Abstract
In vivo confocal microscopy (IVCM) imaging is increasingly popular in ocular surface disease diagnosis and management. We conducted a systematic review to update the use of IVCM in the diagnosis and treatment of dry eye and meibomian gland dysfunction (MGD). A literature review was conducted on IVCM studies in MGD, dry eye disease, systemic disease causing dry eye, dry eye in glaucoma patients, contact lens-associated ocular conditions, graft-versus-host disease, and Sjogren’s syndrome-related dry eye. The articles were identified through PubMed and a total number of 63 eligible publications were analyzed in detail. All primary research studies on confocal microscopy on dry eye and related conditions from 2017 onwards were included. The reports were reviewed for their contribution to the existing literature as well as potential biases and drawbacks. Despite limitations such as small field of view, lack of population-based norms, and lack of standardization of image acquisition, interpretation, and quantification, IVCM is useful as a complementary technique for clinical diagnosis in various ocular surface disorders related to dry eye. With advances in hardware and software in the near future, it has the potential for further practical impact.
1. Introduction
There are many common ocular surface disorders (OSD), such as Dry Eye Disease (DED), blepharitis, and meibomian gland dysfunction (MGD), whose management requires visualization of certain ocular surface structures via slit-lamp biomicroscopy. In vivo confocal microscopy (IVCM), a more recent imaging technique has been evaluated in clinics for similar visualization [1].
The ocular surface consists of the conjunctiva, cornea, and the ocular mucosal adnexa (eyelid margins, eyelid glands, and lacrimal apparatus). The cornea, though uniquely suitable for IVCM due to its transparency, is not the only structure that can be visualized with this technique. It consists of the epithelium, basement membrane, Bowman’s layer, stroma, pre-Descemet, Descemet membrane, and endothelium [2]. The stroma contains keratocytes, dendritic cells (DCs), and nerve bundles that give rise to multiple branches which penetrate the epithelium [1]. Many of these corneal structures are not visible at the cellular level by conventional slit-lamp biomicroscopy but are clearly visible on IVCM. Unlike conventional light microscopy, IVCM directs light with a 670-nm wavelength laser [3] to pass to the desired focal spot using a pinhole aperture, which overcomes the problem of light scattering and provides clearer images at the cellular level. The resolution and magnification of IVCM (800-fold) are also much better than that of slit-lamp microscopy (40 fold), thus allowing improved and even cellular resolution of the ocular surface [4]. The resolution is also superior, with a lateral resolution of 1–2 μm and axial resolution of 5–10 μm [5]. Previously, Cruzat et al. [6] wrote an extensive review in 2017 on the pathological changes of corneal nerves in various ocular surface diseases. Other recent reviews [7,8] published in 2020 have not delved in-depth into various clinical applications of IVCM nor covered the spectrum of non-infective ocular surface diseases, and hence, there is a need for an update. In this article, we aim to review the literature on the clinical use of IVCM, focusing on studies published after 2017.
2. Materials and Methods
2.1. Study Objective and Definition of Reference Standard
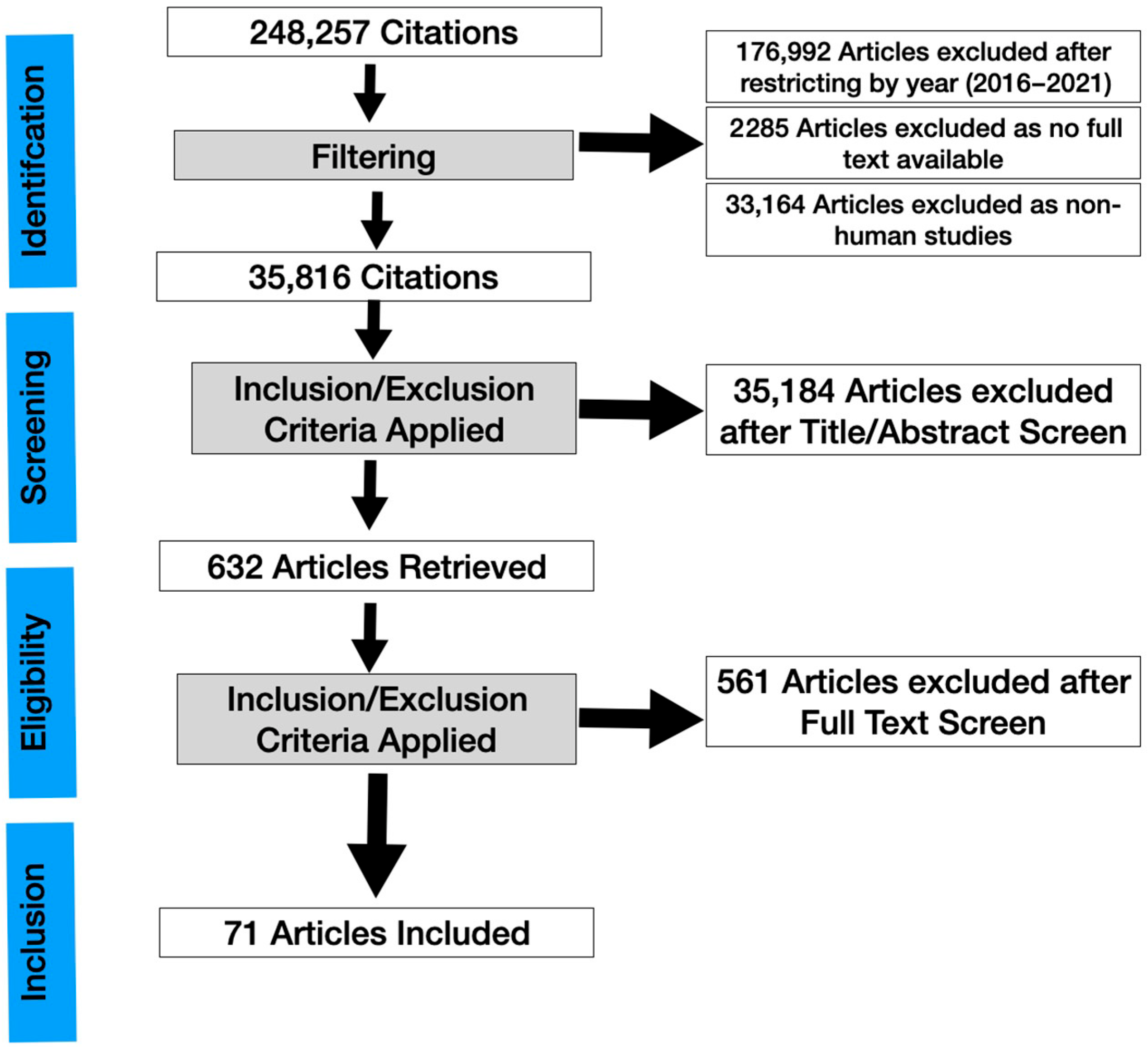
A systematic review was conducted (Figure 1) and the results were reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement. The main objective of this systematic review and meta-analysis is to evaluate the clinical applications of IVCM in dry eye, focusing on new observations. Because this study did not directly involve patients, an ethics committee approval was not required.
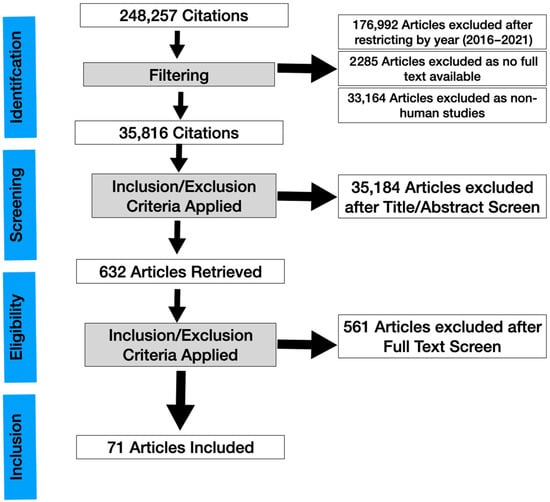
Figure 1.
