Abstract
Despite the progress of its management, COVID-19 maintains an ominous condition which constitutes a threat, especially for the susceptible population. The cardiac injury occurs in approximately 30% of COVID-19 infections and is associated with a worse prognosis. The clinical presentation of cardiac involvement can be COVID-19-related myocarditis. Our review aims to summarise current evidence about that complication. The research was registered at PROSPERO (CRD42022338397). We performed a systematic analysis using five different databases, including i.a. MEDLINE. Further, the backward snowballing technique was applied to identify additional papers. Inclusion criteria were: full-text articles in English presenting cases of COVID-19-related myocarditis diagnosed by the ESC criteria and patients over 18 years old. The myocarditis had to occur after the COVID-19 infection, not vaccination. Initially, 1588 papers were screened from the database search, and 1037 papers were revealed in the backward snowballing process. Eventually, 59 articles were included. Data about patients’ sex, age, ethnicity, COVID-19 confirmation technique and vaccination status, reported symptoms, physical condition, laboratory and radiological findings, applied treatment and patient outcome were investigated and summarised. COVID-19-related myocarditis is associated with the risk of sudden worsening of patients’ clinical status, thus, knowledge about its clinical presentation is essential for healthcare workers.
1. Introduction
The pandemic of COVID-19 had an immense impact on nearly every aspect of the modern world. Until August 2022 585 million people have been infected, and 6.4 million died [1]. Initially, it was considered a respiratory disease, but it soon occurred to be a multidimensional condition, dangerous for several systems, including cardiovascular [2,3,4]. Cardiac manifestations of COVID are varied and include i.a., arrhythmias, acute coronary syndrome, heart failure decompensation and myocarditis [5]. Myocarditis is an inflammatory disease of the myocardium with vast symptomatology and not entirely ascertained pathogenesis [6]. Since myocarditis can lead to life-threatening conditions and its incidence is interwound with the current most crucial epidemiological problem–COVID-19—w decided to perform a systematic review of the available literature. The aim of our study was to show the up-to-date evidence of epidemiology, clinical course, diagnostics, treatment and prognosis of COVID-related myocarditis.
2. Materials and Methods
The study was conducted following PRISMA guidelines [7] and was registered in PROSPERO (no (CRD42022338397).
2.1. Search Strategy
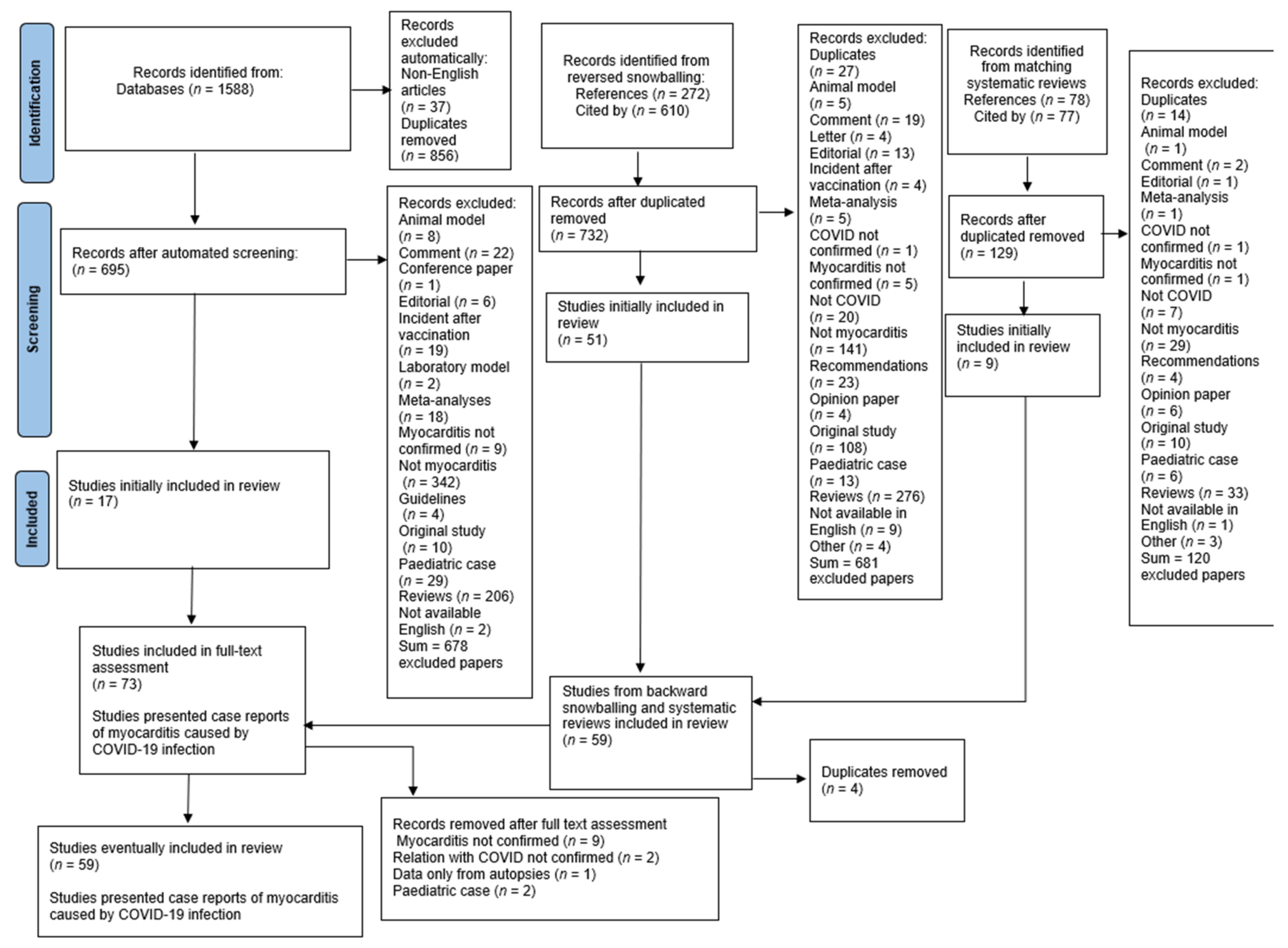
A systematic search was performed using EBSCOhost searching engine. MEDLINE, MEDLINE Ultimate, Health Source—Consumer Edition, Health Source: Nursing/Academic Edition, Academic Search Ultimate and ERIC databases were screened. The research included papers published from 1 December 2019 until 1 June 2022. Two researchers (S.U. and M.B.) independently searched databases using the following keywords: (COVID-19 or coronavirus or 2019-ncov or SARS-CoV-2 or CoV-19 or novel coronavirus OR SARS CoV OR SARS-CoV-19) AND (“cardiac damage” OR “cardiac injury” OR “acute cardiac injury” OR “acute myocardial injury” Myocarditis OR perimyocarditis OR endomyocarditis OR endoperimyocarditis). Initially, 1588 papers were identified. After the automatic exclusion of non-English articles and duplicate removal, 695 papers were assessed and eventually, 17 articles were included in the analysis. Studies which one researcher positively qualified were left to the decision of the third investigator (M.F.). Further, reference lists, papers which cited selected articles and two similar systematic reviews [8,9] were screened for relevant articles, which resulted in identifying 1037 and eventually including 57 papers (Figure 1). After the thorough full-text assessment, 59 studies and 70 patients were included in the analysis.
Figure 1.
Flow chart of the systematic literature search according to PRISMA guidelines.
2.2. Eligibility Criteria
Inclusion criteria were: full-text peer-reviewed articles in English, case-report formula, age of patient ≥18 years, myocarditis after the COVID infection, laboratory-confirmed COVID infection (including COVID in medical history), diagnosis of myocarditis by ESC criteria [6]. Exclusion criteria were: incident of myocarditis after the vaccination, unsure relation of myocarditis with COVID (e.g., positive serological results of different viruses), and unfulfilling ESC criteria.
2.3. Data Extraction and Studies Quality Assessment
Similarly to the study selection, data were extracted independently by two researchers (GI and MJ) and discrepancies were solved by the third (SU). Data regarding demographics and medical history, COVID presentation, applied diagnostics, treatment methods and outcome were collected (Table 1 and Table 2). We have analyzed exclusively case-report studies, which are per se associated with a high risk of bias, given that we have not applied a standardized tool for the quality assessment.

Table 1.
Demographics, clinical characteristics and laboratory findings of studied patients. All descriptive parameters are obtained from the original papers.

Table 2.
Magnetic resonance, biopsy, complications and outcomes of studied patients. All descriptive parameters are obtained from the original papers.
3. Results
After the full-text assessment, 59 studies and 70 patients were included in the analysis. All the counted percentages were calculated from the number of patients who had reported specific parameters.
3.1. Demographics
Men constituted 50 out of 69 (73%) cases which reported sex. Age was reported in every case; the mean age was 44 (SD 15.67). Only in 1 reported case patient was vaccinated; however, the vaccination scheme was not fulfilled—the patient received one dose of an unspecified vaccine 9 days before admission. Prior medical history and comorbidity status were reported in 46 cases—15 patients (33%) had no previous significant medical history. The most frequent comorbidities were hypertension (n = 16, 35%), obesity (n = 5, 11%), dyslipidaemia (n = 4, 9%) and asthma (n = 3, 7%).
