Japan Environment and Children’s Study Group. Adverse obstetrical outcomes for women with endometriosis and adenomyosis: A large cohort of the Japan Environment and Children’s Study. PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Jacques, M.; Freour, T.; Barriere, P.; Ploteau, S. Adverse pregnancy and neo-natal outcomes after assisted reproductive treatment in patients with pelvic endometriosis: A case–control study. Reprod. Biomed. Online 2016, 32, 626–634. [Google Scholar] [CrossRef]
- Kortelahti, M.; Anttila, M.A.; Hippeläinen, M.I.; Heinonen, S.T. Obstetric Outcome in Women with Endometriosis—A Matched Case-Control Study. Gynecol. Obstet. Investig. 2003, 56, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Kuivasaari-Pirinen, P.; Raatikainen, K.; Hippeläinen, M.; Heinonen, S. Adverse Outcomes of IVF/ICSI Pregnancies Vary Depending on Aetiology of Infertility. ISRN Obstet. Gynecol. 2012, 2012, 1–5. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.-L.; Chang, X.-H.; Li, Y.; Wang, Y.; Guan, J.; Cui, H. Effects of Previous Laparoscopic Surgical Diagnosis of Endometriosis on Pregnancy Outcomes. Chin. Med. J. 2017, 130, 428–433. [Google Scholar] [CrossRef]
- Lin, H.; Leng, J.H.; Liu, J.T.; Lang, J.H. Obstetric outcomes in chinese women with endometriosis: A retrospective cohort study. Chin. Med J. 2015, 128, 455–458. [Google Scholar] [CrossRef]
- Luke, B.; Stern, J.E.; Kotelchuck, M.; Declercq, E.R.; Cohen, B.; Diop, H. Birth Outcomes by Infertility Diagnosis Analyses of the Massachusetts Outcomes Study of Assisted Reproductive Technologies (MOSART). J. Reprod. Med. 2016, 60, 480–490. [Google Scholar] [CrossRef][Green Version]
- Mannini, L.; Sorbi, F.; Noci, I.; Ghizzoni, V.; Perelli, F.; Di Tommaso, M.; Mattei, A.; Fambrini, M. New adverse obstetrics outcomes associated with endometriosis: A retrospective cohort study. Arch. Gynecol. Obstet. 2016, 295, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Mekaru, K.; Masamoto, H.; Sugiyama, H.; Asato, K.; Heshiki, C.; Kinjyo, T.; Aoki, Y. Endometriosis and pregnancy outcome: Are pregnancies complicated by endometriosis a high-risk group? Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 172, 36–39. [Google Scholar] [CrossRef]
- Miura, M.; Ushida, T.; Imai, K.; Wang, J.; Moriyama, Y.; Nakano-Kobayashi, T.; Osuka, S.; Kikkawa, F.; Kotani, T. Adverse effects of endometriosis on pregnancy: A case-control study. BMC Pregnancy Childbirth 2019, 19, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Nirgianakis, K.; Gasparri, M.L.; Radan, A.-P.; Villiger, A.; McKinnon, B.; Mosimann, B.; Papadia, A.; Mueller, M.D. Obstetric complications after laparoscopic excision of posterior deep infiltrating endometriosis: A case–control study. Fertil. Steril. 2018, 110, 459–466. [Google Scholar] [CrossRef]
- Pan, M.-L.; Chen, L.-R.; Tsao, H.-M.; Chen, K.-H. Risk of gestational hypertension-preeclampsia in women with preceding endometriosis: A nationwide population-based study. PLoS ONE 2017, 12, e0181261. [Google Scholar] [CrossRef] [PubMed]