PRISMA flowchart of a systematic review of in vivo confocal microscopy in different types of dry eye and meibomian gland dysfunction.
2.2. Literature Search Strategy
We determined the criteria and search strategy before the start of the study. An entrez PubMed literature search of the PubMed database using the terms and a detailed combination of keywords to capture ocular surface disease and dry eye was performed from inception until 9 November 2021. The search terms included the following keywords and keyword combinations (((“in vivo” OR in vivo OR in-vivo) AND confocal) OR IVCM) AND keywords to identify a set of ocular surface articles (Please refer to Supplementary File S1).
2.3. Eligibility Criteria
Two reviewers (R.S. and K.Y.) screened all retrieved articles by title and abstract initially. Only original research articles written in English were included and not analysis reviews, editorials, opinions, single case reports, and ex-vivo studies. The articles found were curated manually for their relevance to the ocular surface. The following conditions were not considered to be ocular surface disease: infectious keratitis, bullous keratopathy, ocular cystinosis, and Iridocorneal Endothelial Syndrome (ICE) syndrome.
The full text of the remaining studies was curated. Additionally, the reference lists of the remaining studies were checked to identify further relevant articles that may have been overlooked during the initial process. Hard copies of all of the eligible articles were obtained and fully read.
We excluded articles where IVCM findings were not mentioned in the results of the full-text article. Studies where recovery of the full text was not possible, even after searching the available medical databases and contacting the corresponding authors, were excluded. Disagreements were settled through discussion with a third reviewer (L.T.).
2.4. Data Extraction and Quality Evaluation of the Studies
The initial database search with the above keywords identified 248,257 papers. The database search for publication dates from 2016 to 22 November 2021 identified 71,265 studies.
After excluding articles where full text was not available (2285), 68,980 studies were left. After excluding non-human studies (33,164), 35,816 studies were left. After going through the title and screening through the abstract and applying our inclusion/exclusion criteria (4281 were reviews, 799 were systematic reviews, and 5056 were case reports), 632 studies were left. After full text-retrieval and further curation, 71 studies remained. A risk of bias assessment was then evaluated using the AMSTAR2 tool (Supplementary Table S1) [9] and the methodological quality of eligible articles was assessed using bias using the Revised Cochrane risk-of-bias tool for randomized trials RoB2 tool (Supplementary Table S2) [10]. Supplementary Table S2 presents the risk of bias summary per domain for individual studies and for all 4 included studies. The overall risk of biased judgment has been ascertained to reflect concerns in these studies as they have some concerns in at least one domain.
3. Results
3.1. Meibomian Gland Dysfunction
Meibomian glands (MG) have been classically described to compose of acini constituted by convoluted borders lined by large cells with fine cellular material within the lumen [11], interstitial space between acini, ductules, and terminal ducts. Abnormal meibum quality and quantity can lead to a decreased or altered tear film lipid layer, tear hyperosmolarity, tear instability, and inflammation, leading to ocular surface damage and DED [12]. Significant fibrosis (demonstrated via loss of MG architecture with extensive fibrotic tissue surrounding MG remnants) has been observed in chronic MG dysfunction [13]. Recent studies on MGD are summarized in Table 1. A decrease in the size of the MG acinar unit was also observed [14]. IVCM has also been used to analyze the palpebral conjunctiva to visualize and quantify the density of immune cells [15]. These cells have been evaluated in different locations: epithelial (EIC), intraglandular (IGIC), stromal (SIC), and periglandular (PGIC) regions. The immune cells in EIC and IGIC were increased in MGD patients with more severe dry eye symptoms, even in those with minimal corneal staining [16]. Basal epithelial cell density was also found to be reduced with greater stromal nerve thickness in the MGD group [17]. Hence IVCM may provide reliable and clinically relevant metrics of inflammation and serve as clinical endpoints in future clinical trials targeting inflammation in MGD.

Table 1.
Studies on Meibomian Gland Dysfunction (MGD).
3.2. Dry Eye Disease
Dry eye disease (DED) is defined as a “multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles” [16]. The ocular surface, epithelial sensory receptors, the innervation of the epithelial sensory receptors, secretory centers in the brain, and efferent nerves supplying the meibomian glands, goblet cells, and the main lacrimal gland form a functional unit. Any or all of these structures may be affected in DED [18]. The cornea is the most densely innervated part of the body. The cornea nerves serve the protective blink reflexes, help in tear secretion, and release neurotransmitters necessary for epithelial and stromal support as well as ocular homeostasis. They also serve the nociceptors associated with mechanical stimuli, pain, and cold sensations. Corneal nerves and their morphological changes can be seen under IVCM. With the help of analytic software, the corneal nerve plexuses can be evaluated quantitatively, typically by measuring the nerve fiber density, length, nerve branch density, and tortuosity [19,20]. The IVCM studies related to dry eye are summarized in Table 2.

Table 2.
Studies on Sjogren Syndrome (SS) and Non-Sjogren Syndrome Dry Eye (NSSDE).
Corneal dendritic cells (DC) have been shown to be increased in dry eye patients compared to controls [21]. In the pathogenesis of dry eye, DCs play an important role in inducing the activation of T cells [22], thus triggering an inflammatory cascade reaction.
Reduced corneal nerve density and length indicate a greater degree of neural damage induced by ocular pathology [23]. It has been shown that reduced density of corneal nerves results in impairment of protective functions such as tear secretion and blink reflexes. This results in a reduction in the tear quality and even aqueous tear deficiency. A study has suggested that patients with DED (diagnosed using TBUT and Schirmer’s, as well as the presence of symptoms) had decreased corneal nerve density [24].
Nerve tortuosity, defined by the frequency and the amplitude of the variations in the nerve fiber orientation, suggests active regeneration of nerve fibers in damaged nerves [25]. Studies by Liu et al. [26], Tepelus et al. [24], and Baikai et al. [27] have shown that nerve tortuosity is positively correlated with the diagnosis of DED. A greater nerve tortuosity is linked to ocular discomfort, visual function disturbance, and tear film instability [27].
3.3. Sjogren’s-Related Dry Eye (SSDE)
Sjogren syndrome (SS) is a systemic autoimmune disease that initially targets the lacrimal and salivary glands primarily, resulting in keratoconjunctivitis sicca (SSDE) and stomatitis sicca (dry mouth). The prevalence of primary SS in the USA approaches 1.3 million, with a range of 0.4–3.1 million [28]. The IVCM studies related to Sjogren’s-related dry eye are summarized in Table 2 as well. Certain IVCM parameters in sub-basal nerves have been reported to be altered in SS. Nerve fiber density is significantly decreased in SS [29,30], and SSDE is associated with greater nerve tortuosity than non-Sjogren’s Syndrome Dry Eye (NSSDE) [29].
Light backscattering (LB) measured in light reflectivity unit (LRU) at the Bowman’s membrane (BM) at 50 μm, 100 μm, and 200 μm deep to the BM has been evaluated in SS using IVCM—this is a measure of corneal inflammation [31]. Higher levels of LB in each corneal layer compared with healthy controls could indicate increased levels of corneal inflammation in SSDE [32].