3.2. Clinical Characteristics
Symptoms were reported in 68 patients. Most frequent symptoms were dyspnoea (n = 36, 53%), cough (n = 22, 32%), chest pain/discomfort (n = 19, 28%), diarrhoea (n = 11, 16%) and weakness/fatigue (n = 10, 15%).
Fever status was reported in 43 cases, and the exact value of temperature was reported in 39 cases, mean reported temperature was 38 C (SD 1.42) and 18 patients (42%) were febrile (over 38 C). Heart rate (HR) was reported in 46 cases, and the exact value was shown in 44 cases. The mean HR value was 112 beats per minute (SD 26.5), and tachycardia (over 100) was reported in 33 patients (75%). Blood pressure was described in 44 patients, and the exact value was shown in 40 cases. Mean systolic blood pressure was 105 mmHg (SD 26.17), and diastolic was 66 mmHg (SD 16.83). Hypotension (mean arterial pressure below 65 mmHg) occurred in 15 patients (34%). Oxygen saturation without oxygen supply was reported in 37 cases; the mean value was 90% (SD 8.06), and 21 (57%) patients were considered to have low saturation (below 95%).
3.3. Time of COVID-Related Myocarditis
The time relationship between COVID and myocarditis, i.e., myocarditis concurrent with the infection or after a specific period, was reported in 69 cases. In 46 patients (67%), myocarditis occurred during the infection. In the remaining 23 patients (33%), myocarditis followed infection. The time between infection and myocarditis was reported in 20 cases (87% of cases of myocarditis after COVID), and the mean time was 52 days (SD 49.13).
3.4. Laboratory Markers
Method of confirmation of COVID infection was declared in 56 patients. Polymerase chain reaction (PCR) showed the virus presence in 45 cases (80%), COVID was diagnosed based on serology results in 5 patients (9%), biopsy analysis revealed infection in 4 patients (7%), antigen test was performed in 2 cases (4%).
The status of inflammatory markers was reported in 62 patients The reported markers included: C-reactive protein (CRP) (reported in 58 cases, elevated in 54 (93%)); (white blood count (43 cases, elevated in 25 cases (58%)); procalcitonin (20 cases, elevated in 19 cases (95%)); ferritin (18 cases, elevated in 15 (83%)); interleukin-6 (13 cases, elevated in 100%); erythrocyte sedimentation rate (4 cases, elevated in 100%) and fibrinogen (reported and elevated in 1 case).
Most frequently reported cardiac biomarkers included: troponin (reported in 69 cases, elevated in 66 (96%)), brain natriuretic peptide (BNP) or N-terminal BNP (NT-proBNP) (reported in 49 cases, elevated in 47 (96%)) and DDimer (reported in 32 cases, elevated in 29 (91%)).
All the values are from the earliest assessment during patients a hospital stay. We decided not to provide mean or median values of the reported markers due to different normal range values and laboratory assessment methods.
3.5. ECG
Electrocardiography results were reported in 58 cases. No pathological changes were reported in 3 patients (5%). Most frequent changes were: ST wave abnormalities (n = 22, 38%), sinus tachycardia (n = 22, 38%), T wave abnormalities (n = 12, 21%), low QRS voltage or low QRS progression (n = 10, 17%), ectopic rhythm origin—including atrial fibrillation (n = 7, 12%). Ventricular tachycardia occurred in 4 patients (7%).
3.6. Imaging Diagnostics
3.6.1. X-ray
Chest X-ray findings have been reported in 30 cases (43%). The radiograph description of the heart has been mentioned in 3 cases, out of which 3 demonstrated cardiomegaly as a cardiac abnormality. Changes in lungs, however, have been reported in 19 case reports (27%), primarily multifocal opacities suggestive of viral pneumonia.
3.6.2. CA
The result of coronary angiography (CA) has been reported in 23 cases (33%). None of them revealed any relevant coronary artery stenosis.
3.6.3. CT
Computed Tomography (CT) has been performed in 42 cases (60%). Changes in heart have been stated in 11 patients (26%), out of which pericardial effusion was described in 6 cases and cardiomegaly in 4 patients. The changes in lungs have been observed in 31 cases (74%), mostly bilateral ground-glass opacities and pleural effusion characteristic of viral pneumonia. 1 case report mentioned evidence of pulmonary embolism in CT findings.
3.6.4. MRI
The findings observed in Magnetic Resonance Imaging (MRI) have been reported in 33 cases (46%). The number of cases fulfilling Lake Louis criteria constituted 22 cases (67%), whilst 11 cases (33%) did not meet Lake Luis criteria. Nonischemic myocardial injury has been observed in 25 cases (76%). Myocardial oedema was confirmed in 17 cases (52%), systolic dysfunction in 11 cases (33%), hypokinesia in 7 cases (21%) and pericardial effusion in 5 of them (15%).
3.6.5. ECHO
The echocardiography results have been mentioned in 66 out of 70 cases (94%). Systolic dysfunction has been described in 55 patients (83%), among which 13 (24%) cases reported on biventricular. The Left ventricular ejection fraction (LVEF) has been measured in 46 cases, and the average LVEF turned out to be 28%. Heart wall hypokinesia or akinesia was reported in 24 cases (36%). The observation of pericardial effusion has been described in 19 cases (29%), heart hypertrophy in 13 cases (20%) and mitral or tricuspidal regurgitations in 6 cases (9%).
3.7. Biopsy
Myocardial biopsy has been performed in 23 out of 70 cases (33%). The Dallas Criteria for myocarditis have been fulfilled in 21 (91%). The most commonly reported changes were diffuse lymphocytes infiltration which was found in 21 cases (91%), myocyte necrosis, mentioned in 5 cases (21%) and oedema in 4 described cases (17%).
3.8. ESC Criteria
The sufficient information for establishing fulfilment of criteria of clinically suspected myocarditis, according to the European Society of Cardiology (ESC) guidelines [6], has been provided in 68 out of 70 cases (97%). The cases fulfilling only one criterium constituted 3 out of 68 cases (4%). The fulfilment of two criteria has been observed in 29 out of 68 cases (43%), three of them in 25 out of 68 cases (37%) and four of them were met in 11 out of 68 cases (16%). The mean number of fulfilled criteria was 3.
The first criterium, I. ECG/Holter/stress test features was fulfilled in 34 out of 68 cases (50%), the second criterium, II. Myocardiocytolysis markers was fulfilled in 64 out of 68 cases (94%), the third criterium, III. Functional and structural abnormalities on imaging (echo/angiography/CMR) was fulfilled in 58 out of 68 cases (85%) and the fourth one, IV. Tissue characterization by CMR was met in 24 out of 68 cases 35%).
3.9. Treatment
3.9.1. Pharmacotherapy
The analysis of treatment implemented in the care of described patients revealed the applied pharmacotherapy in 65 out of 70 cases (92%). Antibiotics were used in 43 out 65 cases (66%), among which azithromycin in 14, piperacillin/tazobactam in 10, meropenem in 8, vancomycin in 7, doxycycline in 7, colchicine in 7 and ceftriaxone in 6 cases. Steroids have been used in 42 cases (65%), with methylprednisone being most often prescribed—in 24 cases. Vasopressors were used in 24 out of 65 cases (37%). Anti-viral drugs were applied in 18 out of 65 cases (28%), among which remdesivir in 9 and Lopinavir + Ritonavir in 5 cases. Inotropics were used in 16 out of 65 cases (25%), among which dobutamine was prescribed in 10 cases. Furtherly, anti-hypertensive drugs in 14 cases, hydroxychloroquine in 14 cases, IVIG in 14 cases, diuretics in 11 cases, anticoagulants in 11 cases, antiplatelet therapy in 10 cases, tocilizumab in 5 cases, NSAIDs in 4 cases and PPIs in 4 cases.
3.9.2. Procedures
The information about medical procedures performed in the care of described patients was mentioned in 34 out of 70 cases (49%). Mechanical ventilation has been applied in 14 patients (41%), and breathing support, meaning CPAP, high-flow or just oxygen, in 8 cases (24%). Extracorporeal membrane oxygenation (ECMO) was used in 16 patients (47%), and Continuous Renal Replacement Therapy (CRRT) in 7 patients (21%). Cardiac devices (ex. Impella CP) have been implemented in 10 cases (29%). The performance of pericardiocentesis or pericardiotomy has been mentioned in 6 cases (18%). Resuscitation was performed in 3 out of 34 cases (9%) and heart transplant in 1 out of 34 patients (3%).
3.10. Complications
Adverse events complicating the course of illness have been described in 38 out of 70 cases (54%). The cardiogenic shock occurred in 15 out of 38 cases (40%), acute respiratory distress syndrome (ARDS) in 12 (32%), heart failure in 5 (13%), acute kidney injury (AKI) in 5 (13%), mixed distributive and cardiogenic shock in 4 (11%), septic shock in 3 (8%). Other complications mentioned included: disseminated intravascular coagulation (DIC), lower limb ischemia, lower limb myositis, facial nerve palsy and pulmonary infection.