- Porpora, M.G.; Tomao, F.; Ticino, A.; Piacenti, I.; Scaramuzzino, S.; Simonetti, S.; Imperiale, L.; Sangiuliano, C.; Masciullo, L.; Manganaro, L.; et al. Endometriosis and Pregnancy: A Single Institution Experience. Int. J. Environ. Res. Public Health 2020, 17, 401. [Google Scholar] [CrossRef]
- Saraswat, L.; Ayansina, D.T.; Cooper, K.G.; Bhattacharya, S.; Miligkos, D.; Horne, A.W. Pregnancy outcomes in women with endometriosis: A national record linkage study. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Scala, C.; Maggiore, U.L.R.; Barra, F.; Tantari, M.; Ferrero, S. Impact of Endometriomas and Deep Infiltrating Endometriosis on Pregnancy Outcomes and on First and Second Trimester Markers of Impaired Placentation. Medicina 2019, 55, 550. [Google Scholar] [CrossRef]
- Shmueli, A.; Salman, L.; Hiersch, L.; Ashwal, E.; Hadar, E.; Wiznitzer, A.; Yogev, Y.; Aviram, A. Obstetrical and neonatal outcomes of pregnancies complicated by endometriosis. J. Matern. Neonatal Med. 2017, 32, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Stephansson, O.; Kieler, H.; Granath, F.; Falconer, H. Endometriosis, assisted reproduction technology, and risk of adverse pregnancy outcome. Hum. Reprod. 2009, 24, 2341–2347. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.E.; Luke, B.; Tobias, M.; Gopal, D.; Hornstein, M.D.; Diop, H. Adverse pregnancy and birth outcomes associated with underlying diagnosis with and without assisted reproductive technology treatment. Fertil. Steril. 2015, 103, 1438–1445. [Google Scholar] [CrossRef] [PubMed]
- Sunkara, S.K.; Antonisamy, B.; Redla, A.C.; Kamath, M.S. Female causes of infertility are associated with higher risk of preterm birth and low birth weight: Analysis of 117 401 singleton live births following IVF. Hum. Reprod. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tzur, T.; Weintraub, A.Y.; Gutman, O.A.; Baumfeld, Y.; Soriano, D.; Mastrolia, S.A.; Sheiner, E. Pregnancy outcomes in women with endometriosis. Minerva Ginecol. 2017, 70, 144–149. [Google Scholar]
- Uccella, S.; Manzoni, P.; Cromi, A.; Marconi, N.; Gisone, B.; Miraglia, A.; Biasoli, S.; Zorzato, P.C.; Ferrari, S.; Lanzo, G.; et al. Pregnancy after Endometriosis: Maternal and Neonatal Outcomes according to the Location of the Disease. Am. J. Perinatol. 2019, 36, S91–S98. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, D.; Pietrzak, B.; Szymusik, I.; Smiech, Z.; Wielgos, M. Should the patients with endometriosis be treated as a risk group of pregnancy complications? Single center experience and literature review and literature review. Ginekol. Polska 2020, 91, 383–388. [Google Scholar] [CrossRef]
- Yi, K.W.; Cho, G.J.; Park, K.; Han, S.W.; Shin, J.-H.; Kim, T.; Hur, J.-Y. Endometriosis Is Associated with Adverse Pregnancy Outcomes: A National Population-Based Study. Reprod. Sci. 2020, 27, 1175–1180. [Google Scholar] [CrossRef]
- WHO Guidelines Approved by the Guidelines Review Committee. In WHO Recommendations for Prevention and Treatment of Pre-Eclampsia and Eclampsia; World Health Organization: Geneva, Switzerland, 2011.