The corneal epithelium of DED patients shows morphological changes, such as areas of enlarged and irregularly-shaped cells, which can be quantified by IVCM. Compared to controls, the density of superficial epithelial cells was decreased in both the NSDE and SSDE groups [33].
In summary, IVCM represents a reliable technique for examining nerve tortuosity in DED, as well as documenting corneal epithelial changes and immune cell densities in SSDE.
3.4. The Use of IVCM to Evaluate the Treatment for Dry Eye
While disease outcomes have typically been measured using symptomatic questionnaires and clinical tools such as Schirmer Test, corneal and conjunctival staining, tear break up time (TBUT), and tear osmolarity, there has been increasing interest to document treatment outcomes with IVCM [34,35,36].
The IVCM studies performed as part of interventional trials in DED are summarized in Table 3. The most common anti-inflammatory treatment for DED is cyclosporin A (CsA), an immunosuppressant and a calcineurin inhibitor. It has been used in several trials since 1986 and continues to be the major anti-inflammatory drug in the treatment of DED. Six months following treatment with topical CsA in SSDE patients [37], symptoms of dry eye documented by the Ocular Surface Disease Index (OSDI) score improved together with a decrease in corneal nerve tortuosity. The same study reported an increase in sub-basal nerve plexus (SNP) density and a decrease in DC density after treatment. Though increased nerve reflectivity was found, the association was not significant. The decrease in DC density was attributed to the decrease in antigen-presenting cells and local inflammation, and the increase in SNP density was due to the normalization of innervation by controlling the inflammatory reaction [37].

Table 3.
Studies on Treatment-Induced Changes in Dry Eye.
Similarly, Laccheri et al. [38] also found a decrease in nerve tortuosity in SSDE after treatment with cyclosporine. They also found a decrease in nerve reflectivity, the number of sub-basal nerves, and an increase in intermediate corneal epithelial cells. The reduction of sub-basal nerves, reflectivity, and nerve tortuosity could be related to decreased nerve growth factor (NGF) post-treatment, though NGF levels were not checked for in this study. This is uniquely expressed in the human limbal basal epithelium, along with its two corresponding receptors: the low-affinity receptor p75NTR and the high-affinity receptor TrkA. The first receptor, when activated, transmits a signal mainly of apoptosis; the latter, when activated, promotes a molecular cascade aimed at the proliferation and cell activation, which replaces apoptotic cells. NGF secretion is stimulated by high levels of IL-1 and TNF during inflammation and hence treatment of DED would reduce inflammation and the level of NGF. The increase in epithelial cell density after using cyclosporine has also been postulated to be due to decreased NGF and reduction of apoptotic signals through the p75NTR [38].
The apparent discrepancies in these studies could be due to the difference in severities of the disease or demographic differences in the patients recruited.
More recently, homologous sera obtained from healthy donors (i.e., allogeneic peripheral blood serum [allo-PBS] and cord blood serum [CBS]) have been proposed as treatment alternatives in patients with severe DED. Both treatments significantly improved corneal SNP parameters, and in particular, nerve density, length, width, and fractal dimension [39]. Giant epithelial cells, beading, and neuromas have also been shown to be decreased. Corneal nerve fractal dimension (CNFrD) is a novel IVCM metric that measures the structural complexity of corneal nerves, and its reduction represents nerve degeneration. It has been demonstrated that the CNFrD value has a diagnostic efficiency comparable with conventional IVCM parameters for identifying diabetic corneal neuropathy [40]. On the other hand, corticosteroids did not alter the quantitative measurements of the corneal SNP even though they decreased corneal DC density [41]. IVCM was used in a clinical trial that evaluated the use of omega-3 fatty acid supplements in DED [42]. These supplementations may have neuroprotective effects on corneal nerves, shown by an increase in corneal total nerve branch density (CTBD) and corneal nerve branch density on the main fiber (CNBD) after 90 days.
3.5. Systemic Disease
Certain systemic diseases associated with DED are shown in Table 4.

Table 4.
Studies on Systemic Disease-related Dry Eye.
Diabetic neuropathy, including diabetic corneal neuropathy, is one of the most common microvascular complications in diabetes [43]. There is increasing evidence to show that impairment of microvascular components is preceded by early neurodegenerative alterations primarily involving small nerve fibers, which can be demonstrated by IVCM [44,45]. Moreover, as small-fiber neuropathic changes can be picked up by IVCM, corneal nerve metrics have been used as surrogate markers for diabetic peripheral neuropathy [46]. Studies have shown that IVCM parameters such as CNFL [47,48], CNBD, CNFD, and CNFrD are reduced in patients with diabetes compared to controls, especially at the inferior whorl site [49,50]. A significant reduction in nerve beading frequency was also reported, which may be due to reduced metabolomic activity in diabetic patients [51].
IVCM is useful in analyzing the cornea and MG structures as well as the skin epidermis and dermis in ocular rosacea. It can quantify MG alterations based on meibum reflectivity, inflammation, and fibrosis, which correlated with the number of Demodex mites in both MG and cheek. However, no correlation was found between IVCM scores and both subjective and objective tests of dry eye [52].
Graves’ ophthalmopathy (GO) is often associated with DED, the most frequent cause of ocular discomfort in such patients [53]. GO is an autoimmune disease in which autoantibodies to the thyroid-stimulating hormone receptor lead to an inflammatory response in the orbital tissues [54]. Recent studies with IVCM have found changes in corneal nerves and MGs. Abnormal corneal SNP has been reported in active and inactive GO, suggesting nerve degeneration in GO. These central corneal SNP parameters of GO patients were significantly decreased compared with those of controls: corneal nerve fiber density (CNFD), corneal nerve branch density (CNBD), corneal nerve fiber length (CNFL), corneal nerve fiber total branch density (CTBD), corneal nerve fiber area (CNFA), corneal nerve fiber width (CNFW) and corneal nerve fiber fractal dimension (CNFrD). In addition, CNFD and ACNFrD values were significantly lower in the active GO compared with inactive GO patients. However, this study did not adjust for potential differences in DED between GO states [55]. Hence, further studies could further stratify the active TAO further into mild, moderate, and severe states before comparing the difference in nerve parameters.
IVCM also effectively revealed microstructural changes of MGs in eyes with GO and provided strong in vivo evidence for the roles of obstruction and inflammation in the disease process [56]. However, the patients in both groups had differing OSDI scores. Hence, it is unclear if the MG changes are related to concomitant DED in the GO patients or related to an extension of GO orbitopathy.
Previous studies have discovered an association between DED and migraine headaches [57,58]. It has been hypothesized that the trigeminal system plays a critical role in the pathogenesis of ocular symptoms in migraine. The pain or photophobia associated with migraine is believed to arise from the release of vasoactive neuropeptides at the peripheries of the three main branches of the trigeminal nerve, which innervates the dura, cranium, face, and eye. The ophthalmic branch (V1), in particular, also serves as the afferent for ocular discomfort associated with dry eye [59,60]. IVCM can also study the structural changes in nociceptive corneal axons in the SNP, which showed a decrease in corneal nerve fiber length, total branch density, nerve branch density, and fiber area in patients with migraine and photophobia compared to patients with migraine without photophobia. Hence, SNP changes on IVCM may serve as a potential imaging marker for ocular symptoms of chronic migraine [61]. Unfortunately, the study did not include controls without migraine and did not assess the DED parameters in the participants. Hence, it is difficult to conclude whether the presence of DED parameters is indicative of the severity of migraines.