3.11. Outcome
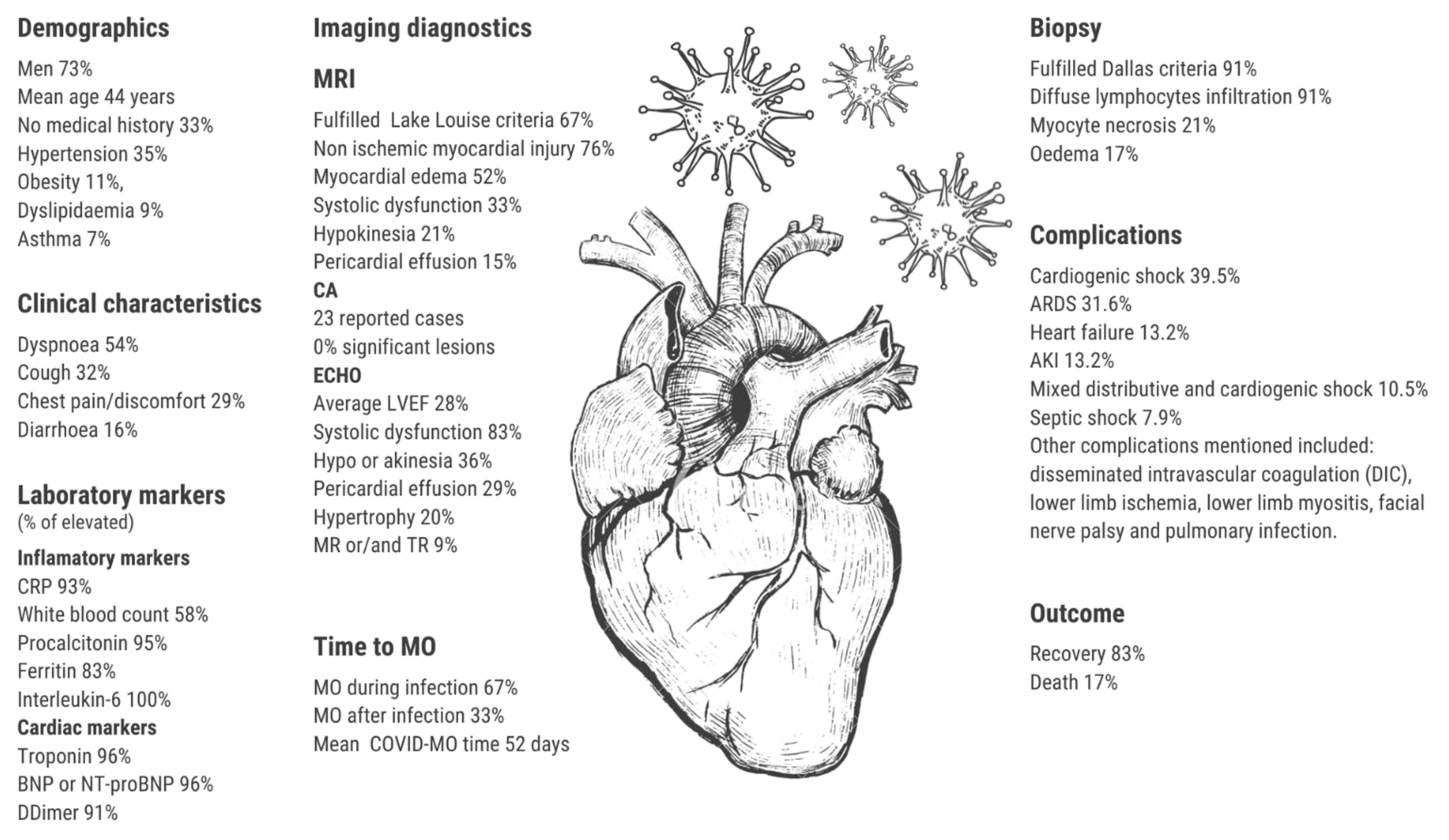
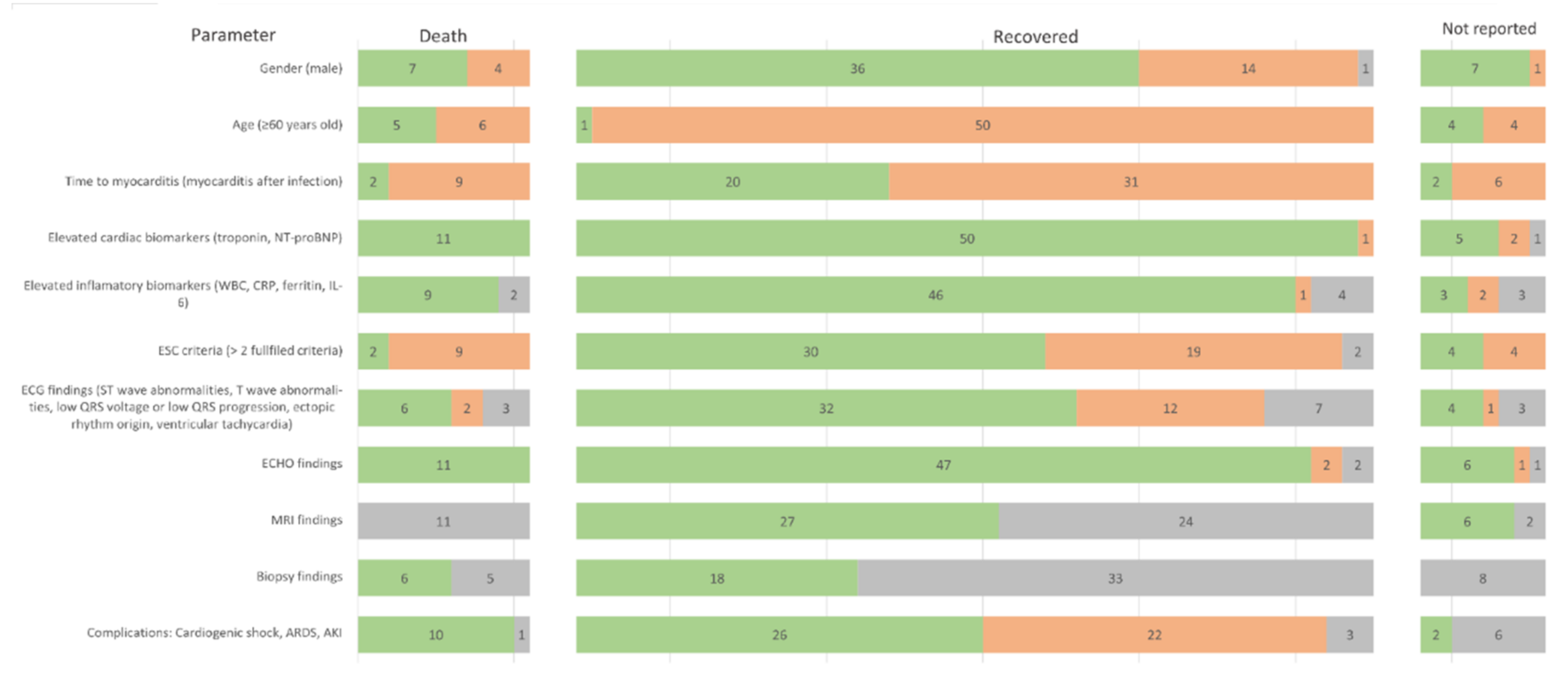
The final outcome has been reported in 63 out of 70 cases (90%). In 53 cases (84%), recovery has been achieved. The number of days till the patient’s discharge was stated in 22 out of 53 cases (42%), with the average number of 16 days and the range from 3 to 52 days. Death was the outcome in 11 out of 63 cases (17%). The number of days from admission to the hospital till death has been given in 7 out of 11 cases (64%), with the average number of 9 days with the range from <1 day to 33 days. Most important features are shown in Figure 2. Clinical features by outcome groups are summarized in the harvest plot (Figure 3).
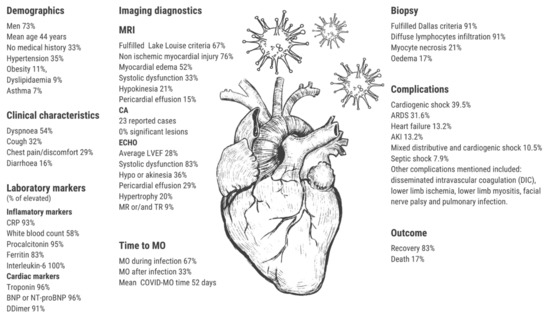
Figure 2.
The central illustration of the findings. Abbreviations: MO—myocarditis, CRP—c-reactive protein, NT-proBNP—N-terminal brain natriuretic peptide, ARDS—acute respiratory distress syndrome, AKI—acute kidney injury, MRI—magnetic resonance imaging., CA—coronary angiography, ECHO—chocardiography.
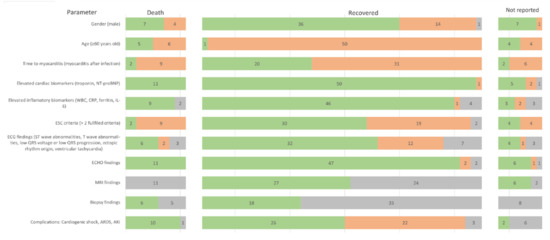
Figure 3.
Harvest plot summarising the most important features of the studied patients by groups of outcomes. Green colour indicates the presence of the feature, orange its absence, and grey a missing value. The numbers on the bars show the number of patients in each group. Abbreviations: NT-proBNP—N-terminal brain natriuretic peptide, WBC—white blood count, CRP—c-reactive protein, IL-6—interleukin 6, ESC—European Society of Cardiology, ECG -electrocardiogram, ECHO—echocardiography, MRI- magnetic resonance imaging, ARDS—acute respiratory distress syndrome, AKI—acute kidney injury.
A summary of the most important findings is shown in Table 1 and Table 2 and displayed in Figure 1. The full report of reviewed cases is in Supplementary Materials (Supplementary Table S1).