- Mol, B.W.J.; Roberts, C.T.; Thangaratinam, S.; Magee, L.A.; De Groot, C.J.; Hofmeyr, G.J. Pre-eclampsia. Lancet 2016, 387, 999–1011. [Google Scholar] [CrossRef]
- Cutland, C.L.; Lackritz, E.M.; Mallett-Moore, T.; Bardají, A.; Chandrasekaran, R.; Lahariya, C.; Nisar, M.I.; Tapia, M.D.; Pathirana, J.; Kochhar, S.; et al. Low birth weight: Case definition & guidelines for data collection, analysis, and presentation of maternal immunization safety data. Vaccine 2017, 35, 6492–6500. [Google Scholar] [CrossRef]
- Lau, C.; Rogers, J.M.; Desai, M.; Ross, M.G. Fetal programming of adult disease: Implications for prenatal care. Obstet. Gynecol. 2011, 117, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Howson, C.P.; The Born Too Soon Preterm Birth Action Group; Kinney, M.V.; McDougall, L.; Lawn, E.J. Born Too Soon: Preterm birth matters. Reprod. Health 2013, 10, S1. [Google Scholar] [CrossRef]
- Mercier, F.J.; Van de Velde, M. Major obstetric hemorrhage. Anesthesiol. Clin. 2008, 26, 53–66, vi. [Google Scholar] [CrossRef]
- Villar, J.; Carroli, G.; Zavaleta, N.; Donner, A.; Wojdyla, D.M.; Faundes, A.; Velazco, A.; Bataglia, V.; Langer, A.; Narváez, A.; et al. Maternal and neonatal individual risks and benefits associated with caesarean delivery: Multicentre prospective study. BMJ 2007, 335, 1025. [Google Scholar] [CrossRef] [PubMed]
- Cousens, S.; Blencowe, H.; Stanton, C.; Chou, D.; Ahmed, S.; Steinhardt, L.; Creanga, A.A.; Tunçalp, Ö.; Balsara, Z.P.; Gupta, S.; et al. National, regional, and worldwide estimates of stillbirth rates in 2009 with trends since 1995: A systematic analysis. Lancet 2011, 377, 1319–1330. [Google Scholar] [CrossRef]
- Say, L.; Chou, D.; Gemmill, A.; Tunçalp, Ö.; Moller, A.-B.; Daniels, J.; Gülmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A WHO systematic analysis. Lancet Glob. Health 2014, 2, e323–e333. [Google Scholar] [CrossRef]
- Brosens, I.A.; Fusi, L.; Brosens, J.J. Endometriosis is a risk factor for spontaneous hemoperitoneum during pregnancy. Fertil. Steril. 2009, 92, 1243–1245. [Google Scholar] [CrossRef] [PubMed]
- Lier, M.C.; Malik, R.F.; Ket, J.C.; Lambalk, C.B.; Brosens, I.A.; Mijatovic, V. Spontaneous hemoperitoneum in pregnancy (SHiP) and endometriosis—A systematic review of the recent literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 219, 57–65. [Google Scholar] [CrossRef]
- Gao, F.-M.; Liu, G.-L. Four Case Reports of Endometriosis-Related Hemoperitoneum in Pregnancy. Chin. Med. J. 2018, 131, 502–504. [Google Scholar] [CrossRef]
- Feld, Z.; Rowen, T.; Callen, A.; Goldstein, R.; Poder, L. Uterine artery pseudoaneurysm in the setting of deep endometriosis: An uncommon cause of hemoperitoneum in pregnancy. Emerg. Radiol. 2017, 25, 107–110. [Google Scholar] [CrossRef]
- Gonzalez, A.; Artazcoz, S.; Elorriaga, F.; Palin, H.; Carugno, J. Massive recurrent hemoperitoneum with encapsulating peritonitis: Another enigmatic clinical feature of endometriosis. Fertil. Steril. 2019, 112, 1190–1192. [Google Scholar] [CrossRef]
- Naeh, A.; Bruchim, I.; Hallak, M.; Gabbay-Benziv, R. Endometriosis-related Hemoperitoneum in Late Pregnancy. ISR Med. Assoc. J. 2019, 21, 557–559. [Google Scholar]
- Kim, B.H.; Park, S.N.; Kim, B.R. Endometriosis-induced massive hemoperitoneum misdiagnosed as ruptured ectopic pregnancy: A case report. J. Med. Case Rep. 2020, 14. [Google Scholar] [CrossRef] [PubMed]