3.6. Glaucoma Treatment-Related Dry Eyes
Glaucoma is the leading cause of global irreversible blindness. The number of people with glaucoma worldwide will increase to 111.8 million in 2040, disproportionally affecting people residing in Asia and Africa [62]. Glaucoma is the leading cause of global irreversible blindness. The most common initial treatment for glaucoma is topical medical therapy and about half of glaucoma patients on topical anti-glaucomatous medications have the ocular surface disease [63]. Previous studies have demonstrated that toxic and proinflammatory effects of antiglaucoma ophthalmic solutions are mainly due to preservatives, though prostaglandins by themselves can cause periorbitopathy [64,65].
IVCM studies related to glaucoma are summarized in Table 5. Such imaging is useful in evaluating proinflammatory ocular surface changes induced by anti-glaucoma eye drops. These parameters may be affected: basal epithelial cell density, stromal reflectivity, number of sub-basal nerves, sub-basal nerve tortuosity, sub-basal nerve reflectivity, and endothelial cell density. One study found increased basal epithelial cells density, stromal reflectivity, sub-basal nerve tortuosity, and reduced sub-basal nerves in patients using glaucoma drops compared to healthy controls [66].

Table 5.
Studies on Glaucoma-Related Dry Eye.
IVCM can also document changes in the cornea after glaucoma filtration surgery to evaluate for surgical success. For instance, preoperative DC density and goblet cell density (GCD) are correlated with filtration surgery outcomes [67]. These parameters were measured at the upper bulbar conjunctiva corresponding to the bleb site pre-operatively and at the bleb site postoperatively. Images were acquired from the epithelium and subepithelium (10–50 microns of depth). GCs may transport aqueous humor through the bleb wall [68] and DCs are the source of immune-regulatory cytokines [69], so increased GC and decreased DC are predictors of good outcomes. Hence, IVCM of the conjunctiva may represent an imaging tool to predict surgical success in glaucoma [67]. However, the study did not assess objective markers of dry eye, such as Schirmer’s test or TBUT.
In addition, IVCM can be used to describe and compare the conjunctival filtering bleb features after XEN gel implantation and trabeculectomy, providing objective evaluation at a cellular level. For instance, IVCM was used to evaluate parameters like stromal meshwork reflectivity (SMR). As SMR represents an indirect indicator of the collagen content within the conjunctival stroma, a hyper-reflective pattern was a sign of collagen deposition, scarring, and potentially poorer clinical outcomes. After trabeculectomy, blebs showing a low degree of reflectivity and a thick wall are more likely to have a good filtering function [70]. However, this study did not evaluate the success or failure of these procedures in the long term as it only included blebs with a completely successful filtering function.
3.7. Corneal Graft Versus Host Disease (GVHD)
IVCM studies related to inflammation-related dry eyes from immune, toxic, or environmental causes are summarized in Table 6. For example, DED can also be mediated by severe immune reactions such as Graft-versus-host disease (GVHD), which is an inflammatory immune disease arising from an immunologic attack by donor alloreactive T cells that result in damage to vital organs, including the ocular surface of the eye [71].

Table 6.
Studies on Inflammation-Related Dry Eye.
Patients with ocular GVHD adjusted for ODSI and corneal staining displayed significantly decreased corneal epithelial cell density, SNP fiber density, and reflectivity compared to DED from other causes and healthy controls, while nerve tortuosity and epithelial DC density were increased in both oGVHD and DED groups [72]. This is in agreement with previous cross-sectional studies done [73,74,75]. As patients with DED unrelated to GVHD and ocular GVHD typically present with similar symptoms, IVCM could be used to evaluate and monitor patients with dry eyes due to GVHD and non-GVHD.
Patients with chronic GVHD had worse meibography scores, reduced corneal sub-basal nerve plexus densities, lower TBUT scores, lower Schirmer I values and higher corneal staining scores. There was extensive loss of meibomian glands in both superior and inferior eyelids. In patients with chronic GVHD, the ensuing long-term inflammation often results in fibrosis of the ocular surface and cicatrizing conjunctivitis [76]. Hence, patients with chronic GVHD are at high risk for developing DED and MG dysfunction [77]. It is unclear if the IVCM signs of GVHD are linked to the more severe MG dysfunction compared to the DED group.
3.8. Contact Lens-Related Conditions
Clinical studies using IVCM for contact lens-related problems are summarized in Table 7. Estimates of total contact lens (CL) wearers worldwide in 2005 were as high as 140 million and hence even complications with a low incidence may affect a large number of individuals [78]. While the majority of complications are minor such as conjunctival hyperemia and corneal edema from overwear, there are serious sight-threatening complications such as infectious keratitis [79]. IVCM of the central cornea observed a higher density of DCs in contact lens wearers compared with non–contact lens wearers. CL lens has been known to activate and increase DC, contributing to ocular surface inflammation and a decrease in SNP. This decrease in SNP has been hypothesized to be due to increased DC and activated inflammation [80]. This finding has also been confirmed in soft lens wearers [81].

Table 7.
Studies on CL-Related Dry Eye.
The precise etiology of “corneal infiltrative events” (CIE) which arise during CL wear, including both corneal infections and noninfectious inflammatory events [82], is not well understood. The incidence of symptomatic CIEs during extended soft lens wear ranges from 2.5 to 6%; when asymptomatic CIEs are included, the incidence can be as high as 20–25% [83].
IVCM can thus be potentially used to assess the subclinical response of the ocular surface in CL wearer. The risk of developing CIEs is 12.5 times higher in reusable lenses (those stored overnight in disinfecting solution throughout their usage period, which is typically 2 weeks or 1 month) compared with daily disposable lenses [80]. Interestingly, DC density was higher in reusable lens wearers than in daily disposable CL wearers [82].
IVCM can also study changes in corneal nerves associated with contact lenses. Orthokeratology (OK) involves using specially designed and fitted GP contact lenses to reshape the corneal surface for the temporary correction of refractive error. Lenses are only worn at night during sleep and removed on waking to provide clear, unaided vision throughout the day. IVCM has found that nerve fiber density (NFD) is decreased in OK wear [84,85]. This reduced NFD is associated with reduced corneal sensitivity and increased nerve tortuosity as well [85].
4. Discussion
4.1. Limitations
Several studies were limited by their cross-sectional nature and hence inability to prove causation between the disease and IVCM parameters studied [14,38,52,55,56,61,70,77]. In addition, many studies were limited by their small sample size [14,24,55,61,66,68,70,77,80].
Some study designs include biases, with either no placebo group for comparison [37] or a lack of standardized treatment [68]. A study had no control groups or insufficient control (controls had dry eye symptoms) [52]. Others had study populations that may not be representative. For instance, in one study, most participants were female, and hence the results may not be extrapolated to the general population [24].
There were also limitations in the IVCM technique. A central area for analysis was selected to ensure consistent measurements across patient groups [66], but multiple scans in different areas could potentially give a more comprehensive assessment.
There may be significant inter-observer and intra-observer variability with poor repeatability and manual processing is laborious and time-consuming. Furthermore, IVCM can only image a very small field of view, hindering reproducible imaging of the same areas over time.
A limitation of our study was complying with all items for a systematic review. We did not register the protocol for this review in a review registry which is a flaw according to AMSTAR-2 (Supplementary Table S1). We have tried to reflect, in the material and methods section, the entire search protocol as it was carried out and the search strategy designed. For conducting a proper systematic review, out of sixteen questions, we answered “Yes” to nine questions, with partial “Yes” to one and “No” to five questions. We missed item 7 (justification for excluding individual studies—we did not provide the full list of excluded articles, but this can be made available on request), item 10 (funding sources of papers as the paper would be too lengthy), item 11, 12, and 15 as these questions are not relevant when meta-analysis are not performed. We have a partial “Yes” to item 4 (we searched only 1 database instead of 2, but we have provided the keywords and search strategy used).