4. Discussion
4.1. Clinical Characteristics of Patients
Reported patients were predominantly men (73%). Myocarditis occurred in relatively young patients (mean age 44 years). Notably, 33% of patients had no previous medical history. Given that, the phenotype of the myocarditis patient—a mid-aged man with no or a few comorbidities, does not suggest severe illness and can be falsely comforting for a physician. In 33% of cases, myocarditis occurred sometime after the COVID infection. Center for Disease Control and Prevention reports that myocarditis can be diagnosed several months after the infection. In the observed cohort of 36,005,294 patients, 89.6% were diagnosed with myocarditis in the same month as COVID infection, 6.6% 1 month after, and 3.9% ≥ 2 months after [69]. In our case, the mean time between infection and myocardium affection was 52 days. However, one case occurred after 6 months after the initial disease. This highlights the necessity of watchful surveillance of susceptible patients after the COVID, even up to half a year. Most of the published case reports describe patients who experienced myocarditis when the vaccines were not yet available. Only one patient was vaccinated, however, he did not finish the complete vaccination scheme. Further, some studies showed an increased incidence of myocarditis after COVID vaccines [70,71]. A recent analysis performed by the Center for Disease Control and Prevention, which included 40 health care systems, revealed that the risk of myocarditis after vaccination is lower than the risk of myocarditis after COVID infection regardless of sex and age group [72]. The impact of the vaccination on the incidence and severity of myocarditis after COVID infection—despite prior immunization—needs further cohort studies.
4.2. Clinical Presentation
Some of the most reported symptoms were dyspnoea, cough, chest pain and reported weakness. These are the typical myocarditis manifestations, and as they overlap the symptomatology of the COVID infection, making an appropriate diagnosis can be challenging [73]. Noteworthy, we observed a relatively high occurrence of diarrhoea (16%). In every, except one, cases diarrhoea accompanied myocarditis at the time of COVID. Given that, it can be associated rather with the infection than the myocarditis [74].
Inflammatory markers, including white blood count, CRP, procalcitonin, ferritin, and interleukin-6, were typically elevated. The precise pathophysiology of COVID-related myocarditis remains unsolved, and a cytokine storm remains one of the hypotheses [75]. Significant elevation of inflammatory markers (CRP, ferritin, DDimer) were also associated with a more aggressive course [8,76].
Noteworthy, cytokine storm as a reflection of the hyperactive immune response is not the only proposed hypothesis for explaining myocardial injury by COVID-19. COVID molecules invade i.a. heart cells via the protein receptor angiotensin-converting enzyme 2 (ACE2). ACE2 is also associated with the modulation of the myocardial inflammatory response. The affected heart cells were noted to have a higher expression of the ACE2 receptors [77,78]. Given that, the role of the ACE2 receptors in COVID-related myocarditis pathophysiology seems to be significant.
Cardiac markers, such as troponin or NT-proBNP, were elevated in most cases. The rise of both biomarkers in COVID infection was associated with a poor prognosis. Notably, the prognosis of the patients with preceding cardiovascular disease remained favorable [79]. The high troponin level was also associated with the arrhythmia prevelance [80]. Cardiac biomarkers, especially troponin, should be analyzed in the context of other laboratory results. While the isolated rise of troponin reflects the isolated cardiac involvement, the rise of troponin with the surrounding intensive rise of inflammatory biomarkers reflects the hyperinflammatory state with possible multiorgan dysfunciton [8]. This distinction has important clinical implications, as physicians should always recognize which therapeutical pathway would be more rewarding, i.e., targeting inflammation by using steroids, immunoglobulins, biological treatment (e.g., anakinra, tocilizumab, sarilumab, canakinumab, JAK inhibition) or focusing on the cardioprotective treatment [81,82,83,84,85].
Described electrocardiographic findings present a wide range of changes and are not characteristic of myocarditis. On the other hand, physiological ECG was reported only in 5% of cases, suggesting that the ECG can successfully serve as a screening tool for myocarditis.
4.3. Imaging Diagnostics
Although endomyocardial biopsy (EMB) has lost its popularity [86], it remains the gold standard in diagnosing myocarditis. As it is an invasive procedure, it requires professional training and has complications, but when done correctly, it has invaluable diagnostic and prognostic value [87,88]. The EMB was performed in 33% of all reviewed cases. Lymphocytic infiltrates associated with myocyte necrosis, which stands for Dallas Criteria, were present in 91% of cases (n = 21). With the evolution of immunohistochemistry technics, more detailed analysis allows detecting more myocarditis cases and overcoming the limitations present in the traditional sample examination [89].
The deterioration of left ventricular systolic function was present in 83% of the described patients. This finding remains consistent with other systematic reviews: Jaisiwal et al.—74% [73], Ho et al.—66% [9]. Similarly to what has been shown in previous systematic review performed by Castiello T. et al. [8], one of the most common echocardiographic findings was global and regional wall hypokinesia 36.4% (n = 24), suggestive of myocardial oedema. Even though there are no echocardiographic changes unique to myocarditis, the assessment of echocardiographic parameters is recommended prior to the EMB procedure. It allows excluding pericardial effusion or intracavitary thrombus, as well as it could help to exclude other causes of heart failure and may have a prognostic value [89]. Moreover, the initial ECHO diagnostic is also an excellent parameter to be controlled in follow-up [90].
Cardiac magnetic resonance is considered a noninvasive gold standard for diagnosing myocarditis [91]. Late gadolinum enhancement, indicating the most inflamed areas, was present in 66,7% (n = 22) of patients. It is also a prognostic marker for the increased risk of adverse cardiac outcomes in patients with myocarditis [92]. Myocardial oedema, which was present in 51% (n = 17) of reviewed cases, may affect myocardial function. It can be an expression of diffuse inflammation due to systematic response, vascular leakage induced by endothelial barrier dysfunction or direct myocardial damage caused by SARS-CoV-2 [93,94].
The most often quoted abnormal finding in X-Ray was cardiomegaly (25%, n = 9) which can be a non-specific manifestation of various primary or acquired cardiomiopathies [95]. The most common X-Ray findings were opacities suggesting COVID-19 pneumonia (61% n = 19). Normal X-Ray findings were described in 12% (n = 4). The 22 patients (31%) who underwent CA were predominantly men (n = 16), and their age ranged from 19 to 69, with a median of 42.5 years. Although CA is not a standard procedure in the diagnostics of myocarditis, the patients presenting acute retrosternal pain and dyspnoea at a relatively young age raise the alarm for myocardial infarct procedures [96].
4.4. Treatment
The pharmacotherapy strategies varied among the described patients, depending on comorbidities and centre-specific procedures. It is impossible to enucleate specific myocarditis treatment, as the strategy should focus first on the disease source and unique patient’s symptomatic [89].
One of the main targets for the pharmacotherapy of COVID-19 myocarditis was cytokine storm. Incorporating the steroids (65% of our cases) into the COVID treatment was an attempt to address this pathological pathway. Indeed, the RECOVERY platform trial has proven that steroids play an important role in reducing COVID-19 mortality [97]. Dexamethasone was also found to diminish myocardial oedema and improve systolic function [93]. Another drug considered to dump patients’ immune response was tocilizumab (5 reviewed cases), which was shown to reduce mortality even in the group already treated with steroids [84,85,98]. Intravenous immunoglobulins were used in 14 patients, yet their use to modulate inflammation in COVID is still under debate [99,100]. Other conceptions for the immunomodulatory treatment in COVID tested interleukin-1β inhibitor—canakinumab; and colchicine. These conceptions did not show clinical or prognostic benefit in COVID setting [83,101].
Treatment with non-steroidal anti-inflammatory drugs (NSAIDs) in myopericarditis is another unsolved problem. There are studies supporting the hypothesis that it can be potentially harmful and should be avoided in myocarditis [102], as well as studies where NSAIDs neither affected all-cause mortality rate nor left ventricle function [103]. Nevertheless, none of the studies showed the benefit of NSAIDs in myocarditis treatment, and generally, they should not be widely implemented [6].
As ARDS was the most common complication, antibiotics were used, i.a., to prevent bacterial superinfection. The most willingly used antibiotic was azithromycin [9,73,104]. It seems that using antibiotics in standard COVID care is unjustified [105]. The anti-viral treatment was applied in 27% of the reviewed cases. None of the tested antiviral agents has proven its efficacy in improving COVID survival [106,107,108]. Further, there is a lack of evidence about antiviral treatment for myocarditis in general [89,109]. Thus, the symptomatic treatment according to NYHA functional class (with beta-blockers, diuretics, angiotensin-converting enzyme inhibitors or angiotensin-II receptor blockers) should be applied [6,89].
Surgical procedures like mechanical circulatory support or heart transplant are warranted particularly for patients with cardiogenic shock in the course of fulminant myocarditis, whose clinical state is deteriorating despite optimal medical treatment. Similarly, cardiac devices implementation should be considered in patients with myocarditis-related atrioventricular blocks or after a myocardial infarct. These technics should be considered early, when the highest possible pharmaceutics doses are insufficient to facilitate recovery [6,89].
4.5. Prognosis and Outcome
The overall incidence of combined in-hospital mortality and heart transplantations associated with myocarditis, presented in 2018 in the study from Multicenter Lombardy Registry, was 3.2% [110], much lower than in our work (17% of deaths). Higher mortality was also reported in the corresponding systematic reviews [8,9,73,104], ranging from 15.2% to 31.8%. It can be explained by the specific course of COVID-related myocarditis, which is overlapped by the aggressive, infectious disease. Noteworthy, case reports are the low-quality evidence and reported cases are usually severe and complicated, which may artificially increase the reported mortality. Given that, forthcoming registries are warranted to establish reliable COVID-related myocarditis mortality.