- Setubal, A.; Sidiropoulou, Z.; Torgal, M.; Casal, E.; Lourenço, C.; Koninckx, P. Bowel complications of deep endometriosis during pregnancy or in vitro fertilization. Fertil. Steril. 2014, 101, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Sartini, A.; Garibaldi, S.; Cencini, M. Deep Endometriosis Induced Spontaneous Colon Rectal Perforation in Pregnancy: Laparoscopy Is Advanced Tool to Confirm Diagnosis. Case Reports in Obstetrics and Gynecology 2014, 2014, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Albareda, J.; Albi, M.V.; Sosa, G.; Cano, A.; Macello, M.E.; Martin, B.A. Puerperal ileal perforation secondary to endometriosis: Case report and literature review. Taiwan. J. Obstet. Gynecol. 2016, 55, 121–124. [Google Scholar] [CrossRef]
- Carneiro, M.M.; Costa, L.M.P.; Torres, M.D.G.; Gouvea, P.S.; De Ávila, I. Intestinal Perforation due to Deep Infiltrating Endometriosis during Pregnancy: Case Report. RBGO Gynecol. Obstet. 2018, 40, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Schisterman, E.F.; Cole, S.R.; Platt, R.W. Overadjustment Bias and Unnecessary Adjustment in Epidemiologic Studies. Epidemiology 2009, 20, 488–495. [Google Scholar] [CrossRef]
- Landi, S.; Mereu, L.; Pontrelli, G.; Stepniewska, A.; Romano, L.; Tateo, S.; Dorizzi, C.; Minelli, L. The influence of adenomyosis in patients laparoscopically treated for deep endometriosis. J. Minim. Invasive Gynecol. 2008, 15, 566–570. [Google Scholar] [CrossRef]
- Brosens, I.; Derwig, I.; Brosens, J.; Fusi, L.; Benagiano, G.; Pijnenborg, R. The enigmatic uterine junctional zone: The missing link between reproductive disorders and major obstetrical disorders? Hum. Reprod. 2010, 25, 569–574. [Google Scholar] [CrossRef]
- Romero, R.; Dey, S.K.; Fisher, S.J. Preterm labor: One syndrome, many causes. Science 2014, 345, 760–765. [Google Scholar] [CrossRef]
- Marquardt, R.M.; Kim, T.H.; Shin, J.H.; Jeong, J. Progesterone and Estrogen Signaling in the Endometrium: What Goes Wrong in Endometriosis? Int. J. Mol. Sci. 2019, 20, 3822. [Google Scholar] [CrossRef]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Kunz, G.; Beil, D.; Huppert, P.; Noe, M.; Kissler, S.; Leyendecker, G. Adenomyosis in endometriosis—prevalence and impact on fertility. Evidence from magnetic resonance imaging. Hum. Reprod. 2005, 20, 2309–2316. [Google Scholar] [CrossRef]
- Bruun, M.R.; Arendt, L.H.; Forman, A.; Ramlau-Hansen, C.H. Endometriosis and adenomyosis are associated with increased risk of preterm delivery and a small-for-gestational-age child: A systematic review and meta-analysis. Acta Obstet. Gynecol. Scand. 2018, 97, 1073–1090. [Google Scholar] [CrossRef]
- Lier, M.C.; Brosens, J.J.; Mijatovic, V.; Habiba, M.; Benagiano, G. Decidual Bleeding as a Cause of Spontaneous Hemoperitoneum in Pregnancy and Risk of Preterm Birth. Gynecol. Obstet. Investig. 2017, 82, 313–321. [Google Scholar] [CrossRef]
- Navarro, R.; Poder, L.; Sun, D.; Jha, P. Endometriosis in pregnancy. Abdom. Radiol. 2020, 45, 1741–1753. [Google Scholar] [CrossRef]
- Pisanu, A.; Deplano, D.; Angioni, S.; Ambu, R.; Uccheddu, A. Rectal perforation from endometriosis in pregnancy: Case report and literature review. World J. Gastroenterol. 2010, 16, 648–651. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).