However, this review is about an imaging diagnostic tool, IVCM, not about specific therapy. As there were no studies that evaluated health outcomes based on the use of IVCM vs. the lack of such imaging, the AMSTAR2 and ROB2 could only be applied to study outcomes of specific treatment interventions.
4.2. Future Research Directions
Due to the high cost of this technology, the widespread deployment and use of IVCM in clinical practice are limited. Hopefully, improvement in hardware and wider use may bring the cost down. Analysis of corneal SNP on IVCM images can be fully automated, semi-automated, or manual. The fully automated technique requires no manual input from the observer and is faster and more suited for large trials and longitudinal studies requiring analysis of a large number of images. However, there might be more false-negative and false-positive errors that require improvement of these algorithms in the future [86].
As mentioned earlier, one of the drawbacks of IVCM is its small field of view, preventing an overview of SNP architecture and necessitating subjective image sampling of small areas of the SNP for analysis. Hence, future directions also include large-area imaging and mapping or mosaic technique. Corneal SNP can be reconstructed by automated mosaicking, with an average mosaic image size corresponding to 48 individual IVCM fields of view [87].
The use of artificial intelligence (AI) and increasing automation will improve the speed and accuracy of image analysis. Freeware, including ImageJ (NIH) and image processing packages for python (e.g., scikit-image, OpenCV) and others, have many built-in functions which allow for custom scripting. Future advances are likely to include advances in machine-learning algorithms, which are currently making their way into commercial software packages [88]. High-speed networks will also improve the ease of using IVCM images in digital medicine.
5. Conclusions
IVCM is useful as a complementary technique for clinical diagnosis in various ocular surface disorders related to dry eye. With advances in hardware and software in the near future, it has the potential for further practical impact and can be used for a multitude of OSD for diagnosis, management, and prognostication.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jcm11092349/s1, Supplementary File S1: TFOS Lifestyle Workshop 2021—Standardised Electronic Searches. Table S1: Table to summarize the results of AMSTAR-2. Table S2: Risk of bias summary for individual studies (n = 4) in accordance with Rob2.
Author Contributions
Conceptualization, L.T.; methodology, L.T.; literature review, R.S., K.Y.; data curation; writing—original draft preparation, K.Y. and R.S.; writing—review and editing, R.S., L.T., and Y.-C.L.; visualization, L.T., R.S.; supervision, L.T. All authors have read and agreed to the published version of the manuscript.
Funding
National Medical Research Council\Clinician Scientist Award\017\2017.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Singapore National Eye Centre, Singapore Eye Research Institute.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cruzat, A.; Pavan-Langston, D.; Hamrah, P. In Vivo Confocal Microscopy of Corneal Nerves: Analysis and Clinical Correlation. Semin. Ophthalmol. 2010, 25, 171–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dua, H.S.; Faraj, L.; Said, D.G.; Gray, T.; Lowe, J. Human Corneal Anatomy Redefined. Ophthalmology 2013, 120, 1778–1785. [Google Scholar] [CrossRef] [PubMed]
- Bohn, S.; Sperlich, K.; Stahnke, T.; Schünemann, M.; Stolz, H.; Guthoff, R.F.; Stachs, O. Multiwavelength confocal laser scanning microscopy of the cornea. Biomed. Opt. Express 2020, 11, 5689–5700. [Google Scholar] [CrossRef] [PubMed]
- Stachs, O.; Guthoff, R.F.; Aumann, S. In Vivo Confocal Scanning Laser Microscopy. In High Resolution Imaging in Microscopy and Ophthalmology; Springer: Berlin/Heidelberg, Germany, 2019; pp. 263–284. [Google Scholar] [CrossRef] [Green Version]
- Khamar, P.; Nair, A.P.; Shetty, R.; Vaidya, T.; Subramani, M.; Ponnalagu, M.; Dhamodaran, K.; D’Souza, S.; Ghosh, A.; Pahuja, N.; et al. Dysregulated Tear Fluid Nociception-Associated Factors, Corneal Dendritic Cell Density, and Vitamin D Levels in Evaporative Dry Eye. Investig. Opthalmol. Vis. Sci. 2019, 60, 2532–2542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cruzat, A.; Qazi, Y.; Hamrah, P. In Vivo Confocal Microscopy of Corneal Nerves in Health and Disease. Ocul. Surf. 2016, 15, 15–47. [Google Scholar] [CrossRef] [Green Version]
- Schmidl, D.; Schlatter, A.; Chua, J.; Tan, B.; Garhöfer, G.; Schmetterer, L. Novel Approaches for Imaging-Based Diagnosis of Ocular Surface Disease. Diagnostics 2020, 10, 589. [Google Scholar] [CrossRef]
- Binotti, W.W.; Bayraktutar, B.; Ozmen, M.C.; Cox, S.M.; Hamrah, P. A Review of Imaging Biomarkers of the Ocular Surface. Eye Contact Lens Sci. Clin. Pract. 2020, 46, S84–S105. [Google Scholar] [CrossRef]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [Green Version]
- Efron, N.; Al-Dossari, M.; Pritchard, N. In Vivo Confocal Microscopy of the Palpebral Conjunctiva and Tarsal Plate. Optom. Vis. Sci. 2009, 86, E1303–E1308. [Google Scholar] [CrossRef]
- Chhadva, P.; Goldhardt, R.; Galor, A. Meibomian Gland Disease: The Role of Gland Dysfunction in Dry Eye Disease. Ophthalmology 2017, 124, S20–S26. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Chen, J.-Y.; Wang, Y.-Q.; Lin, Z.-R.; Wang, S. In vivo Confocal Microscopy Evaluation of Meibomian Gland Dysfunction in Dry Eye Patients with Different Symptoms. Chin. Med. J. 2016, 129, 2617–2622. [Google Scholar] [CrossRef] [PubMed]
- Qazi, Y.; Kheirkhah, A.; Blackie, C.; Cruzat, A.; Trinidad, M.; Williams, C.; Korb, D.R.; Hamrah, P. In vivo detection of clinically non-apparent ocular surface inflammation in patients with meibomian gland dysfunction-associated refractory dry eye symptoms: A pilot study. Eye 2015, 29, 1099–1110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qazi, Y.; Kheirkhah, A.; Blackie, C.; Trinidad, M.; Williams, C.; Cruzat, A.; Korb, D.R.; Hamrah, P. Clinically Relevant Immune-Cellular Metrics of Inflammation in Meibomian Gland Dysfunction. Investig. Opthalmol. Vis. Sci. 2018, 59, 6111–6123. [Google Scholar] [CrossRef]