4.6. Limitations
Our study is not free from limitations. The most important one is that the data for our analysis come from the case-report papers, which provide poor quality evidence. Further, most of the reported cases demonstrate very dramatic situations—patients with severe and sophisticated course and complications—which is attractive for the publication reasons but does not reflect the typical clinical situation. Given that, the generalizability of our conclusions is limited, especially in epidemiology. Conversely, presented descriptions give a detailed, patient-level evaluation of each case. Submitted data was frequently incomplete—not every parameter was regularly described. We tried to choose and analyze only the most repetitive factors. Laboratory parameters were unsuitable for statistical analysis, as laboratories use different evaluation techniques and normal range values. Considering it all, quality or bias assessments could not be performed. Our study provides a current, interim summary of the data, while the results of more extensive, well-designed studies are unavailable.
5. Conclusions
This systematic review provides the most recent summary of the reported COVID-related myocarditis. We present the complex summary of COVID-related myocarditis, which can serve as a reliable source of knowledge on that topic. Importantly, in approximately one-third of patients, myocarditis occurs a few weeks after infection, even up to 6 months. The necessity of considering COVID as a cause of myocarditis in relatively young and healthy populations should be strongly highlighted. Susceptible patients should be carefully observed after the COVID infection, even the poorly symptomatic one.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jcm11195519/s1. Table S1.
Author Contributions
Conceptualization S.U., M.B., M.J., G.I., M.F., K.F., M.G. (Mateusz Guzik), M.G. (Mateusz Garus) and P.G.; methodology, S.U., M.F., K.F., G.I., M.B. and M.J.; software, S.U., M.F., G.I., M.B. and Ł.L.; validation K.F., M.J. and M.G. (Mateusz Guzik), M.G. (Mateusz Garus) and P.G.; formal analysis S.U., M.B., M.J., G.I., M.F., K.F., M.G. (Mateusz Guzik), M.G. (Mateusz Garus) and P.G.; investigation, S.U., M.B., M.J., G.I., M.F., K.F., M.G. (Mateusz Garus), M.G. (Mateusz Guzik), P.G. and Ł.L.; resources, S.U., M.F., G.I. and M.B.; data curation, S.U., M.J., K.F., M.F., M.B. and G.I.; writing—original draft preparation, S.U., M.B., M.J., G.I., M.F., K.F., M.G. (Mateusz Guzik), M.G. (Mateusz Garus) and P.G.; writing—review and editing, R.Z., J.B., P.P., A.T. and P.T.; visualization, S.U. and G.I.; Supervision, R.Z., J.B., P.P., A.T. and P.T.; project administration, S.U. and R.Z.; funding acquisition, J.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Wroclaw Medical University, Institute of Heart Diseases. Grant nuM.B.er S.U.BZ.A460.22.055.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Full report of screened papers is available in Table S1.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Coronavirus Death Toll and Trends—Worldometer, (n.d.). Available online: https://www.worldometers.info/coronavirus/coronavirus-death-toll/ (accessed on 9 August 2022).
- Ahmad, I.; Rathore, F.A. Neurological manifestations and complications of COVID-19: A literature review. J. Clin. Neurosci. 2020, 77, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Sarfraz, Z.; Sarfraz, A.; Barrios, A.; Garimella, R.; Dominari, A.; KC, M.; Pandav, K.; Pantoja, J.C.; Retnakumar, V.; Cherrez-Ojeda, I. Cardio-Pulmonary Sequelae in Recovered COVID-19 Patients: Considerations for Primary Care. J. Prim. Care Community Health 2021, 12, 21501327211023726. [Google Scholar] [CrossRef] [PubMed]
- Sokolski, M.; Gajewski, P.; Zymliński, R.; Biegus, J.; Berg, J.M.T.; Bor, W.; Braunschweig, F.; Caldeira, D.; Cuculi, F.; D’Elia, E.; et al. Impact of Coronavirus Disease 2019 (COVID-19) Outbreak on Acute Admissions at the Emergency and Cardiology Departments Across Europe. Am. J. Med. 2021, 134, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Bielecka-Dabrowa, A.; Cichocka-Radwan, A.; Lewek, J.; Pawliczak, F.; Maciejewski, M.; Banach, M. Cardiac manifestations of COVID-19. Rev. Cardiovasc. Med. 2021, 22, 365–371. [Google Scholar] [CrossRef]
- Caforio, A.L.P.; Pankuweit, S.; Arbustini, E.; Basso, C.; Gimeno-Blanes, J.; Felix, S.B.; Fu, M.; Heliö, T.; Heymans, S.; Jahns, R.; et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases, (n.d.). Eur. Heart J. 2013, 34, 2636–2648. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Altman, D.; Antes, G.; Atkins, D.; Barbour, V.; Barrowman, N.; Berlin, J.A.; et al. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Castiello, T.; Georgiopoulos, G.; Finocchiaro, G.; Claudia, M.; Gianatti, A.; Delialis, D.; Aimo, A.; Prasad, S. COVID-19 and myocarditis: A systematic review and overview of current challenges. Heart Fail. Rev. 2022, 27, 251–261. [Google Scholar] [CrossRef]
- Ho, J.S.; Sia, C.H.; Chan, M.Y.; Lin, W.; Wong, R.C. Coronavirus-induced myocarditis: A meta-summary of cases. Heart Lung 2020, 49, 681–685. [Google Scholar] [CrossRef]
- Doyen, D.; Moceri, P.; Ducreux, D.; Dellamonica, J. Myocarditis in a patient with COVID-19: A cause of raised troponin and ECG changes. Lancet 2020, 395, 1516. [Google Scholar] [CrossRef]
- Luetkens, J.A.; Isaak, A.; Zimmer, S.; Nattermann, J.; Sprinkart, A.M.; Boesecke, C.; Rieke, G.J.; Zachoval, C.; Heine, A.; Velten, M.; et al. Diffuse Myocardial Inflammation in COVID-19 Associated Myocarditis Detected by Multiparametric Cardiac Magnetic Resonance Imaging. Circ. Cardiovasc. Imaging 2020, 13, 10897. [Google Scholar] [CrossRef]
- Kim, I.C.; Kim, J.Y.; Kim, H.A.; Han, S. COVID-19-related myocarditis in a 21-year-old female patient. Eur. Heart J. 2020, 41, 1859. [Google Scholar] [CrossRef]
- Sala, S.; Peretto, G.; Gramegna, M.; Palmisano, A.; Villatore, A.; Vignale, D.; de Cobelli, F.; Tresoldi, M.; Cappelletti, A.M.; Basso, C.; et al. Acute myocarditis presenting as a reverse Tako-Tsubo syndrome in a patient with SARS-CoV-2 respiratory infection. Eur. Heart J. 2020, 41, 1861–1862. [Google Scholar] [CrossRef]
- Bonnet, M.; Champagnac, A.; Lantelme, P.; Harbaoui, B. Endomyocardial biopsy findings in Kawasaki-like disease associated with SARS-CoV-2. Eur. Heart J. 2020, 41, 3863–3864. [Google Scholar] [CrossRef]
- Ishikura, H.; Maruyama, J.; Hoshino, K.; Matsuoka, Y.; Yano, M.; Arimura, T.; Katano, H.; Kato, S.; Kitamura, T.; Nakamura, Y. Coronavirus disease (COVID-19) associated delayed-onset fulminant myocarditis in patient with a history of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. J. Infect. Chemother. 2021, 27, 1760–1764. [Google Scholar] [CrossRef]
- Meyer, M.; Vogel, T.; Meyer, A.; Constancias, F.; Porter, L.F.; Kaltenbach, G.; Schmitt, E.; Chayer, S.; Zeyons, F.; Riou, M.; et al. Presence of active myocarditis at the 6 month follow-up appointment for a severe form of COVID-19: A case report. ESC Heart Fail. 2021, 8, 4307–4312. [Google Scholar] [CrossRef]
- Roest, S.; Brugts, J.J.; van Kampen, J.J.A.; von der Thüsen, J.H.; Constantinescu, A.A.; Caliskan, K.; Hirsch, A.; Manintveld, O.C. COVID-19-related myocarditis post-heart transplantation. Int. J. Infect. Dis. 2021, 107, 34–36. [Google Scholar] [CrossRef]
- Inciardi, R.M.; Lupi, L.; Zaccone, G.; Italia, L.; Raffo, M.; Tomasoni, D.; Cani, D.S.; Cerini, M.; Farina, D.; Gavazzi, E.; et al. Cardiac Involvement in a Patient with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 819–824. [Google Scholar] [CrossRef]