- Craig, J.P.; Nichols, K.K.; Akpek, E.K.; Caffery, B.; Dua, H.S.; Joo, C.-K.; Liu, Z.; Nelson, J.D.; Nichols, J.J.; Tsubota, K.; et al. TFOS DEWS II Definition and Classification Report. Ocul. Surf. 2017, 15, 276–283. [Google Scholar] [CrossRef]
- Azizi, S.; Uçak, T.; Yaşar, I.; Karakurt, Y.; Erdogan, E.; Salman, I. Evaluation of the Corneal Layers in Meibomian-Gland-Dysfunction-Related Dry Eye by In Vivo Slit-Scanning Confocal Microscopy. Semin. Ophthalmol. 2017, 32, 377–383. [Google Scholar] [CrossRef]
- Stern, M.E.; Schaumburg, C.S.; Pflugfelder, S.C. Dry Eye as a Mucosal Autoimmune Disease. Int. Rev. Immunol. 2013, 32, 19–41. [Google Scholar] [CrossRef]
- Al-Aqaba, M.A.; Dhillon, V.K.; Mohammed, I.; Said, D.G.; Dua, H.S. Corneal nerves in health and disease. Prog. Retin. Eye Res. 2019, 73, 100762. [Google Scholar] [CrossRef]
- Liu, Y.-C.; Lin, M.T.-Y.; Mehta, J.S. Analysis of corneal nerve plexus in corneal confocal microscopy images. Neural Regen. Res. 2021, 16, 690–691. [Google Scholar] [CrossRef]
- Xu, J.; Chen, P.; Yu, C.; Liu, Y.; Hu, S.; Di, G. In vivo Confocal Microscopic Evaluation of Corneal Dendritic Cell Density and Subbasal Nerve Parameters in Dry Eye Patients: A Systematic Review and Meta-analysis. Front. Med. 2021, 8, 578233. [Google Scholar] [CrossRef]
- Maruoka, S.; Inaba, M.; Ogata, N. Activation of Dendritic Cells in Dry Eye Mouse Model. Investig. Opthalmol. Vis. Sci. 2018, 59, 3269–3277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallinikos, P.; Berhanu, M.; O’Donnell, C.; Boulton, A.J.M.; Efron, N.; Malik, R. Corneal Nerve Tortuosity in Diabetic Patients with Neuropathy. Investig. Opthalmol. Vis. Sci. 2004, 45, 418–422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tepelus, T.C.; Chiu, G.B.; Huang, J.; Huang, P.; Sadda, S.R.; Irvine, J.; Lee, O.L. Correlation between corneal innervation and inflammation evaluated with confocal microscopy and symptomatology in patients with dry eye syndromes: A preliminary study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 1771–1778. [Google Scholar] [CrossRef] [PubMed]
- Jalbert, I.; Stapleton, F.; Papas, E.; Sweeney, D.; Coroneo, M. In vivo confocal microscopy of the human cornea. Br. J. Ophthalmol. 2003, 87, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chou, Y.; Dong, X.; Liu, Z.; Jiang, X.; Hao, R.; Li, X. Corneal Subbasal Nerve Analysis Using In Vivo Confocal Microscopy in Patients with Dry Eye: Analysis and Clinical Correlations. Cornea 2017, 38, 1253–1258. [Google Scholar] [CrossRef]
- Ma, B.; Xie, J.; Yang, T.; Su, P.; Liu, R.; Sun, T.; Zhou, Y.; Wang, H.; Feng, X.; Ma, S.; et al. Quantification of Increased Corneal Subbasal Nerve Tortuosity in Dry Eye Disease and Its Correlation with Clinical Parameters. Transl. Vis. Sci. Technol. 2021, 10, 26. [Google Scholar] [CrossRef]
- Nguyen, C.Q.; Peck, A.B. Unraveling the Pathophysiology of Sjogren Syndrome-Associated Dry Eye Disease. Ocul. Surf. 2009, 7, 11–27. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Zhang, Q.; Ying, X.; He, J.; Jin, Y.; Xu, H.; Cheng, Y.; Zhao, M. Corneal nerve structure in patients with primary Sjögren’s syndrome in China. BMC Ophthalmol. 2021, 21, 211. [Google Scholar] [CrossRef]
- McNamara, N.A.; Ge, S.; Lee, S.M.; Enghauser, A.M.; Kuehl, L.; Chen, F.Y.-T.; Gallup, M.; McKown, R.L. Reduced Levels of Tear Lacritin Are Associated With Corneal Neuropathy in Patients With the Ocular Component of Sjögren’s Syndrome. Investig. Opthalmol. Vis. Sci. 2016, 57, 5237–5243. [Google Scholar] [CrossRef]
- Hillenaar, T.; van Cleynenbreugel, H.; Verjans, G.M.; Wubbels, R.J.; Remeijer, L. Monitoring the Inflammatory Process in Herpetic Stromal Keratitis: The Role of In Vivo Confocal Microscopy. Ophthalmology 2012, 119, 1102–1110. [Google Scholar] [CrossRef]
- Lanza, M.; Iaccarino, S.; Varricchi, G.; D’Errico, T.; Carnevale, U.A.G.; Bifani, M. Corneal confocal microscopy alterations in Sjögren’s syndrome dry eye. Acta Ophthalmol. 2016, 95, e366–e372. [Google Scholar] [CrossRef] [PubMed]
- Lee, O.L.; Tepelus, T.C.; Huang, J.; Irvine, A.G.; Irvine, C.; Chiu, G.B.; Sadda, S.R. Evaluation of the corneal epithelium in non-Sjögren’s and Sjögren’s dry eyes: An in vivo confocal microscopy study using HRT III RCM. BMC Ophthalmol. 2018, 18, 309. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, M.; Sano, K.; Takechi, S.; Tsubota, K. Impact of lifestyle intervention on dry eye disease in office workers: A randomized controlled trial. J. Occup. Health 2018, 60, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Giannaccare, G.; Pellegrini, M.; Sebastiani, S.; Bernabei, F.; Roda, M.; Taroni, L.; Versura, P.; Campos, E.C. Efficacy of Omega-3 Fatty Acid Supplementation for Treatment of Dry Eye Disease: A Meta-Analysis of Randomized Clinical Trials. Cornea 2019, 38, 565–573. [Google Scholar] [CrossRef]
- Leyva, I.M.; Molina-Leyva, A.; Bueno-Cavanillas, A. Efficacy of nutritional supplementation with omega-3 and omega-6 fatty acids in dry eye syndrome: A systematic review of randomized clinical trials. Acta Ophthalmol. 2017, 95, e677–e685. [Google Scholar] [CrossRef] [Green Version]
- Levy, O.; Labbé, A.; Borderie, V.; Hamiche, T.; Dupas, B.; Laroche, L.; Baudouin, C.; Bouheraoua, N. Increased corneal sub-basal nerve density in patients with Sjögren syndrome treated with topical cyclosporine A. Clin. Exp. Ophthalmol. 2017, 45, 455–463. [Google Scholar] [CrossRef] [Green Version]
- Iaccheri, B.; Torroni, G.; Cagini, C.; Fiore, T.; Cerquaglia, A.; Lupidi, M.; Cillino, S.; Dua, H.S. Corneal confocal scanning laser microscopy in patients with dry eye disease treated with topical cyclosporine. Eye 2017, 31, 788–794. [Google Scholar] [CrossRef] [Green Version]
- Giannaccare, G.; Buzzi, M.; Fresina, M.; Velati, C.; Versura, P. Efficacy of 2-Month Treatment With Cord Blood Serum Eye Drops in Ocular Surface Disease: An In Vivo Confocal Microscopy Study. Cornea 2017, 36, 915–921. [Google Scholar] [CrossRef]
- Chen, X.; Graham, J.; Petropoulos, I.N.; Ponirakis, G.; Asghar, O.; Alam, U.; Marshall, A.; Ferdousi, M.; Azmi, S.; Efron, N.; et al. Corneal Nerve Fractal Dimension: A Novel Corneal Nerve Metric for the Diagnosis of Diabetic Sensorimotor Polyneuropathy. Investig. Opthalmol. Vis. Sci. 2018, 59, 1113–1118. [Google Scholar] [CrossRef]
- Villani, E.; Garoli, E.; Termine, V.; Pichi, F.; Ratiglia, R.; Nucci, P. Corneal Confocal Microscopy in Dry Eye Treated with Corticosteroids. Optom. Vis. Sci. 2015, 92, e290–e295. [Google Scholar] [CrossRef] [Green Version]
- Chinnery, H.R.; Golborne, C.N.; Downie, L.E. Omega-3 supplementation is neuroprotective to corneal nerves in dry eye disease: A pilot study. Ophthalmic Physiol. Opt. 2017, 37, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, H.; Tan, H.C.; Lin, M.T.-Y.; Mehta, J.S.; Liu, Y.-C. Diabetic Corneal Neuropathy. J. Clin. Med. 2020, 9, 3956. [Google Scholar] [CrossRef] [PubMed]
- Clerck, E.E.B.D.; Schouten, J.S.; Berendschot, T.T.; Kessels, A.G.H.; Nuijts, R.M.M.A.; Beckers, H.J.M.; Schram, M.; Stehouwer, C.D.A.; Webers, C.A.B. New ophthalmologic imaging techniques for detection and monitoring of neurodegenerative changes in diabetes: A systematic review. Lancet Diabetes Endocrinol. 2015, 3, 653–663. [Google Scholar] [CrossRef]
- Perkins, B.A.; Lovblom, L.E.; Bril, V.; Scarr, D.; Ostrovski, I.; Orszag, A.; Edwards, K.; Pritchard, N.; Russell, A.; Dehghani, C.; et al. Corneal confocal microscopy for identification of diabetic sensorimotor polyneuropathy: A pooled multinational consortium study. Diabetologia 2018, 61, 1856–1861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.-C.; So, W.; Wong, N.Q.; Tan, H.; Lin, M.Y.; Lee, I.Y.; Mehta, J. Diabetic corneal neuropathy as a surrogate marker for diabetic peripheral neuropathy. Neural Regen. Res. 2022, 17, 2172. [Google Scholar] [CrossRef] [PubMed]
- Lagali, N.S.; Allgeier, S.; Guimarães, P.; Badian, R.A.; Ruggeri, A.; Köhler, B.; Utheim, T.P.; Peebo, B.; Peterson, M.; Dahlin, L.B.; et al. Reduced Corneal Nerve Fiber Density in Type 2 Diabetes by Wide-Area Mosaic Analysis. Investig. Opthalmol. Vis. Sci. 2017, 58, 6318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Utsunomiya, T.; Nagaoka, T.; Hanada, K.; Omae, T.; Yokota, H.; Abiko, A.; Haneda, M.; Yoshida, A. Imaging of the Corneal Subbasal Whorl-like Nerve Plexus: More Accurate Depiction of the Extent of Corneal Nerve Damage in Patients With Diabetes. Investig. Opthalmol. Vis. Sci. 2015, 56, 5417–5423. [Google Scholar] [CrossRef] [Green Version]
- Cozzini, T.; Piona, C.; Marchini, G.; Merz, T.; Brighenti, T.; Bonetto, J.; Marigliano, M.; Olivieri, F.; Maffeis, C.; Pedrotti, E. In vivo confocal microscopy study of corneal nerve alterations in children and youths with Type 1 diabetes. Pediatr. Diabetes 2021, 22, 780–786. [Google Scholar] [CrossRef]
- Petropoulos, I.N.; Ferdousi, M.; Marshall, A.; Alam, U.; Ponirakis, G.; Azmi, S.; Fadavi, H.; Efron, N.; Tavakoli, M.; Malik, R.A. The Inferior Whorl For Detecting Diabetic Peripheral Neuropathy Using Corneal Confocal Microscopy. Investig. Opthalmol. Vis. Sci. 2015, 56, 2498–2504. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Liu, D.; Li, L.; Li, Y.; Li, Q.; An, Z.; Sun, X.; Tian, H. Circulating Betatrophin in Patients with Type 2 Diabetes: A Meta-Analysis. J. Diabetes Res. 2016, 2016, 6194750. [Google Scholar] [CrossRef] [Green Version]
- Liang, H.; Randon, M.; Michee, S.; Tahiri, R.; Labbe, A.; Baudouin, C. In vivo confocal microscopy evaluation of ocular and cutaneous alterations in patients with rosacea. Br. J. Ophthalmol. 2016, 101, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Gürdal, C.; Saraç, Ö.; Genç, I.; Kırımlıoğlu, H.; Takmaz, T.; Can, I. Ocular Surface and Dry Eye in Graves’ Disease. Curr. Eye Res. 2011, 36, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Harrison, A.R.; Bothun, E.D.; Scheuer, R.A.; Lee, M.S. Update on thyroid eye disease and management. Clin. Ophthalmol. 2009, 3, 543–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.-Q.; Mou, P.; Chen, Z.-Y.; Cheng, J.-W.; Le, Q.-H.; Cai, J.-P.; Wei, R.-L. Altered Corneal Nerves in Chinese Thyroid-Associated Ophthalmopathy Patients Observed by In Vivo Confocal Microscopy. Med. Sci. Monit. 2019, 25, 1024–1031. [Google Scholar] [CrossRef]
- Cheng, S.; Yu, Y.; Chen, J.; Ye, L.; Wang, X.; Jiang, F. In vivo confocal microscopy assessment of meibomian glands microstructure in patients with Graves’ orbitopathy. BMC Ophthalmol. 2021, 21, 261. [Google Scholar] [CrossRef]
- Yang, S.; Kim, W.; Kim, H.S.; Na, K.-S. Epidemiologic Survey Committee of the Korean Ophthalmologic Society Association Between Migraine and Dry Eye Disease: A Nationwide Population-Based Study. Curr. Eye Res. 2017, 42, 837–841. [Google Scholar] [CrossRef]
- Sarac, O.; Kosekahya, P.; Tasci, Y.Y.; Keklikoglu, H.D.; Deniz, O.; Erten, Ş.; Çağıl, N. The Prevalence of Dry Eye and Sjögren Syndrome in Patients with Migraine. Ocul. Immunol. Inflamm. 2017, 25, 370–375. [Google Scholar] [CrossRef]
- Situ, P.; Simpson, T.L.; Jones, L.W.; Fonn, D. Conjunctival and Corneal Hyperesthesia in Subjects with Dryness Symptoms. Optom. Vis. Sci. 2008, 85, 867–872. [Google Scholar] [CrossRef]
- Edvinsson, J.C.A.; Viganò, A.; Alekseeva, A.; Alieva, E.; Arruda, R.; De Luca, C.; D’Ettore, N.; Frattale, I.; Kurnukhina, M.; Macerola, N.; et al. The fifth cranial nerve in headaches. J. Headache Pain 2020, 21, 65. [Google Scholar] [CrossRef]
- Shetty, R.; Deshmukh, R.; Shroff, R.; Dedhiya, C.; Jayadev, C. Subbasal Nerve Plexus Changes in Chronic Migraine. Cornea 2017, 37, 72–75. [Google Scholar] [CrossRef]
- Tham, Y.-C.; Li, X.; Wong, T.Y.; Quigley, H.A.; Aung, T.; Cheng, C.-Y. Global Prevalence of Glaucoma and Projections of Glaucoma Burden through 2040: A systematic review and meta-analysis. Ophthalmology 2014, 121, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Fechtner, R.D.; Godfrey, D.G.; Budenz, D.; Stewart, J.A.; Stewart, W.C.; Jasek, M.C. Prevalence of Ocular Surface Complaints in Patients With Glaucoma Using Topical Intraocular Pressure-Lowering Medications. Cornea 2010, 29, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Servat, J.J.; Bernardino, C.R. Effects of Common Topical Antiglaucoma Medications on the Ocular Surface, Eyelids and Periorbital Tissue. Drugs Aging 2011, 28, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Kucukevcilioglu, M.; Bayer, A.; Uysal, Y.; Altinsoy, H.I. Prostaglandin associated periorbitopathy in patients using bimatoprost, latanoprost and travoprost. Clin. Exp. Ophthalmol. 2013, 42, 126–131. [Google Scholar] [CrossRef]