- Hu, H.; Ma, F.; Wei, X.; Fang, Y. Coronavirus fulminant myocarditis treated with glucocorticoid and human immunoglobulin. Eur. Heart J. 2021, 42, 206. [Google Scholar] [CrossRef]
- Zeng, J.H.; Liu, Y.X.; Yuan, J.; Wang, F.X.; Wu, W.B.; Li, J.X.; Wang, L.F.; Gao, H.; Wang, Y.; Dong, C.F.; et al. First case of COVID-19 complicated with fulminant myocarditis: A case report and insights. Infection 2020, 48, 773–777. [Google Scholar] [CrossRef]
- Aldeghaither, S.; Qutob, R.; Assanangkornchai, N.; Issa-Chergui, B.; Tam, M.; Larotondo, R.; Samoukovic, G. Clinical and Histopathologic Features of Myocarditis in Multisystem Inflammatory Syndrome (Adult)-Associated COVID-19. Crit. Care Explor. 2022, 10, e0630. [Google Scholar] [CrossRef]
- Amin, A.; Eftekhar, S.P.; Ziaie, N.; Roudbari, S.; Salehi, P.; Jalali, F.; Jafaripour, I.; Ghaffari, S.; Salehi, M.M.; Ebadi, R. Clinically suspected myocarditis in COVID-19 patients: Case series and review of the literature. Clin. Case Rep. 2021, 9, e05236. [Google Scholar] [CrossRef]
- Menter, T.; Cueni, N.; Gebhard, E.C.; Tzankov, A. Case Report: Co-occurrence of Myocarditis and Thrombotic Microangiopathy Limited to the Heart in a COVID-19 Patient. Front. Cardiovasc. Med. 2021, 8, 695010. [Google Scholar] [CrossRef]
- Ashok, V.; Loke, W.I.; Dinov, B.; Robert, S.; David, N.; Camm, C.F.; Ross, T. Case report: High-grade atrioventricular block in suspected COVID-19 myocarditis. Eur. Heart J. Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Li, A.; Garcia-Bengochea, Y.; Stechel, R.; Azari, B.M.; Aboumarie, H.S.; Miglioranza, M.H.; Cameli, M.; Camm, C.F.; Thomson, R. Management of COVID-19 myopericarditis with reversal of cardiac dysfunction after blunting of cytokine storm: A case report. Eur. Heart J. Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Nakatani, S.; Ohta-Ogo, K.; Nishio, M.; Amemiya, K.; Sato, S.; Sawano, H.; Hatakeyama, K.; Katano, H.; Suzuki, T.; Hirooka, K. Microthrombosis as a cause of fulminant myocarditis-like presentation with COVID-19 proven by endomyocardial biopsy. Cardiovasc. Pathol. 2022, 60, 107435. [Google Scholar] [CrossRef]
- Ismayl, M.; Abusnina, W.; Thandra, A.; Sabri, A.; Groh, D.; Kanmanthareddy, A.; Alla, V.M. Delayed acute myocarditis with COVID-19 infection. Proc. Bayl. Univ. Med. Cent. 2022, 35, 366–368. [Google Scholar] [CrossRef]
- Thomson, A.; Totaro, R.; Cooper, W.; Dennis, M. Fulminant Delta COVID-19 myocarditis: A case report of fatal primary cardiac dysfunction. Eur. Heart J. Case Rep. 2022, 6, ytac142. [Google Scholar] [CrossRef]
- Campoamor, D.; Seixas, R.; Gama, L.; Duarte, J.; Araújo, T. Arrhythmogenic Cardiomyopathy as a Late Complication of COVID-19-Induced Myocarditis. Cureus 2022, 14, e21941. [Google Scholar] [CrossRef]
- Flagiello, M.; Al Harthy, A.; Boccalini, S.; Jacquemet, L.; Obadia, J.F.; Baudry, G.; Pozzi, M. Veno-arterial extracorporeal membrane oxygenation for COVID-19-associated acute myocardial injury complicated by refractory cardiogenic shock. J. Card. Surg. 2021, 36, 4396. [Google Scholar] [CrossRef]
- Pascariello, G.; Cimino, G.; Calvi, E.; Bernardi, N.; Grigolato, M.; Garyfallidis, P.; Fabbricatore, D.; Pezzola, E.; Lombardi, C.M.; Metra, M.; et al. Cardiogenic Shock due to COVID-19-Related Myocarditis in a 19-Year-Old Autistic Patient. J. Med. Cases 2020, 11, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Gaudriot, B.; Mansour, A.; Thibault, V.; Lederlin, M.; Cauchois, A.; Lelong, B.; Ross, J.T.; Leurent, G.; Tadié, J.M.; Revest, M.; et al. Successful heart transplantation for COVID-19-associated post-infectious fulminant myocarditis. ESC Heart Fail. 2021, 8, 2625–2630. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.Z.; Kumar, S.A.; Patel, A.A. Myocarditis and Pericarditis in Patients with COVID-19. Heart Views 2020, 21, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Shabbir, A.; Camm, C.F.; Elkington, A.; Tilling, L.; Stirrup, J.; Chan, A.; Bull, S. Myopericarditis and myositis in a patient with COVID-19: A case report. Eur. Heart J. Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Gauchotte, G.; Venard, V.; Segondy, M.; Cadoz, C.; Esposito-Fava, A.; Barraud, D.; Louis, G. SARS-CoV-2 fulminant myocarditis: An autopsy and histopathological case study. Int. J. Legal Med. 2021, 135, 577–581. [Google Scholar] [CrossRef]
- Bernal-Torres, W.; Herrera-Escandón, Á.; Hurtado-Rivera, M.; Plata-Mosquera, C.A.; Savarese, G.; David, S.; Ferrannini, G.; Camm, C.F. COVID-19 fulminant myocarditis: A case report. Eur. Heart J. Case Rep. 2020, 4, 1–6. [Google Scholar] [CrossRef]
- Oleszak, F.; Maryniak, A.; Botti, E.; Abrahim, C.; Salifu, M.O.; Youssef, M.; Henglein, V.L.; McFarlane, S.I. Myocarditis Associated with COVID-19. Am. J. Med. Case Rep. 2020, 8, 498. [Google Scholar] [CrossRef]
- Gay, H.C.; Sinha, A.; Michel, E.; Mozer, A.B.; Budd, A.; Feinstein, M.J.; Benzuly, K.H.; Al-Qamari, A.; Pawale, A.A.; Vorovich, E.E. Fulminant myocarditis in a patient with coronavirus disease 2019 and rapid myocardial recovery following treatment. ESC Heart Fail. 2020, 7, 4367–4370. [Google Scholar] [CrossRef]
- Gomila-Grange, A.; Espasa, M.; Moglia, E. Cardiogenic Shock Caused by SARS-CoV-2 in a Patient with Serial Negative Nucleic Acid Amplification Tests. Case Report. SN Compr. Clin. Med. 2020, 2, 1903–1905. [Google Scholar] [CrossRef]
- Richard, I.; Robinson, B.; Dawson, A.; Aya, A.; Ali, R. An Atypical Presentation of Fulminant Myocarditis Secondary to COVID-19 Infection. Cureus 2020, 12, e9179. [Google Scholar] [CrossRef]
- Wenzel, P.; Kopp, S.; Gobel, S.; Jansen, T.; Geyer, M.; Hahn, F.; Kreitner, K.F.; Escher, F.; Schultheiss, H.P.; Münzel, T. Evidence of SARS-CoV-2 mRNA in endomyocardial biopsies of patients with clinically suspected myocarditis tested negative for COVID-19 in nasopharyngeal swab. Cardiovasc. Res. 2020, 116, 1661–1663. [Google Scholar] [CrossRef]
- Bemtgen, X.; Klingel, K.; Hufnagel, M.; Janda, A.; Bode, C.; Staudacher, D.L.; Supady, A.; Jandova, I. Case Report: Lymphohistiocytic Myocarditis with Severe Cardiogenic Shock Requiring Mechanical Cardiocirculatory Support in Multisystem Inflammatory Syndrome Following SARS-CoV-2 Infection. Front. Cardiovasc. Med. 2021, 9, 1091. [Google Scholar] [CrossRef]
- Beşler, M.S.; Arslan, H. Acute myocarditis associated with COVID-19 infection. Am. J. Emerg. Med. 2020, 38, 2489.e1. [Google Scholar] [CrossRef]
- Meel, R.; Ramsamy, T.D.; Narsing, R.; Wong, M. Focal myocarditis in a young male with SARS-CoV-2 infection. Oxf. Med. Case Rep. 2021, 2021, 72–75. [Google Scholar] [CrossRef]
- Paul, J.F.; Charles, P.; Richaud, C.; Caussin, C.; Diakov, C. Myocarditis revealing COVID-19 infection in a young patient. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 776. [Google Scholar] [CrossRef]
- Coyle, J.; Igbinomwanhia, E.; Sanchez-Nadales, A.; Danciu, S.; Chu, C.; Shah, N. A Recovered Case of COVID-19 Myocarditis and ARDS Treated with Corticosteroids, Tocilizumab, and Experimental AT-001. JACC Case Rep. 2020, 2, 1331–1336. [Google Scholar] [CrossRef]
- Irabien-Ortiz, Á.; Carreras-Mora, J.; Sionis, A.; Pàmies, J.; Montiel, J.; Tauron, M. Fulminant myocarditis due to COVID-19. Rev. Española De Cardiol. 2020, 73, 503–504. [Google Scholar] [CrossRef]
- Frustaci, A.; Francone, M.; Verardo, R.; Capobianchi, M.R.; Chimenti, C. Virus-Negative Necrotizing Coronary Vasculitis with Aneurysm Formation in Human SARS-CoV-2 Infection. Infect. Dis. Rep. 2021, 13, 597–601. [Google Scholar] [CrossRef]
- Sardari, A.; Tabarsi, P.; Borhany, H.; Mohiaddin, R.; Houshmand, G. Myocarditis detected after COVID-19 recovery. Eur. Heart J. Cardiovasc. Imaging 2021, 22, 131–132. [Google Scholar] [CrossRef]
- Warchoł, I.; Dȩbska-Kozłowska, A.; Karcz-Socha, I.; Książczyk, M.; Szymańska, K.; Lubiński, A. Terra incognita: Clinically suspected myocarditis in a patient with severe acute respiratory syndrome coronavirus 2 infection. Pol. Arch. Intern. Med. 2020, 130, 446–448. [Google Scholar] [CrossRef]
- Shahrami, B.; Davoudi-Monfared, E.; Rezaie, Z.; Gheibi, S.; Vakili-Ardabili, A.; Arabzadeh, A.A.; Talebi, A.; Mojtahedzadeh, M. Management of a critically ill patient with COVID-19-related fulminant myocarditis: A case report. Respir. Med. Case Rep. 2022, 36, 101611. [Google Scholar] [CrossRef]
- Gaine, S.; Devitt, P.; Coughlan, J.J.; Pearson, I. COVID-19-associated myocarditis presenting as new-onset heart failure and atrial fibrillation. BMJ Case Rep. CP 2021, 14, e244027. [Google Scholar] [CrossRef]
- Taouihar, S.; Bouabdallaoui, A.; Aabdi, M.; Elaidouni, G.; Bkiyar, H.; Aichouni, N.; Smaili, N.; ElOuafi, N.; Skiker, I.; Housni, B. ST elevation revealing acute myocarditis with SARS-CoV-2 infection: Case report. Ann. Med. Surg. 2021, 65, 102313. [Google Scholar] [CrossRef]
- Dahl, E.H.; Mosevoll, K.A.; Cramariuc, D.; Vedeler, C.A.; Blomberg, B. COVID-19 myocarditis and postinfection Bell’s palsy. BMJ Case Rep. CP 2021, 14, e240095. [Google Scholar] [CrossRef]
- Matsumura, K.; Kawano, H.; Kurobe, M.; Akashi, R.; Yoshimuta, T.; Ikeda, S.; Ueki, N.; Nakashima, M.; Maemura, K. Delayed Acute Perimyocarditis and Bilateral Facial Nerve Palsy in a Patient with COVID-19. Intern. Med. 2022, 61, 2327–2332. [Google Scholar] [CrossRef]
- Van Praet, J.T.; Hoste, L.; Haerynck, F. Acute Perimyocarditis in a Case of Multisystem Inflammatory Syndrome in Adults. J. Rheumatol. 2022, 49, 538–539. [Google Scholar] [CrossRef]
- Masiak, A.; Lass, A.; Kowalski, J.; Hajduk, A.; Zdrojewski, Z. Self-limiting COVID-19-associated Kikuchi-Fujimoto disease with heart involvement: Case-based review. Rheumatol. Int. 2022, 42, 341–348. [Google Scholar] [CrossRef]
- Noori, M.A.M.; Rushdy, A.; Fichadiya, H.; Sherif, R.; Younes, I.; Lenchur, P.D.; Lenchur, P.M.; Shamoon, F. Stemi mimicking post COVID: A case of focal myopericarditis post COVID infection. J. Community Hosp. Intern. Med. Perspect. 2021, 11, 769. [Google Scholar] [CrossRef] [PubMed]
- Okor, I.; Sleem, A.; Zhang, A.; Kadakia, R.; Bob-Manuel, T.; Krim, S.R. Suspected COVID-19-Induced Myopericarditis. Ochsner J. 2021, 21, 181–186. [Google Scholar] [CrossRef]
- Bulbul, R.F.; Al Suwaidi, J.; Al-Hijji, M.; Al Tamimi, H.; Fawzi, I. COVID-19 Complicated by Acute Respiratory Distress Syndrome, Myocarditis, and Pulmonary Embolism. A Case Report. J. Crit. Care Med. (Univ. De Med. Si Farm. Din Targu Mures) 2021, 7, 123–129. [Google Scholar] [CrossRef]
- Khatri, A.; Wallach, F. Coronavirus disease 2019 (COVID-19) presenting as purulent fulminant myopericarditis and cardiac tamponade: A case report and literature review. Heart Lung 2020, 49, 858–863. [Google Scholar] [CrossRef] [PubMed]
- Nicol, M.; Cacoub, L.; Baudet, M.; Nahmani, Y.; Cacoub, P.; Cohen-Solal, A.; Henry, P.; Adle-Biassette, H.; Logeart, D. Delayed acute myocarditis and COVID-19-related multisystem inflammatory syndrome. ESC Heart Fail. 2020, 7, 4371–4376. [Google Scholar] [CrossRef] [PubMed]
- Hudowenz, O.; Klemm, P.; Lange, U.; Rolf, A.; Schultheiss, H.P.; Hamm, C.; Müller-Ladner, U.; Wegner, F. Case report of severe PCR-confirmed COVID-19 myocarditis in a European patient manifesting in mid January 2020. Eur. Heart J. Case Rep. 2020, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, W.; Lammens, M.; Kerckhofs, A.; Voets, E.; van San, E.; van Coillie, S.; Peleman, C.; Mergeay, M.; Sirimsi, S.; Matheeussen, V.; et al. Fatal lymphocytic cardiac damage in coronavirus disease 2019 (COVID-19): Autopsy reveals a ferroptosis signature. ESC Heart Fail. 2020, 7, 3772–3781. [Google Scholar] [CrossRef]
- Hussain, H.; Fadel, A.; Alwaeli, H.; Guardiola, V. Coronavirus (COVID-19) Fulminant Myopericarditis and Acute Respiratory Distress Syndrome (ARDS) in a Middle-Aged Male Patient. Cureus 2020, 12, e8808. [Google Scholar] [CrossRef]
- Rehman, M.; Gondal, A.; Rehman, N.U. Atypical Manifestation of COVID-19-Induced Myocarditis. Cureus 2020, 12, e8685. [Google Scholar] [CrossRef]
- Monmeneu, J.V.; Mafe, E.D.; Soler, J.A.; Perez, B.V.; Caravaca, J.S.; Torres, R.B.; García-Gonzalez, P.; Ortega, L.H.; Lopez-Lereu, M.P.; MacEira, A.M.; et al. Subacute perimyocarditis in a young patient with COVID-19 infection. Eur. Heart J. Case Rep. 2020, 4, 1–3. [Google Scholar] [CrossRef]
- Tavazzi, G.; Pellegrini, C.; Maurelli, M.; Belliato, M.; Sciutti, F.; Bottazzi, A.; Sepe, P.A.; Resasco, T.; Camporotondo, R.; Bruno, R.; et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur. J. Heart Fail. 2020, 22, 911–915. [Google Scholar] [CrossRef]
- Boehmer, T.K.; Kompaniyets, L.; Lavery, A.M.; Hsu, J.; Ko, J.Y.; Yusuf, H.; Harris, A.M. Association Between COVID-19 and Myocarditis Using Hospital-Based Administrative Data—United States, March 2020–January 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1228–1232. [Google Scholar] [CrossRef]
- Wong, H.L.; Hu, M.; Zhou, C.K.; Lloyd, P.C.; Amend, K.L.; Beachler, D.C.; Secora, A.; McMahill-Walraven, C.N.; Lu, Y.; Wu, Y.; et al. Risk of myocarditis and pericarditis after the COVID-19 mRNA vaccination in the USA: A cohort study in claims databases. Lancet 2022, 399, 2191–2199. [Google Scholar] [CrossRef]
- Le Vu, S.; Bertrand, M.; Jabagi, M.-J.; Botton, J.; Drouin, J.; Baricault, B.; Weill, A.; Dray-Spira, R.; Zureik, M. Age and sex-specific risks of myocarditis and pericarditis following COVID-19 messenger RNA vaccines. Nat. Commun. 2022, 13, 1–9. [Google Scholar] [CrossRef]
- Block, J.P.; Boehmer, T.K.; Forrest, C.B.; Carton, T.W.; Lee, G.M.; Ajani, U.A.; Christakis, D.A.; Cowell, L.G.; Draper, C.; Ghildayal, N.; et al. Cardiac Complications After SARS-CoV-2 Infection and mRNA COVID-19 Vaccination—PCORnet, United States, January 2021–January 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, V.; Sarfraz, Z.; Sarfraz, A.; Mukherjee, D.; Batra, N.; Hitawala, G.; Yaqoob, S.; Patel, A.; Agarwala, P.; Ruchika; et al. COVID-19 Infection and Myocarditis: A State-of-the-Art Systematic Review. J. Prim. Care Community Health 2021, 12, 21501327211056800. [Google Scholar] [CrossRef] [PubMed]
- Maslennikov, R.; Poluektova, E.; Ivashkin, V.; Svistunov, A. Diarrhoea in adults with coronavirus disease-beyond incidence and mortality: A systematic review and meta-analysis. Infect. Dis. 2021, 53, 348–360. [Google Scholar] [CrossRef]
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Oudit, G.Y.; Kassiri, Z.; Jiang, C.; Liu, P.P.; Poutanen, S.M.; Penninger, J.M.; Butany, J. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur. J. Clin. Investig. 2009, 39, 618–625. [Google Scholar] [CrossRef]
- Nicin, L.; Abplanalp, W.T.; Mellentin, H.; Kattih, B.; Tombor, L.; John, D.; Schmitto, j.; Heineke, J.; Emrich, F.; Arsalan, M.; et al. Cell type-specific expression of the putative SARS-CoV-2 receptor ACE2 in human hearts. Eur. Heart J. 2020, 41, 1804–1806. [Google Scholar] [CrossRef]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 811–818. [Google Scholar] [CrossRef]