- Rolle, T.; Spinetta, R.; Nuzzi, R. Long term safety and tolerability of Tafluprost 0.0015% vs Timolol 0.1% preservative-free in ocular hypertensive and in primary open-angle glaucoma patients: A cross sectional study. BMC Ophthalmol. 2017, 17, 136. [Google Scholar] [CrossRef] [Green Version]
- Mastropasqua, R.; Fasanella, V.; Brescia, L.; Oddone, F.; Mariotti, C.; Di Staso, S.; Agnifili, L. In Vivo Confocal Imaging of the Conjunctiva as a Predictive Tool for the Glaucoma Filtration Surgery Outcome. Investig. Opthalmol. Vis. Sci. 2017, 58, 114–120. [Google Scholar] [CrossRef]
- Amar, N.; Labbé, A.; Hamard, P.; Dupas, B.; Baudouin, C. Filtering Blebs and Aqueous Pathway: An Immunocytological and In Vivo Confocal Microscopy Study. Ophthalmology 2008, 115, 1154–1161.e4. [Google Scholar] [CrossRef]
- Agrawal, A.; Agrawal, S.; Gupta, S. Role of Dendritic Cells in Inflammation and Loss of Tolerance in the Elderly. Front. Immunol. 2017, 8, 896. [Google Scholar] [CrossRef]
- Sacchi, M.; Agnifili, L.; Brescia, L.; Oddone, F.; Villani, E.; Nucci, P.; Mastropasqua, L. Structural imaging of conjunctival filtering blebs in XEN gel implantation and trabeculectomy: A confocal and anterior segment optical coherence tomography study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2020, 258, 1763–1770. [Google Scholar] [CrossRef]
- Herretes, S.; Ross, D.B.; Duffort, S.; Barreras, H.; Yaohong, T.; Saeed, A.M.; Murillo, J.C.; Komanduri, K.V.; Levy, R.B.; Perez, V.L. Recruitment of Donor T Cells to the Eyes During Ocular GVHD in Recipients of MHC-Matched Allogeneic Hematopoietic Stem Cell Transplants. Investig. Opthalmol. Vis. Sci. 2015, 56, 2348–2357. [Google Scholar] [CrossRef] [Green Version]
- Tepelus, T.C.; Chiu, G.B.; Maram, J.; Huang, J.; Chopra, V.; Sadda, S.R.; Lee, O.L. Corneal features in ocular graft-versus-host disease by in vivo confocal microscopy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 255, 2389–2397. [Google Scholar] [CrossRef]
- Niederer, R.; Perumal, D.; Sherwin, T.; McGhee, C.N.J. Corneal Innervation and Cellular Changes after Corneal Transplantation: An In Vivo Confocal Microscopy Study. Investig. Opthalmol. Vis. Sci. 2007, 48, 621–626. [Google Scholar] [CrossRef] [Green Version]
- Patel, S.V.; Erie, J.C.; McLaren, J.W.; Bourne, W.M. Keratocyte and subbasal nerve density after penetrating keratoplasty. Trans. Am. Ophthalmol. Soc. 2007, 105, 180–189. [Google Scholar]
- Al-Aqaba, M.A.; Otri, A.M.; Fares, U.; Miri, A.; Dua, H.S. Organization of the Regenerated Nerves in Human Corneal Grafts. Am. J. Ophthalmol. 2012, 153, 29–37.e4. [Google Scholar] [CrossRef]
- Ogawa, Y. Sjögren’s Syndrome, Non-Sjögren’s Syndrome, and Graft-Versus-Host Disease Related Dry Eye. Investig. Opthalmol. Vis. Sci. 2018, 59, 71–79. [Google Scholar] [CrossRef]
- Dikmetas, O.; Kocabeyoglu, S.; Mocan, M.C. The Association between Meibomian Gland Atrophy and Corneal Subbasal Nerve Loss in Patients with Chronic Ocular Graft-versus-host Disease. Curr. Eye Res. 2021, 46, 796–801. [Google Scholar] [CrossRef]
- Stapleton, F.; Keay, L.; Jalbert, I.; Cole, N. The Epidemiology of Contact Lens Related Infiltrates. Optom. Vis. Sci. 2007, 84, 257–272. [Google Scholar] [CrossRef]
- Poggio, E.C.; Glynn, R.J.; Schein, O.D.; Seddon, J.M.; Shannon, M.J.; Scardino, V.A.; Kenyon, K.R. The Incidence of Ulcerative Keratitis among Users of Daily-Wear and Extended-Wear Soft Contact Lenses. N. Engl. J. Med. 1989, 321, 779–783. [Google Scholar] [CrossRef]
- Chalmers, R.L.; Keay, L.; McNally, J.; Kern, J. Multicenter Case-Control Study of the Role of Lens Materials and Care Products on the Development of Corneal Infiltrates. Optom. Vis. Sci. 2012, 89, 316–325. [Google Scholar] [CrossRef]
- Golebiowski, B.; Chao, C.; Bui, K.A.; Lam, W.Y.W.; Richdale, K.; Stapleton, F. Effect of age and contact lens wear on corneal epithelial dendritic cell distribution, density, and morphology. Contact Lens Anterior Eye 2019, 43, 84–90. [Google Scholar] [CrossRef]
- Saliman, N.H.; Morgan, P.; MacDonald, A.S.; Maldonado-Codina, C. Subclinical Inflammation of the Ocular Surface in Soft Contact Lens Wear. Cornea 2019, 39, 146–154. [Google Scholar] [CrossRef]
- Steele, K.R.; Szczotka-Flynn, L. Epidemiology of contact lens-induced infiltrates: An updated review. Clin. Exp. Optom. 2017, 100, 473–481. [Google Scholar] [CrossRef] [Green Version]
- Lum, E.; Golebiowski, B.; Swarbrick, H.A. Reduced Corneal Sensitivity and Sub-Basal Nerve Density in Long-Term Orthokeratology Lens Wear. Eye Contact Lens Sci. Clin. Pract. 2017, 43, 218–224. [Google Scholar] [CrossRef]
- Nombela-Palomo, M.; Felipe-Márquez, G.; Teus, M.; Hernández-Verdejo, J.L.; Bona, A.N. Long-Term Impacts of Orthokeratology Treatment on Sub-Basal Nerve Plexus and Corneal Sensitivity Responses and Their Reversibility. Eye Contact Lens Sci. Clin. Pract. 2018, 44, 91–96. [Google Scholar] [CrossRef]
- Turuwhenua, J.T.; Patel, D.; McGhee, C.N. Fully Automated Montaging of Laser Scanning In Vivo Confocal Microscopy Images of the Human Corneal Subbasal Nerve Plexus. Investig. Opthalmol. Vis. Sci. 2012, 53, 2235–2242. [Google Scholar] [CrossRef] [Green Version]
- Badian, R.A.; Allgeier, S.; Scarpa, F.; Andréasson, M.; Bartschat, A.; Mikut, R.; Colonna, A.; Bellisario, M.; Utheim, T.P.; Köhler, B.; et al. Wide-field mosaics of the corneal subbasal nerve plexus in Parkinson’s disease using in vivo confocal microscopy. Sci. Data 2021, 8, 306. [Google Scholar] [CrossRef]
- Bohr, A.; Memarzadeh, K. The rise of artificial intelligence in healthcare applications. In Artificial Intelligence in Healthcare; Academic Press: Cambridge, MA, USA, 2020; Chapter 2; pp. 25–60. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).