- Rav-Acha, M.; Orlev, A.; Itzhaki, I.; Zimmerman, S.F.; Fteiha, B.; Bohm, D.; Kurd, R.; Samuel, T.Y.; Asher, E.; Helviz, Y.; et al. Cardiac arrhythmias amongst hospitalised Coronavirus 2019 (COVID-19) patients: Prevalence, characterisation, and clinical algorithm to classify arrhythmic risk. Int. J. Clin. Pract. 2021, 75, e13788. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Calabrese, L.H. Cytokine storm and the prospects for immunotherapy with COVID-19. Clevel. Clin. J. Med. 2020, 87, 389–393. [Google Scholar] [CrossRef]
- Cremer, P.C.; Sheng, C.C.; Sahoo, D.; Dugar, S.; Prada, R.A.; Wang, T.K.M.; Hassan, O.K.A.; Hernandez-Montfort, J.; Wolinsky, D.A.; Culver, D.A.; et al. Double-blind randomized proof-of-concept trial of canakinumab in patients with COVID-19 associated cardiac injury and heightened inflammation. Eur. Heart J. Open 2021, 1, oeab002. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. Targeting cytokine storm in COVID-19: What have we learned? Eur. Heart J. Open 2021, 1, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.C.; Angus, D.C.; Derde, L.P.G. Interleukin-6 Receptor Antagonists in Critically Ill Patients with COVID-19. N. Engl. J. Med. 2021, 385, 1147–1149. [Google Scholar] [CrossRef]
- Veinot, J.P. Diagnostic endomyocardial biopsy—still useful after all these years. Can. J. Cardiol. 2009, 25, e55. [Google Scholar] [CrossRef]
- Kindermann, I.; Kindermann, M.; Kandolf, R.; Klingel, K.; Bültmann, B.; Müller, T.; Lindinger, A.; Böhm, M. Predictors of outcome in patients with suspected myocarditis. Circulation 2008, 118, 639–648. [Google Scholar] [CrossRef]
- Leone, O.; Veinot, J.P.; Angelini, A.; Baandrup, U.T.; Basso, C.; Berry, G.; Bruneval, P.; Burke, M.; Butany, J.; Calabrese, F.; et al. 2011 consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovasc. Pathol. 2012, 21, 245–274. [Google Scholar] [CrossRef]
- Kindermann, I.; Barth, C.; Mahfoud, F.; Ukena, C.; Lenski, M.; Yilmaz, A.; Klingel, K.; Kandolf, R.; Sechtem, U.; Cooper, L.T.; et al. Update on myocarditis. J. Am. Coll. Cardiol. 2012, 59, 779–792. [Google Scholar] [CrossRef]
- Tymińska, A.; Ozierański, K.; Caforio, A.L.P.; Marcolongo, R.; Marchel, M.; Kapłon-Cieślicka, A.; Baritussio, A.; Filipiak, K.J.; Opolski, G.; Grabowski, M. Myocarditis and inflammatory cardiomyopathy in 2021: An update. Pol. Arch. Intern. Med. 2021, 131, 594–606. [Google Scholar] [CrossRef]
- Tschöpe, C.; Ammirati, E.; Bozkurt, B.; Caforio, A.L.P.; Cooper, L.T.; Felix, S.B.; Hare, J.M.; Heidecker, B.; Heymans, S.; Hübner, N.; et al. Myocarditis and inflammatory cardiomyopathy: Current evidence and future directions. Nat. Rev. Cardiol. 2021, 18, 169–193. [Google Scholar] [CrossRef]
- Georgiopoulos, G.; Figliozzi, S.; Sanguineti, F.; Aquaro, G.D.; di Bella, G.; Stamatelopoulos, K.; Chiribiri, A.; Garot, J.; Masci, P.G.; Ismail, T.F. Prognostic Impact of Late Gadolinium Enhancement by Cardiovascular Magnetic Resonance in Myocarditis: A Systematic Review and Meta-Analysis. Circ. Cardiovasc. Imaging 2021, 14, E011492. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, M.J.; Fard, F.B.; Friedrich, M.G. COVID-19, myocardial edema and dexamethasone. Med. Hypotheses 2020, 145, 110307. [Google Scholar] [CrossRef]
- Liguori, C.; Farina, D.; Vaccher, F.; Ferrandino, G.; Bellini, D.; Carbone, I. Myocarditis: Imaging up to date. La Radiol. Med. 2020, 125, 1124. [Google Scholar] [CrossRef]
- Amin, H.; Siddiqui, W.J. Cardiomegaly. In Diagnostic Atlas of Pediatric Imaging: Chest and Mediastinum; Jaypee Brothers Medical Pub.: New Delhi, India, 2021; p. 88. [Google Scholar] [CrossRef]
- Tymińska, A.; Ozierański, K.; Skwarek, A.; Kapłon-Cieślicka, A.; Baritussio, A.; Grabowski, M.; Marcolongo, R.; Caforio, A.L.P. Personalized Management of Myocarditis and Inflammatory Cardiomyopathy in Clinical Practice. J. Pers. Med. 2022, 12, 183. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with COVID-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2021, 397, 1637–1645. [Google Scholar] [CrossRef]
- Mazeraud, A.; Jamme, M.; Mancusi, R.L.; Latroche, C.; Megarbane, B.; Siami, S.; Zarka, J.; Moneger, G.; Santoli, F.; Argaud, L.; et al. Intravenous immunoglobulins in patients with COVID-19-associated moderate-to-severe acute respiratory distress syndrome (ICAR): Multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Respir. Med. 2022, 10, 158. [Google Scholar] [CrossRef]
- De la Concepción, M.L.R.; Ainsua-Enrich, E.; Reynaga, E.; Ávila-Nieto, C.; Santos, J.R.; Roure, S.; Mateu, L.; Paredes, R.; Puig, J.; Jimenez, J.M.; et al. High-dose intravenous immunoglobulins might modulate inflammation in COVID-19 patients. Life Sci. Alliance 2021, 4, lsa.202001009. [Google Scholar] [CrossRef]
- Tardif, J.C.; Bouabdallaoui, N.; L’Allier, P.L.; Gaudet, D.; Shah, B.; Pillinger, M.H.; Lopez-Sendon, J.; da Luz, P.; Verret, L.; Audet, S.; et al. Colchicine for community-treated patients with COVID-19 (COLCORONA): A phase 3, randomised, double-blinded, adaptive, placebo-controlled, multicentre trial. Lancet Respir. Med. 2021, 9, 924–932. [Google Scholar] [CrossRef]
- Maisch, B.; Alter, P. Treatment options in myocarditis and inflammatory cardiomyopathy: Focus on i. v. immunoglobulins. Herz 2018, 43, 423–430. [Google Scholar] [CrossRef]
- Mirna, M.; Schmutzler, L.; Topf, A.; Boxhammer, E.; Sipos, B.; Hoppe, U.C.; Lichtenauer, M. Treatment with Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) Does Not Affect Outcome in Patients with Acute Myocarditis or Myopericarditis. J. Cardiovasc. Dev. Dis. 2022, 9, 32. [Google Scholar] [CrossRef]
- Rathore, S.S.; Rojas, G.A.; Sondhi, M.; Pothuru, S.; Pydi, R.; Kancherla, N.; Singh, R.; Ahmed, N.K.; Shah, J.; Tousif, S.; et al. Myocarditis associated with COVID-19 disease: A systematic review of published case reports and case series. Int. J. Clin. Pract. 2021, 75, e14470. [Google Scholar] [CrossRef]
- Chedid, M.; Waked, R.; Haddad, E.; Chetata, N.; Saliba, G.; Choucair, J. Antibiotics in treatment of COVID-19 complications: A review of frequency, indications, and efficacy. J. Infect. Public Health 2021, 14, 570–576. [Google Scholar] [CrossRef]
- Horby, P.W.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Emberson, J.; Palfreeman, A.; Raw, J.; Elmahi, E.; Prudon, B.; et al. Lopinavir–ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2020, 396, 1345–1352. [Google Scholar] [CrossRef]
- WHO Solidarity Trial Consortium. Repurposed Antiviral Drugs for COVID-19—Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021, 384, 497–511. [Google Scholar] [CrossRef]
- Vegivinti, C.T.R.; Evanson, K.W.; Lyons, H.; Akosman, I.; Barrett, A.; Hardy, N.; Kane, B.; Keesari, P.R.; Pulakurthi, Y.S.; Sheffels, E.; et al. Efficacy of antiviral therapies for COVID-19: A systematic review of randomized controlled trials. BMC Infect. Dis. 2022, 22, 1–45. [Google Scholar] [CrossRef]
- Pollack, A.; Kontorovich, A.R.; Fuster, V.; Dec, G.W. Viral myocarditis—diagnosis, treatment options, and current controversies. Nat. Rev. Cardiol. 2015, 12, 670–680. [Google Scholar] [CrossRef]
- Ammirati, E.; Cipriani, M.; Moro, C.; Raineri, C.; Pini, D.; Sormani, P.; Mantovani, R.; Varrenti, M.; Pedrotti, P.; Conca, C.; et al. Clinical Presentation and Outcome in a Contemporary Cohort of Patients with Acute Myocarditis: Multicenter Lombardy Registry. Circulation 2018, 138, 1088–1099. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).